Renal Impairment in Chronic Hepatitis B: A Review
Abstract
1. Introduction
2. HBV-Associated Nephropathy
2.1. Pathology of HBV-Associated Nephropathy
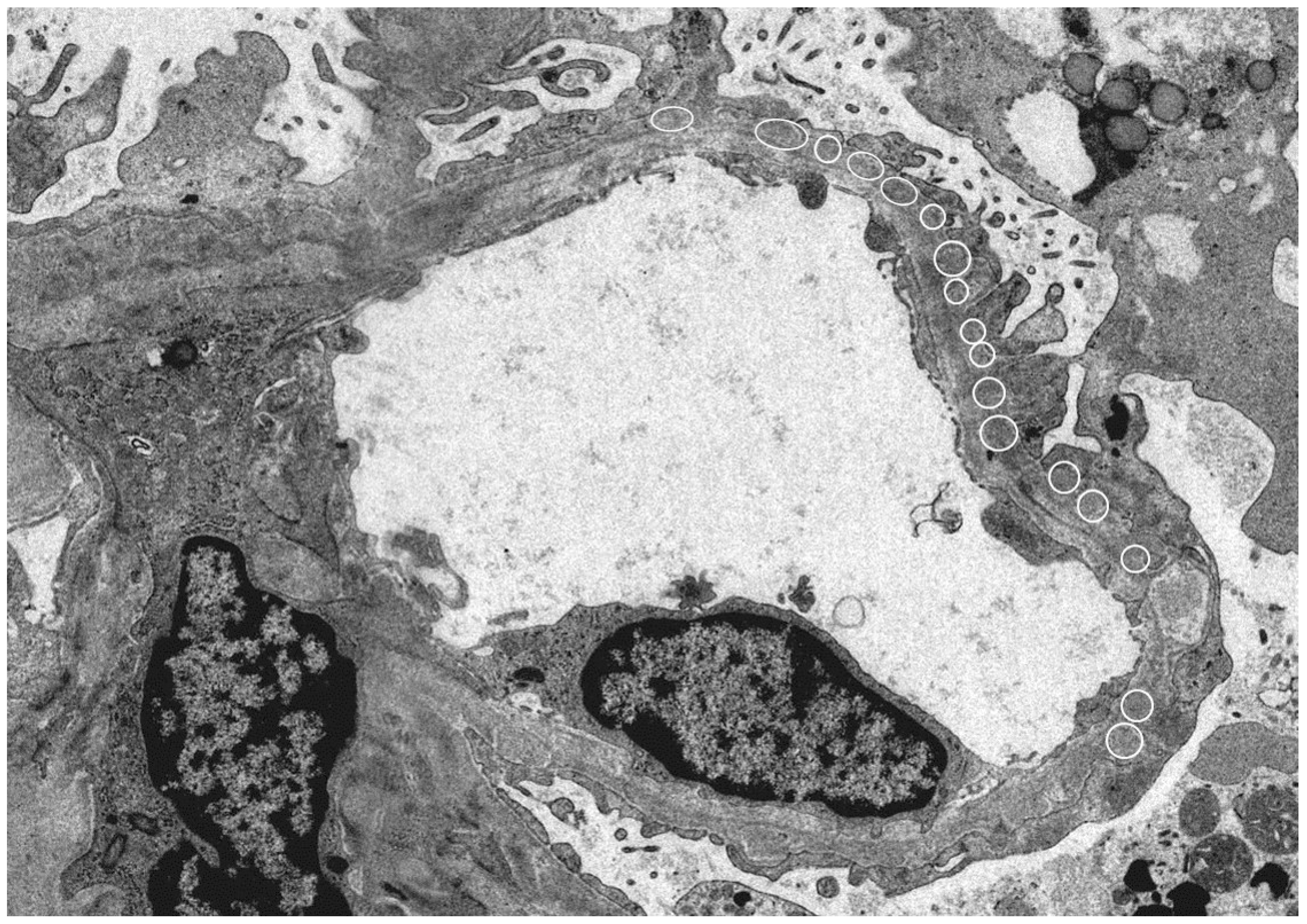
2.1.1. Membranous Nephropathy (MN)
2.1.2. Membranoproliferative Glomerulonephritis (MPGN)
2.1.3. Polyarteritis Nodosa (PAN)
2.2. Treatment of HBV-Associated Nephropathy
3. Kidney Injury with the Hepatitis B Treatment Drug
4. Hepatitis B Cirrhosis and Renal Failure
4.1. Mechanism of HRS
4.2. Treatment of HRS
5. Equivalent Renal Failure in Liver Cirrhosis
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Combes, B.; Shorey, J.; Barrera, A.; Stastny, P.; Eigenbrodt, E.H.; Hull, A.R.; Carter, N.W. Glomerulonephritis with deposition of Australia antigen-antibody complexes in glomerular basement membrane. Lancet 1971, 2, 234–237. [Google Scholar] [CrossRef]
- Kahn, J.; Lagakos, S.; Wulfsohn, M.; Cherng, D.; Miller, M.; Cherrington, J.; Hardy, D.; Beall, G.; Cooper, R.; Murphy, R.; et al. Efficacy and safety of adefovir dipivoxil with antiretroviral therapy: A randomized controlled trial. JAMA 1999, 282, 2305–2312. [Google Scholar] [CrossRef] [PubMed]
- Magalhães-Costa, P.; Matos, L.; Barreiro, P.; Chagas, C. Fanconi syndrome and chronic renal failure in a chronic hepatitis B monoinfected patient treated with tenofovir. Rev. Esp. Enferm. Dig. 2015, 107, 512–514. [Google Scholar] [PubMed]
- Lai, K.N.; Li, P.K.; Lui, S.F.; Au, T.C.; Tam, J.S.; Tong, K.L.; Lai, F.M. Membranous nephropathy related to hepatitis B virus in adults. N. Engl. J. Med. 1991, 324, 1457–1463. [Google Scholar] [CrossRef] [PubMed]
- Chan, T.M. Hepatitis B and renal disease. Curr. Hepatol. Rep. 2010, 9, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.J.; Couser, W.G. Hepatitis B infection and renal disease: Clinical, immunopathogenetic and therapeutic considerations. Kidney Int. 1990, 37, 663–676. [Google Scholar] [CrossRef] [PubMed]
- Takekoshi, Y.; Tochimaru, H.; Nagata, Y.; Itami, N. Immunopathogenetic mechanisms of hepatitis B virus-related glomerulopathy. Kidney Int. 1991, 40, 34–39. [Google Scholar]
- Conjeevaram, H.S.; Hoofnagle, J.H.; Austin, H.A.; Park, Y.; Fried, M.W.; Di Bisceglie, A.M. Long-term outcome of hepatitis B virus-related glomerulonephritis after therapy with interferon alfa. Gastroenterology 1995, 109, 540–546. [Google Scholar] [CrossRef]
- He, X.Y.; Fang, L.J.; Zhang, Y.E.; Sheng, F.Y.; Zhang, X.R.; Guo, M.Y. In situ hybridization of hepatitis B DNA in hepatitis B-associated glomerulonephritis. Pediatr. Nephrol. 1998, 12, 117–120. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.C.; Su, Y.C.; Li, C.Y.; Wu, C.P.; Lee, M.S. A nationwide cohort study suggests chronic hepatitis B virus infection increases the risk of end-stage renal disease among patients in Taiwan. Kidney Int. 2015, 87, 1030–1038. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Shen, P.; Li, X.; Xu, Y.; Pan, X.; Wang, W.; Chen, X.; Zhang, W.; Ren, H.; Chen, N. Re-evaluation of the classification system for membranoproliferative glomerulonephritis. Contrib. Nephrol. 2013, 181, 175–184. [Google Scholar] [PubMed]
- Mouthon, L.; Deblois, P.; Sauvaget, F.; Meyrier, A.; Callard, P.; Guillevin, L. Hepatitis B virus-related polyarteritis nodosa and membranous nephropathy. Am. J. Nephrol. 1995, 15, 266–269. [Google Scholar] [CrossRef] [PubMed]
- Lai, N.; Lai, F.M.; Chan, K.W.; Chow, C.B.; Tong, K.L.; Vallance-Owen, J. The clinico-pathologic features of hepatitis B virus-associated glomerulonephritis. Q. J. Med. 1987, 63, 323–333. [Google Scholar] [PubMed]
- Lai, K.N.; Tam, J.S.; Lin, H.J.; Lai, F.M. The therapeutic dilemma of the usage of corticosteroid in patients with membranous nephropathy and persistent hepatitis B virus surface antigenaemia. Nephron 1990, 54, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Shouval, D.; Shibolet, O. Immunosuppression and HBV reactivation. Semin. Liver Dis. 2013, 33, 167–177. [Google Scholar] [PubMed]
- Bhimma, R.; Coovadia, H.M.; Kramvis, A.; Adhikari, M.; Kew, M.C. Treatment of hepatitis B virus-associated nephropathy in black children. Clin. Nephrol. 1993, 40, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Knecht, G.L.; Chrisari, F.V. Reversibility of hepatitis B virus-induced glomerulonephritis and chronic active hepatitis after spontaneous clearance of serum hepatitis B surface antigen. Gastroenterology 1978, 75, 1152–1154. [Google Scholar] [PubMed]
- Elewa, U.; Sandri, A.M.; Kim, W.R.; Fervenza, F.C. Treatment of hepatitis B virus-associated nephropathy. Nephron Clin. Pract. 2011, 119, c41–c49. [Google Scholar] [CrossRef] [PubMed]
- Cooksley, W.G.; Piratvisuth, T.; Lee, S.D.; Mahachai, V.; Chao, Y.C.; Tanwandee, T.; Chutaputti, A.; Chang, W.Y.; Zahm, F.E.; Pluck, N. Peginterferon alpha-2a (40 kDa): An advance in the treatment of hepatitis B e antigen-positive chronic hepatitis B. J. Viral Hepat. 2003, 10, 298–305. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Ma, Y.; Chen, D.; Zhuo, L.; Li, W. A Meta-analysis of antiviral therapy for hepatitis B virus-associated membranous nephropathy. PLoS ONE 2016, 11, e0160437. [Google Scholar] [CrossRef] [PubMed]
- Salvaggio, S.E.; Giacomelli, A.; Falvella, F.S.; Oreni, M.L.; Meraviglia, P.; Atzori, C.; Clementi, E.G.I.; Galli, M.; Rusconi, S. Clinical and genetic factors associated with kidney tubular dysfunction in a real-life single centre cohort of HIV-positive patients. BMC Infect. Dis. 2017, 17, 396. [Google Scholar] [CrossRef] [PubMed]
- Cihlar, T.; Ho, E.S.; Lin, D.C.; Mulato, A.S. Human renal organic anion transporter 1 (hOAT1) and its role in the nephrotoxicity of antiviral nucleotide analogs. Nucleic Acids 2001, 20, 641–648. [Google Scholar] [CrossRef] [PubMed]
- Cortez, K.J.; Proschan, M.A.; Barrett, L.; Brust, D.G.; Weatherley, B.; Formentini, E.; Davey, R.T.; Masur, H.; Polis, M.A.; Neumann And, A.U.; et al. Baseline CD4+ T-cell counts predict HBV viral kinetics to adefovir treatment in lamivudine-resistant HBV-infected patients with or without HIV infection. HIV Clin. Trials 2013, 14, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Karras, A.; Lafaurie, M.; Furco, A.; Bourgarit, A.; Droz, D.; Sereni, D.; Legendre, C.; Martinez, F.; Molina, J.M. Tenofovir-related nephrotoxicity in human immunodeficiency virus-infected patients: Three cases of renal failure, Fanconi syndrome, and nephrogenic diabetes insipidus. Clin. Infect. Dis. 2003, 36, 1070–1073. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.C.; Shih, Y.F.; Liu, C.J. Viral factors affecting the clinical outcomes of chronic hepatitis B. J. Infect. Dis. 2017, 216, S757–S764. [Google Scholar] [CrossRef] [PubMed]
- Sax, P.E.; Wohl, D.; Yin, M.T.; Post, F.; DeJesus, E.; Saag, M.; Pozniak, A.; Thompson, M.; Podzamczer, D.; Molina, J.M.; et al. Tenofovir alafenamide versus tenofovir disoproxil fumarate, coformulated with elvitegravir, cobicistat, and emtricitabine, for initial treatment of HIV-1 infection: Two randomised, double-blind, phase 3, non-inferiority trials. Lancet 2015, 385, 2606–2615. [Google Scholar] [CrossRef]
- Garcia-Tsao, G.; Parikh, C.R.; Viola, A. Acute kidney injury in cirrhosis. Hepatology 2008, 48, 2064–2077. [Google Scholar] [CrossRef] [PubMed]
- Salerno, F.; Gerbes, A.; Ginès, P.; Wong, F.; Arroyo, V. Diagnosis, prevention and treatment of hepatorenal syndrome in cirrhosis. Gut 2007, 56, 1310–1318. [Google Scholar] [CrossRef] [PubMed]
- Bataller, R.; Ginès, P.; Guevara, M.; Arroyo, V. Hepatorenal syndrome. Semin. Liver Dis. 1997, 17, 233–247. [Google Scholar] [CrossRef] [PubMed]
- Wong, F.; Nadim, M.K.; Kellum, J.A.; Salerno, F.; Bellomo, R.; Gerbes, A.; Angeli, P.; Moreau, R.; Davenport, A.; Jalan, R.; et al. Working Party proposal for a revised classification system of renal dysfunction in patients with cirrhosis. Gut 2011, 60, 702–709. [Google Scholar] [CrossRef] [PubMed]
- Martin-Llahi, M.; Pépin, M.N.; Guevara, M.; Díaz, F.; Torre, A.; Monescillo, A.; Soriano, G.; Terra, C.; Fábrega, E.; Arroyo, V.; et al. Terlipressin and albumin vs. albumin in patients with cirrhosis and hepatorenal syndrome: A randomized study. Gastroenterology 2008, 134, 1352–1359. [Google Scholar] [CrossRef] [PubMed]
- Cavallin, M.; Piano, S.; Romano, A.; Fasolato, S.; Frigo, A.C.; Benetti, G.; Gola, E.; Morando, F.; Stanco, M.; Rosi, S.; et al. Terlipressin given by continuous intravenous infusion versus intravenous boluses in the treatment of hepatorenal syndrome: A randomized controlled study. Hepatology 2016, 63, 983–992. [Google Scholar] [CrossRef] [PubMed]
- De Souza, V.; Hadj-Aissa, A.; Dolomanova, O.; Rabilloud, M.; Rognant, N.; Lemoine, S.; Radenne, S.; Dumortier, J.; Chapuis-Cellier, C.; Beyerle, F. Creatinine- versus cystatine C-based equations in assessing the renal function of candidates for liver transplantation with cirrhosis. Hepatology 2014, 59, 1522–1531. [Google Scholar] [CrossRef] [PubMed]
- Mindikoglu, A.L.; Opekun, A.R.; Mitch, W.E.; Magder, L.S.; Christenson, R.H.; Dowling, T.C.; Weir, M.R.; Seliger, S.L.; Howell, C.D.; Raufman, J.P. Cystatin C is a gender-neutral glomerular filtration rate biomarker in patients with cirrhosis. Dig. Dis. Sci. 2018, 63, 665–675. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, K.; Iwasa, M.; Eguchi, A.; Kojima, S.; Yoshizawa, N.; Tempaku, M.; Sugimoto, R.; Yamamoto, N.; Sugimoto, K.; Kobayashi, Y.; et al. Neutrophil gelatinase-associated lipocalin level is a prognostic factor for survival in rat and human chronic liver diseases. Hepatol. Commun. 2017, 1, 946–956. [Google Scholar] [CrossRef] [PubMed]
- Hörstrup, J.H.; Gehrmann, M.; Schneider, B.; Plöger, A.; Froese, P.; Schirop, T.; Kampf, D.; Frei, U.; Neumann, R.; Eckardt, K.U. Elevation of serum and urine levels of TIMP-1 and tenascin in patients with renal disease. Nephrol. Dial. Transpl. 2002, 17, 1005–1013. [Google Scholar] [CrossRef]
- Waanders, F.; Vaidya, V.S.; van Goor, H.; Leuvenink, H.; Damman, K.; Hamming, I.; Bonventre, J.V.; Vogt, L.; Navis, G. Effect of renin-angiotensin-aldosterone system inhibition, dietary sodium restriction, and/or diuretics on urinary kidney injury molecule 1 excretion in nondiabetic proteinuric kidney disease: A post hoc analysis of a randomized controlled trial. Am. J. Kidney Dis. 2009, 53, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Aregger, F.; Uehlinger, D.E.; Witowski, J.; Brunisholz, R.A.; Hunziker, P.; Frey, F.J.; Jörres, A. Identification of IGFBP-7 by urinary proteomics as a novel prognostic marker in early acute kidney injury. Kidney Int. 2014, 85, 909–919. [Google Scholar] [CrossRef] [PubMed]
- Kamijo, A.; Sugaya, T.; Hikawa, A.; Yamanouchi, M.; Hirata, Y.; Ishimitsu, T.; Numabe, A.; Takagi, M.; Hayakawa, H.; Tabei, F.; et al. Urinary liver-type fatty acid binding protein as a useful biomarker in chronic kidney disease. Mol. Cell. Biochem. 2006, 284, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Parikh, C.R.; Jani, A.; Melnikov, V.Y.; Faubel, S.; Edelstein, C.L. Urinary interleukin-18 is a marker of human acute tubular necrosis. Am. J. Kidney Dis. 2004, 43, 405–414. [Google Scholar] [CrossRef] [PubMed]
| Biomarker | Pathophysiological Process | Authors | Characteristic |
|---|---|---|---|
| Cystatin C | Kidney function (GFR) | Mindikoglu, A.L. et al. [34] | Serum cystatin C avoids the limitations of glomerular filtration rate related to the patients with cirrhosis, particularly in women and those with sarcopenia. |
| Neutrophil gelatinase-associated lipocalin (NGAL) | Tubulointerstitial injury | Yoshikawa, K. et al. [35] | NGAL, a ubiquitous lipocalin iron-carrying protein, 45 is highly expressed in the tubular epithelium of the distal nephron and released from tubular epithelial cells following damage such as AKI. |
| Tissue inhibitor of metalloproteinases-2 (TIMP-2) | Inflammation | Horstrup, J.H. et al. [36] | Urinary concentrations of tenascin and TIMP-1 are elevated in association with renal disease and may reflect specific aspects of renal fibrosis. |
| Kidney injury molecule-1 (KIM-1) | Tubulointerstitial injury | Waanders, F. et al. [37] | KIM-1 is a transmembrane tubular protein with uncertain function, not detectable in the normal kidney, but elevated in experimental and clinical kidney damage. |
| IGF-binding protein-7 (IGFBP7) | Inflammation | Aregger, F. et al. [38] | The protein has been implicated in these processes it is believed editing might affect apoptosis, regulation of cell growth and angiogenesis. |
| Liver-type fatty acid binding protein (L-FABP) | Tubulointerstitial injury | Kamijo, A. et al. [39] | L-FABP is expressed in proximal tubular cells and is a biomarker of inflammation investigated in diabetes, diabetic nephropathy, hypertension, and early CKD. |
| Interleukin-18 (IL-18) | Inflammation | Chirag R. Parikh et al. [40] | IL-18 was markedly increased in patients with established AKI from ischemia, but not in patients with AKI from urinary tract infection, CKD, nephrotic syndrome, or those with prerenal failure. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kamimura, H.; Setsu, T.; Kimura, N.; Yokoo, T.; Sakamaki, A.; Kamimura, K.; Tsuchiya, A.; Takamura, M.; Yamagiwa, S.; Terai, S. Renal Impairment in Chronic Hepatitis B: A Review. Diseases 2018, 6, 52. https://doi.org/10.3390/diseases6020052
Kamimura H, Setsu T, Kimura N, Yokoo T, Sakamaki A, Kamimura K, Tsuchiya A, Takamura M, Yamagiwa S, Terai S. Renal Impairment in Chronic Hepatitis B: A Review. Diseases. 2018; 6(2):52. https://doi.org/10.3390/diseases6020052
Chicago/Turabian StyleKamimura, Hiroteru, Toru Setsu, Naruhiro Kimura, Takeshi Yokoo, Akira Sakamaki, Kenya Kamimura, Atsunori Tsuchiya, Masaaki Takamura, Satoshi Yamagiwa, and Shuji Terai. 2018. "Renal Impairment in Chronic Hepatitis B: A Review" Diseases 6, no. 2: 52. https://doi.org/10.3390/diseases6020052
APA StyleKamimura, H., Setsu, T., Kimura, N., Yokoo, T., Sakamaki, A., Kamimura, K., Tsuchiya, A., Takamura, M., Yamagiwa, S., & Terai, S. (2018). Renal Impairment in Chronic Hepatitis B: A Review. Diseases, 6(2), 52. https://doi.org/10.3390/diseases6020052





