Phytoconstituents with Radical Scavenging and Cytotoxic Activities from Diospyros shimbaensis
Abstract
:1. Introduction
2. Results and Discussion
2.1. Isolation and Identification of Compounds
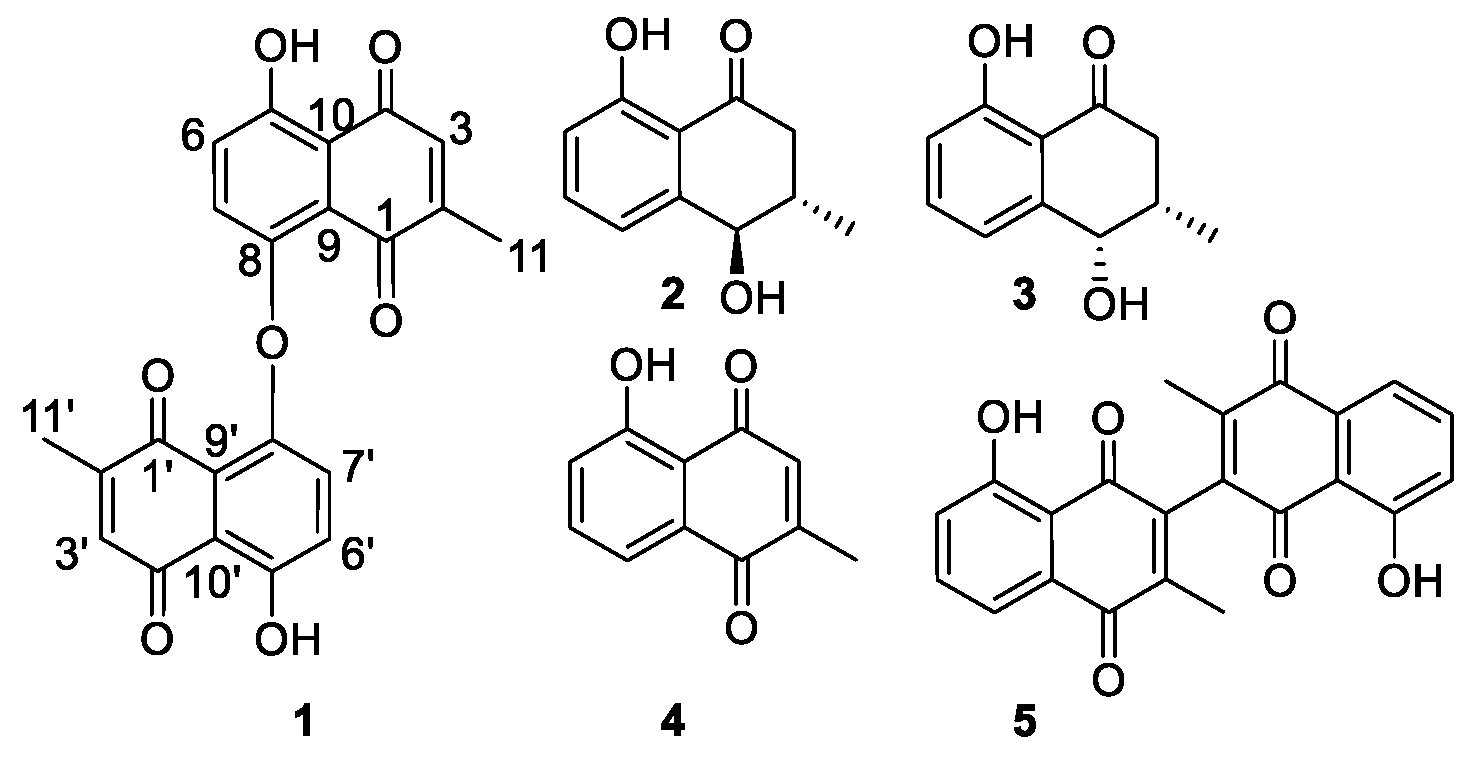
| Position | δH (I, m, J in Hz) | δC | HMBC (2J, 3J) |
|---|---|---|---|
| 1,1′ | - | 185.2 | - |
| 2,2′ | - | 150.0 | - |
| 3,3′ | 6.80, (2H, q, 1.6) | 134.9 | C1, C1′, C10, C10′, C11, C11′ |
| 4,4′ | - | 190.5 | - |
| 5,5′ | - | 161.3 | - |
| 6,6′ | 7.29, (2H, d, 8.6) | 124.3 | C10, C10′, C5, C5′, C7, C7′, C8, C8′ |
| 7,7′ | 7.20 (2H, d, 8.6) | 137.9 | C5, C5′, C6, C6′, C8, C8′, C9,C9′ |
| 8,8′ | - | 135.6 | - |
| 9, 9′ | - | 128.2 | - |
| 10, 10′ | - | 115.5 | - |
| 11,11′ | 2.01 (6H, d, 1.6) | C1, C1′, C2, C2′, C3, C3′ | |
| 5-OH, 5′-OH | 12.58 (2H, brs) | - | C10, C10′, C5, C5′, C6, C6′ |

2.2. Cytotoxicity and Radical Scavenging Activities
| Tested Compound/Extract | Cytotoxicity IC50 a | RSA EC50 b (µM) |
|---|---|---|
| 8,8′-Oxo-biplumbagin (1) | NT | >25 |
| trans-Isoshinanolone (2) | >520 | >25 |
| cis-Isoshinanolone (3) | NT | 12.5 |
| Plumbagin (4) | 130.8 | >25 |
| 3,3′-Biplumbagin (5) | 82.1 | >25 |
| Methanol crude extract from the stem barks | 59.6 | NT |
| Isohexane crude extract from the root barks | 16.1 | NT |
| Dichloromethane crude extract from the root barks | 29.7 | NT |
| Methanol crude extract from the root barks | >100 | NT |
| Dichloromethane crude extract from the leaves | 73.0 | NT |
| Methanol crude extract from the leaves | 44.7 | NT |
3. Experimental Section
3.1. General Information
3.2. Plant Materials
3.3. Extraction and Isolation
3.4. Cytotoxicity Assay
3.5. Radical Scavenging Activities Assay
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Thuong, P.T.; Lee, C.H.; Dao, T.T.; Nguyen, P.H.; Kim, W.G.; Lee, S.J.; Oh, W.K. Triterpenoids from the leaves of Diospyros kaki (Perimmon) and their inhibitory effects on protein tyrosine phosphatase 1B. J. Nat. Prod. 2008, 71, 1775–1778. [Google Scholar] [CrossRef] [PubMed]
- Li, X.-C.; van der Bijl, P.; Wu, C.D. Binaphthalenone glycosides from African chewing sticks, Diospyros lycioides. J. Nat. Prod. 1998, 61, 817–820. [Google Scholar] [CrossRef] [PubMed]
- Cai, L.; Wei, G.-X.; van der Bijl, P.; Wu, C.D. Namibian chewing stick, Diospyros lycioides, contains antibacterial compounds against oral pathogens. J. Agric. Food Chem. 2000, 48, 909–914. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.-G.; Graf, T.N.; Lee, D.; Chai, H.-G.; Mi, Q.; Kardono, L.B.S.; Setyowati, F.M.; Ismail, R.; Riswan, S.; Farnsworth, N.R.; et al. Cytotoxic and antimicrobial constituents of the bark of Diospyros maritima collected in two geographical locations in Indonesia. J. Nat. Prod. 2004, 67, 1156–1161. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.-I.; Chen, C.-R.; Chiu, H.-L.; Kuo, C.-L.; Kuo, Y.-H. Chemical constituents from the stems of Diospyros maritima. Molecules 2009, 14, 5281–5288. [Google Scholar] [CrossRef] [PubMed]
- Mallavadhani, U.V.; Panda, A.K.; Rao, Y.R. Pharmacology and chemotaxonomy of Diospyros. Phytochemistry 1998, 49, 901–951. [Google Scholar] [CrossRef]
- Watt, J.M.; Breyer-Brandwijk, M.G. Medicinal and Poisonous Plants of Southern and Eastern Africa; E&S Livingstone limited: Edinburgh, UK, 1962. [Google Scholar]
- Kokwaro, J.O. Medicinal Plants of East Africa; Kenya Literature Bureau: Nairobi, Kenya, 1993. [Google Scholar]
- Chhabra, S.C.; Mahunnah, R.L.A.; Mshiu, E.N. Plants used in traditional medicine in Eastern Tanzania. II. Angiospersm (Capparidaceae to Ebenaceae). J. Ethnopharmacol. 1989, 25, 339–359. [Google Scholar] [CrossRef]
- Hook, I.; Mills, C.; Sheridan, H. Bioactive naphthoquinones from higher plants. Studies Nat. Prod. Chem. 2014, 41, 119–160. [Google Scholar]
- Wallnöfer, B. The biology and systematics of Ebenaceae: A review. Ann. Naturhist. Mus. Wien. 2001, 103B, 485–512. [Google Scholar]
- Diospyros shimbaensis. The IUCN red list of threatened species 1998: e.T33174A9756690. Available online: http://dx.doi.org/10.2305/IUCN.UK.1998.RLTS.T33174A9756690.en (accessed on 13 November 2015).
- Bringmann, G.; Wohlfarth, M.; Rischer, H.; Ruckert, M.; Schlauer, J. The polyketide folding mode in the biogenesis of isoshinanolone and plumbagin from Ancistrocladus heyneanus (Ancistrocladaceae). Tetrahedron Lett. 1998, 39, 8445–8448. [Google Scholar] [CrossRef]
- Fournet, A.; Angelo, A.; Muñoz, V.; Roblot, F.; Hocquemiller, R.; Cavé, A. Biological and chemical studies of Pera benensis, a Bolivian plant used in folk medicine as a treatment of cutaneous leishmaniasis. J. Ethnopharmacol. 1992, 37, 159–164. [Google Scholar] [CrossRef]
- Uc-Cachón, A.H.; Borges-Argáez, R.; Said-Fernández, S.; Vargas-Villarreal, J.; González-Salazar, F.; Méndez-González, M.; Cáceres-Farfán, M.; Molina-Salinas, G.M. Naphthoquinones isolated from Diospyros anisandra exhibit potent activity against pan-resistant first-line drugs Mycobacterium tuberculosis strains. Pulm. Pharmacol. Ther. 2014, 27, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Yue, J.-M.; Xu, J.; Zhao, Y.; Sun, H.-D.; Lin, Z.-W. Chemical components from Ceratostigma willmottianum. J. Nat. Prod. 1997, 60, 1031–1033. [Google Scholar] [CrossRef]
- Anh, NH.; Ripperger, H.; Porzel, A.; van Sung, T.; Adam, G. Tetralones from Ancistrocladus cochinchinensis. Phytochemistry 1997, 41, 549–551. [Google Scholar]
- Gunaherath, G.M.K.B.; Gunatilaka, A.A.L.; Sultanbawa, M.U.S.; Balasubramaniam, S. 1,2(3)-Tetrahydro-3,3′-biplumbagin: A naphthalenone and other constituents from Plumbago zeylanica. Phytochemistry 1983, 22, 1245–1247. [Google Scholar] [CrossRef]
- Zakaria, B.M.; Jeffreys, J.A.D.; Waterman, P.G.; Zhong, S.-M. Naphthoquinones and triterpenes from some Asian Diospyros species. Phytochemistry 1984, 23, 1481–1484. [Google Scholar] [CrossRef]
- Kumar, V.; Meepagala, K.M.; Balasubramaniam, S. Quinonoid and other constituents of Aristea ecklonii. Phytochemistry 1985, 24, 1118–1119. [Google Scholar] [CrossRef]
- Bhattacharyya, J.; de Carvalho, V.R. Epi-isoshinanolone from Plumbago scandens. Phytochemistry 1986, 25, 764–765. [Google Scholar] [CrossRef]
- Zhong, S.-M.; Waterman, P.G.; Jeffreys, J.A.D. Naphthoquinones and triterpenes from African Diospyros species. Phytochemistry 1984, 23, 1067–1072. [Google Scholar] [CrossRef]
- Hanson, S.W.; Crawford, M.; Thanasingh, D.P.J. (+)-Isoshinanolone and 2-methylbenzofuran-4-carbaldehyde from the fish-stunning plant Habropetalum dawei. Phytochemistry 1981, 20, 1162–1164. [Google Scholar] [CrossRef]
- Bringmann, G.; Münchbach, M.; Messer, K.; Koppler, D.; Michel, M.; Schupp, O.; Wenzel, M.; Louis, A.M. cis- and trans-Isoshinanolone from Dioncophyllum thollonii: Absolute configuration of two ′known′, wide-spread natural products. Phytochemistry 1999, 51, 693–699. [Google Scholar]
- Tezuka, M.; Takahashi, C.; Kuroyanagi, M.; Satake, M.; Yoshihira, K.; Natori, S. New naphthoquinones from Diospyros. Phytochemistry 1973, 12, 175–183. [Google Scholar] [CrossRef]
- Sreelatha, T.; Hymavathi, A.; Murthy, M.J.; Rani, P.U.; Rao, J.M.; Babu, K.S. Bioactivity-guided isolation of mosquitocidal constituents from the rhizomes of Plumbago capensis Thunb. Bioorg. Med. Chem. Lett. 2010, 20, 2974–2977. [Google Scholar] [CrossRef] [PubMed]
- Kumagai, Y.; Shinkai, Y.; Miura, T.; Cho, A.K. The chemical biology of naphthoquinones and its environmental implications. Annu. Rev. Pharmacol. Toxicol. 2012, 52, 221–247. [Google Scholar] [CrossRef] [PubMed]
- Padhye, S.; Dandawate, P.; Yusufi, M.; Ahmad, A.; Sarkar, F.H. Perspectives on medicinal properties of plumbagin and its analogs. Med. Res. Rev. 2012, 32, 1131–1158. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.; Banerjee, S.; Wang, Z.; Kong, D.; Sarkar, F.H. Plumbagin-induced apoptosis of human breast cancer cells is mediated by inactivation of NF-κB and Bcl-2. J. Cell. Biochem. 2008, 105, 1461–1471. [Google Scholar] [CrossRef] [PubMed]
- Kuo, P.-L.; Hsu, Y.-L.; Cho, C.-Y. Plumbagin induces G2-M arrest and autophagy by inhibiting the AKT/mammalian target of rapamycin pathway in breast cancer cells. Mol. Cancer Ther. 2006, 12, 3209–3221. [Google Scholar] [CrossRef] [PubMed]
- Cicero, D.O.; Barbato, G.; Bazzo, R. NMR analysis of molecular flexibility in solution: A new method for the study of complex distributions of rapidly exchanging conformations. Application to a 13-residue peptide with an 8-residue loop. J. Am. Chem. Soc. 1995, 117, 1027–1033. [Google Scholar] [CrossRef]
- Erdelyi, M.; Pfeiffer, B.; Hauenstein, K.K.; Forher, J.; Gertsch, J.; Altmann, K.H.; Carlomagno, T. Conformational preferences of natural and C3-modified epothilones in aqueous solution. J. Med. Chem. 2008, 51, 1469–1473. [Google Scholar] [CrossRef] [PubMed]
- Danelius, E.; Brath, U.; Erdelyi, M. Insight into beta-hairpin stability: Intrastrand hydrogen bonding. Synlett 2013, 24, 2407–2410. [Google Scholar]
- Andersson, H.; Demaegdt, H.; Vauquelin, G.; Lindeberg, G.; Karlén, A.; Hallberg, M.; Erdelyi, M.; Hallberg, A.J. Disulfide cyclized tripeptide analogues of angiotensin IV as potent and selective inhibitors of insulin-regulated aminopeptidase (IRAP). J. Med. Chem. 2010, 53, 8059–8071. [Google Scholar] [CrossRef] [PubMed]
- Friden-Saxin, M.; Seifert, T.; Hansen, L.K.; Grötli, M.; Erdelyi, M.; Luthman, K. Proline-mediated formation of novel chroman-4-one tetrahydropyrimidines. Tetrahedron 2012, 68, 7035–7040. [Google Scholar] [CrossRef]
- Monks, T.J.; Jones, D.C. The metabolism and toxicity of quinones, quinonimines, quinine methides and quinone-thioethers. Curr. Drug Metable 2002, 3, 425–438. [Google Scholar] [CrossRef]
- Huang, Q.; Lu, G.; Sben, H.M.; Cbung, M.C.M.; Ong, C.N. Anti-cancer properties of anthraquinones from rhubarb. Med. Res. Rev. 2007, 27, 609–630. [Google Scholar] [CrossRef] [PubMed]
- Endale, M.; Alao, J.P.; Akala, H.M.; Rono, N.K.; Eyase, F.L.; Derese, S.; Ndakala, A.; Mbugua, M.; Walsh, D.S.; Sunnerhagen, P.; et al. Antiplasmodial quinones from Pentas longiflora and Pentas lanceolata. Planta Med. 2012, 78, 31–35. [Google Scholar] [CrossRef] [PubMed]
- Queiroz, M.L.; Valadares, M.C.; Torello, C.O.; Ramos, A.L.; Oliveira, A.B.; Rocha, F.D.; Arruda, V.A.; Accorci, W.R. Comparative studies of the effects of Tabebuia avellanedae bark extract and β-lapachone on the hematopoietic response of tumour-bearing mice. J. Ethnopharmacol. 2008, 117, 228–235. [Google Scholar] [CrossRef] [PubMed]
- Lim, E.S.; Rhee, Y.H.; Park, M.K.; Shim, B.S.; Ahn, K.S.; Kang, H.; Yoo, H.S.; Kim, S.H. DMNQ S-64 induces apoptosis via caspase activation and cyclooxygenase-2 inhibition in human non-small lung cancer cells. Ann. N. Y. Acad. Sci. 2007, 1095, 7–18. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.J.; Bao, J.L.; Wu, G.S.; Xu, W.S.; Huang, M.Q.; Chen, X.P.; Wang, Y.T. Quinones derived from plant secondary metabolites as anti-cancer agents. Anticancer Agents Med. Chem. 2013, 13, 456–463. [Google Scholar] [CrossRef] [PubMed]
- Sinha, S.; Pal, K.; Elkhanany, A.; Dutta, S.; Cao, Y.; Mondal, G.; Iyer, S.; Somasundaram, V.; Couch, F.J.; Shridhar, V. Plumbagin inhibits tumorigenesis and angiogenesis of ovarian cancer cells in vivo. Int. J. Cancer 2013, 132, 1201–1212. [Google Scholar] [CrossRef] [PubMed]
- Fujii, N.; Yamashita, Y.; Arima, Y.; Nagashima, M.; Nakano, H. Induction of topoisomerase II-mediated DNA cleavage by the plant naphthoquinones, plumbagin and shikonin. Antimicrob. Agents Chemother. 1992, 36, 2589–2594. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-H.; Yeon, J.-H.; Kim, H.; Roh, W.; Chae, J.; Park, H.-O.; Kim, D.-M. The natural anticancer agent plumbagin induces potent cytotoxicity in MCF-7 human breast cancer cells by inhibiting a PI-5 kinase for ROS generation. PLoS ONE 2012, 7, e45023. [Google Scholar] [CrossRef] [PubMed]
- Hussein, M.A. A convenient mechanism for the free radical scavenging activity of resveratrol. Int. J. Phytomed. 2011, 3, 459–469. [Google Scholar]
- Hsieh, T.-C.; Elangovan, S.; Wu, J.M. Differential suppression of proliferation in MCF-7 and MDA-MB-231 breast cancer cells exposed to α-, γ- and δ-tocotrienols is accompanied by altered expression of oxidative stress modulatory enzymes. Anticancer Res. 2010, 30, 4169–4176. [Google Scholar] [PubMed]
- Srinivas, P.; Patra, C.R.; Bhattacharya, S.; Mukhopadhyay, D. Cytotoxicity of naphthoquinones and their capacity to generate reactive oxygen species is quenched when conjugated with gold nanoparticles. Int. J. Nanonmed. 2011, 6, 2113–2122. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, A.-S.H.; Mohammed, A.S.; Rasedee, A.; Mirghani, M.E.S.; Al-Qubaisi, M.S. Induction of apoptosis and oxidative stress in estrogen receptor-negative breast cancer, MDA-MB231 cells, by ethanolic mango seed extract. BMC Complement. Altern. Med. 2010, 15, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Dinicola, S.; Cucina, A.; Antonacci, D.; Bizzarri, M. Anticancer effects of grape seed extract on human cancers: A review. J. Carcinog. Mutagen. 2014. [Google Scholar] [CrossRef]
- Wokaun, A.; Ernst, R.R. Selective detection of multiple quantum transitions in NMR by 2-dimensional spectroscopy. Chem. Phys. Lett. 1977, 52, 407–412. [Google Scholar] [CrossRef]
- Kumar, A.; Ernst, R.R.; Wüthrich, K. A two-dimensional nuclear Overhauser enhancement (2D NOE) experiment for the elucidation of complete proton-proton cross-relaxation networks in biological macromolecules. Biochem. Biophys. Res. Commun. 1980, 95, 1–6. [Google Scholar] [CrossRef]
- Hurd, R.E.; John, B.K. Gradient-enhanced proton-detected heteronuclear multiple-quantum coherence spectroscopy. J. Magn. Reson. 1991, 91, 648–653. [Google Scholar] [CrossRef]
- Perpickdumont, M.; Reynolds, W.F.; Enriquez, R.G. 13C—1H shift correlation with full 1h—1H decoupling. Magn. Reson. Chem. 1988, 26, 358–361. [Google Scholar] [CrossRef]
- Negera, A.; Induli, M.; Fitzpatrick, P.; Alao, J.P.; Sunnerhagen, P.; Landberg, G.; Yenesew, A.; Erdélyi, M. Cytotoxic quinones from the roots of Aloe dawei. Molecules 2014, 19, 3264–3273. [Google Scholar]
- Irungu, B.N.; Orwa, J.A.; Gruhonjic, A.; Fitzpatrick, P.A.; Landberg, G.; Kimani, F.; Midiwo, J.; Erdélyi, M.; Yenesew, A. Constituents of the roots and leaves of Ekebergia capensis and their potential antiplasmodial and cytotoxic activities. Molecules 2014, 19, 14235–14246. [Google Scholar] [CrossRef] [PubMed]
- Ohnishi, S.T.; Kojima, R. In vitro antioxidant activity of an aged garlic extract. FASEB J. 1994, 8, A596. [Google Scholar]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aronsson, P.; Munissi, J.J.E.; Gruhonjic, A.; Fitzpatrick, P.A.; Landberg, G.; Nyandoro, S.S.; Erdelyi, M. Phytoconstituents with Radical Scavenging and Cytotoxic Activities from Diospyros shimbaensis. Diseases 2016, 4, 3. https://doi.org/10.3390/diseases4010003
Aronsson P, Munissi JJE, Gruhonjic A, Fitzpatrick PA, Landberg G, Nyandoro SS, Erdelyi M. Phytoconstituents with Radical Scavenging and Cytotoxic Activities from Diospyros shimbaensis. Diseases. 2016; 4(1):3. https://doi.org/10.3390/diseases4010003
Chicago/Turabian StyleAronsson, Per, Joan J. E. Munissi, Amra Gruhonjic, Paul A. Fitzpatrick, Göran Landberg, Stephen S. Nyandoro, and Mate Erdelyi. 2016. "Phytoconstituents with Radical Scavenging and Cytotoxic Activities from Diospyros shimbaensis" Diseases 4, no. 1: 3. https://doi.org/10.3390/diseases4010003
APA StyleAronsson, P., Munissi, J. J. E., Gruhonjic, A., Fitzpatrick, P. A., Landberg, G., Nyandoro, S. S., & Erdelyi, M. (2016). Phytoconstituents with Radical Scavenging and Cytotoxic Activities from Diospyros shimbaensis. Diseases, 4(1), 3. https://doi.org/10.3390/diseases4010003






