Incidental Cardiac Uptake on 99mTc-HMDP Bone Scintigraphy in Oncology Patients: Two Cases of Transthyretin Amyloid Cardiomyopathy with Literature Review
Abstract
1. Introduction
2. Aim and Methods of the Present Review
3. Case Report
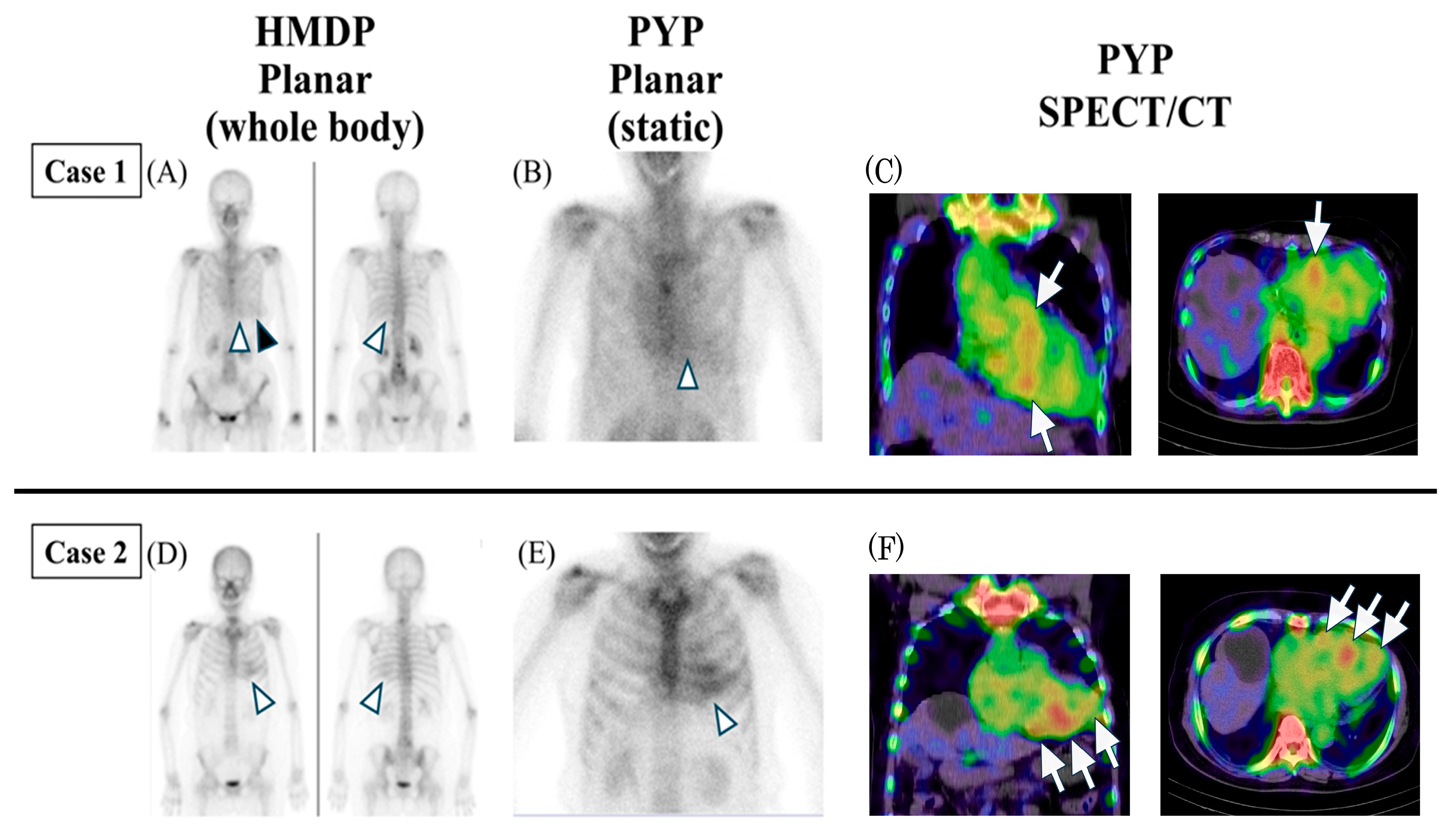
3.1. Case 1
3.2. Case 2
4. Review of Cohort Studies and Case Reports on Incidental Cardiac Uptake for Bone Scintigraphy in Oncology
4.1. Diagnostic Role of Bone Scintigraphy in ATTR-CM
| Authors [Ref.] | Tracers | Age (Years) | Men | Cardiac Uptake (Grade ≥ 2) in Overall Patients | Oncology Patients | Cardiac Uptake in Oncology Patients |
|---|---|---|---|---|---|---|
| Suomalainen et al. [5] | 99mTc-HMDP | 78 ± 6 | 69% | 3.5% (69/2000) | 90% | 3.6% (65/1810) |
| Halme et al. [13] | 99mTc-HMDP | 77 ± 10 | 82% | 3.5% (47/1334) | 94% | 2.9% (36/1253) |
| Mohamed-Salem et al. [14] | 99mTc-HMDP 99mTc-HDP 99mTc-DPD | 80 (77–83) | 65% | 2.8% (31/1114) | 95.7% | Not reported |
| Navarro-Saez et al. [15] | 99mTc-DPD | 78 (74–84) | 51% | 2.3% (82/3629) | 60% | Not reported |
| Nitsche et al. [16] | 99mTc-DPD | 64 (51–73) | 37% | 1.5% (167/11,527) | 73.6% | Not reported |
| de Haro Del Moral et al. [17] | 99mTc-HMDP 99mTc-HDP 99mTc-DPD 99mTc-PYP | 83.2 ± 6.1 | 85.9% | 0.55% (54/9864) | 91.5% | Not reported |
| Bianco et al. [18] | 99mTc-HDP 99mTc-DPD | 83 ± 5 | 78% | 0.54% (23/4228) | 47.9% | Not reported |
| Cuscaden et al. [19] † | 99mTc-HMDP | – | 53% | 0.43% (15/3472) | Not specified | Not reported |
| Longhi et al. [20] | 99mTc-DPD | 74 (65–82) | 37% | 0.36% (45/12,400) | 95% | Not reported |
| Salvalaggio et al. [21] ‡ | 99mTc-HMDP 99mTc-DPD | 79 ± 8 | – | 0.21% (20/9616) | Not specified | Not reported |
| Son et al. [22] | 99mTc-DPD 99mTc-HMDP | 55 ± 17 | 54% | 0.07% (23/32,245) | 53% | Not reported |
| Kim et al. [23] § | 99mTc-DPD | – | – | 0.06% (6/9580) | Not specified | Not reported |
| Authors [Ref.] | Tracers | Age (Years) | Gender | Oncology Subtype | Amyloid Subtype |
|---|---|---|---|---|---|
| Tanaka H et al. [25] | 99mTc-HMDP | 73 | Men | Lung cancer | ATTRwt |
| Delaney et al. [26] | 99mTc-DPD | 77 | Men | Prostate cancer | ATTR |
| Chono T et al. [27] | 99mTc-HMDP | 70s | Men | Prostate cancer | ATTR |
| Lu Y et al. [28] | 99mTc-MDP | 68 | Men | Prostate cancer | ATTR |
| Fathala A. [29] | 99mTc-MDP | 86 | Men | Prostate cancer | ATTR |
| Lin et al. [30] | 99mTc-MDP | 63 | Men | Lung cancer | AL |
| Ikebe et al. [31] | 99mTc-HMDP | 83 | Men | Prostate cancer | ATTRwt |
4.2. Prevalence of Incidental Cardiac Uptake on Bone Scintigraphy
4.3. Prevalence and Predictors of Incidental Cardiac Uptake in Cancer Patients
4.4. Diagnostic Limitations of 99mTc-MDP for ATTR-CM
5. Serial Bone Scintigraphy in Oncology Patients: Early Signs of Cardiac Amyloidosis and Diagnostic Pitfalls
6. Thromboembolic Risk and Stroke in ATTR-CM: Clinical Implications and the Role of LAAC
7. Current and Emerging Therapies for ATTR-CM
8. Management of Heart Failure in ATTR-CM
9. Early Diagnosis as a Catalyst for Social Impact: Improving Health Outcomes and Reducing Care Burden
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 99mTc-HMDP | Technetium-99m hydroxymethylene diphosphonate |
| ATTRwt-CM | Wild-type transthyretin cardiac amyloidosis |
| 99mTc-PYP | Technetium-99m pyrophosphate |
| 99mTc-DPD | Technetium-99m-3,3-diphosphono-1,2- propanodicarboxylic acid |
| ATTR-CM | Transthyretin amyloid cardiomyopathy |
| siRNA | Small interfering RNA |
| AF | Atrial fibrillation |
| hsTnT | High-sensitivity troponin T |
| NT-proBNP | N-terminal pro-brain natriuretic peptide |
| FLC | Free light chains |
| SPECT/CT | Single-photon emission computed tomography/computed tomography |
| LAAC | Left atrial appendage closure |
| 99mTc-MDP | Technetium-99m methylene diphosphonate |
| TTR | Transthyretin |
| ASO | Antisense oligonucleotide |
| CRISPR-Cas9 | Clustered regularly interspaced short palindromic repeats and associated Cas9 endonuclease |
| H/CL | Heart-to-contralateral |
| EF | Ejection fraction |
| MRA | Mineralocorticoid receptor antagonists |
| ARNI | Angiotensin receptor–neprilysin inhibitors |
| ARB | Angiotensin receptor blockers |
| SGLT2i | Sodium–glucose cotransporter 2 inhibitors |
| AQP | Aquaporin |
| QoL | Quality of life |
References
- Ruberg, F.L.; Grogan, M.; Hanna, M.; Kelly, J.W.; Maurer, M.S. Transthyretin Amyloid Cardiomyopathy: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2019, 73, 2872–2891. [Google Scholar] [CrossRef]
- Ungericht, M.; Groaz, V.; Messner, M.; Schuetz, T.; Brunelli, L.; Zaruba, M.M.; Lener, D.; Stocker, E.; Bauer, A.; Kroiss, A.S.; et al. Correlation of 99mTc-DPD bone scintigraphy with histological amyloid load in patients with ATTR cardiac amyloidosis. Amyloid 2024, 31, 22–31. [Google Scholar] [CrossRef]
- Dorbala, S.; Ando, Y.; Bokhari, S.; Dispenzieri, A.; Falk, R.H.; Ferrari, V.A.; Fontana, M.; Gheysens, O.; Gillmore, J.D.; Glaudemans, A.; et al. ASNC/AHA/ASE/EANM/HFSA/ISA/SCMR/SNMMI Expert Consensus Recommendations for Multimodality Imaging in Cardiac Amyloidosis: Part 1 of 2-Evidence Base and Standardized Methods of Imaging. Circ. Cardiovasc. Imaging 2021, 14, e000029. [Google Scholar] [CrossRef]
- Van den Wyngaert, T.; Strobel, K.; Kampen, W.U.; Kuwert, T.; van der Bruggen, W.; Mohan, H.K.; Gnanasegaran, G.; Delgado-Bolton, R.; Weber, W.A.; Beheshti, M.; et al. The EANM practice guidelines for bone scintigraphy. Eur. J. Nucl. Med. Mol. Imaging 2016, 43, 1723–1738. [Google Scholar] [CrossRef] [PubMed]
- Suomalainen, O.; Pilv, J.; Loimaala, A.; Mätzke, S.; Heliö, T.; Uusitalo, V. Prognostic significance of incidental suspected transthyretin amyloidosis on routine bone scintigraphy. J. Nucl. Cardiol. 2022, 29, 1021–1029. [Google Scholar] [CrossRef]
- Engebretsen, I.; Oteiza, F.; Birkelund, E.F.; Brandal, S.M.; Bugge, C.; Halvorsen, S. Cardiovascular disease in breast cancer patients: A nationwide real-world evidence study 2013-20. Eur. Heart J. Open 2025, 5, oeaf043. [Google Scholar] [CrossRef]
- Weiner, A.B.; Li, E.V.; Desai, A.S.; Press, D.J.; Schaeffer, E.M. Cause of death during prostate cancer survivorship: A contemporary, US population-based analysis. Cancer 2021, 127, 2895–2904. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Pavia, P.; Rapezzi, C.; Adler, Y.; Arad, M.; Basso, C.; Brucato, A.; Burazor, I.; Caforio, A.L.P.; Damy, T.; Eriksson, U.; et al. Diagnosis and treatment of cardiac amyloidosis: A position statement of the ESC Working Group on Myocardial and Pericardial Diseases. Eur. Heart J. 2021, 42, 1554–1568. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.G.M.; Li, P.; Badrish, N.; Kesari, A.; Shah, K.B. Transthyretin Cardiac Amyloidosis: Current and Emerging Therapies. Curr. Cardiol. Rep. 2025, 27, 33. [Google Scholar] [CrossRef]
- Vogel, J.; Jura, S.; Settelmeier, S.; Buehning, F.; Lerchner, T.; Carpinteiro, A.; Rassaf, T.; Michel, L. Delays in diagnosis and treatment of ATTR cardiac amyloidosis: A real-world data analysis. ESC Heart Fail. 2025, 12, 2969–2975. [Google Scholar] [CrossRef]
- Stats, M.A.; Stone, J.R. Varying levels of small microcalcifications and macrophages in ATTR and AL cardiac amyloidosis: Implications for utilizing nuclear medicine studies to subtype amyloidosis. Cardiovasc. Pathol. 2016, 25, 413–417. [Google Scholar] [CrossRef]
- Pilebro, B.; Suhr, O.B.; Näslund, U.; Westermark, P.; Lindqvist, P.; Sundström, T. (99m)Tc-DPD uptake reflects amyloid fibril composition in hereditary transthyretin amyloidosis. Ups. J. Med. Sci. 2016, 121, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Halme, H.L.; Ihalainen, T.; Suomalainen, O.; Loimaala, A.; Mätzke, S.; Uusitalo, V.; Sipilä, O.; Hippeläinen, E. Convolutional neural networks for detection of transthyretin amyloidosis in 2D scintigraphy images. EJNMMI Res. 2022, 12, 27. [Google Scholar] [CrossRef]
- Mohamed-Salem, L.; Santos-Mateo, J.J.; Sanchez-Serna, J.; Hernández-Vicente, Á.; Reyes-Marle, R.; Castellón Sánchez, M.I.; Claver-Valderas, M.A.; Gonzalez-Vioque, E.; Haro-Del Moral, F.J.; García-Pavía, P.; et al. Prevalence of wild type ATTR assessed as myocardial uptake in bone scan in the elderly population. Int. J. Cardiol. 2018, 270, 192–196. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Saez, M.D.C.; Feijoo-Massó, C.; Bravo Ferrer, Z.D.C.; Oliva Morera, J.C.; Balado González, A.M.; Palau-Domínguez, A.; Guillamon Toran, L.; Comet Monte, R.; Fernández-Codina, A. Trends in diagnosis of cardiac transthyretin amyloidosis: 3-year analysis of scintigraphic studies: Prevalence of myocardial uptake and its predictor factors. Int. J. Cardiovasc. Imaging 2023, 39, 1397–1404. [Google Scholar] [CrossRef]
- Nitsche, C.; Mascherbauer, K.; Calabretta, R.; Koschutnik, M.; Dona, C.; Dannenberg, V.; Hofer, F.; Halavina, K.; Kammerlander, A.A.; Traub-Weidinger, T.; et al. Prevalence and Outcomes of Cardiac Amyloidosis in All-Comer Referrals for Bone Scintigraphy. J. Nucl. Med. 2022, 63, 1906–1911. [Google Scholar] [CrossRef]
- de Haro Del Moral, F.J.; Aguadé Bruix, S.; Tabuenca Mateo, M.J.; Pilar Tamayo Alonso, M.; Mohamed Salem, L.; Bernal, L.; Primiano, D.; Tarilonte, P. Assessment of incidental cardiac uptake in bone scintigraphy across Spain: The ECCINGO study. Rev. Esp. Med. Nucl. Imagen. Mol. (Engl. Ed.) 2024, 43, 500020. [Google Scholar] [CrossRef]
- Bianco, M.; Parente, A.; Biolè, C.; Righetti, C.; Spirito, A.; Luciano, A.; Destefanis, P.; Nangeroni, G.; Angusti, T.; Anselmino, M.; et al. The prevalence of TTR cardiac amyloidosis among patients undergoing bone scintigraphy. J. Nucl. Cardiol. 2021, 28, 825–830. [Google Scholar] [CrossRef]
- Cuscaden, C.; Ramsay, S.C.; Prasad, S.; Goodwin, B.; Smith, J. Estimation of prevalence of transthyretin (ATTR) cardiac amyloidosis in an Australian subpopulation using bone scans with echocardiography and clinical correlation. J. Nucl. Cardiol. 2021, 28, 2845–2856. [Google Scholar] [CrossRef] [PubMed]
- Longhi, S.; Guidalotti, P.L.; Quarta, C.C.; Gagliardi, C.; Milandri, A.; Lorenzini, M.; Potena, L.; Leone, O.; Bartolomei, I.; Pastorelli, F.; et al. Identification of TTR-related subclinical amyloidosis with 99mTc-DPD scintigraphy. JACC Cardiovasc. Imaging 2014, 7, 531–532. [Google Scholar] [CrossRef]
- Salvalaggio, A.; Cipriani, A.; Righetto, S.; Artioli, P.; Sinigiani, G.; De Michieli, L.; Cason, M.; Pilichou, K.; Cecchin, D.; Briani, C. Incidental cardiac uptake of (99m)Tc-diphosphonates is predictive of poor outcome: Data from 9616 bone scintigraphies. J. Nucl. Cardiol 2022, 29, 3419–3425. [Google Scholar] [CrossRef]
- Son, J.; Han, Y.H.; Lee, S.H. Prevalence and characteristics of patients with incidental cardiac uptake on bone scintigraphy. J. Cardiovasc. Imaging 2024, 32, 19. [Google Scholar] [CrossRef]
- Kim, H.M.; Sohn, D.W.; Paeng, J.C. Prevalence of Positive (99 m)Tc-DPD Scintigraphy as an Indicator of the Prevalence of Wild-type Transthyretin Amyloidosis in the Elderly. Int. Heart J. 2019, 60, 643–647. [Google Scholar] [CrossRef]
- Gillmore, J.D.; Maurer, M.S.; Falk, R.H.; Merlini, G.; Damy, T.; Dispenzieri, A.; Wechalekar, A.D.; Berk, J.L.; Quarta, C.C.; Grogan, M.; et al. Nonbiopsy Diagnosis of Cardiac Transthyretin Amyloidosis. Circulation 2016, 133, 2404–2412. [Google Scholar] [CrossRef]
- Tanaka, H.; Hosono, M.; Kanagaki, M.; Shimizu, M.; Matsubara, N.; Kawabata, K.; Miyamoto, T.; Itoi, K. A case of cardiac amyloidosis incidentally detected by bone scintigraphy. Asia Ocean. J. Nucl. Med. Biol. 2021, 9, 71–75. [Google Scholar] [CrossRef]
- Delaney, F.T.; Dempsey, P.; Welaratne, I.; Buckley, B.; O’Sullivan, D.; O’Connell, M. Incidental cardiac uptake in bone scintigraphy: Increased importance and association with cardiac amyloidosis. BJR Case Rep. 2021, 7, 20200161. [Google Scholar] [CrossRef] [PubMed]
- Chono, T.; Harada, K.; Asanuma, O. Incidental Cardiac Uptake in Bone Scintigraphy Establishing a Diagnosis of Transthyretin Amyloid Cardiomyopathy: A Case Report. Nihon Hoshasen Gijutsu Gakkai Zasshi 2023, 79, 1370–1374. [Google Scholar] [CrossRef]
- Lu, Y.; Groth, J.V.; Emmadi, R. Cardiac amyloidosis detected on tc-99m bone scan. Nucl. Med. Mol. Imaging 2015, 49, 78–80. [Google Scholar] [CrossRef]
- Fathala, A. Incidentally detected cardiac amyloidosis on (99m)Tc-MDP bone scintigraphy. Radiol. Case Rep. 2020, 15, 705–708. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Zhang, X.; Einstein, A.J.; Tang, G. Serial Tc-99m MDP scintigraphy demonstrating increasing cardiac uptake over time in a patient with light-chain cardiac amyloidosis. J. Nucl. Cardiol. 2022, 29, 2024–2028. [Google Scholar] [CrossRef]
- Ikebe, S.; Takashio, S.; Nishi, M.; Morioka, M.; Tsujita, K. Transthyretin Amyloid Cardiomyopathy Diagnosed on Incidental Myocardial Uptake During Bone Scintigraphy. Circ. J. 2020, 84, 679. [Google Scholar] [CrossRef]
- Banydeen, R.; Signate, A.; Tran, T.H.; Monfort, A.; Neviere, R.; Inamo, J. Cerebral Ischemic Events: An Overlooked Complication of Transthyretin Cardiac Amyloidosis in Afro-Caribbean Patients. Front. Neurol. 2022, 13, 878292. [Google Scholar] [CrossRef]
- Fukuzawa, S.; Okino, S.; Ishiwaki, H.; Iwata, Y.; Uchiyama, T.; Kuroiwa, N.; Oka, N.; Shibayama, N.; Inagaki, M. Positive Myocardial Uptake of Bone Scintigraphic Agents Associated with Cardiac Amyloidosis: Frequency of Positive Uptake Data Based on Daily Clinical Practice. Ann. Nucl. Cardiol. 2020, 6, 27–32. [Google Scholar] [CrossRef] [PubMed]
- al-Nahhas, A.M.; Jinnouchi, S.; Anagnostopoulos, C.; Hirsch, W.; Heary, T.; McCready, V.R. Clinical significance of technetium-99m methylene diphosphonate myocardial uptake: Association with carcinoma of the prostate. Eur. J. Nucl. Med. 1995, 22, 148–153. [Google Scholar] [CrossRef] [PubMed]
- Lee, V.W.; Caldarone, A.G.; Falk, R.H.; Rubinow, A.; Cohen, A.S. Amyloidosis of heart and liver: Comparison of Tc-99m pyrophosphate and Tc-99m methylene diphosphonate for detection. Radiology 1983, 148, 239–242. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.C.; Better, N. Tc-99m HMDP versus MDP discrepancy in cardiac amyloidosis. J. Nucl. Cardiol. 2021, 28, 2424. [Google Scholar] [CrossRef]
- Grigoratos, C.; Aimo, A.; Rapezzi, C.; Genovesi, D.; Barison, A.; Aquaro, G.D.; Vergaro, G.; Pucci, A.; Passino, C.; Marzullo, P.; et al. Diphosphonate single-photon emission computed tomography in cardiac transthyretin amyloidosis. Int. J. Cardiol. 2020, 307, 187–192. [Google Scholar] [CrossRef]
- Sperry, B.W.; Vranian, M.N.; Tower-Rader, A.; Hachamovitch, R.; Hanna, M.; Brunken, R.; Phelan, D.; Cerqueira, M.D.; Jaber, W.A. Regional Variation in Technetium Pyrophosphate Uptake in Transthyretin Cardiac Amyloidosis and Impact on Mortality. JACC Cardiovasc. Imaging 2018, 11, 234–242. [Google Scholar] [CrossRef]
- Uno, K.; Uchida, Y.; Sakata, S.; Minoshima, S.; Okada, J.; Yoshikawa, K.; Arimizu, N.; Karikomi, M.; Kitakata, Y.; Suzuki, Y. Visualization of the female breast in bone scintigraphy. Kaku Igaku 1992, 29, 1201–1206. [Google Scholar]
- Inanir, S.; Kiliç, A.K.; Ozdemir, E.; Zorlutuna, Y. Technetium-99m-methylene diphosphonate scintimammography in male breast cancer. J. Nucl. Med. 1998, 39, 28–29. [Google Scholar]
- Nurkalem, Z.; Sahin, S.; Uslu, N.; Emre, A.; Alper, A.T.; Gorgulu, S.; Yardi, F.; Eren, M. Predicting breast attenuation in patients undergoing myocardial perfusion scintigraphy: A digital x-ray study. J. Digit. Imaging 2008, 21, 446–451. [Google Scholar] [CrossRef]
- Holcman, K.; Chmura, Ł.; Rubiś, P.; Stepień, A.; Graczyk, K.; Kostkiewicz, M. Glu112Lys: A rare pathogenic transthyretin gene variant associated with negative scintigraphy findings. Pol. Arch. Intern. Med. 2025, 135. [Google Scholar] [CrossRef]
- Adams, C.; Banks, K.P. Bone Scan. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2024. Available online: https://www.ncbi.nlm.nih.gov/books/NBK531486/ (accessed on 15 May 2025).
- Holcman, K.; Rubiś, P.; Ćmiel, B.; Szot, W.; Stępień-Wroniecka, A.; Graczyk, K.; Mróz, K.; Dziewięcka, E.; Mateusz, W.; Szczepara, S.; et al. [(99m)Tc]Tc-DPD SPECT/CT evaluation of right and left ventricular involvement in cardiac transthyretin amyloidosis. Int. J. Cardiol. 2025, 431, 133227. [Google Scholar] [CrossRef] [PubMed]
- Wollenweber, T.; Rettl, R.; Kretschmer-Chott, E.; Rasul, S.; Kulterer, O.C.; Kluge, K.; Duca, F.; Bonderman, D.; Hacker, M.; Traub-Weidinger, T. Cardiac DPD-uptake time dependency in ATTR patients verified by quantitative SPECT/CT and semiquantitative planar parameters. J. Nucl. Cardiol. 2023, 30, 1363–1371. [Google Scholar] [CrossRef]
- Scully, P.R.; Morris, E.; Patel, K.P.; Treibel, T.A.; Burniston, M.; Klotz, E.; Newton, J.D.; Sabharwal, N.; Kelion, A.; Manisty, C.; et al. DPD Quantification in Cardiac Amyloidosis: A Novel Imaging Biomarker. JACC Cardiovasc. Imaging 2020, 13, 1353–1363. [Google Scholar] [CrossRef] [PubMed]
- Donnellan, E.; Wazni, O.M.; Hanna, M.; Elshazly, M.B.; Puri, R.; Saliba, W.; Kanj, M.; Vakamudi, S.; Patel, D.R.; Baranowski, B.; et al. Atrial Fibrillation in Transthyretin Cardiac Amyloidosis: Predictors, Prevalence, and Efficacy of Rhythm Control Strategies. JACC Clin. Electrophysiol. 2020, 6, 1118–1127. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.D.; Liu, Y.X.; Yan, X.W.; Fang, L.G.; Fang, Q.; Zhao, D.C.; Wang, Y.N. Cerebral embolism secondary to cardiac amyloidosis: A case report and literature review. Exp. Ther. Med. 2017, 14, 6077–6083. [Google Scholar] [CrossRef]
- Zubkov, A.Y.; Rabinstein, A.A.; Dispenzieri, A.; Wijdicks, E.F. Primary systemic amyloidosis with ischemic stroke as a presenting complication. Neurology 2007, 69, 1136–1141. [Google Scholar] [CrossRef]
- de Frutos, F.; Gonzalez-Lopez, E. New challenges in the management of patients with transthyretin cardiac amyloidosis: Beyond oral anticoagulation. Rev. Esp. Cardiol. (Engl. Ed.) 2023, 76, 494–496. [Google Scholar] [CrossRef]
- Elliott, P.; Drachman, B.M.; Gottlieb, S.S.; Hoffman, J.E.; Hummel, S.L.; Lenihan, D.J.; Ebede, B.; Gundapaneni, B.; Li, B.; Sultan, M.B.; et al. Long-Term Survival With Tafamidis in Patients With Transthyretin Amyloid Cardiomyopathy. Circ. Heart Fail. 2022, 15, e008193. [Google Scholar] [CrossRef]
- Gillmore, J.D.; Judge, D.P.; Cappelli, F.; Fontana, M.; Garcia-Pavia, P.; Gibbs, S.; Grogan, M.; Hanna, M.; Hoffman, J.; Masri, A.; et al. Efficacy and Safety of Acoramidis in Transthyretin Amyloid Cardiomyopathy. N. Engl. J. Med. 2024, 390, 132–142. [Google Scholar] [CrossRef]
- Garcia-Pavia, P.; Sultan, M.B.; Gundapaneni, B.; Sekijima, Y.; Perfetto, F.; Hanna, M.; Witteles, R. Tafamidis Efficacy Among Octogenarian Patients in the Phase 3 ATTR-ACT and Ongoing Long-Term Extension Study. JACC Heart Fail. 2024, 12, 150–160. [Google Scholar] [CrossRef]
- Penchala, S.C.; Connelly, S.; Wang, Y.; Park, M.S.; Zhao, L.; Baranczak, A.; Rappley, I.; Vogel, H.; Liedtke, M.; Witteles, R.M.; et al. AG10 inhibits amyloidogenesis and cellular toxicity of the familial amyloid cardiomyopathy-associated V122I transthyretin. Proc. Natl. Acad. Sci. USA 2013, 110, 9992–9997. [Google Scholar] [CrossRef]
- Judge, D.P.; Alexander, K.M.; Cappelli, F.; Fontana, M.; Garcia-Pavia, P.; Gibbs, S.D.J.; Grogan, M.; Hanna, M.; Masri, A.; Maurer, M.S.; et al. Efficacy of Acoramidis on All-Cause Mortality and Cardiovascular Hospitalization in Transthyretin Amyloid Cardiomyopathy. J. Am. Coll. Cardiol. 2025, 85, 1003–1014. [Google Scholar] [CrossRef] [PubMed]
- Fontana, M.; Berk, J.L.; Gillmore, J.D.; Witteles, R.M.; Grogan, M.; Drachman, B.; Damy, T.; Garcia-Pavia, P.; Taubel, J.; Solomon, S.D.; et al. Vutrisiran in Patients with Transthyretin Amyloidosis with Cardiomyopathy. N. Engl. J. Med. 2025, 392, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Aimo, A.; Castiglione, V.; Rapezzi, C.; Franzini, M.; Panichella, G.; Vergaro, G.; Gillmore, J.; Fontana, M.; Passino, C.; Emdin, M. RNA-targeting and gene editing therapies for transthyretin amyloidosis. Nat. Rev. Cardiol. 2022, 19, 655–667. [Google Scholar] [CrossRef] [PubMed]
- Gillmore, J.D.; Gane, E.; Taubel, J.; Kao, J.; Fontana, M.; Maitland, M.L.; Seitzer, J.; O’Connell, D.; Walsh, K.R.; Wood, K.; et al. CRISPR-Cas9 In Vivo Gene Editing for Transthyretin Amyloidosis. N. Engl. J. Med. 2021, 385, 493–502. [Google Scholar] [CrossRef]
- Fontana, M.; Solomon, S.D.; Kachadourian, J.; Walsh, L.; Rocha, R.; Lebwohl, D.; Smith, D.; Täubel, J.; Gane, E.J.; Pilebro, B.; et al. CRISPR-Cas9 Gene Editing with Nexiguran Ziclumeran for ATTR Cardiomyopathy. N. Engl. J. Med. 2024, 391, 2231–2241. [Google Scholar] [CrossRef]
- Kitai, T.; Kohsaka, S.; Kato, T.; Kato, E.; Sato, K.; Teramoto, K.; Yaku, H.; Akiyama, E.; Ando, M.; Izumi, C.; et al. JCS/JHFS 2025 Guideline on Diagnosis and Treatment of Heart Failure. Circ. J. 2025, 89, 1579–1581. [Google Scholar] [CrossRef]
- Izumiya, Y.; Kubo, T.; Endo, J.; Takashio, S.; Minamisawa, M.; Hamada, J.; Ishii, T.; Abe, H.; Konishi, H.; Tsujita, K. Transthyretin amyloid cardiomyopathy: Literature review and red-flag symptom clusters for each cardiology specialty. ESC Heart Fail. 2025, 12, 955–967. [Google Scholar] [CrossRef]
- Porcari, A.; Cappelli, F.; Nitsche, C.; Tomasoni, D.; Sinigiani, G.; Longhi, S.; Bordignon, L.; Masri, A.; Serenelli, M.; Urey, M.; et al. SGLT2 Inhibitor Therapy in Patients With Transthyretin Amyloid Cardiomyopathy. J. Am. Coll. Cardiol. 2024, 83, 2411–2422. [Google Scholar] [CrossRef]
- Byer, S.H.; Sivamurugan, A.; Grewal, U.S.; Fradley, M.G.; Dominic, P. Impact of Sodium-Glucose Cotransporter-2 Inhibitors on Cardiovascular Outcomes in Transthyretin Amyloid Cardiomyopathy. Am. J. Cardiol. 2025, 243, 15–18. [Google Scholar] [CrossRef]
- Kurita, T.; Nakamura, K.; Tabuchi, M.; Orita, M.; Ooshima, K.; Higashino, H. Effects of Gorei-san: A Traditional Japanese Kampo Medicine, on Aquaporin 1, 2, 3, 4 and V2R mRNA Expression in Rat Kidney and Forebrain. J. Med. Sci. 2011, 11, 30–38. [Google Scholar] [CrossRef]
- Isogai, T.; Morita, K.; Okada, A.; Michihata, N.; Matsui, H.; Miyawaki, A.; Jo, T.; Yasunaga, H. Association between complementary use of Goreisan (a Japanese herbal Kampo medicine) and heart failure readmission: A nationwide propensity score-matched study. J. Cardiol. 2025, 85, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Rozenbaum, M.H.; Tran, D.; Bhambri, R.; Nativi-Nicolau, J. Annual Cardiovascular-Related Hospitalization Days Avoided with Tafamidis in Patients with Transthyretin Amyloid Cardiomyopathy. Am. J. Cardiovasc. Drugs 2022, 22, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Ponti, L.; Hsu, K.; Damy, T.; Villacorta, E.; Verheyen, N.; Keohane, D.; Wang, R.; Ines, M.; Kumar, N.; Munteanu, C.; et al. Burden of untreated transthyretin amyloid cardiomyopathy on patients and their caregivers by disease severity: Results from a multicenter, non-interventional, real-world study. Front. Cardiovasc. Med. 2023, 10, 1238843. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Matsuki, N.; Awaya, T.; Endo, J.; Kunimasa, T.; Gomi, T.; Okamoto, Y.; Hara, H. Incidental Cardiac Uptake on 99mTc-HMDP Bone Scintigraphy in Oncology Patients: Two Cases of Transthyretin Amyloid Cardiomyopathy with Literature Review. Diseases 2026, 14, 23. https://doi.org/10.3390/diseases14010023
Matsuki N, Awaya T, Endo J, Kunimasa T, Gomi T, Okamoto Y, Hara H. Incidental Cardiac Uptake on 99mTc-HMDP Bone Scintigraphy in Oncology Patients: Two Cases of Transthyretin Amyloid Cardiomyopathy with Literature Review. Diseases. 2026; 14(1):23. https://doi.org/10.3390/diseases14010023
Chicago/Turabian StyleMatsuki, Naoya, Toru Awaya, Jin Endo, Taeko Kunimasa, Tatsuya Gomi, Yasushi Okamoto, and Hidehiko Hara. 2026. "Incidental Cardiac Uptake on 99mTc-HMDP Bone Scintigraphy in Oncology Patients: Two Cases of Transthyretin Amyloid Cardiomyopathy with Literature Review" Diseases 14, no. 1: 23. https://doi.org/10.3390/diseases14010023
APA StyleMatsuki, N., Awaya, T., Endo, J., Kunimasa, T., Gomi, T., Okamoto, Y., & Hara, H. (2026). Incidental Cardiac Uptake on 99mTc-HMDP Bone Scintigraphy in Oncology Patients: Two Cases of Transthyretin Amyloid Cardiomyopathy with Literature Review. Diseases, 14(1), 23. https://doi.org/10.3390/diseases14010023






