Vitamin D Deficiency as a Risk Factor of Preeclampsia during Pregnancy
Abstract
1. Introduction
1.1. Preeclampsia
1.2. Pathophysiology of Preeclampsia
1.3. Diagnosis of Preeclampsia
1.4. Vitamin D Sources
1.5. Biological Role and Forms
1.6. The Role of Vitamin D in Bone Health
1.7. Pregnancy and Breastfeeding
1.8. Vitamin D, Immune System, and Other Effects
1.9. Vitamin D and Diabetes Mellitus
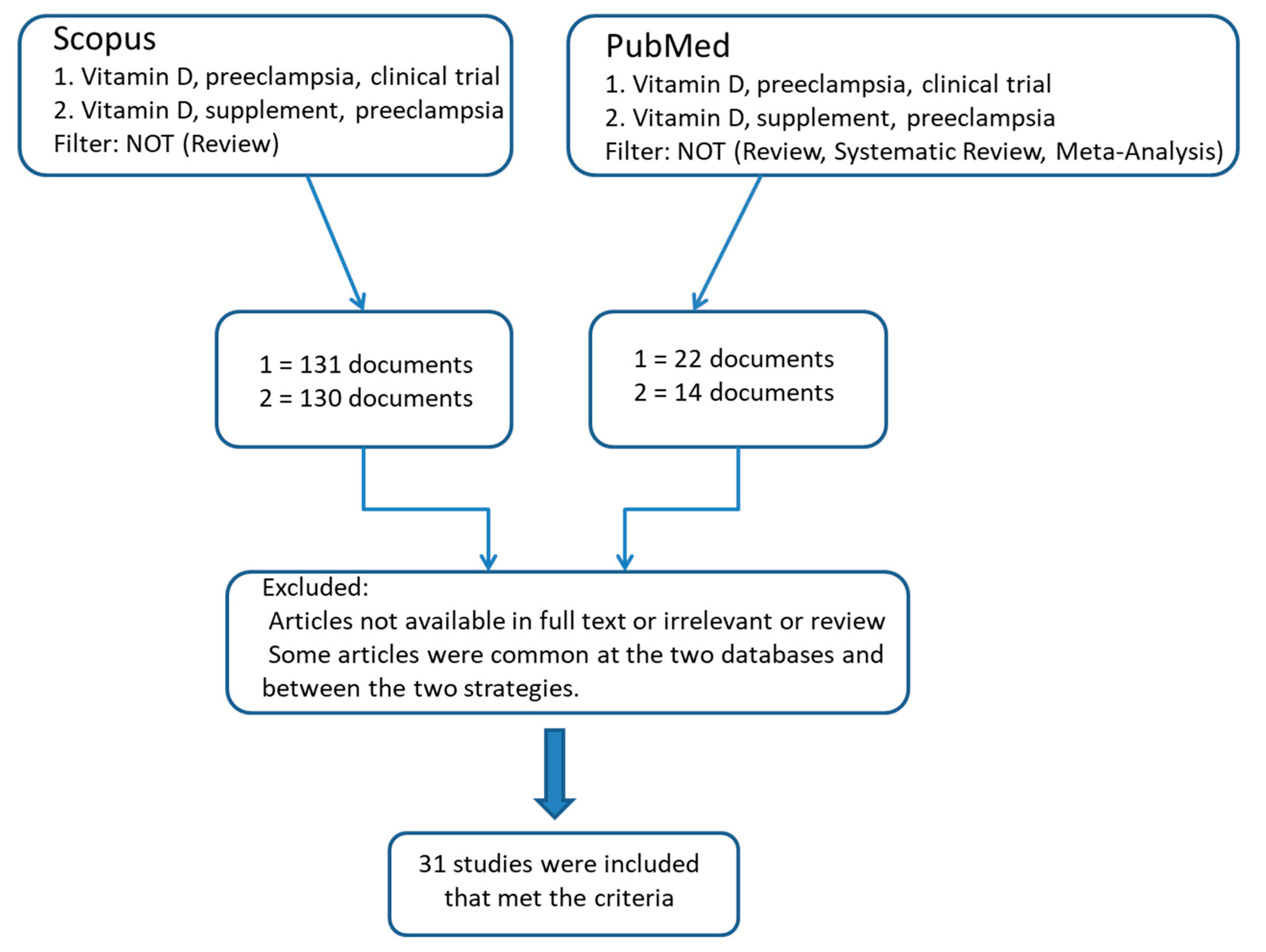
2. Materials and Methods
3. Results
3.1. Vitamin D Supplementation
3.2. Observational Studies
3.3. Effect of a Low-Glycemic-Index Diet on Vitamin D
| Authors | Country | Study Design | Maternal Age | N Total | Duration of Investigation | Eligibility Criteria or Exclusion Criteria | Methodology of the Study | N/Group | Mean Age/Group | Preeclampsia | Statistically Significant Correlation between Preeclampsia and Vitamin D? (Yes/No) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Anderson, 2015 [116] | USA, Dakota | Case–control study | 24–26 | 48 | n/a | Eligibility: Nulliparous women, >18 years (<14 w of pregnancy) | Dietary intake- supplementation of vitamin D in the previous 3 months. Normotensive and PE groups determined by blood pressure and proteinuria. RIA to adjust the maternal [25(OH)D] serum. | Normotensive: n = 37 Gestational hypertension: n = 11 | Normotensive: 24.2 ± 0.62 Gestational hypertension: 25.3± 0.72 | There is reference to SBP/DBP and hypertension during pregnancy | Yes |
| Bärebring, 2016 [60] | Sweden | Cohort study | 31.3 (mean) | 2000 | Fall 2013 (2 September–8 November) Spring 2014 (24 February–13 June) | Exclusion: pregnancy exceeding 16 w gestation, miscarried before 20 w, lost to follow-up | Blood samples <16 w (8–12) and after 31 w (32–35), questionnaire for lifestyle. After delivery: BP, proteinuria, preexisting medical conditions, weight, height, etc. Available samples T1, T3 (N = 1827) | n/a | n/a | 4% | No |
| Benachi, 2020 [45] | Belgium and France | Nested case–control study/prospective observational cohort | n/a | 402 | April 2012–February 2015 | Patients were excluded from selection of controls if we could not be sure whether preeclampsia occurred or not (patients not followed up to delivery or no data on blood pressure or proteinuria), if pregnancy was interrupted (abortion, intrauterine fetal death) or if there were no data on delivery. The sample of eligible controls was obtained from controls without preterm delivery (37 WA), whose newborn was alive in the delivery room and presented no intrauterine growth restriction <5th percentile) at birth, with vitamin D measurement available in both the first and third trimesters and with no missing data on any matching factors. | Vitamin D insufficiency in the first trimester and preeclampsia later in pregnancy. A bolus vitamin D dose (100,000 IU of cholecalciferol) was prescribed to the patients at the seventh month of pregnancy according to current French recommendations. The main outcome measure was serum 25(OH)D status in the first trimester. | Cases n = 93 controls n = 319 | cases 32.2 ± 5.9, controls 31.7 ± 5.0 | No percentage per group | No |
| Robinson, 2010 [14] | USA | Case–control study | 26 (24 + 28) (mean) | 150 | n/a | Eligibility criteria for EOSPE: American College of Obstetrics and Gynecology and diagnosis prior to 34 w Exclusion for EOSPE: chronic hypertension, pregestational diabetes, renal disease, lupus, multiple gestation Exclusion criteria for controls: EOSPE criteria | Demographic (BMI, gestational age, maternal, SBP, DBP, urine protein) and outcome data of plasma 25(OH)D analysis | Cases: n = 50 Controls: n = 100 | Cases: 24 (21–30) Controls: 28 (23–32) | Cases: 42% Controls: 10% | Yes |
| Tamblyn, 2017 [43] | United Kingdom | Cross sectional analysis | n/a | 88 | n/a | n/a | Placental biopsies, analysis of DBP, albumin and free vitamin D metabolites, blood samples, demographic characteristics | First trimester—healthy: n = 25 third trimester—healthy: n = 21 PET: n = 22 Non pregnant: n = 20 | No significant differences between ages | n/a | Yes |
| Pashapour, 2019 [117] | Iran, West Azerbaijan | Case–control study | 24–35 | 160 | January–May 2016 | Inclusion criteria: singleton pregnancy, no medical disorders (diabetes, kidney, hypertension), no history of vitamin intake during pregnancy, no smoking, no BMI > 30 kg/m2 | Measurement od 25(OH)D in pregnant women with and without preeclampsia | Preeclampsia group: n = 80 Healthy group: n = 80 | Preeclampsia group: 28.83 ± 6.7 Healthy group: 24.13 ± 8.4 | Preeclamptic women tend to be older, >BMI, 30–35 years old more prone to get PE. Lower vitamin D increased PE | Yes |
| Forde, 2021 [118] | Dublin | Observational study | 32.1 (mean) | 415 | 2007–2011 (5-year follow up) | Exclusion criteria: history of gestational diabetes, concurrent medication, age < 18, multiple pregnancy | Follow up women in low-glycemic-index diet, maternal concentration of vitamin D and mother’s age | The is no separation into subgroups | 32.1 | No correlation between vitamin D consumption and blood pressure during first–third trimester and 5 y follow up | No |
| Huang, 2022 [18] | China | Matched control study | n/a | 1180 | March 2016–June 2019 | Cases inclusion criteria: diagnosed with preeclampsia according to the guidelines for hypertensive disorders in pregnancy Controls criteria: no preeclampsia | Serum concentrations of 25(OH)D2 and 25(OH)D3 | Cases: n = 532 Controls: n = 648 | Cases: 30.88 (mean) Controls: 31.03 (mean) | Dietary intake was negatively associated with preeclampsia risk. Higher vitamin d intake or serum concentration are associated with lower risk of preeclampsia | Yes |
| Boyle, 2016 [72] | Australia | Cohort study | 30.3 (mean) | 1710 | 2005–2008 | n/a | Non-fasting serum samples were collected at 15 weeks of gestation. Women were screened for GDM between 24 and 28 weeks of gestation with a non-fasting 50 g polycose challenge in community laboratories, according to the Auckland District Healt Board Guidelines | Cases: n = 93, controls n = 319 | cases 32.2 ± 5.9, controls 31.7 ± 5.0 | Vitamin D did not predict preeclampsia | No |
| Raia-Barjat, 2021 [30] | France, Nimes | Ad hoc study of a previous cohort | 32 ± 5 (mean) | 200 | June 2008–October 2010 | Inclusion criteria: High risk for preeclampsia that included diabetes, chronic hypertension, systemic lupus, antiphospholipid syndrome, history of cardiovascular disease, etc. Exclusion criteria: twin pregnancy, history of fetal death, chromosomal IUGR, venous thromboembolism | Monitoring blood samples for analysis of serum 25(OH)D at 20, 24, 28, 32, 36 w | PMC: n = 43 No PMC: n = 139 | PMC: 32 ± 5.3 No PMC: 32.1 ± 5.1 | 43% developed PMC. Vitamin D levels at 32 w were lower between patients with PMC. Strong relationship between vitamin D levels and the risk of PMC at 32 w | Yes |
| Yuan, 2021 [55] | China | Nested case–control study | 29–34 | 610 | March 2012–February 2015 | Exclusion criteria: loss of basic information, medical abortion, multiple pregnancy, and the use of assisted reproductive technology or 25(OH)D concentrations beyond the assay detection limit | Serum 25(OH)D was at first examination in second or third trimester. Diagnosis of preeclampsia during other examination | Cases: n = 122 Controls: n = 488 | Cases: 30.6 ± 3.7 Controls: 29.6 ± 2.8 | Women with preeclampsia were older, >BMI. Maternal vitamin D levels were lower in women with preeclampsia | Yes |
| Serrano, 2013 [32] | Colombia | Case–control study | <25 years except HELLP syndrome | 2028 | December 2000–February 2012 | Inclusion criteria for cases: BP ≥ 140/90 mmHg, 24 h proteinuria ≥ 300 mg or ≥2+ in urine after 20th week Exclusion criteria: history of hypertension, diabetes mellitus, renal or autoimmune diseases | Sociodemographic data for all participants, samples of maternal 25(OH)D and quantification | Cases: n = 1013 Controls: n = 1015 | Cases: 19.1 Controls: 18.7 | 52% of women with preeclampsia were vitamin D deficient. | Yes |
| Bodnar, 2007 [73] | Pennsylvania | Prospective cohort study | 14–44 | 274 | 1997–2001 | Inclusion criteria: <16 w gestation, singleton pregnancy, 14–44 y.o., planning deliver at Magee- Women’s Hospital | Quantitation of serum 25(OH)D, examination of predisposing factors for preeclampsia | Cases: n = 55 Controls: n = 219 | Cases: <20 y 20, 20–29 y 52.7, >30 y 27.3 Controls: <20 y 37.4, 20–29 y 47.5, >30 y 15.1 | Serum [25(OH)D] were lower 15% in cases | Yes |
| Haile, 2021 [31] | Ethiopia | Unmatched case–control | <20, 20-34, ≥35 | 344 | n/a | n/a | Blood samples from both cases and controls | Cases: n = 86 Controls: n = 258 | Cases: <20 y 9, 20–34 y 64, ≥35 y 13 Controls: <20 y 26, 20–34 y 200, ≥35 y 32 | n/a | Yes |
| Chia, 2018 [121] | Singapore | GUSTO study | 18–50 | 1051 | June 2009–September 2010 | Exclusion criteria: Type 1 diabetes mellitus, chemotherapy, psychotropic drugs, parents or spouses had different ethnicities | Dietary intakes and analyze diet quality at 26–28 w of pregnancy. Infant outcomes | No separation into groups | n/a | Maternal diet quality does not have any relationship with SGA or prematurity | No |
| Bodnar, 2014 [119] | USA (12 States) | Case–cohort study | n/a | 3703 | 1959–1966 | Exclusion criteria: preexisting conditions, entry to care at ≥26 w | In both cases and controls, measurement of [25(OH)D] at ≤26 w | Subcohort: n = 3068 Cases: n = 717 | Subcohort: <20 23%, 20–29 61%, ≥30 16% Cases: <20 37%, 20–29 43%, ≥30 20% | Mothers who developed preeclampsia tended to be black, nulliparous, <20/ ≥ 30 years old, smokers, less educated, overweight, etc. There is no significant association between maternal [25(OH)D] and preeclampsia. Although it may be a risk factor | Yes |
| Thorsen, 2021 [120] | Australia | Cohort study | n/a | 1074 pregnant women- and 1074 infants | 2010–2013 | Exclusion criteria: birth <32 w, serious disease, genetic abnormality | Vitamin D metabolite data at 28–32 w and birth, and Treg proportions in CB | No separation into groups | n/a | Higher maternal 25(OH)D3 levels were associated with increased free Treg, and aTreg. A positive correlation was found between CB 25(OH)D3 levels and CB aTreg. 25(OH)D3 may play a role in neonatal immunity | Yes |
| Burris, 2014 [122] | USA | Prospective prenatal cohort study | 32.1 (mean) | 1591 | n/a | n/a | Among women enrolled in the Project Viva prenatal cohort in Massachusetts, we examined associations of 25(OH)D levels obtained at 16.4–36.9 weeks of gestation (mean 27.9 weeks) with hypertensive disorders of pregnancy, including preeclampsia (56/1591, 3.5%) and gestational hypertension (109/1591, 6.9%) | category per plasma [25(OH)D] (nmol/L): <25 n = 81, 25-<50 n = 472, 50-<75 n = 743, ≥75 n = 295 | 3.8% | No, it was not detected an association at mean 27.9 w gestation and preeclampsia | No |
| Kiely, 2016 [123] | Ireland | Cohort study | n/a | 1768 | March 2008–January 2011 | n./a | Non-fasting serum samples were collected at 15 weeks of gestation. Women were screened for GDM between 24 and 28 weeks of gestation with a non-fasting 50 g polycose challenge in community laboratories, according to the Auckland District Health Board Guidelines. | cases n = 93, controls n = 319 | 3.8% | Yes, it was protective at levels >75 nmol/L | Yes |
3.4. Vitamin D Levels and Preeclampsia Incidence in Case of Comorbidities
3.5. Studies Showing No Association between Vitamin D Levels and Preeclampsia
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mccollum, E.V.; Pitz, W.; Simmonds, N.; Becker, J.E.; Shipley, P.G.; Bunting, R.W. The effect of additions of fluorine to the diet of the rat on the quality of the teeth. 1925. Studies on experimental rickets. XXI. An experimental demonstration of the existence of a vitamin which promotes calcium deposition. 1922. The effect of additions of fluorine to the diet of the rat on the quality of the teeth. 1925. J. Biol. Chem. 2002, 277, E8. [Google Scholar]
- Mozolowski, W. Jedrzej Sniadecki (1768–1838) on the cure of rickets. Nature 1939, 143, 121e4. [Google Scholar]
- Finberg, L. Vitamin D Deficiency and Rickets. Lond. J. Pediatr. Endocrinol. Metab. 2006, 19, 203. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.W.; Lee, H.C. Vitamin D and health—The missing vitamin in humans. In Pediatrics and Neonatology; Elsevier Pte Ltd.: Singapore, 2019; Volume 60, pp. 237–244. [Google Scholar] [CrossRef]
- O’riordan, J.L.H.; Bijvoet, O.L.M. Rickets before the discovery of vitamin D. BoneKEy Rep. 2014, 3, 478. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Glisson, F.; Armin, P.; Bate, G.; Regemorter, A. A Treatise of the Rickets: Being a Disease Common to Children; John Streater: London, UK, 1951. [Google Scholar]
- Holick, M.F. McCollum Award Lecture, 1994: Vitamin D—New horizons for the 21st century. Am. J. Clin. Nutr. 1994, 60, 619–630. [Google Scholar] [CrossRef] [PubMed]
- Moore, S. Dissertatio Medica Inauguralis de Rachitide; Balfour et Smellie: Edinburgh, UK, 1728; pp. 1–35. [Google Scholar]
- Trousseau, A. Lectures on Clinical Medicine; Lecture 86; Rickets 90–94; Translated by Cormack K.B.; The New Sydenham Society: Sydenham, UK, 1868; Volume 5. [Google Scholar]
- Hess, A.F.; Unger, L.J. Prophylactic therapy for rickets in a Negro community. J. Am. Med. Assoc. 1917, 69, 1583–1586. [Google Scholar] [CrossRef]
- Mellanby, E. Experimental Rickets; H.M. Stationery Office: London, UK, 1921; Available online: https://books.google.gr/books?id=yLMfAAAAIAAJ (accessed on 3 March 2018).
- Kulda, V. Metabolizmus vitaminu D [Vitamin D metabolism]. Vnitr. Lek. 2012, 58, 400–404. [Google Scholar] [PubMed]
- Hollis, B.W. Circulating 25-hydroxyvitamin D levels indicative of vitamin D sufficiency: Implications for establishing a new effective dietary intake recommendation for vitamin D. J. Nutr. 2005, 135, 317–322. [Google Scholar] [CrossRef] [PubMed]
- Robinson, C.J.; Alanis, M.C.; Wagner, C.L.; Hollis, B.W.; Johnson, D.D. Plasma 25-hydroxyvitamin D levels in early-onset severe preeclampsia. Am. J. Obstet. Gynecol. 2010, 203, 366.e1–366.e6. [Google Scholar] [CrossRef]
- De-Regil, L.M.; Palacios, C.; Lombardo, L.K.; Peña-Rosas, J.P. Vitamin D supplementation for women during pregnancy. In Cochrane Database of Systematic Reviews; John Wiley and Sons Ltd.: Hoboken, NJ, USA, 2016; Volume 2016. [Google Scholar] [CrossRef]
- World Health Organization. Vitamin and Mineral Requirements in Human Nutrition, 2nd ed.; World Health Organization: Geneva, Switzerland, 2004; p. 341. [Google Scholar]
- DeLuca, H.F. History of the discovery of vitamin D and its active metabolites. BoneKEy Rep. 2014, 3, 479. [Google Scholar] [CrossRef]
- Huang, X.M.; Liu, Y.H.; Zhang, H.; Cao, Y.; Dou, W.F.; Duan, D.D.; Chen, H.N.; Bo, Y.C.; Amoah, A.N.; Fu, W.J.; et al. Dietary and serum vitamin D and preeclampsia risk in Chinese pregnant women: A matched case-control study. Br. J. Nutr. 2022, 128, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Rosen, C.J. Clinical practice Vitamin D Insufficiency. N. Engl. J. Med. 2011, 364, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Mithal, A.; Bansal, B.; Kyer, C.S.; Ebeling, P. The Asia-Pacific Regional Audit-Epidemiology, costs, and burden of osteoporosis in India 2013: A report of International Osteoporosis Foundation. Indian J. Endocrinol. Metab. 2014, 18, 449–454. [Google Scholar] [CrossRef]
- Accortt, E.E.; Mirocha, J.; Schetter, C.D.; Hobel, C.J. Adverse Perinatal Outcomes and Postpartum Multi-Systemic Dysregulation: Adding Vitamin D Deficiency to the Allostatic Load Index. Matern. Child Health J. 2017, 21, 398–406. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.Q.; Qi, H.P.; Luo, Z.C.; Fraser, W.D. Maternal vitamin D status and adverse pregnancy outcomes: A systematic review and meta-analysis. J. Matern.-Fetal Neonatal Med. 2013, 26, 889–899. [Google Scholar] [CrossRef] [PubMed]
- Redman, C.W.G.; Jacobson, S.-L.; Russell, R. Hypertension in pregnancy. In De Swiet’s Medical Disorders in Obstetric Practice, 5th ed.; Powrie, R., Greene, M., Camann, W., Eds.; Blackwell Publishing: Oxford, UK, 2010; pp. 153–181. [Google Scholar]
- Brown, M.A.; Magee, L.A.; Kenny, L.C.; Karumanchi, S.A.; McCarthy, F.P.; Saito, S.; Hall, D.R.; Warren, C.E.; Adoyi, G.; Ishaku, S. Hypertensive disorders of pregnancy: ISSHP classification, diagnosis, and management recommendations for international practice. Hypertension 2018, 72, 24–43. [Google Scholar] [CrossRef] [PubMed]
- Hypertension in Pregnancy. Report of the American College of Obstetricians and Gynecologists’ Task Force on Hypertension in Pregnancy. Obstet. Gynecol. 2013, 122, 1122–1131. [Google Scholar] [CrossRef] [PubMed]
- Tranquilli, A.L.; Brown, M.A.; Zeeman, G.G.; Dekker, G.; Sibai, B.M. The definition of severe and early-onset preeclampsia. Statements from the International Society for the Study of Hypertension in Pregnancy (ISSHP). Pregnancy Hypertens. 2013, 3, 44–47. [Google Scholar] [CrossRef]
- Nirupama, R.; Divyashree, S.; Janhavi, P.; Muthukumar, S.; Ravindra, P. Preeclampsia: Pathophysiology and management. J. Gynecol. Obstet. Hum. Reprod. 2021, 50, 101975. [Google Scholar] [CrossRef]
- Wójtowicz, A.; Zembala-Szczerba, M.; Babczyk, D.; Kołodziejczyk-Pietruszka, M.; Lewaczyńska, O.; Huras, H. Early-and Late-Onset Preeclampsia: A Comprehensive Cohort Study of Laboratory and Clinical Findings according to the New ISHHP Criteria. Int. J. Hypertens. 2019, 2019, 4108271. [Google Scholar] [CrossRef]
- Wadhwani, P.; Saha, P.K.; Kalra, J.K.; Gainder, S.; Sundaram, V. A study to compare maternal and perinatal outcome in early vs. late onset preeclampsia. Obstet. Gynecol. Sci. 2020, 63, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Raia-Barjat, T.; Sarkis, C.; Rancon, F.; Thibaudin, L.; Gris, J.-C.; Alfaidy, N.; Chauleur, C. Vitamin D deficiency during late pregnancy mediates placenta-associated complications. Sci. Rep. 2021, 11, 20708. [Google Scholar] [CrossRef]
- Haile, T.G.; Assefa, N.; Alemayehu, T.; Mariye, T.; Geberemeskel, G.G.; Bahrey, D.; Mebrahtom, G.; Demisse, B.; Gebrekidan, H.; Getachew, T. Determinants of Preeclampsia among Women Attending Delivery Services in Public Hospitals of Central Tigray, Northern Ethiopia: A Case-Control Study. J. Pregnancy 2021, 19, 4654828. [Google Scholar] [CrossRef]
- Serrano, N.C.; Guío, E.; Quintero-Lesmes, D.C.; Becerra-Bayona, S.; Luna-Gonzalez, M.L.; Herrera, V.M.; Prada, C.E. Vitamin D deficiency and pre-eclampsia in Colombia: PREVitD study. Pregnancy Hypertens. 2018, 14, 240–244. [Google Scholar] [CrossRef] [PubMed]
- Jeyabalan, A. Epidemiology of preeclampsia: Impact of obesity. Nutr. Rev. 2013, 71 (Suppl. S1), S18–S25. [Google Scholar] [CrossRef] [PubMed]
- Behjat Sasan, S.; Zandvakili, F.; Soufizadeh, N.; Baybordi, E. The Effects of Vitamin D Supplement on Prevention of Recurrence of Preeclampsia in Pregnant Women with a History of Preeclampsia. Obstet. Gynecol. Int. 2017, 2017, 8249264. [Google Scholar] [CrossRef]
- Brantsaeter, A.L.; Myhre, R.; Haugen, M.; Myking, S.; Sengpiel, V.; Magnus, P.; Jacobsson, B.; Meltzer, H.M. Intake of probiotic food and risk of preeclampsia in primiparous women. Am. J. Epidemiol. 2011, 174, 807–815. [Google Scholar] [CrossRef]
- World Health Organization International Collaborative Study of Hypertensive Disorders of Pregnancy. Geographic variation in the incidence of hypertension. 1n pregnancy. Am. J. Obstet. Gynecol. 1986, 158, 80–83. [Google Scholar] [CrossRef]
- Sibai, B.; Dekker, G.; Kupferminc, M. Pre-eclampsia. Lancet 2005, 365, 785–799. [Google Scholar] [CrossRef]
- Duley, L. The Global Impact of Pre-eclampsia and Eclampsia. Semin. Perinatol. 2009, 33, 130–137. [Google Scholar] [CrossRef]
- Wallis, A.B.; Saftlas, A.F.; Hsia, J.; Atrash, H.K. Secular trends in the rates of preeclampsia, eclampsia, and gestational hypertension, United States, 1987–2004. Am. J. Hypertens. 2008, 21, 521–526. [Google Scholar] [CrossRef] [PubMed]
- Sibai, B.M. Diagnosis and management of gestational hypertension and preeclampsia. Obstet. Gynecol. 2003, 102, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Karpova, N.; Dmitrenko, O.; Arshinova, E.; Nurbekov, M. Review: Influence of 25(OH)D Blood Concentration and Supplementation during Pregnancy on Preeclampsia Development and Neonatal Outcomes. Int. J. Mol. Sci. 2022, 23, 12935. [Google Scholar] [CrossRef] [PubMed]
- Ayoubi. Pre-eclampsia: Pathophysiology, diagnosis, and management. Vasc. Health Risk Manag. 2011, 7, 467–474. [Google Scholar] [CrossRef]
- Tamblyn, J.; Susarla, R.; Jenkinson, C.; Jeffery, L.; Ohizua, O.; Chun, R.; Chan, S.; Kilby, M.D.; Hewison, M. Dysregulation of maternal and placental vitamin D metabolism in preeclampsia. Placenta 2017, 50, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Fisher, S.J. Why is placentation abnormal in preeclampsia? Am. J. Obstet. Gynecol. 2015, 213, S115–S122. [Google Scholar] [CrossRef]
- Benachi, A.; Baptiste, A.; Taieb, J.; Tsatsaris, V.; Guibourdenche, J.; Senat, M.V.; Haidar, H.; Jani, J.; Guizani, M.; Jouannic, J.M.; et al. Relationship between vitamin D status in pregnancy and the risk for preeclampsia: A nested case-control study. Clin. Nutr. 2020, 39, 440–446. [Google Scholar] [CrossRef] [PubMed]
- Brosens, I.; Renaer, M. On the pathogenesis of placental infarcts in pre-eclampsia. J. Obstet. Gynaecol. Br. Commonw. 1972, 79, 794–799. [Google Scholar] [CrossRef]
- Roccella, E.J. Report of the National High Blood Pressure Education Program Working Group on High Blood Pressure in Pregnancy. Am. J. Obstet. Gynecol. 2000, 183, S1–S22. [Google Scholar] [CrossRef]
- Kanasaki, K.; Palmsten, K.; Sugimoto, H.; Ahmad, S.; Hamano, Y.; Xie, L.; Parry, S.; Augustin, H.G.; Gattone, V.H.; Folkman, J.; et al. Deficiency in catechol-O-methyltransferase and 2-methoxyoestradiol is associated with pre-eclampsia. Nature 2008, 453, 1117–1121. [Google Scholar] [CrossRef]
- Rana, S.; Lemoine, E.; Granger, J.P.; Karumanchi, S.A. Preeclampsia: Pathophysiology, Challenges, and Perspectives. Circ. Res. 2019, 124, 1094–1112. [Google Scholar] [CrossRef]
- Jauniaux, E.; Watson, A.L.; Hempstock, J.; Bao, Y.-P.; Skepper, J.N.; Burton, G.J. Onset of Maternal Arterial Blood Flow and Placental Oxidative Stress A Possible Factor in Human Early Pregnancy Failure. Am. J. Pathol. 2000, 157, 2111–2122. [Google Scholar] [CrossRef] [PubMed]
- Karrar, S.A.; Hong, P.L. Preeclampsia. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Tranquilli, A.L.; Dekker, G.; Magee, L.; Roberts, J.; Sibai, B.M.; Steyn, W.; Zeeman, G.G.; Brown, M.A. The classification, diagnosis and management of the hypertensive disorders of pregnancy: A revised statement from the ISSHP. Pregnancy Hypertens. 2014, 4, 97–104. [Google Scholar] [CrossRef]
- Roberts, J.M.; Pearson, G.; Cutler, J.; Lindheimer, M. Summary of the NHLBI Working Group on research on hypertension during pregnancy. Hypertension 2003, 41, 437–445. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.W. Reproductive Tract Abnormalities. In Williams Obstetrics; McGraw Hill/Medical: New York, NY, USA, 2014; Volume 40. [Google Scholar]
- Yuan, Y.; Tai, W.; Xu, P.; Fu, Z.; Wang, X.; Long, W.; Guo, X.; Ji, C.; Zhang, L.; Zhang, Y.; et al. Association of maternal serum 25-hydroxyvitamin D concentrations with risk of preeclampsia: A nested case-control study and meta-analysis. J. Matern. -Fetal Neonatal Med. 2021, 34, 1576–1585. [Google Scholar] [CrossRef]
- Antonucci, R.; Locci, C.; Clemente, M.G.; Chicconi, E.; Antonucci, L. Vitamin D deficiency in childhood: Old lessons and current challenge. J. Pediatr. Endocrinol. Metab. 2018, 31, 247–260. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Cohen, W.R.; Silva, P.; Epstein, F.H. Rapid Publications Elevated 1,25-Dihydroxyvitamin D Plasma Levels in Normal Human Pregnancy and Lactation. J. Clin. Investig. 1979, 63, 342–344. [Google Scholar] [CrossRef] [PubMed]
- Misra, M.; Pacaud, D.; Petryk, A.; Collett-Solberg, P.F.; Kappy, M. Vitamin D deficiency in children and its management: Review of current knowledge and recommendations. Pediatrics 2008, 122, 398–417. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.S.; Hewison, M. Update in vitamin D. J. Clin. Endocrinol. Metab. 2010, 95, 471–478. [Google Scholar] [CrossRef]
- Bärebring, L.; Bullarbo, M.; Glantz, A.; Leu Agelii, M.; Jagner, Å.; Ellis, J.; Hulthén, L.; Schoenmakers, I.; Augustin, H. Preeclampsia and Blood Pressure Trajectory during Pregnancy in Relation to Vitamin D Status. PLoS ONE 2016, 11, e0152198. [Google Scholar] [CrossRef]
- Kelly, C.B.; Wagner, C.L.; Shary, J.R.; Leyva, M.J.; Yu, J.Y.; Jenkins, A.J.; Nankervis, A.J.; Hanssen, K.F.; Garg, S.K.; Scardo, J.A.; et al. Vitamin D metabolites and binding protein predict preeclampsia in women with type 1 diabetes. Nutrients 2020, 12, 2048. [Google Scholar] [CrossRef] [PubMed]
- Yao, P.; Bennett, D.; Mafham, M.; Lin, X.; Chen, Z.; Armitage, J.; Clarke, R. Vitamin D and Calcium for the Prevention of Fracture: A Systematic Review and Meta-analysis. JAMA Netw. Open 2019, 2, e1917789. [Google Scholar] [CrossRef] [PubMed]
- Bachrach, L.K. Acquisition of optimal bone mass in childhood and adolescence. Trends Endocrinol. Metab. 2001, 12, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Aydın, C.G.; Dinçel, Y.M.; Arıkan, Y.; Taş, S.K.; Deniz, S. The effects of indoor and outdoor sports participation and seasonal changes on vitamin D levels in athletes. SAGE Open Med. 2019, 7, 2050312119837480. [Google Scholar] [CrossRef] [PubMed]
- Kanis, J.A.; Odén, A.; McCloskey, E.V.; Johansson, H.; Wahl, D.A.; Cooper, C. A systematic review of hip fracture incidence and probability of fracture worldwide. Osteoporos. Int. 2012, 23, 2239–2256. [Google Scholar] [CrossRef] [PubMed]
- Voulgaridou, G.; Papadopoulou, S.K.; Detopoulou, P.; Tsoumana, D.; Giaginis, C.; Kondyli, F.S.; Lymperaki, E.; Pritsa, A. Vitamin D and Calcium in Osteoporosis, and the Role of Bone Turnover Markers: A Narrative Review of Recent Data from RCTs. Diseases 2023, 11, 29. [Google Scholar] [CrossRef]
- Lane, N.E. Epidemiology, etiology, and diagnosis of osteoporosis. Am. J. Obstet. Gynecol. 2006, 194, S3–S11. [Google Scholar] [CrossRef] [PubMed]
- Fischer, V.; Haffner-Luntzer, M.; Amling, M.; Ignatius, A. Calcium and vitamin D in bone fracture healing and post-traumatic bone turnover. Eur. Cells Mater. 2018, 35, 365–385. [Google Scholar] [CrossRef]
- Liu, C.; Kuang, X.; Li, K.; Guo, X.; Deng, Q.; Li, D. Effects of combined calcium and vitamin D supplementation on osteoporosis in postmenopausal women: A systematic review and meta-analysis of randomized controlled trials. Food Funct. 2020, 11, 10817–10827. [Google Scholar] [CrossRef]
- Steer, P.J.; Little, M.P.; Kold-Jensen, T.; Chapple, J.; Elliott, P. Maternal blood pressure in pregnancy, birth weight, and perinatal mortality in first births: Prospective study. Br. Med. J. 2004, 329, 1312–1314. [Google Scholar] [CrossRef]
- Khan, K.S.; Wojdyla, D.; Say, L.; Metin Gülmezoglu, A.; Van Look, P.F.A. Articles WHO analysis of causes of maternal death: A systematic review. Lancet 2006, 367, 1066–1074. [Google Scholar] [CrossRef] [PubMed]
- Boyle, V.T.; Thorstensen, E.B.; Mourath, D.; Jones, M.B.; McCowan, L.M.E.; Kenny, L.C.; Baker, P.N. The relationship between 25-hydroxyvitamin D concentration in early pregnancy and pregnancy outcomes in a large, prospective cohort. Br. J. Nutr. 2016, 116, 1409–1415. [Google Scholar] [CrossRef] [PubMed]
- Bodnar, L.M.; Catov, J.M.; Simhan, H.N.; Holick, M.F.; Powers, R.W.; Roberts, J.M. Maternal vitamin D deficiency increases the risk of preeclampsia. J. Clin. Endocrinol. Metab. 2007, 92, 3517–3522. [Google Scholar] [CrossRef] [PubMed]
- Abedi, P.; Mohaghegh, Z.; Afshary, P.; Latifi, M. The relationship of serum vitamin D with pre-eclampsia in the Iranian women. Matern. Child Nutr. 2014, 10, 206–212. [Google Scholar] [CrossRef]
- Hyppönen, E.; Cavadino, A.; Williams, D.; Fraser, A.; Vereczkey, A.; Fraser, W.D.; Bánhidy, F.; Lawlor, D.; Czeizel, A.E. Vitamin D and Pre-Eclampsia: Original data, systematic review and meta-analysis. Ann. Nutr. Metab. 2014, 63, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Ullah, M.I.; Koch, C.A.; Tamanna, S.; Rouf, S.; Shamsuddin, L. Vitamin D deficiency and the risk of preeclampsia and eclampsia in Bangladesh. Horm. Metab. Res. 2013, 45, 682–687. [Google Scholar] [CrossRef] [PubMed]
- Wagner, C.L.; McNeil, R.; Hamilton, S.A.; Winkler, J.; Cook, C.R.; Warner, G.; Bivens, B.; Davis, D.J.; Smith, P.G.; Murphy, M.; et al. A randomized trial of vitamin D supplementation in 2 community health center networks in South Carolina. Am. J. Obstet. Gynecol. 2013, 208, 137.e1–137.e13. [Google Scholar] [CrossRef] [PubMed]
- Mackillop, L. Vitamin D Supplementation in Pregnancy; Oxford University Hospitals NHS Foundation Trust: Oxford, UK, 2017. [Google Scholar]
- Basile, L.A.; Taylor, S.N.; Wagner, C.L.; Horst, R.L.; Hollis, B.W. The Effect of High-Dose Vitamin D Supplementation on Serum Vitamin D Levels and Milk Calcium Concentration in Lactating Women and Their Infants. Breastfeed. Med. 2006, 1, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Hollis, B.W.; Wagner, C.L. Vitamin D requirements during lactation: High-dose maternal supplementation as therapy to prevent hypovitaminosis D for both the mother and the nursing infant 1–4. Am. J. Clin. Nutr. 2004, 80, 1752S–1758S. [Google Scholar] [CrossRef]
- Roth, D.E.; Abrams, S.A.; Aloia, J.; Bergeron, G.; Bourassa, M.W.; Brown, K.H.; Calvo, M.S.; Cashman, K.D.; Combs, G.; De-Regil, L.M.; et al. Global prevalence and disease burden of vitamin D deficiency: A roadmap for action in low-and middle-income countries. Ann. N. Y. Acad. Sci. 2018, 1430, 44–79. [Google Scholar] [CrossRef]
- Nnoaham, K.E.; Clarke, A. Low serum vitamin D levels and tuberculosis: A systematic review and meta-analysis. Int. J. Epidemiol. 2008, 37, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Martineau, A.R.; Timms, P.M.; Bothamley, G.H.; Hanifa, Y.; Islam, K.; Claxton, A.P.; Packe, G.E.; Moore-Gillon, J.C.; Darmalingam, M.; Davidson, R.N.; et al. High-dose vitamin D3 during intensive-phase antimicrobial treatment of pulmonary tuberculosis: A double-blind randomised controlled trial. Lancet 2011, 377, 242–250. [Google Scholar] [CrossRef] [PubMed]
- Camargo, C.A., Jr.; Ingham, T.; Wickens, K.; Thadhani, R.; Silvers, K.M.; Epton, M.J.; Town, G.I.; Pattemore, P.K.; Espinola, J.A.; Crane, J.; et al. Cord-blood 25-hydroxyvitamin D levels and risk of respiratory infection, wheezing, and asthma. Pediatrics 2011, 127, e180–e187. [Google Scholar] [CrossRef]
- Belderbos, M.E.; Houben, M.L.; Wilbrink, B.; Lentjes, E.; Bloemen, E.M.; Kimpen, J.L.L.; Rovers, M.; Bont, L. Cord blood vitamin D deficiency is associated with respiratory syncytial virus bronchiolitis. Pediatrics 2011, 127, e1513–e1520. [Google Scholar] [CrossRef]
- Alyasin, S.; Momen, T.; Kashef, S.; Alipour, A.; Amin, R. The relationship between serum 25 hydroxy vitamin D levels and asthma in children. Allergy Asthma Immunol. Res. 2011, 3, 251–255. [Google Scholar] [CrossRef]
- Mirzakhani, H.; Carey, V.J.; McElrath, T.F.; Laranjo, N.; O’Connor, G.; Iverson, R.E.; Lee-Parritz, A.; Strunk, R.C.; Bacharier, L.B.; Macones, G.A.; et al. The Association of Maternal Asthma and Early Pregnancy Vitamin D with Risk of Preeclampsia: An Observation From Vitamin D Antenatal Asthma Reduction Trial (VDAART). J. Allergy Clin. Immunol. Pract. 2018, 6, 600–608.e2. [Google Scholar] [CrossRef]
- Kim, M.J.; Kim, S.-N.; Lee, Y.W.; Choe, Y.B.; Ahn, K.J. Vitamin D status and efficacy of vitamin D supplementation in atopic dermatitis: A systematic review and meta-analysis. Nutrients 2016, 8, 789. [Google Scholar] [CrossRef]
- Camargo, C.A.; Ganmaa, D.; Sidbury, R.; Erdenedelger, K.; Radnaakhand, N.; Khandsuren, B. Randomized trial of vitamin D supplementation for winter-related atopic dermatitis in children. J. Allergy Clin. Immunol. 2014, 134, 831–835.e1. [Google Scholar] [CrossRef]
- Holick, M.F. Resurrection of vitamin D deficiency and rickets. J. Clin. Investig. 2006, 116, 2062–2072. [Google Scholar] [CrossRef] [PubMed]
- Weydert, J.A. Vitamin d in children’s health. Children 2014, 1, 208–226. [Google Scholar] [CrossRef] [PubMed]
- Poniedziałek-Czajkowska, E.; Mierzyński, R. Could vitamin D be effective in prevention of preeclampsia? Nutrients 2021, 13, 3854. [Google Scholar] [CrossRef] [PubMed]
- Jafarzadeh, L.; Motamedi, A.; Behradmanesh, M.; Hashemi, R. A Comparison of Serum Levels of 25-hydroxy Vitamin D in Pregnant Women at Risk for Gestational Diabetes Mellitus and Women Without Risk Factors. Mater. Socio Medica 2015, 27, 318–322. [Google Scholar] [CrossRef] [PubMed]
- Ede, G.; Keskin, U.; Yenen, M.C.; Samur, G. Lower vitamin D levels during the second trimester are associated with developing gestational diabetes mellitus: An observational cross-sectional study. Gynecol. Endocrinol. 2019, 35, 525–528. [Google Scholar] [CrossRef]
- Weissgerber, T.L.; Mudd, L.M. Preeclampsia and Diabetes. Curr. Diabetes Rep. 2015, 15, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Valdés, E.; Sepúlveda-Martínez, Á.; Manukián, B.; Parra-Cordero, M. Assessment of pregestational insulin resistance as a risk factor of preeclampsia. Gynecol. Obstet. Investig. 2014, 77, 111–116. [Google Scholar] [CrossRef]
- Hauth, J.C.; Clifton, R.G.; Roberts, J.M.; Myatt, L.; Spong, C.Y.; Leveno, K.J.; Varner, M.W.; Wapner, R.J.; Thorp, J.M.; Mercer, B.M.; et al. Maternal insulin resistance and preeclampsia. Am. J. Obstet. Gynecol. 2011, 204, 327.e1–327.e6. [Google Scholar] [CrossRef]
- Alsnes, I.V.; Janszky, I.; Forman, M.R.; Vatten, L.J.; Økland, I. A population-based study of associations between preeclampsia and later cardiovascular risk factors. Am. J. Obstet. Gynecol. 2014, 211, 657.e1–657.e7. [Google Scholar] [CrossRef]
- Engeland, A.; Bjørge, T.; Daltveit, A.K.; Skurtveit, S.; Vangen, S.; Vollset, S.E.; Furu, K. Risk of diabetes after gestational diabetes and preeclampsia. A registry-based study of 230,000 women in Norway. Eur. J. Epidemiol. 2011, 26, 157–163. [Google Scholar] [CrossRef]
- Feig, D.S.; Shah, B.R.; Lipscombe, L.L.; Wu, C.F.; Ray, J.G.; Lowe, J.; Hwee, J.; Booth, G.L. Preeclampsia as a Risk Factor for Diabetes: A Population-Based Cohort Study. PLOS Med. 2013, 10, e1001425. [Google Scholar] [CrossRef]
- Libby, G.; Murphy, D.J.; McEwan, N.F.; Greene, S.A.; Forsyth, J.S.; Chien, P.W.; Morris, A.D. Pre-eclampsia and the later development of type 2 diabetes in mothers and their children: An intergenerational study from the Walker cohort. Diabetologia 2007, 50, 523–530. [Google Scholar] [CrossRef]
- Männistö, T.; Mendola, P.; Vääräsmäki, M.; Järvelin, M.-R.; Hartikainen, A.-L.; Pouta, A.; Suvanto, E. Elevated blood pressure in pregnancy and subsequent chronic disease risk. Circulation 2013, 127, 681–690. [Google Scholar] [CrossRef] [PubMed]
- Savitz, D.A.; Danilack, V.A.; Elston, B.; Lipkind, H.S. Pregnancy-induced hypertension and diabetes and the risk of cardiovascular disease, stroke, and diabetes hospitalization in the year following delivery. Am. J. Epidemiol. 2014, 180, 41–44. [Google Scholar] [CrossRef] [PubMed]
- Freimane, K.Z.; Kerrigan, L.; Eastwood, K.-A.; Watson, C.J. Pre-Eclampsia Biomarkers for Women With Type 1 Diabetes Mellitus: A Comprehensive Review of Recent Literature. Front. Bioeng. Biotechnol. 2022, 10, 809528. [Google Scholar] [CrossRef]
- Azar, M.; Basu, A.; Jenkins, A.J.; Nankervis, A.J.; Hanssen, K.F.; Scholz, H.; Henriksen, T.; Garg, S.K.; Hammad, S.M.; Scardo, J.A.; et al. Serum carotenoids and fat-soluble vitamins in women with type 1 diabetes and preeclampsia: A longitudinal study. Diabetes Care 2011, 34, 1258–1264. [Google Scholar] [CrossRef]
- Vestgaard, M.; Secher, A.L.; Ringholm, L.; Jensen, J.B.; Damm, P.; Mathiesen, E.R. Vitamin D insufficiency, preterm delivery and preeclampsia in women with type 1 diabetes—An observational study. Acta Obstet. Et Gynecol. Scand. 2017, 96, 1197–1204. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.M.; Alobaid, A.; Malhis, T.N.; Khattab, A.F. Effect of vitamin D3 supplementation in pregnancy on risk of pre-eclampsia—Randomized controlled trial. Clin. Nutr. 2019, 38, 557–563. [Google Scholar] [CrossRef]
- Manasova, G.S.; Kuzmin, N.V.; Didenkul, N.V.; Derishov, S.V.; Badiuk, N.S. Supplementation of vitamin D in pregnant women with 25(OH) D deficiency and risk of preeclampsia development improves perinatal outcomes. PharmacologyOnLine 2021, 3, 1107–1116. Available online: http://pharmacologyonline.silae.it (accessed on 30 April 2021).
- Naghshineh, E.; Sheikhaliyan, S. Effect of vitamin D supplementation in the reduce risk of preeclampsia in nulliparous women. Adv. Biomed. Res. 2016, 5, 7. [Google Scholar] [CrossRef]
- Dahma, G.; Neamtu, R.; Nitu, R.; Gluhovschi, A.; Bratosin, F.; Grigoras, M.L.; Silaghi, C.; Citu, C.; Orlu, I.N.; Bhattarai, S.; et al. The Influence of Maternal Vitamin D Supplementation in Pregnancies Associated with Preeclampsia: A Case-Control Study. Nutrients 2022, 14, 3008. [Google Scholar] [CrossRef]
- Rostami, M.; Tehrani, F.R.; Simbar, M.; Bidhendi Yarandi, R.; Minooee, S.; Hollis, B.W.; Hosseinpanah, F. Effectiveness of prenatal Vitamin D deficiency screening and treatment program: A stratified randomized field trial. J. Clin. Endocrinol. Metab. 2018, 103, 2936–2948. [Google Scholar] [CrossRef]
- Jiang, X.; Wei, Y. Effect of vitamin D3 supplementation during pregnancy on high risk factors—A randomized controlled trial. J. Perinat. Med. 2021, 49, 480–484. [Google Scholar] [CrossRef]
- Mirzakhani, H.; Litonjua, A.A.; McElrath, T.F.; O’connor, G.; Lee-Parritz, A.; Iverson, R.; Macones, G.; Strunk, R.C.; Bacharier, L.B.; Zeiger, R.; et al. Early pregnancy Vitamin D status and risk of preeclampsia. J. Clin. Investig. 2016, 126, 4702–4715. [Google Scholar] [CrossRef]
- Samimi, M.; Kashi, M.; Foroozanfard, F.; Karamali, M.; Bahmani, F.; Asemi, Z.; Hamidian, Y.; Talari, H.R.; Esmaillzadeh, A. The effects of vitamin D plus calcium supplementation on metabolic profiles, biomarkers of inflammation, oxidative stress and pregnancy outcomes in pregnant women at risk for pre-eclampsia. J. Hum. Nutr. Diet. Off. J. Br. Diet. Assoc. 2016, 29, 505–515. [Google Scholar] [CrossRef] [PubMed]
- Azami, M.; Azadi, T.; Sc, M.; Farhang, S.; Rahmati, S.; Pourtaghi, K. The effects of multi mineral-vitamin D and vitamins (C+E) supplementation in the prevention of preeclampsia: An RCT. Int. J. Reprod. BioMedicine 2017, 15, 273–278. [Google Scholar] [CrossRef]
- Anderson, C.M.; Ralph, J.L.; Johnson, L.; Scheett, A.; Wright, M.L.; Taylor, J.Y.; Ohm, J.E.; Uthus, E. First trimester vitamin D status and placental epigenomics in preeclampsia among Northern Plains primiparas. Life Sci. 2015, 129, 10–15. [Google Scholar] [CrossRef]
- Pashapour, S.; Golmohammadlou, S.; Behroozi-Lak, T.; Ghasemnejad-Berenji, H.; Sadeghpour, S.; Ghasemnejad-Berenji, M. Relationship between low maternal vitamin D status and the risk of severe preeclampsia: A case control study. Pregnancy Hypertens. 2019, 15, 161–165. [Google Scholar] [CrossRef]
- Forde, H.; Crowley, R.K.; McKenna, M.J.; Kilbane, M.T.; Conway, M.; McDonnell, C.M.; Twomey, P.J.; McAuliffe, F.M. No effect of calcium and vitamin D intake on maternal blood pressure in a healthy pregnant population. Eur. J. Obstet. Gynecol. Reprod. Biol. 2021, 264, 8–14. [Google Scholar] [CrossRef]
- Bodnar, L.M.; Simhan, H.N.; Catov, J.M.; Roberts, J.M.; Platt, R.W.; Diesel, J.C.; Klebanoff, M.A. Maternal vitamin D status and the risk of mild and severe preeclampsia. Epidemiology 2014, 25, 207–214. [Google Scholar] [CrossRef]
- Thorsen, S.U.; Collier, F.; Pezic, A.; O’hely, M.; Clarke, M.; Tang, M.L.K.; Burgner, D.; Vuillermin, P.; Ponsonby, A.-L. Maternal and Cord Blood 25-Hydroxyvitamin D3 Are Associated with Increased Cord Blood and Naive and Activated Regulatory T Cells: The Barwon Infant Study. J. Immunol. 2021, 206, 874–882. [Google Scholar] [CrossRef] [PubMed]
- Chia, A.R.; Tint, M.T.; Han, C.Y.; Chen, L.W.; Colega, M.; Aris, I.M.; Chua, M.C.; Tan, K.H.; Yap, F.; Shek, L.P.C.; et al. Adherence to a healthy eating index for pregnant women is associated with lower neonatal adiposity in a multiethnic Asian cohort: The Growing Up in Singapore Towards healthy Outcomes (GUSTO) Study. Am. J. Clin. Nutr. 2018, 107, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Burris, H.H.; Rifas-Shiman, S.L.; Huh, S.Y.; Kleinman, K.; Litonjua, A.A.; Oken, E.; Rich-Edwards, J.W.; Camargo, C.A.; Gillman, M.W. Vitamin D status and hypertensive disorders in pregnancy. Ann. Epidemiology 2014, 24, 399–403.e1. [Google Scholar] [CrossRef] [PubMed]
- Kiely, M.E.; Zhang, J.Y.; Kinsella, M.; Khashan, A.S.; Kenny, L.C. Vitamin D status is associated with uteroplacental dysfunction indicated by pre-eclampsia and small-for-gestational-age birth in a large prospective pregnancy cohort in Ireland with low Vitamin D status. Am. J. Clin. Nutr. 2016, 104, 354–361. [Google Scholar] [CrossRef] [PubMed]
- Fogacci, S.; Fogacci, F.; Cicero, A.F. Nutraceuticals and hypertensive disorders in pregnancy: The available clinical evidence. Nutrients 2020, 12, 378. [Google Scholar] [CrossRef] [PubMed]
- Borghi, C.; Cicero, A.F.G.; Degli Esposti, D.; Immordino, V.; Bacchelli, S.; Rizzo, N.; Santi, F.; Ambrosioni, E. Hemodynamic and neurohumoral profile in patients with different types of hypertension in pregnancy. Intern. Emerg. Med. 2011, 6, 227–234. [Google Scholar] [CrossRef]
- Cicero, A.F.; Degli Esposti, D.; Immordino, V.; Morbini, M.; Baronio, C.; Rosticci, M.; Borghi, C. Independent Determinants of Maternal and Fetal Outcomes in a Sample of Pregnant Outpatients With Normal Blood Pressure, Chronic Hypertension, Gestational Hypertension, and Preeclampsia. J. Clin. Hypertens. 2015, 17, 777–782. [Google Scholar] [CrossRef]
- Liu, N.; Hewison, M. Vitamin D, the placenta and pregnancy. Arch. Biochem. Biophys. 2012, 523, 37–47. [Google Scholar] [CrossRef] [PubMed]
| Authors | Country | Study Design | Maternal Age | N Total | Duration of Investigation | Intervention-Groups | N/Group | Mean Age/Group | Preeclampsia | Statistically Significant Correlation between Preeclampsia and Vitamin D? (Yes/No) |
|---|---|---|---|---|---|---|---|---|---|---|
| Ali, 2019 [107] | Saudi Arabia | Randomized controlled study | 20–40 | 164 | October 2012– October 2015 | Group 1: 400 IU vitamin D3; group 2: 4000 IU vitamin D3 | Group 1: n = 81 Group 2: n = 83 | Group 1: 29.3 Group 2: 29.4 | Group 1: 7.4% Group 2: 1.2% | Yes |
| Behjat Sasan, 2017 [34] | Iran | Randomized controlled clinical trial | 24–35 | 142 | n/a | Group 1: 50,000 IU vitamin D3/2 weeks; group 2: placebo | Group 1: n = 70 Group 2: n = 72 | Group 1: 32.04 ± 5.901 Group 2: 29.77 ± 5.21 | Total: 23.2% Group 1: 15.7% Group 2: 30.6% | Yes |
| Manasova, 2021 [108] | Ukraine | Randomized controlled clinical trial | 27–33 | 54 | 2017–2020 | Group 1: multivitamin mineral complex (including cholecalciferol 500 IU); group 2: multivitamin mineral complex until 16 w and then 2000 IU until the end of the pregnancy | Group 1: n = 25 Group 2: n = 29 | Group 1: 27.4 ± 4.4 Group 2: 28.2 ± 4.6 | n/a | Yes |
| Naghshineh, 2016 [109] | Iran | Double-blind randomized controlled trial | 25 ± 4.1 (mean) | 140 | May–January 2012 | Group 1: 600 IU vitamin D/day; group 2: placebo | Group 1: n = 68 Group 2: n = 70 | Group 1: 25 ± 3.8 Group 2: 24.8 ± 4.4 | Group 1: 2 women Group 2: 7 women | No |
| Dahma, 2022 [110] | Romania | Single-centric case–control | 25–34 | 198 | 2018–2022 | Group 1: No supplementation; group 2: Low dose, 2000 IU during first trimester; group 3: High dose, 4000 IU | Group 1: n = 59 Group 2: n = 63 Group 3: n = 76 | n/a | Group 1: 18.6% Group 2: 9.5% Group 3: 5.3% | Yes |
| Rostami, 2018 [111] | Iran | Stratified randomized field trial | 18–40 | Phase 1: 2500 Phase 2: 800 | n/a | Moderate-deficiency: Group 1: 50,000 IU D3/w for 6 w; group 2: same as Group 1 + 50,000 IU D3/month until delivery; group 3: single dose of 300,000 IU D3; group 4: same as Group 3 + 50,000 IU/month until delivery. Severe-deficiency: Group 5: 50,000 IU/w for 12 w; group 6: same as Group 5 + 50,000 IU/month until delivery; group 7: two doses of 300,000 IU for 6 w; group 8: same as Group 7 + 50,000/month until delivery | n/a | Screening site: 25–32 (mean 29) Non-screening site: 25–32 (mean 29) | Moderate-deficiency screening site: 7% Severe-deficiency screening site: 8% Total screening site: 8% Moderate-deficiency non-screening site: 13% Severe-deficiency-non-screening site: 23% Total non-screening site: 17% | Yes |
| Xiaomang, 2021 [112] | Finland | Open label randomized study | 20–40 | 450 | January 2016– December 2018 | Group 1: low dose 400 IU/d; group 2: middle dose 1500 IU/d; group 3: high dose 4000 IU/d | Group 1: n = 135 Group 2: n = 134 Group 3: n = 138 | Group 1: 27.76 ± 3.16 Group 2: 28.54 ± 3.27 Group 3: 28.94 ± 3.21 | Group 1: 9.63% Group 2: 6.72% Group 3: 1.45% | Yes |
| Mirzakhani, 2016 [113] | Boston Massachusetts, Missouri, California | VDAART study | 18–40 | 881, available data for 816 | October 2009– July 2011 | Prenatal vitamins 400 IU cholecalciferol G1:4400 IU/day; G2: placebo/day | G1: n = 408 G2: n = 408 | Group 1: 27.5 Group 2: 27.2 | For 8.2% of all subjects, supplementation at week 10–18 did not reduce preeclampsia. Sufficient vitamin during enrollment and pregnancy lower the risk of preeclampsia | Yes |
| Samimi, 2016 [114] | Iran | Randomized double-blind placebo-controlled clinical trial | 18–40 | 60 | September 2014–February 2015 | Group 1: 50,000 IU vitamin D3; Group 2: placebo | Group 1: 50,000 IU vitamin D3 n = 30: Group 2: placebo n = 30 | Group 2: n = 27.1, Group 1: n = 27.3 | Vitamin D+ Ca for 12 w affected blood pressure | Yes |
| Azami, 2017 [115] | Iran | Randomized controlled clinical trial | 31.63 ± 6.13 (mean) | 90 | n/a | Group 1 received Ferrous sulfate (1 tablet/day) + one tablet of Claci-care multimineral-vitamin D containing 800 mg calcium, 200 mg magnesium, 8 mg zinc and 400 IU Vitamin D3 per day, group 2 received Ferrous sulfate (1 tablet/day) + 250 mg vitamin C + 55 mg vitamin E, and group 3 (controls) received only one Ferrous sulfate tablet daily | Three equal (n = 30) groups | Group 1: 33.03 ± 6.49, Group 2: 31.73 ± 6.41, Group 3 (control): 30.15 ± 5.28 | n/a percentage per group | Yes |
| Authors | Country | Study Design | Maternal Age | N total | Duration of Study | Eligibility Criteria or Exclusion Criteria | Methodology of the Study | N/Group | Mean Age/Group | Preeclampsia | Statistically Significant Correlation between Preeclampsia and Vitamin D? (Yes/No) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Kelly, 2020 | Norway, Australia, USA | Cohort study | n/a | 66 | n/a | Eligibility criteria for health: hypertension free, no proteinuria, no microalbuminuria at enrollment. Eligibility criteria for preeclampsia: new- onset hypertension and proteinuria at >20 w | Total, bioavailable, free 25(OH)D, 1,25(OH)2D and VDBP at week 12, 22 and 32 | Type 1 diabetes mellitus subgroups: Diabetes mellitus with preeclampsia: n = 23. Normotensive with diabetes: n = 24. Normotensive and non-diabetic as controls: n = 19. | Diabetes mellitus with preeclampsia: 28.5 ± 5.6. Normotensive with diabetes: 29.9 ± 3.8. Normotensive and non-diabetic: 31.4 ± 4.5. | Vitamin D deficiency was more common in women with T1DM but no concentration predicted preeclampsia. At visit 2 and 3, the 1,25(OH)2D concentration was related with preeclampsia in women with T1DM such as lower concentrations of VDBP | Yes |
| Mirzakhani, 2018 | Boston Massachusetts, Missouri, California | VDAART study | 18–39 | 816 | September 2009–July 2011 | Group 1: 4000 IU of vitamin D/day + multivitamin with 400 IU vitamin D; group 2: placebo + multivitamin with 400 IU vitamin D | Group 1: 4000 IU of vitamin D/day + multivitamin with 400 IU vitamin D; group 2: placebo + multivitamin with 400 IU vitamin D | Asthma group: n = 327. No asthma group: n = 489 | Asthma group: 25.67 ± 5.45 No asthma group: 27.79 ± 5.49 | +PE+asthma: n = 29 +PE-asthma: n = 38 Statistical difference among women with controlled and uncontrolled asthma, Vitamin D supplementation reduced risk of preeclampsia by controlling the asthmatic condition | Yes |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giourga, C.; Papadopoulou, S.K.; Voulgaridou, G.; Karastogiannidou, C.; Giaginis, C.; Pritsa, A. Vitamin D Deficiency as a Risk Factor of Preeclampsia during Pregnancy. Diseases 2023, 11, 158. https://doi.org/10.3390/diseases11040158
Giourga C, Papadopoulou SK, Voulgaridou G, Karastogiannidou C, Giaginis C, Pritsa A. Vitamin D Deficiency as a Risk Factor of Preeclampsia during Pregnancy. Diseases. 2023; 11(4):158. https://doi.org/10.3390/diseases11040158
Chicago/Turabian StyleGiourga, Chrysoula, Sousana K. Papadopoulou, Gavriela Voulgaridou, Calliope Karastogiannidou, Constantinos Giaginis, and Agathi Pritsa. 2023. "Vitamin D Deficiency as a Risk Factor of Preeclampsia during Pregnancy" Diseases 11, no. 4: 158. https://doi.org/10.3390/diseases11040158
APA StyleGiourga, C., Papadopoulou, S. K., Voulgaridou, G., Karastogiannidou, C., Giaginis, C., & Pritsa, A. (2023). Vitamin D Deficiency as a Risk Factor of Preeclampsia during Pregnancy. Diseases, 11(4), 158. https://doi.org/10.3390/diseases11040158








