E-textiles in Clinical Rehabilitation: A Scoping Review
Abstract
:1. Introduction
- 1
- Sewable and washable microcontrollers like the commercially available, Arduino Lilypad (lilypadarduino.org) offer researchers affordable and fabric-friendly embedded platforms for quickly developing robust prototypes [2].
- 2
- Flexible circuits have allowed the textile integration of advanced electronics capable of sensing and information transmission, without compromising the comfort of the wearer. Likewise, smaller and flexible power sources for e-textile applications have emerged [3] while energy-harvesting power electronics are already under development by companies like PowerLeap.(www.powerleap.com).
- 3
- Conductive threads and yarns, usually composed of stainless steel or conductive silver with a nylon core, have become widely available. The properties of such threads and fabric transmission lines for textile computing applications have been documented [4,5,6], facilitating their selection and implementation in e-textile solutions.
- 4
1.1.Degree of Integration
| Rating | Description |
|---|---|
| 0→ | Wearable computer: no textile integration |
| 1→ | Superficial integration: components in pockets or connected to fabric with snaps |
| 2→ | Partial integration: some sensing components incorporated (e.g., woven, knit, printed, embroidered etc.) into fabric |
| 3→ | Partial integration: all sensing components incorporated (e.g., woven, knit, printed, embroidered etc.) into fabric |
| 4→ | Partial integration: wiring and sensing woven into fabric |
| 5→ | Towards full integration: sensing, wiring and power supply woven into fabric |
2. Methods
| Textile | Electronic | Rehab |
|---|---|---|
| fabric clothing | smart intelligent sensing | physiology biomechanics bio- |
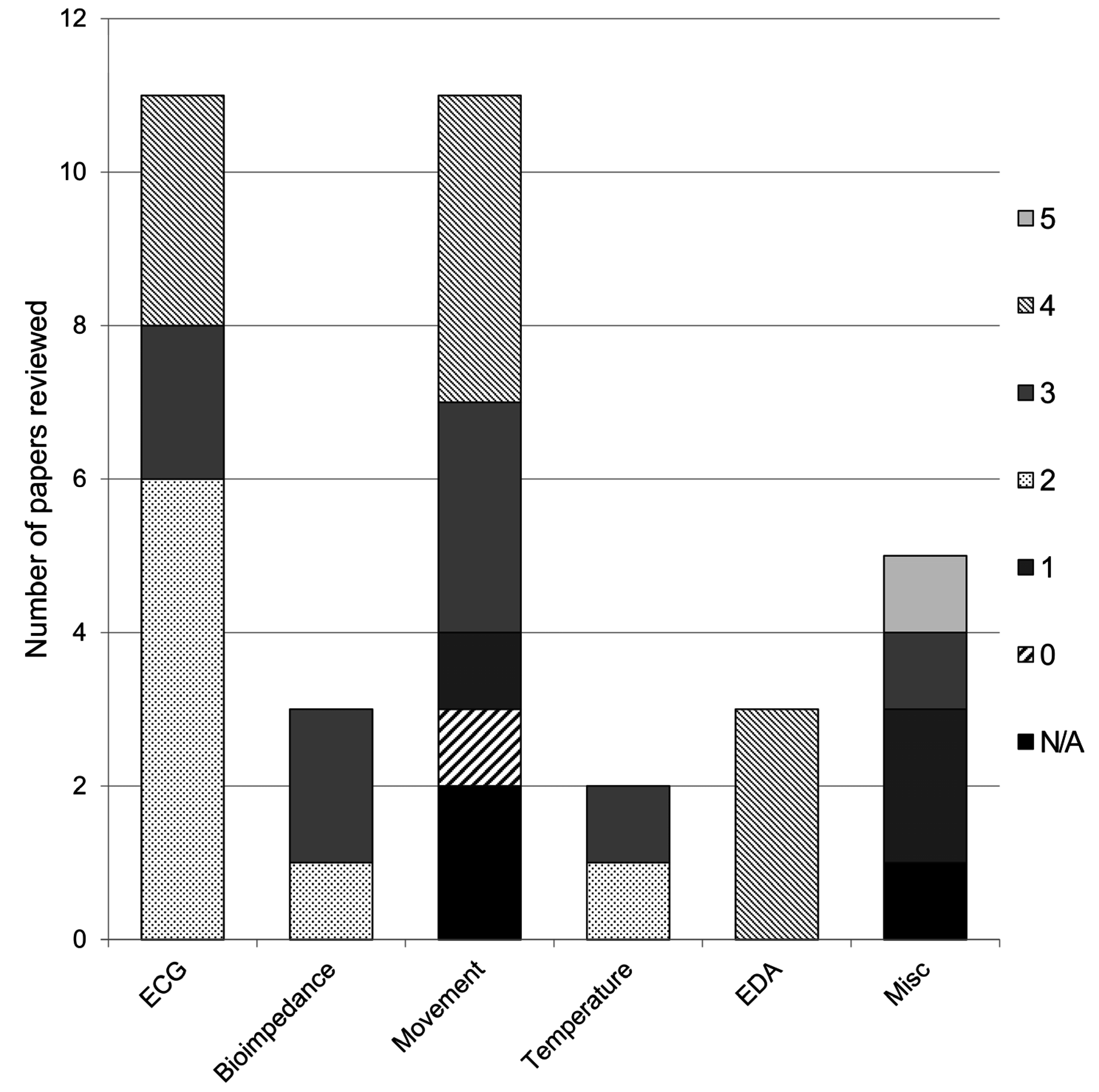
3. Results
| Reference | Stage of Development | # Study Participants | Participant Type | Sensed phenomenon | Sensor validation | DoI | Area Application of |
|---|---|---|---|---|---|---|---|
| Adnane et al. [15] | Empirical testing | 1 | Target | ECG, respiration | Non-fabric alternative: 3-lead ECG; pneumography | 2 | Sleep disorders |
| Angelidis [16] | Conceptual design | N/A | N/A | ECG, BP, SpO2, temperature, sweat | N/A | 5 | Healthcare: general |
| Baek et al. [17] | Empirical testing | 5 | Adult, male, healthy | ECG, pulse, BP | Non-fabric alternative: Biopac ECG& PPG, Finometerpro blood pressure | 2 | Hospital monitoring, Remote monitoring |
| Bianchi et al. [18] | Empirical testing | 24 | Adult, healthy | ECG | Gold standard; clinical polysomnography | 4 | Sleep disorders |
| Empirical testing | 50 | Target, database | ECG | ||||
| Cho et al. [19] | Empirical testing | 2 | Adult, male, healthy | ECG | None | 4 | Remote monitoring |
| Fletcher et al. [20] | Lab testing | N/A | N/A | EDA, pulse | Gold standard | 4 | Emotion |
| Empirical testing | 12 | Adult, healthy | EDA, acceleration, tempreature | Gold standard | |||
| Gioberto and Dunne [21] | Lab testing | N/A | N/A | Strain | None | 3 | Monitoring: general |
| Giorgino et al. [22] | Empirical testing | 3 | Unknown | Strain | Human expert | 4 | Motor rehabilitation |
| Goy et al. [23] | Lab testing | N/A | N/A | VOP | N/A | 2 | Remote monitoring |
| Empirical testing | 5 | Adult, healthy | VOP | Gold standard; 4 Ag/AgCl electrodes | |||
| Hannikainen et al. [24] | Empirical testing | 9 | Adult, healthy; Target | Bioimpedance | None | 3 | Remote monitoring |
| Harms et al. [25] | Computer model | 5 | Adult, healthy | Acceleration | None | N/A | Motor rehabilitation |
| Hong et al. [26] | Empirical testing | 18 | Adult, healthy | ECG | Non-fabric alternative: 3 lead ECG | 4 | Remote monitoring |
| Kim and Cho [27] | Empirical testing | 12 | Target | BP, HR | N/A | 2 | Treatment |
| Lanata et al. [28] | Lab testing | N/A | N/A | EDA | Platinum electrodes | N/A | Emotion detection |
| Empirical testing | 35 | Adult, healthy | EDA | Ag/AgCl electrode | 4 | ||
| Lee et al. [29] | Empirical testing | Unknown | Unknown | ECG, respiration, pulse wave velocity, EMG, pressure | Non-fabric alternatives: commercial sensors | 3 | Remote monitoring |
| Lee et al. [30] | Empirical testing | 15 | Adult, male, healthy | Knee joint movements via bioimpedance | Non-fabric alternative: tilt sensor | 3 | Motor rehabilitation |
| Lee et al. [31] | Empirical testing | 1 | Adult, healthy | EDA, pulse wave | None | 4 | Remote monitoring |
| Lee and Chung [32] | Empirical testing | 1 | Adult, healthy | ECG, acceleration | None | 2 | Remote monitoring |
| Li et al. [33] | Lab testing (mathematical modeling) | N/A | N/A | Temperature | Mathematical model | 2 | Remote monitoring; diagnostic tool |
| Lofhede et al. [34] | Empirical testing | 5 | Adult, healthy | EEG | Standard EEG electrodes | 3 | Neonatal monitoring |
| Lopez et al. [35] | Lab testing | N/A | N/A | ECG, temperature, acceleration, position | Simulated signals | 2 | Hospital monitoring |
| Empirical testing | 5 | Target | ECG temperature, acceleration, position | None | |||
| Lorussi et al. [36] | Lab testing | N/A | N/A | Bend angle | Non-fabric alternative: electrogoniometer | 0 | Motor rehabilitation |
| Empirical testing | 1 | ||||||
| Marquez et al. [37] | Empirical testing | 3 | Adult, male, healthy | Bioimpedance | Gold standard: clinical bioimpedance spectrometer | 3 | Remote monitoring |
| Preece et al. [38] | Empirical testing | 20 | Adult, healthy | Strain | Non-fabric alternative: AMTI force platforms | 4 | Motor rehabilitation |
| Di Renzo et al. [39] | Lab testing | N/A | N/A | Posture | Non-fabric alternative: “traditional” ECG (no further details provided) | N/A | General e-textiles |
| Empirical testing | Target | ECG, respiration | 4 | ||||
| Schwarz et al. [40] | Lab testing | N/A | N/A | Electroconductivity | Mathematical model | N/A | General e-textile |
| Shu et al. [41] | Empirical testing | 8 | Adult, male, healthy & Target | Pressure | Gold standard: force platform; commercial in-sole pressure system | 4 | Hospital monitoring |
| Song et al. [42] | Empirical testing | 3 | Adult, healthy | ECG | None | 3 | Healthcare: general |
| Tormene et al. [43] | Empirical testing | 1 | Adult, male, healthy | Strain | Non-fabric alternative: triaxial accelerometer & magnetometer | 4 | Motor rehabilitation |
| Vuorela et al. [44] | Empirical testing | 1 | Adult, healthy | ECG, respiration | Gold standard: pneumotachograph; clinical ECG (# leads not specified) | 2 | Remote monitoring |
| Yamada et al. [45] | Lab & empirical testing | 1 | N/A | Strain | None | 1 | Motor rehabilitation |
| Zhang et al. [46] | Lab testing | N/A | N/A | ECG, respiration, SpO2 | Signal database, lung simulator & patient simulator | 2 | Remote monitoring |
| Empirical testing | 15 | Adult, male, healthy | Respiration | Gold standard: clinical ventilator tester | |||
| Empirical testing | 10 | Adult, male, healthy | ECG | Non-fabric alternative: Polar HR monitor | |||
| Zheng et al. [47] | Lab testing | N/A | N/A | Power | Conventional discharge policies | 1 | General e-textiles |
| Zysset et al. [48] | Empirical testing | N/A | N/A | Temperature, acceleration | None | 3 | General e-textiles |
3.1. Electrocardiogram (ECG)
3.1.1. Phenomenological Background
3.1.2. Current Practice
3.1.3. Textile Innovations
3.1.3.1. Sensor development
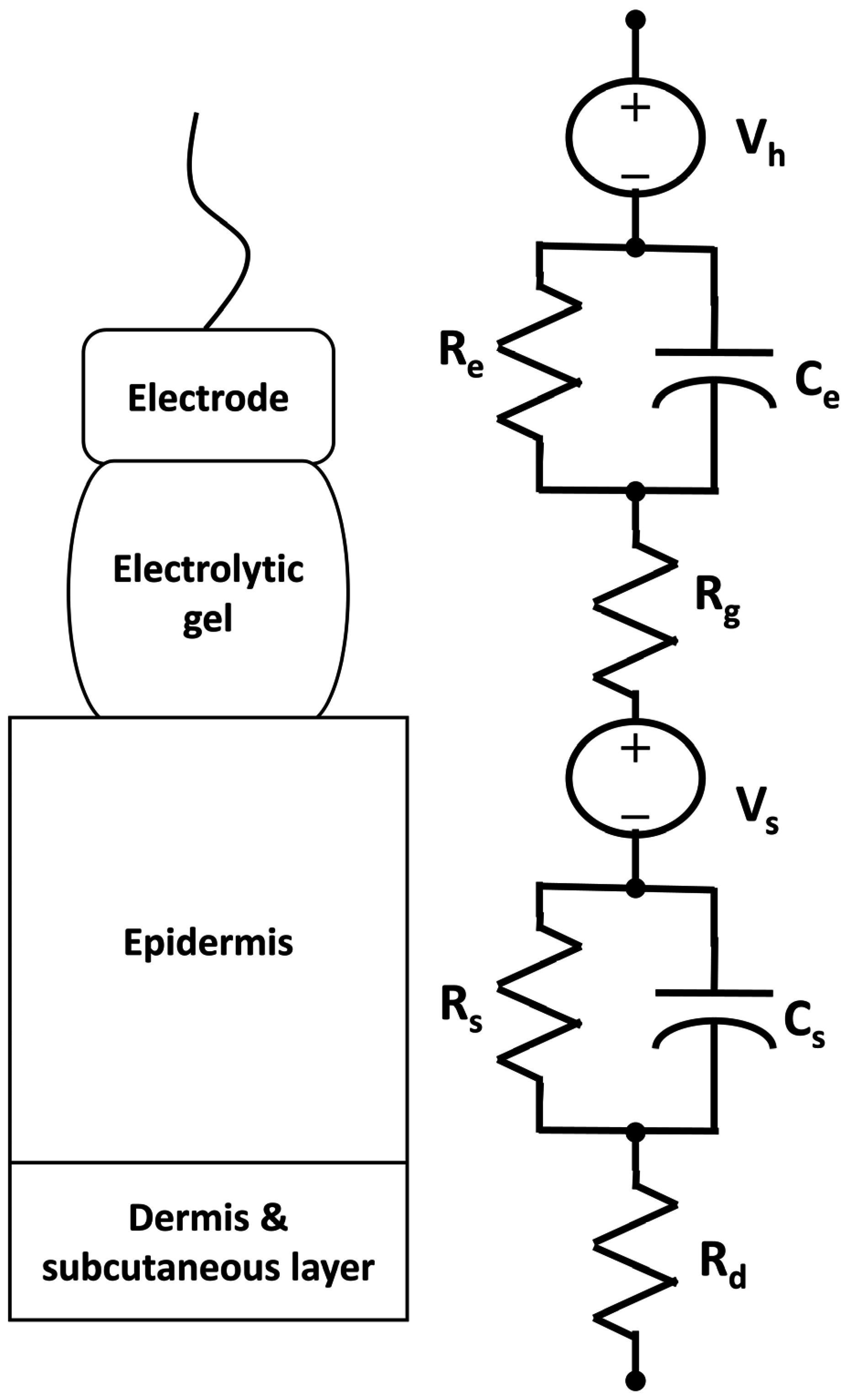
Improving fabric-based ECG signals
PVDF
Active electrodes
Improving skin-electrode interface
Sensing shirt/belt for sleep measurements
Monitoring
3.1.4. Merits and Limitations
3.2. Bioimpedance
3.2.1. Phenomenological Background
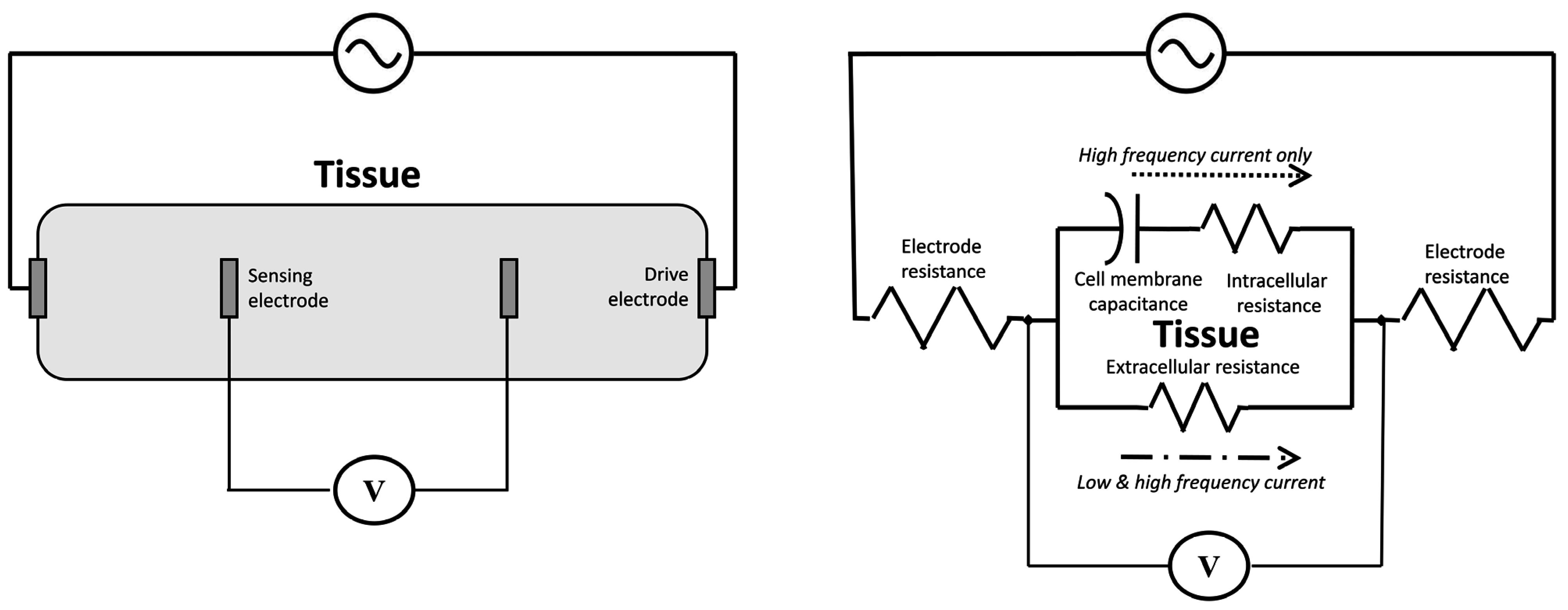
3.2.2. Current Practice
3.2.3. Textile Innovations
3.2.4. Merits and Limitations
3.3. Movement and Posture
3.3.1. Phenomological Background
3.3.2. Current Practice
3.3.3. Textile Innovations
3.3.4. Merits and Limitations
3.4. Temperature
3.4.1. Phenomenological Background
3.4.2. Current Practice
3.4.3. Textile Innovations
3.4.4. Merits and Limitations
3.5. Electrodermal Activity
3.5.1. Phenomenological Background
3.5.2. Current Practice
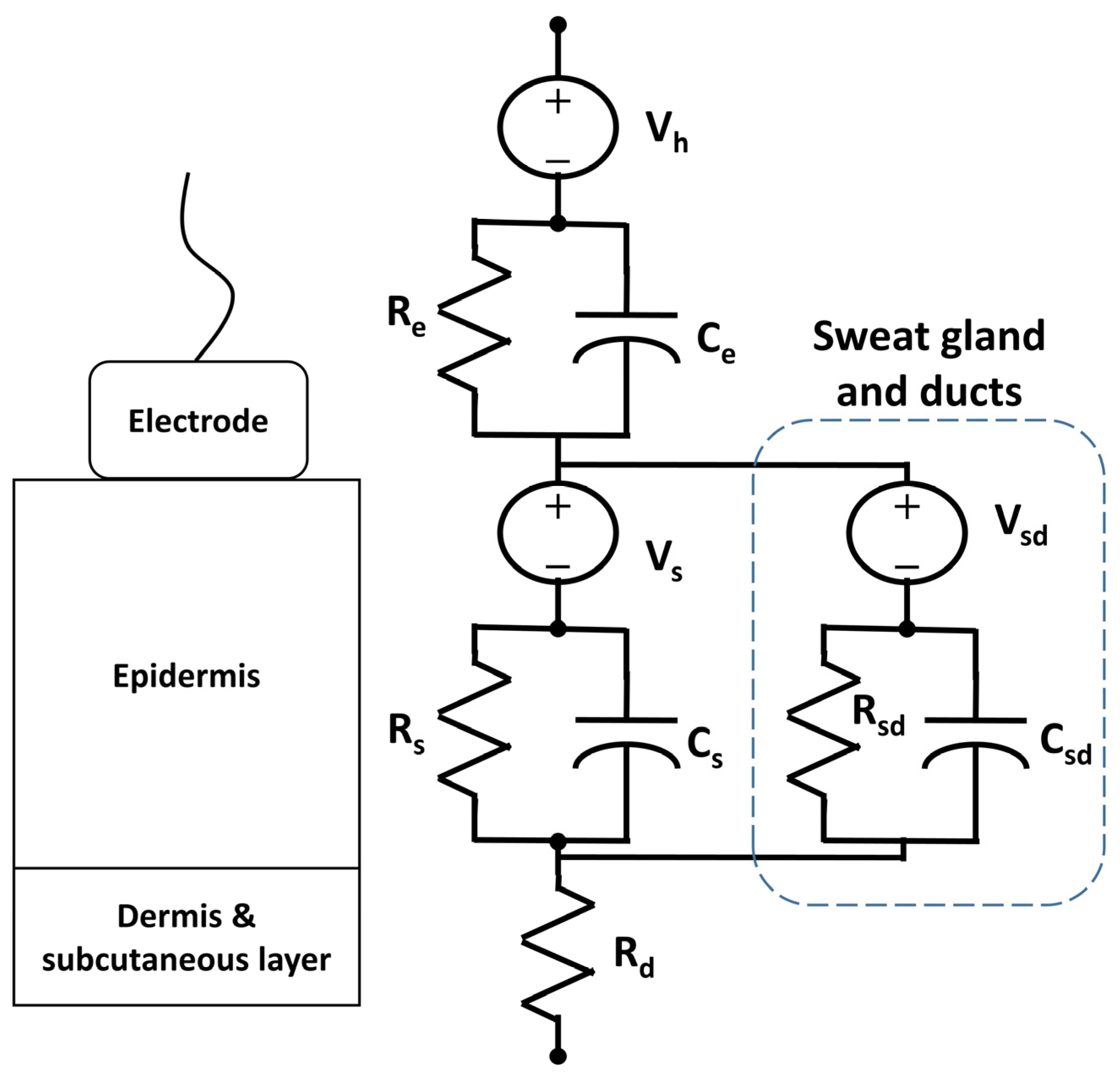
3.5.3. Textile Innovations
3.5.4. Merits and Limitations
3.6. Miscellaneous
4. Discussion
5. Recommendations
- 1
- Future research ought to validate textile sensing of a particular physiological or biomechanical phenomenon against its corresponding clinical gold standard. This was a common gap across the reviewed papers.
- 2
- In the spirit of patient self-management, future work may entertain the potential of incorporating textile actuation and hence, immediate sensory feedback to the wearer.
- 3
- While sensing elements have experienced a boon in fabric integration, connecting circuitry and power sources still lag behind in textile assimilation. Truly imperceptible e-textiles will require full system integration.
- 4
- With continued improvements in e-textile signal quality and system integration, it would behoove researchers to initiate testing with clinical populations in ambulatory settings. In particular, future research should consider issues of signal stability over time and across user activities as well as textile sensor integrity with wear and wash.
6. Limitations
Conclusions
Conflicts of Interest
Author Contributions
References
- Park, S.; Mackenzie, K.; Jayaraman, S. The Wearable Motherboard: A Framework for Personalized Mobile Information Processing (PMIP). In Proceedings of the 39th Design Automation Conference, New Orleans, LA, USA, 10–14 June 2002.
- Buechley, L.; Eisenberg, M. Fabric PCBs, electronic sequins, and socket buttons: Techniques for e-textile craft. Pers. Ubiquitous Comput. 2009, 13, 133–150. [Google Scholar] [CrossRef]
- Meoli, D.; May-Plumlee, T. Interactive electronic textile development: A review of technologies. J. Textile Appar. Technol. Manag. 2002, 2, 1–12. [Google Scholar]
- Post, E.R.; Orth, M.; Russo, R.R.; Gershenfeld, N. E-broidery: Design and fabrication of textile-based computing. IBM Syst. J. 2000, 39, 840–860. [Google Scholar] [CrossRef]
- Cottet, D.; Grzyb, J.; Kirstein, T.; Tröster, G. Electrical characterization of textile transmission lines. IEEE Trans. Adv. Packag. 2003, 26, 182–190. [Google Scholar] [CrossRef]
- Maggie, O. Defining Flexibility and Sewability in Conductive Yarns. In Proceedings of the Materials Research Society (MRS) Symposium, San Francisco, CA, USA, 25–29 April 2011; Volume 736, pp. 37–48.
- Bonderover, E.; Wagner, S. A woven inverter circuit for e-textile applications. IEEE Electr. Device Lett. 2004, 25, 295–297. [Google Scholar] [CrossRef]
- Lee, J.B.; Subramanian, V. Organic Transistors on Fiber: A First Step towards Electronic Textiles. In Proceedings of the Technical Digest—International Electron Devices Meeting, Washington, WA, USA, 8–10 December 2003.
- Lee, J.B.; Subramanian, V. Weave patterned organic transistors on fiber for e-textiles. IEEE Trans. Electr. Devices 2005, 52, 269–275. [Google Scholar] [CrossRef]
- Edmison, J.; Jones, M.; Nakad, Z.; Martin, T. Using piezoelectric materials for wearable electronic textiles. In Proceedings of the Sixth International Symposium on Wearable Computers (ISWC), Seattle, WD, USA, 7–10 October 2002; pp. 41–48.
- Pacelli, M.; Loriga, G.; Taccini, N.; Paradiso, R.M.D. 3rd IEEE EMBS International Summer School on, B. Sensing fabrics for monitoring physiological and biomechanical variables: E-textile solutions. In Proceedings of the 3rd IEEE/EMBS International Summer School on Medical Devices and Biosensors, Cambridge, MA, USA, 4–6 September 2006.
- Pantelopoulos, A.; Bourbakis, N.G. A survey on wearable sensor-based systems for health monitoring and prognosis. IEEE Trans. Syst. Man Cybern. Part C Appl. Rev. 2010, 40, 1–12. [Google Scholar] [CrossRef]
- Chan, M.; Esteve, D.; Fourniols, J.Y.; Escriba, C.; Campo, E. Smart wearable systems: Current status and future challenges. Artif. Intell. Med. 2012, 56, 137–156. [Google Scholar] [CrossRef] [PubMed]
- Bonato, P. Advances in wearable technology and applications in physical medicine and rehabilitation. J. NeuroEng. Rehabil. 2005, 2. [Google Scholar] [CrossRef]
- Adnane, M.; Jiang, Z.; Choi, S.; Jang, H. Detecting specific health-related events using an integrated sensor system for vital sign monitoring. Sensors 2009, 9, 6897–6912. [Google Scholar] [CrossRef] [PubMed]
- Angelidis, P.A. Personalised physical exercise regime for chronic patients through a wearable ICT platform. Int. J. Electron. Healthc. 2010, 5, 355–370. [Google Scholar] [CrossRef] [PubMed]
- Baek, H.J.; Chung, G.S.; Kim, K.K.; Park, K.S. A smart health monitoring chair for nonintrusive measurement of biological signals. IEEE Trans. Inf. Technol. Biomed. 2012, 16, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, A.M.; Mendez, M.O.; Cerutti, S. Processing of signals recorded through smart devices: Sleep-quality assessment. IEEE Trans. Inf. Technol. Biomed. 2010, 14, 741–747. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.S.; Koo, S.M.; Lee, J.; Cho, H.; Kang, D.H.; Song, H.Y.; Lee, J.W.; Lee, K.H.; Lee, Y.J. Heart monitoring garments using textile electrodes for healthcare applications. J. Med. Syst. 2011, 35, 189–201. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, R.R.; Dobson, K.; Goodwin, M.S.; Eydgahi, H.; Wilder-Smith, O.; Fernholz, D.; Kuboyama, Y.; Hedman, E.B.; Poh, M.Z.A.; Picard, R.W.A. ICalm: Wearable sensor and network architecture for wirelessly communicating and logging autonomic activity. IEEE Trans Inf. Technol Biomed. 2010, 14, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Gioberto, G.; Dunne, L.E. Overlock-stitched stretch sensors: Characterization and effect of fabric property. J. Textile Appar. Technol. Manag. 2013, 8, 1–14. [Google Scholar]
- Giorgino, T.; Tormene, P.; Lorussi, F.; de Rossi, D.; Quaglini, S. Sensor evaluation for wearable strain gauges in neurological rehabilitation. IEEE Trans. Neural Syst. Rehabil. Eng. 2009, 17, 409–415. [Google Scholar] [CrossRef] [PubMed]
- Goy, C.B.; Dominguez, J.M.; Gómez López, M.A.; Madrid, R.E.; Herrera, M.C. Electrical characterization of conductive textile materials and its evaluation as electrodes for venous occlusion plethysmography. J. Med. Eng. Technol. 2013, 37, 359–367. [Google Scholar] [CrossRef] [PubMed]
- Hannikainen, J.; Vuorela, T.; Vanhala, J. Physiological measurements in smart clothing: A case study of total body water estimation with bioimpedance. Trans. Inst. Meas. Control 2007, 29, 337–354. [Google Scholar] [CrossRef]
- Harms, H.; Amft, O.; Troester, G. Estimating posture-recognition performance in sensing garments using geometric wrinkle modeling. IEEE Trans. Inf. Technol. Biomed. 2010, 14, 1436–1445. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.; Yang, Y.; Kim, S.; Shin, S.; Lee, I.; Jang, Y.; Kim, K.; Yi, H. Performance study of the wearable one-lead wireless electrocardiographic monitoring system. Telemed. J. E-Health 2009, 15, 166–175. [Google Scholar] [CrossRef] [PubMed]
- Kim, R.H.; Cho, G. Effectiveness of the smart healthcare glove system for elderly persons with hypertension. Hum. Factors Ergon. Manuf. 2013, 23, 198–212. [Google Scholar] [CrossRef]
- Lanata, A.; Gaetano, V.; Scilingo, E. A novel EDA glove based on textile-integrated electronics for affective computing. Med. Biol. Eng. Comput. 2012, 50, 1163–1172. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Ahn, Y.; Prabu, A.; Kim, K. Piezoelectric Polymer and Piezocapacitive Nanoweb Based Sensors for Monitoring Vital Signals and Energy Expenditure in Smart Textiles. J. Fiber Bioeng. Inf. 2013, 6, 369–381. [Google Scholar]
- Lee, B.W.; Lee, C.; Kim, J.; Lee, M. Optimum conductive fabric sensor sites for evaluating the status of knee joint movements using bio-impedance. Biomed. Eng. Online 2011, 10, 48. [Google Scholar] [CrossRef] [PubMed]
- Fowles, D.; Coles, M.; Donchin, E.; Porges, S. The eccrine system and electrodermal activity. In Psychophysiology; Guildford Press: New York, NY, USA, 1985; pp. 51–96. [Google Scholar]
- Lee, Y.D.; Chung, W.Y. Wireless sensor network based wearable smart shirt for ubiquitous health and activity monitoring. Sens. Actuators B Chem. 2009, 140, 390–395. [Google Scholar] [CrossRef]
- Li, H.; Yang, H.; Li, E.; Liu, Z.; Wei, K. Wearable sensors in intelligent clothing for measuring human body temperature based on optical fiber Bragg grating. Opt. Express 2012, 20, 11740–11752. [Google Scholar] [CrossRef] [PubMed]
- Lofhede, J.; Seoane, F.; Thordstein, M. Textile electrodes for EEG recording—A pilot study. Sensors 2012, 12, 16907–16919. [Google Scholar] [CrossRef] [PubMed]
- Lopez, G.; Custodio, V.; Ignacio Moreno, J. LOBIN: E-textile and wireless-sensor-network-based platform for healthcare monitoring in future hospital environments. IEEE Trans. Inf. Technol. Biomed. 2010, 14, 1446–1458. [Google Scholar] [CrossRef] [PubMed]
- Lorussi, F.; Galatolo, S.; Bartalesi, R. Modeling and characterization of extensible wearable textile-based electrogoniometers. IEEE Sens. J. 2013, 13, 217–228. [Google Scholar] [CrossRef]
- Marquez, J.C.; Seoane, F.; Lindecrantz, K. Textrode functional straps for bioimpedance measurements -experimental results for body composition analysis. Eur. J. Clin. Nutr. 2013, 67 (Suppl. 1), S22–S27. [Google Scholar] [CrossRef]
- Preece, S.J.; Kenney, L.P.; Major, M.J.; M.J., T.; Lay, E.; Fernandes, B.T. Automatic identification of gait events using an instrumented sock. J. NeuroEng. Rehabil. 2011, 8, 32. [Google Scholar]
- Di Rienzo, M.; Rizzo, F.; Meriggi, P.; Castiglioni, P.; Mazzoleni, P.; Parati, G.; Bordoni, B.; Brambilla, G.; Ferratini, M. MagIC system. IEEE Eng. Med. Biol. Mag. 2009, 28, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, A.; Cuny, L.; Hertleer, C.; Ghekiere, F.; Kazani, I.; de Clercq, G.; de Mey, G.; van Langenhove, L.A. Electrical circuit model of elastic and conductive yarns produced by hollow spindle spinning. Mater. Technol. 2011, 26, 121–127. [Google Scholar] [CrossRef]
- Shu, L.; Hua, T.; Wang, Y.; Li, Q.; Feng, D.D.; Tao, X. In-shoe plantar pressure measurement and analysis system based on fabric pressure sensing array. IEEE Trans. Inf. Technol. Biomed. 2010, 14, 767–775. [Google Scholar] [CrossRef] [PubMed]
- Song, H.Y.; Lee, J.H.; Kang, D.; Cho, H.; Cho, H.S.; Lee, J.W.; Lee, Y.J. Textile electrodes of jacquard woven fabrics for biosignal measurement. J. Textile Inst. 2010, 101, 758–770. [Google Scholar] [CrossRef]
- Tormene, P.; Bartolo, M.; De Nunzio, A.M.; Fecchio, F.; Quaglini, S.; Tassorelli, C.; Sandrini, G. Estimation of human trunk movements by wearable strain sensors and improvement of sensor’s placement on intelligent biomedical clothes. Biomed. Eng. Online 2012, 11, 95. [Google Scholar] [CrossRef] [PubMed]
- Vuorela, T.; Seppa, V.P.; Vanhala, J.; Hyttinen, J. Design and implementation of a portable long-term physiological signal recorder. IEEE Trans. Inf. Technol. Biomed. 2010, 14, 718–725. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Hayamizu, Y.; Yamamoto, Y.; Yomogida, Y.; Izadi-Najafabadi, A.; Futaba, D.N.; Hata, K. A stretchable carbon nanotube strain sensor for human-motion detection. Nat. Nanotechnol. 2011, 6, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.B.; Shen, Y.H.; Wang, W.D.; Wang, B.Q.; Zheng, J.W. Design and implementation of sensing shirt for ambulatory cardiopulmonary monitoring. J. Med. Biol. Eng. 2011, 31, 207–216. [Google Scholar] [CrossRef]
- Zheng, N.; Wu, Z.; Lin, M.; Yang, L.T. Enhancing battery efficiency for pervasive health-monitoring systems based on electronic textiles. IEEE Trans. Inf. Technol. Biomed. 2010, 14, 350–359. [Google Scholar] [CrossRef] [PubMed]
- Zysset, C.; Kinkeldei, T.; Münzenrieder, N.; Petti, L.; Salvatore, G.; Tröster, G. Combining electronics on flexible plastic strips with textiles. Textile Res. J. 2013, 83, 1130–1142. [Google Scholar]
- Mirvis, D.; Goldberger, A. Electrocardiography. In Braunwald’s Heart Disease, 10th ed.; Saunders Elsevier: Philadelphia, PA, USA, 2015; pp. 114–154. [Google Scholar]
- Camm, A.; Malik, M.; Bigger, J.; Breithardt, G.; Cerutti, S.; Cohen, R.; Coumel, P.; Fallen, E.; Kennedy, H.; Kleiger, R.; et al. Heart rate variability: standards of measurement, physiological interpretation and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Circulation 1996, 93, 1043–1065. [Google Scholar]
- Neumann, M. Biopotential electrodes. In Clinical Engineering; David, Y., von Maltzahn, W., Neuman, M., Bronzino, J., Eds.; CRC Press: Boca Raton, FL, USA, 2003; pp. 10:1–10:12. [Google Scholar]
- Webster, J. Medical Instrumentation: Application and Design, 4th ed.; Wiley: Hoboken, NJ, USA, 2010. [Google Scholar]
- Su, L.; Borov, S.; Zrenner, B. 12-lead Holter electrocardiography: Review of the literature and clinical application update. Herzschrittmachertherapie Elektrophysiol. 2013, 24, 92–96. [Google Scholar] [CrossRef]
- Acharya, U.R.; Joseph, K.P.; Kannathal, N.; Lim, C.M.; Suri, J.S. Heart rate variability: A review. Med. Biol. Eng. Comput. 2006, 44, 1031–1051. [Google Scholar] [CrossRef] [PubMed]
- Appelhans, B.; Luecken, L. Heart rate variability as an index of regulated emotional responding. Rev. General Psychol. 2006, 10, 229–240. [Google Scholar] [CrossRef]
- Coyle, M. Ambulatory cardiopulmonary data capture. In Proceedings of the 2nd Annual International IEEE-EMBS Special Topic Conference on Microtechnologies in Medicine & Biology, Madison, WI, USA, 2–4 May 2002; pp. 297–300.
- Coosemans, J.; Hermans, B.; Puers, R. Integrating wireless ECG monitoring in textiles. Sens. Actuators A 2006, 130–131, 48–53. [Google Scholar]
- Lim, Y.; Kim, K.; Park, K. ECG recording on a bed during sleep without direct skin-contact. IEEE Trans. Biomed. Eng. 2007, 54, 718–725. [Google Scholar] [CrossRef] [PubMed]
- Grimnes, S.; Martinsen, O. Bioimpedance Biolectricity Basics, 2nd ed.; Academic: London, UK, 2008. [Google Scholar]
- Kyle, U.; Bosaeus, I.; Lorenzo, A.D.; Deurenberg, P.; Elia, M.; Gomez, J.; Heitmann, B.; Kent-Smith, L.; Melchoir, J.; Pirlich, M.; et al. Bioelectrical impedance analysis—Part I: Review of principles and methods. Clin. Nutr. 2004, 23, 1226–1243. [Google Scholar]
- Kushner, R. Bioelectrical impedance analysis: A review of principles and applications. J. Am. Coll. Nutr. 1992, 11, 199–209. [Google Scholar] [PubMed]
- Togawa, T.; Tamura, T.; Oberg, P.A. Biomedical Transducers and Instruments; CRC Press: Boca Raton, FL, USA, 1997. [Google Scholar]
- Cornish, B.; Jacobs, A.; Thomas, B.; Ward, L. Optimizing electrode sites for segmental bioimpedance measurements. Physiol. Meas. 1999, 20, 241–250. [Google Scholar] [CrossRef] [PubMed]
- Lescolarde, L.; Yanguas, J.; Lukaski, H.; Alomar, X.; Rosell-Ferrer, J.; Rodas, G. Localized bioimpedance to assess muscle injury. Physiol. Meas. 2013, 34, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Kushner, R.F.; Schoeller, D.A. Estimation of total body water by bioelectrical impedance analysis. Am. J. Clin. Nutr. 1986, 44, 417–424. [Google Scholar] [PubMed]
- Agarwal, R.; Yadav, R.; Anand, S.; Suri, J.C.; Girija, J. Electrical impedance plethysmography technique in estimating pulmonary function status. J. Med. Eng. Technol. 2007, 31, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Azar, R.; al-Moubarak, I.; Barsumau, J.; Smessaert, C.; Vairon, M.X. Assessment and follow-up of nutritional status in hemodialysis patients. Néphrol. Thér. 2009, 5, S317–S322. [Google Scholar] [CrossRef] [PubMed]
- Medrano, G.; Eitner, F.; Floege, J.; Leonhardt, S. A novel bioimpedance technique to monitor fluid volume state during hemodialysis treatment. ASAIO J. 2010, 56, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Sjedic, E.; Fu, Y.; Pak, A.; Fairley, J.; Chau, T. The effects of rhythmic sensory cues on the temporal dynamics of human gait. PLoS One 2012, 7, e43104. [Google Scholar] [CrossRef] [PubMed]
- Masani, K.; Vette, A.; Popovic, M. Controlling balance during quiet standing: proportional and derivative controller generates preceding motor command to body sway position observed in experiments. Gait Posture 2006, 23, 164–172. [Google Scholar] [CrossRef] [PubMed]
- Lesperance, A.; Blain, S.; Chau, T. An integrated approach to detecting communicative intent amid hyperkinetic movements in children. Augment. Altern. Commun. 2011, 27, 150–162. [Google Scholar] [CrossRef] [PubMed]
- Tong, L.; Song, Q.; Ge, Y.; Liu, M. HMM-based human fall detection and prediction method using tri-axial accelerometer. IEEE Sens. J. 2013, 13, 1848–1856. [Google Scholar]
- Gibbs, P.T.; Asada, H.H. Wearable conductive fiber sensors for multi-axis human joint angle measurements. J. NeuroEng. Rehabil. 2005, 2, 18. [Google Scholar] [CrossRef] [PubMed]
- Edmison, J.; Jones, M.; Lockhart, T.; Martin, T. An e-textile system for motion analysis. Stud. Health Technol. Inform. 2004, 108, 292–301. [Google Scholar] [PubMed]
- Dunne, L.E.; Walsh, P.; Hermann, S.; Smyth, B.; Caulfield, B. Wearable monitoring of seated spinal posture. IEEE Trans. Biomed. Circuits Syst. 2008, 2, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Kuang, K.; Cantwell, W.J. An evaluation of a novel plastic optical fibre sensor for axial strain and bend measurements. Meas. Sci. Technol. 2002, 13, 1523. [Google Scholar] [CrossRef]
- Lorussi, F.; Galatolo, S.; de Rossi, D.E. Textile-based electrogoniometers for wearable posture and gesture capture systems. IEEE Sens. J. 2009, 9, 1014–1024. [Google Scholar] [CrossRef]
- Hall, M.A.; Holmes, G. Benchmarking attribute selection techniques for discrete class data mining. IEEE Trans. Knowl. Data Eng. 2003, 15, 1437–1447. [Google Scholar] [CrossRef]
- Gioberto, G.; Dunne, L. Theory and characterization of a top-thread coverstitched stretch sensor. In Proceedings of the IEEE International Conference on Systems, Man and Cybernetics, Seoul, Korea, 14–17 October 2012; pp. 3275–3280.
- Sund-Levander, M.; Forsberg, C.; Wahren, L. Normal oral, rectal, tympanic and axillary body temperature in adult men and women: A systematic literature review. Scand. J. Caring Sci. 2002, 16, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Charkoudian, N. Skin blood flow in adult human thermoregulation: How it works, when it does not and why. Mayo Clin. Proc. 2003, 78, 603–612. [Google Scholar] [CrossRef] [PubMed]
- Oberle, J.; Elam, M.; Karlsson, T.; Wallin, B. Temperature-dependent interaction between vasoconstrictor and vasodilator mechanisms in human skin. Acta Physiol. Scand. 1988, 132, 459–469. [Google Scholar] [CrossRef] [PubMed]
- Sund-Levander, M.; Grodzinsky, E. Assessment of body temperature measurement options. Br. J. Nurs. 2013, 22, 944–950. [Google Scholar] [CrossRef]
- Nhan, B.; Chau, T. Classifying affective states using thermal infrared imaging of the human face. IEEE Trans. Biomed. Eng. 2010, 57, 979–987. [Google Scholar] [CrossRef] [PubMed]
- Allegaert, K.; Casteels, K.; van Gorp, I.; Bogaert, G. Tympanic, infrared skin and temporal artery scan thermometers compared with rectal measurement in children: A real-life assessment. Curr. Ther. Res. 2014, 76, 34–38. [Google Scholar] [CrossRef] [PubMed]
- Keefe, F.; Gardner, E. Learned control of skin temperature: Effects of short- and long-term biofeedback training. Behav. Ther. 1979, 10, 202–210. [Google Scholar] [CrossRef]
- Blain, S.; Mihailidis, A.; Chau, T. Assessing the potential of electrodermal activity as an alternative access pathway. Med. Eng. Phys. 2008, 30, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Boccaletti, C.; Castrica, F.; Fabbri, G.; Santello, M. A non-invasive biopotential electrode for the correct detection of bioelectric currents. In Proceedings of the Sixth IASTED International Conference on Biomedical Engineering, Innsbruck, Austria, 13–15 February 2008; pp. 353–358.
- Blain, S.; Chau, T.; Mihailidis, A. Peripheral autonomic signals as access pathways for individuals with severe disabilities: A literature appraisal. Open Rehabil. J. 2008, 1, 27–37. [Google Scholar] [CrossRef]
- Kushki, A.; Fairley, J.; Merja, S.; King, G.; Chau, T. Comparison of blood volume pulse and skin conductance responses to mental and affective stimuli at different anatomical sites. Physiol. Meas. 2011, 32, 1529–1539. [Google Scholar] [CrossRef] [PubMed]
- Leung, B.; Chau, T. Autonomic responses to correct outcomes and interaction errors during single-switch scanning among children with severe spastic quadriplegic cerebral palsy. J. NeuroEng. Rehabil. 2014, 11, 13. [Google Scholar] [CrossRef] [PubMed]
- Falk, T.; Guirgis, M.; Power, S.; Chau, T. Taking NIRS-BCI outside the lab: towards achieving robustness against environmental noise. IEEE Trans. Neural Syst. Rehabil. Eng. 2010, 19, 136–146. [Google Scholar] [CrossRef] [PubMed]
- Kushki, A.; Drumm, E.; Mobarak, M.P.; Tanel, N.; Dupuis, A.; Chau, T.; Anagnostou, E. Investigating the autonomic nervous system response to anxiety in children with autism spectrum disorders. PLoS One 2013, 8, e59730. [Google Scholar] [CrossRef] [PubMed]
- Grangeon, M.; Charvier, K.; Guillot, A.; Rode, G.; Collet, C. Using sympathetic skin responses in individuals with spinal cord injury as a quantitative evaluation of motor imagery abilities. Phys. Ther. 2012, 92, 831–840. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Lee, B.; Lee, M. Wearable sensor glove based on conducting fabric using electrodermal activity and pulse-wave sensors for e-health application. Telemed. J. E-Health 2010, 16, 209–217. [Google Scholar] [CrossRef] [PubMed]
- Curone, D.; Secco, E.L.; Tognetti, A.; Loriga, G.; Dudnik, G.; Risatti, M.; Whyte, R.; Bonfiglio, A.; Magenes, G. Smart garments for emergency operators: The ProeTEX project. IEEE Trans. Inf. Technol. Biomed. 2010, 14, 694–701. [Google Scholar] [CrossRef] [PubMed]
- Habetha, J. The MyHeart project-fighting cardiovascular diseases by prevention and early diagnosis. In Proceedings of the IEEE EMBS Annual International Conference, New York, NY, USA, 30 August–3 September 2006; pp. 6746–6749.
- Shumaker, S.; Ockene, J.; Riekert, K. The Handbook of Health Behavior Change, 3rd ed.; Springer Publishing Company: New York, NY, USA, 2009. [Google Scholar]
- Baber, C. Wearable computers: A human factors review. Int. J. Hum. Comput. Interact. 2001, 13, 123–145. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fleury, A.; Sugar, M.; Chau, T. E-textiles in Clinical Rehabilitation: A Scoping Review. Electronics 2015, 4, 173-203. https://doi.org/10.3390/electronics4010173
Fleury A, Sugar M, Chau T. E-textiles in Clinical Rehabilitation: A Scoping Review. Electronics. 2015; 4(1):173-203. https://doi.org/10.3390/electronics4010173
Chicago/Turabian StyleFleury, Amanda, Maddy Sugar, and Tom Chau. 2015. "E-textiles in Clinical Rehabilitation: A Scoping Review" Electronics 4, no. 1: 173-203. https://doi.org/10.3390/electronics4010173
APA StyleFleury, A., Sugar, M., & Chau, T. (2015). E-textiles in Clinical Rehabilitation: A Scoping Review. Electronics, 4(1), 173-203. https://doi.org/10.3390/electronics4010173





