Fine-Tuned Aggregation Control in Perylene Diimide-Based Organic Solar Cells via a Mixed-Acceptor Strategy Using Planar and Twisted Acceptors
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Fabrication of Devices
2.3. Characterizations
3. Results and Discussion
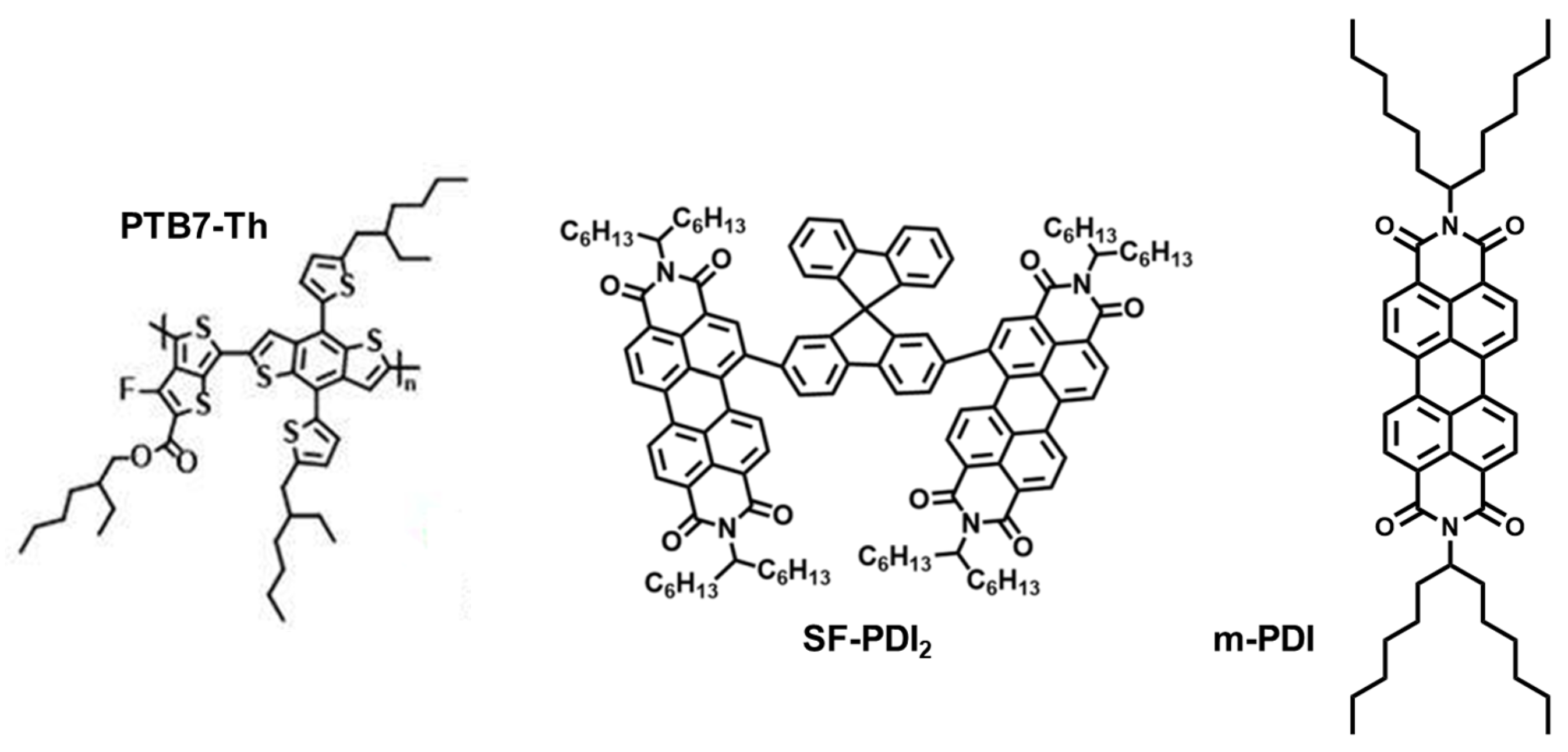
3.1. Molecular Structure of PDI Acceptors
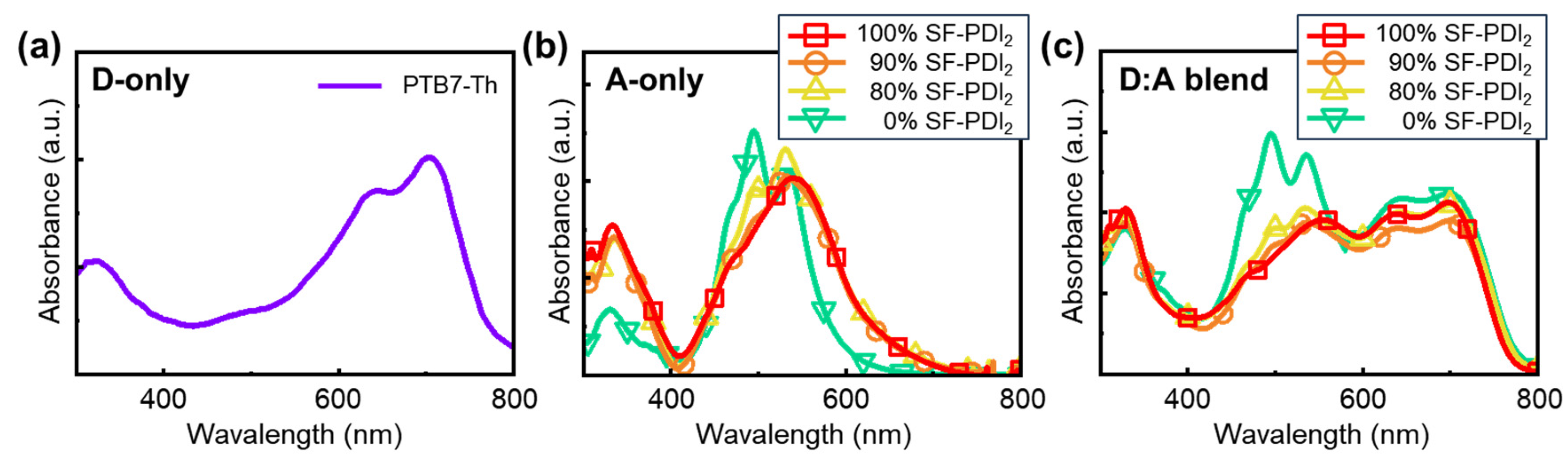
3.2. Aggregation Behavior of Mixed-Acceptor Systems
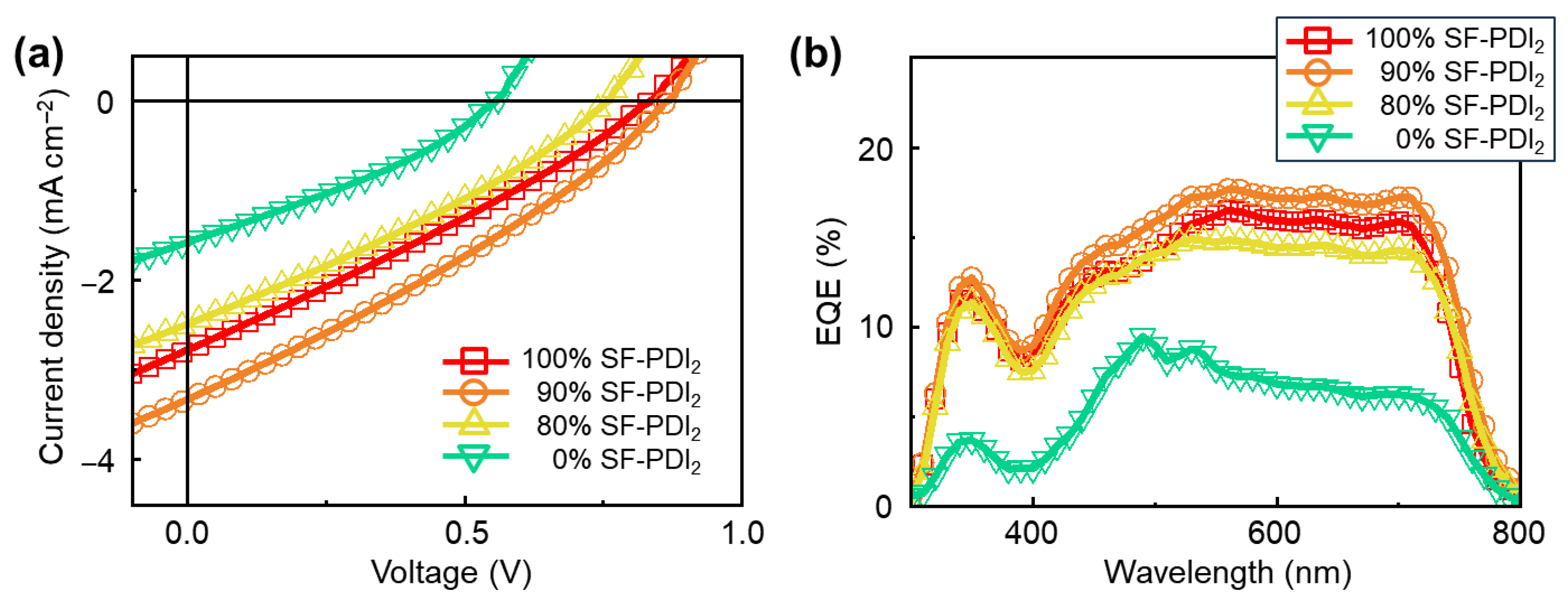
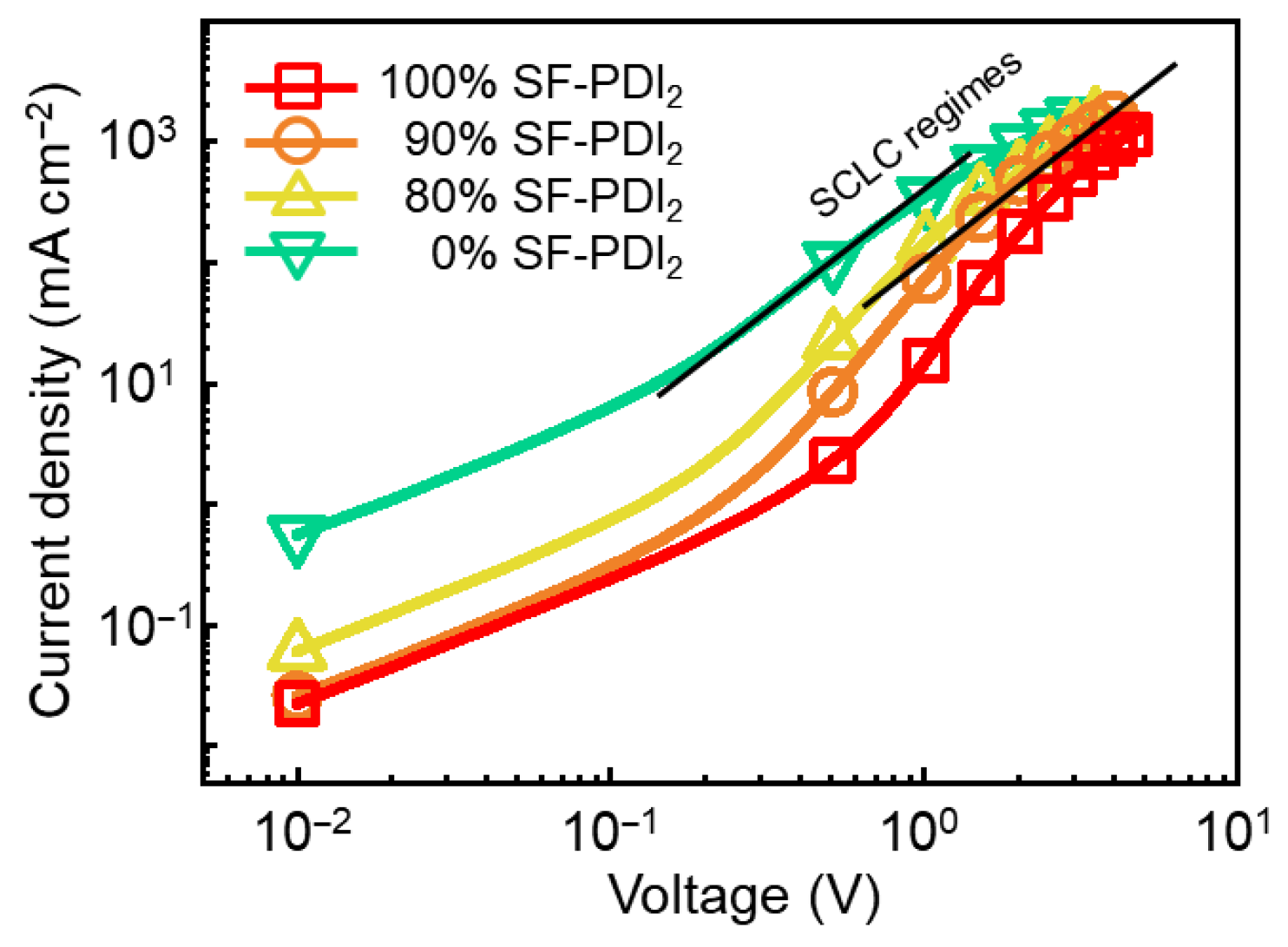
3.3. Photovoltaic Performance
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Zhang, X.; Lu, Z.; Ye, L.; Zhan, C.; Hou, J.; Zhang, S.; Jiang, B.; Zhao, Y.; Huang, J.; Zhang, S.; et al. A Potential Perylene Diimide Dimer-Based Acceptor Material for Highly Efficient Solution-Processed Non-Fullerene Organic Solar Cells with 4.03% Efficiency. Adv. Mater. 2013, 25, 5791–5797. [Google Scholar] [CrossRef]
- Lee, H.; Park, C.; Sin, D.H.; Park, J.H.; Cho, K. Recent Advances in Morphology Optimization for Organic Photovoltaics. Adv. Mater. 2018, 30, 1800453. [Google Scholar] [CrossRef]
- Cusumano, P.; Arnone, C.; Giambra, M.A.; Parisi, A. Donor/Acceptor Heterojunction Organic Solar Cells. Electronics 2020, 9, 70. [Google Scholar] [CrossRef]
- Kesava, S.V.; Fei, Z.; Rimshaw, A.D.; Wang, C.; Hexemer, A.; Asbury, J.B.; Heeney, M.; Gomez, E.D. Domain Compositions and Fullerene Aggregation Govern Charge Photogeneration in Polymer/Fullerene Solar Cells. Adv. Energy Mater. 2014, 4, 1400116. [Google Scholar] [PubMed]
- Schubert, M.; Dolfen, D.; Frisch, J.; Roland, S.; Steyrleuthner, R.; Stiller, B.; Chen, Z.; Scherf, U.; Koch, N.; Facchetti, A.; et al. Influence of Aggregation on the Performance of All-Polymer Solar Cells Containing Low-Bandgap Naphthalenediimide Copolymers. Adv. Energy Mater. 2012, 2, 369–380. [Google Scholar] [CrossRef]
- Kwon, O.K.; Park, J.H.; Park, S.K.; Park, S.Y. Soluble Dicyanodistyrylbenzene-Based Non-Fullerene Electron Acceptors with Optimized Aggregation Behavior for High-Efficiency Organic Solar Cells. Adv. Energy Mater. 2014, 5, 1400929. [Google Scholar] [CrossRef]
- Ye, L.; Sun, K.; Jiang, W.; Zhang, S.; Zhao, W.; Yao, H.; Wang, Z.; Hou, J. Enhanced Efficiency in Fullerene-Free Polymer Solar Cell by Incorporating Fine-Designed Donor and Acceptor Materials. ACS Appl. Mater. Interfaces 2015, 7, 9274–9280. [Google Scholar] [CrossRef]
- Yin, H.; Ma, L.K.; Wang, Y.; Huang, J.; Yu, H.; Zhang, J.; Chow, P.C.Y.; Ma, W.; So, S.K.; Yan, H. Donor Polymer Can Assist Electron Transport in Bulk Heterojunction Blends with Small Energetic Offsets. Adv. Mater. 2019, 31, e1903998. [Google Scholar] [CrossRef]
- Akash; Tiwari, J.P. Recent Advancements in Perylene Diimide as an Electron Acceptor in Organic Solar Cells. J. Mater. Chem. C 2024, 12, 838–853. [Google Scholar] [CrossRef]
- Eynaud, Q.; Koganezawa, T.; Sekimoto, H.; Kramdi, M.E.A.; Quéléver, G.; Margeat, O.; Ackermann, J.; Yoshimoto, N.; Videlot-Ackermann, C. Ternary Polymer Solar Cells: Impact of Non-Fullerene Acceptors on Optical and Morphological Properties. Electronics 2024, 13, 1752. [Google Scholar] [CrossRef]
- Cooling, N.A.; Feron, K.; Jones, T.W.; Belcher, W.J.; Dastoor, P.C. Controlling Charge Generation in Organic Photovoltaic Ternary Blends: How Trace Ternary Additives Determine Mechanism. Electronics 2025, 14, 1655. [Google Scholar] [CrossRef]
- Xu, W.; Tian, H.; Ni, Y.; Xu, Y.; Zhang, L.; Zhang, F.; Wu, S.; Young Jeong, S.; Huang, T.; Du, X.; et al. Eco-Friendly Solvent-Processed Layer-by-Layer Ternary All-Polymer Solar Cells Exhibiting over 18.5% Efficiency. Chem. Eng. J. 2024, 493, 152558. [Google Scholar] [CrossRef]
- Zhang, C.; Sun, S.; Han, T.; Chi, J.; Dong, R.; Han, S.; Zhou, H.; Xu, Y.; Cai, L.; Du, X.; et al. Superior Charge Dynamics Via Ternary Doping Layer-by-Layer Strategy in High-Efficiency Organic Solar Cells. Chem. Eng. J. 2025, 522, 167959. [Google Scholar] [CrossRef]
- Zhong, Y.; Trinh, M.T.; Chen, R.; Wang, W.; Khlyabich, P.P.; Kumar, B.; Xu, Q.; Nam, C.Y.; Sfeir, M.Y.; Black, C.; et al. Efficient Organic Solar Cells with Helical Perylene Diimide Electron Acceptors. J. Am. Chem. Soc. 2014, 136, 15215–15221. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.H.; Chueh, C.C.; Xi, Y.Y.; Zhong, H.L.; Gao, G.P.; Wang, Z.H.; Pozzo, L.D.; Wen, T.C.; Jen, A.K.Y. Influence of Molecular Geometry of Perylene Diimide Dimers and Polymers on Bulk Heterojunction Morphology toward High-Performance Nonfullerene Polymer Solar Cells. Adv. Funct. Mater. 2015, 25, 5326–5332. [Google Scholar] [CrossRef]
- Lee, J.; Singh, R.; Sin, D.H.; Kim, H.G.; Song, K.C.; Cho, K. A Nonfullerene Small Molecule Acceptor with 3d Interlocking Geometry Enabling Efficient Organic Solar Cells. Adv. Mater. 2016, 28, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Yang, S. Progress in Perylene Diimides for Organic Solar Cell Applications. RSC Adv. 2022, 12, 6966–6973. [Google Scholar] [CrossRef] [PubMed]
- Cheng, P.; Zhao, X.; Zhan, X. Perylene Diimide-Based Oligomers and Polymers for Organic Optoelectronics. Acc. Mater. Res. 2022, 3, 309–318. [Google Scholar] [CrossRef]
- Sugie, A.; Han, W.; Shioya, N.; Hasegawa, T.; Yoshida, H. Structure-Dependent Electron Affinities of Perylene Diimide-Based Acceptors. J. Phys. Chem. C 2020, 124, 9765–9773. [Google Scholar] [CrossRef]
- Sun, J.-P.; Hendsbee, A.D.; Dobson, A.J.; Welch, G.C.; Hill, I.G. Perylene Diimide Based All Small-Molecule Organic Solar Cells: Impact of Branched-Alkyl Side Chains on Solubility, Photophysics, Self-Assembly, and Photovoltaic Parameters. Org. Electron. 2016, 35, 151–157. [Google Scholar] [CrossRef]
- Qu, J.; Mu, Z.; Lai, H.; Xie, M.; Liu, L.; Lu, W.; Chen, W.; He, F. Effect of the Molecular Configuration of Perylene Diimide Acceptors on Charge Transfer and Device Performance. ACS Appl. Energy Mater. 2018, 1, 833–840. [Google Scholar] [CrossRef]
- Singh, R.; Suranagi, S.R.; Lee, J.; Lee, H.; Kim, M.; Cho, K. Unraveling the Efficiency-Limiting Morphological Issues of the Perylene Diimide-Based Non-Fullerene Organic Solar Cells. Sci. Rep. 2018, 8, 2849. [Google Scholar] [CrossRef]
- Lin, Y.; Wang, Y.; Wang, J.; Hou, J.; Li, Y.; Zhu, D.; Zhan, X. A Star-Shaped Perylene Diimide Electron Acceptor for High-Performance Organic Solar Cells. Adv. Mater. 2014, 26, 5137–5142. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Wu, Q.; Cai, Z.; Zheng, T.; Chen, W.; Lu, J.; Yu, L. Electron Acceptors Based on A-Substituted Perylene Diimide (Pdi) for Organic Solar Cells. Chem. Mater. 2016, 28, 1139–1146. [Google Scholar] [CrossRef]
- Liu, Z.; Zhang, L.; Shao, M.; Wu, Y.; Zeng, D.; Cai, X.; Duan, J.; Zhang, X.; Gao, X. Fine-Tuning the Quasi-3d Geometry: Enabling Efficient Nonfullerene Organic Solar Cells Based on Perylene Diimides. ACS Appl. Mater. Interfaces 2018, 10, 762–768. [Google Scholar] [CrossRef]
- Wu, M.; Yi, J.P.; Chen, L.; He, G.; Chen, F.; Sfeir, M.Y.; Xia, J. Novel Star-Shaped Helical Perylene Diimide Electron Acceptors for Efficient Additive-Free Nonfullerene Organic Solar Cells. ACS Appl. Mater. Interfaces 2018, 10, 27894–27901. [Google Scholar] [CrossRef]
- Luo, Z.; Liu, T.; Chen, Z.; Xiao, Y.; Zhang, G.; Huo, L.; Zhong, C.; Lu, X.; Yan, H.; Sun, Y.; et al. Isomerization of Perylene Diimide Based Acceptors Enabling High-Performance Nonfullerene Organic Solar Cells with Excellent Fill Factor. Adv. Sci. 2019, 6, 1802065. [Google Scholar] [CrossRef] [PubMed]
- Ding, K.; Wang, Y.; Shan, T.; Xu, J.; Bao, Q.; Liu, F.; Zhong, H. Propeller-Like Acceptors with Difluoride Perylene Diimides for Organic Solar Cells. Org. Electron. 2020, 78, 105569. [Google Scholar] [CrossRef]
- Liu, W.; Zhang, C.; Liu, J.; Bo, Z. Pdi-Based Hexapod-Shaped Nonfullerene Acceptors for the High-Performance as-Cast Organic Solar Cells. ACS Appl. Mater. Interfaces 2020, 12, 37409–37417. [Google Scholar] [CrossRef]
- Li, Z.; Li, D.; Bao, Y.; Guo, P.; Liang, Z.; Yang, C.; Tong, J. Acceptor-Acceptor Type Perylenediimide-Based Polymeric Acceptors with Large 2-Decyltetradecyl Flexible Side Chain: Synthesis and Impact of Fluorination. Opt. Mater. 2025, 162, 116932. [Google Scholar] [CrossRef]
- Song, K.C.; Singh, R.; Lee, J.; Sin, D.H.; Lee, H.; Cho, K. Propeller-Shaped Small Molecule Acceptors Containing a 9,9′-Spirobifluorene Core with Imide-Linked Perylene Diimides for Non-Fullerene Organic Solar Cells. J. Mater. Chem. C 2016, 4, 10610–10615. [Google Scholar] [CrossRef]
- Singh, R.; Lee, J.; Kim, M.; Keivanidis, P.E.; Cho, K. Control of the Molecular Geometry and Nanoscale Morphology in Perylene Diimide Based Bulk Heterojunctions Enables an Efficient Non-Fullerene Organic Solar Cell. J. Mater. Chem. A 2017, 5, 210–220. [Google Scholar] [CrossRef]
- Chen, R.; Wang, H.; Yuan, S.; Jia, T.; Luo, Y.; Lin, T.; Xu, Y.; Li, Q.; Li, Y.; Xiao, B.; et al. Modulated Aggregation on Terpolymer Towards 19.2% Green Solvent Processed Binary Organic Solar Cells. Nano Energy 2025, 134, 110576. [Google Scholar] [CrossRef]
- Weng, K.; Li, C.; Bi, P.; Ryu, H.S.; Guo, Y.; Hao, X.; Zhao, D.; Li, W.; Woo, H.Y.; Sun, Y. Ternary Organic Solar Cells Based on Two Compatible Pdi-Based Acceptors with an Enhanced Power Conversion Efficiency. J. Mater. Chem. A 2019, 7, 3552–3557. [Google Scholar] [CrossRef]
- Lu, H.; Liu, J.; Liu, Y.; Xu, X.; Bo, Z. Improving the Efficiency of Organic Solar Cells by Introducing Perylene Diimide Derivative as Third Component and Individually Dissolving Donor/Acceptor. ChemSusChem 2021, 14, 5442–5449. [Google Scholar] [CrossRef]
- Liu, M.; Ge, X.; Jiang, X.; Chen, D.; Guo, F.; Gao, S.; Peng, Q.; Zhao, L.; Zhang, Y. 18% Efficiency of Ternary Organic Solar Cells Enabled by Integrating a Fused Perylene Diimide Guest Acceptor. Nano Energy 2023, 112, 108501. [Google Scholar] [CrossRef]
- Lee, H.; Lee, D.; Sin, D.H.; Kim, S.W.; Jeong, M.S.; Cho, K. Effect of Donor–Acceptor Molecular Orientation on Charge Photogeneration in Organic Solar Cells. npg Asia Mater. 2018, 10, 469–481. [Google Scholar] [CrossRef]
- Farahat, M.E.; Welch, G.C. N-Annulated Perylene Diimide Non-Fullerene Acceptors for Organic Photovoltaics. Colorants 2023, 2, 151–178. [Google Scholar] [CrossRef]
- An, Q.; Wang, J.; Gao, W.; Ma, X.; Hu, Z.; Gao, J.; Xu, C.; Hao, M.; Zhang, X.; Yang, C.; et al. Alloy-Like Ternary Polymer Solar Cells with over 17.2% Efficiency. Sci. Bull. 2020, 65, 538–545. [Google Scholar] [CrossRef]
- Naqvi, S.; Kumar, M.; Kumar, R. Facile Synthesis and Evaluation of Electron Transport and Photophysical Properties of Photoluminescent Pdi Derivatives. ACS Omega 2019, 4, 19735–19745. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.; Wang, Z.; Chen, Y.; Zhang, Q.; Zhang, J.; Zhu, L.; Zhang, M.; Cui, Z.; Wei, Z.; Ade, H.; et al. Revealing Aggregation of Non-Fullerene Acceptors in Intermixed Phase by Ultraviolet-Visible Absorption Spectroscopy. Cell Rep. Phys. Sci. 2022, 3, 100983. [Google Scholar] [CrossRef]
- Guo, C.; Fu, Y.; Li, D.; Wang, L.; Zhou, B.; Chen, C.; Zhou, J.; Sun, Y.; Gan, Z.; Liu, D.; et al. A Polycrystalline Polymer Donor as Pre-Aggregate toward Ordered Molecular Aggregation for 19.3% Efficiency Binary Organic Solar Cells. Adv. Mater. 2023, 35, e2304921. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhong, J.; Chung, S.; Bai, L.; Zhao, Z.; Tan, L.; Liu, Y.; Zhang, M.; Lyu, S.; Lee, J.; et al. Π-Conjugation-Driven Aggregation Enhances Charge Transport in Y6 Derivatives for Bilayer Organic Solar Cells with 20 % Efficiency. Chem. Eng. J. 2025, 522, 167311. [Google Scholar] [CrossRef]
- Montero, J.M.; Bisquert, J. Interpretation of Trap-Limited Mobility in Space-Charge Limited Current in Organic Layers with Exponential Density of Traps. J. Appl. Phys. 2011, 110, 043705. [Google Scholar] [CrossRef]
- Nicolai, H.T.; Mandoc, M.M.; Blom, P.W.M. Electron Traps in Semiconducting Polymers: Exponential Versus Gaussian Trap Distribution. Phys. Rev. B 2011, 83, 195204. [Google Scholar] [CrossRef]

| Device | JSC | VOC | FF | PCE |
|---|---|---|---|---|
| [mA cm−2] | [V] | [%] | ||
| 100% SF-PDI2 | 2.67 (±0.154) | 0.830 (±0.027) | 0.282 (±0.004) | 0.624 (±0.042) |
| 90% SF-PDI2 | 3.28 (±0.139) | 0.845 (±0.050) | 0.303 (±0.005) | 0.840 (±0.072) |
| 80% SF-PDI2 | 2.39 (±0.115) | 0.773 (±0.012) | 0.294 (±0.004) | 0.544 (±0.026) |
| 0% SF-PDI2 | 1.62 (±0.163) | 0.567 (±0.012) | 0.313 (±0.003) | 0.287 (±0.022) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Hwang, H.; Lee, H. Fine-Tuned Aggregation Control in Perylene Diimide-Based Organic Solar Cells via a Mixed-Acceptor Strategy Using Planar and Twisted Acceptors. Electronics 2026, 15, 1039. https://doi.org/10.3390/electronics15051039
Hwang H, Lee H. Fine-Tuned Aggregation Control in Perylene Diimide-Based Organic Solar Cells via a Mixed-Acceptor Strategy Using Planar and Twisted Acceptors. Electronics. 2026; 15(5):1039. https://doi.org/10.3390/electronics15051039
Chicago/Turabian StyleHwang, Hyeongjin, and Hansol Lee. 2026. "Fine-Tuned Aggregation Control in Perylene Diimide-Based Organic Solar Cells via a Mixed-Acceptor Strategy Using Planar and Twisted Acceptors" Electronics 15, no. 5: 1039. https://doi.org/10.3390/electronics15051039
APA StyleHwang, H., & Lee, H. (2026). Fine-Tuned Aggregation Control in Perylene Diimide-Based Organic Solar Cells via a Mixed-Acceptor Strategy Using Planar and Twisted Acceptors. Electronics, 15(5), 1039. https://doi.org/10.3390/electronics15051039







