1. Introduction
As global health systems increasingly emphasize preventive care and chronic disease management, the demand for accessible and scalable health monitoring tools has grown exponentially. Wearable computing is at the heart of this demand. Rapid advancement in sensor technologies, miniaturized electronics, and wireless communication has led to the proliferation of wearable computing systems designed for real-time health monitoring. Wearable computing has emerged as a transformative approach in healthcare, enabling continuous, unobtrusive monitoring of physiological and behavioral parameters. These systems, typically comprising smart sensors embedded in garments, wristbands, patches, or headgear, support real-time data collection relevant to both mental and physical health. Among the most promising developments is digital phenotyping [
1] an innovative health monitoring method that utilizes smart devices, sensors, and mobile apps to enable continuous, noninvasive, and personalized health evaluation outside traditional clinical settings [
2,
3,
4]. Having a capable processing platform and enabling persistent measurements of physiological and behavioral parameters, wearable health monitoring systems can provide critical insights into both physical conditions, such as cardiovascular health, respiratory function, and metabolic activity, and mental states, including stress, anxiety, and depression [
5,
6]. The integration of artificial intelligence (AI) and machine-learning (ML) algorithms further enhances the value of wearables by enabling real-time data analysis, anomaly detection, and personalized feedback.
In the context of mental health, wearables offer a unique opportunity to detect subtle behavioral signals that are often missed in episodic clinical visits. Metrics such as heart rate variability (HRV), electrodermal activity (EDA), activity levels, and sleep quality have been correlated with emotional and cognitive states [
7,
8]. On the physical health front, wearable technologies support the continuous monitoring of chronic conditions such as diabetes, hypertension, and cardiovascular disease, and facilitate timely intervention [
9,
10].
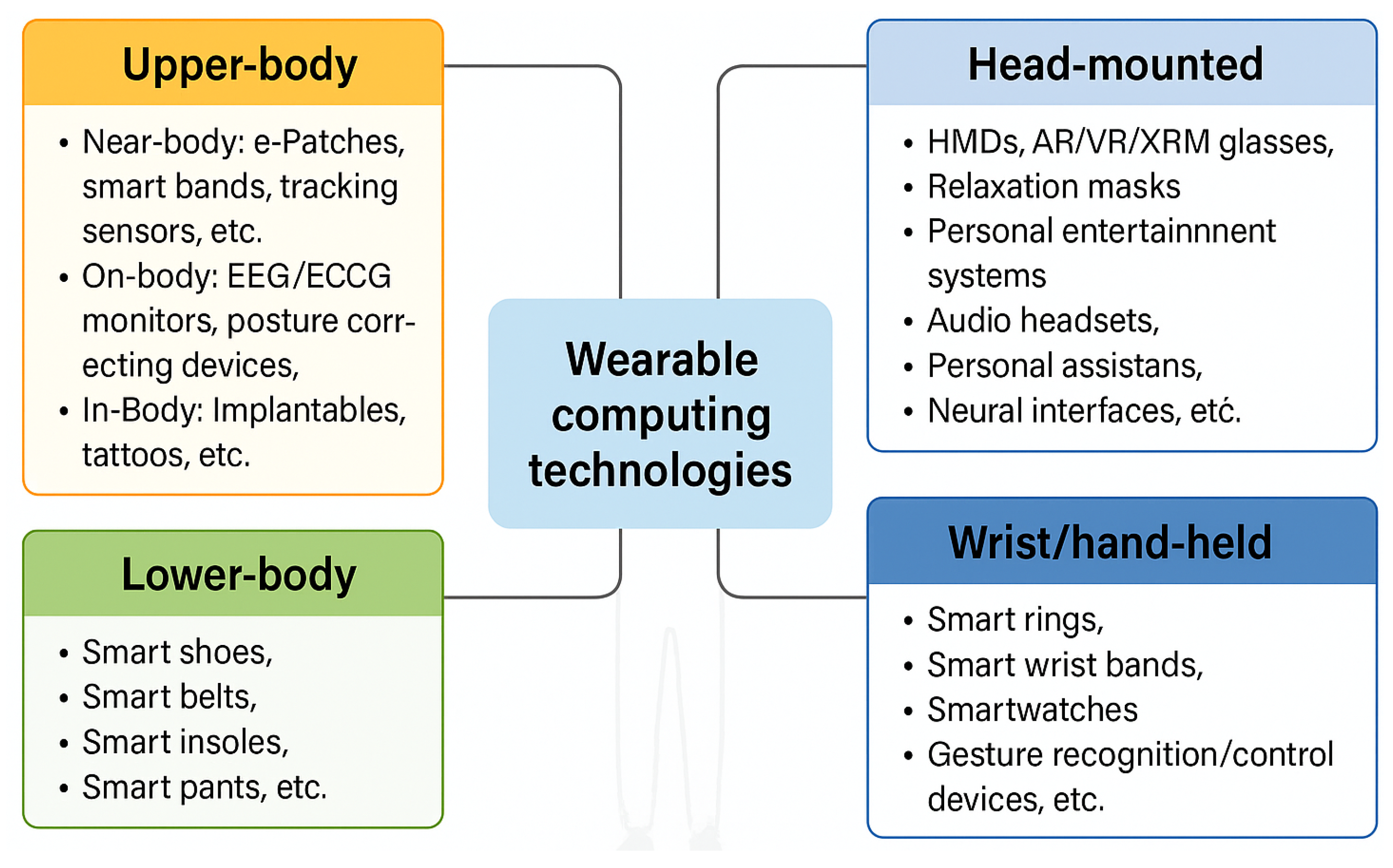
Figure 1 provides a structured overview of wearable computing technologies, organized by their placement on the human body. It categorizes devices into four main groups: head-mounted (e.g., AR/VR headsets, relaxation masks, audio headsets), upper body (e.g., smart clothes, posture monitors, safety devices, implantables), wrist/hand-held (e.g., smartwatches, smart rings, gesture control devices), and lower body (e.g., smart shoes, belts, insoles, pants).
To contextualize the novelty and breadth of our survey,
Table 1 presents a comparative analysis of recent review articles in the domain of wearable computing for monitoring mental and physical health. Each column represents an existing review, while the rows list key thematic and technical dimensions, including sensor types, artificial intelligence integration, wireless communication methods, high-performance energy-efficient edge computing, and future research directions. The presence or absence of coverage in each paper is indicated, enabling readers to quickly discern the relative scope and focus of prior works. This structured comparison highlights the comprehensive nature of our survey, which uniquely integrates mental and physical health perspectives with emerging topics such as federated learning, context-aware multimodal fusion, and edge-AI acceleration.
This paper is divided as follows:
Section 2 presents the methodology used for selecting the references and their sources.
Section 3 discusses the background and enabling technologies, including wearable sensors, communication protocols, and data modalities.
Section 4 presents a comparison between the four most common computational frameworks used.
Section 5 covers applications in physical health monitoring, such as cardiovascular assessment, activity tracking, chronic disease management, and sleep analysis.
Section 6 focuses on mental health applications, highlighting stress, mood, and cognitive state detection.
Section 7 outlines the machine-learning techniques used in wearable systems.
Section 8 reviews evaluation metrics and benchmarks, while
Section 9 addresses challenges and limitations.
Section 10 presents future research directions, followed by conclusions in
Section 11.
2. Paper Selection Methodology
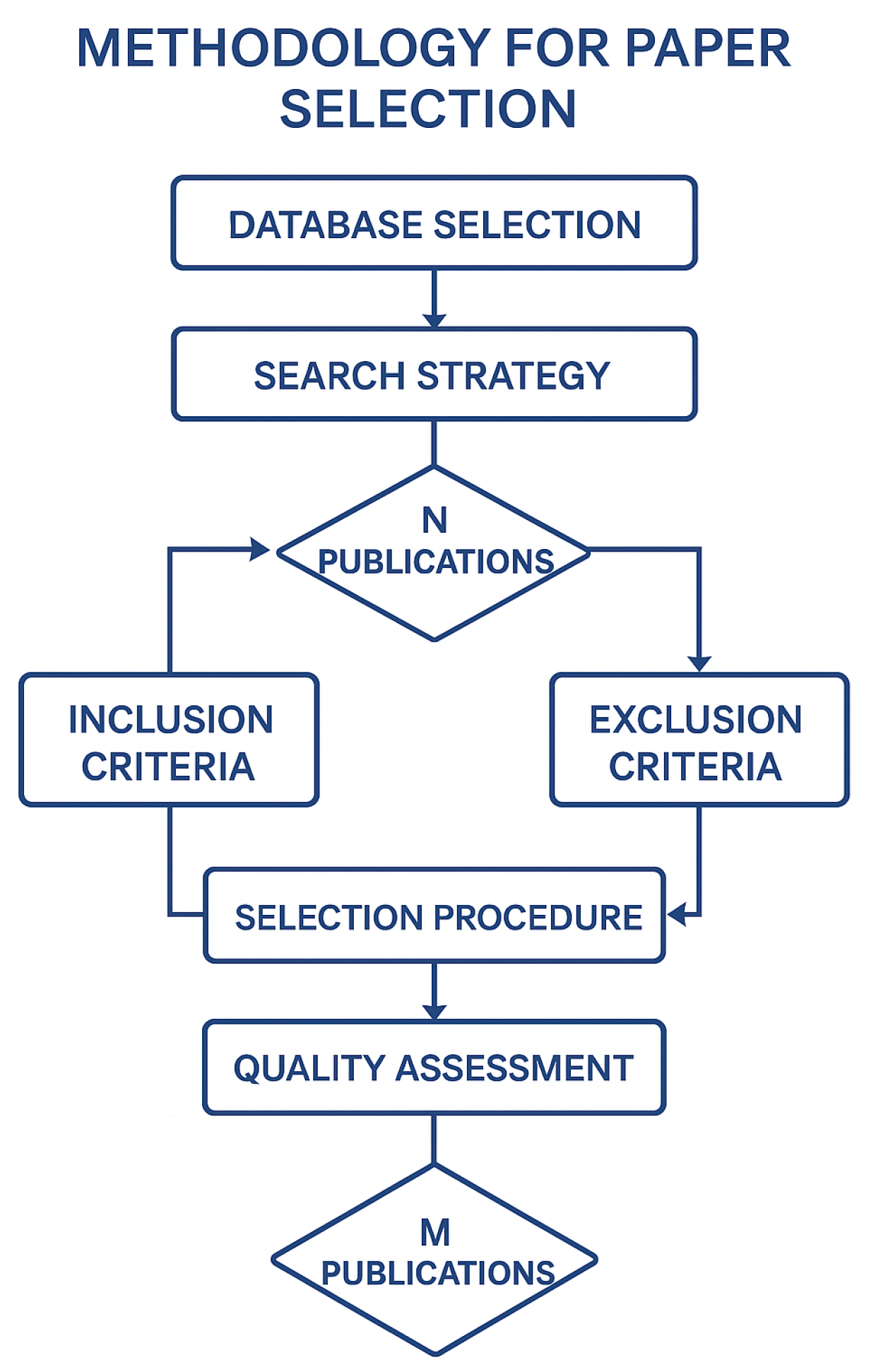
To ensure a comprehensive and objective literature review, a systematic paper selection methodology was adopted as shown in
Figure 2. The process involved the following steps:
Database Selection: We queried multiple reputable academic databases, including IEEE Xplore, MDPI, ACM Digital Library, SpringerLink, Elsevier ScienceDirect, and PubMed. Additionally, Google Scholar was used to capture relevant works not indexed in specialized repositories. Crossref had been used to obtain the DOI of some papers.
Search Strategy: A combination of controlled vocabulary and free-text search terms was employed, focusing on keywords such as “wearable computing”, “mental health monitoring”, “physical health monitoring”, “physiological sensors”, “context-aware systems”, “Wearable devices used for monitoring respiratory and pulmonary function”, “Wearable devices used for monitoring joint kinematics”, and “advancement in bipolar disorder management using wearable technologies”. Boolean operators and wildcard symbols were applied to capture variations of terms.
Inclusion Criteria: Peer-reviewed journal articles, conference proceedings, and high-impact surveys published between 2015 and 2025, 165 of them from 2021 and after. Studies that focus on hardware, software, or integrated systems relevant to wearable computing in health monitoring. Papers providing experimental results, prototype implementations, or systematic evaluations.
Exclusion Criteria: Non-peer-reviewed articles, opinion pieces, and patents. Studies focusing solely on unrelated wearable applications such as gaming or any application that does not have a health-monitoring component. Redundant publications with overlapping content from the same research group was also excluded.
Selection Procedure: The initial search returned more than 1000 publications. Titles and abstracts were screened for relevance, followed by a full-text review for the selected papers to confirm compliance with inclusion criteria.
Quality Assessment: Each paper was evaluated for methodological rigor, novelty, and contribution to the field. Priority was given to studies presenting measurable performance metrics (e.g., accuracy, power consumption, latency) or novel design approaches.
Final Dataset: After applying the above criteria, over 250 publications were included in the final review, representing a balanced coverage of both mental and physical health monitoring applications.
3. Background and Enabling Technologies
Wearable computing has undergone significant advancements in the past two decades, driven by developments in low-power electronics, miniaturized sensors, wireless communication, and embedded AI. These technologies have enabled the development of unobtrusive, continuous health monitoring systems capable of tracking both physical and mental health metrics in real time [
4,
19]. The core architecture of wearable systems typically integrates sensing, data acquisition, wireless communication as well as data types and their modalities.
3.1. Wearable Sensor Technologies
Wearable health monitoring systems are fundamentally enabled by a wide array of miniaturized sensors capable of capturing physiological and biomechanical signals in real time. These sensors are integral to both mental and physical health monitoring, as they provide objective, continuous, and non-invasive data collection essential for health assessment and intervention [
2,
4].
3.1.1. Physiological Sensors
Physiological sensors monitor internal body signals that are indicative of physical health status and mental well-being, see
Figure 3.
Electrocardiogram (ECG): ECG sensors measure the electrical activity of the heart. They are widely used for detecting cardiac anomalies such as arrhythmias, monitoring HRV, and analyzing stress-related responses [
6,
7].
Photoplethysmography (PPG): PPG sensors, traditionally used for heart rate (HR) and oxygen saturation (SpO
2) estimation, have recently evolved toward high-precision, low-power, and always-on architectures that overcome earlier limitations such as motion artifacts and limited penetration depth [
20]. Advances in emerging organic and flexible materials, photodetector designs [
21], and adaptive signal processing now enable continuous cardiovascular monitoring with reduced energy consumption, making PPG suitable for long-term wear in both clinical and consumer health contexts. These sensors are commonly embedded in smartwatches and wristbands [
8]. Charlton et al. [
22] present a road-map that outlines directions for research and development to realize the full potential of wearable photoplethysmography. It shows and propose many new sensor technologies such as using emerging and organic materials in sensors.
EDA: Also known as galvanic skin response (GSR), EDA sensors detect changes in skin conductance linked to sweat gland activity, which correlates with sympathetic nervous system arousal and is frequently used to infer stress, anxiety, and emotional states [
5].
Electroencephalogram (EEG): EEG sensors measure brain wave activity by measuring electrical potentials generated by synchronized neuronal firing, captured as voltage fluctuations on the scalp. These sensors are increasingly used in wearable form factors to assess mental states, cognitive workload, and emotional responses [
23].
Skin Temperature: These sensors provide insights into thermoregulation and hydration status. Changes in skin temperature can reflect fever, stress responses, or metabolic activity, while sweat analysis can yield biochemical markers for hydration and electrolyte balance [
24].
Microfluidic and Sweat Sensors: Microfluidic devices allow continuous, non-invasive collection and analysis of biofluids such as sweat, saliva, and interstitial fluid to detect biomarkers including glucose, lactate, cortisol, and electrolytes in real time. By integrating flexible microfluidic channels with low-power electronics and wireless communication, these systems can provide on-body biochemical sensing that complements physiological data from conventional wearable sensors [
25,
26], enabling more comprehensive health assessment and personalized interventions. Recent advances in stretchable substrates, capillary-driven flow, and on-chip multiplexed assays have further enhanced their suitability for long-term, unobtrusive monitoring in applications ranging from stress tracking to athletic performance optimization [
27]. Also recent trends in electro-microfluidic devices for wireless monitoring of biomarker levels unlocked new possibilities for personalized healthcare [
28,
29].
Table 2 presents a comparison between the different physiological sensors in terms of there application areas in health monitoring. It also shows the advantages and limitations of each sensor.
3.1.2. Motion and Activity Sensors
Motion sensors allow for monitoring of physical activity, posture, gait, and general mobility patterns, which are critical indicators for both physical health and mental state, see
Figure 4.
Table 3 presents a comparison between the different motion and activity sensors in terms of there measured parameters and applications. It also shows the advantages and limitations of each sensor. Together, physiological and motion sensors provide a holistic view of the user’s health, enabling accurate monitoring, early diagnosis, and personalized intervention strategies.
3.2. Communication and Processing
The effectiveness of wearable devices in mental and physical health monitoring hinges not only on the sensor modalities but also on the supporting communication and computational infrastructure. Seamless data transmission, real-time analytics, and system integration are vital for continuous, low-latency, and context-aware health assessment. This section outlines the key wireless technologies, edge/cloud processing paradigms, and architectural considerations for integrating wearable health systems [
2,
32,
33,
34].
3.2.1. Wireless Communication Technologies
Wearable devices commonly utilize short-range, low-power wireless communication protocols that are well-suited for body-area networks and personal health devices.
Bluetooth Low Energy (BLE): BLE is the most prevalent protocol for health wearables due to its low energy consumption and wide compatibility with smartphones. It supports intermittent data exchange, making it ideal for periodic health monitoring [
35].
Zigbee: Zigbee offers mesh networking capabilities and is used in scenarios requiring multiple wearables or sensor nodes in close proximity, such as home-based elder care systems.
Wi-Fi: For high-throughput and continuous streaming applications, Wi-Fi provides reliable communication over broader ranges but at a higher power cost.
Near Field Communication (NFC): NFC is used for passive data transfer or authentication purposes in secure health applications.
Long Range (LoRa): LoRa (Long Range) communication is increasingly used in wearable computing for mental and physical health monitoring due to its ability to transmit physiological data—such as heart rate, stress indicators, sleep metrics, and activity levels—over several kilometers while consuming minimal power. Compared to Bluetooth and Wi-Fi, LoRa supports scalable networks of wearables for applications like telehealth, elderly care, air pollution and chronic disease management, with devices often lasting months or years on a single battery [
36,
37,
38].
Cellular (4G/5G): These capabilities enable wearables to stream rich physiological and behavioral data—such as continuous ECG, EEG, stress indicators, and activity metrics—to cloud platforms in real time, supporting telemedicine [
39,
40], remote rehabilitation, and AI-driven health analytics [
41]. 5G’s ultra-reliable low-latency communication (URLLC) is particularly beneficial for critical health applications, including closed-loop systems for cardiac monitoring or neurofeedback, where immediate response is essential.
Ultra-Wideband (UWB): UWB’s centimeter-level accuracy enables wearables to track mobility patterns [
42], gait parameters, and fall detection with far greater reliability than Bluetooth or Wi-Fi [
43], which is crucial for elderly care and rehabilitation programs. Additionally, UWB-based wearables can support stress and mental health monitoring by contextualizing physiological data [
40] such as HRV and EDA—with movement and location insights [
44], facilitating more comprehensive behavioral analysis.
Table 4 presents a comparison between all the wireless communication technologies used in wearable devices used for mental and physical health monitoring.
3.2.2. Edge and Cloud Processing
Wearable systems are increasingly adopting hybrid architectures combining edge and cloud computing to balance processing load, latency, and privacy.
Edge Computing: Data are processed locally on the wearable or a nearby device, such as a smartphone or dedicated edge processor (e.g., Coral TPU). This allows real-time analytics, reduces latency, and mitigates the need for constant cloud connectivity [
32,
33,
34,
45].
Cloud Computing: Cloud servers are used for long-term storage, advanced modeling, and global pattern recognition. This is crucial for tracking chronic conditions, conducting cohort analyses, or enabling remote healthcare provider access.
Federated Learning and Privacy: Modern systems integrate federated learning to allow decentralized model training across edge devices without transferring raw health data, thereby preserving user privacy [
46,
47].
3.2.3. System Integration and Middleware
Wearable health platforms must integrate sensor data acquisition, communication, processing, and visualization into a cohesive system.
Middleware frameworks such as AWARE and Open mHealth facilitate sensor data abstraction, synchronization, and interoperability across different platforms [
48].
Application-specific integration includes linking wearables to electronic health records (EHRs), mHealth apps, or AI dashboards for decision support.
Security and reliability remain major concerns, necessitating secure data encryption, authentication mechanisms, and redundancy for critical health services.
The synergy of communication, computation, and integration technologies enables wearable systems to transition from isolated health gadgets to scalable, intelligent health platforms.
3.3. Data Types and Modalities
Wearable devices are capable of capturing diverse forms of data that collectively enable a multifaceted understanding of a user’s health. These data types are broadly categorized into physiological, contextual, and behavioral domains, each offering distinct insights into mental and physical well-being [
49,
50].
3.3.1. Physiological Data
Physiological signals are direct measurements of internal bodily functions, crucial for assessing vital signs, detecting anomalies, and inferring stress or fatigue.
3.3.2. Contextual Data
Contextual information describes the external environment or user’s situational setting, enhancing the interpretation of raw physiological and behavioral signals.
Location and Mobility: GPS modules track spatial movement, enabling correlation between location-based behavior and health outcomes (e.g., sedentary patterns) [
54].
Ambient Conditions: Sensors for light, humidity, and noise contextualize user behavior, such as sleep quality or work productivity.
Device Usage Patterns: Data from smartphones or smartwatches, including app usage and screen time, have been linked to mood and cognitive states [
55].
3.3.3. Behavioral Data
Behavioral metrics capture user habits, movements, and routines, which are critical in identifying early symptoms of mental and physical health decline.
Physical Activity and Gait: Data from accelerometers and gyroscopes help detect motion patterns, postural control, and walking anomalies [
31].
Sleep Patterns: Sleep stages, duration, and disruptions are tracked using multi-sensor fusion approaches and are indicative of mental health status [
56].
Social Interaction: Proximity sensing and communication logs (e.g., Bluetooth, call/text frequency) reveal patterns of social engagement or isolation [
57].
Routine Regularity: Inconsistencies in daily routines may serve as early warning signs of depression, anxiety, or cognitive disorders [
58].
By integrating these three categories of data, wearable systems enable personalized, adaptive health monitoring capable of delivering actionable insights and timely interventions.
4. Computation Frameworks
The deployment of wearable computing systems for mental and physical health monitoring relies on robust computation frameworks capable of processing diverse, high-frequency physiological and behavioral data streams in real time. These frameworks must balance latency, energy efficiency, scalability, and privacy to meet the demands of continuous monitoring and personalized feedback. Four primary computational paradigms dominate this domain: cloud-based frameworks, edge computing, hybrid edge–cloud architectures, and federated learning systems.
4.1. Cloud-Centric Frameworks
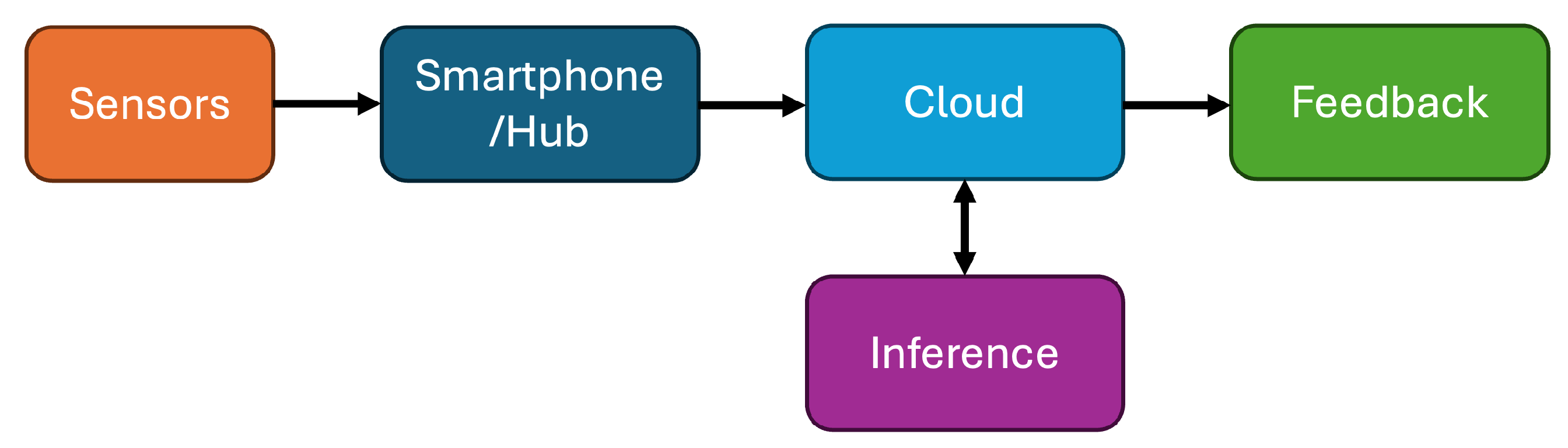
Cloud-centric computation frameworks represent the earliest and most widely adopted architecture for wearable computing systems designed to monitor mental [
59] and physical health [
60]. In this paradigm, as shown in
Figure 5, raw or minimally processed sensor data—captured from devices such as PPG wristbands, accelerometers, EDA sensors, or continuous glucose monitors (CGMs)—is transmitted to centralized cloud servers for data storage, feature extraction, model training, and inference. The results, such as health assessments, anomaly detection, or personalized recommendations, are then fed back to the user or clinician via the connected device. These frameworks leverage the virtually unlimited computational resources and storage capacity of cloud infrastructures to enable advanced analytics, including deep-learning models for multi-sensor fusion, stress detection [
61], sleep staging, arrhythmia classification, and long-term trend analysis.
The primary strength of cloud-centric frameworks lies in their scalability [
62]. Centralized platforms can support large-scale health monitoring deployments, aggregating data from thousands of devices to uncover population-level trends or refine predictive models. Additionally, they facilitate seamless integration with (EHRs) and telemedicine systems [
63], enabling clinicians to access longitudinal data and support remote diagnostics and intervention [
64]. Cloud-based analytics also allow for rapid prototyping and iteration of machine-learning algorithms, as models can be retrained frequently without constraints posed by wearable device hardware. However, cloud-centric systems face notable limitations [
65]. Continuous transmission of raw physiological and contextual data imposes significant energy demands on wearable devices, reducing battery life and limiting the feasibility of long-term, continuous monitoring. Latency is another concern; while acceptable for retrospective analyses, cloud pipelines struggle to support real-time interventions—such as stress-triggered breathing exercises or arrhythmia alerts—that require millisecond-scale feedback. Furthermore, the transmission and centralized storage of sensitive mental health and physiological data introduce privacy and security vulnerabilities, raising the risk of breaches or misuse by third parties, including insurers or employers. Compliance with data protection standards such as GDPR and HIPAA adds regulatory complexity [
66,
67].
Overall, while cloud-centric frameworks have driven the initial scalability and analytical capabilities of wearable computing in healthcare, their inherent challenges in energy efficiency, latency, and privacy have prompted the exploration of edge, hybrid, and federated approaches that can complement or replace purely cloud-based solutions.
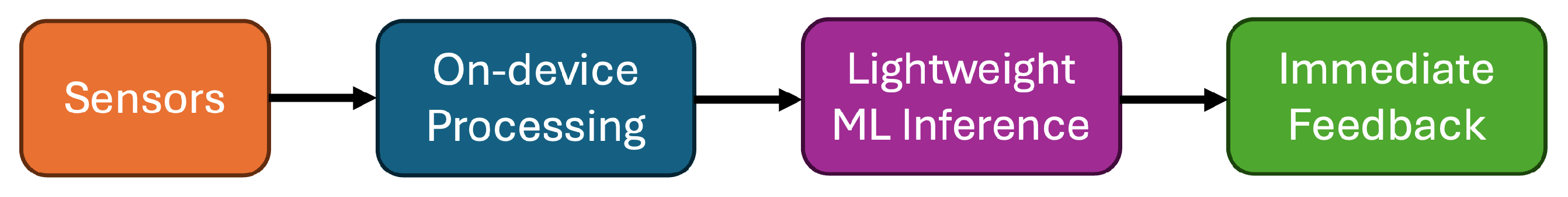
4.2. Edge Computing Frameworks
To address the latency and energy concerns inherent in cloud-centric systems, edge computing shifts computation closer to the user by leveraging the processing capabilities of wearable devices or nearby gateways. Unlike cloud-centric architectures, where raw sensor streams are transmitted to remote servers for processing, edge computing leverages the computational capabilities of wearable devices, smartphones, or nearby gateways to perform data processing, feature extraction, and machine-learning inference locally. This decentralized approach enables real-time analytics and feedback, which is particularly critical for time-sensitive health applications such as arrhythmia detection [
68,
69], fall detection [
70,
71,
72], fall prevention [
72], stress-triggered breathing interventions [
73], and seizure alert systems [
74,
75].
Edge computing frameworks as shown in
Figure 6 process multi-modal sensor data—including accelerometer, gyroscope, PPG, EDA, and sometimes electromyography (EMG)—to generate cleaned, feature-rich representations in real time. Preprocessing tasks typically include filtering, normalization, segmentation, and artifact removal, which help reduce the complexity of the processing algorithm. Given the resource constraints of edge devices (limited CPU/GPU, battery, and memory), many frameworks rely on lightweight models [
76], such as quantized neural networks, Support Vector Machines (SVMs), and Random Forests, or deploy model compression and pruning techniques for deep neural networks to ensure feasibility on-device. Beyond performance benefits, edge computing offers substantial privacy advantages [
77], as raw data (e.g., sensitive physiological or behavioral patterns) often remains on-device, with only processed results or alerts transmitted to the cloud when necessary. This architecture is increasingly integrated with event-driven synchronization, where data are uploaded selectively—such as when anomalies are detected—reducing bandwidth and extending device battery life.
In summary, the edge computing paradigm is transforming wearable health monitoring by enabling low-latency, energy-efficient, and privacy-preserving analytics directly on user devices. Future research is poised to enhance its capabilities through hardware-software co-design, energy-optimized AI models, and synergistic integration with federated and hybrid architectures, enabling scalable and clinically robust monitoring solutions for mental and physical health.
4.3. Hybrid Edge–Cloud Architectures
Hybrid edge–cloud architectures have become the dominant computational paradigm for wearable computing systems supporting mental and physical health monitoring. These frameworks aim to balance the low-latency, energy efficiency, and privacy benefits of edge processing with the scalability and analytic depth of cloud computing. By partitioning computational tasks between the edge (wearable devices, smartphones, or local gateways) and the cloud, these systems enable real-time interventions while still supporting longitudinal trend analysis, advanced AI modeling, and multi-patient aggregation [
78,
79].
In a typical hybrid framework as in
Figure 7, sensor signals from accelerometers, gyroscopes, PPG, EDA, and other biosensors are first preprocessed and partially analyzed at the edge. Tasks like filtering, segmentation, anomaly detection, and basic classification (e.g., detecting falls, arrhythmias, or stress events) are performed locally to minimize latency. For example, an edge device might detect elevated heart rate and galvanic skin response indicative of acute stress and trigger real-time interventions such as haptic breathing prompts without relying on cloud connectivity. Only summarized or event-driven data, such as flagged anomalies or periodic health summaries, are transmitted to the cloud, where resource-intensive tasks—including deep learning-based mood prediction, multimodal data fusion, and population-level analytics—are executed.
This division of labor addresses several limitations of fully cloud-based systems, including high energy consumption from constant data transmission, latency in feedback loops, and heightened privacy risks. By reducing communication overhead and keeping sensitive raw data localized, hybrid architectures can extend battery life and comply with data privacy regulations (e.g., GDPR, HIPAA) [
80,
81]. Furthermore, leveraging the cloud for heavy analytics enables the use of complex models such as transformers, large-scale convolutional networks, or multimodal fusion frameworks that would otherwise be infeasible on constrained edge hardware. Emerging research is enhancing hybrid frameworks through adaptive computing models, where AI-driven orchestration dynamically shifts workloads between edge and cloud based on user state, network conditions, and computational demand. Integration with federated learning [
82] is also gaining traction, allowing hybrid systems to collaboratively update global models while preserving user privacy and maintaining local personalization [
83].
In summary, hybrid edge–cloud architectures provide a balanced, scalable, and adaptive solution for wearable health monitoring, capable of supporting real-time, personalized feedback while enabling sophisticated analytics. As wearable health ecosystems expand, hybrid frameworks are expected to serve as the foundational architecture for next-generation digital health systems, facilitating the convergence of personalization, population health insights, and privacy-aware intelligence.
4.4. Federated Learning Frameworks
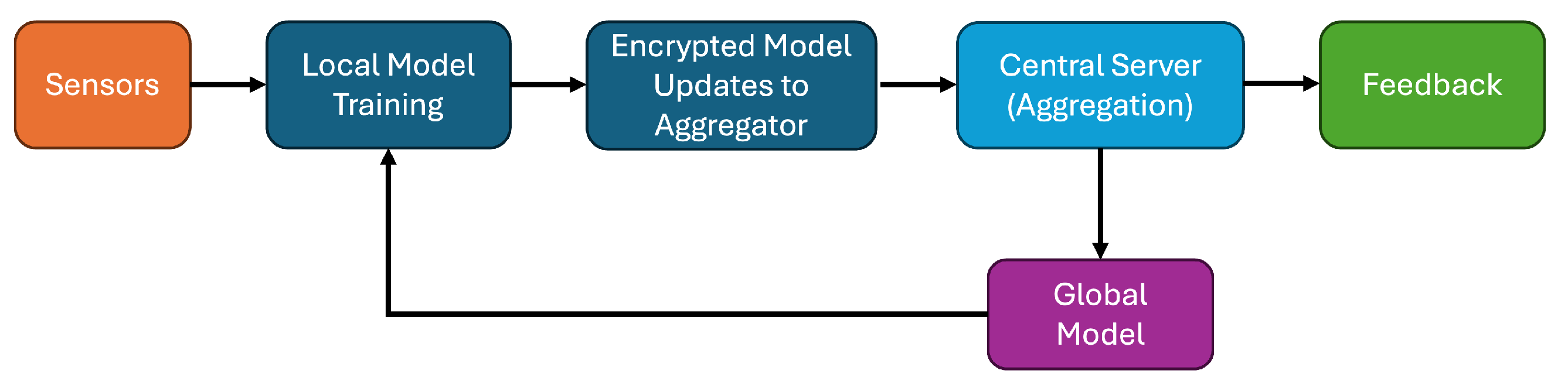
Given the sensitivity of physiological and mental health data, federated learning (FL) has emerged as a privacy-preserving computation framework. In FL as shown in
Figure 8, wearable devices collaboratively train a shared global model by transmitting only model updates or gradients rather than raw data to a central aggregator. In wearable health monitoring, FL is particularly valuable for managing sensitive data streams used to detect conditions such as stress, depression, cardiac irregularities, sleep disturbances, and mobility impairments. By retaining raw data on-device, FL aligns with regulatory frameworks such as HIPAA and GDPR, while supporting continuous, real-world data collection without centralized data pooling [
84]. Furthermore, FL inherently supports personalization [
83], as global models can be fine-tuned locally to reflect individual physiological baselines and behavioral patterns, improving prediction accuracy for diverse populations with heterogeneous sensor characteristics and health conditions. Beyond privacy, FL improves scalability and efficiency by reducing bandwidth usage—critical for low-power wearables—since only compact parameter updates are communicated rather than raw sensor data [
82]. This allows large networks of wearables to collectively contribute to model improvements without overwhelming cloud infrastructure or draining device batteries. Some systems employ federated averaging (FedAvg) [
85] as the core aggregation algorithm, while more advanced frameworks integrate adaptive weighting to account for non-independent and identically distributed (non-IID) data [
86], which is common when users differ in sensor types, activity levels, or health states.
Despite its promise, FL in wearable health monitoring faces significant technical challenges [
82]. Devices vary widely in computational capacity, connectivity, and energy availability, making synchronous training difficult. Communication delays and dropouts can lead to stale or biased updates, slowing model convergence. Moreover, non-IID data distributions pose challenges for maintaining global model generalizability, as models risk overfitting to dominant user profiles or device types. Research is increasingly focused on personalized federated learning, where techniques such as meta-learning, clustered FL, and differential privacy are employed to ensure robust, fair, and privacy-preserving outcomes.
Looking forward, FL is expected to become a cornerstone framework for large-scale, privacy-conscious wearable health ecosystems. Integration with edge and hybrid architectures is gaining momentum, enabling real-time local inference combined with secure global model refinement. Advances in communication-efficient FL (e.g., gradient compression, sparse updates) and robust aggregation algorithms will further enhance the viability of FL for mental and physical health monitoring, paving the way for scalable, personalized, and trustworthy AI-driven healthcare systems.
Overall, these computational paradigms offer distinct trade-offs in scalability, latency, privacy, and computational efficiency as shown in
Table 5. Future wearable health systems will likely combine federated learning and hybrid edge–cloud architectures with energy-optimized AI models to balance performance, privacy, and accessibility across diverse healthcare applications.
5. Applications in Physical Health Monitoring
These health monitoring devices, often integrated into smartwatches, fitness bands, smart textiles, and skin patches, are equipped with sensors capable of measuring heart rate, respiration, body temperature, blood oxygen saturation, and movement [
24,
87]. Applications range from chronic disease management—such as glucose monitoring for diabetes [
2] and arrhythmia detection for cardiovascular patients—to rehabilitation [
4] and post-operative care. Wearables also enable early detection of anomalies and support preventive medicine by analyzing trends in vital signs and behavior patterns. Advances in sensor miniaturization, battery efficiency, and wireless communication have facilitated seamless integration into daily life, making these devices increasingly user-friendly and unobtrusive. Moreover, coupling wearables with cloud-based analytics and AI algorithms enhances diagnostic accuracy and supports personalized healthcare interventions. As a result, wearable computing is reshaping how physical health is monitored, shifting from episodic clinical visits to proactive, data-driven wellness management.
5.1. Cardiovascular Health Monitoring
Wearable computing technologies have emerged as a transformative force in cardiovascular health monitoring, offering continuous, unobtrusive, and real-time data acquisition that surpasses the limitations of traditional point-in-time clinical assessments. These systems typically integrate advanced biosensors, such as PPG [
88,
89,
90,
91,
92], electrocardiography (ECG) [
93,
94,
95], and ballistocardiography (BCG) [
96,
97,
98,
99], to monitor key cardiovascular parameters including (HR), respiratory rate, (SpO
2), and peripheral circulation dynamics. PPG-based devices, commonly used in smartwatches and fitness trackers, estimate pulse wave characteristics by analyzing light absorption changes in the skin, while ECG-enabled wearables like chest straps [
100] or adhesive patches capture electrical activity for arrhythmia detection and waveform analysis [
6,
101] (see
Figure 9 for examples). These data streams are processed locally using embedded microcontrollers or offloaded to mobile phones and cloud platforms, where machine-learning algorithms perform real-time anomaly detection, such as early identification of atrial fibrillation (AF) [
90], premature ventricular contractions, or bradycardia [
102,
103,
104]. In large-scale clinical studies, such as the mSToPS trial, wearable ECG patches demonstrated statistically significant improvements in detecting previously undiagnosed AF when compared to standard care, validating their utility in population-level screening and preventative cardiology [
105].
Moreover, modern wearable cardiovascular systems incorporate edge analytics for low-latency decision support and energy efficiency, and are often synchronized with electronic health record (EHR) systems to support clinician-driven interventions [
106]. The continuous nature of these measurements provides rich longitudinal datasets that facilitate trend analysis, risk stratification, and personalized treatment plans [
7]. Despite challenges such as motion artifacts, data privacy concerns, and regulatory compliance, the integration of wearable cardiovascular monitors into mainstream healthcare is rapidly expanding. Their potential to enable proactive health management, remote patient monitoring, and closed-loop therapeutic feedback makes them indispensable in addressing chronic cardiovascular diseases, which remain a leading global cause of mortality [
4,
10].
Table 6 presents a comparative analysis of five clinical studies evaluating wearable systems for cardiovascular health monitoring. Most studies report on metrics such as heart rate (HR), heart rate variability (HRV), respiratory rate (RR), and electrocardiogram (ECG) signals, with data primarily collected using wrist-worn or PPG-based sensors. Sample sizes vary considerably, ranging from a single test subject to 160 participants, with clinical validation levels spanning ECG comparisons to hospital-based evaluations. While some studies employed robust validation frameworks, others lacked proper benchmarking or were limited by small and homogenous samples. Common limitations include poor data quality, technical issues during activity monitoring, and limited generalizability due to narrow demographic representation or testing in restricted clinical settings.
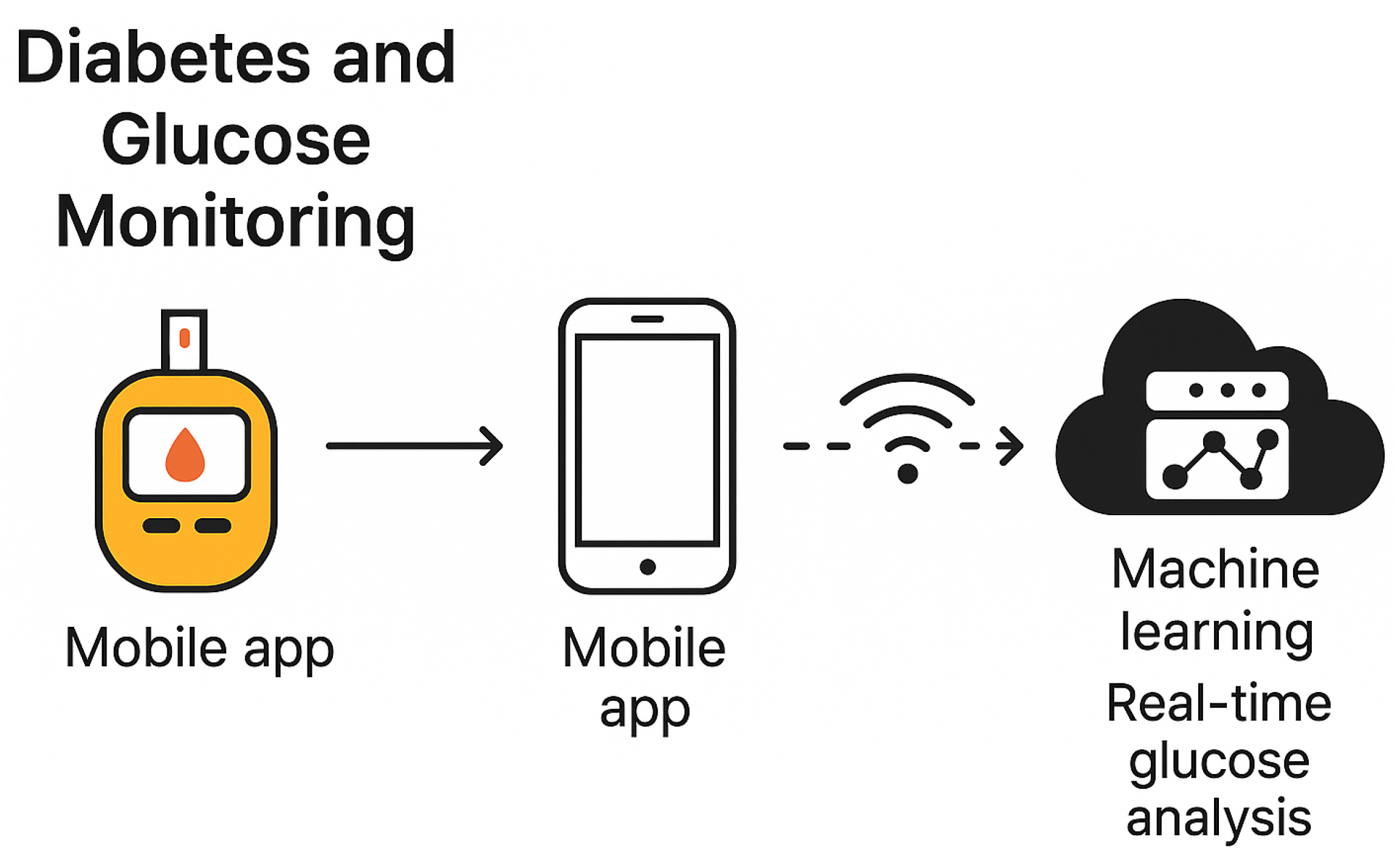
5.2. Diabetes and Glucose Monitoring
Wearable computing technologies have significantly advanced the field of diabetes management by enabling continuous glucose monitoring (CGM) and personalized feedback systems (see
Figure 10). Traditional glucose monitoring methods rely on intermittent finger-prick blood tests, which are invasive and provide only momentary data snapshots. In contrast, wearable CGM devices [
112,
113], often based on electrochemical biosensors, continuously measure glucose concentrations in interstitial fluid, offering real-time data trends that enable timely interventions [
87]. These systems improve glycemic regulation by leveraging activity pattern data collected from wearable devices to predict fluctuations in blood glucose levels among adults with prediabetes [
114], reduce hypoglycemic episodes [
115,
116], and improve the quality of life for individuals with diabetes. Integration of CGM with insulin pumps and mobile health (mHealth) applications has paved the way for closed-loop insulin delivery, known as artificial pancreas systems [
117,
118,
119], which automatically modulate insulin doses based on dynamic glucose readings [
113,
120]. Recent advancements have also led to improved sensor accuracy and longevity, with several commercial devices achieving Mean Absolute Relative Difference (MARD) values below 10%, a benchmark for clinical reliability [
121]. Additionally, non-invasive CGM technologies employing nanomaterial-based sensors [
122,
123,
124] are under development, aiming to further reduce user discomfort while maintaining high sensitivity and specificity [
125]. These innovations signal a paradigm shift in diabetes care, moving from episodic to continuous and personalized disease management.
The compared studies, see
Table 7 collectively explore the feasibility, accuracy, and user interaction with wearable glucose monitoring systems. Most studies rely on CGM devices—either commercial (e.g., Dexcom) or novel wearable patches—and evaluate clinical validity through metrics like TIR, TAR, TBR, HbA1c, or direct glucose level correlations. Sample sizes vary widely—from 6 to 108 participants—with some studies focusing on qualitative feedback while others conduct longitudinal or lab-based validations. Common limitations include small sample sizes, lack of real-world diversity, and short duration of evaluation, signaling a need for broader clinical validation in future work.
5.3. Respiratory and Pulmonary Function
Wearable computing technologies have emerged as powerful tools for monitoring respiratory and pulmonary function, offering continuous, non-invasive assessment of respiratory health parameters. These systems typically incorporate a range of physiological sensors such as piezoelectric belts, respiratory inductance plethysmography (RIP), strain sensors, and acoustic microphones to measure key indicators like respiratory rate [
131,
132], tidal volume, and breathing patterns in real time [
52] (see
Figure 11). More advanced wearable platforms utilize flexible and stretchable materials [
133] to conform to the thoracic cavity, enabling precise detection of chest wall movements during inhalation and exhalation [
134]. The integration of these sensors with wireless transmission modules and edge-processing capabilities allows data to be processed locally or transmitted to cloud platforms for clinical evaluation and remote health monitoring. In particular, wearables have proven beneficial in managing chronic respiratory diseases such as asthma [
135,
136,
137,
138], chronic obstructive pulmonary disease (COPD) [
139,
140,
141], and sleep apnea [
142,
143,
144,
145], where early detection of anomalies can prevent exacerbations and reduce hospitalizations [
53]. Additionally, wearable respiratory monitors are being increasingly adopted in post-COVID rehabilitation and environmental exposure studies, where continuous assessment of pulmonary function under ambulatory conditions is crucial [
50,
146,
147]. These systems are also being equipped with machine learning algorithms that can classify respiratory events and detect anomalies such as wheezing or apnea, enabling more accurate, personalized health interventions. Thus, wearable respiratory monitoring technologies are reshaping pulmonary diagnostics and chronic care management by offering scalable, comfortable, and context-aware solutions for long-term health tracking.
The comparison presented in
Table 8 of five clinical studies reveals the growing diversity and capability of wearable health monitoring systems in measuring metrics such as respiratory rate (RR), heart rate (HR), ECG, and oxygen saturation (SpO
2). Devices ranged from PPG-based patches and dry-electrode health patches to eddy current and multimodal biosensors. Validation efforts varied, with sample sizes ranging from 4 to 70 participants and comparisons made against clinical-grade devices like VitalPatch and Cosmed K5. Despite demonstrating promising sensor performance, the studies were limited by small and often homogenous participant groups, short study durations, and a lack of real-time clinical integration, underscoring the need for larger, standardized validation trials to support widespread clinical adoption.
5.4. Musculoskeletal and Gait Analysis
Wearable computing technologies play a crucial role in musculoskeletal and gait analysis by enabling real-time, unobtrusive monitoring of joint kinematics, limb dynamics, and biomechanical parameters across various settings, including clinical, athletic, and rehabilitation environments. These systems typically integrate inertial measurement units (IMUs), accelerometers, gyroscopes, magnetometers, and pressure sensors to capture motion patterns and joint angles with high temporal resolution [
153,
154,
155]. Wearable sensors offer a flexible alternative to traditional lab-based motion capture systems, such as optical tracking and force platforms, which are limited by their cost, complexity, and constrained measurement environments [
156]. Advances in textile-based electronics and flexible sensor integration have further facilitated the design of smart garments that provide full-body motion capture without impeding natural movement [
157]. These wearables are widely used in gait analysis for diagnosing neurological disorders like Parkinson’s disease, monitoring rehabilitation progress after orthopedic surgeries, and assessing fall risk in elderly populations [
158,
159]. Embedded machine-learning algorithms allow for on-device pattern recognition and classification of abnormal movement patterns, contributing to early diagnosis [
160] and personalized therapy plans [
161,
162]. This is valuable for fall detection in elderly populations [
163,
164], rehabilitation tracking after surgeries, and orthopedic diagnostics [
165]. Furthermore, cloud-based platforms enable long-term tracking and remote monitoring by clinicians, enhancing patient adherence and enabling continuous assessment beyond clinical visits [
4]. As the demand for at-home rehabilitation and telehealth services grows, wearable technologies are becoming indispensable in musculoskeletal assessment, offering scalable and cost-effective tools for precision movement analysis.
5.5. Sleep and Circadian Rhythm Monitoring
Wearable computing technologies have revolutionized the assessment of sleep and circadian rhythms by offering continuous, non-invasive, and user-friendly tools for long-term monitoring in naturalistic settings. Traditional polysomnography (PSG), while the gold standard, is limited by its clinical setting, complexity, and discomfort for users. In contrast, wearable devices leverage sensors such as accelerometers, PPG, EDA, and skin temperature sensors to estimate sleep stages, circadian patterns, and related physiological signals like HR) and respiratory rate [
166,
167]. Wrist-worn actigraphy has been widely adopted for sleep-wake detection, showing strong correlations with PSG for sleep duration and efficiency, making it suitable for population-scale studies [
168]. Newer multimodal wearables, such as the Oura Ring and WHOOP Strap, integrate machine-learning algorithms to classify sleep stages (light, deep, REM) [
169,
170] and identify disorders such as insomnia or sleep apnea [
171] with increasing accuracy by combining motion and PPG-derived metrics. Additionally, circadian rhythm monitoring through skin temperature and ambient light sensors enables detection of chronotype shifts [
172], jet lag [
173], and sleep disorders [
174,
175] such as delayed sleep phase syndrome [
176,
177]. These data-rich systems support behavioral interventions and personalized sleep hygiene programs by providing real-time feedback via mobile apps or cloud platforms. Furthermore, research has explored the use of wearables for detecting early signs of cognitive decline and mood disorders [
178], given the strong bidirectional relationship between sleep and mental health [
179]. Long-term monitoring can provide insights into lifestyle patterns and overall health outcomes [
56,
180]. Despite their promise, challenges remain in achieving PSG-level accuracy for clinical diagnosis, standardizing algorithms across platforms, and addressing privacy and data integrity in remote health monitoring. Nonetheless, wearable sleep monitoring systems are paving the way for accessible, scalable, and personalized sleep health solutions.
To critically assess the maturity and applicability of wearable technologies in real-world health monitoring scenarios, we synthesized findings from representative clinical studies into a unified comparative table (
Table 9). This table presents a structured overview based on four criteria: metrics reported, sensor modalities, clinical validation characteristics, and study limitations. Metrics such as apnea–hypopnea index (AHI) correlation, accuracy in sleep staging or disorder detection, and total sleep time mean absolute error (TST MAE) were consistently used to evaluate performance across studies. The sensors utilized ranged from PPG and ECG to multimodal platforms including inertial and ambient sensors. Validation settings spanned lab-controlled environments to hybrid and home-based deployments, with participant numbers ranging from 35 to 75 and comparisons against gold-standard PSG benchmarks. Finally, the table highlights common limitations such as signal quality degradation due to motion artifacts, reduced specificity in certain populations, lack of real-time analytics, and power or communication constraints. This summary offers an evidence-based reference for researchers and developers aiming to benchmark or enhance future wearable health monitoring systems.
Critical assessment (sleep): Wearables show good agreement with PSG for macro-sleep metrics (e.g., TST, SE), but stage classification and apnea severity estimation still lag gold standards. More standardized protocols and longitudinal validation are required.
5.6. Fitness and Activity Tracking
Wearable computing technologies have revolutionized fitness and activity tracking by enabling continuous, non-intrusive monitoring of physical performance metrics such as step count, energy expenditure, heart rate, oxygen saturation, and sleep quality. These devices—typically worn as wristbands, smartwatches, or smart clothing—leverage integrated sensors such as accelerometers, gyroscopes, magnetometers, and optical PPG to capture multidimensional data in real time [
184,
185,
186]. Advanced algorithms, often driven by machine learning, transform raw sensor signals into actionable insights, allowing users to assess their fitness level, set personalized goals, and detect anomalies indicative of overtraining or underlying health issues [
187]. For example, the combination of inertial measurement units (IMUs) with heart rate monitors has enabled estimation of VO
2 max and caloric burn with high reliability [
188]. Cloud-based platforms further enhance these systems by enabling tracking, trend visualization, and social interaction, while edge-processing techniques are increasingly employed to reduce latency and power consumption during high-frequency data capture [
189]. Recently, more advanced systems provide real-time coaching or AI-driven insights based on historical data trends [
10].
5.7. Rehabilitation and Recovery
Wearable computing has emerged as a transformative tool in rehabilitation and recovery by enabling continuous, remote, and personalized monitoring of patients undergoing physical therapy or recovering from musculoskeletal injuries, neurological disorders, and post-surgical procedures. These systems typically integrate inertial measurement units (IMUs), electromyography (EMG) sensors, and pressure sensors into wearable formats such as smart garments, knee braces, or wristbands to capture kinematic and biomechanical data during rehabilitation exercises [
4,
190]. The collected data allows clinicians to assess joint angles, gait symmetry, range of motion, and muscle activation patterns in real time, facilitating early detection of abnormal movement and poor compliance to prescribed regimens [
191]. Advanced machine-learning algorithms further enhance these systems by enabling automated classification of movement quality, performance scoring, and adaptive feedback generation [
192]. For instance, wearable devices have been shown to support telerehabilitation programs by allowing patients to perform exercises at home while transmitting relevant metrics to remote healthcare providers for feedback and progress evaluation [
193]. Moreover, closed-loop systems combining wearable sensors with haptic actuators [
194] or electrical stimulators can deliver corrective feedback or neuromuscular stimulation [
195], thus accelerating motor relearning and improving functional outcomes, especially in stroke or spinal cord injury recovery. Post-operative or post-stroke rehabilitation is increasingly enhanced through wearable technologies that guide therapeutic exercises and assess motor improvements. Motion tracking wearables [
196] provide biofeedback and allow remote supervision by clinicians. It can also monitor upper extremity functions during daily life in neurologically impaired individuals [
197]. These technologies reduce the burden on healthcare systems, lower rehabilitation costs, and increase patient engagement by offering gamified interfaces and progress visualization tools.
Table 10 presents a ranking of the different sensors relevance for physical health monitoring applications in wearable computing
6. Applications in Mental Health Monitoring
Wearable computing has increasingly become integral in monitoring and improving mental health, providing real-time, objective, and continuous assessment of psychological states (see
Figure 12). These technologies offer promising tools for early detection, intervention, and management of mental disorders such as depression, anxiety, bipolar disorder, and stress-related conditions [
198,
199].
6.1. Stress and Anxiety Detection
Stress and anxiety disorders affect a significant portion of the global population. Wearables equipped with physiological sensors such as EDA, and skin temperature can non-invasively capture autonomic nervous system responses associated with stress [
5] and provide an assessment of physiological and behavioral markers [
200] associated with emotional and cognitive states. Elevated EDA, for instance, is correlated with sympathetic arousal and has been widely used in stress quantification, while HRV offers insights into parasympathetic activity, with lower HRV often linked to higher stress and anxiety levels [
201,
202]. Advanced wearable devices such as Empatica E4 and Fitbit Sense utilize multi-modal data streams and embedded machine-learning algorithms to detect stress episodes and provide feedback for self-regulation and intervention [
203]. Recent studies have demonstrated the effectiveness of deep-learning models and personalized calibration techniques in improving detection accuracy by accounting for individual variability and contextual information such as time of day, activity level, and social interactions [
8,
204]. Furthermore, the integration of ecological momentary assessment (EMA) frameworks into wearable platforms [
205] allows for correlating sensor-derived stress indices with subjective reports, improving the reliability of detection and offering rich insights into daily mental health fluctuations [
6].
6.2. Depression Monitoring
Depression, characterized by persistent sadness, loss of interest, and cognitive and physiological dysregulation, often manifests in measurable changes in sleep patterns [
57,
58], physical activity, circadian rhythm, and autonomic nervous system signals. Wearable sensors, including accelerometers, heart rate monitors, skin conductance sensors, and actigraphy-based sleep trackers, enable high-resolution data collection that can be used to model depressive behaviors in real-world settings [
206,
207,
208]. For example, reduced step count, increased sedentary behavior, and irregular sleep-wake cycles have been correlated with depressive symptom severity [
58]. In addition, alterations in HRV, which reflect reduced vagal tone, have been consistently associated with major depressive disorder (MDD) and can be measured via ECG-equipped wearable devices [
209]. Advances in machine learning allow for the integration of these multimodal signals into predictive models capable of identifying individuals at risk, monitoring symptom trajectories, and potentially guiding personalized interventions [
198].
6.3. Bipolar Disorder Management
Wearables aid in tracking mood fluctuations and activity rhythms in individuals with bipolar disorder, a chronic and recurrent mood disorder characterized by alternating episodes of depression and mania or hypomania. These mood fluctuations are often associated with physiological and behavioral changes that can be unobtrusively captured through wearable devices, enabling early detection of mood episodes [
7,
210] and more responsive intervention strategies [
211]. Wearable sensors such as accelerometers, EDA monitors, HRV sensors, and sleep trackers provide continuous streams of data related to circadian rhythms, physical activity, autonomic function, and sleep quality—all of which have been found to vary significantly across mood states in individuals with BD [
212,
213]. For example, reduced sleep duration and increased motor activity often precede manic episodes, while extended inactivity and disturbed circadian patterns are typically observed during depressive phases [
214]. Leveraging these insights, machine-learning algorithms trained on multimodal sensor data have demonstrated potential in distinguishing between mood states, forecasting transitions, and supporting mood stabilization through digital phenotyping [
215,
216]. Moreover, the integration of ecological momentary assessments (EMAs) with wearable sensor feedback allows for real-time mood monitoring and personalized behavioral feedback, reducing the reliance on subjective self-reporting and enhancing clinical decision making [
217].
6.4. Cognitive Load and Attention Assessment
Wearable computing technologies offer a robust and scalable approach to assessing cognitive load and attention by continuously capturing physiological and behavioral indicators linked to mental effort and attentional focus. Cognitive load, defined as the total amount of mental effort being used in the working memory, can significantly influence learning outcomes, task performance, and error rates in high-stakes environments. Wearable devices equipped with electroencephalography (EEG), HRV, EDA, and eye-tracking [
218] sensors enable real-time monitoring of neurophysiological signals that reflect attentional engagement and mental workload [
219,
220]. For instance, increased theta and decreased alpha band power in EEG signals are commonly associated with heightened cognitive workload, while reduced HRV and elevated EDA levels typically indicate sympathetic nervous system activation due to increased mental effort [
220,
221]. Eye-tracking wearables further enrich the assessment by capturing metrics like fixation duration, pupil dilation, and saccadic velocity, which are sensitive to attentional demand and task complexity [
222]. These multi-modal data streams, when analyzed through machine learning and signal processing techniques, can enable robust and individualized models for detecting cognitive states across diverse contexts such as aviation, education, and neurorehabilitation [
223,
224]. Wearable EEG devices such as the Muse headband or NeuroSky MindWave have shown utility in monitoring cognitive load, attention levels, and neurofeedback training [
225,
226]. These applications support populations with ADHD or occupational stress, providing actionable feedback and enhancing mental resilience [
227,
228]. Additionally, wearable-based cognitive assessment tools offer the advantage of mobility, allowing researchers and clinicians to measure cognitive responses in naturalistic settings rather than constrained lab environments.
6.5. Sleep and Mental Well-Being
Sleep plays a vital role in emotional regulation, cognitive performance, and mental health, and disruptions in sleep architecture are closely linked to conditions such as depression, anxiety, and bipolar disorder. Modern wearables equipped with accelerometers, PPG, EDA, and HRV sensors can effectively estimate sleep stages, total sleep time, sleep efficiency [
56,
171], and nocturnal awakenings without requiring clinical polysomnography setups [
229,
230]. Given the strong bidirectional link between sleep and mental health [
231,
232,
233], wearable sleep trackers are valuable in diagnosing and treating sleep-related disorders such as insomnia and hypersomnia.
6.6. Behavioral Intervention and Feedback Systems
Modern wearables can deliver just-in-time adaptive interventions (JITAIs) by combining sensor data with context-aware systems [
55]. These platforms deliver personalized prompts, cognitive-behavioral therapy (CBT) support, or relaxation guidance to manage psychological distress. Integration with smartphones and cloud analytics enhances accessibility and scalability. For example, wearables can identify prolonged sedentary behavior and prompt users to take activity breaks, or monitor stress indicators via HRV and deliver relaxation prompts or breathing exercises [
234]. Through machine-learning algorithms and context modeling, these devices can infer high-risk states—such as excessive alcohol consumption, smoking urges, or emotional dysregulation—and deliver just-in-time adaptive interventions (JITAIs) via haptic, visual, or auditory feedback mechanisms [
235]. Moreover, when combined with smartphone interfaces and cloud-based analytics, wearable feedback systems can be integrated into digital therapeutics and cognitive-behavioral therapy (CBT) platforms, supporting habit formation, self-efficacy, and long-term behavioral change [
236]
In conclusion, wearable technologies offer a multidimensional approach to mental health care, enabling unobtrusive monitoring, early detection, and personalized interventions that augment traditional psychiatric treatment paradigms.
Table 11 presents a ranking of the different sensors relevance for mental health monitoring applications in wearable computing.
7. Machine-Learning and AI Techniques
The proliferation of wearable computing devices for mental and physical health monitoring has spurred the development of advanced machine-learning (ML) and artificial intelligence (AI) techniques that can transform raw sensor data into actionable health insights. These techniques address challenges such as heterogeneous sensor data, signal noise, personalized health baselines, and the need for privacy-preserving analytics in large-scale deployments. The AI pipeline for wearable health monitoring typically involves four core stages: data preprocessing, modeling, sensor fusion, and federated learning for distributed intelligence.
7.1. Data Preprocessing
Raw data streams from wearable sensors—including accelerometers, gyroscopes, PPG, EDA, electrocardiography (ECG), and electromyography (EMG)—are often noisy, irregularly sampled, and susceptible to motion artifacts. Preprocessing involves filtering, normalization, and segmentation to improve signal quality and extract meaningful features [
237]. Time-domain (e.g., HRV metrics), frequency-domain (e.g., spectral energy bands for stress detection), and time-frequency domain features (e.g., wavelet transforms for EEG or EMG) are commonly derived [
238]. Data augmentation techniques, such as synthetic oversampling and jittering, help mitigate class imbalance in mental health datasets [
239], where clinically labeled events (e.g., panic attacks) are rare. Preprocessing pipelines increasingly leverage unsupervised denoising autoencoders and adaptive filtering to automate signal enhancement and reduce manual intervention.
7.2. Modeling and Predictive Analytics
AI models applied to wearable data range from traditional machine-learning algorithms to deep learning architectures. Classical models such as Support Vector Machines (SVMs), Random Forests, and k-Nearest Neighbors (k-NN) have been widely used for activity classification, stress detection, and physiological state prediction due to their interpretability and low computational requirements [
204,
240]. More recently, deep-learning approaches—Convolutional Neural Networks (CNNs) and Recurrent Neural Networks (RNNs) including Long Short-Term Memory (LSTM) models—have shown superior performance in capturing temporal dependencies and spatial patterns from multimodal signals [
241], making them well-suited for applications like sleep stage classification [
242] and mood prediction [
243]. Transfer learning techniques [
244] further enable the reuse of pre-trained physiological models, addressing the challenge of limited labeled health data.
7.3. Sensor Fusion for Multimodal Insights
Sensor fusion techniques integrate heterogeneous data streams to enhance the robustness and accuracy of health assessments. Early fusion approaches concatenate synchronized sensor features (e.g., accelerometer and PPG signals) before feeding them into a unified model, while late fusion combines the outputs of modality-specific models through ensemble techniques or probabilistic weighting [
245]. Advanced fusion leverages attention mechanisms and graph neural networks (GNNs) [
246] to dynamically weigh sensor contributions based on contextual relevance, such as prioritizing EDA during stress episodes or motion sensors during activity transitions. Multimodal fusion [
247] has proven particularly effective in complex mental health monitoring tasks, where combining physiological, contextual, and behavioral signals improves the detection of depression, anxiety, and cognitive load beyond what single modalities can achieve.
7.4. Federated Learning and Privacy-Preserving Analytics
Given the sensitive nature of mental and physical health data, federated learning (FL) has emerged as a key paradigm for enabling collaborative model training without centralizing raw data. In FL, wearable devices train local models on-device and share only encrypted model updates with a central aggregator, significantly reducing privacy risks [
248] while maintaining scalability [
82,
249]. FL frameworks tailored for wearables address challenges such as device heterogeneity, variable connectivity, and energy constraints [
250] by employing adaptive aggregation strategies and lightweight neural architectures. Moreover, federated transfer learning and personalization layers [
84] allow models to adapt to individual physiological baselines, which is crucial for mental health monitoring where inter-individual variability is pronounced. Combining FL with on-device inference [
223] supports real-time, privacy-conscious applications such as early stress alerts, seizure detection, and cognitive state tracking.
Overall, the integration of ML and AI techniques—from preprocessing and deep modeling to multimodal fusion and federated learning—has transformed wearable computing into a powerful platform for scalable, adaptive, and privacy-aware health monitoring. Future research will likely focus on explainable AI (XAI) approaches to enhance clinical trust, energy-efficient deep learning for continuous monitoring, and hybrid edge-cloud intelligence to balance latency, power, and privacy requirements.
8. Evaluation Metrics and Benchmarks
The growing integration of wearable computing into mental and physical health monitoring has necessitated the development of standardized evaluation metrics and benchmarks to assess system accuracy, usability, reliability, and clinical relevance. Since these devices often operate outside controlled environments, their evaluation must encompass both technical performance and human-centered factors to ensure their long-term utility in research, clinical, and consumer contexts.
8.1. Performance Metrics
Performance metrics gauge how accurately and reliably wearable devices measure and interpret physiological and behavioral parameters. Signal fidelity is a primary metric, often quantified using mean absolute error (MAE), root mean square error (RMSE), or correlation coefficients relative to clinical gold standards such as electrocardiography (ECG), polysomnography (PSG) [
101], or spirometry for respiratory assessment. In classification tasks—such as stress detection, activity recognition, or sleep stage classification—metrics such as precision, recall, specificity, sensitivity, F1-score, accuracy, and the area under the receiver operating characteristic curve (AUC-ROC) are widely adopted [
251,
252]. For time-series analysis and longitudinal monitoring, techniques like dynamic time warping (DTW) and Bland–Altman plots are used to capture temporal alignment and measurement bias across modalities. Additionally, for AI-driven wearables, model latency, computational complexity (FLOPs, memory requirements), and energy efficiency are critical benchmarks, particularly for real-time and edge-based systems where device resources are constrained.
8.2. Usability and User Experience
Usability is fundamental to the adoption and sustained use of wearable devices. Metrics for usability include wear comfort, form factor, and intrusiveness, as these directly influence adherence and data continuity. Subjective tools such as the System Usability Scale (SUS) and NASA Task Load Index (NASA-TLX) [
253,
254] assess perceived ease of use, mental workload, and user satisfaction. Battery life and charging frequency are also central usability indicators; excessive maintenance burdens reduce device compliance, especially in continuous monitoring applications. Moreover, ease of data visualization and clinician integration impacts the utility of wearables in real-world healthcare workflows. Systems that enable seamless interoperability with EHRs and provide actionable, interpretable insights are more likely to achieve clinical adoption [
255].
8.3. Adherence and Engagement
The clinical value of wearable health systems hinges on user adherence—the consistency with which individuals wear the device and engage with associated software. Adherence is typically quantified through wear-time percentage, data completeness rates, and behavioral engagement metrics such as goal completions, app interactions, or response rates to prompts [
256,
257]. Sustained engagement [
258] is often undermined by factors like false positives, frequent alerts, discomfort, privacy concerns, or user fatigue. Studies often set minimum wear-time [
259] thresholds—commonly 70–80% over a study period—as benchmarks for data validity, although thresholds may vary by application. To bolster adherence, many modern platforms incorporate just-in-time adaptive interventions (JITAIs) [
235], gamification, and personalized feedback to maintain motivation and reduce burden.
8.4. Benchmark Datasets and Validation Frameworks
Robust benchmarking requires standardized datasets and validation protocols. Datasets like WESAD (Wearable Stress and Affect Detection) [
260] and PAMAP2 (Physical Activity Monitoring) [
261] serve as foundational benchmarks for stress detection and activity classification, respectively, offering multimodal signals for model training and comparison. However, the field still lacks large-scale, diverse, and longitudinal datasets that capture complex, multi-condition scenarios, particularly for mental health states such as depression, bipolar disorder, and cognitive stress. Validation frameworks must also account for cross-device variability, differences in sampling rates [
262], and sensor placement [
263], which can significantly skew reported performance. To address these gaps, recent initiatives call for open, standardized benchmarks and clinically validated reference protocols that integrate multi-site, demographically representative cohorts, enabling reproducibility and generalizability across studies.
8.5. Toward Standardization
Current disparities in evaluation protocols hinder cross-study comparability and slow clinical translation. Future benchmarking efforts should emphasize harmonized performance metrics, open-access datasets, and multi-dimensional evaluation frameworks that jointly consider technical accuracy, usability, and user adherence. Incorporating explainable AI (XAI) [
264,
265] for interpretability, alongside energy and privacy metrics for federated and edge-based systems, will further align evaluation practices with real-world deployment needs.
9. Challenges and Limitations
While wearable computing has revolutionized the monitoring of mental and physical health by enabling continuous, real-time, and unobtrusive data collection, its widespread deployment faces a range of technical, ethical, and practical challenges that hinder its effectiveness and large-scale adoption.
9.1. Technical Challenges
Wearable devices face persistent challenges in data accuracy, reliability, and signal integrity due to their dependence on physiological and motion sensors that are susceptible to noise and artifacts. Motion-induced interference significantly impacts signals like PPG and EDA, especially during high-mobility activities, leading to unreliable measurements of HRV and stress markers [
101]. Sensor calibration and placement variability across users exacerbate these issues, resulting in inconsistent baselines for vital signs or activity metrics. Battery limitations impose further constraints [
189], as the need for long-term continuous monitoring conflicts with power-intensive sensing, computation, and wireless transmission, particularly in multi-sensor systems or devices employing high-frequency data acquisition [
266,
267]. Additionally, algorithmic robustness and generalizability remain hurdles; AI models trained on limited, demographically skewed datasets [
268] often underperform when deployed across diverse populations, leading to inaccurate health predictions and reduced clinical reliability. Interoperability is another barrier [
269], as proprietary hardware and data formats limit integration across devices, platforms, and healthcare systems, complicating multi-modal sensor fusion and longitudinal tracking.
9.2. Ethical and Privacy Concerns
The continuous capture of sensitive physiological and behavioral data raises critical privacy [
270], security [
271], and ethical issues. Health-related data [
272], including stress biomarkers, sleep patterns, or activity profiles, can reveal highly personal insights, and breaches or misuse pose risks of discrimination, stigmatization, or unauthorized profiling by insurers or employers. Although encryption and anonymization techniques are standard, federated learning [
273] and differential privacy [
274] are still emerging solutions, and their deployment at scale is challenged by computational and communication overheads on resource-constrained wearables [
275]. Furthermore, ethical questions arise regarding informed consent and user autonomy [
276]; many users may not fully comprehend the extent of data being collected, shared, and analyzed, particularly when wearables integrate with third-party applications or cloud analytics. Addressing these concerns requires robust legal frameworks, transparent consent mechanisms, and adherence to standards such as GDPR and HIPAA [
67].
9.3. Practical and User-Centric Issues
Beyond technical and ethical challenges, practical factors often limit the usability and adoption of wearable health monitoring systems. User compliance remains a significant issue, as comfort, aesthetics, and intrusiveness influence adherence; bulky or conspicuous devices tend to see reduced long-term engagement, undermining the continuity of data streams critical for mental health monitoring [
277,
278]. False positives or inaccurate alerts, arising from imperfect sensor readings or algorithmic models [
279], can lead to user fatigue and reduced trust, discouraging individuals from sustained use. Cost is another barrier, as advanced wearables with multi-sensor capabilities and integrated AI analytics remain expensive, limiting accessibility for underserved populations and widening health inequities [
9]. Finally, the integration of wearable-derived insights into clinical workflows remains limited due to lack of standardized validation protocols and skepticism among healthcare professionals regarding data accuracy and clinical utility.
9.4. Barriers to Clinical Validation of Wearable and Digital Health Systems
Clinical validation remains a crucial bottleneck for the widespread adoption of wearable and AI-enabled health monitoring systems. Despite promising performance in controlled settings, these technologies often face significant translational barriers when moving toward real-world clinical use. These barriers span regulatory, technical, clinical, and sociocultural domains, as detailed in
Table 12. Addressing these barriers requires a multi-stakeholder effort, including regulatory harmonization, robust data standardization, long-term outcome studies, and transparent explainable AI frameworks. Enhanced collaboration between device developers, clinicians, patients, and regulators is vital for sustainable clinical adoption.
Obtaining FDA approval for wearable devices designed for health monitoring is a complex and multifaceted process that poses several significant challenges. One major hurdle is establishing robust clinical validity and utility, which requires extensive clinical trials to demonstrate that the device reliably measures or detects specific health conditions in real-world environments. For novel metrics, the absence of gold-standard benchmarks further complicates validation. Developers must also navigate evolving regulatory pathways such as the 510 (k) clearance or De Novo classification, each with distinct evidence requirements. Wearables integrating AI or machine learning face added scrutiny under the FDA’s SaMD framework, particularly concerning algorithm transparency, performance stability, and post-market update controls. Ensuring secure data transmission and privacy compliance is another critical barrier, especially when devices rely on wireless communication or cloud-based storage. In addition, developers must address interoperability with EHRs using standards like HL7 or FHIR to enable integration into clinical workflows. Finally, usability and human factors testing are essential to confirm that the device can be safely and effectively used by both patients and healthcare providers. Collectively, these technical, regulatory, and clinical validation barriers extend development timelines and necessitate cross-disciplinary expertise, making FDA approval a rigorous endeavor for wearable health technologies.
Although it is difficult to obtain FDA approval, many wearable devices have obtained that approval. These devices utilize a range of embedded computing technologies that enable real-time physiological monitoring, data processing, and wireless communication. At the hardware level, these devices often incorporate ultra-low-power microcontrollers (MCUs) or system-on-chips (SoCs) tailored for energy-efficient operation, such as ARM Cortex-M series chips. These chips support onboard data acquisition from biosensors (e.g., ECG, PPG, accelerometers) and perform initial signal conditioning and feature extraction. Some advanced wearables integrate dedicated DSPs or hardware accelerators for tasks like arrhythmia detection or sleep stage classification. For communication, BLE is commonly used for data transmission to companion mobile applications or edge gateways. On the software side, these systems frequently employ real-time operating systems (RTOS) to manage tasks such as sensor sampling, data buffering, and anomaly detection. Furthermore, FDA-cleared wearables like the Apple Watch (for atrial fibrillation detection) or the BioIntelliSense BioSticker utilize encrypted data transfer protocols and secure APIs for integration with electronic health records (EHRs), complying with HIPAA and FDA cybersecurity requirements. This layered computing architecture enables these devices to operate autonomously while supporting cloud connectivity and clinician-facing dashboards, forming a secure and clinically validated digital health ecosystem.
Overall, while wearable computing offers immense promise for advancing personalized mental and physical health monitoring, overcoming these technical, ethical, and practical challenges is critical for achieving reliable, equitable, and clinically integrated solutions.
10. Future Directions
Despite substantial advancements in wearable computing for health monitoring, the field is undergoing a rapid transformation driven by sensor miniaturization, AI acceleration, material innovation, and deeper clinical integration. We foresee wearables evolving from passive trackers into active, intelligent health companions that seamlessly merge sensing, computation, and intervention in real time. The following directions outline where research and innovation are poised to make the greatest impact:
10.1. High-Performance, Energy-Efficient Edge Computing
The limitations of current wearable platforms often stem from the inability to run complex AI models locally. Near-future architectures such as ultra-low-power neuromorphic chips, analog in-memory computing arrays, and domain-specific AI accelerators will enable advanced analytics directly on-device, eliminating latency and preserving data privacy. Work such as [
280] demonstrates the feasibility of integrating specialized accelerators within the strict energy envelopes of wearables. This will allow real-time disease detection, multimodal fusion, and personalized feedback without cloud dependency.
10.2. Personalized and Precision Medicine
We anticipate a shift toward individualized baselines for health metrics, moving beyond population norms. This will be fueled by multi-omic data integration (genomics, proteomics, metabolomics) combined with long-term behavioral tracking. AI models trained on these datasets could detect pre-symptomatic disease onset, mental health deterioration, or medication side effects with high specificity. Federated and transfer learning will be essential to achieving personalization while preserving privacy, especially in heterogeneous global populations.
10.3. Smart Fabrics and Material Innovations
The next wave of wearables will be textile-native, embedding biosensors, stretchable circuits, and energy-harvesting fibers into garments for continuous, unobtrusive monitoring. Smart fabrics capable of detecting ECG, EMG, hydration levels, and sweat metabolites are moving toward mass production [
281,
282,
283]. Key challenges include washable, durable electronics and low-power wireless modules that sustain long-term use.
10.4. Context-Aware and Multimodal Intelligence
Future systems will integrate environmental, physiological, and behavioral sensing into adaptive feedback loops. Deep-learning architectures including graph neural networks will fuse multimodal data streams to detect complex states like cognitive overload, emotional distress, or early mobility decline [
246,
284]. Explainable AI will ensure these insights remain clinically interpretable, while digital twin models could simulate patient responses to guide interventions.
10.5. Integration with Digital Therapeutics and Clinical Workflows
Wearables will increasingly operate as active treatment delivery platforms rather than just monitors. Closed-loop therapeutic systems may modulate rehabilitation programs, deliver haptic feedback for anxiety control, or adjust physical therapy routines based on sensor-driven progress [
285,
286]. Clinical integration will depend on interoperability standards, regulatory frameworks, and robust validation pipelines.
10.6. Cross-Cutting Challenges
Across all domains, ensuring equitable access, data security, and scalable manufacturing remains essential. Privacy-preserving computation, adaptive gamification for long-term engagement, and culturally aware AI design will be critical in ensuring wearable computing achieves its full potential without reinforcing health disparities.
11. Conclusions
Wearable computing is transitioning from a data collection paradigm to an active, precision-guided health orchestration system. This review has presented developments across sensing modalities, wireless protocols, computational frameworks, and AI-driven analytics that collectively shape the state of the art. Going beyond existing surveys, we linked advances in microfluidic sweat sensing, smart textiles, on-device AI acceleration, and context-aware multimodal fusion to emerging clinical and consumer use cases.
To guide future developments in wearable health monitoring systems, we propose the following roadmap prioritizing urgent gaps and priority areas:
Urgent Gaps
- -
Standardization of Benchmarks and Metrics: There is a critical need to define unified performance metrics and clinical validation protocols for comparing wearable systems across studies.
- -
Explainable AI (XAI) Integration: Making AI models interpretable is essential for clinical trust and regulatory compliance, especially in high-risk scenarios like cardiovascular anomaly detection or mental health diagnostics.
- -
Edge AI under Power Constraints: Development of low-power, high-performance AI accelerators for wearable devices is vital to support real-time analytics without relying on cloud offloading.
- -
Neuromorphic and analog AI processors: Enables complex analytics under wearable power constraints.
Priority Research Areas
- -
Smart Fabrics and Materials: Embedding sensors directly into textiles promises long-term, comfortable, and unobtrusive monitoring, but requires progress in durability, washability, and power autonomy.
- -
Personalization via Longitudinal and Multimodal Data: Leveraging continuous data to personalize baselines and thresholds can improve diagnostic accuracy and user adherence.
- -
Federated and Transfer Learning: These privacy-preserving techniques are crucial for scaling models across diverse populations while accounting for demographic, physiological, and behavioral heterogeneity.
- -
Closed-loop therapeutic wearables: Adapt interventions dynamically to physiological and psychological states.
This roadmap is intended to focus on the design of next-generation wearable systems that are clinically trustworthy, scalable, and aligned with the principles of personalized and precision medicine.
By grounding these insights in current research trajectories, we position wearable computing as a cornerstone of preventive, personalized, and participatory healthcare. Addressing the outlined technical and ethical challenges will ensure that these systems evolve into trusted, equitable, and clinically indispensable tools for mental and physical health management in the coming decade.