Mobile Robot Gas Source Localization Using SLAM-GDM with a Graphene-Based Gas Sensor
Abstract
1. Introduction
2. Research Methodology
2.1. Mobile Robot Setup
2.1.1. Hardware Architecture
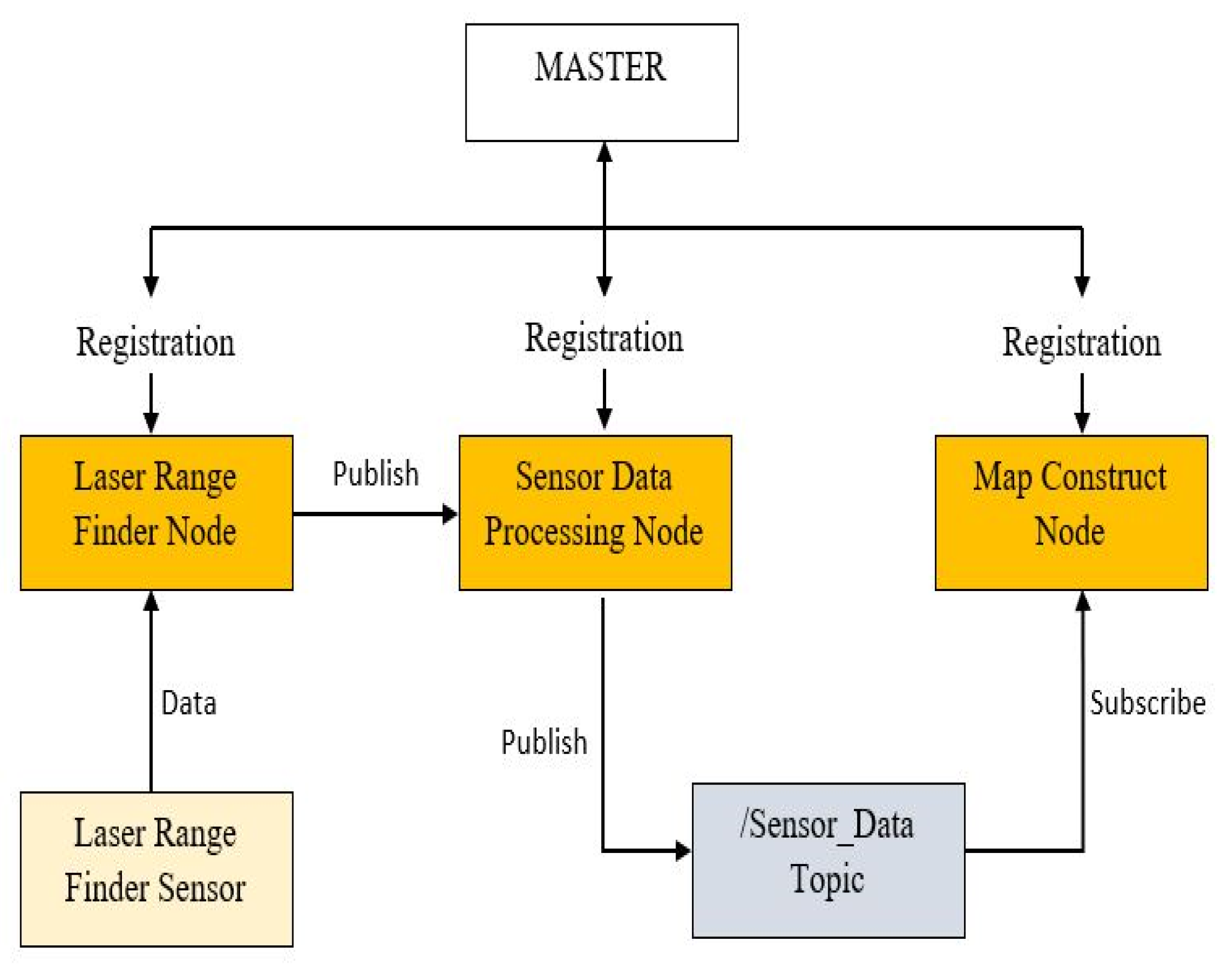
2.1.2. Software Architecture
2.2. Preparation of Reduced Graphene Oxide (RGO)
2.3. Simultaneous Localization and Mapping (SLAM)
2.4. Gas Distribution Mapping (GDM)
2.5. SLAM-GDM
3. Results and Discussion
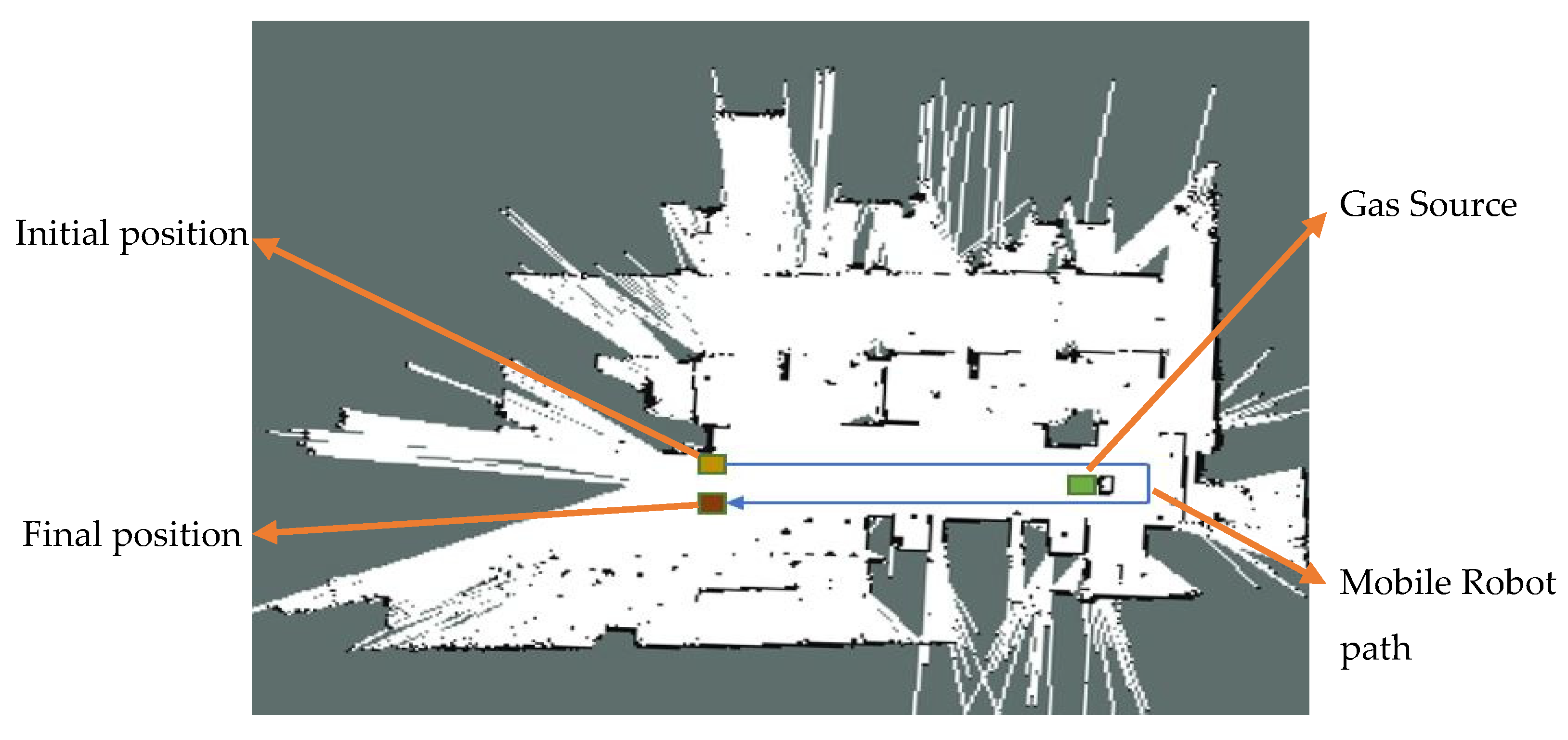
3.1. Experimental Setup
3.2. Experimental Results
3.2.1. Sensor Lab Testing
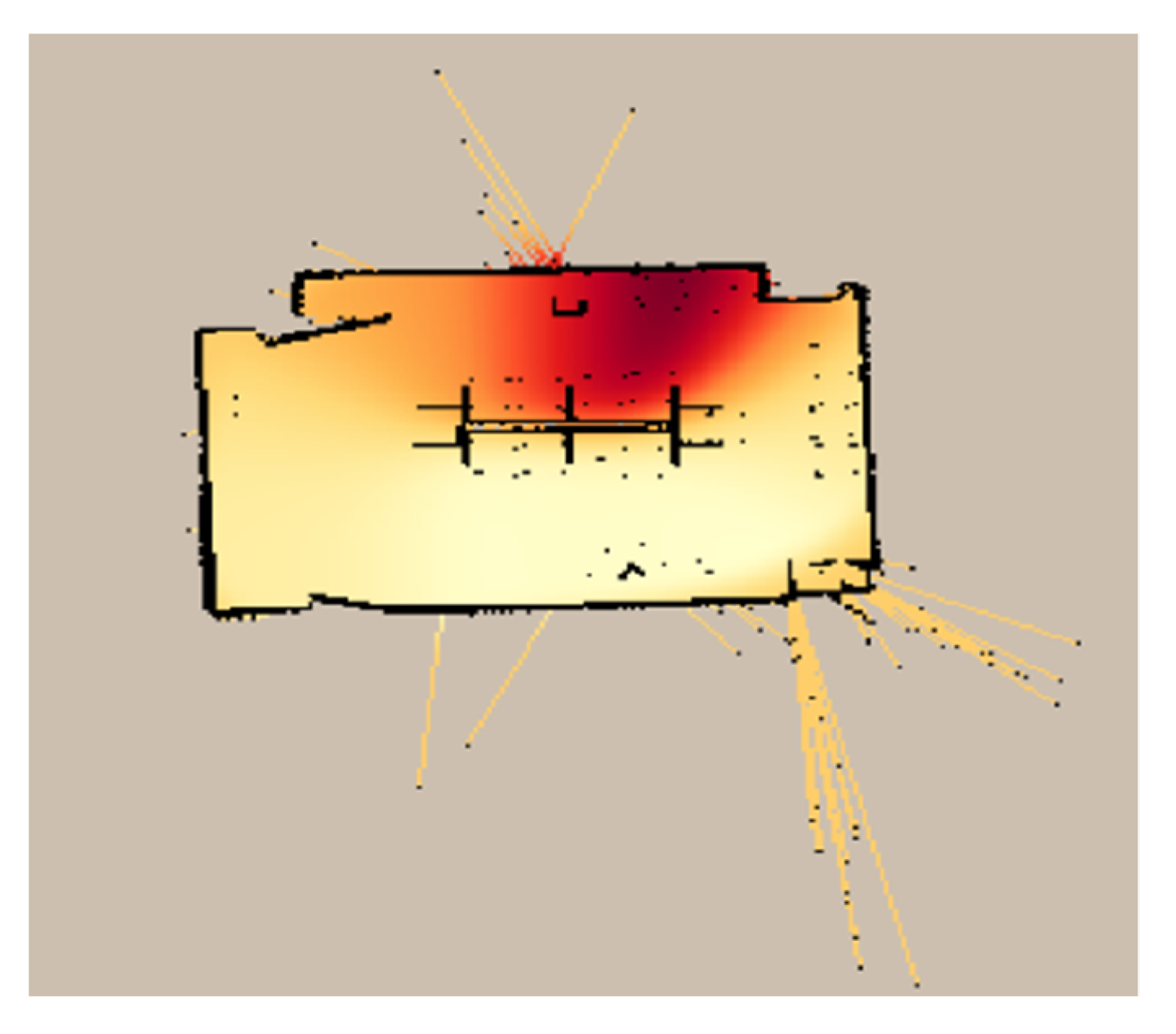
3.2.2. SLAM-GDM Test Results
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Örnek, Ö.; Karlık, B. An Overview of Metal Oxide Semiconducting Sensors in Electronic Nose Applications. In Proceedings of the 3rd International Symposium on Sustainable Development, Sarajevo, Bosnia and Herzegovina, 31 May–1 June 2012; Volume 2, pp. 506–515. [Google Scholar]
- Lee, E.B.; Hwang, I.S.; Cha, J.H.; Lee, H.J.; Lee, W.B.; Pak, J.J.; Lee, J.H.; Ju, B.K. Micromachined catalytic combustible hydrogen gas sensor. Sens. Actuators B Chem. 2011, 153, 392–397. [Google Scholar] [CrossRef]
- Neri, G. First fifty years of chemoresistive gas sensors. Chemosensors 2015, 3, 1–20. [Google Scholar] [CrossRef]
- Filipenko, M.; Afanasyev, I. Comparison of Various SLAM Systems for Mobile Robot in an Indoor Environment. In Proceedings of the 2018 International Conference on Intelligent Systems (IS), Funchal, Portugal, 25–27 September 2018; pp. 400–407. [Google Scholar]
- Alrammouz, R.; Podlecki, J.; Abboud, P.; Sorli, B.; Habchi, R. A review on flexible gas sensors: From materials to devices. Sens. Actuators A Phys. 2018, 284, 209–231. [Google Scholar] [CrossRef]
- Rossi, M.; Brunelli, D. Analyzing the transient response of MOX gas sensors to improve the lifetime of distributed sensing systems. In Proceedings of the 2013 5th IEEE International Workshop on Advances in Sensors and Interfaces (IWASI), Bari, Italy, 13–14 June 2013; pp. 211–216. [Google Scholar]
- Chutia, R.; Bhuyan, M. Study of temperature modulated tin oxide gas sensor and identification of chemicals. In Proceedings of the 2012 2nd National Conference on Computational Intelligence and Signal Processing (CISP), Guwahati, India, 2–3 March 2012; pp. 181–184. [Google Scholar]
- Loutfi, A.; Coradeschi, S.; Lilienthal, A.J.; Gonzalez, J. Gas distribution mapping of multiple odour sources using a mobile robot. Robotica 2009, 27, 311–319. [Google Scholar] [CrossRef]
- Palacín, J.; Martínez, D.; Clotet, E.; Pallejà, T.; Burgués, J.; Fonollosa, J.; Pardo, A.; Marco, S. Application of an Array of Metal-Oxide Semiconductor Gas Sensors in an Assistant Personal Robot for Early Gas Leak Detection. Sensors 2019, 19, 1957. [Google Scholar] [CrossRef] [PubMed]
- Vincent, T.A.; Xing, Y.; Cole, M.; Gardner, J.W. Investigation of the response of high-bandwidth MOX sensors to gas plumes for application on a mobile robot in hazardous environments. Sens. Actuators B Chem. 2018, 279, 351–360. [Google Scholar] [CrossRef]
- Lilienthal, A.; Ulmer, H.; Frohlich, H.; Stutzle, A.; Werner, F.; Zell, A. Gas source declaration with a mobile robot. In Proceedings of the IEEE International Conference on Robotics and Automation (ICRA), New Orleans, LA, USA, 26 April–1 May 2004; Volume 2, pp. 1430–1435. [Google Scholar]
- Shooshtari, M.; Salehi, A.; Vollebregt, S. Effect of temperature and humidity on the sensing performance of TiO2 nanowire-based ethanol vapor sensors. Nanotechnology 2021, 32, 325501. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.D.; Rothschild, A.; Lee, B.H.; Kim, D.Y.; Jo, S.M.; Tuller, H.L. Ultrasensitive chemiresistors based on electrospun TiO2 nanofibers. Nano Lett. 2006, 6, 2009–2013. [Google Scholar] [CrossRef] [PubMed]
- Gessner, T.; Gottfried, K.; Hoffmann, R.; Kaufmann, C.; Weiss, U.; Charetdinov, E.; Hauptmann, P.; Lucklum, R.; Zimmermann, B.; Dietel, U.; et al. Metal oxide gas sensor for high temperature application. Microsyst. Technol. 2000, 6, 169–174. [Google Scholar] [CrossRef]
- Korotcenkov, G.; Cho, B.K. Metal oxide composites in conductometric gas sensors: Achievements and challenges. Sens. Actuators B Chem. 2017, 244, 182–210. [Google Scholar] [CrossRef]
- Anderson, M.J.; Sullivan, J.G.; Horiuchi, T.K.; Fuller, S.B.; Daniel, T.L. A bio-hybrid odor-guided autonomous palm-sized air vehicle. Bioinspiration Biomim. 2020, 16, 026002. [Google Scholar] [CrossRef] [PubMed]
- Terutsuki, D.; Uchida, T.; Fukui, C.; Sukekawa, Y.; Okamoto, Y.; Kanzaki, R. Electroantennography-based bio-hybrid odor-detecting drone using silkmoth antennae for odor source localization. J. Vis. Exp. 2021, 174, e62895. [Google Scholar] [CrossRef] [PubMed]
- Terutsuki, D.; Uchida, T.; Fukui, C.; Sukekawa, Y.; Okamoto, Y.; Kanzaki, R. Real-time odor concentration and direction recognition for efficient odor source localization using a small bio-hybrid drone. Sens. Actuators B Chem. 2021, 339, 129770. [Google Scholar] [CrossRef]
- Yuan, W.; Shi, G. Graphene-Based Gas Sensors. Mater. Chem. A 2013, 1, 10078–10091. [Google Scholar] [CrossRef]
- Priyandoko, G.; Ming, T.Y.; Achmad, M.S.H. Mapping of unknown industrial plant using ROS-based navigation mobile robot. In Proceedings of the IOP Conference Series: Material Science and Engineering, Pahang, Malaysia, 1–2 August 2017; IOP Publishing: Philadelphia, PA, USA, 2017; Volume 257, p. 012088. [Google Scholar]
- Gupta, M.; Hawari, H.F.; Kumar, P.; Burhanudin, Z.A.; Tansu, N. Functionalized Reduced Graphene Oxide Thin Films for Ultrahigh CO2 Gas Sensing Performance at Room Temperature. Nanomaterials 2021, 11, 623. [Google Scholar] [CrossRef] [PubMed]
- Gupta, M.; Hawari, H.F.; Kumar, P.; Burhanudin, Z.A. Copper Oxide/Functionalized Graphene Hybrid Nanostructures for Room Temperature Gas Sensing Applications. Crystals 2022, 12, 264. [Google Scholar] [CrossRef]
- Lee, Z.Y.; Hawari, H.F.; Djaswadi, G.W.B.; Kamarudin, K. A Highly Sensitive Room Temperature CO2 Gas Sensor Based on SnO2-rGO Hybrid Composite. Materials 2021, 14, 522. [Google Scholar] [CrossRef] [PubMed]
- Hussein, N.A.A.; Hawari, H.F.; Wong, Y.H. Synthesis of Iron Oxide/Polyaniline/Reduced Graphene Oxide Nanocomposite Materials as Active Sensing Material. In Proceedings of the International Conference on Intelligent and Advanced Systems: Enhance the Present for a Sustainable Future (ICIAS), Kuching, Malaysia, 13–15 July 2021; pp. 1–5. [Google Scholar]
- Noda, K.; Aizawa, H. Indoor environmental monitoring system using a robot vacuum cleaner. Sens. Mater. 2020, 32, 1133–1140. [Google Scholar] [CrossRef]
- Kohlbrecher, S.; Von Stryk, O.; Meyer, J.; Klingauf, U. A flexible and scalable SLAM system with full 3D motion estimation. In Proceedings of the 9th IEEE International Symposium on Safety, Security, and Rescue Robotics (SSRR), Kyoto, Japan, 1–5 November 2011; pp. 155–160. [Google Scholar]
- Eliwa, M.; Adham, A.; Sami, I.; Eldeeb, M. A critical comparison between Fast and Hector SLAM algorithms. REST J. Emerg. Trends Model. Manuf. 2017, 3, 44–49. [Google Scholar]
- Xuexi, Z.; Guokun, L.; Genping, F.; Dongliang, X.; Shiliu, L. SLAM algorithm analysis of mobile robot based on lidar. In Proceedings of the Chinese Control Conference (CCC), Guangzhou, China, 27–30 July 2019; pp. 4739–4745. [Google Scholar]
- Lilienthal, A.; Duckett, T. Gas Source Localisation by Constructing Concentration Gridmaps with a Mobile Robot. In Proceedings of the European Conference on Mobile Robots (ECMR), Warsaw, Poland, 4–6 September 2003. [Google Scholar]
- Kamarudin, K.; Md Shakaff, A.Y.; Bennetts, V.H.; Mamduh, S.M.; Zakaria, A.; Visvanathan, R.; Ali Yeon, A.S.; Kamarudin, L.M. Integrating SLAM and gas distribution mapping (SLAM-GDM) for real-time gas source localization. Adv. Robot. 2018, 32, 903–917. [Google Scholar] [CrossRef]
- Norzam, W.A.S.; Hawari, H.F.; Kamarudin, K. Analysis of Mobile Robot Indoor Mapping using GMapping Based SLAM with Different Parameter. In IOP Conference Series: Materials Science and Engineering, Pulau Pinang, Malaysia, 26–27 August 2019; IOP Publishing: Philadelphia, PA, USA, 2019. [Google Scholar]



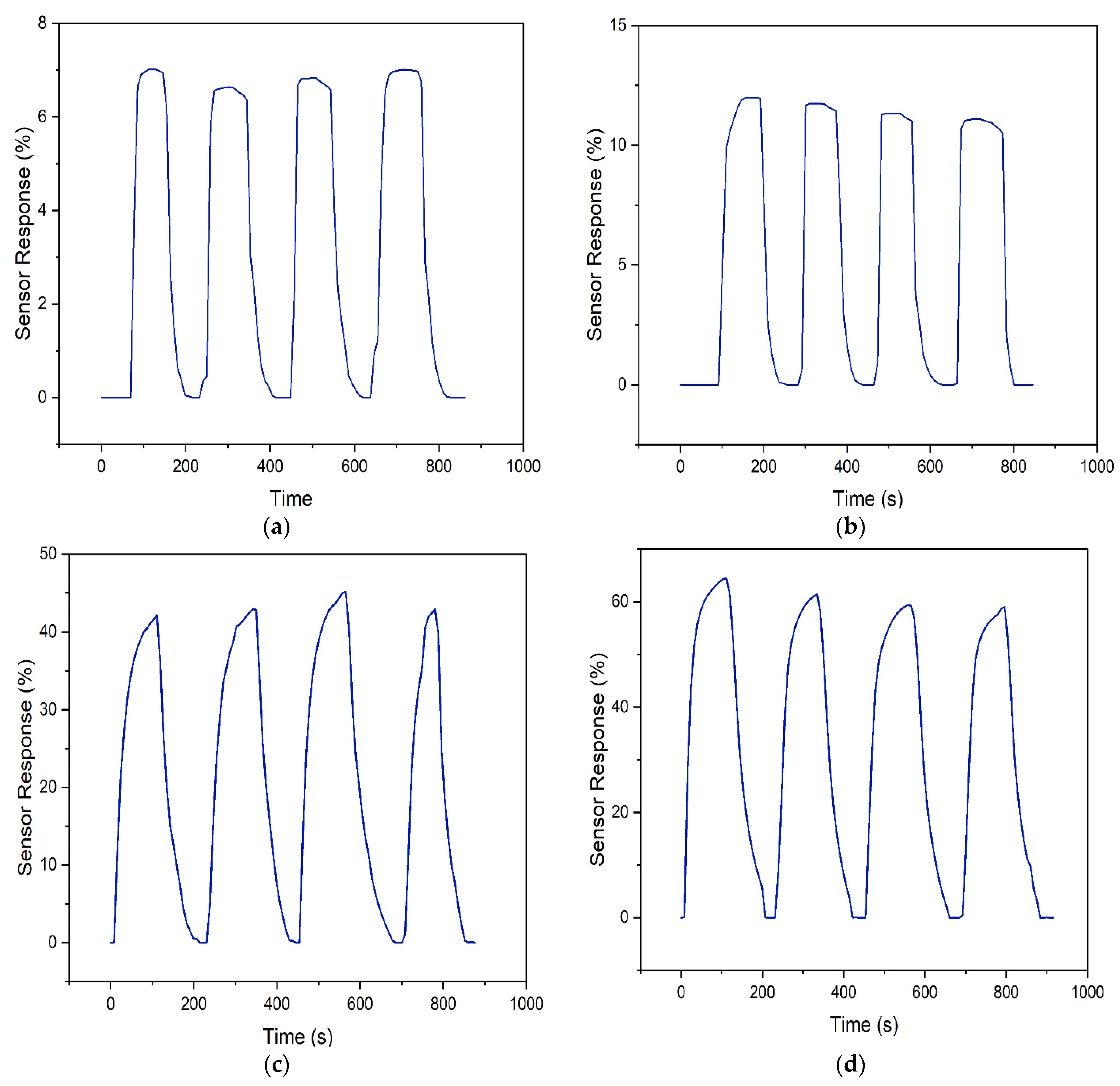
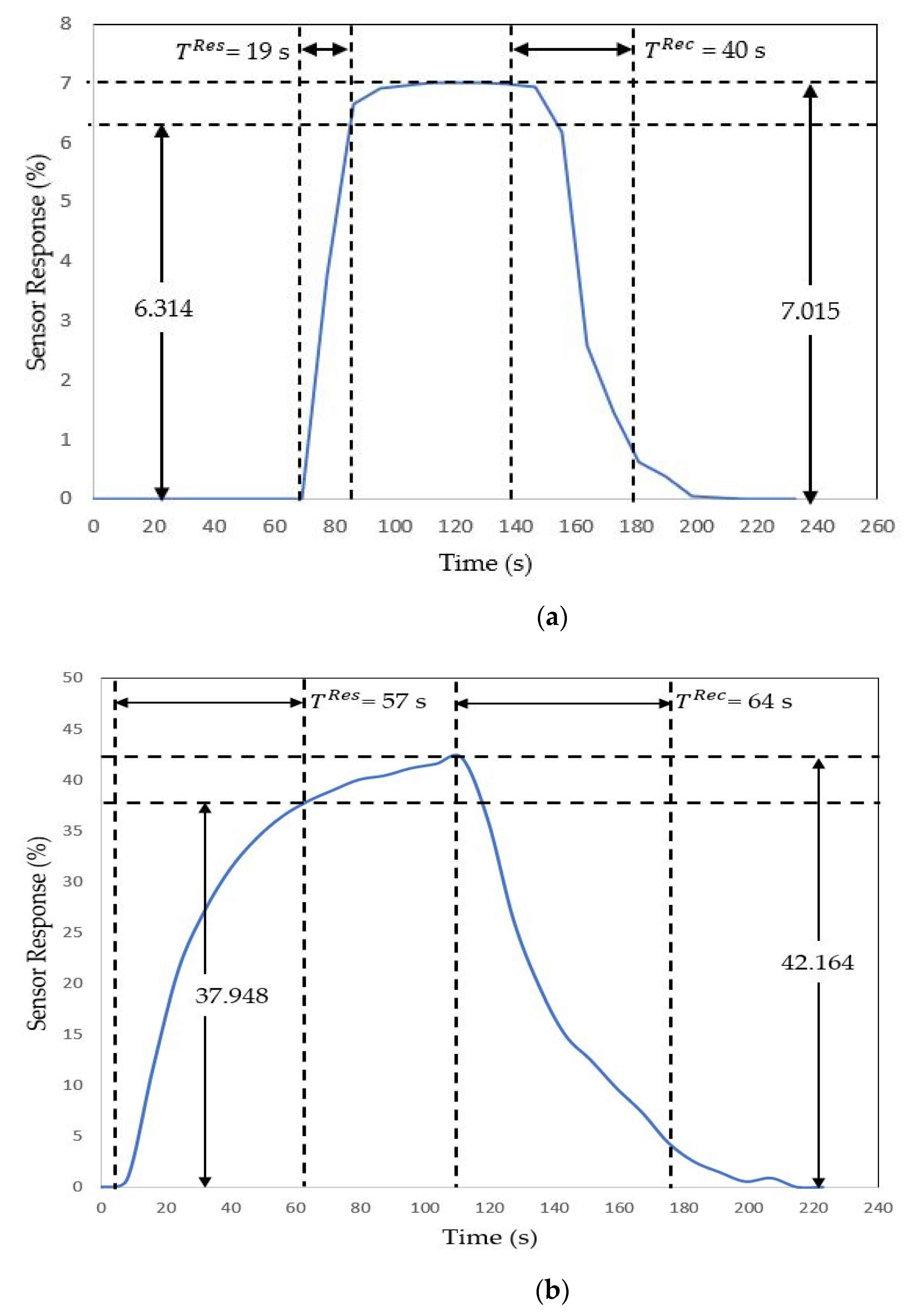
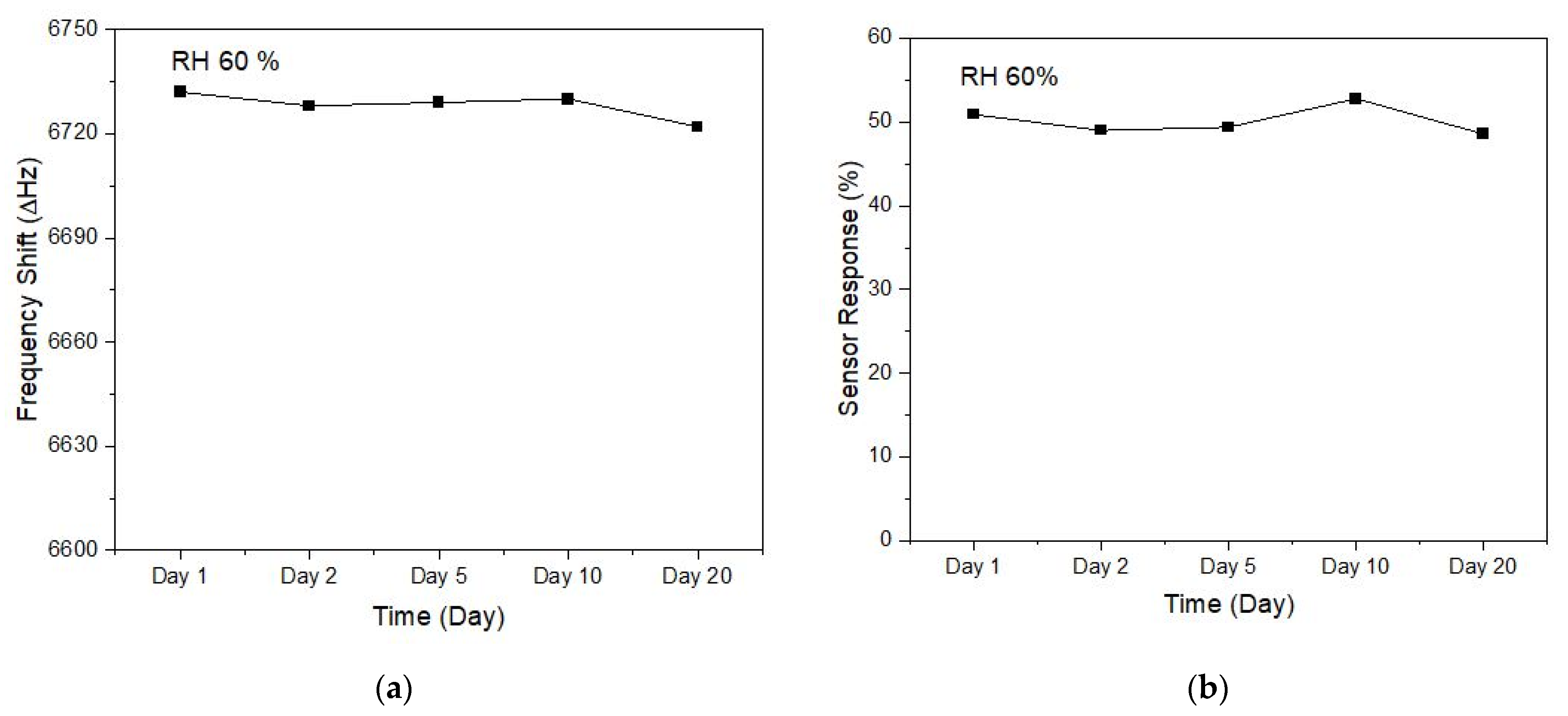
| Sensor | Trial | 100 ppm | 300 ppm | ||
|---|---|---|---|---|---|
| Response Time (s) | Recovery Time (s) | Response Time (s) | Recovery Time (s) | ||
| RGO | 1 | 19 | 40 | 28 | 28 |
| 2 | 25 | 43 | 17 | 29 | |
| 3 | 18 | 50 | 17 | 36 | |
| 4 | 32 | 43 | 18 | 25 | |
| MOX | 1 | 57 | 64 | 37 | 85 |
| 2 | 63 | 61 | 50 | 72 | |
| 3 | 51 | 79 | 49 | 74 | |
| 4 | 45 | 59 | 40 | 70 | |
| Duration of Gas Spread (min) | Gas Sensor | Legends | |
|---|---|---|---|
| MOX | RGO | ||
| 7 | 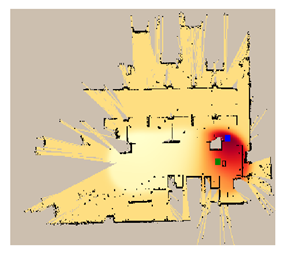 | 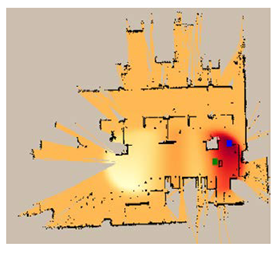 |  |
| 12 | 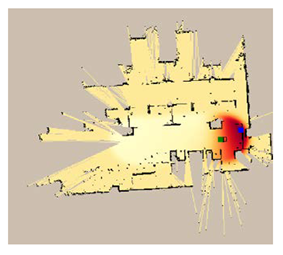 | 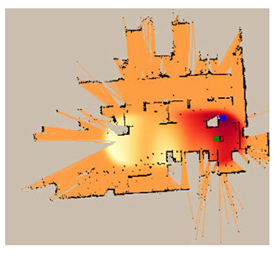 | |
| Trial | Distance Error (cm) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | Average | |||||||
| Duration of gas spread (min) | 7 | 12 | 7 | 12 | 7 | 12 | 7 | 12 | 7 | 12 | 7 | 12 |
| MOX | 31.9 | 30.5 | 5.4 | 31.8 | 28.3 | 27.3 | 26.9 | 30.2 | 5.1 | 31.6 | 19.52 | 30.28 |
| RGO | 6.3 | 33.2 | 27.7 | 28.5 | 35.4 | 26.2 | 28.0 | 33.2 | 28.8 | 31.9 | 25.24 | 30.60 |
| Duration of Gas Source Spread (min) | Gas Sensor | |
|---|---|---|
| MOX | RGO | |
| 7 |  |  |
| 12 |  |  |
| Trial | Duration of Gas Source Spread (min) | MOX | RGO | Differences (%) |
|---|---|---|---|---|
| 1 | 7 | 24,321 | 26,530 | 8.33 |
| 12 | 25,326 | 25,477 | 0.59 | |
| 2 | 7 | 24,621 | 24,708 | 0.35 |
| 12 | 24,628 | 26,467 | 6.95 | |
| 3 | 7 | 25,377 | 24,919 | 1.80 |
| 12 | 25,139 | 25,736 | 2.32 | |
| 4 | 7 | 24,716 | 26,124 | 5.39 |
| 12 | 24,442 | 25,323 | 3.48 | |
| 5 | 7 | 24,410 | 24,695 | 1.15 |
| 12 | 24,623 | 25,045 | 1.68 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Norzam, W.A.S.; Hawari, H.F.; Kamarudin, K.; Juffry, Z.H.M.; Hussein, N.A.A.; Gupta, M.; Abdullah, A.N. Mobile Robot Gas Source Localization Using SLAM-GDM with a Graphene-Based Gas Sensor. Electronics 2023, 12, 171. https://doi.org/10.3390/electronics12010171
Norzam WAS, Hawari HF, Kamarudin K, Juffry ZHM, Hussein NAA, Gupta M, Abdullah AN. Mobile Robot Gas Source Localization Using SLAM-GDM with a Graphene-Based Gas Sensor. Electronics. 2023; 12(1):171. https://doi.org/10.3390/electronics12010171
Chicago/Turabian StyleNorzam, Wan Abdul Syaqur, Huzein Fahmi Hawari, Kamarulzaman Kamarudin, Zaffry Hadi Mohd Juffry, Nurul Athirah Abu Hussein, Monika Gupta, and Abdulnasser Nabil Abdullah. 2023. "Mobile Robot Gas Source Localization Using SLAM-GDM with a Graphene-Based Gas Sensor" Electronics 12, no. 1: 171. https://doi.org/10.3390/electronics12010171
APA StyleNorzam, W. A. S., Hawari, H. F., Kamarudin, K., Juffry, Z. H. M., Hussein, N. A. A., Gupta, M., & Abdullah, A. N. (2023). Mobile Robot Gas Source Localization Using SLAM-GDM with a Graphene-Based Gas Sensor. Electronics, 12(1), 171. https://doi.org/10.3390/electronics12010171






