Effect of Addition of PVA/PG to Oil-in-Water Nanoemulsion Kojic Monooleate Formulation on Droplet Size: Three-Factors Response Surface Optimization and Characterization
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Selection of the Polymer
2.3. Selection of Solvent
2.4. Formulation of Kojic Monooleate Nanoemulsion Formulation in Thin Film System (TFS)
2.5. Optimization Using the Response Surface Methodology (RSM)
2.5.1. Experimental Design
2.5.2. Statistical Analysis
2.5.3. Verification of Models
2.6. Characterization of the Thin Film System (TFS)
2.6.1. Film Formation
2.6.2. Drying Time
2.7. Physicochemical Characterization
2.7.1. Viscosity
2.7.2. Droplet Size
2.7.3. Transmission Electron Microscopy (TEM)
2.7.4. pH
2.7.5. Stability Study
3. Results and Discussion
3.1. Combination of Polymer and Solvent to Develop KMO Formulation in TFS
3.2. Optimization Using the Response Surface Methodology (RSM)
3.3. Characterization of the Thin Film System (TFS)
3.3.1. Film Formation
3.3.2. Drying Time
3.4. Physicochemical Characterization
3.4.1. Viscosity
3.4.2. Particle Size, polydispersity index (PDI), and Zeta Potential
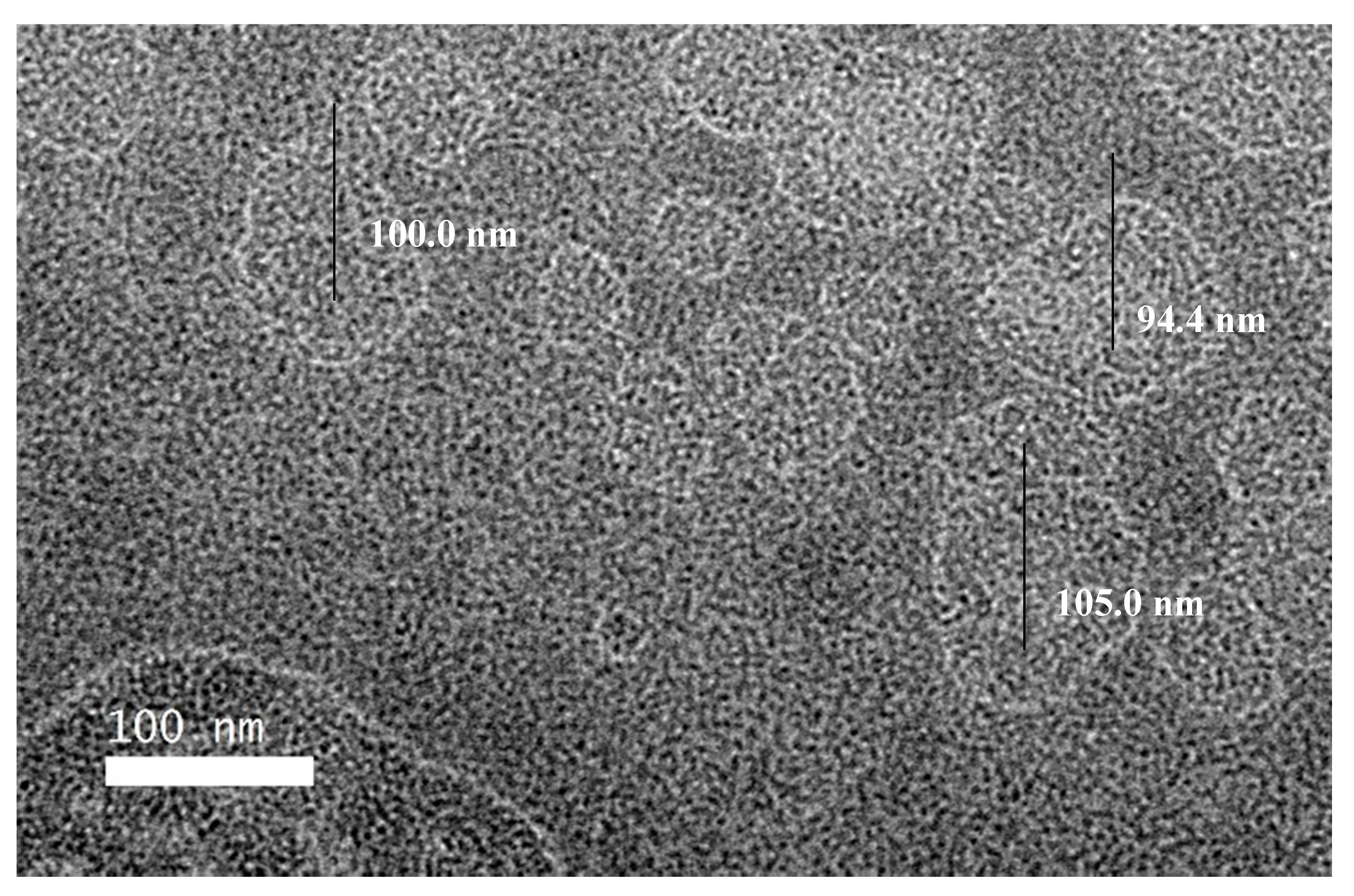
3.4.3. Transmission Electron Microscopy (TEM)
3.4.4. pH and Conductivity
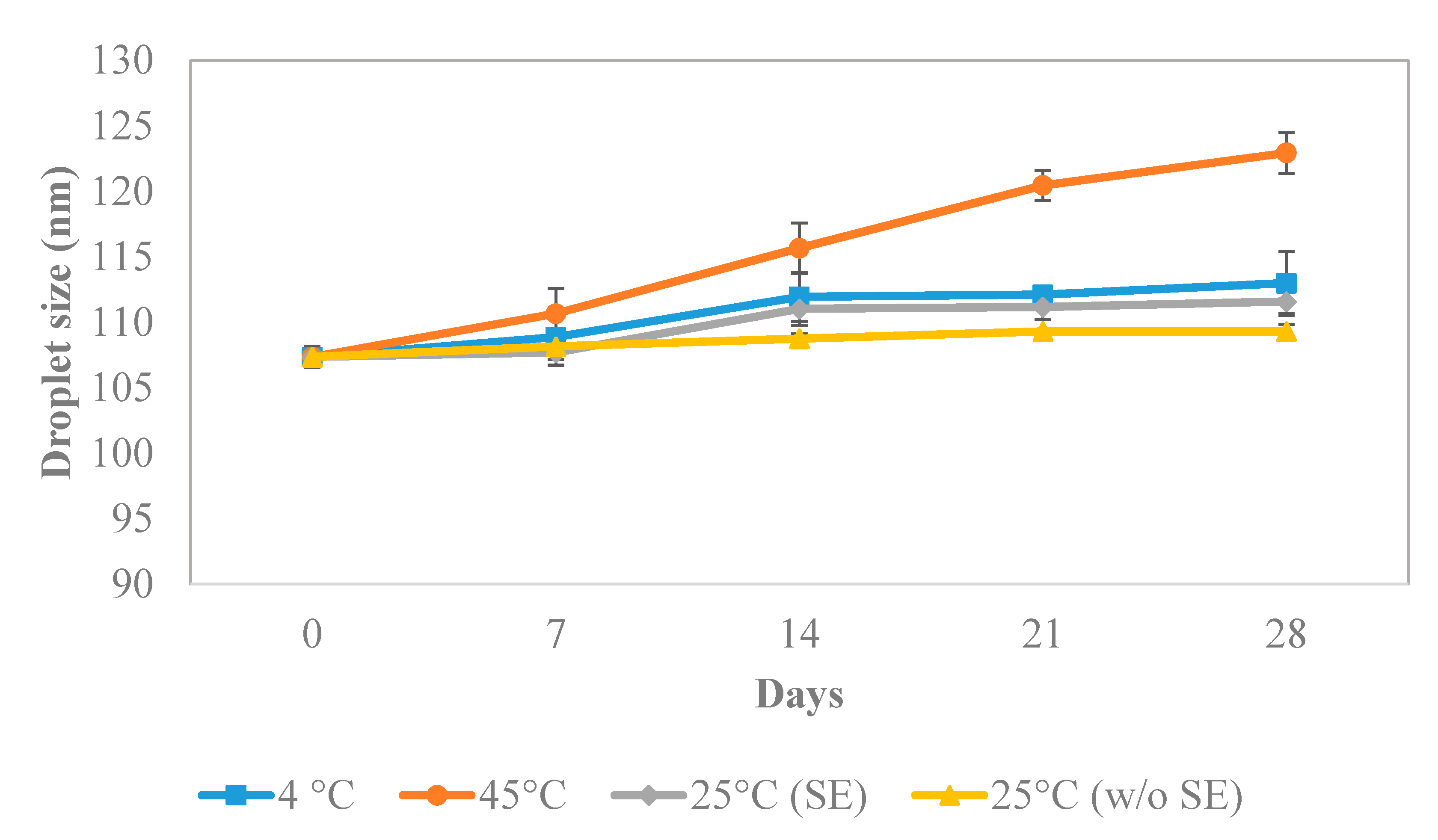
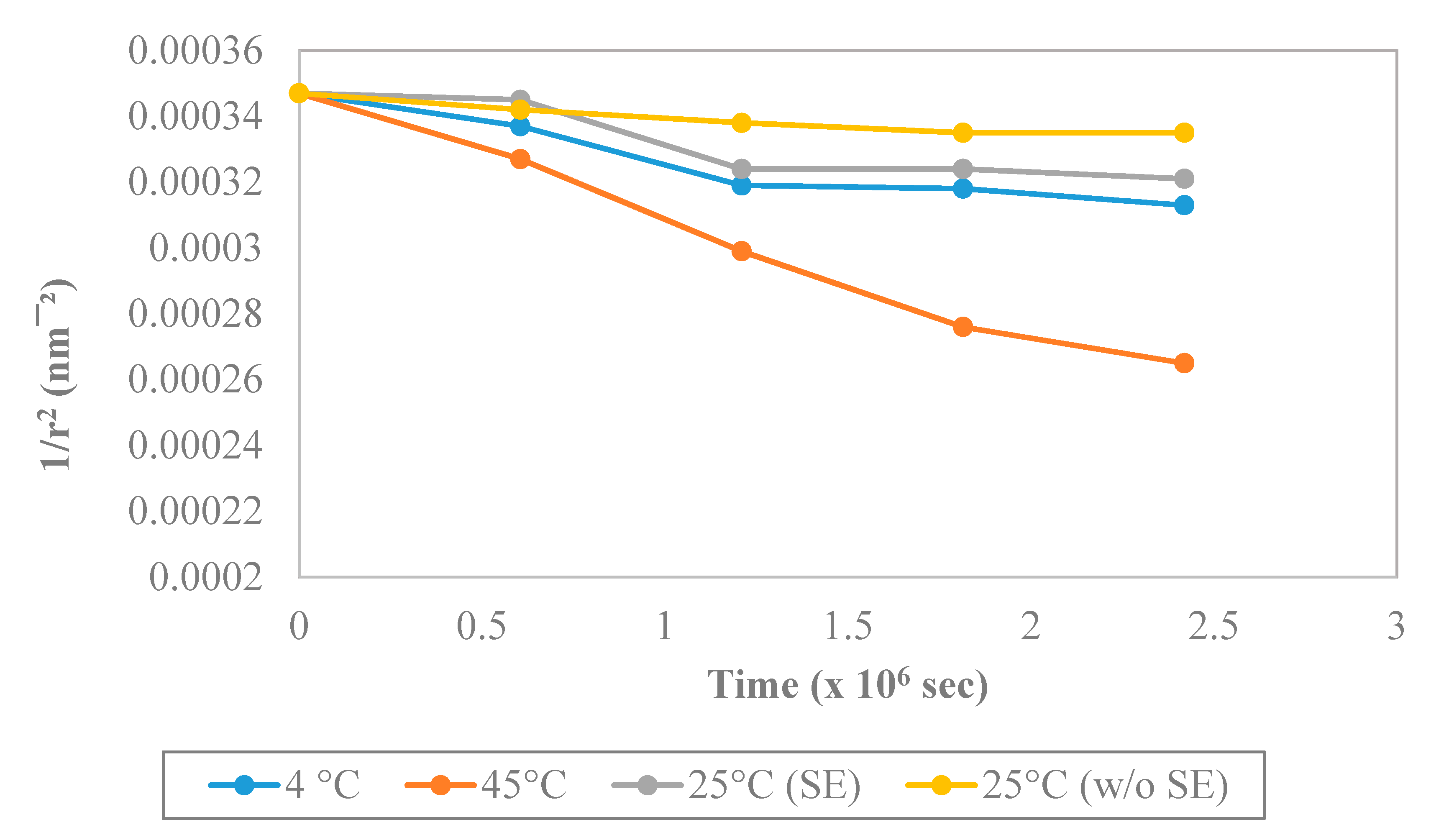
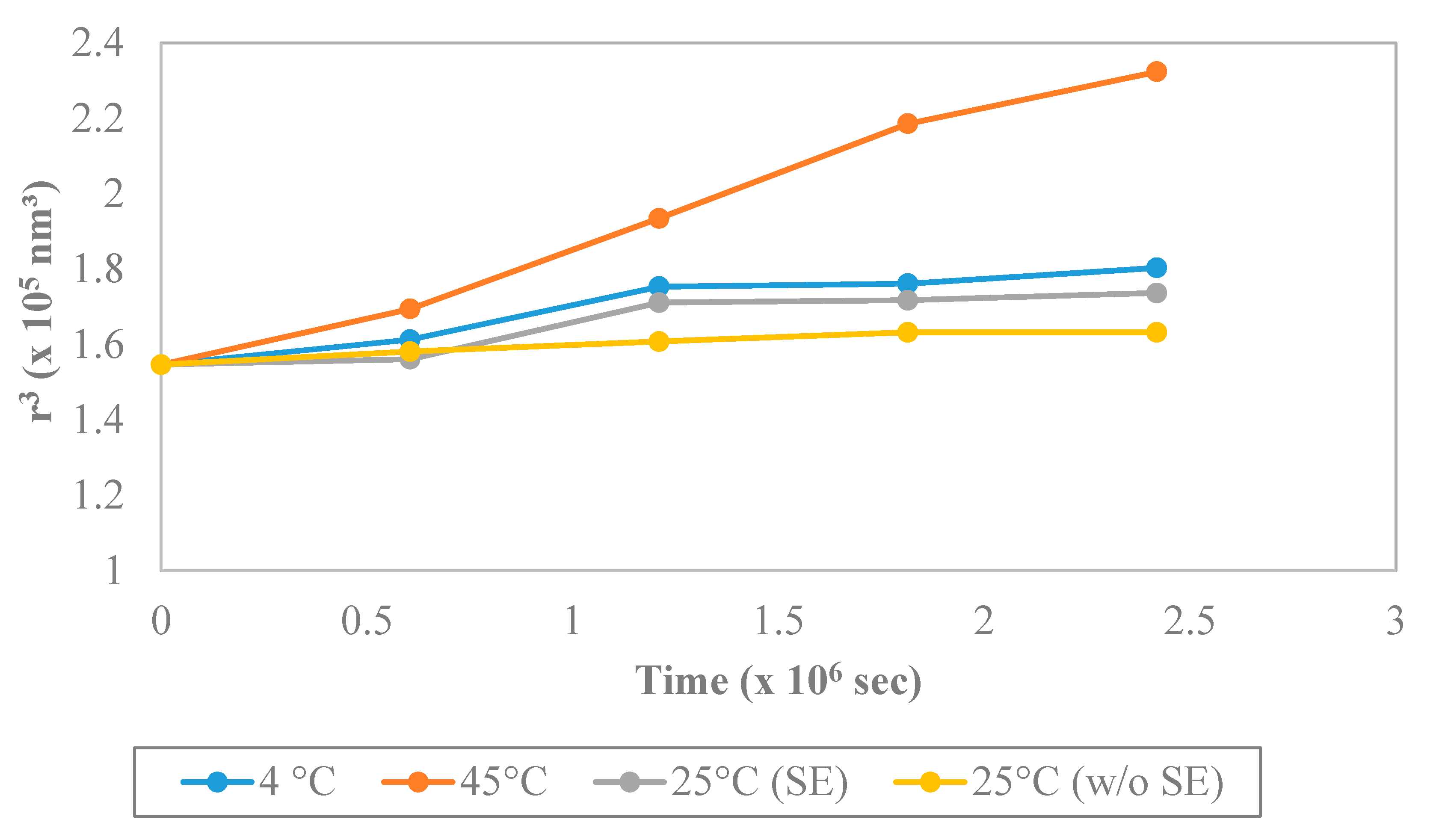
3.4.5. Stability Study
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ashari, S.E.; Mohamad, R.; Ariff, A.; Basri, M.; Salleh, A.B. Optimization of enzymatic synthesis of palm-based kojic acid ester using response surface methodology. J. Oleo Sci. 2009, 58, 503–510. [Google Scholar] [CrossRef] [PubMed]
- Lajis, A.F.B.; Hamid, M.; Ariff, A.B. Depigmenting effect of kojic acid esters in hyperpigmented B16F1 melanoma cells. BioMed. Res. Int. 2012, 2012, 9. [Google Scholar] [CrossRef] [PubMed]
- Yukuyama, M.N.; Ghisleni, D.D.M.; Pinto, T.D.J.A.; Bou-Chacra, N.A. Nanoemulsion: Process selection and application in cosmetics—A review. Int. J. Cosm. 2016, 38, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Bishnoi, R.S.; Shukla, A.K.; Jain, C.P. Techniques for Formulation of Nanoemulsion Drug Delivery System: A Review. Prev. Nutr. Food Sci. 2019, 24, 225–234. [Google Scholar] [CrossRef]
- Nikam, T.H.; Patil, M.P.; Patil, S.S.; Vadnere, G.P.; Lodhi, S. Nanoemulsion: A brief review on development and application in Parenteral Drug Delivery. Adv. Pharm. J. 2018, 3, 43–54. [Google Scholar] [CrossRef]
- Jaiswal, M.; Dudhe, R.; Sharma, P.K. Nanoemulsion: An advanced mode of drug delivery system. 3 Biotech 2015, 5, 123–127. [Google Scholar] [CrossRef]
- Gutiérrez, J.M.; González, C.; Maestro, A.; Solè, I.M.; Pey, C.M.; Nolla, J. Nano-emulsions: New applications and optimisation of their preparation. Curr. Opi. Col. Interface Sci. 2008, 13, 245–251. [Google Scholar] [CrossRef]
- Karki, S.; Kim, H.; Na, S.-J.; Shin, D.; Jo, K.; Lee, J. Thin films as an emerging platform for drug delivery. Asian J. Pharm. Sci. 2016, 11, 559–574. [Google Scholar] [CrossRef]
- Frederiksen, K.; Guy, R.H.; Petersson, K. Formulation considerations in the design of topical, polymeric film-forming systems for sustained drug delivery to the skin. Eur. J. Pharm. Biopharm. 2015, 91, 9–15. [Google Scholar] [CrossRef]
- Kathe, K.; Kathpalia, H. Film forming systems for topical and transdermal drug delivery. Asian J. Pharm. Sci. 2017, 12, 487–497. [Google Scholar] [CrossRef]
- Budiman, A.; Aulifa, D.L.; Kusuma, A.S.W.; Kurniawan, I.S.; Sulastri, A. Peel-off gel formulation from black mulberries (Morus nigra) extract as anti-acne mask. Nat. J. Physiol. Pharm. Pharmacol. 2017, 7, 987–994. [Google Scholar] [CrossRef]
- Velasco, M.V.R.; Vieira, R.P.; Fernandes, A.R.; Dario, M.F.; Pinto, C.A.S.D.O.; Pedriali, C.A.; Kaneko, T.M.; Baby, A.R. Short-term clinical of peel-off facial mask moisturisers. Int. J. Cosm. Sci. 2014, 36, 355–360. [Google Scholar] [CrossRef] [PubMed]
- Lunter, D.; Daniels, R. In vitro Skin Permeation and Penetration of Nonivamide from Novel Film-Forming Emulsions. Ski. Pharmacol. Physiol. 2013, 26, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Aydar, A. Statistical Methods in Optimisation of Food Materials. Eur. Int. J. Sci. Tech. 2019, 8, 33–40. [Google Scholar]
- Latha, S.; Sivaranjani, G.; Dhanasekaran, D. Response surface methodology: A non-conventional statistical tool to maximise the throughput of Streptomyces species biomass and their bioactive metabolites. Crit. Rev. Microbiol. 2017, 43, 567–582. [Google Scholar] [CrossRef] [PubMed]
- Ong, S.T.; Ong, S.T.; Hung, Y.T.; Phung, Y.P. Application of conventional and statistical experimental methodology to optimise malachite green dye removal from aqueous solutions. Desalin. Water Treat. 2015, 55, 1359–1371. [Google Scholar]
- Habib, S.; Ahmad, S.A.; Johari, W.L.W.; Shukor, M.Y.A.; Alias, S.A.; Khalil, K.A.; Yasid, N.A. Evaluation of conventional and response surface level optimisation of n-dodecane (n-C12) mineralisation by psychrotolerant strains isolated from pristine soil at Southern Victoria Island, Antarctica. Microbial. Cell Fact. 2018, 17, 44. [Google Scholar] [CrossRef]
- Singh, S.K.; Singh, S.K.; Tripathi, V.R.; Khare, S.K.; Garg, S.K. Comparative one-factor-at-a-time, response surface (statistical) and bench-scale bioreactor level optimisation of thermoalkaline protease production from a psychrotrophic Pseudomonas putida SKG-1 isolate. Microbial. Cell Fact. 2011, 10, 114. [Google Scholar] [CrossRef]
- Galooyak, S.S.; Dabir, B. Three-factor response surface optimization of nano-emulsion formation using a microfluidizer. J. Food Sci. Technol. 2015, 52, 2558–2571. [Google Scholar] [CrossRef]
- Khan, M.J.H.; Hussain, M.A.; Mujtaba, I.M. Process parameters optimization for polypropylene production in a pilot scale fluidized bed catalytic reactor. In Proceedings of the Chemeca 2014: Processing Excellence; Powering Our Future, Perth, Australia, 28 September–1 October 2014; pp. 1393–1401. [Google Scholar]
- Beringhs, A.O.; Rosa, J.M.; Stulzer, H.K.; Budal, R.M.; Sonaglio, D. Green Clay and Aloe Vera Peel-Off Facial Masks: Response Surface Methodology Applied to the Formulation Design. AAPS PharmSciTech 2013, 14, 445–455. [Google Scholar] [CrossRef]
- Azhar, S.N.A.S.; Ashari, S.; Salim, N. Development of a kojic monooleate-enriched oil-in-water nanoemulsion as a potential carrier for hyperpigmentation treatment. Int. J. Nanomed. 2018, 13, 6465–6479. [Google Scholar] [CrossRef] [PubMed]
- Beringhs, A.O.; Dalmina, M.; Creczynski-Pasa, T.B.; Sonaglio, D. Response Surface Methodology IV-Optimal design applied to the performance improvement of an RP-HPLC-UV method for the quantification of phenolic acids in Cecropia glaziovii products. Rev. Bras. Farm. 2015, 25, 513–521. [Google Scholar] [CrossRef]
- Ganz, M. Formulation and Evaluation of Hydrous and Anhydrous Skin Whitening Products Containing Sodium Ascorbyl pHospHate and Kojic Acid Dipalmitate. Doctoral Thesis, North-West University, Xian, Shaanxi, 2006. [Google Scholar]
- Kulkarni, S.; Bairagee, D.; Choudhary, N. Formulation and Evaluation of activated charcoal peel off mask. Int. J. Phytopharm. Res. 2018, 9, 40–44. [Google Scholar]
- Vieira, R.; Fernandes, A.R.; Kaneko, T.M.; Consiglieri, V.; Pinto, C.A.S.D.O.; Pereira, C.S.C.; Baby, A.; Velasco, M.R. Physical and physicochemical stability evaluation of cosmetic formulations containing soybean extract fermented by Bifidobacterium animalis. Braz. J. Pharm. Sci. 2009, 45, 515–525. [Google Scholar] [CrossRef][Green Version]
- Thottoli, A.K.; Achuthanunni, A.K. Effect of polyvinyl alcohol concentration on the ZnS nanoparticles and wet chemical synthesis of wurtzite ZnS nanoparticles. J. Nanostruct. Chem. 2013, 3, 31. [Google Scholar] [CrossRef]
- Krishna, A.; Kumar, A.; Singh, R.K. Effect of Polyvinyl Alcohol on the Growth, Structure, Morphology, and Electrical Conductivity of Polypyrrole Nanoparticles Synthesized via Microemulsion Polymerization. ISRN Nanomater. 2012, 2012, 1–6. [Google Scholar] [CrossRef]
- Oh, D.H.; Balakrishnan, P.; Oh, Y.-K.; Kim, D.-D.; Yong, C.S.; Choi, H.-G. Effect of process parameters on nanoemulsion droplet size and distribution in SPG membrane emulsification. Int. J. Pharm. 2011, 404, 191–197. [Google Scholar] [CrossRef]
- Lyoo, W.S.; Cho, S.H.; Shin, D.S.; Han, S.S.; Park, C.S.; Kim, J.H.; Noh, S.K. Effect of processing parameters on the size and size distribution of high molecular weight atactic poly (vinyl alcohol) gel particles prepared by water-in-oil emulsion method using high pressure homogeniser. Polym. Polym. Compos. 2006, 14, 47–53. [Google Scholar] [CrossRef]
- Safiah Mohamad, J.A.; Rozida, M.K.; Rizafizah, O. Coconut Oil Based Microemulsion Formulations for Hair Care Product Application. Sains Malays. 2019, 48, 599–605. [Google Scholar] [CrossRef]
- Saberi, A.H.; Fang, Y.; McClements, D.J. Fabrication of vitamin E-enriched nanoemulsions by spontaneous emulsification: Effect of propylene glycol and ethanol on formation, stability, and properties. Food Res. Int. 2013, 54, 812–820. [Google Scholar] [CrossRef]
- Suárez, M.J.; Lévy, H.; Lang, J. Effect of addition of polymer to water-in-oil microemulsions on droplet size and exchange of material between droplets. J. Phys. Chem. 1993, 97, 9808–9816. [Google Scholar] [CrossRef]
- Marzuki, N.H.C.; Wahab, R.A.; Hamid, M.A. An overview of nanoemulsion: Concepts of development and cosmeceutical applications. Biotechnol. Biotechnol. Equip. 2019, 33, 779–797. [Google Scholar] [CrossRef]
- Martínez-Pla, J.J.; Martín-Biosca, Y.; Sagrado, S.; Villanueva-Camañas, R.M.; Medina-Hernández, M.J. Evaluation of the pH effect of formulations on the skin permeability of drugs by biopartitioning micellar chromatography. J. Chromatogr. A. 2004, 1047, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.; Yosipovitch, G. Skin pH: From Basic SciencE to Basic Skin Care. Acta Derm. Venereol. 2013, 93, 261–267. [Google Scholar] [CrossRef]
- Mohamed, A.I.; Sultan, A.S.; Hussein, I.A.; Al-Muntasheri, G.A. Influence of surfactant structure on the stability of water-in-oil emulsions under high-temperature high-salinity conditions. J. Chem. 2017, 2017, 1–11. [Google Scholar] [CrossRef]
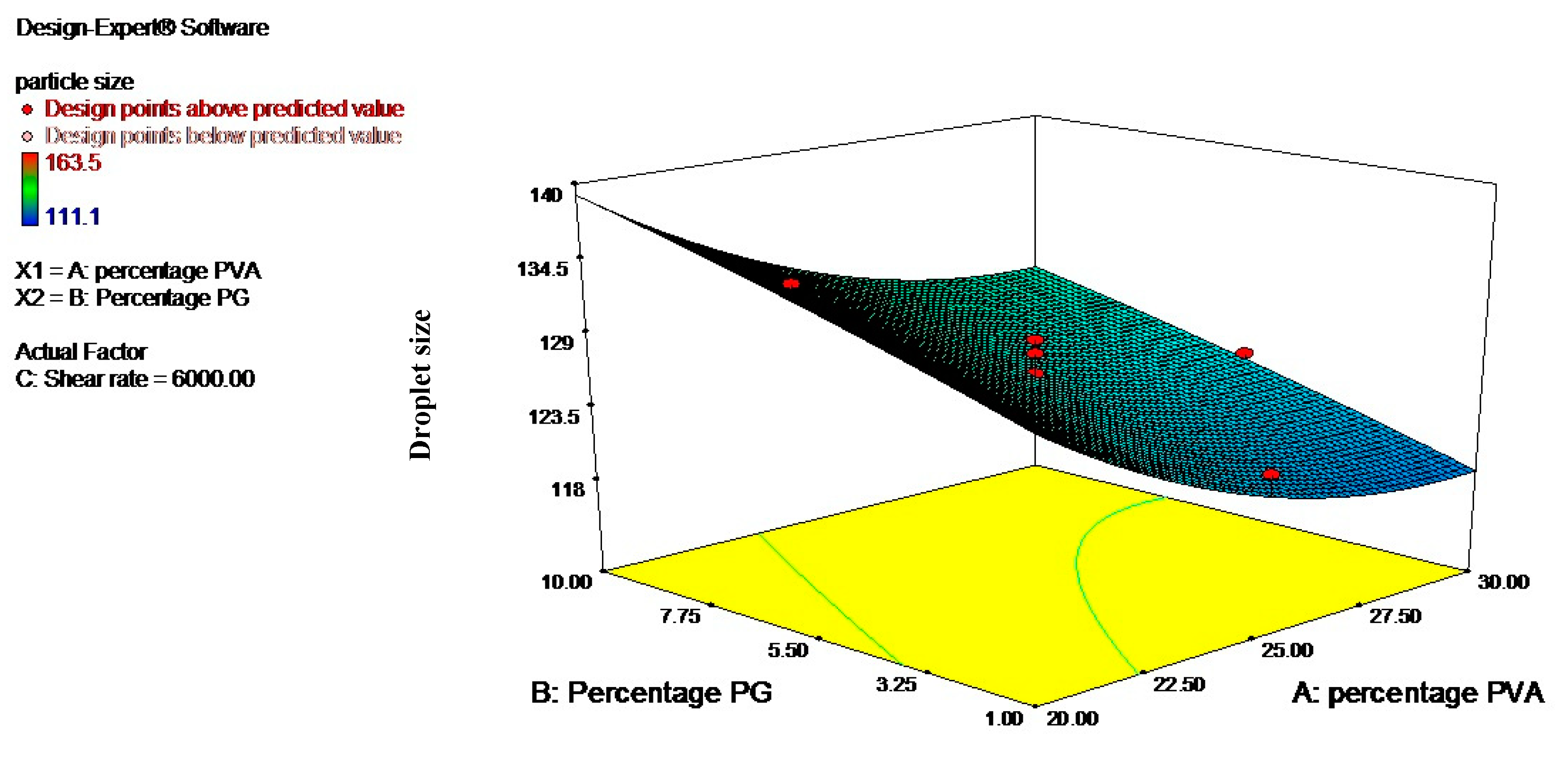
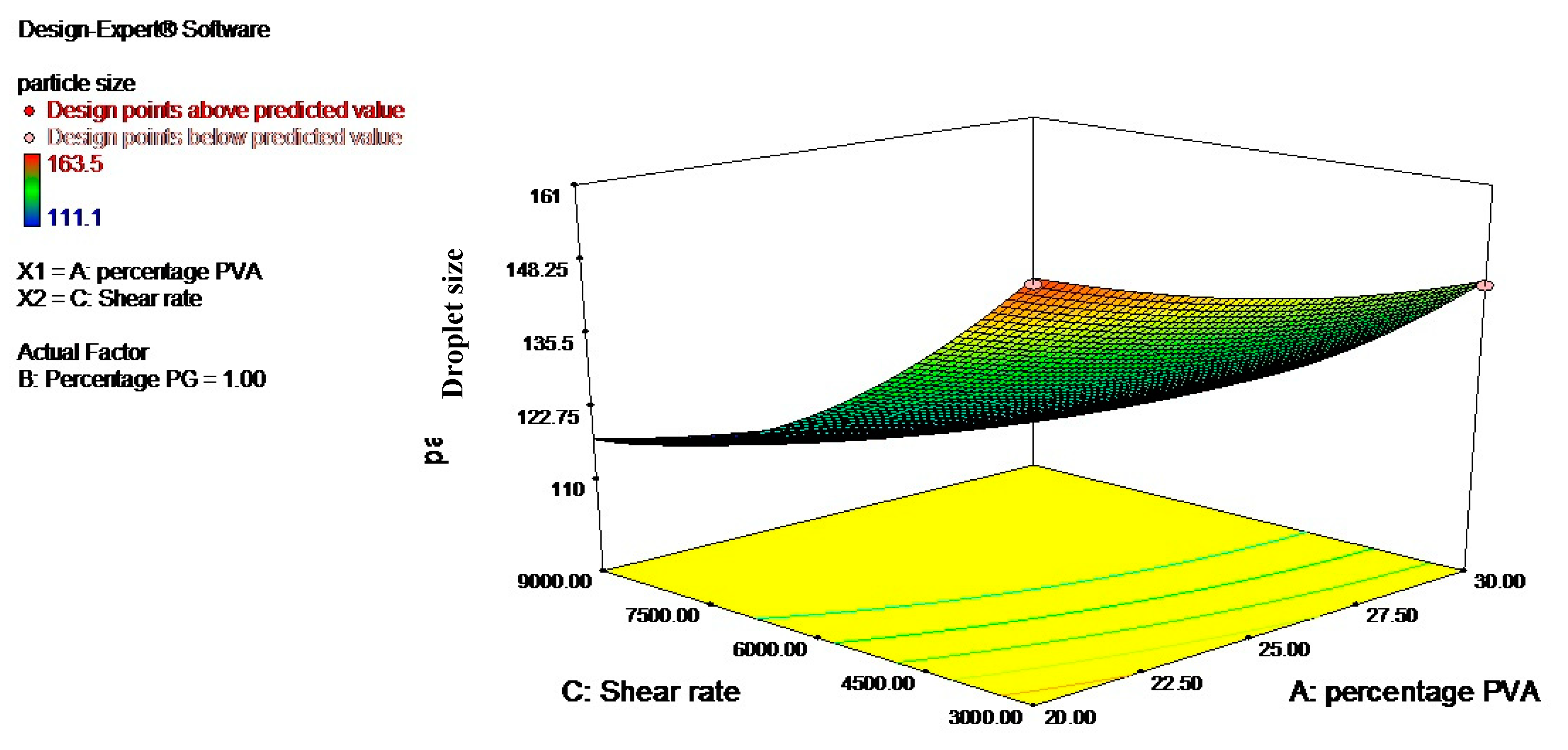
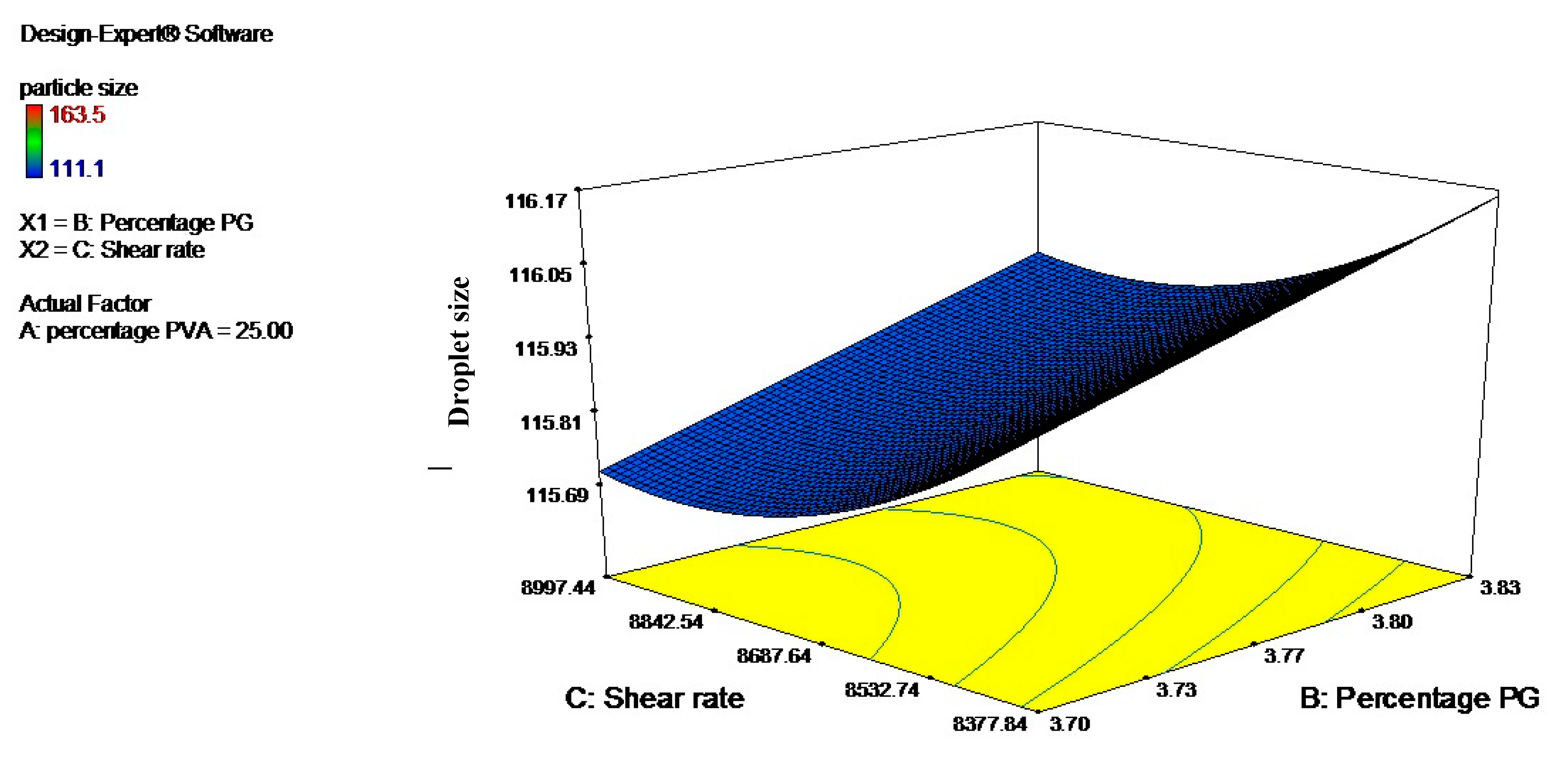
| Levels | Independent Variables | ||
|---|---|---|---|
| Percentage PVA (A) (%) | Percentage PG (B) (%) | Shear rate (C) (rpm) | |
| Lower limit, Ll Upper limit, Ul | 20 | 1 | 3000 |
| 30 | 10 | 9000 | |
| Run No | PVA A | PG B | Shear Rate C | Droplet Size (nm) | |
|---|---|---|---|---|---|
| Actual | Predicted | ||||
| 1 | 25.0 | 10.0 | 6000.0 | 146.1 | 130.3 |
| 2 | 20.0 | 1.0 | 3000.0 | 159.3 | 160.3 |
| 3 | 30.0 | 10.0 | 3000.0 | 147.9 | 148.3 |
| 4 | 20.0 | 5.5 | 6000.0 | 136.0 | 134.8 |
| 5 | 30.0 | 5.5 | 6000.0 | 124.0 | 123.6 |
| 6 | 25.0 | 5.5 | 6000.0 | 126.1 | 126.1 |
| 7 | 20.0 | 10.0 | 3000.0 | 163.5 | 164.2 |
| 8 | 25.0 | 5.5 | 6000.0 | 123.1 | 126.1 |
| 9 | 20.0 | 10.0 | 9000.0 | 133.1 | 132.6 |
| 10 | 25.0 | 5.5 | 9000.0 | 117.3 | 118.8 |
| 11 | 25.0 | 5.5 | 6000.0 | 122.4 | 126.1 |
| 12 | 20.0 | 1.0 | 9000.0 | 142.3 | 117.2 |
| 13 | 25.0 | 5.5 | 6000.0 | 125.4 | 126.1 |
| 14 | 25.0 | 1.0 | 6000.0 | 122.6 | 121.0 |
| 15 | 25.0 | 5.5 | 6000.0 | 127.6 | 126.1 |
| 16 | 25.0 | 5.5 | 3000.0 | 154.8 | 151.7 |
| 17 | 30.0 | 1.0 | 9000.0 | 111.1 | 110.8 |
| 18 | 30.0 | 10.0 | 9000.0 | 126.1 | 125.5 |
| 19 | 25.0 | 5.5 | 6000.0 | 128.6 | 126.1 |
| 20 | 30.0 | 1.0 | 3000.0 | 144.0 | 144.9 |
| Source | Sum of Squares | df | Mean Square | F-Value | p-Value | Significant |
|---|---|---|---|---|---|---|
| Model | 3725.55 | 9 | 413.95 | 65.30 | <0.0001 | significant |
| A | 208.05 | 1 | 208.05 | 32.82 | 0.0004 | |
| B | 117.19 | 1 | 117.19 | 18.49 | 0.0026 | |
| C | 1796.80 | 1 | 1796.80 | 283.44 | <0.0001 | |
| AB | 0.10 | 1 | 0.10 | 0.016 | 0.9012 | |
| AC | 24.11 | 1 | 24.11 | 3.80 | 0.0869 | |
| BC | 39.60 | 1 | 39.60 | 6.25 | 0.0370 | |
| A2 | 21.57 | 1 | 21.57 | 3.40 | 0.1023 | |
| B2 | 0.28 | 1 | 0.28 | 0.045 | 0.8375 | |
| C2 | 185.44 | 1 | 185.44 | 29.25 | 0.0006 | |
| Residual | 50.71 | 8 | 6.34 | |||
| Lack-of-Fit | 20.96 | 3 | 6.99 | 1.17 | 0.4071 | not significant |
| Pure Error | 29.75 | 5 | 5.95 | |||
| Cor Total | 3776.26 | 17 |
| Factor | Coefficient Estimate | ||
|---|---|---|---|
| Intercept | 126.07 | Standard deviation | 2.52 |
| A | −5.60 | Mean | 132.94 |
| B | 4.67 | C.V. % | 1.89 |
| C | −16.47 | PRESS | 787.73 |
| AB | −0.15 | R2 | 0.9866 |
| AC | 2.22 | Adjusted R2 | 0.9715 |
| BC | 2.85 | Predicted R2 | 0.7914 |
| A2 | 3.13 | Adequate precision | 28.498 |
| B2 | −0.40 | ||
| C2 | 9.18 |
| Independent Variables | Size Particle | ||||
|---|---|---|---|---|---|
| A | B | C | Actual Value (nm) | Predicted Value (nm) | Relative Standard Error (% RSE) |
| 22 | 1 | 9000 | 105.20 ± 0.36 | 113.93 | 0.18 |
| 22 | 1 | 8000 | 112.57 ± 0.64 | 115.72 | 0.32 |
| 24 | 2.5 | 9000 | 107.23 ± 1.79 | 114.38 | 0.91 |
| 24 | 2.5 | 8000 | 110.87 ± 1.19 | 115.54 | 0.60 |
| 26 | 5 | 6000 | 122.87 ± 0.29 | 124.52 | 0.13 |
| Goal | Lower Limit | Upper Limit | |
|---|---|---|---|
| PVA | In range | 20.00 | 30.00 |
| PG | In range | 1.00 | 10.00 |
| Shear rate | In range | 3000 | 9000 |
| Droplet size | Minimize | 111.1 | 163.5 |
| Variables | Droplet Size (nm) | Desirability | |||
|---|---|---|---|---|---|
| PVA | PG | Shear Rate | Actual | Predicted | |
| 27.61 | 1.05 | 8656.17 | 105.93 ± 0.21 | 110.21 | 1.00 |
| Formulations | k | n | R2 |
|---|---|---|---|
| KMO formulation | 1.1252 | 0.6803 | 0.8962 |
| Property | KMO Formulation | % RSE |
|---|---|---|
| Droplet Size (nm) | 105.93 ± 0.21 | 0.11 |
| PDI | 0.13 ± 4.50 × 10−3 | - |
| Zeta potential (mV) | −37.37 ± 0.86 | - |
| Conductivity (µS/cm) | 7.47 ± 4.05 × 10−3 | - |
| pH | 4.74 ± 0.02 | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jaslina, N.F.; Faujan, N.H.; Mohamad, R.; Ashari, S.E. Effect of Addition of PVA/PG to Oil-in-Water Nanoemulsion Kojic Monooleate Formulation on Droplet Size: Three-Factors Response Surface Optimization and Characterization. Cosmetics 2020, 7, 73. https://doi.org/10.3390/cosmetics7040073
Jaslina NF, Faujan NH, Mohamad R, Ashari SE. Effect of Addition of PVA/PG to Oil-in-Water Nanoemulsion Kojic Monooleate Formulation on Droplet Size: Three-Factors Response Surface Optimization and Characterization. Cosmetics. 2020; 7(4):73. https://doi.org/10.3390/cosmetics7040073
Chicago/Turabian StyleJaslina, Nur Farzana, Nur Hana Faujan, Rosfarizan Mohamad, and Siti Efliza Ashari. 2020. "Effect of Addition of PVA/PG to Oil-in-Water Nanoemulsion Kojic Monooleate Formulation on Droplet Size: Three-Factors Response Surface Optimization and Characterization" Cosmetics 7, no. 4: 73. https://doi.org/10.3390/cosmetics7040073
APA StyleJaslina, N. F., Faujan, N. H., Mohamad, R., & Ashari, S. E. (2020). Effect of Addition of PVA/PG to Oil-in-Water Nanoemulsion Kojic Monooleate Formulation on Droplet Size: Three-Factors Response Surface Optimization and Characterization. Cosmetics, 7(4), 73. https://doi.org/10.3390/cosmetics7040073





