1. Introduction
Alopecia, also referred to as hair loss, is the reduction in the quantity of visible hair. Although hair loss may not result in physical harm, it can have a substantial impact on mental well-being by reducing self-esteem and self-confidence. Currently, the available treatments for alopecia encompass pharmaceutical alternatives like minoxidil, finasteride, and anti-androgens, alongside surgical interventions such as hair transplant surgeries and laser therapy [
1]. Although these therapies are successful for specific types of hair loss, these treatments also have challenges and limitations, such as side effects, cost, and long-term commitment [
2]. Consequently, there is a growing demand for new bioactive substances that can be used in pharmaceutical or cosmetic products to prevent hair loss and promote new hair growth.
Anti-hair loss therapies primarily aim to inhibit or minimize hair shedding, whereas hair growth promotion products facilitate the regeneration of hair or the development of new hair strands. They are closely related to hair growth stimulation and anti-hair loss products since they both contribute to the maintenance of healthy hair. In order to inhibit and diminish the process of hair aging, extensive research has been undertaken globally to identify active compounds that can effectively minimize hair thinning and loss. Plant extracts are commonly utilized in hair care cosmetics due to their diverse range of functions, which include antioxidant, radical-scavenging and immunostimulant effects; 5α-reductase (5-AR) inhibition; hair growth promotion; and targeting various modes of action [
3].
Resveratrol (trans-3, 5, 4′-trihydroxystilbene) is a polyphenolic phytoalexin that helps accelerate wound healing by enhancing the epidermis, collagen deposition, and hair follicles [
4]. Moreover, it suppresses prostaglandin D2, which causes male baldness and hair lengthening inhibition [
5]. Therefore, it might be considered one of the possible compounds for treating hair loss, although further research is necessary.
Peanut (
Arachis hypogaea L.) is among the dietary sources of resveratrol. Peanut contains a variety of bioactive substances, including flavonoids, phenolic acids, phytosterols, and alkaloids. In vivo and in vitro studies on the biological effects of peanut show that it has anti-microbial, gut flora-regulating, anti-inflammatory, cancer preventing, and cardiovascular protecting properties, with resveratrol showing the most significant impact [
6]. The extracts derived from various parts of the peanut plant have special qualities and possible health advantages. Peanut sprout reduces inflammation and protects human dermal fibroblasts from oxidative stress damage when exposed to ultraviolet radiation [
7]. Biotechnological progress has made it possible to produce resveratrol from unconventional sources. Peanut callus tissue is capable of synthesizing resveratrol in response to stress condition. This approach not only provides a sustainable way to harvest resveratrol without depleting natural resources but also yields extracts that are abundant in resveratrol and potentially other beneficial compounds. The induction of peanut callus enhances phenolic compounds (especially resveratrol) and bioactivities including anti-tyrosinase and antioxidant activities that may have beneficial uses in dietary supplements, foods, pharmaceuticals, and cosmetics [
8]. However, it is important to recognize that while resveratrol and peanuts have shown promise in these areas, more research is needed to fully understand their potential benefits for hair health and their utilization for hair loss treatments. This study aimed to investigate the hair growth promoting effects of peanut callus compared to its seed and sprout, and standard resveratrol.
2. Materials and Methods
Ethanol (≥99.9%), trichloroacetic acid (≤100%), and sulfuric acid (98%), were acquired from Merck, Darmstadt, Germany. Murashige and Skoog medium, 2,4-dichlorophenoxyacetic acid (>99%), 1-naphthaleneacetic acid (>97%), and 6-benzylaminopurine (>98%) and gallant gum (100%) were purchased from Phyto Technology Laboratories, Lenexa, KS, USA. Penicillin streptomycin solution (10,000 U/mL), trypsin/EDTA solution (0.25%), and phosphate-buffered saline (PBS; 1X, pH 7.4) were purchased from Gibco, MA, USA. 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT; 95–100%) and dimethyl sulfoxide (DMSO; ≥99.9%) were acquired from Bio Basic Inc., Markham, ON, Canada. The human hair follicle dermal papilla cells (C-12071), fetal bovine serum, and follicle dermal papilla cell growth media (C26501) were obtained from PromoCell GmbH, Heidelberg, Baden-Württemberg, Germany.
2.1. Plant Material and Extract Preparation
The peanut seeds cv.KK5 were collected in Chiang Rai Province, Thailand. Peanut sprouts were generated by surface-sterilizing peanut seeds with 70% ethanol solution and then subjecting them on three layers of moist filter paper (soaked in 3 mL of distilled water) in Petri dishes. The dishes were incubated at ambient temperature and kept in a wet environment until nearly all peanut seeds sprouted radicles measuring over 1 mm in length (for a duration of 3 days). Callus culture was initiated by inoculating sterilized leaf explants from peanut seedlings onto agar solidified basal Murashige and Skoog medium supplemented with sugar (30 g/L), agar (2 g/L), 2,4-dichlorophenoxyacetic acid (2 mg/L), 1-naphthaleneacetic acid (1 mg/L), and 6-benzylaminopurine (1 mg/L). Subsequently, the plates were placed in a sterilized environment at a controlled temperature condition (25 ± 2 °C) for 28 days. The callus formation was then collected for extraction. Next, a portion (1 g) of samples (peanut seed, sprout, and callus) was extracted using 10 mL of 95% ethanol for 24 h at room temperature. The extracts underwent filtration using Whatman® filter paper no. 1 (Cytiva, Marlborough, MA, USA) and was thereafter kept at a temperature of 4 °C until analysis.
2.2. Determination of Resveratrol
The resveratrol content in the samples was analyzed by reversed-phase high-performance liquid chromatography (HPLC) [
8]. The extracts were analyzed by using Poroshell 120 EC-C18 (250 × 4.6 mm, 4 µm) with a Poroshell 120 guard column (5 × 4.6 mm, 4 µm) at 40 °C and a detection wavelength at 320 nm. The elution program was performed as a mixture of 40%
v/
v of methanol and 60%
v/
v of water at 1 mL/min flow rate for 30 min. The resveratrol content was quantified using the linear regression equation. The regions of the peaks were integrated automatically, and the chromatograms were displayed and processed by a computer using Agilent software (OpenLab CDS version 2.3). The standard calibration curves were generated by graphing the peak areas obtained from HPLC analysis against the concentrations of standard resveratrol solution. The data obtained from the usual linear regression had a strong linear correlation, and the resulting equation remained accurate within the concentration range of interest. The linear calibration equations were y = 1.003x – 1.1996, where Y is peak area and X is concentration of resveratrol. The correlation coefficients (r
2) were 0.9999.
2.3. Cell Culture
hDPCs were cultivated in a culture medium containing 10% fetal bovine serum and 1% penicillin streptomycin solution. The cells were cultivated until they achieved a state of 90% confluence. Subsequently, a volume of 20,000 cells per milliliter was transferred to a fresh well of microplates and subjected to incubation in a humidified incubator containing 5% CO2 at a temperature of 37 °C for 24 h prior to subsequent analysis.
2.3.1. Cytotoxicity
The cytotoxicity of the test samples was investigated using hDPCs through the MTT assay [
9]. The cells were exposed to the diluted extract at concentrations ranging from 0% to 20% and resveratrol at concentrations ranging from 0% to 0.5%, for a duration of 24 h. Consequently, 50 µL of MTT solution with a concentration of 0.1 mg/mL was placed there, and the plate underwent incubation for a further 4 h. Then, DMSO (100 µL) was introduced, and the plate was allowed to remain at ambient for an additional 30 min. The measurement of absorbance was thereafter conducted at 570 nm using a microplate reader (Biochrom, Holliston, MA, USA). The following equation was used to calculate the percentage of cell viability:
where A
sample represents the absorbance of the cell when the test sample is present and A
control represents the absorbance of the cell when the test sample is absent (as a control).
The cytotoxicity was quantified using the CC50 value, which represents the concentration of the test sample that is necessary to reduce 50% of the living cells compared to the cells that were not treated as a control. Afterwards, subsequent studies selected the sample at non-toxic concentration as the further test concentration.
2.3.2. Cell Proliferation
The proliferation-promoting activity of the materials was assessed using the MTT proliferation technique [
10]. The samples containing the highest non-toxic dose were added to cells at a concentration of 20,000 cells/mL. The plate was then cultured at 37 °C in a 5% CO
2 humidified incubator for 72 h. Subsequently, all medium used for cultivation was taken out, and a volume of 50 µL of MTT solution, which had been filtered and sterilized, was added and incubated for a duration of 4 h. After that, DMSO (100 µL) was added and incubated for another half an hour. A microplate reader (Biochrom, USA) was used to measure the absorbance at 570 nm. This equation was used to calculate cell proliferation:
2.3.3. Growth Factor Gene Expression
hDPCs were grown in serum-free medium for a duration of 24 h before being exposed to the test samples for a further 24 h. To extract total RNAs, the acid guanidinium thiocyanate–phenol–chloroform method was applied. M-MLV reverse transcriptase was used to reverse transcribe total RNAs. The RT mixes underwent further exposure to PCR cycles. The cyclic protocol was 40 cycles for IGF-1, HGF, and VEGF and 35 cycles for KGF for 30 s at 94 °C, 30 s at 58 °C, and 1 min at 72 °C [
11].
2.3.4. 5α-Reductase Inhibition
hDPCs were grown in serum-free medium for a duration of 24 h before being exposed to the test samples for a further 24 h. To extract total RNAs, the acid guanidinium thiocyanate–phenol–chloroform method was chosen. M-MLV reverse transcriptase was used to reverse transcribe total RNAs. In PCR reactions, the Cdna produced by this reaction serves as a DNA template. The first step in the PCR-thermal profile was denaturation at 94 °C for 3 min. Then, there were 35 cycles of denaturation, annealing, and extension at 72 °C for 30 s, 2 min, and 10 min, respectively. The PCR results underwent processing and densitometer measurement by 1% agarose gel electrophoresis [
11].
2.4. Clinical Study
2.4.1. Ethical Aspects
The procedure was permitted by the Ethics Committee of Mae Fah Luang University and it was assigned approval number EC19312-17. The Helsinki Declaration on the use of human subjects was adhered to in the research protocols. Each participant in the clinical trial signed a written informed consent form before participating, outlining the purpose of the investigation, the procedures to be followed, the general characteristics of the substances under evaluation, and any known or anticipated adverse effects that might result from involvement.
2.4.2. Participants
A total of 60 healthy volunteers (51 females and 9 males) with pattern hair loss in mild to moderate alopecia (females: Ludwig Scale I to II; males: Norwood Pattern II to IV) aged between 22 and 65 years were treated. All volunteers had no history of hypersensitivity and none of them were pregnant or lactating. They were excluded if they had used any topical or systemic medications for hair regrowth in the last 6 months. The volunteer consisted of healthy adults that had no scalp disease and no previous experience of intolerance or allergic reactions to cosmetic products.
2.4.3. Skin Irritation Testing
Skin irritation testing was performed using a Draize model [
12]. The test products were applied to forearm skin for up to 24 h under occlusive patches. The evaluation of skin irritation was performed at 30 min and 24 h after patch removal. The severity of skin irritation was assessed on a scale ranging from 0 to 4, and the average irritation index (M.I.I.) was computed. A score below 0.2 indicated the absence of irritation or non-irritation.
2.4.4. Efficacy of Anti-Hair Loss Serums
Anti-hair loss serums were tested in a randomized, double-blind, placebo-controlled clinical trial with healthy female and male volunteers that experienced hair loss. The volunteers were assigned to one of the three treatment groups using a random selection process. The following groups were formed: F1 (20 participants that applied a placebo hair serum); F2 (20 participants that applied a hair serum containing 15% peanut callus extract); and F3 (20 participants that applied a hair serum containing 0.5% resveratrol). All volunteers were advised to apply and massage 2 g of the anti-hair loss product over the entire scalp daily at night for 12 weeks. All volunteers received cleansing shampoos that contained no bioactive ingredients to normalize external factors. Participants were not allowed to make any changes owing to their normal hair care routine or to use any additional hair care products during the trial, including those intended to prevent hair loss or dandruff. The participants were advised against modifying their hairstyles throughout the course of the study.
Anti-hair loss effects were evaluated via daily hair shedding counts, one-minute combing, and the forcible hair pluck test. The daily hair shedding count involved volunteers collecting and counting all the hair shed in a day from the shower, sink, and brush. The one-minute combing test involved the volunteers visiting the research laboratory for a 2-week interval without washing their hair one day prior to the examination; the volunteer’s hair was combed for 1 min over a white cloth sheet. The quantity of hair on the sheet and in the comb was counted and recorded compared with the baseline during application of the products and after examination. The percentage change in hair fall values was determined for each participant using the following equation:
To determine the anagen-to-telogen counts ratio (AT ratio), ten hair shafts were plucked with forceps from the treated region of the scalp and classified the shafts as either anagen (with bulb) or telogen (without bulb).
2.5. Statistical Analysis
The measurements were conducted three times. The collected data were subjected to statistical analysis using SPSS 11.5 for Windows (SPSS Inc., Chicago, IL, USA). Statistical significance was determined at p < 0.05. The data comparison was examined using a one-way analysis of variance (ANOVA) with Duncan’s multiple range test.
3. Results and Discussion
3.1. Plant Material and Extract Preparation
A peanut sprout was successfully germinated from peanut seeds. Radicle emergence occurred from the peanut seed (
Figure 1A), and
Figure 1B shows the morphology of the peanut sprout after 3 days of germination. Peanut callus was effectively generated from the explant leaf after 4 weeks of induction on the callus media. The callus exhibits a dense structure and has a yellow color (
Figure 1C). The peanut sprout and callus extracts were obtained as light-yellow liquid, whereas the seed extract was a colorless solution (
Figure 1).
3.2. Determination of Resveratrol
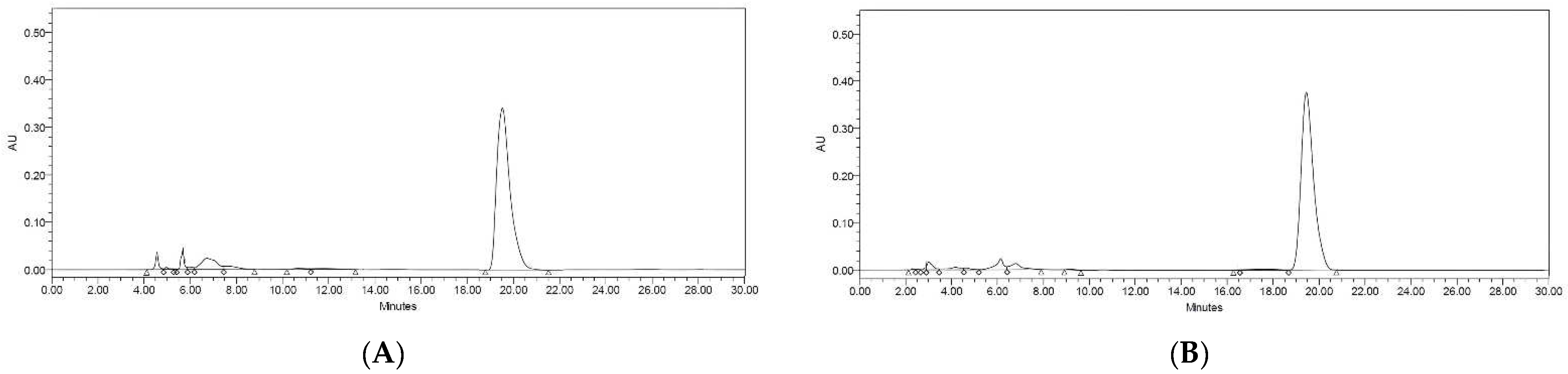
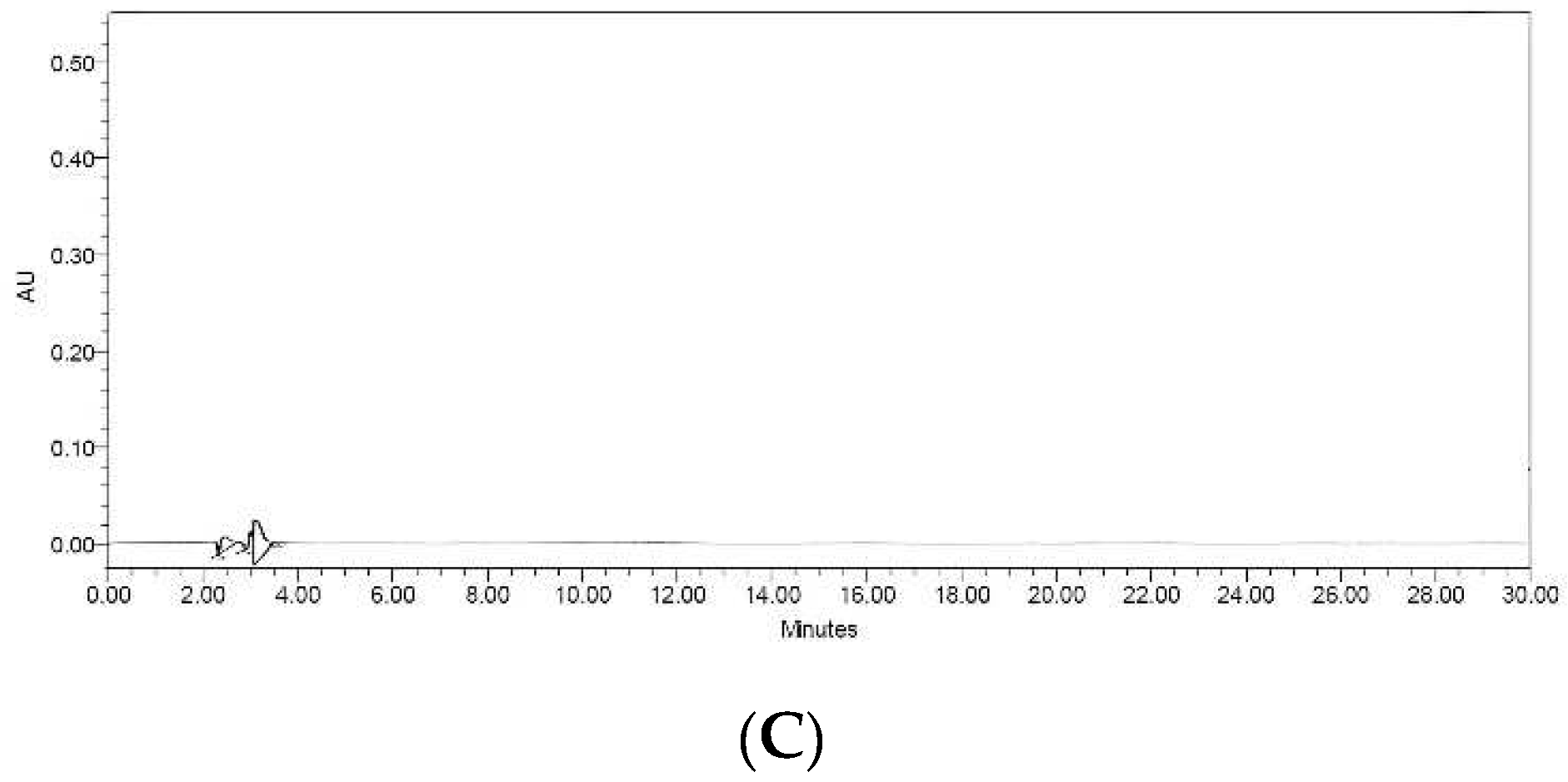
Resveratrol is bioactive compound present in peanuts, particularly in the red seed coat. Resveratrol content is dependent on a number of variables, including plant variety, the part used, growth environment, and processing techniques [
13]. Therefore, in this investigation, resveratrol levels were determined via HPLC analysis of the extracts. The HPLC chromatograms of the resveratrol standard showed a peak at a retention time of 19.44 min. The chromatogram obtained from the sprout and callus extracts exhibited a prominent peak corresponding to resveratrol (
Figure 2A,B). The amounts of resveratrol in the sprout and callus extracts were calculated by comparing them to the standard calibration curve. The sprout sample had a concentration of 131.37 ± 0.01 ppm, whereas the callus sample had a concentration of 191.97 ± 2.56 ppm. However, no discernible peak was observed in the seed extract (
Figure 2C). The findings provided evidence that peanut seeds have the capacity to produce resveratrol through the regulated sprout germination and manipulation of callus culture.
3.3. Hair Growth Promoting and Anti-Hair Loss Effects
The number of people experiencing hair loss is rising in the modern day. Therefore, creating cosmetics to reverse hair loss and promote hair growth is quite pertinent. Specialized mesenchymal cells are found at the base of hair follicles and are crucial to the development of hair follicles and postnatal hair growth cycles. Consequently, hDPCs cells were employed to examine the potential of various compounds, including natural extracts, growth factors, cytokines, peptides, and hormones, for their ability to prevent hair loss and promote hair growth in order to demonstrate the efficacy of these chemicals as innovative therapies in hair care products [
14]. This study utilized hDPCs as a model to assess the extract’s efficacy in hair growth promotion by evaluating cell proliferation, growth factor expression, and its capacity to inhibit 5α-reductase, a key enzyme associated with hair loss.
3.3.1. Cytotoxicity
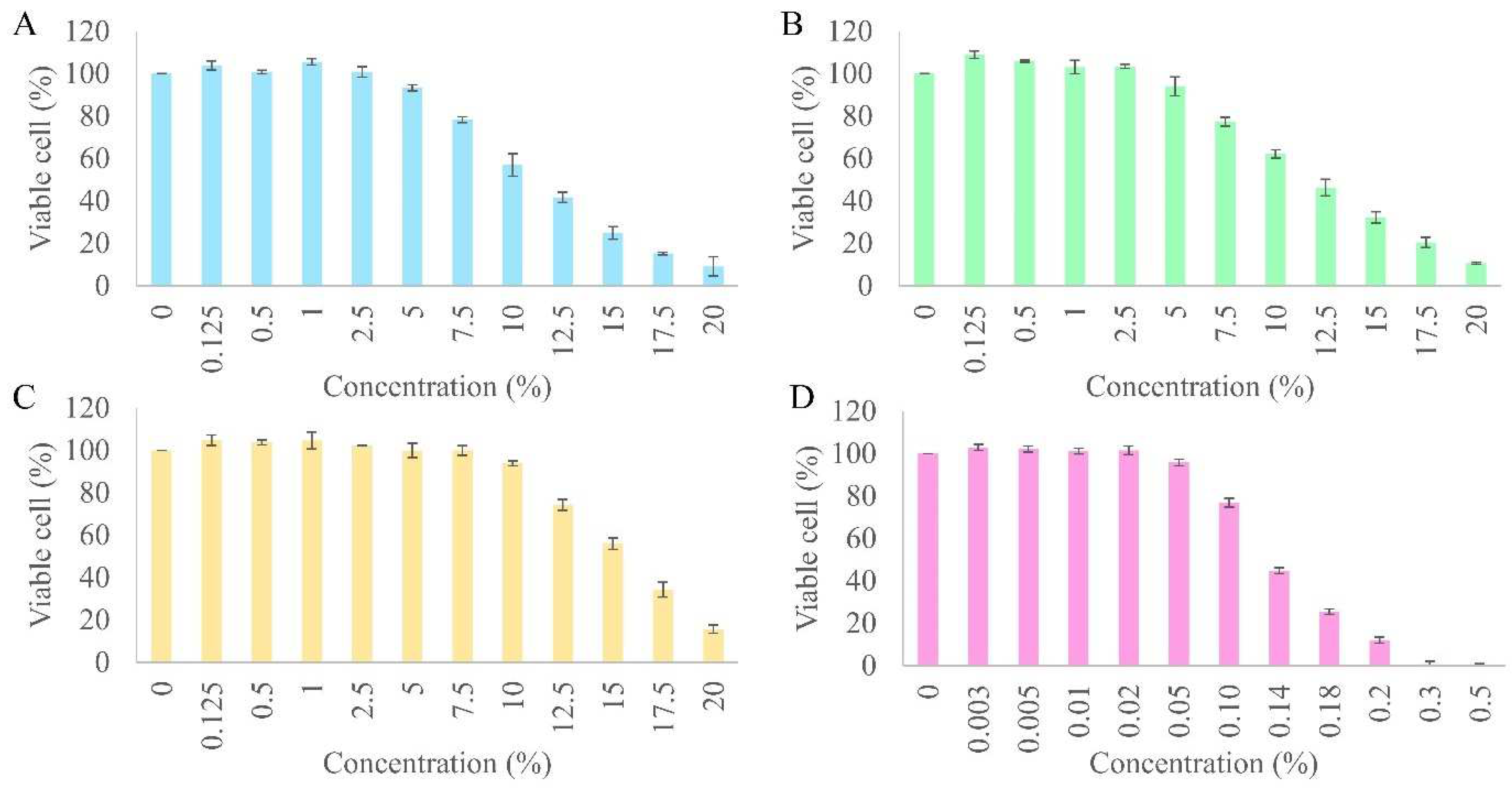
Initially, the cytotoxicity of all the samples, the extracts, and resveratrol was assessed by subjecting different concentrations (0–20%) of the extracts and (0–0.5%) resveratrol to hDPCs. All the extracts and resveratrol showed cytotoxicity in a dose-dependent manner on hDPCs (
Figure 3). The cytotoxicity of the samples was calculated and expressed as the 50% cytotoxic concentration (CC
50). The toxicity of the peanut callus extract (CC
50 of 15.50 ± 0.05%) was found to be significantly lower in comparison to the sprout (CC
50 of 12.30 ± 0.14%) and seed (CC
50 of 11.69 ± 0.26%) extracts when tested on hDPCs (
p < 0.05). The toxicity levels of all the samples were found to be lower in comparison to that of resveratrol (CC
50 of 0.14 ± 0.00%), as evidenced by a tenfold higher CC
50 value. The cytotoxicity curve demonstrated that 0.02% resveratrol and 2.5% of all the extracts represented the highest non-cytotoxic concentration. Hence, these concentrations were selected as the primary concentrations for further examinations of cellular proliferation, enhancement of expression of growth factor genes, and inhibition of 5-alpha reductase.
3.3.2. hDPCs Proliferation
Several studies have demonstrated a strong correlation between the proliferation of dermal papilla cells and the hair development cycle. An elevation in the number of dermal papilla cells during the anagen phase leads to a reduction in hair loss [
14]. Moreover, hDPCs from androgenic alopecia patients have a slower growth rate and a tendency to lose their proliferative potential compared to healthy hDPCs [
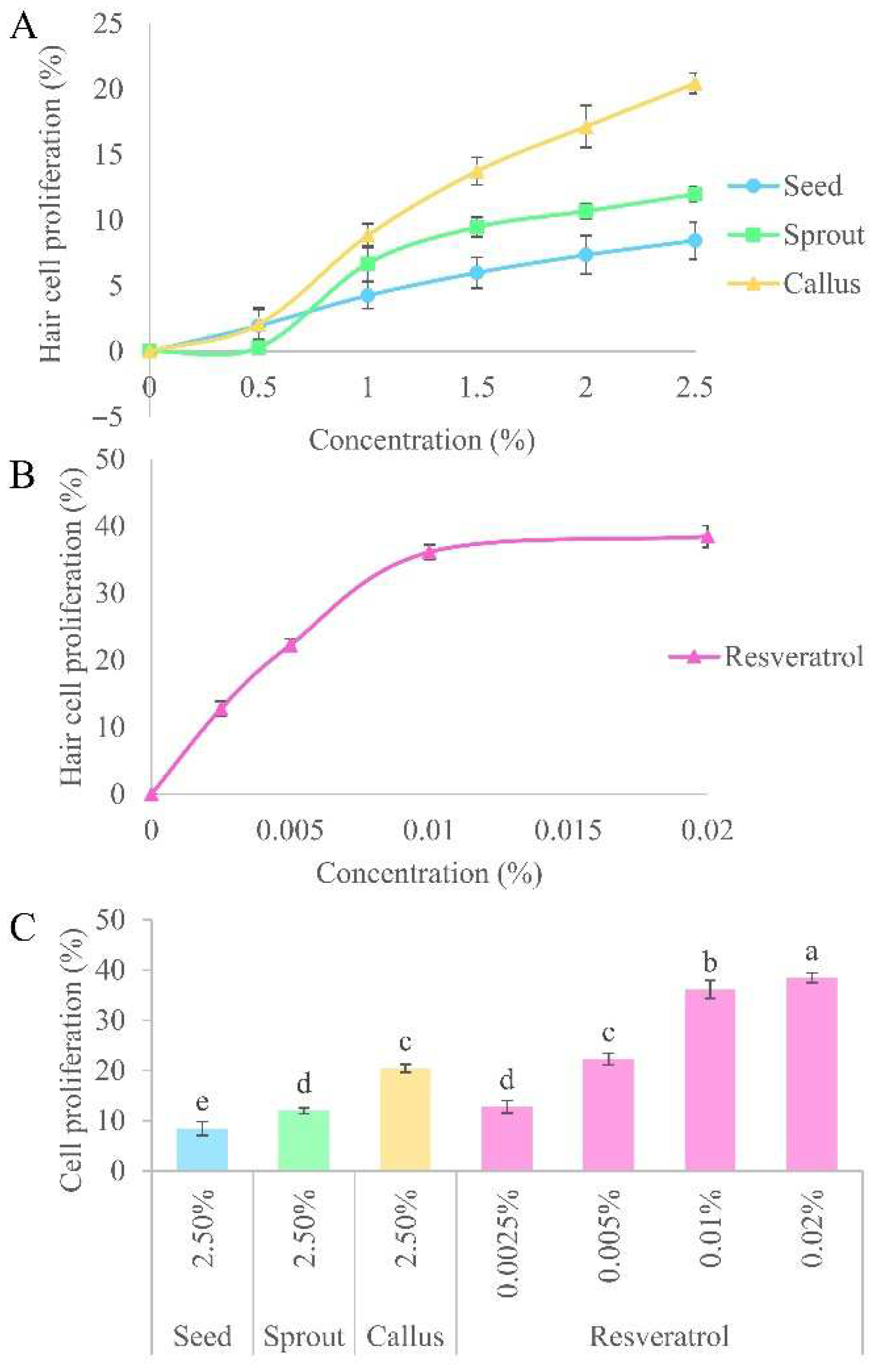
15]. The proliferation of hDPCs has been extensively examined to assess the impact of natural extracts on encouraging hair growth or suppressing hair loss. The proliferation of hDPCs extended the anagen phase of the hair cycle, resulting in enhanced hair density and decreased hair loss. The results indicated that all of the samples had a dose-dependent effect on the proliferation of hDPCs (
Figure 4A,B). The percentage of hDPCs exposed to the callus extract (20.44 ± 0.78%) was significantly greater in comparison to the sprout (11.99 ± 0.55%) and seed (8.45 ± 1.41%) extracts. Of all the tested extracts, callus extract had the greatest beneficial effect on the growth of human hair cells. These results may be due to it has the highest amount of resveratrol. The efficacy of the callus extract at a concentration of 2.5% was found to be almost equivalent to that of 0.005% resveratrol (22.29 ± 1.14%;
Figure 4C). This finding indicated that resveratrol treatment can accelerate hair growth in vitro. Juchaux et. al. and Zhang et. al. documented that resveratrol diminishes the generation of reactive oxygen species in hair follicles, shields them from oxidative harm, and stimulates the growth of hDPCs and hair matrix cells [
16,
17]. Therefore, extracts containing resveratrol may stimulate hair development by accelerating the proliferation of crucial cells in hair follicles, protecting them from harm caused by the environment. Furthermore, the extracts may stimulate cell proliferation, leading to the shift from the telogen phase to the anagen phase, ultimately boosting the regeneration of hair.
3.3.3. Growth Factor Gene Expression
Measuring the gene expression level of growth factor is a way to understand how substances contribute to encouraging hair growth. Multiple growth factors, including IGF-1, KGF, HGF, and VEGF have a role in regulating hair development [
14]. The regulation of hair morphogenesis and hair development involves the expression of several growth factors. IGF-1 promotes the growth of epithelial cells and controls the process of cellular proliferation and migration in the creation of hair follicles. IGF-1 stimulates hair growth and is crucial for the growth and renewal of hair follicles. It also plays a significant role in tissue restructuring and the hair growth cycle. The reduced production of IGF-1 has been linked to androgenetic alopecia [
18,
19]. KGF serves as an internal signaling molecule that promotes the growth of hair follicles and enhances the development of sebaceous glands and keratinocytes in the hair follicles. Moreover, KGF is a growth factor that promotes the elongation of hair fibers, safeguards hair follicles against cell death, and has a direct impact on the formation of hair follicles [
20,
21]. HGF functions as a paracrine factor by stimulating the growth of follicles through its effects on nearby follicular epithelial cells. Research has demonstrated that dermal papilla cells release HGF, which is used to regulate the sheath fibroblasts surrounding hair follicles to promote hair regeneration [
22]. VEGF plays a crucial role in promoting the growth of blood vessels around hair follicles, leading to improved blood flow and larger hair follicles [
23]. VEGF expression also stimulates the growth and specialization of fibroblast cells, as well as the regeneration and movement of epithelial cells within hair follicles [
24]. In order to understand the molecular mechanism of the extracts that promote hair growth, the expression of growth factors (IGF-1, KGF, HGF, and VEGF) was examined.
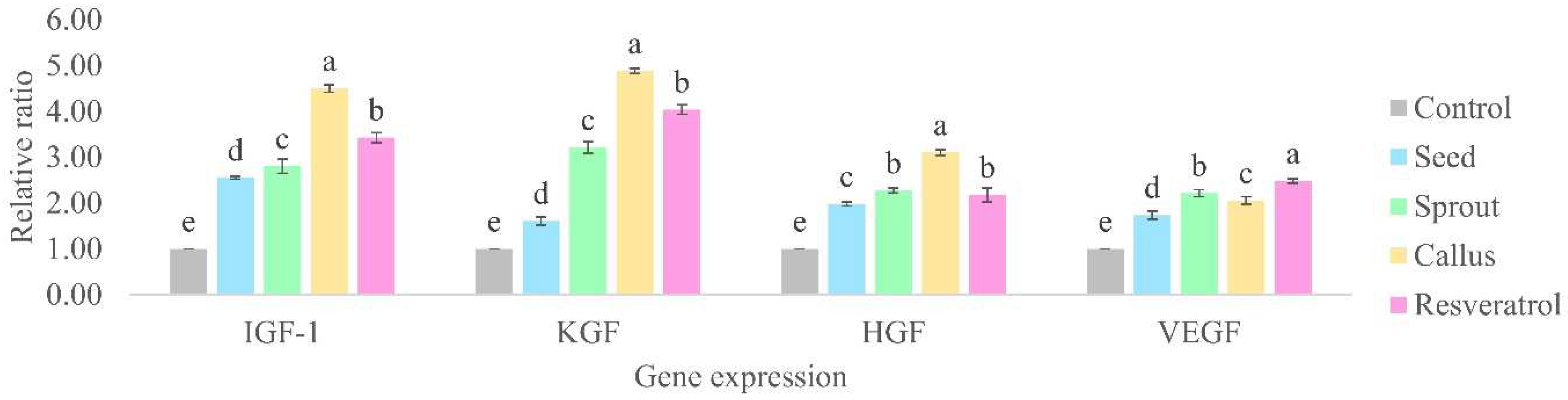
The mRNA expression levels of all growth factors were found to be elevated when treated with extracts (2.5%) and resveratrol (0.02%) compared to untreated cells (
Figure 5). The expression profiles of each growth factor in the treatment exhibited diverse ranges. IGF-1 and KGF expression showed greater increases, ranging from 2.5- to 4.5-fold and 1.6- to 4.8-fold, respectively, whereas HGF and VEGF showed increases from 1.9- to 3.1-fold and 1.73- to 2.21-fold, respectively. The standard resveratrol increased levels of IGF-1, KGF, HGF, and VEGF expression, with fold changes of 3.43 ± 0.11, 4.04 ± 0.11, 2.18 ± 0.15, and 2.48 ± 0.05, respectively. This result is in accordance with Kubo’s work on the impact of polyphenols on hair growth, which revealed that resveratrol enhanced the increase in IGF-1 and KGF expression in HaCaT cells [
25]. In comparison to other treatments, the callus extract exhibited considerably greater levels of IGF-1, KGF, and HGF expression (4.50 ± 0.08-fold, 4.89 ± 0.05-fold, and 3.11 ± 0.06-fold, respectively). The treatment of resveratrol (3.43 ± 0.11- and 4.04 ± 0.11-fold) was a greater expression of IGF-1 and KGF compared to sprout (2.81 ± 0.16- and 3.21 ± 0.12-fold) and seed (2.55 ± 0.03- and 1.61 ± 0.09-fold). There was no significant difference in HGF expression between sprout treatment (2.27 ± 0.05-fold) and resveratrol (2.18 ± 0.15-fold). The higher inducing gene expression levels (IGF-1, KGF, and HGF) of the samples, particularly in the callus extract, are related to a highly positive effect on the proliferation and differentiation of cells in hair follicles and manipulate sheath fibroblasts that surround hair follicles, which are important to maintain normal tissue structure and stimulate hair regeneration. VEGF expression was slightly higher in resveratrol (2.48 ± 0.05-fold) and sprout extract (2.21 ± 0.08-fold) than in the callus extract (2.05 ± 0.08-fold) and the seed extract (1.73 ± 0.09-fold), indicating its impact on the stimulation of the vasodilation of scalp blood vessels. Although callus extract induced lower levels of VEGF gene expression than sprout extract and resveratrol, it increased VEGF expression compared to untreated cells. In this regard, it is noteworthy that the callus and sprout extracts has broader potential to increase hair growth mRNA expression, especially IGF-1, KGF, and HGF in comparison with the untreated cell. The findings demonstrated that the extracts derived from callus and sprouts have the ability to promote hair growth. This is achieved by stimulating the multiplication and specialization of epithelial cells located near the root of the hair follicle. Additionally, these extracts also influence the fibroblasts surrounding the hair follicles and cause the widening of blood vessels in the scalp.
3.3.4. 5α-Reductase Inhibition
In addition to influencing cell proliferation and the expression level of growth factor genes, androgens play a role in hair development. Testosterone and dihydrotestosterone are primary hormones responsible for regulating hair growth. Dihydrotestosterone, a hormone derived from testosterone by the action of 5α-reductase, is responsible for causing hair loss [
26]. Elevating the androgenic signal reduces the length of the anagen phase, and the reaction to the conversion of large follicles to smaller ones triggers gradual thinning of the scalp hair and subsequent hair loss [
27]. Thus, the suppression of 5α-reductase activity in dermal papilla cells has a direct impact on the therapy of androgenic alopecia. Thus, hDPCs were utilized to investigate the quantitative expression and inhibitory function of 5-AR, taking into account its impact on the investigation of human hair growth.
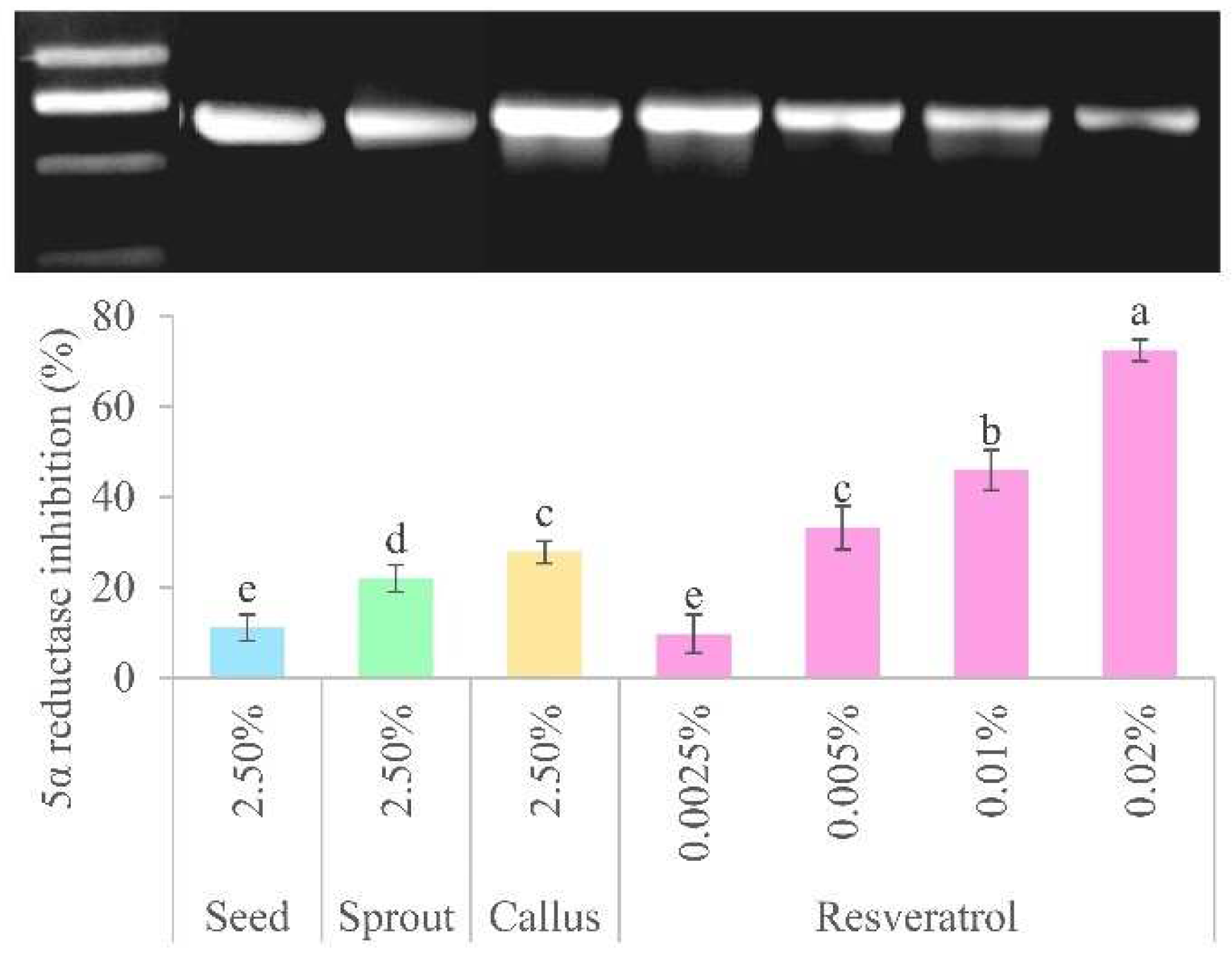
The maximum non-toxic concentration (2.5%) of the extracts and various concentrations (0.0025–0.02%) of resveratrol were used to represent the 5-AR inhibitory activity, as depicted in
Figure 6. The results indicated that the callus exhibited the highest potency (27.83 ± 2.48%) compared to the sprout (21.99 ± 2.99%) and seed (11.07 ± 2.87%). Additionally, the results indicated that the callus had no observable statistically significant distinction when comparing the effects of 0.005% resveratrol (33.22 ± 4.78%). In this investigation, the callus extract was found to have the highest concentration of resveratrol and a higher potency of 5-AR inhibition than other components of the peanut plant, and its impact closely resembles to that of resveratrol. These findings are consistent with previous studies showing that resveratrol acts as a 5-AR inhibitor, with a substantially stronger effect than α-linolenic acid and epigallocatechin gallate in preventing age-related hair loss and stimulating the growth of new hair [
28]. Moreover, trans-resveratrol produced using a hydrogen peroxide/horseradish peroxidase system exhibited potent 5-AR inhibitory activity [
29]. Resveratrol-containing extracts are therefore promising candidates for new non-steroidal 5-AR inhibitors.
The concentration of resveratrol in the extracts showed a strong positive correlation with the proliferation (r = 0.898) and 5-AR inhibition (r = 0.949) of hDPCs. Moreover, there was a favorable correlation between the content and the expression levels of the growth factors IGF-1 (r = 0.815), KGF (r = 0.974), HGF (r = 0.884), and VEGF (r = 0.750). These results indicate that increasing the resveratrol concentration of sprout and callus extracts can increase the potential efficacy of hDPCs. This suggests that resveratrol is the main active ingredient that influences the levels of growth factor expression, the proliferation of 5-AR, and the suppression of 5-AR, all of which are associated with the features of anti-androgenic alopecia. Overall, the function of the peanut callus extract in stimulating hair development has been demonstrated by this in vitro investigation. It shown superior effectiveness in stimulating hair regrowth when compared to the extracts from sprouts and seeds. Therefore, it is chosen as the active ingredient in hair serum formulations for further clinical investigation.
3.4. Clinical Study
3.4.1. Skin Irritation Testing
The hair serum base in this study was composed of water, butylene glycol, glycerin, propylene glycol, polyquaternium-7, phenoxyethanol, and a fragrance, all of which were cosmetic-grade chemicals. This serum had a translucent appearance and was non-greasy and sticky when applied to the scalp and hair. The hair serum formulation contained 15% of the extract, determined based on the CC50 value of the extract (15.50 ± 0.05%). The efficacy of the anti-hair loss treatment was assessed through clinical testing by comparing the extract to resveratrol (a positive control) and a hair serum base (a negative control).
First, a patch test for irritation was conducted on a group of 60 volunteers. The redness and swelling of all examined samples were assessed after 30 min and 24 h after removing the patch. All the tested serums revealed that there was no edema (M.I.I. = 0.00) and no erythema (M.I.I. = 0.00). The tested hair serum products were categorized as non-irritants and no negative side effects were noted.
3.4.2. Efficacy of Anti-Hair Loss Serums
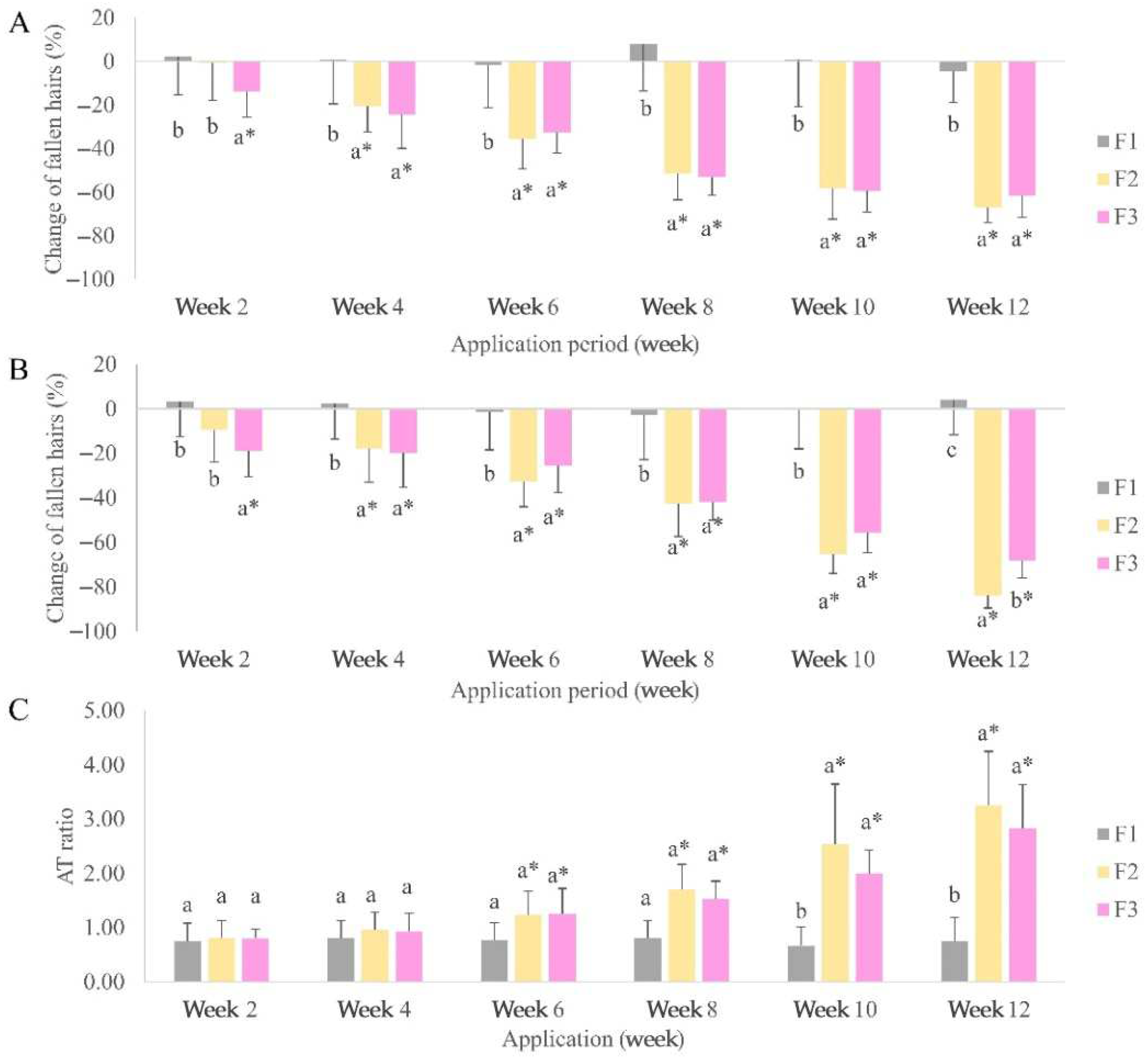
The application of hair serum application containing the peanut callus extract (F2) and resveratrol (F3) showed a reduction in hair shedding in all volunteers (
Figure 7A). The baseline hair count in the F2 group significantly changed by week 4 (20.35%), whereas that in the F3 group significantly changed faster by week 2 (13.84%). No discernible differences were observed between the two groups at the end of the trial (66.98 and 61.47% for F2 and F3, respectively). The combing test demonstrated a significant difference between placebo and active serums at week 2 following the use of a hair serum containing peanut callus extract (F2) and resveratrol (F3) (
Figure 7B). F2 (83.92%) showed significantly better results than F3 (68.19%) in week 12. The AT ratios of each volunteer in the F1 group did not significantly change (0.6–0.8;
Figure 7C). A significant increase in the anagen phase of hair was observed in the F2 and F3 groups at 10 weeks of application. There were no noticeable distinctions observed between F2 (3.3) and F3 (2.8) groups at the end of the trial. The clinical efficacy tests showed that hair serum products with peanut callus extract and resveratrol showed higher anti-hair loss efficacy than the placebo. This study demonstrated that a hair serum formulation containing peanut callus extract or resveratrol are key ingredients that decrease hair fall and increase the anagen phase in volunteers. This finding aligns with previous studies that demonstrated that application of resveratrol topically to shaved C57BL/6 mice effectively stimulated hair growth. On day 18, the length of hair in the resveratrol group was comparable to that of the minoxidil group (positive control group). Resveratrol additionally promoted the shift of the hair cycle from the resting (telogen) phase to the growth (anagen) phase [
17]. Another research trial was carried out to evaluate the efficacy of a hair serum containing peanut shell extract and other natural substances in 42 volunteers for 90 days. There was a significant improvement in the rate of hair growth, density (both fine hair and fully developed hair), and reduction in hair loss (both the presence and absence of a bulb), in comparison to the first assessment [
30].
The mechanism of hair growth activity of the peanut callus extract may involve resveratrol, which is suspected to exhibit anti-hair loss effects via inhibiting DHT synthesis through the regulation of 5-AR activity and stimulate hair growth activity via the hair cycle transition from the telogen to the anagen phase. Additionally, oxidative stress and inflammatory responses contribute to hair loss. Resveratrol showed protection for hDPCs from oxidative damage caused by hydrogen peroxide [
17]. Therefore, the resveratrol’s ability to combat oxidative stress and inflammation may lead to a decrease in hair loss and promote the growth of new hair. The results indicated that the extract from peanut callus, which is rich in resveratrol, showed great potential as a new approach to treating hair loss due to its strong biological effects. It may offer a more efficient natural alternative compared to existing treatments.
4. Conclusions
Resveratrol can be synthesized during the germination of peanut sprouts and the induction of its callus. The experiment results demonstrated that the extracts promoted hair growth via stimulating hDPCs proliferation, inhibiting 5-AR activity, and enhancing gene expression (IGF-1, KGF, HGF, and VEGF), which are associated with the hair cell cycle, proliferation, differentiation, and the stimulation of factors that promote the growth of capillary networks. The callus extract exhibited higher resveratrol content and hair growth promotion compared to the sprout extract. The treatment of the hair serum formulation containing peanut callus extract for a period of three months for hair loss volunteers proved to be both safe and effective in reducing hair shedding and increasing the anagen-to-telogen ratio. These results revealed that peanut callus extract is a potential natural cosmetic ingredient for hair growth stimulation and hair loss prevention.
Author Contributions
Conceptualization, N.S.; Methodology, N.S. and W.V.; Validation, N.S. and W.V.; Formal Analysis, N.S. and W.V., Investigation, N.S. and W.V.; Resources, N.S.; Data Curation, N.S. and W.V.; Writing—Origination Draft Preparation, N.S. and W.V.; Writing—Review and Editing, N.S.; Visualization, W.V.; Supervision, N.S.; Project Administration, N.S.; Funding Acquisition, N.S. All authors have read and agreed to the published version of the manuscript.
Funding
This work was financially supported by the Thailand Research Fund, grant number PHD60I0091.
Institutional Review Board Statement
This study was conducted in accordance with the Declaration of Helsinki and was approved on 27 December 2019 the Ethics Committee of Mae Fah Luang University (Chiang Rai, Thailand), protocol code EC19312-17.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
All of the data are available in the manuscript.
Acknowledgments
The author acknowledges the Cosmetic and Beauty Innovations for Sustainable Development (CBIS) Research Group and Scientific and Technological Instruments Center at Mae Fah Luang University for providing the scientific equipment and facilities for this research.
Conflicts of Interest
The author declares no conflicts of interest.
References
- Salah, M.; Samy, N.; Fawzy, M.M.; Farrag, A.R.; Shehata, H.; Hany, A. The effect of the fractional carbon dioxide laser on improving minoxidil delivery for the treatment of androgenetic alopecia. J. Lasers Med. Sci. 2020, 11, 29–36. [Google Scholar] [CrossRef]
- Kaiser, M.; Abdin, R.; Gaumond, S.I.; Issa, N.T.; Jimenez, J.J. Treatment of androgenetic alopecia: Current guidance and unmet needs. Clin. Cosmet. Investig. Dermatol. 2023, 16, 1387–1406. [Google Scholar] [CrossRef]
- Park, S.; Lee, J. Modulation of hair growth promoting effect by natural products. Pharmaceutics 2021, 13, 2163. [Google Scholar] [CrossRef]
- Hecker, A.; Schellnegger, M.; Hofmann, E.; Luze, H.; Nischwitz, S.P.; Kamolz, L.P.; Kotzbeck, P. The impact of resveratrol on skin wound healing, scarring, and aging. Int. Wound J. 2022, 19, 9–28. [Google Scholar] [CrossRef]
- Kuroyanagi, G.; Mizutani, J.; Kondo, A.; Yamamoto, N.; Matsushima-Nishiwaki, R.; Otsuka, T.; Kozawa, O.; Tokuda, H. Suppression by resveratrol of prostaglandin D2-stimulated osteoprotegerin synthesis in osteoblasts. Prostaglandins Leukot. Essent. Fat. Acids 2014, 91, 73–80. [Google Scholar] [CrossRef]
- Mingrou, L.; Guo, S.; Ho, C.T.; Bai, N. Review on chemical compositions and biological activities of peanut (Arachis hypogeae L.). J. Food Biochem. 2022, 46, e14119. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.Y.; Choi, D.I.; Lee, J.B.; Yun, S.J.; Lee, D.H.; Eun, J.B.; Lee, S.C. Ethanol extract of peanut sprout induces Nrf2 activation and expression of antioxidant and detoxifying enzymes in human dermal fibroblasts: Implication for its protection against UVB-irradiated oxidative stress. Photochem. Photobiol. 2013, 89, 453–460. [Google Scholar] [CrossRef]
- Vichit, W.; Saewan, N. In vitro antioxidant activities and cytotoxicity of peanut callus extract. Food Appl. Biosci. J. 2019, 7, 142–151. [Google Scholar]
- Kim, Y.D.; Pi, L.Q.; Lee, W.S. Effect of Chrysanthemum zawadskii extract on dermal papilla cell proliferation and hair growth. Ann. Dermatol. 2020, 32, 395–401. [Google Scholar] [CrossRef]
- Vichit, W.; Saewan, N. Effect of germination on antioxidant, anti-inflammatory and keratinocyte proliferation of rice. Int. Food Res. J. 2016, 23, 2006–2015. [Google Scholar]
- Saewan, N. Effect of coffee berry extract on anti-aging for skin and hair—In vitro approach. Cosmetics 2022, 9, 66. [Google Scholar] [CrossRef]
- Baldisserotto, A.; Buso, P.; Radice, M.; Dissette, V.; Lampronti, I.; Gambari, R.; Manfredini, S.; Vertuani, S. Moringa oleifera leaf extracts as multifunctional ingredients for “natural and organic” sunscreens and photoprotective preparations. Molecules 2018, 23, 664. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Fan, Z.; Meenu, M.; Xu, B. Impact of germination time on resveratrol, phenolic acids, and antioxidant capacities of different varieties of peanut (Arachis hypogaea Linn.) from China. Antioxidants 2021, 10, 1714. [Google Scholar] [CrossRef]
- Madaan, A.; Verma, R.; Singh, A.T.; Jaggi, M. Review of hair follicle dermal papilla cells as in vitro screening model for hair growth. Int. J. Cosmet. Sci. 2018, 40, 429–450. [Google Scholar] [CrossRef]
- Choi, Y.H.; Shin, J.Y.; Kim, J.; Kang, N.G.; Lee, S. Niacinamide down-regulates the expression of dkk-1 and protects cells from oxidative stress in cultured human dermal papilla cells. Clin. Cosmet. Investig. Dermatol. 2021, 14, 1519–1528. [Google Scholar] [CrossRef] [PubMed]
- Juchaux, F.; Sellathurai, T.; Perrault, V.; Boirre, F.; Delannoy, P.; Bakkar, K.; Albaud, J.; Gueniche, A.; Cheniti, A.; Dal Belo, S.; et al. A combination of pyridine-2, 4-dicarboxylic acid diethyl ester and resveratrol stabilizes hypoxia-inducible factor 1-alpha and improves hair density in female volunteers. Int. J. Cosmet. Sci. 2020, 42, 167–173. [Google Scholar] [CrossRef]
- Zhang, Y.; Ni, C.; Huang, Y.; Tang, Y.; Yang, K.; Shi, X.; Zhang, Y.; Li, Z.; Wang, J.; Zhu, Y.; et al. Hair growth-promoting effect of resveratrol in mice, human hair follicles and dermal papilla cells. Clin. Cosmet. Investig. Dermatol. 2021, 14, 1805–1814. [Google Scholar] [CrossRef]
- Trüeb, R.M. Further clinical evidence for the effect of IGF-1 on hair growth and alopecia. Ski. Appendage Disord. 2018, 4, 90–95. [Google Scholar] [CrossRef]
- Zhao, B.; Li, J.; Chen, Q.; Yang, N.; Bao, Z.; Hu, S.; Chen, Y.; Wu, X. A treatment combination of IGF and EGF promotes hair growth in the angora rabbit. Genes 2021, 12, 24. [Google Scholar] [CrossRef]
- Gentile, P.; Garcovich, S. Advances in regenerative stem cell therapy in androgenic alopecia and hair loss: Wnt pathway, growth-factor, and mesenchymal stem cell signaling impact analysis on cell growth and hair follicle development. Cells 2019, 8, 466. [Google Scholar] [CrossRef]
- Kim, J.; Kim, S.R.; Choi, Y.H.; Shin, J.Y.; Kim, C.D.; Kang, N.G.; Park, B.C.; Lee, S. Quercitrin stimulates hair growth with enhanced expression of growth factors via activation of MAPK/CREB signaling pathway. Molecules 2020, 25, 4004. [Google Scholar] [CrossRef] [PubMed]
- Nicu, C.; O’Sullivan, J.D.B.; Ramos, R.; Timperi, L.; Lai, T.; Farjo, N.; Farjo, B.; Pople, J.; Bhogal, R.; Hardman, J.A.; et al. Dermal adipose tissue secretes HGF to promote human hair growth and pigmentation. J. Investig. Dermatol. 2021, 141, 1633–1645. [Google Scholar] [CrossRef] [PubMed]
- Woo, Y.M.; Kim, O.J.; Jo, E.S.; Jo, M.Y.; Ahn, M.Y.; Lee, Y.H.; Li, C.R.; Lee, S.H.; Choi, J.S.; Ha, J.M.; et al. The effect of Lactobacillus plantarum hydrolysates promoting VEGF production on vascular growth and hair growth of C57BL/6 mice. J. Anal. Sci Technol. 2019, 10, 18. [Google Scholar] [CrossRef]
- Dai, B.; Sha, R.N.; Yuan, J.L.; Liu, D.J. Multiple potential roles of thymosin β4 in the growth and development of hair follicles. J. Cell. Mol. Med. 2021, 25, 1350–1358. [Google Scholar] [CrossRef]
- Kubo, C.; Ogawa, M.; Uehara, N.; Katakura, Y. Fisetin promotes hair growth by augmenting TERT expression. Front. Cell Dev. Biol. 2020, 8, 566617. [Google Scholar] [CrossRef]
- Ma, L.; Shen, H.; Fang, C.; Chen, T.; Wang, J. Camellia seed cake extract supports hair growth by abrogating the effect of dihydrotestosterone in cultured human dermal papilla cells. Molecules 2022, 27, 6443. [Google Scholar] [CrossRef]
- Lao, Z.; Fan, Y.; Huo, Y.; Liao, F.; Zhang, R.; Zhang, B.; Kong, Z.; Long, H.; Xie, J.; Sang, C.; et al. Physcion, a novel inhibitor of 5α-reductase that promotes hair growth in vitro and in vivo. Arch. Dermatol. Res. 2022, 314, 41–51. [Google Scholar] [CrossRef]
- Hirano, Y.; Kondo, R.; Sakai, K. Compounds inhibitory to rat liver 5α-reductase from tropical commercial wood species: Resveratrol trimers from melapi (Shorea sp.) heartwood. J. Wood Sci. 2001, 47, 308–312. [Google Scholar] [CrossRef]
- Hirano, Y.; Kondo, R.; Sakai, K. 5α-reductase inhibitory compounds produced by polymerization of resveratrol with horseradish peroxidase. J. Wood Sci. 2002, 48, 64–68. [Google Scholar] [CrossRef]
- Majeed, M.; Majeed, S.; Nagabhushanam, K.; Mundkur, L.; Neupane, P.; Shah, K. Clinical study to evaluate the efficacy and safety of a hair serum product in healthy adult male and female volunteers with hair fall. Clin. Cosmet. Investig. Dermatol. 2020, 13, 691–700. [Google Scholar] [CrossRef]
| Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).