Abstract
In the current study, human keratinocyte cell line was used as in vitro cell culture model to elucidate the effects of the fatty acid chain length of acylglutamate (amino acid-based surfactant) namely, sodium cocoyl glutamate, sodium lauroyl glutamate, and sodium myristoyl glutamate on their cytotoxicity and the ultraviolet B induced phototoxicity. The endpoint used to assess toxicity was a tetrazolium-based assay whereas, the phototoxic potential of acylglutamate surfactants was predicted using two models namely, the Photo-Irritation Factor and Mean Photo Effect. The results of this study showed that the fatty acid chain length of acylglutamate greatly influences toxic effects on human keratinocyte cells. In addition, all the acylglutamate surfactants tested on human keratinocyte cells demonstrated significantly less cytotoxicity (when irradiated and non-irradiated with ultraviolet B light; p < 0.05) and no phototoxic potential was observed in any of the acylglutamate surfactants, when compared with the positive control chlorpromazine. In conclusion, the in vitro studies confirm the suitability of sodium lauroyl glutamate destined for the synthesis and stabilization of lipid nanoparticles.
1. Introduction
In cosmeceuticals, surfactant has several applications, such as cleansing, foaming wetting, thickening, solubilizing, and conditioning. In addition to these applications, surfactant is also used for the synthesis and stabilization of lipid nanoparticles for improved delivery of cosmeceutical actives [1,2,3]. Cosmeceutical products were frequently designed in order to retain the agents on the skin in spite of rinsing, and the recurrent use of phototoxic surfactant containing cosmeceuticals to the skin would inevitably increase risk. Moreover, the disposal of these products can adversely affect the aquatic environment, their biocompatibility and biodegradability become virtually as important as their functional performance to the consumer. Hence, there is a predilection for utilizing biocompatible and biodegradable surfactants for cosmeceutical formulations [4]. Surfactant of this type can be achieved by designing molecules that mimic natural amphiphilic structures (Nα-acyl amino acids, phopholipids and alkyl-glycosides) [5,6,7]. Surfactant molecules from renewable crude substance (such as amino acids) that mimic natural lipoamino acids are one of the preferred choices for cosmeceutical formulations. The importance of amino acid as crude substance for the synthesis of surfactant came into existence immediately after they were discovered in 1930 [8]. Acylglutamate surfactants are remarkably superior to conventional surfactants in characteristic features. They have an excellent skin moisturization effect without a taut feeling, ability to improve conventional surfactant mildness, and usable even by patients suffering from skin disease [9,10,11,12].
Until 11 March 2013, experimentation of finished cosmetic products and ingredients was prohibited in the EU (European Union), but the 7th amendment (Directive 2003/15/EC [13]) to the cosmetic directive (Directive 76/768/EEC [13]) has now placed a Europe-wide prohibition on the sale of cosmetics if they have been experimented on animals anywhere on the globe. The main aim of the new directive is to safeguard and enhance the welfare of animals for experimental and research purposes by supporting the development of alternative methods, as well as to firmly anchor the principle of “3Rs”, to replace, reduce and refine the use of animals [14]. As a result, Over the past two decades, many different types of cell and tissue culture based in vitro models have been developed, not only in response to the need to find ethical alternatives to the animal-based tests, but also because many in vitro models allow one to obtain more relevant, objective test results [15].
In the current study we chose a human keratinocyte (HaCaT) cell line, as keratinocytes represent the major cell type in the epidermis and in vivo keratinocytes are biological relevant targets for surfactants once they pass through the stratum corneum. Furthermore, this cell line provides a virtually endless supply of look-alike cells, assuring immense reproducibility across-laboratory and is of greater human relevance to epidermal-induced irritation, than animal derived fibroblast cells [16,17]. Earlier studies reported that HaCaT cells are a promising screening tool for predicting the toxic and irritation potential of amino acid-based surfactants, anionic surfactants, certain phytochemicals, and lipid nanocapsules [18,19,20,21].
Phototoxicity is of growing concern in dermatology due to stratospheric ozone depletion resulting in an increasing penetration of ultraviolet (UV) radiation. Particularly, ultraviolet B (UVB; mid wave, 290–320 nm) irradiation is thought to be responsible for more of the UV-induced adverse effects in the skin and serves as an important etiologic factor causing inflammatory skin damages, oxidative stress, DNA damage, cellular, as well as tissue injuries, cell death, skin cancer, and premature skin aging [22,23]. On that account, the ultimate goal of the current study was to elucidate the effects of the fatty acid chain length of acylglutamate surfactants on their cytotoxicity and the UVB-induced phototoxicity using the HaCaT cell line. Ultimately, it was foreseen that the results from this study generate the first set of data which would allow us to (a) categorize acylglutamte surfactants into non-phototoxic, probable phototoxic and phototoxic, (b) pick the most suitable surfactant for the synthesis and stabilization of lipid nanoparticles (unpublished data).
2. Experimental Section
2.1. Chemicals and Reagents
Chlorpromazine (CPZ), crystal violet and dimethyl sulfoxide (DMSO) were obtained from Sigma (St. Louis, MO, USA). Dulbecco’s-Modified Eagle’s Medium (DMEM), l-glutamine, antibiotics, fetal bovine serum (FBS) and phosphate-buffered saline (PBS) and trypsin were purchased from Gibco®, life technologies (Grand Island, NY, USA). 75 cm2 tissue culture flask and flat-bottom polystyrene tissue culture treated 96 well-tissue culture plates were procured from BD Falcon (Becton Dickinson and Company, Franklin Lakes, NJ, USA).
2.2. Surfactants Tested
Anionic amino acid-based, weakly acidic surfactants (Amisoft®, Acylglutamte salt), namely sodium cocoyl glutamate (CS-11), sodium lauroyl glutamate (LS-11) and sodium myristoyl glutamate (MS-11) were generously supplied as gift sample by Western Commercial Corporation (Mumbai, India). Physico-chemical properties of each acylglutamate surfactant tested are shown in Table 1.
Each acylglutamate surfactant solution was prepared by dissolving them in serum-free DMEM supplemented with 2 mM l-glutamine and antibiotic mixture. The CPZ stock solution was prepared in DMSO and further dilutions by serum free DMEM.

Table 1.
Physico-chemical properties of acylglutamate surfactants.
| Surfactant | Average MW | Base amino acid | Charge | Fatty acid chain length (C % by weight) | Foaming power a (After 5 min) mm |
|---|---|---|---|---|---|
| Amisoft CS 11 | 359 | l-glutamic acid | Anionic | C8–14 Mixture (86.9) b | 203 |
| Amisoft LS 11 | 356 | l-glutamic acid | Anionic | C12 (97.3) c | 219 |
| Amisoft MS 11 | 384 | l-glutamic acid | Anionic | C14 (97.8) c | 212 |
MW, Molecular weight; C, Carbon; mm, Milimeter; CS 11, sodium cocoyl glutamate; LS 11, sodium lauroyl glutamate; MS 11, sodium myristoyl glutamate. a Adapted from [12]; b Adapted from [24]; c Adapted from [25].
2.3. Cell Culture
Early-passage HaCaT cell line was acquired from National Center for Cell Sciences (NCCS, Pune, India) was grown as adherent cultures into 75 cm2 culture flasks using, DMEM high glucose (4.5 g/L) as base medium, supplemented with 10% heat-inactivated FBS, 2mM l-glutamine, antibiotics (penicillin 10,000 units/mL and streptomycin 10,000 μg/mL) and maintained at 37 °C with 5% CO2, 95% air in a humidified incubator (model 3193, Forma Scientific Inc., Marietta, OH, USA). When cell line reached nearly 80% confluence, the culture media were removed from the flask and the cells were briefly rinsed with 10 mL of PBS pH 7.4, harvested with 0.25% trypsin and was then used for further experiments.
2.4. UVB-Irradiation and Cell Viability
For cell viability experiment, cells were seeded 100 μL/well from a density of 2 × 105 cells/mL, into 96 well-tissue culture plate and then allowed to attach overnight in an incubator. After 24 h, cells were exposed to UVB light (13, 27, 41, 55, 83, 111, and 138 μW/cm2, which corresponds exactly to 50, 100, 150, 200, 300, 400 and 500 mJ/cm2) without plastic lid. The μW/cm2 doses were achieved after 60 min of irradiation time using CL1000M UV Crosslinker (UVP, Upland, CA, USA). The irradiance was measured using a UV dosimeter (UVX Digital Radiometer, UVX-31 sensor, UVP). To prevent UVB light absorption by the cell culture medium, the medium was removed just prior to irradiation and replenished with a thin layer of PBS to cover the cells [26]. After the UVB irradiation, cells were replenished with fresh cell culture medium and incubated for 24 h. Later, cell viability was assessed by MTS (3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium inner salt) assay (Section 2.6). Non-irradiated cells were served as control. No quantifiable rise in temperature was recorded during the irradiation.
For colony formation assay, cells were seeded (2 × 105 cells/mL), into 24 well-tissue culture plate, allowed to attach overnight and later irradiated. After 240 h (cell media was changed every 48 h), cells were fixed with 3.7% formaldehyde (in PBS) and stained with crystal violet (0.05% in PBS). Excess crystal violet stain was rinsed off by PBS (twice), and air-dried.
2.5. Surfactant Treatment
Cells were seeded 100 μL/well from a density of 2 × 105 cells/mL, into the central 60 wells of 96 well-tissue culture plate. The peripheral wells of plate received 100 μL of supplemented DMEM alone (no cells = blank). One column was used as negative control (cells in supplemented DMEM) and then allowed to attach overnight in an incubator.
After 24 h of attachment, the culture media was decanted and cells were rinsed with 150 μL of culture media used for incubation. The HaCaT cell line was exposed to100 μL of different concentrations of acylglutamate surfactants and CPZ solutions (1, 5, 10, 20, 40, 80, 160 and 320 μg/mL, pH 7.2), followed by incubation for 24 h and cell viability was assessed by MTS assay.
2.6. MTS Assay
Cytotoxicity was evaluated using a ready-to-use The CellTiter 96® Aqueous One Solution Cell Proliferation Assay (MTS) (Promega, Madison, WI). This colorimetric assay is fast, convenient, easy-to-use in comparison with MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide). MTS is bioreduced by cells into a colored formazan product, which is soluble in tissue culture media [27,28].
After 24 h of incubation, 20 μL of MTS solution (according to a protocol proposed by the manufacturer) was added directly into each well of 96-well assay plate containing acylglutamate surfactants and CPZ solutions followed by additional 3 hours of incubation. Finally, the cell viability was quantified by recording the formazan absorbance at 490 nm using a Bio-Rad microplate reader (Model 680, Hercules, CA, USA). Since the absorbance directly correlated with the number of viable cells, the percent viability was calculated from the absorbance.
2.7. Photoirritation Evaluation
The HaCaT cell line was utilized as in vitro model cell system to predict the cutaneous photoirritation [17], determined by MTS assay. Briefly, two 96-well plates were prepared for each surfactant (as per Section 2.5). One plate (without plastic lid) was irradiated (+UVB) at 200 mJ/cm2 for 60 min using CL-1000M UV Crosslinker (UVP) and the other non-irradiated plate (−UVB) was wrapped in foil and place into the crosslinker [29]. Measures were taken to prevent UVB light absorption by the cell culture medium (as described in Section 2.4). After the UVB irradiation, cells were replenished with fresh cell culture medium and incubated for 24 h. Later, cell viability was assessed by MTS assay (Section 2.6).
2.8. Data Analysis
The data obtained from cytotoxicity and photoirritation evaluation was fitted into two models, namely the Photo-Irritation Factor (PIF) and Mean Photo Effect (MPE) is commonly known as predictors for the photoirritation potential of a diverse set of surfactants. The PIF is calculated by dividing the IC50 (the concentration of the surfactant by which the cell viability is reduced by 50%) of −UVB by the IC50 of +UVB and MPE aims to overcome the obvious limitations in the application of the PIF, by comparing the value of the photo effect between two curves at arbitrary doses. The value obtained from two models allows categorize a surfactant into “no photoirritant” (PIF < 2 or MPE < 0.1), “probable photoirritant” (PIF > 2 and <5 or an MPE > 0.1 and <0.15) and “photoirritant” (PIF > 5 or an MPE > 0.15) [30]. The IC50, PIF and MPE values were determined using software, Phototox (ver. 2.0) available from the Secretariat [31]. This program module performs a bootstrap resampling of the original concentration-response data, which results in a set of new computer-generated concentration-response data and can be regarded as equiprobable realizations of the “true” concentration-response data hidden in the experimental observations [32].
2.9. Statistical Analysis
In present study, the series of experiments were performed at least in three independent examinations (same day) with six replicates for each surfactant and data were displayed as mean ± standard deviation (SD). All data compared by one-way ANOVA (analysis of variance) followed by tukey post hoc test (GraphPad Prism 6.0 Software, San Diego, CA, USA). p-value < 0.05 was regarded as statistical significant.
3. Results
To examine the outcome of UVB on HaCaT cell proliferation rates, they were exposed to UVB at doses ranging from 50 to 500 mJ/cm2 and later the viable cells were assessed by MTS assay and colony formation assay. As depicted in Figure 1A,B the viability rate of HaCaT was substantially decreased in dose-dependent manner (P < 0.05) of UVB irradiation. Our data showed that 200 mJ/cm2 UVB dose induced almost 50% cell death compared to the control.
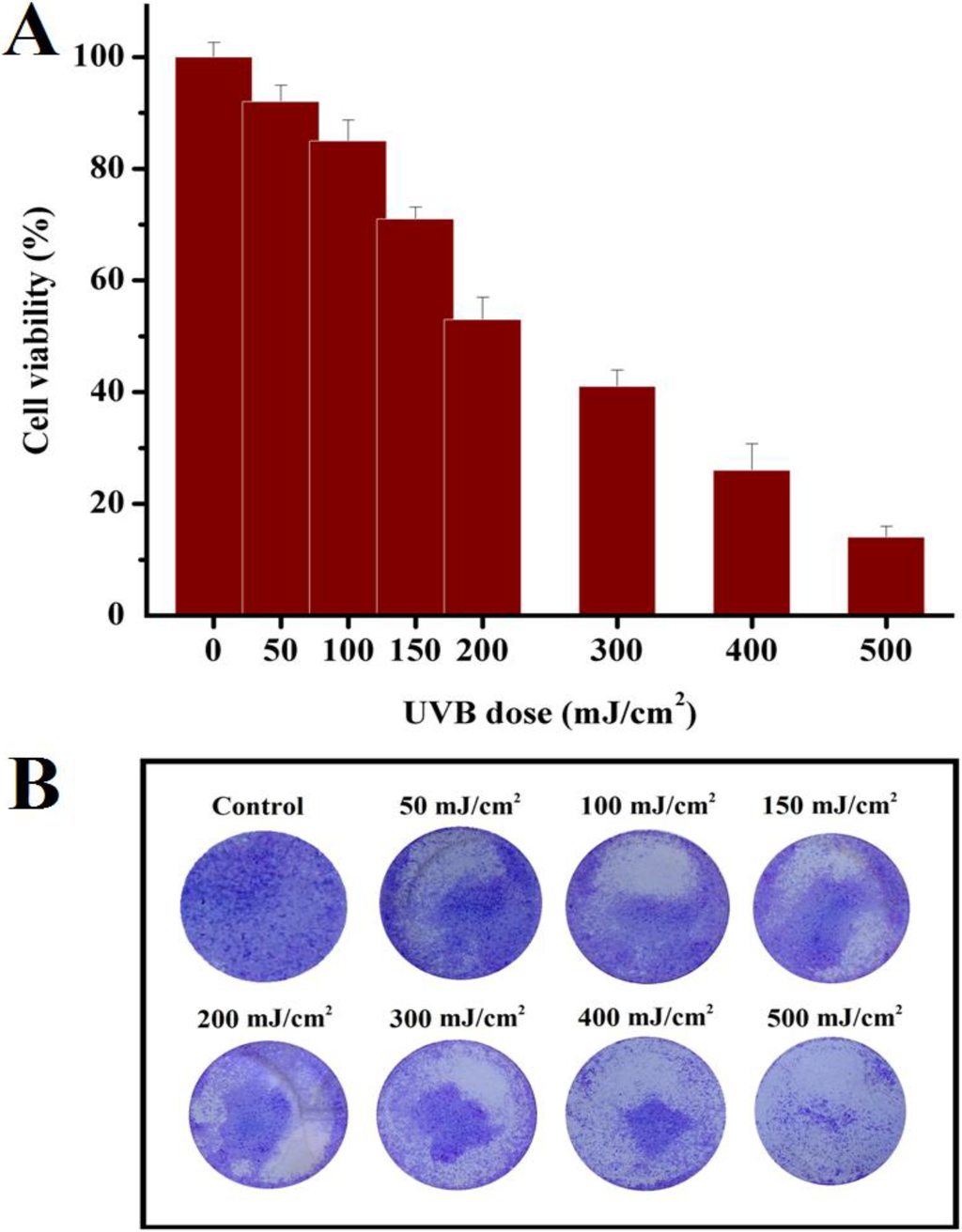
Figure 1.
Cell proliferation rate in UVB irradiated HaCaT cells. (A) Viability assessed by MTS assay 24 h after UVB irradiation. The bar graph depicts the percent cell viability compared to control and represent the mean percentage ± S.D. (standard deviation) of three independent experiments. (B) Colony formation assay. Representative photomicrographs of survival colonies after different doses of UVB, stained with crystal violet solution. Similar results were obtained in two independent experiments.
Figure 1.
Cell proliferation rate in UVB irradiated HaCaT cells. (A) Viability assessed by MTS assay 24 h after UVB irradiation. The bar graph depicts the percent cell viability compared to control and represent the mean percentage ± S.D. (standard deviation) of three independent experiments. (B) Colony formation assay. Representative photomicrographs of survival colonies after different doses of UVB, stained with crystal violet solution. Similar results were obtained in two independent experiments.
The calculated IC50 values for acylglutamate surfactant on HaCaT cells with −UVB (blue points depict data and gray curve depicts statistical curve fitting by phototox software) [32] and +UVB (yellow points depict data and black curve depicts statistical curve fitting by phototox software) [32] exposed group were higher (Table 2, Figure 2A–C) compared to the CPZ (+UVB 1.054 ± 0.23, −UVB 16.00 ± 0.89,) (Figure 2D), also significant differences (p < 0.05) were observed among the groups compared. The IC50 values for acylglutamate surfactants were in the descending order: LS 11 > CS 11 > MS 11.
The results of photoirritation evaluation suggest that all acylglutamate surfactant tested were non-phototoxic with +UVB, which is reflected in its low PIF mean and MPE values (PIF < 2 and MPE < 0.1) (Table 3). Whereas, the resulting PIF mean and MPE values for CPZ tested on HaCaT cells were found to be 15.186 and 0.435, respectively, exhibiting the toxicity potential of a CPZ in the presence of UVB light (Table 3).

Table 2.
IC50 values of acylglutamate surfactants and chlorpromazine (CPZ) on human keratinocyte (HaCaT) cells.
| Surfactant | UVB light | IC50 a ± SD (μg/mL) |
|---|---|---|
| Amisoft CS 11 | − | 43.48 ± 1.09 |
| Amisoft CS 11 | + | 30.62 ± 2.72 |
| Amisoft LS 11 | − | 69.97 ± 1.66 |
| Amisoft LS 11 | + | 54.22 ± 1.81 |
| Amisoft MS 11 | − | 26.48 ± 1.47 |
| Amisoft MS 11 | + | 16.72 ± 0.92 |
| CPZ | − | 16.00 ± 0.89 |
| CPZ | + | 1.054 ± 0.23 |
a Values represent the mean ± standard deviation of three independent examinations, each performed with six replicates. CS 11, sodium cocoyl glutamate; LS 11, sodium lauroyl glutamate; MS 11, sodium myristoyl glutamate; CPZ, chlorpromazine; “−” indicate UVB non-irradiated; “+” indicate UVB irradiated at 200 mJ/cm2 for 60 min; IC50, the concentration of the surfactant by which the cell viability is reduced by 50%; SD, standard deviation.
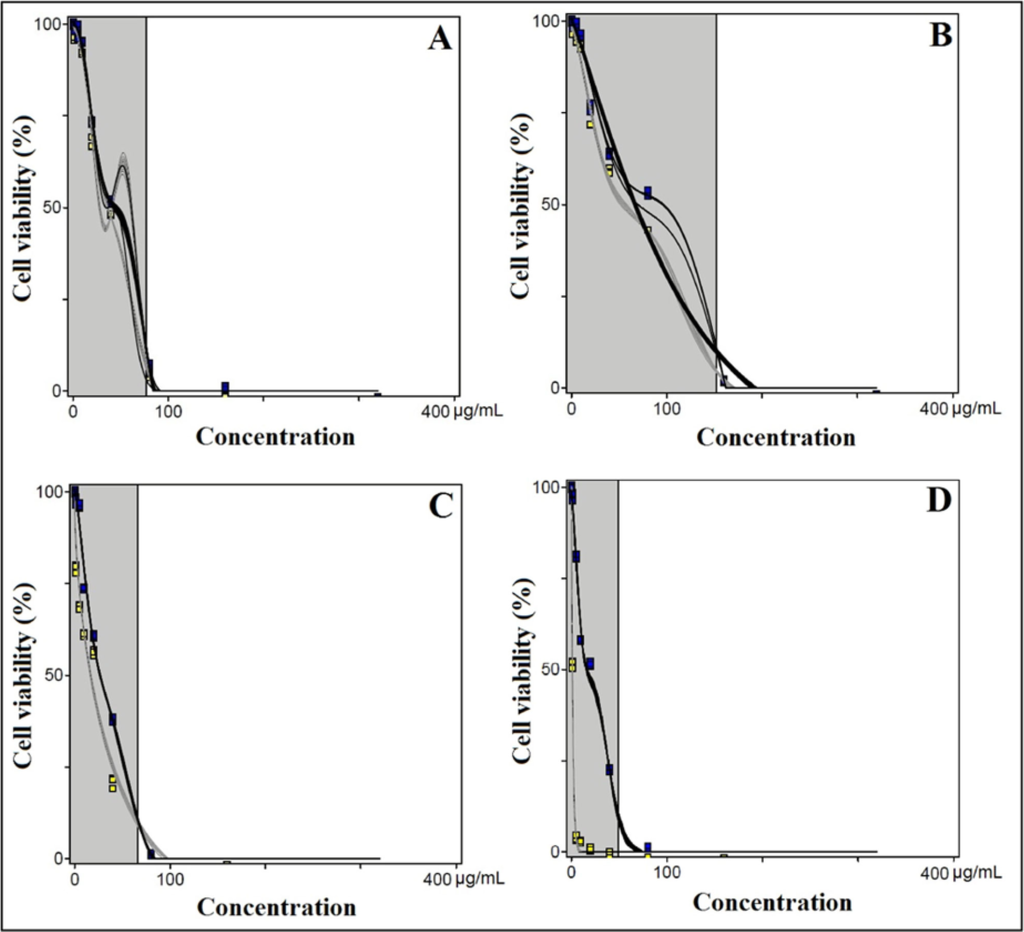
Figure 2.
Effect of increasing concentration of (A) CS-11; (B) LS-11; (C) MS-11 and (D) CPZ on cell viability of HaCaT cells. Yellow and blue points represent the original concentration-response data from +UVB and −UVB, respectively whereas, the black and gray curves depict statistical curve fitting by photox software. CS 11 indicates sodium cocoyl glutamate; LS 11, sodium lauroyl glutamate; MS 11, sodium myristoyl glutamate; CPZ, chlorpromazine; +UVB irradiated at 200 mJ/cm2 for 60 minutes; −UVB non-irradiated. (For interpretation of the references to color in this figure legend, the reader is referred to the web version of this article).
Figure 2.
Effect of increasing concentration of (A) CS-11; (B) LS-11; (C) MS-11 and (D) CPZ on cell viability of HaCaT cells. Yellow and blue points represent the original concentration-response data from +UVB and −UVB, respectively whereas, the black and gray curves depict statistical curve fitting by photox software. CS 11 indicates sodium cocoyl glutamate; LS 11, sodium lauroyl glutamate; MS 11, sodium myristoyl glutamate; CPZ, chlorpromazine; +UVB irradiated at 200 mJ/cm2 for 60 minutes; −UVB non-irradiated. (For interpretation of the references to color in this figure legend, the reader is referred to the web version of this article).

Table 3.
PIF mean and MPE mean values of acylglutamate surfactants and CPZ on HaCaT cells.
| Surfactant | PIF mean a | Toxicity probability | MPE mean a | Toxicity probability | Phototoxic potential |
|---|---|---|---|---|---|
| Amisoft CS 11 | 1.292 | 0 | 0.019 | 0 | No |
| Amisoft LS 11 | 1.442 | 0 | 0.007 | 0 | No |
| Amisoft MS11 | 1.585 | 0 | 0.092 | 0.055 | No |
| CPZ | 15.186 | 1.0 | 0.435 | 1.0 | Yes |
a PIF and MPE values are averages over all bootstrap pairs. CS 11, sodium cocoyl glutamate; LS 11, sodium lauroyl glutamate; MS 11, sodium myristoyl glutamate; CPZ, chlorpromazine; PIF, photo-irritation factor; MPE, mean photo effect.
4. Discussion
The acylglutamate surfactants in all likelihood segregate into polar amino acids (hydrophilic moiety) and non polar fatty acids (hydrophobic moiety) in the presence of water. Since most of these amino acids and fatty acids are found in the foods, oral toxicity is not expected. Dermal toxicity as a result of dermal exposure is not expected to be different from oral exposure [33]. Cutaneous phototoxicity of acylglutamate surfactants is of concern, and the central focus of this paper.
Cutaneous phototoxicity is an “acute” reaction caused by a single treatment with skin irritants, such as UV radiation and surfactant. In vivo, this reaction can be elicited in all subjects provided the suitable dose of radiation and concentration of surfactants. “Acute” include immediate as well as delayed (after 48 h) reactions. Conforming to the diverse classes and chemical structure of surfactants with toxic potential, it seems exceedingly possible that several pathways must be involved in cutaneous irritation. Generally, two different pathways (either alone or in combination) are associated with surfactants which can commence and regulate the cutaneous toxicity. First, by damaging the barrier function of the stratum corneum and second, by straightforward effects of surfactant on cutaneous cells [34].
In this study, we first examined HaCaT cell viability following UVB irradiation, and found that cell viability was related with UVB dose. The observed results are in accordance with previous results [35], which state that maintaining genomic integrity during cellular proliferation is essential for the continued viability of the organism. G2-M checkpoint makes certain that cells don’t start mitosis before they have a chance to repair damaged DNA after replication. Cells that have a defective G2-M checkpoint enter mitosis before repairing their DNA. When cells bypass the normal restrictions on entrance into S phase imposed by DNA damage, the replication of damaged DNA can either result in cell death or an accumulation of genetic changes leading ultimately to cancer. UVB light is known to damage DNA and therefore cells expose to single dose of 200 mJ/cm2 UVB causes G2-M arrest. Moreover, this dose corresponds to 15 min–4 h of solar irradiation [36], a common period of time for out door exposure. Hence, we selected this dose when performing photoirritation experiments examining the effect of acylglutamate surfactants.
Much more is known about the phototoxicity of CPZ and is one of the classic phototoxicants of the HaCaT cells [37]. Upon UV irradiation CPZ produces a variety of free radicals along with the corresponding cation radical, the neutral promazinyl radical, a chlorine radical and super-centered peroxy radical. The neutral promazinyl radical can react covalently with cell membrane, protein, and other macromolecules to yield antigens which could be responsible for the photoallergic response to CPZ resulting phototoxicity, both in vitro and in vivo [38]. Hence, CPZ was used as a positive control in the current study and as expected exhibited the highest cytotoxic and phototoxic activity on HaCaT cell among all surfactant tested. In the current study we also observed that PIF mean and MPE values of CPZ were in the range of the reference values (PIF > 14.4 and MPE 0.33–0.63) indicating that the irradiation system and method for measuring phototoxicity of acylglutamate surfactants were appropriate [30]. This was achieved because we used a wider concentration range of CPZ for both the +UVB and −UVB group.
The data obtained from current studies revealed that the increased cytotoxicity of MS 11 is due to its larger fatty acid chain length (C14, 97.8%) in comparison with shorter fatty acid chain length of LS 11 (C12, 97.3%) and CS 11 (C12, 57.8%). A possible explanation for this is that, as the alkyl side chain in the amino acid becomes larger, the sodium salts of acylglutamate cannot pack closely at water surfaces. As a result, their capacity to lower surface tension becomes less. On the contrary, the overall hydrophobicities of the molecules become larger [12,39]. Hence, causing greater amounts of surfactant adsorbed on the HaCaT cell membrane, leading to cellular disintegration, eventually cell death and the concomitant release of keratinocyte cytoplasm containing the proinflammatory cytokine, IL-1α [40,41].
The non-phototoxic nature of all acylglutamate surfactant is also due to the presence of glutamic acid in it and glutamic acid is one of the precursors for the synthesis of glutathione (GSH). GSH is the main protective mechanism against UVB radiation, as it is the only donor of hydrogen in H2O2 neutralization thus preventing damage to important cellular components [42]. Thus, signify that higher the GSH level in HaCaT cell is expected to confer resistance to UVB irradiation [43].
5. Conclusion
In the light of cytotoxicity and phototoxicity results, we could conclude that shorter the fatty acid chain length of acylglutamate surfactants, the milder it functions on HaCaT cell line and vice versa. In addition, both amino acid and fatty acid are valuable for the barrier effect of the skin cells. Considering their biocompatible properties and low cytotoxicity profile, the sodium lauroyl glutamate would be the most suitable surfactant for the synthesis and stabilization of lipid nanoparticles for improved delivery of various cosmeceutical actives.
Acknowledgements
Varsha Pokharkar would like to thank the University Grants Commission, New Delhi, India, for providing financial assistance in terms of Major Research Project [F.35-101/2008 (SR)].
Author Contributions
Abhay Kyadarkunte and Varsha Pokharkar designed the entire experiment, verified and analyzed the study results, wrote the initial draft of the manuscript and approved the revised version of the manuscript. Milind Patole provided the necessary inputs and cell-culture facilities to perform experiments.
Conflicts of interest
The authors declare that there are no conflicts of interest.
References
- Liu, J.; Hu, W.; Chen, H.; Ni, Q.; Xu, H.; Yang, X. Isotretinoin-loaded solid lipid nanoparticles with skin targeting for topical delivery. Int. J. Pharm. 2007, 328, 91–195. [Google Scholar]
- Kovacevic, A.; Savic, S.; Vuleta, G.; Müller, R.H.; Keck, C.M. Polyhydroxy surfactants for the formulation of lipid nanoparticles (SLN and NLC): Effects on size, physical stability and particle matrix structure. Int. J. Pharm. 2011, 406, 163–172. [Google Scholar]
- Mitri, K.; Shegokar, R.; Gohla, S.; Anselmi, C.; Müller, R.H. Lipid nanocarriers for dermal delivery of lutein: Preparation, characterization, stability and performance. Int. J. Pharm. 2011, 414, 267–275. [Google Scholar] [CrossRef]
- Somasundaran, P.; Soma, C.; Puspendu, D.; Namita, D.; Somasundaran, T. Contribution of surfactants to personal care products. In Surfactant in Personal Care Products and Decorative Cosmetics, 3rd ed.; Rhein, L.D., Olenick, A., Schlossman, M., Somasundaran, P., Eds.; CRC Press: Florida, FL, USA, 2007; pp. 121–135. [Google Scholar]
- Infante, M.R.; Molinero, J.; Erra, P.; Juliá, M.R.; García Domínguez, J.J.; Robert, M. The influence of steric configuration of some Nα-lauroyl amino-acid derivatives on their antimicrobial activity. Fette Seifen Anstrichm. 1986, 88, 108–110. [Google Scholar] [CrossRef]
- Okahata, Y.; Tanamachi, S.; Nagai, M.; Kunitake, T. Synthetic bilayer membranes prepared from dialkyl amphiphiles with nonionic and zwitterionic head groups. J. Colloid Interface Sci. 1981, 82, 401–417. [Google Scholar]
- Kida, T.; Morishima, N.; Masuyama, A.; Nakatsuji, Y. New cleavable surfactants derived from glucono-1,5-lactone. J. Am. Oil Chem. Soc. 1994, 71, 705–710. [Google Scholar] [CrossRef]
- Heutrich, W.; Keppler, H.; Hintzmann, K. Detergents, Wetting, Dispersing and Leveling Agents. German Patent 635522, 18 September 1936. [Google Scholar]
- Amisoft® CS 22. Amino Acid Based Anionic Surfactant. Available online: www.cosmesi.it/portals/7/documenti/amisoft%20CS-22_brochure.pdf (accessed on 15 April 2014).
- Kawasaki, Y.; Quan, D.; Sakamoto, K.; Cooke, R.; Maibach, H.I. Influence of surfactant mixtures on intercellular lipid fluidity and skin barrier function. Skin Res. Technol. 1999, 5, 96–101. [Google Scholar] [CrossRef]
- Lee, C.H.; Kawasaki, Y.; Maibach, H. Effect of surfactant mixtures on irritant contact dermatitis potential in man: Sodium lauroyl glutamate and sodium lauryl sulphate. Contact Dermat. 1994, 30, 205–209. [Google Scholar] [CrossRef]
- Takehara, M. Properties and applications of amino acid based surfactants. Colloids Surf. 1989, 38, 149–167. [Google Scholar]
- Cosmetic Directive. Available online: http://ec.europa.eu/consumers/archive/sectors/cosmetics/documents/directive/index_en.htm (accessed on 8 July 2014).
- Full EU Ban on Animal Testing for Cosmetics Enters into Force. Available online: http://europa.eu/rapid/press-release_IP-13-210_en.htm (accessed on 15 April 2014).
- Botham, P.A.; Earl, L.K.; Fentem, J.H.; Roguet, R.; van de Sandt, J.J.M. Alternative Methods for Skin Irritation Testing: The Current Status. Altern. Lab. Anim. 1998, 26, 195–211. [Google Scholar]
- Wilhelm, K.P.; Böttjer, B.; Siegers, C.-P. Quantitative assessment of primary skin irritants in vitro in a cytotoxicity model: Comparison with in vivo human irritation tests. Br. J. Dermatol. 2001, 145, 709–715. [Google Scholar]
- Benavides, T.; Martínez, V.; Mitjans, M.; Infante, M.R.; Moran, C.; Clapés, P.; Clothier, R.; Vinardell, M.P. Assessment of the potential irritation and photoirritation of novel amino acid-based surfactants by in vitro methods as alternative to the animal tests. Toxicology 2004, 201, 87–93. [Google Scholar]
- Sanchez, L.; Mitjans, M.; Infante, M.R.; Vinardell, M.P. Potential irritation of lysine derivative surfactants by hemolysis and HaCaT cell viability. Toxicol. Lett. 2006, 161, 53–60. [Google Scholar]
- Wilheilm, K.P.; Samblebe, M.; Siegers, C.P. Quantitative in vitro assessment of N-alkylsulphate-induced cytotoxicity in human keratinocytes (HaCaT). Comparison with in vivo human irritation tests. Br. J. Dermatol. 1994, 130, 18–23. [Google Scholar] [CrossRef]
- Lohézic-Le Dévéhat, F.; Legouin, B.; Couteau, C.; Boustie, J.; Coiffard, L. Lichenic extracts and metabolites as UV filters. J. Photochem. Photobiol. B 2013, 120, 17–28. [Google Scholar]
- Maupas, C.; Moulari, B.; Beduneau, A.; Lamprecht, A.; Pellequer, Y. Surfactant dependent toxicity of lipid nanocapsules in HaCaT cells. Int. J. Pharm. 2011, 411, 136–141. [Google Scholar] [CrossRef]
- Nichols, J.A.; Katiyar, S.K. Skin photoprotection by natural polyphenols: Anti-inflammatory, antioxidant and DNA repair mechanisms. Arch. Dermatol. Res. 2010, 302, 71–83. [Google Scholar] [CrossRef]
- Kim, J.K.; Kim, Y.; Na, K.M.; Surh, Y.J.; Kim, T.Y. [6]-Gingerol prevents UVB-induced ROS production and COX-2 expression in vitro and in vivo. Free Radic. Res. 2007, 41, 603–614. [Google Scholar] [CrossRef]
- Yoshida, R.; Yoshimurra, I.; Usuba, Y.; Shibue, A. Surface active N-acylglutamate: V. Application of N-acylglutamates to detergent bars. J. Am. Oil Chem. Soc. 1976, 53, 113–117. [Google Scholar] [CrossRef]
- Takehara, M.; Moriyuki, H.; Yoshimura, I.; Yoshida, R. Surface active N-acylglutamate: II. Physicochemical properties of long chain N-acylglutamic acids and their sodium salts. J. Am. Oil Chem. Soc. 1972, 49, 143–150. [Google Scholar]
- Park, K.; Lee, J.H. Protective effects of resveratrol on UVB-irradiated HaCaT cells through attenuation of the caspase pathway. Oncol. Rep. 2008, 19, 413–417. [Google Scholar]
- Cory, A.H.; Owen, T.C.; Barltrop, J.A.; Cory, J.G. Use of an aqueous soluble tetrazolium/formazan assay for cell growth assays in culture. Cancer Commun. 1991, 3, 207–212. [Google Scholar]
- Yin, J.J.; Liu, J.; Ehrenshaft, M.; Roberts, J.E.; Fu, P.P.; Mason, R.P.; Zhao, B. Phototoxicity of nano titanium dioxides in HaCaT keratinocytes--generation of reactive oxygen species and cell damage. Toxicol. Appl. Pharm. 2012, 263, 81–88. [Google Scholar]
- Vinardell, M.P.; Benavides, T.; Mitjans, M.; Infante, M.R.; Clapés, P.; Clothier, R. Comparative evaluation of cytotoxicity and phototoxicity of mono and diacylglycerol amino acid-based surfactants. Food Chem. Toxicol. 2008, 46, 3837–3841. [Google Scholar]
- Test No. 432: In vitro 3T3 NRU phototoxicity test. In OECD Guidelines for the Testing of Chemicals Section 4: Health Effects; Organisation for Economic Cooperation and Development (OECD) iLibrary: Paris, France, 2004; pp. 1–15.
- OECD. Available online: http://www.oecd.org/document/55/0,2340,en_2649_34377_2349687_1_1_1_1,00.html (accessed on 1 April 2014).
- Peters, B.; Holzhutter, H.G. In vitro phototoxicity testing: development and validation of a new concentration response analysis software and biostatistical analyses related to the use of various prediction models. Altern. Lab. Anim. 2002, 30, 415–432. [Google Scholar]
- Safety Assessment of Amino Acid Alkyl Amides as Used in Cosmetics. Available online: www.cir-safety.org/sites/default/files/alkyl_amides_0.pdf (accessed on 4 April 2014).
- Berardesca, E.; Distante, F. The modulation of skin irritation. Contact Dermat. 1994, 31, 281–287. [Google Scholar] [CrossRef]
- Athar, M.; Kim, A.L.; Ahmad, N.; Mukhtar, H.; Gautier, J.; Bickers, D.R. Mechanism of ultraviolet B-induced cell cycle arrest in G2/M phase in immortalized skin keratinocytes with defective p53. Biochem. Biophys. Res. Commun. 2000, 277, 107–111. [Google Scholar] [CrossRef]
- Kudish, A.I.; Lyubansky, V.; Evseev, E.G.; Ianetz, A. Statistical analysis and inter-comparison of the solar UVB, UVA and global radiation for Beer Sheva and Neve Zohar (Dead Sea), Israel. Theor. Appl. Climatol. 2005, 80, 1–15. [Google Scholar]
- Suh, Y.-W. An Investigation of the Phototoxicity of Decabromodiphenyl Ether and Triclosan. Ph.D. Thesis, University of Iowa, Iowa City, IA, USA, 2010. [Google Scholar]
- Chignell, C.F.; Motten, A.G.; Buettner, G.R. Photoinduced free radicals from chlorpromazine and related phenothiazines: Relationship to phenothiazine-induced photosensitization. Environ. Health Perspect. 1985, 64, 103–110. [Google Scholar] [CrossRef]
- Kanari, M.; Kawasaki, Y.; Sakamoto, K. Acylglutamate as an anti-irritant for mild detergent system. J. Soc. Cosmet. Chem. Jpn. 1993, 27, 498–505. [Google Scholar]
- Osborne, R.; Perkins, M.A. An approach for development of alternative test methods based on mechanisms of skin irritation. Food Chem. Toxicol. 1994, 32, 133–142. [Google Scholar]
- De Brugerolle de Fraissinette, A.; Picarles, V.; Chibout, S.; Kolopp, M.; Medina, J.; Burtin, P.; Ebelin, M.E.; Osborne, S.; Mayer, F.K.; Spake, A.; et al. Predictivity of an in vitro model for acute and chronic skin irritation (SkinEthic) applied to the testing of topical vehicles. Cell Biol. Toxicol. 1999, 15, 121–135. [Google Scholar] [CrossRef]
- Afaq, F.; Mukhtar, H. Effects of solar radiation on cutaneous detoxification pathways. J. Photochem. Photobiol. B 2001, 63, 61–69. [Google Scholar] [CrossRef]
- Leccia, M.T.; Richard, M.J.; Joanny-Crisci, F.; Beani, J.C. UV-A1 cytotoxicity and antioxidant defence in keratinocytes and fibroblasts. Eur. J. Dermatol. 1998, 8, 478–482. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
