Is the Microfiltration Process Suitable as a Method of Removing Suspended Solids from Rainwater?
Abstract
1. Introduction
2. Materials and Methods
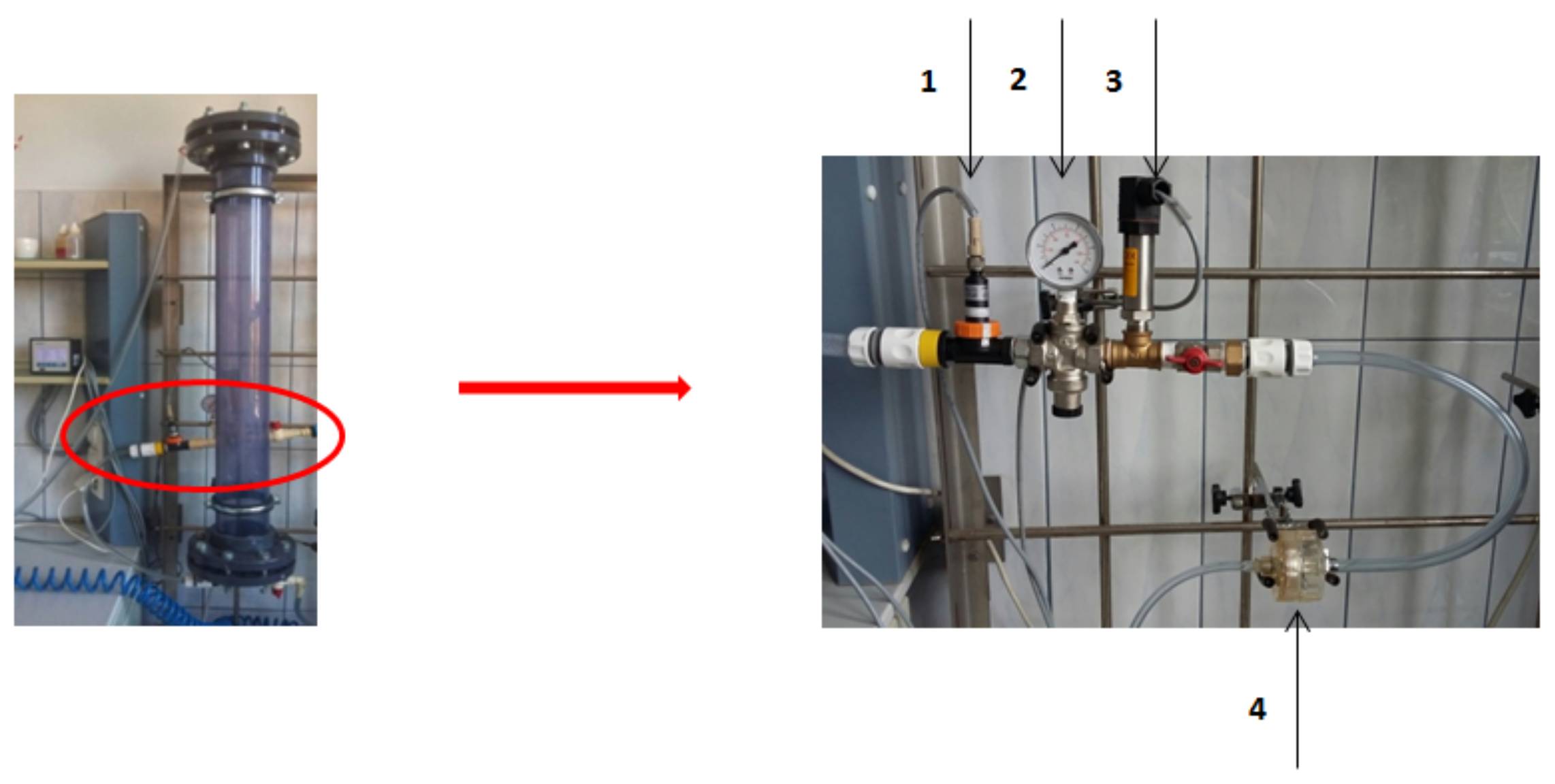
2.1. Analyzed Object and Samples Collection
2.2. Methods
2.2.1. Total Suspended Solids Determination
2.2.2. Turbidity Measurements
2.2.3. Particle Size Analysis
2.2.4. Silt Density Index Test
3. Results
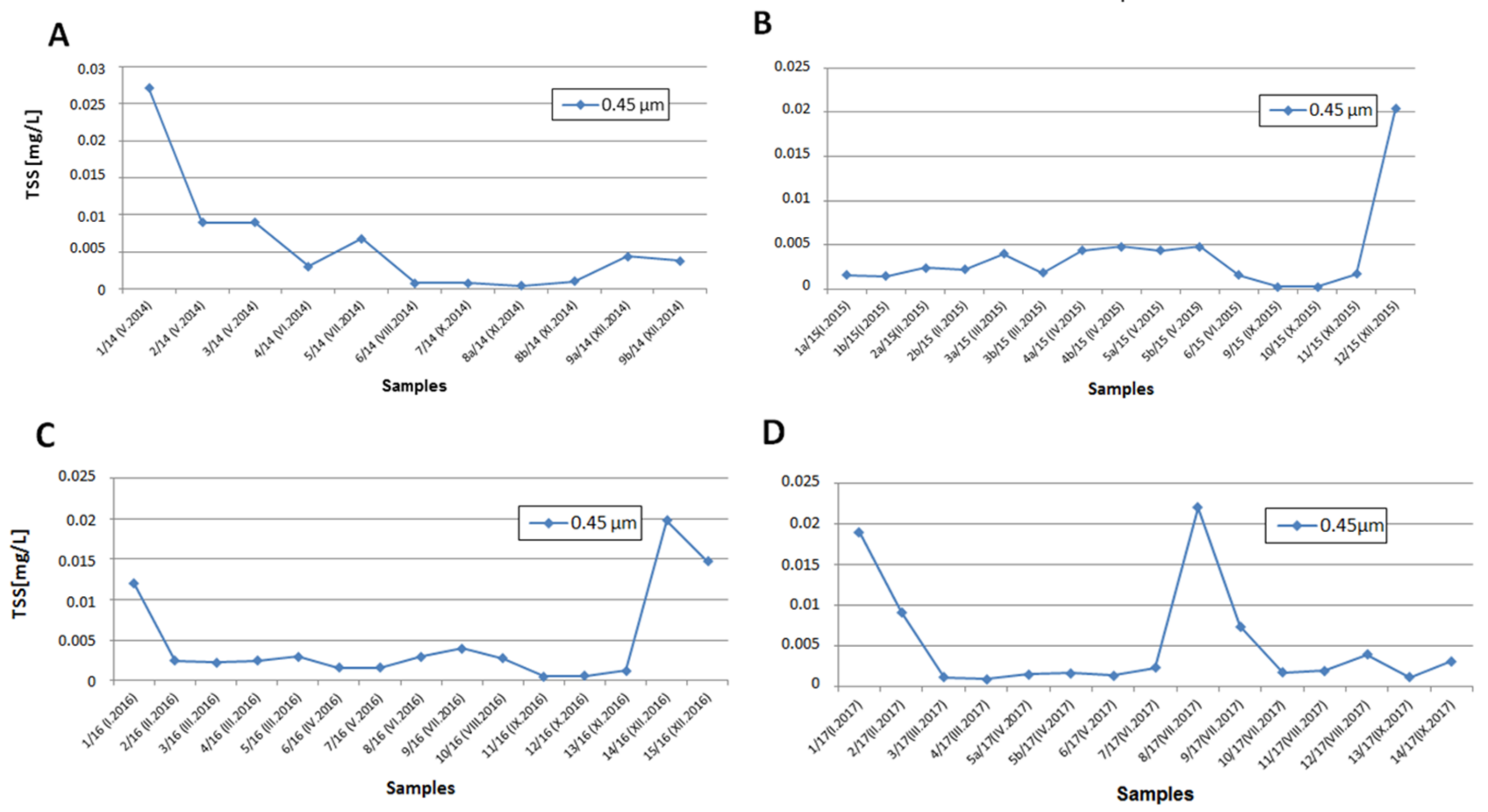
3.1. Total Suspended Solid Concentrations
3.2. Turbidity Changes
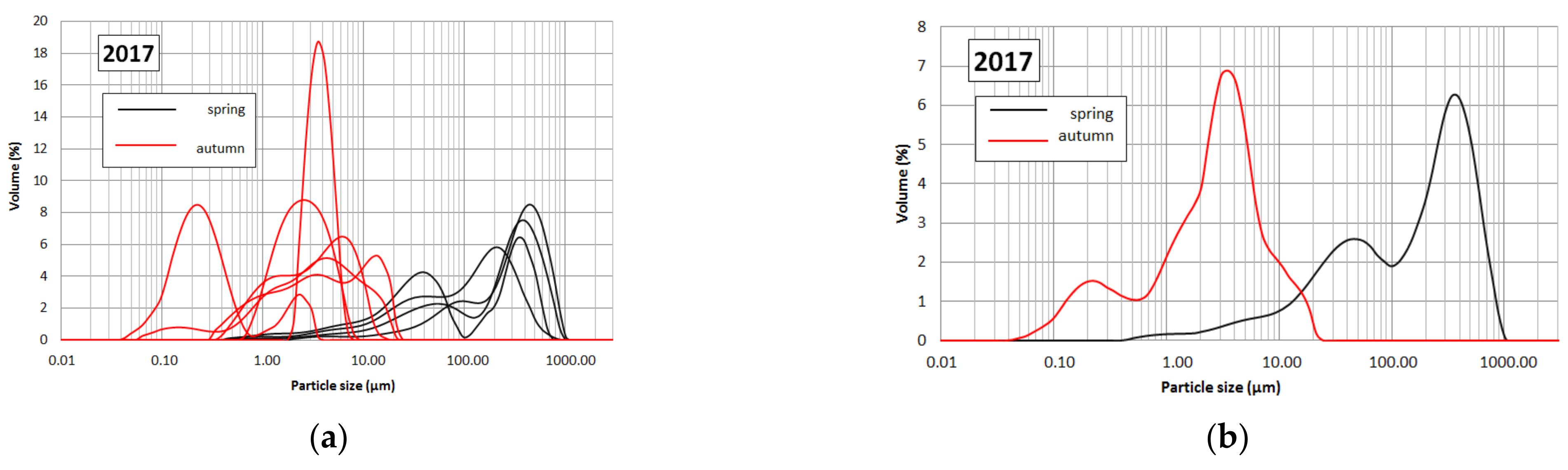
3.3. Granulometric Analysis
3.4. Silt Density Index Measurements
4. Discussion
4.1. Total Suspended Solid Concentration
4.2. Turbidity Changes
4.3. Granulometric Analysis
4.4. Silt Density Index Measurements
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Nika, C.E.; Vasilaki, V.; Expósito, A.; Katsou, E. Water Cycle and Circular Economy: Developing a Circularity Assessment Framework for Complex Water Systems. Water Res. 2020, 187, 116423. [Google Scholar] [CrossRef]
- Liang, X.; van Dijk, M.P. Economic and financial analysis on rainwater harvesting for agricultural irrigation in the rural areas of Beijing. Resour. Conserv. Recycl. 2011, 55, 1100–1108. [Google Scholar] [CrossRef]
- Gatt, K.; Schranz, C. Retrofitting a 3 star hotel as a basis for piloting water minimisation interventions in the hospitality sector. Int. J. Hosp. Manag. 2015, 50, 115–121. [Google Scholar] [CrossRef]
- Ghisi, E. Potential for potable water savings by using rainwater in the residential sector of Brazil. Build. Environ. 2006, 41, 1544–1550. [Google Scholar] [CrossRef]
- Ghisi, E.; da Fonseca Tavares, D.; Rocha, V.L. Rainwater harvesting in petrol stations in Brasília: Potential for potable water savings and investment feasibility analysis. Resour. Conserv. Recycl. 2009, 54, 79–85. [Google Scholar] [CrossRef]
- Nasif, M.S.; Kumar, A.G.A.; Roslan, R. An effective optimization of rainwater harvesting system in hotel and hospital buildings. In Proceedings of the 4th IET Clean Energy and Technology Conference (CEAT 2016), Kuala Lumpur, Malaysia, 14–15 November 2016. [Google Scholar] [CrossRef]
- Rysulová, M.; Káposztásová, D.; Vranayová, Z. Grey water system application—water savings and use in the hotel building. In New Developments in Environmental Science and Geoscience, Proceedings of the International Conference on Environmental Science and Geoscience (ESG 2015), Vienna, Austria, 15–17 March 2015; Mastorakis, N.E., Bulucea, A., Eds.; Energy, Environmental and Structural Engineering Series; INASE: Vienna, Austria, 2015; pp. 61–64. [Google Scholar]
- Bogacz, A.; Woźniczka, P.; Burszta-Adamiak, E.; Kolasińska, K. Methods of enhancing water retention in urban areas. Sci. Rev. Eng. Environ. Sci. 2013, 22, 27–35. [Google Scholar]
- Geiger, W.F. Think global, act local. In New Waterscapes: Planning, Building and Designing with Water; Dreiseitl, H., Grau, D., Eds.; Birkhäuser—Publishers for Architecture: Basel, Switzerland, 2005; pp. 78–111. [Google Scholar]
- Sands, P.; Galizzi, P. (Eds.) Council Directive 98/83/EC of 3 November 1998 on the Quality of Water Intended for Human Consumption; Cambridge University Press: Cambridge, UK, 1998. [Google Scholar] [CrossRef]
- Regulation of the Minister of Health of 7 December 2017 on the Quality of Water Intended for Human Consumption. Available online: http://isap.sejm.gov.pl/isap.nsf/DocDetails.xsp?id=WDU20170002294 (accessed on 28 February 2021).
- Khayan, K.; Heru Husodo, A.; Astuti, I.; Sudarmadji, S.; Sugandawaty Djohan, T. Rainwater as a source of drinking water: Health impacts and rainwater treatment. J. Environ. Public Health 2019, 2019, 1–10. [Google Scholar] [CrossRef]
- Kim, T.; Lye, D.; Donohue, M.; Mistry, J.H.; Pfaller, S.; Vesper, S.; Kirisits, M.J. Harvested rainwater quality before and after treatment and distribution in residential systems. J. Am. Water Work. Assoc. 2016, 108, E571–E584. [Google Scholar] [CrossRef]
- Stefanakis, A.I. The role of constructed wetlands as green infrastructure for sustainable urban water management. Sustainability 2019, 11, 6981. [Google Scholar] [CrossRef]
- Nicolaisen, B. Developments in membrane technology for water treatment. Desalination 2003, 153, 355–360. [Google Scholar] [CrossRef]
- Bodzek, M.; Konieczny, K.; Rajca, M. Membranes in water and wastewater disinfection. Arch. Environ. Prot. 2019, 45, 3–18. [Google Scholar] [CrossRef]
- Guo, W.; Ngo, H.H.; Li, J. A mini-review on membrane fouling. Bioresour. Technol. 2012, 122, 27–34. [Google Scholar] [CrossRef]
- Howe, K.J.; Clark, M.M. Fouling of microfiltration and ultrafiltration membranes by natural waters. Environ. Sci. Technol. 2002, 36, 3571–3576. [Google Scholar] [CrossRef]
- Water Quality—Determination of Turbidity—Part 1: Quantitative Methods; PN-EN ISO 7027-1:2016-09 Polish Standard. Available online: https://infostore.saiglobal.com/preview/98709941385.pdf?sku=879938_SAIG_NSAI_NSAI_2090663 (accessed on 28 February 2021).
- Vitton, S.J.; Sadler, L.Y. Particle size analysis of soils using laser light scattering and X-ray absorption technology. Geotech. Test. J. 1997, 20, 63–73. [Google Scholar] [CrossRef]
- Bray, R.T.; Fitobór, K. Sizes of iron hydroxide particles formed during ferric coagulation processes. Desalination Water Treat. 2017, 64, 419–424. [Google Scholar] [CrossRef]
- Bartmiński, P.; Krusińska, A.; Bieganowski, A.; Ryżak, M. Preparation of soil samples for grain size distribution analysis using the laser diffraction method. Rocz. Glebozn. 2011, 62, 9–15. [Google Scholar]
- ASTM International. Standard Test Method for Silt Density Index (SDI) of Water; D4189-07, An American National Standard; ASTM International: West Conshohocken, PA, USA, 2014; Available online: https://www.techstreet.com/standards/astm-d4189-07-2014?product_id=1873862#jumps (accessed on 28 February 2021).
- Promat-Woda. Operating Instructions—SDI Measuring Device; Promat-Woda: Gdynia, Poland, 2015. [Google Scholar]
- Ociepa, E.; Mrowiec, M.; Deska, I.; Okoniewska, E. Snow cover as a medium for deposition of pollution. Annu. Set Environ. Prot. 2015, 17, 560–575. [Google Scholar]
- Polkowska, Ż.; Demkowska, I.; Cichała-Kamrowska, K.; Namieśnik, J. Pollutants present in snow samples collected from various layers of snow cover as a source of information about the state of environment in a big urban agglomeration. Ecol. Chem. Eng. S 2010, 17, 203–231. [Google Scholar]
- Zhu, H.; Xu, Y.; Yan, B.; Guan, J. Snowmelt runoff: A new focus of urban nonpoint source pollution. Int. J. Environ. Res. Public Health 2012, 9, 4333–4345. [Google Scholar] [CrossRef] [PubMed]
- Meera, V.; Ahammed, M.M. Water quality of rooftop rainwater harvesting systems: A review. J. Water Supply: Res. Technol. AQUA 2006, 55, 257–268. [Google Scholar] [CrossRef]
- Sakson, G. The use of rainwater in buildings. Rynek Instal. 2010, 5, 50–53. [Google Scholar]
- Skwarzyńska, A.; Sosnowska, B.; Pytka, A.; Marzec, M.; Gizińska, M.; Słowik, T.; Zając, G. The quality of rainwater and evaluation of its use in individual households. Technol. Wody 2014, 6, 20–23. [Google Scholar]
- Todeschini, S.; Manenti, S.; Creaco, E. Testing an innovative first flush identification methodology against field data from an Italian catchment. J. Environ. Manag. 2019, 246, 418–425. [Google Scholar] [CrossRef] [PubMed]
- Bertrand-Krajewski, J.L.; Chebbo, G.; Saget, A. Distribution of pollutant mass vs volume in stormwater discharges and the first flush phenomenon. Water Res. 1998, 32, 2341–2356. [Google Scholar] [CrossRef]
- Sansalone, J.J.; Cristina, C.M. First flush concepts for suspended and dissolved solids in small impervious watersheds. J. Environ. Eng. 2004, 130, 1301–1314. [Google Scholar] [CrossRef]
- Friedler, E.; Gilboa, Y.; Muklada, H. Quality of Roof-Harvested Rainwater as a Function of Environmental and Air Pollution Factors in a Coastal Mediterranean City (Haifa, Israel). Water 2017, 9, 896. [Google Scholar] [CrossRef]
- Gikas, G.D.; Tsihrintzis, V.A. Assessment of water quality of first-flush roof runoff and harvested rainwater. J. Hydrol. 2012, 466–467, 115–126. [Google Scholar] [CrossRef]
- Burszta-Adamiak, E.; Stodolak, R. An assesment of grain size structure of dust in wet precipitation and its physico-chemical properties. Woda-Środowisko-Obsz. Wiej. 2007, 7, 83–94. [Google Scholar]
- Lye, D.J. Rooftop runoff as a source of contamination: A review. Sci. Total Environ. 2009, 407, 5429–5434. [Google Scholar] [CrossRef]
- Zdeb, M.; Zamorska, J.; Papciak, D.; Słyś, D. The quality of rainwater collected from roofs and the possibility of its economic use. Resources 2020, 9, 12. [Google Scholar] [CrossRef]
- Dobrowsky, P.H.; Lombard, M.; Cloete, W.J.; Saayman, M.; Cloete, T.E.; Carstens, M.; Khan, S.; Khan, W. Efficiency of microfiltration systems for the removal of bacterial and viral contaminants from surface and rainwater. Water Air Soil Pollut. 2015, 226, 1–14. [Google Scholar] [CrossRef]
- Rajca, M.; Bray, R.T.; Fitobór, K.; Gołombek, K. Laser Granulometer as an Useful Tool for Selection of Appropriate Membranes Used in the MIEX® DOC-UF/MF Hybrid Process. Arch. Metall. Mater. 2018, 63, 1133–1140. [Google Scholar] [CrossRef]
- Blott, S.J.; Pye, K. Particle size scales and classification of sediment types based on particle size distributions: Review and recommended procedures. Sedimentology 2012, 59, 2071–2096. [Google Scholar] [CrossRef]
- Pye, K.; Blott, S.J. Particle size analysis of sediments, soils and related particulate materials for forensic purposes using laser granulometry. Forensic Sci. Int. 2004, 144, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.D.; Lee, S.H.; Jo, M.H.; Park, P.K.; Lee, C.H.; Kwak, J.W. Effect of coagulation conditions on membrane filtration characteristics in coagulation-microfiltration process for water treatment. Environ. Sci. Technol. 2000, 34, 3780–3788. [Google Scholar] [CrossRef]
- Sakol, D.; Konieczny, K. Application of coagulation and conventional filtration in raw water pretreatment before microfiltration membranes. Desalination 2004, 162, 61–73. [Google Scholar] [CrossRef]
- Schippers, J.C.; Verdouw, J. The modified fouling index, a method of determining the fouling characteristics of water. Desalination 1980, 32, 137–148. [Google Scholar] [CrossRef]
- Boerlage, S.F.; Kennedy, M.D.; Dickson, M.R.; El-Hodali, D.E.; Schippers, J.C. The modified fouling index using ultrafiltration membranes (MFI-UF): Characterisation, filtration mechanisms and proposed reference membrane. J. Membr. Sci. 2002, 197, 1–21. [Google Scholar] [CrossRef]
- Alhadidi, A.; Kemperman, A.J.B.; Blankert, B.; Schippers, J.C.; Wessling, M.; van der Meer, W.G.J. Silt density index and modified fouling index relation, and effect of pressure, temperature and membrane resistance. Desalination 2011, 273, 48–56. [Google Scholar] [CrossRef]
- Khirani, S.; Aim, R.B.; Manero, M.H. Improving the measurement of the Modified Fouling Index using nanofiltration membranes (NF–MFI). Desalination 2006, 191, 1–7. [Google Scholar] [CrossRef]

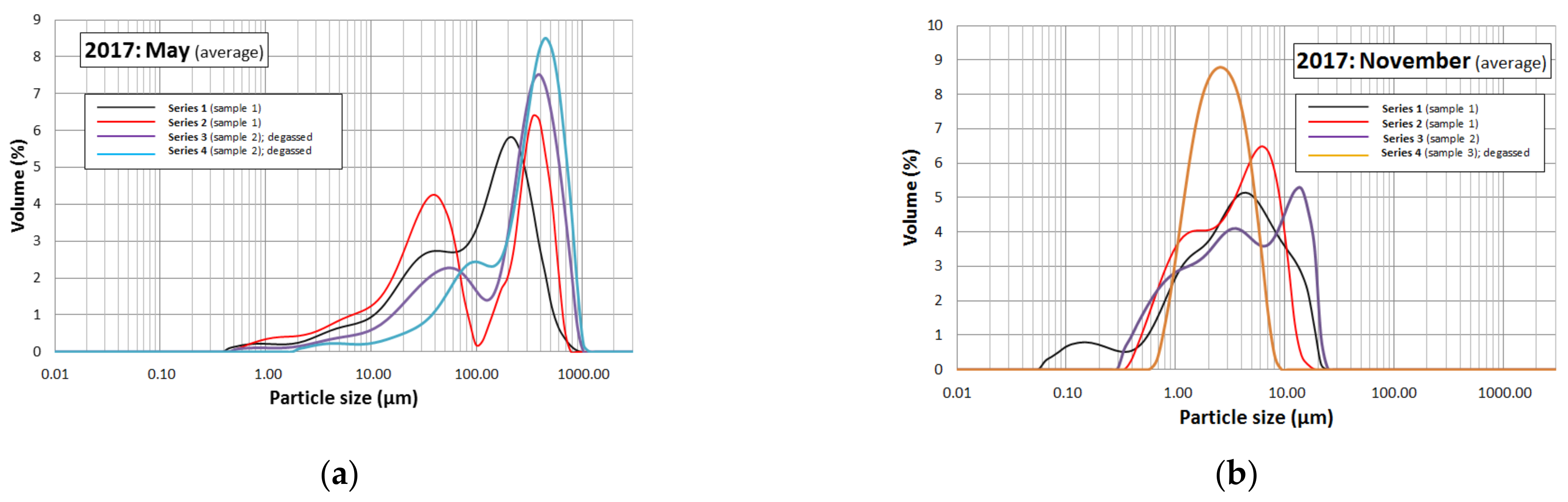
| May 2017 |
| Series 1: sample 1, beginning of May (600 rpm; higher sensitivity) |
| Series 2: sample 1, beginning of May (1200 rpm; normal sensitivity) |
| Series 3: sample 2, mid-May (1200 rpm; normal sensitivity); subjected to degassing |
| Series 4: sample 2, mid-May (1200 rpm; higher sensitivity); subjected to degassing |
| November 2017 |
| Series 1: sample 1, beginning of November (2000 rpm; normal sensitivity) |
| Series 2: sample 1, beginning of November (1600 rpm; normal sensitivity) |
| Series 3: sample 2, mid-November (600 rpm; higher sensitivity) |
| Series 4: sample 3, end of November (1600 rpm; normal sensitivity); subjected to degassing. |
| Measurement | t0 | tx | SDI |
|---|---|---|---|
| [Min] | [Min] | [-] | |
| TEST 1 | |||
| 1 | 01:39 | 13:10 | 5.96 |
| 2 | 00:13 | 24:55 | 6.63 |
| 3 | 02:32 | 29:25 | 6.14 |
| TEST 2 | |||
| 1 | 01:40 | 38:47 | 6.42 |
| 2 | 01:53 | 23:47 | 6.23 |
| TEST 3 | |||
| 1 | 01:08 | 48:37 | 6.52 |
| 2 | 01:37 | 15:14 | 6.06 |
| 3 | 02:26 | 46.52 | 6.34 |
| TEST 4 | |||
| 1 | 01:41 | 49:24 | 6.48 |
| 2 | 01:35 | 11:08 | 5.85 |
| 3 | 01:56 | 36:03 | 6.38 |
| TEST 5 | |||
| 1 | 01:24 | 20:45 | 6.26 |
| 2 | 01:57 | 30:58 | 6.32 |
| 3 | 01:12 | 17:11 | 6.23 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fitobór, K.; Quant, B. Is the Microfiltration Process Suitable as a Method of Removing Suspended Solids from Rainwater? Resources 2021, 10, 21. https://doi.org/10.3390/resources10030021
Fitobór K, Quant B. Is the Microfiltration Process Suitable as a Method of Removing Suspended Solids from Rainwater? Resources. 2021; 10(3):21. https://doi.org/10.3390/resources10030021
Chicago/Turabian StyleFitobór, Karolina, and Bernard Quant. 2021. "Is the Microfiltration Process Suitable as a Method of Removing Suspended Solids from Rainwater?" Resources 10, no. 3: 21. https://doi.org/10.3390/resources10030021
APA StyleFitobór, K., & Quant, B. (2021). Is the Microfiltration Process Suitable as a Method of Removing Suspended Solids from Rainwater? Resources, 10(3), 21. https://doi.org/10.3390/resources10030021






