Pandæsim: An Epidemic Spreading Stochastic Simulator
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Overview
- Based on the quantities , the rules and their kinetics, compute stochastically at what time each reaction is triggered .
- Let being the next reaction: .
- Apply ; i.e., update the vector by decreasing the quantities of the substrates of and increasing the quantities of its products.
- Update the time: .
2.2. Pandæsim Model
2.3. Simulation Data and Parameters
2.4. Evolution Algorithm
- Soft quarantine: People do not use public transportation at all and do not go to restaurants during the midday break.
- Full quarantine: This corresponds to what actually happened in France; people were confined at home except for a one hour stroll per day in low populated areas (public parks, forests, etc., were forbidden). Again, to reduce the number of parameters, we assumed that the probability of contagion during the stroll was the same as at work. This also allowed us to take into account errands made to get food in more populated places such as groceries or supermarkets.
- First, the infection rate at time t, , is computed as the product of the global daily rate of infection, , by the infection factor of the current location (home, workplace, public transportation) . This infection rate is used the same way the propensity is in the standard SSA.
- Then, for each of the four age slices the deterministic continuous solver computes the average number of individuals of that age that will go from susceptible to asymptomatic state, , as the product of the population in that state and the infection rate at time t:The stochastic discrete solver (SDS) computes stochastic integer numbers such that, on the long run, they will average to the same values as the continuous solver. Even when the population is an integer number of individuals, this product, , is generally a floating point number because the infection rate is itself a floating point number. This number has an integral part (≥0) and a fractional part (between 0 and 1). The (discrete) number of new asymptomatic hosts is then computed as the integer part of the average number, plus 1 if a uniform random number taken into the interval … is below the fractional part:As the difference is 0.5 on the average, the higher the value is, the lower the relative impact of this stochastic discretisation becomes and the result is equivalent to a discrete averaged approach. Conversely, the lower the value is, the more important the stochastic discretisation becomes. This mechanism allows the simulator to automatically choose the best strategy to adapt to the value range of the population [26].
- Finally, when the current time indicates the beginning of a new day, (mod 24), individuals in each state either remain in the same state but shifted by one day, or change to another state. All the states transitions are computed stochastically by the SDS (or deterministically by the DCS) using the method described earlier.
- The population in the asymptomatic state that has on average reached the 5/6 day limit is moved to the first day of the ill state.
- According to the illness duration by age slice parameter, a proportion of the population in the ill state is moved to the hospitalised or to the recovered state. The others remaining in the ill state one more day.
- According to the disease severity by age slice parameter, a proportion of the population in the hospitalised state is moved to the deceased or recovered state. The others remain in the hospitalised state one more day.
The global daily rate of infection is then simply computed by multiplying the constant of propagation of the virus, , by the proportion of the total contagious population:By fitting the simulation results after the beginning of the lockdown to the data gathered from hospital statistics, we empirically found a good estimation of for the SARS-CoV-2 to 0.75. We think that using Pandæsim to model another type of epidemic, only this constant, along with the severity parameters, needs to be changed.
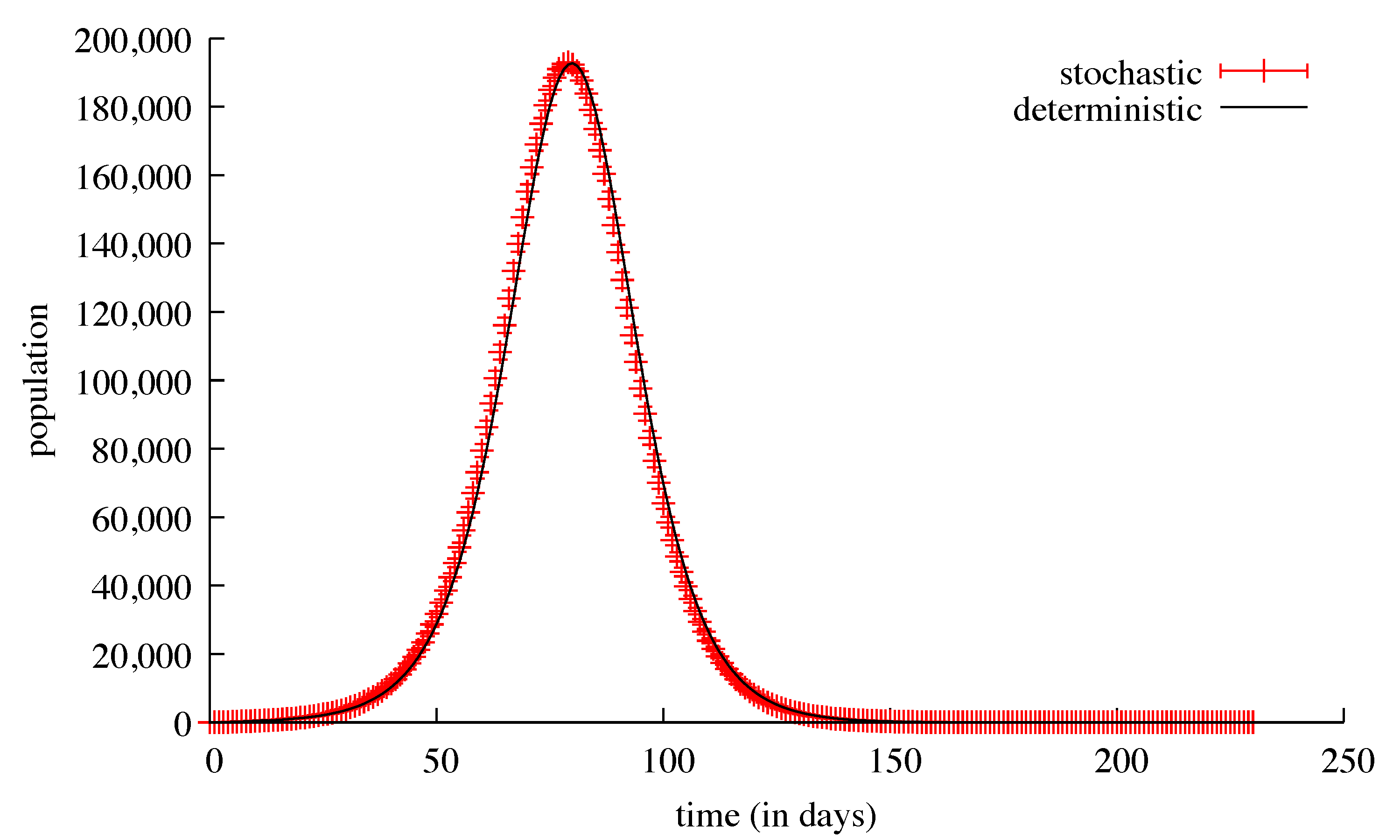
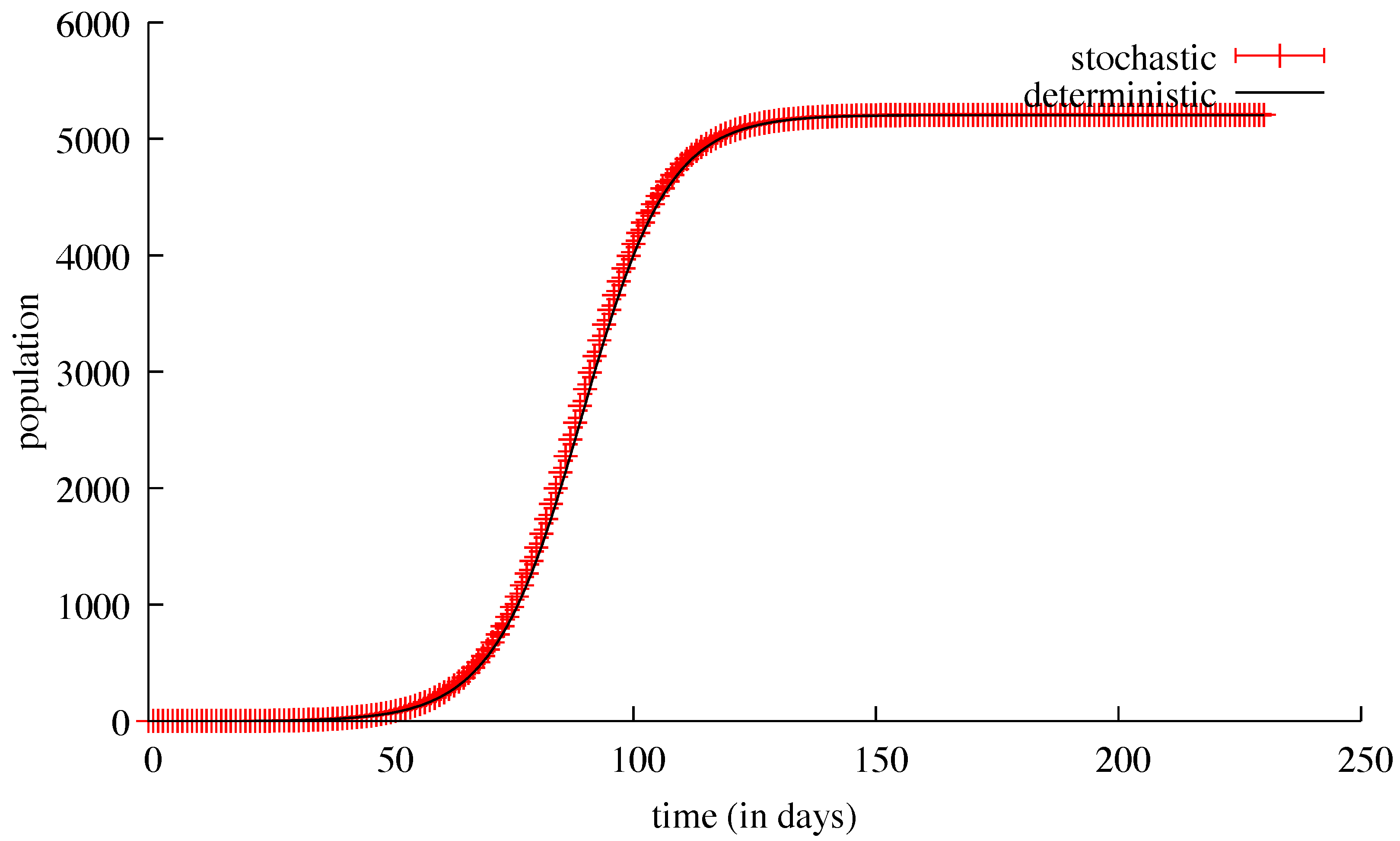
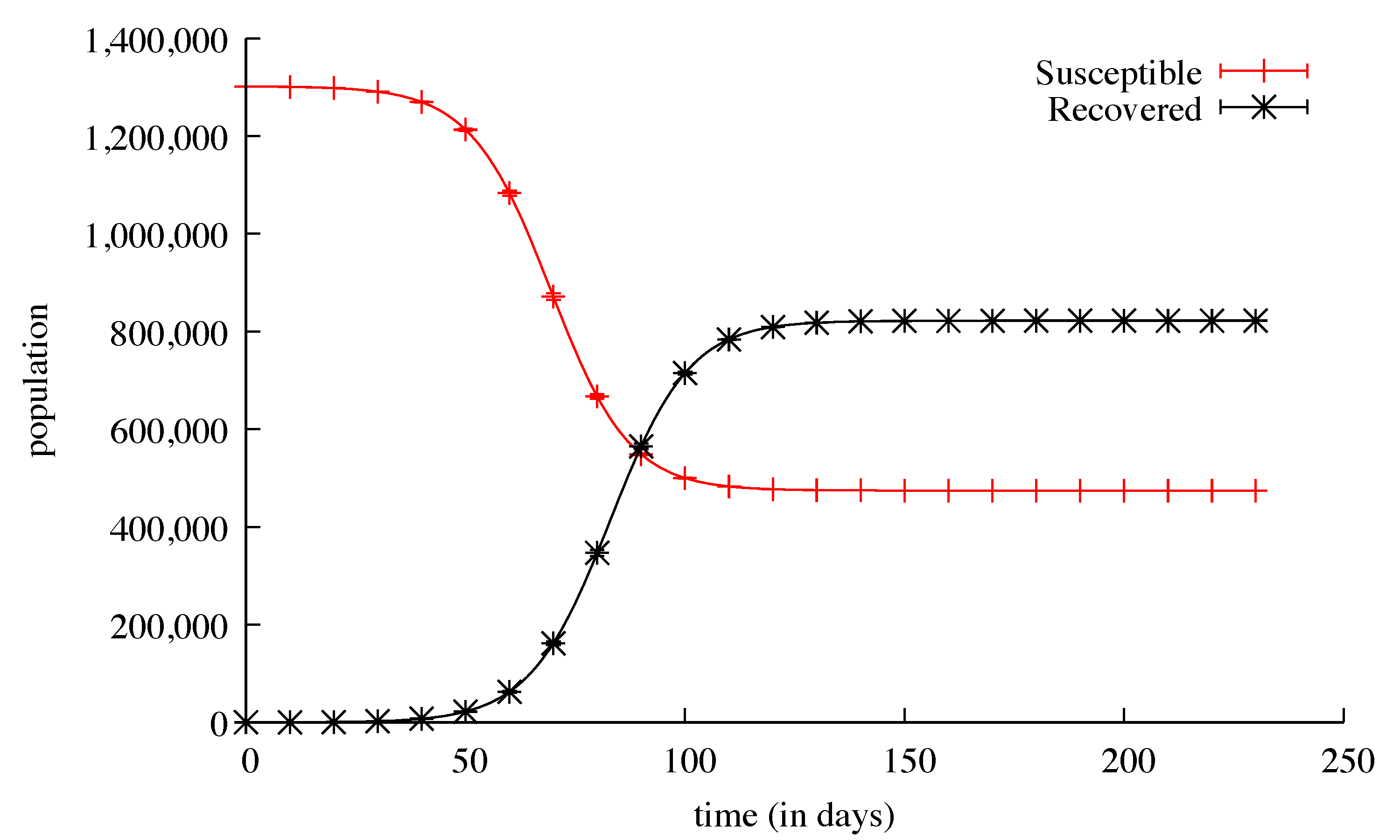
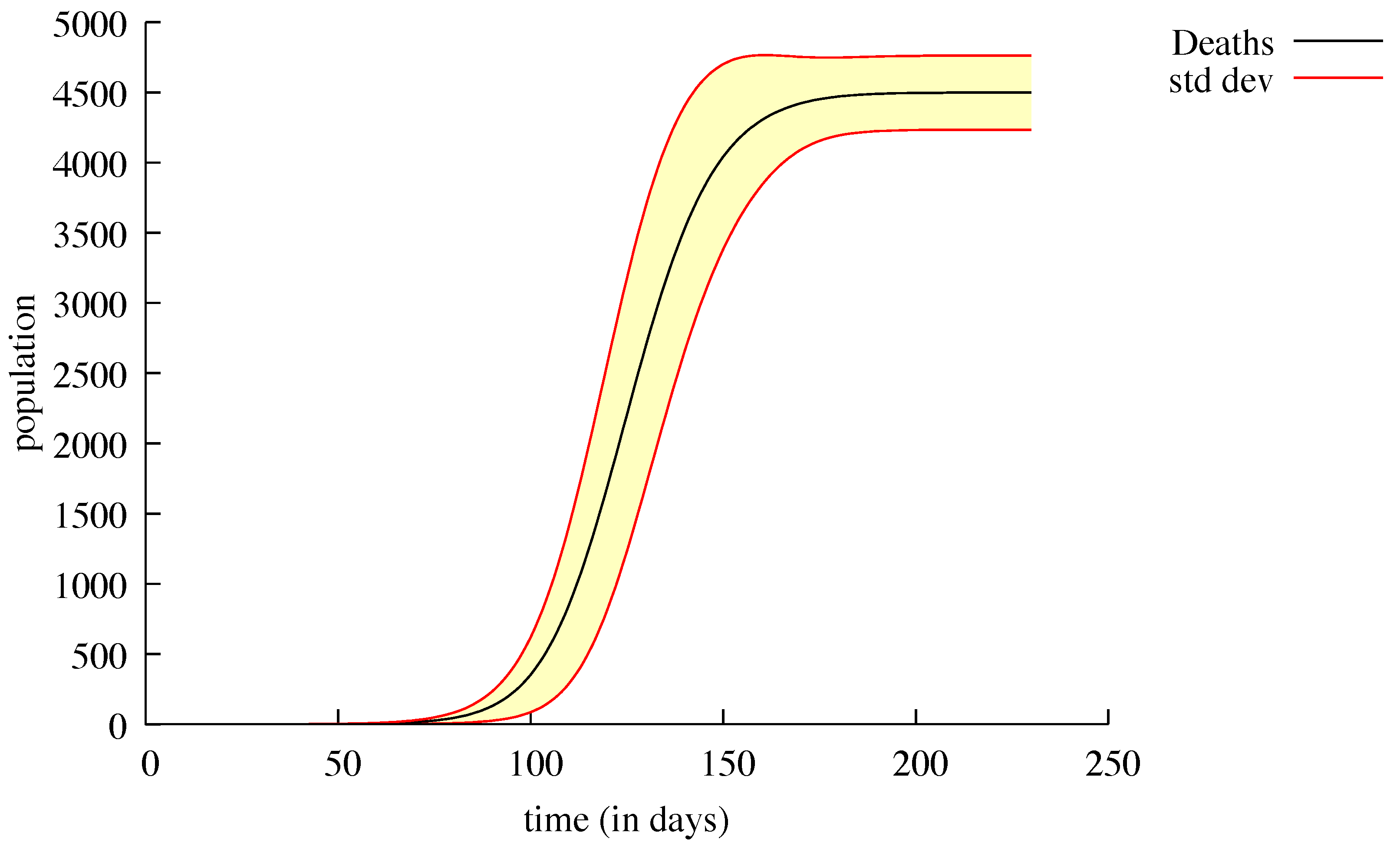
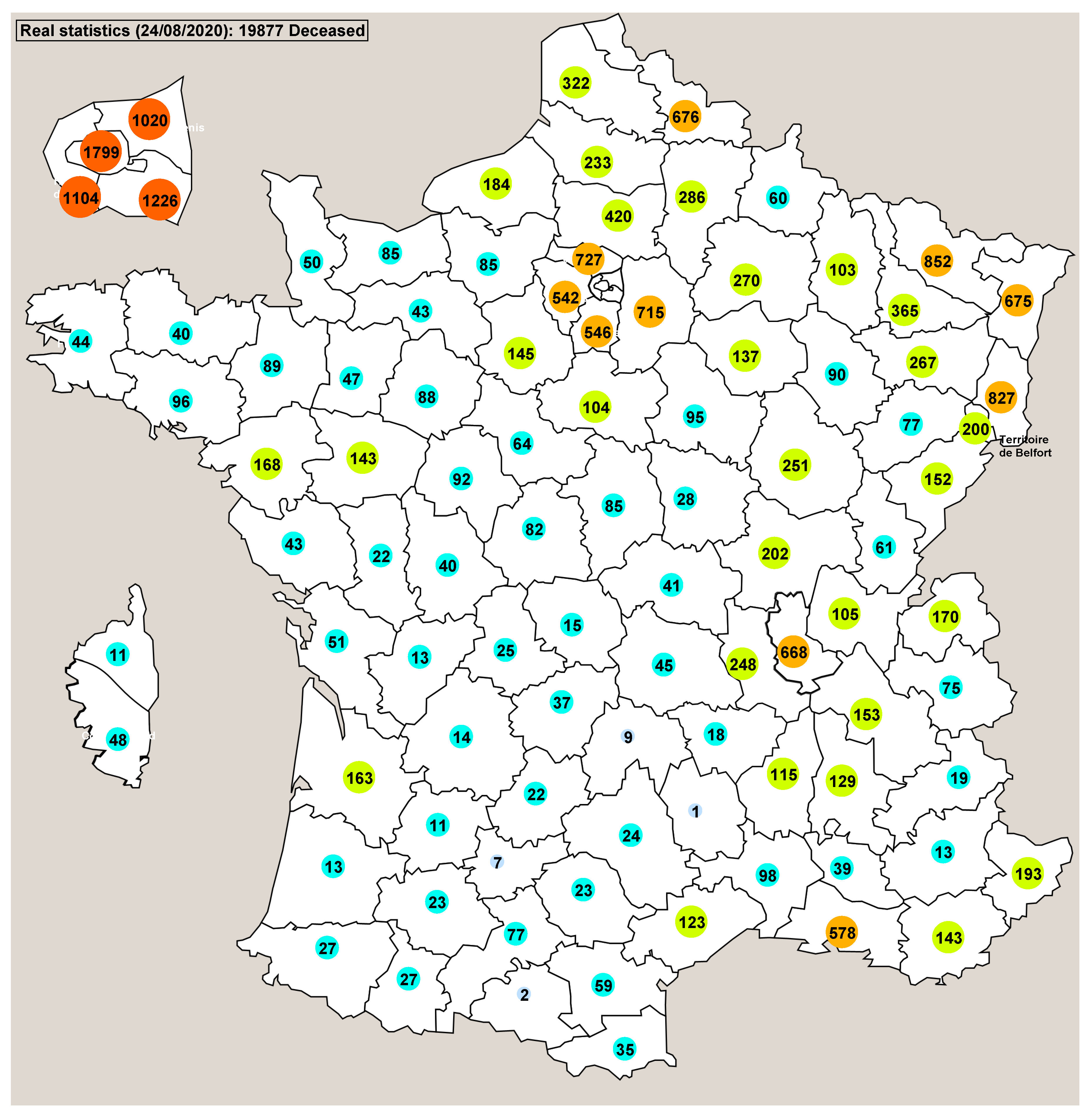
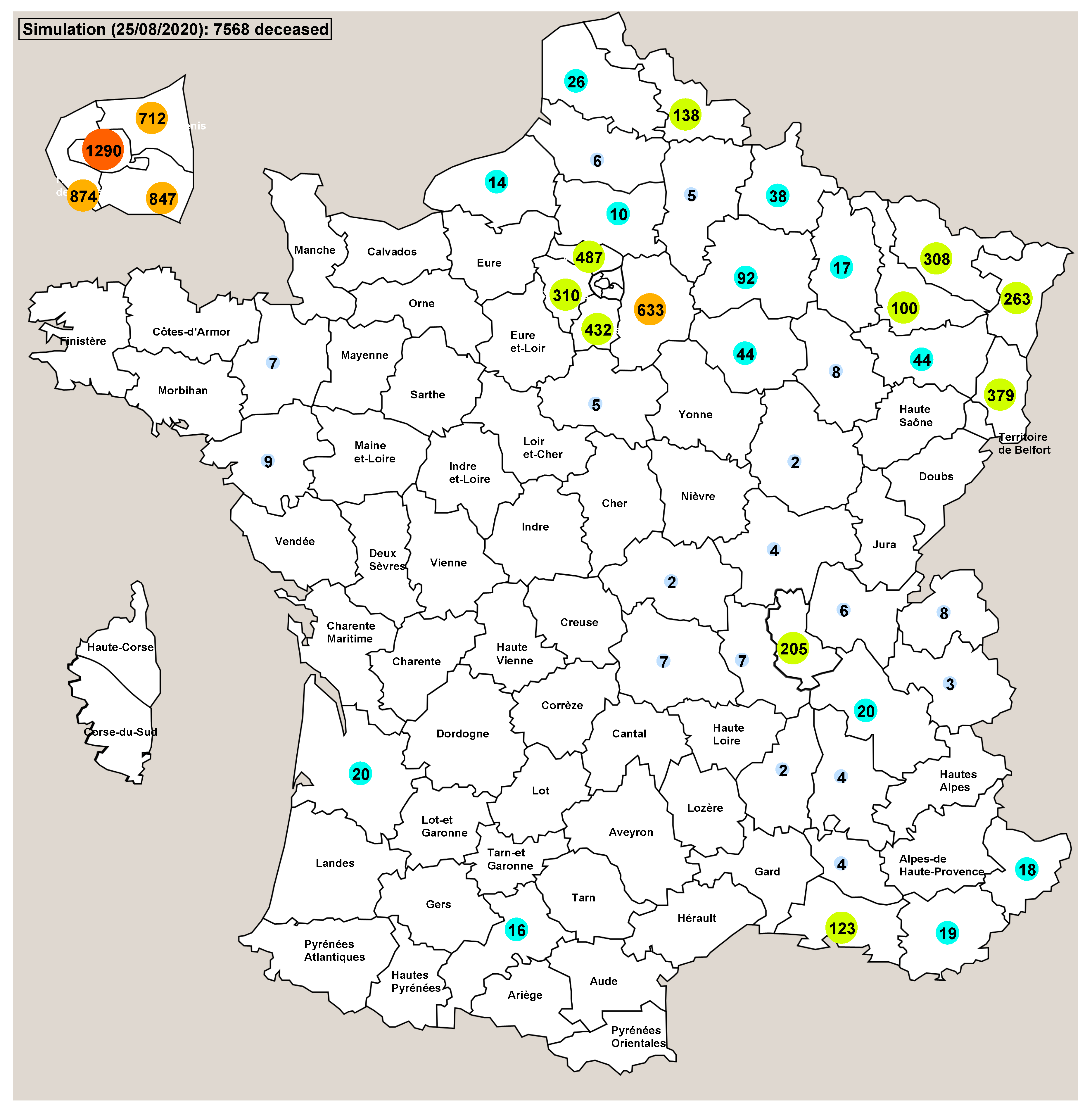
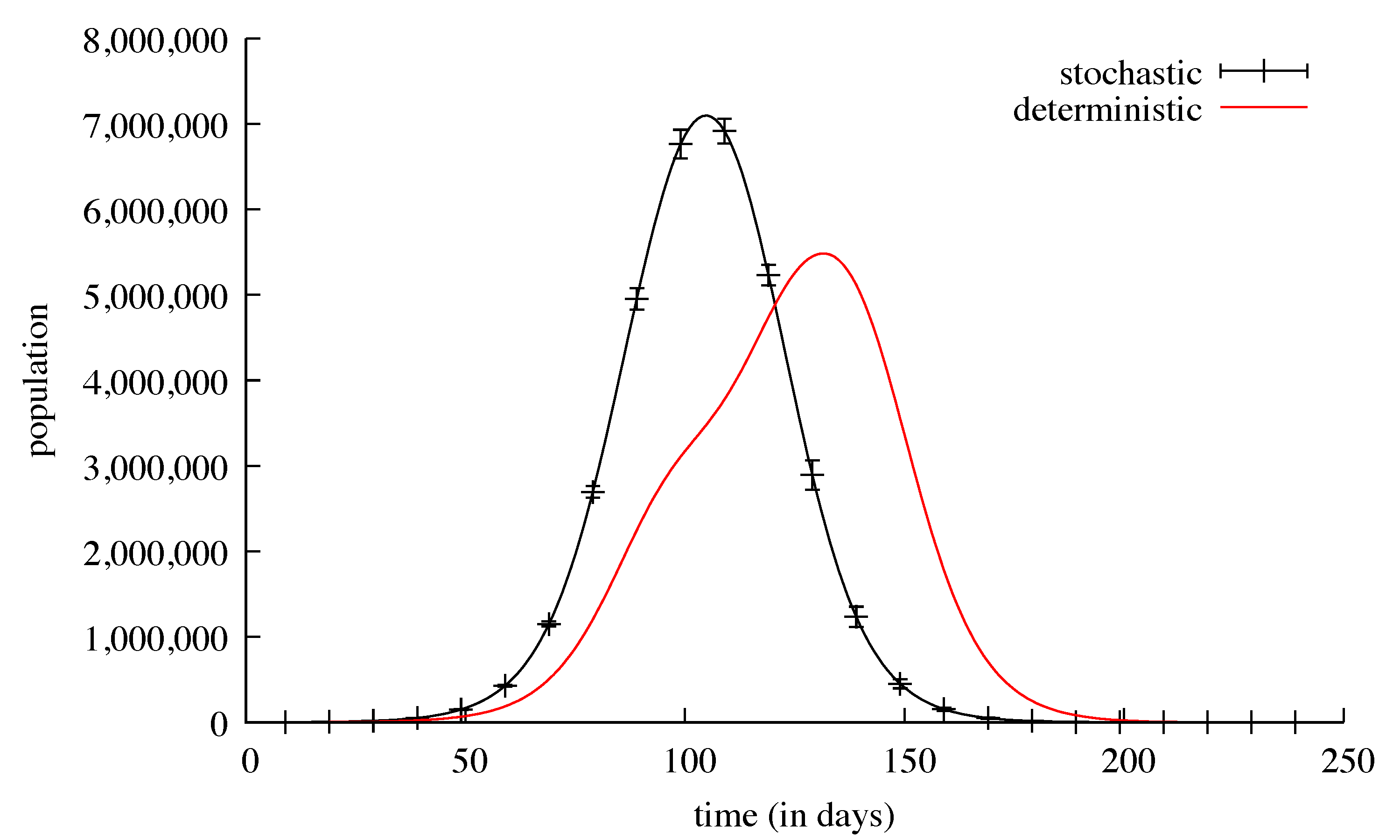
3. Results
4. Discussion
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ICU | Intensive Care Unit |
| SSA | Stochastic Simulation Algorithm |
| ODE | Ordinary Differential Equations |
| SDE | Stochastic Differential Equations |
| WHO | World Health Organization |
| DCS | Deterministic Continuous Solver |
| SDS | Stochastic Discrete Solver |
Appendix A
Appendix A.1.
| Région | Départements | |||||||
|---|---|---|---|---|---|---|---|---|
| Ile de France | Paris | 65 | Val-de-Marne | 180 | Val-d’Oise | 70 | Yvelines | 20 |
| Seine-Saint-Denis | 140 | |||||||
| Hauts de France | Somme | 10 | Nord | 40 | Oise | 10 | Aisne | 10 |
| Normandie | Seine-Maritime | 10 | ||||||
| Bretagne | Morbihan | 2 | Ille-et-Vilaine | 5 | ||||
| Pays de Loire | Loire-Atlantique | 8 | Maine-et-Loire | 8 | ||||
| Centre | Eure-et-Loir | 6 | Loiret | 2 | ||||
| Aquitaine | Charente-Maritime | 2 | Gironde | 6 | ||||
| Occitanie | Haute-Garonne | 2 | Hérault | 10 | Gard | 1 | ||
| Corse | Corse-du-Sud | 2 | ||||||
| Provence Alpes Cote d’Azur | Bouches-du-Rhône | 40 | ||||||
| Auvergne Rhone Alpes | Rhône | 60 | Ardèche | 3 | Drôme | 3 | Loire | 4 |
| Bourgogne Franche-Comté | Territoire de Belfort | 3 | Côte-d’Or | 30 | Doubs | 2 | ||
| Grand Est | Meuse | 1 | Moselle | 70 | Bas-Rhin | 20 | Haut-Rhin | 60 |
| Age | Lethality % | Illness Duration (days) | Local Travel % | Remote Travel % |
|---|---|---|---|---|
| 0–25 | 10 | 7 | 5 | 1 |
| 26–50 | 15 | 8 | 6 | 1 |
| 51–75 | 20 | 10 | 6 | 1 |
| 76+ | 55 | 14 | 0.5 | 0.4 |
| Location | Rate % |
|---|---|
| Home | 0.02 |
| Workplace | 2 |
| Public transportation | 4 |
| Contagious patients % | 1 |
| Severe form % | 20 |
| Propagation constant | 0.75 |
| Region | 0–25 | 26–50 | 51–75 | 76+ |
|---|---|---|---|---|
| Ile de France | 3,164,218 | 4,177,466 | 2,982,661 | 683,650 |
| Hauts de France | 1,597,206 | 1,846,011 | 1,477,121 | 418,273 |
| Normandie | 917,615 | 808,834 | 926,890 | 318,070 |
| Bretagne | 733,777 | 868,726 | 1026,010 | 300,503 |
| Pays de Loire | 1,052,858 | 958,431 | 1,150,747 | 268,259 |
| Centre | 652,748 | 617,614 | 717,309 | 256,969 |
| Aquitaine | 1,483,728 | 1,435,736 | 1,869,051 | 661,315 |
| Occitanie | 1,471,676 | 1,527,461 | 1,873,453 | 594,186 |
| Corse | 69,362 | 96,471 | 91,685 | 40,289 |
| Provence Alpes Cote d’Azur | 1,270,520 | 1,185,877 | 1,562,820 | 473,619 |
| Auvergne Rhone Alpes | 2,152,246 | 2,272,047 | 2,202,878 | 693,612 |
| Bourgogne Franche-Comté | 572,106 | 719,857 | 811,137 | 291,031 |
| Grand Est | 1,403,834 | 1,556,258 | 1,567,738 | 446,914 |
References
- Bernard-Stoecklin, S.; Rolland, P.; Silue, Y.; Mailles, A.; Campese, C.; Simondon, A.; Mechain, M.; Meurice, L.; Nguyen, M.; Bassi, C.; et al. First cases of coronavirus disease 2019 (Covid-19) in France: Surveillance, investigations and control measures. Eurosurveillance 2020, 25, 2000094. [Google Scholar] [CrossRef] [PubMed]
- Décret no 2020-260 du 16 Mars 2020 Portant Réglementation des Déplacements dans le Cadre de la Lutte Contre la Propagation du Virus Covid-19. Legifrance. Available online: https://www.legifrance.gouv.fr/affichTexte.do?cidTexte=JORFTEXT000041728476&categorieLien=id (accessed on 8 June 2020).
- Kraemer, M.U.G.; Yang, C.-H.; Gutierrez, B.; Wu, C.-H.; Klein, B.; Pigott, D.M.; du Plessis, L.; Faria, N.R.; Li, R.; Hanage, W.P.; et al. The effect of human mobility and control measures on the COVID-19 epidemic in China. Science 2020, 368, 493–497. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tian, H.; Liu, Y.; Li, Y.; Wu, C.-H.; Chen, B.; Kraemer, M.U.G.; Li, B.; Cai, J.; Xu, B.; Yang, Q.; et al. An investigation of transmission control measures during the first 50 days of the COVID-19 epidemic in China. Science 2020, 368, 638–642. [Google Scholar] [CrossRef] [Green Version]
- Diekmann, O.; Heesterbeek, J.A.; Metz, J.A. On the definition and the computation of the basic reproduction ratio R0 in models for infectious diseases in heterogeneous populations. J. Math. Biol. 1990, 28, 365–382. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, S.; Lin, Q.; Ran, J.; Musa, S.S.; Yang, G.; Wang, W.; Lou, Y.; Gao, D.; Yang, L.; He, D.; et al. Preliminary estimation of the basic reproduction number of novel coronavirus (2019-nCoV) in China, from 2019 to 2020: A data-driven analysis in the early phase of the outbreak. Int. J. Infect. Dis. 2020, 92, 214–217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roberts, M.G.; Nishiura, H. Early estimation of the reproduction number in the presence of imported cases: Pandemic influenza H1N1-2009 in New Zealand. PLoS ONE 2011, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Q.; Guan, X.; Wu, P.; Wang, X.; Zhou, L.; Tong, Y.; Ren, R.; Leung, K.S.; Lau, E.H.; Wong, J.Y.; et al. Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef]
- Read, J.M.; Bridgen, J.R.E.; Cummings, D.A.T.; Ho, A.; Jewell, C.P. Novel coronavirus 2019-nCoV: Early estimation of epidemiological parameters and epidemic predictions. medRxiv 2020. [Google Scholar] [CrossRef] [Green Version]
- Wu, J.T.; Leung, K.; Leung, G.M. Nowcasting and forecasting the potential domestic and international spread of the 2019-nCoV outbreak originating in Wuhan, China: A modelling study. Lancet 2020, 395, 689–697. [Google Scholar] [CrossRef] [Green Version]
- Tindale, L.; Coombe, M.; Stockdale, J.E.; Garlock, E.; Lau, W.Y.V.; Saraswat, M.; Lee, Y.-H.B.; Zhang, L.; Chen, D.; Wallinga, J.; et al. Transmission interval estimates suggest pre-symptomatic spread of COVID-19. medRxiv 2020. [Google Scholar] [CrossRef] [Green Version]
- Nature. Coronavirus Latest: Scientists Scramble to Study Virus Samples. Available online: https://www.nature.com/articles/d41586-020-00154-w (accessed on 4 February 2020).
- Fang, Y.; Nie, Y.; Penny, M. Transmission dynamics of the COVID-19 outbreak and effectiveness of government interventions: A data-driven analysis. J. Med. Virol. 2020. [Google Scholar] [CrossRef] [Green Version]
- Tang, B.; Xia, F.; Tang, S.; Bragazzi, N.L.; Li, Q.; Sun, X.; Liang, J.; Xiao, Y.; Wu, J. The effectiveness of quarantine and isolation determine the trend of the COVID-19 epidemics in the final phase of the current outbreak in China. Int. J. Infect. Dis. Ijid Off. Publ. Int. Soc. Infect. Dis. 2020, 95, 288–293. [Google Scholar] [CrossRef]
- Prem, K.; Liu, Y.; Russell, T.W.; Kucharski, A.J.; Eggo, R.M.; Davies, N.; Centre for the Mathematical Modelling of Infectious Diseases COVID-19 Working Group; Jit, M.; Klepac, P. The effect of control strategies to reduce social mixing on outcomes of the COVID-19 epidemic in Wuhan, China: A modelling study. Lancet Public Health 2020, 5, e261–e270. [Google Scholar] [CrossRef] [Green Version]
- Ji, C.; Jiang, D.; Shi, N. The Behavior of an SIR Epidemic Model with Stochastic Perturbation. Stoch. Anal. Appl. 2012, 30, 755–773. [Google Scholar] [CrossRef]
- Jiang, D.; Ji, C.; Shi, N.; Yu, J. The long time behavior of DI SIR epidemic model with stochastic perturbation. J. Math. Anal. Appl. 2010, 372, 162–180. [Google Scholar] [CrossRef] [Green Version]
- Cai, Y.; Kang, Y.; Banerjee, M.; Wang, W. A stochastic SIRS epidemic model with infectious force under intervention strategies. J. Differ. Equ. 2015, 259, 7463–7502. [Google Scholar] [CrossRef]
- Gray, A.; Greenhalgh, D.; Hu, L.; Mao, X.; Pan, J. A stochastic differential equation SIS epidemic model. SIAM J. Appl. Math. 2011, 71, 876–902. [Google Scholar] [CrossRef] [Green Version]
- Zakary, O.; Rachik, M.; Elmouki, I. A multi-regional epidemic model for controlling the spread of Ebola: Awareness, treatment, and travel-blocking optimal control approaches. Math. Methods Appl. Sci. 2017, 40, 1265–1279. [Google Scholar] [CrossRef]
- Abouelkheir, I.; El Kihal, F.; Rachik, M.; Zakary, O.; Elmouki, I. A multi-regions SIRS discrete epidemic model with a travel-blocking vicinity optimal control approach on cells. Br. J. Math. Comput. Sci. 2017, 20, 1–16. [Google Scholar] [CrossRef] [Green Version]
- El Kihal, F.; Abouelkheir, I.; Rachik, M.; Elmouki, I. Role of Media and Effects of Infodemics and Escapes in the Spatial Spread of Epidemics: A Stochastic Multi-Region Model with Optimal Control Approach. Mathematics 2019, 7, 304. [Google Scholar] [CrossRef] [Green Version]
- Park, H.; Kim, S.H. A Study on Herd Immunity of COVID-19 in South Korea: Using a Stochastic Economic-Epidemiological Model. Environ. Resour. Econ. 2020, 76, 665–670. [Google Scholar] [CrossRef] [PubMed]
- Hackl, J.; Dubernet, T. Epidemic Spreading in Urban Areas Using Agent-Based Transportation Models. Future Internet 2019, 11, 92. [Google Scholar] [CrossRef] [Green Version]
- Hunter, E.; Mac Namee, B.; Kelleher, J. An open-data-driven agent-based model to simulate infectious disease outbreaks. PLoS ONE 2019, 14, e0211245. [Google Scholar] [CrossRef] [PubMed]
- Amar, P.; Paulevé, L. HSIM: An hybrid stochastic simulation system for systems biology. In Proceedings of the Third International Workshop on Static Analysis and Systems Biology, Deauville, France, 10 September 2012; pp. 3–21. [Google Scholar] [CrossRef]
- Gillespie, D.T. A General Method for Numerically Simulating the Stochastic Time Evolution of Coupled Chemical Reactions. J. Comput. Phys. 1976, 22, 403–434. [Google Scholar] [CrossRef]
- Rathinam, M.; Petzold, L.R.; Cao, Y.; Gillespie, D.T. Stiffness in stochastic chemically reacting systems: The implicit tau-leaping method. J. Chem. Phys. 2003, 119, 12784–12794. [Google Scholar] [CrossRef] [Green Version]
- GÉODES—Géo Données en Santé Publiques. Santé Publique France. Available online: https://geodes.santepubliquefrance.fr/ (accessed on 25 August 2020).
- French Government Website. Info Coronavirus Covid 19. Available online: https://www.gouvernement.fr/info-coronavirus/carte-et-donnees (accessed on 25 August 2020). (In French).
- Guan, W.-J.; Ni, Z.-Y.; Hu, Y.; Liang, W.-H.; Ou, C.-Q.; He, J.-X.; Liu, L.; Shan, H.; Lei, C.-L.; Hui, D.S.; et al. Clinical characteristics of coronavirus disease 2019 in china. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef]
- Grasselli, G.; Pesenti, A.; Cecconi, M. Critical Care Utilization for the COVID-19 Outbreak in Lombardy, Italy: Early Experience and Forecast During an Emergency Response. JAMA 2020. [Google Scholar] [CrossRef] [Green Version]
- Fontanet, A.; Tondeur, L.; Madec, Y.; Grant, R.; Besombes, C.; Jolly, N.; Pellerin, S.F.; Ungeheuer, M.-N.; Cailleau, I.; Kuhmel, L.; et al. Cluster of COVID-19 in northern France: A retrospective closed cohort study. medRxiv 2020. [Google Scholar] [CrossRef]
- Salje, H.; Kiem, C.T.; Lefrancq, N.; Courtejoie, N.; Bosetti, P.; Paireau, J.; Andronico, A.; Hoze, N.; Richet, J.; Dubost, C.-L.; et al. Estimating the burden of SARS-CoV-2 in France. Sciences 2020. [Google Scholar] [CrossRef]
- Béraud, G.; Kazmercziak, S.; Beutels, P.; Levy-Bruhl, D.; Lenne, X.; Mielcarek, N.; Yazdanpanah, Y.; Boëlle, P.-Y.; Hens, N.; Dervaux, B. The French connection: The first large population-based contact survey in France relevant for the spread of infectious diseases. PLoS ONE 2015, 10, e0133203. [Google Scholar] [CrossRef] [Green Version]
- Russell, T.W.; Hellewell, J.; Jarvis, C.I.; van Zandvoort, K.; Abbott, S.; Ratnayake, R.; Flasche, S.; Eggo, R.M.; Edmunds, W.J.; Kucharski, A.J. Cmmid Covid-Working Group, Estimating the infection and case fatality ratio for coronavirus disease (COVID-19) using age-adjusted data from the outbreak on the Diamond Princess cruise ship, February 2020. Euro Surveill. 2020, 25, 2000256. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mizumoto, K.; Kagaya, K.; Zarebski, A.; Chowell, G. Estimating the asymptomatic proportion of coronavirus disease 2019 (COVID-19) cases on board the Diamond Princess cruise ship, Yokohama, Japan, 2020. Euro Surveill. 2020, 25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Verity, R.; Okell, L.C.; Dorigatti, I.; Winskill, P.; Whittaker, C.; Imai, N.; Cuomo-Dannenburg, G.; Thompson, H.; Walker, P.G.; Fu, H.; et al. Estimates of the severity of coronavirus disease 2019: A model-based analysis. Lancet Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Bao, L.; Deng, W.; Gao, H.; Xiao, C.; Liu, J.; Xue, J.; Lv, Q.; Liu, J.; Yu, P.; Xu, Y.; et al. Reinfection could not occur in SARS-CoV-2 infected rhesus macaques. bioRxiv 2020. [Google Scholar] [CrossRef] [Green Version]
- Lauer, S.A.; Grantz, K.H.; Bi, Q.; Jones, F.K.; Zheng, Q.; Meredith, H.R.; Azman, A.S.; Reich, N.G.; Lessler, J. The Incubation Period of Coronavirus Disease 2019 (COVID-19) From Publicly Reported Confirmed Cases: Estimation and Application. Ann. Intern. Med. 2020, 172, 577–582. [Google Scholar] [CrossRef] [Green Version]
- Du, Z.; Xu, X.; Wu, Y.; Wang, L.; Cowling, B.J.; Meyers, L.A. Serial interval of COVID-19 among publicly reported confirmed cases. Emerg. Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Worldometer. 2020. Available online: https://www.worldometers.info/coronavirus (accessed on 24 August 2020).
- Kissler, S.M.; Tedijanto, C.; Goldstein, E.; Grad, Y.H.; Lipsitch, M. Projecting the transmission dynamics of SARS-CoV-2 through the postpandemic period. Science 2020, 368, 860–868. [Google Scholar] [CrossRef]
Sample Availability:Pandæsim can be downloaded on its dedicated website: https://pandaesim.lri.fr. |



© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amar, P. Pandæsim: An Epidemic Spreading Stochastic Simulator. Biology 2020, 9, 299. https://doi.org/10.3390/biology9090299
Amar P. Pandæsim: An Epidemic Spreading Stochastic Simulator. Biology. 2020; 9(9):299. https://doi.org/10.3390/biology9090299
Chicago/Turabian StyleAmar, Patrick. 2020. "Pandæsim: An Epidemic Spreading Stochastic Simulator" Biology 9, no. 9: 299. https://doi.org/10.3390/biology9090299
APA StyleAmar, P. (2020). Pandæsim: An Epidemic Spreading Stochastic Simulator. Biology, 9(9), 299. https://doi.org/10.3390/biology9090299





