Spectral Distribution of Ultra-Weak Photon Emission as a Response to Wounding in Plants: An In Vivo Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Arabidopsis Plants and Leaves
2.2. Wound Treatment for Ultra-Weak Photon Emission Measurement
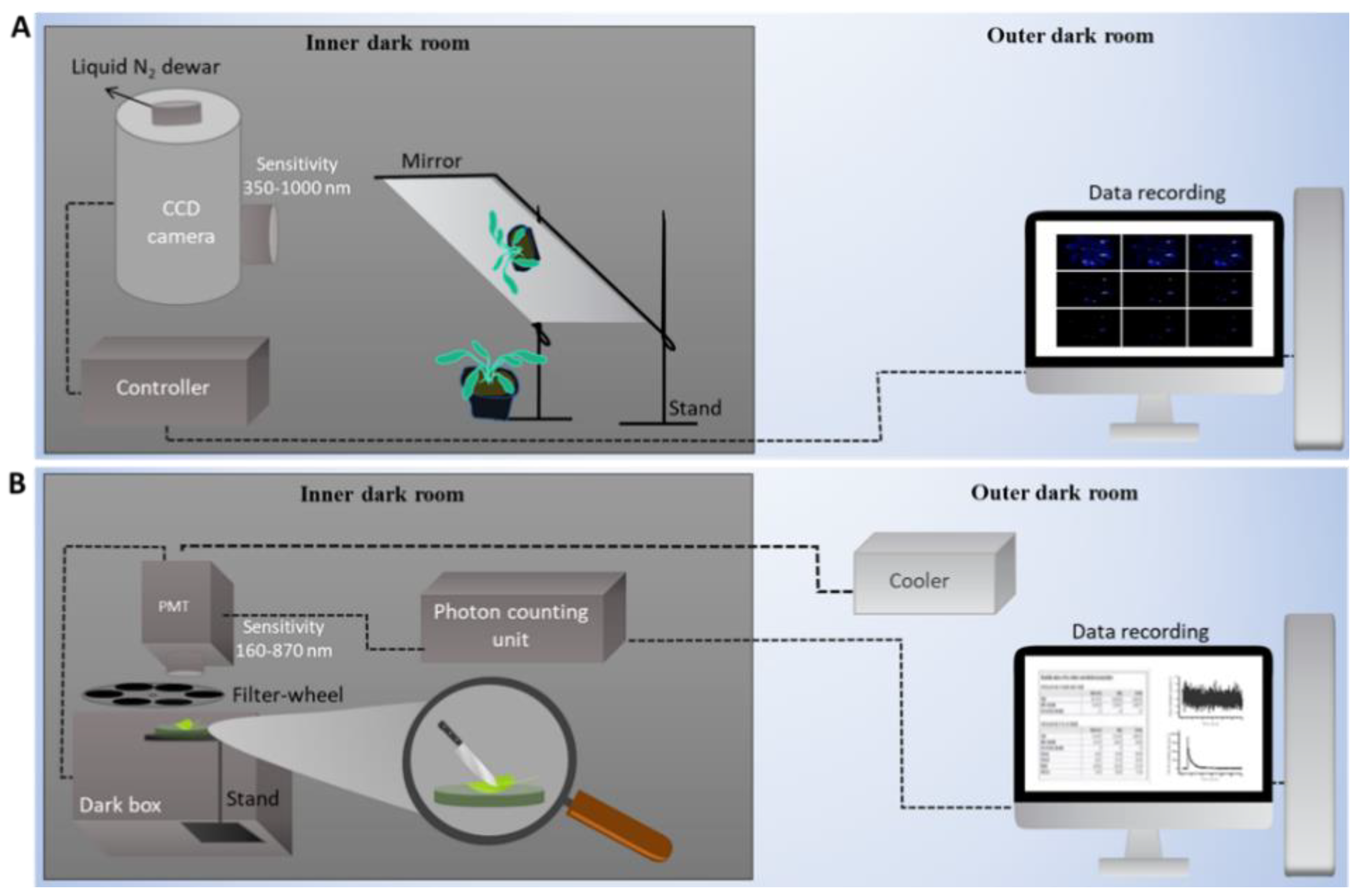
2.3. Ultra-Weak Photon Emission
2.4. Procedure for Spectral Measurement and Data Evaluation
3. Results
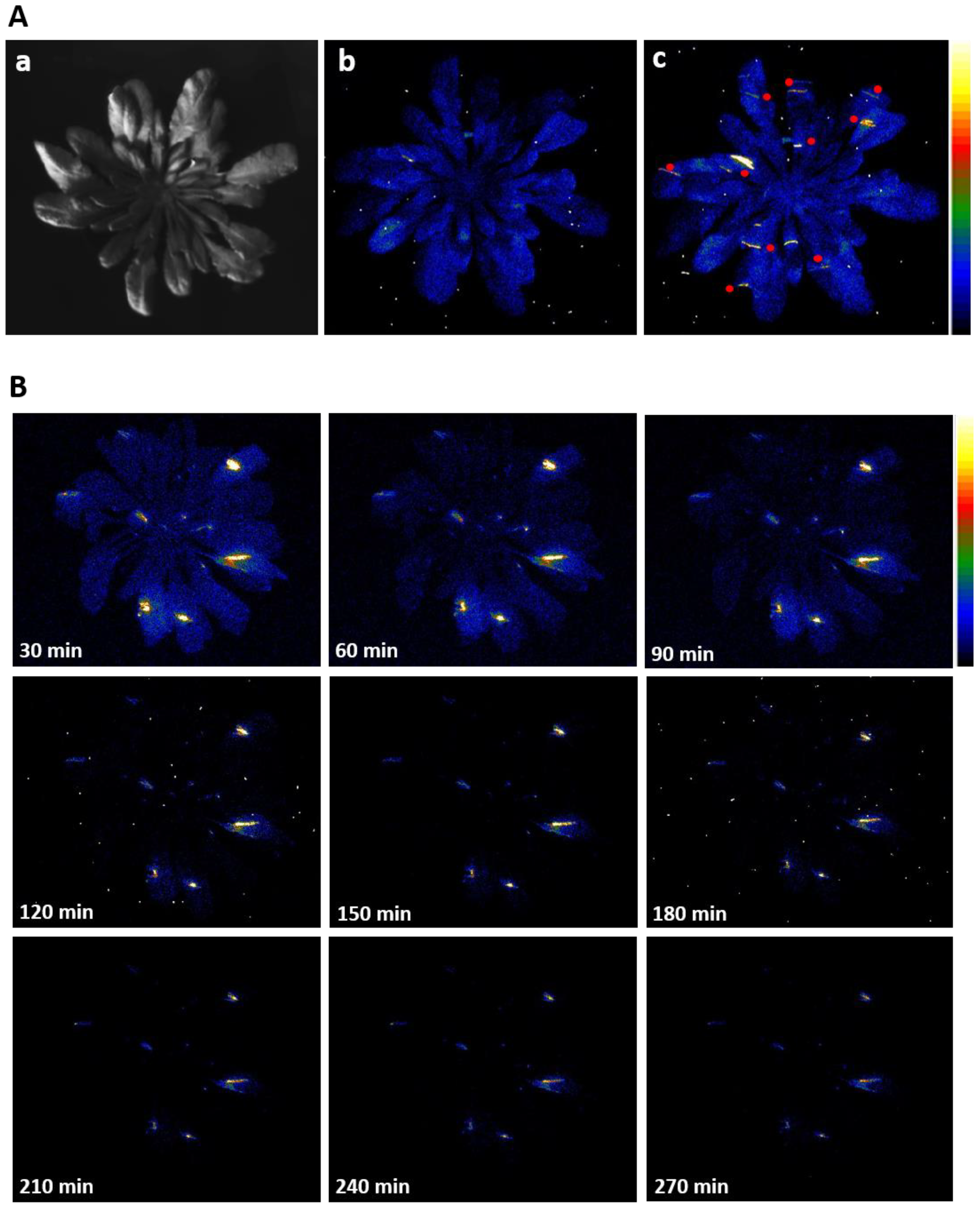
3.1. Wounding and Ultra-Weak Photon Emission Imaging
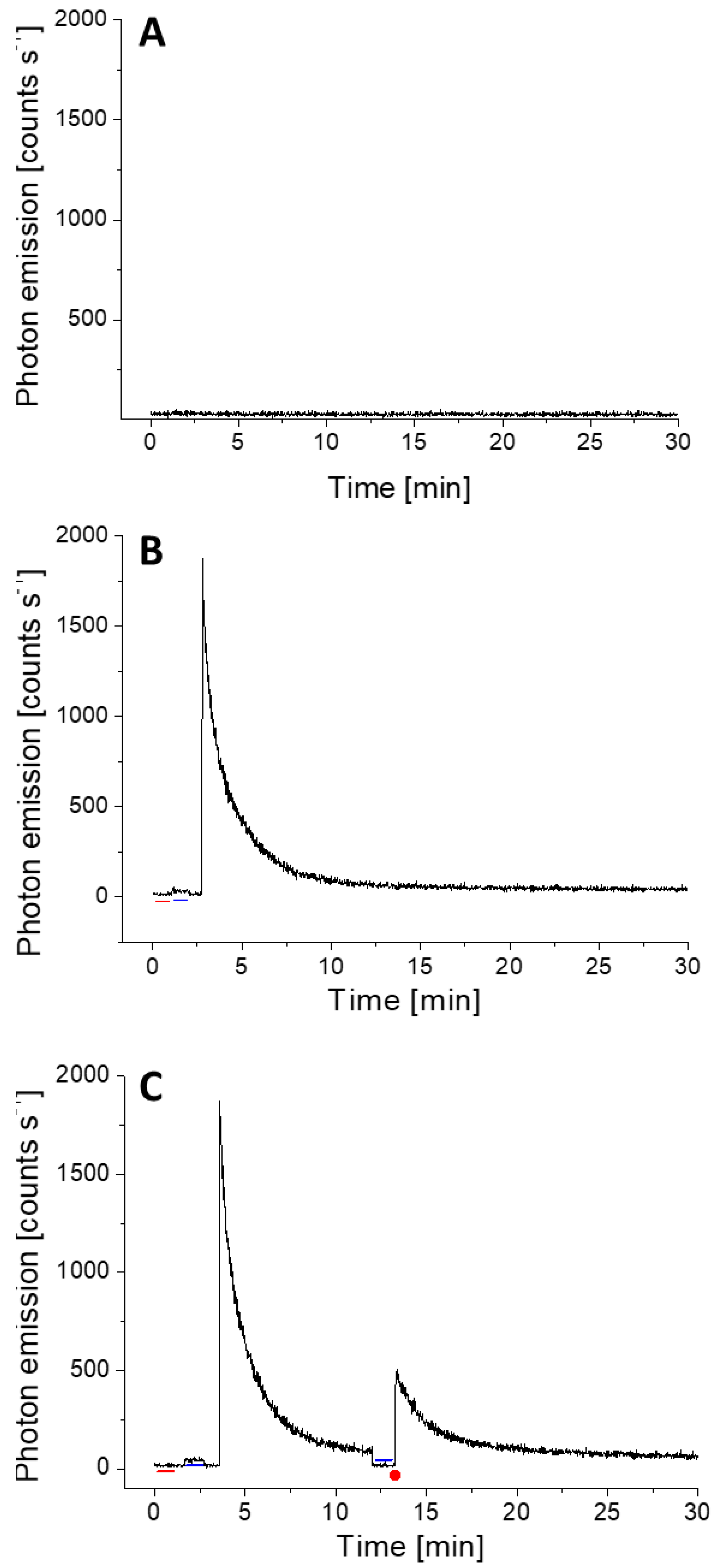
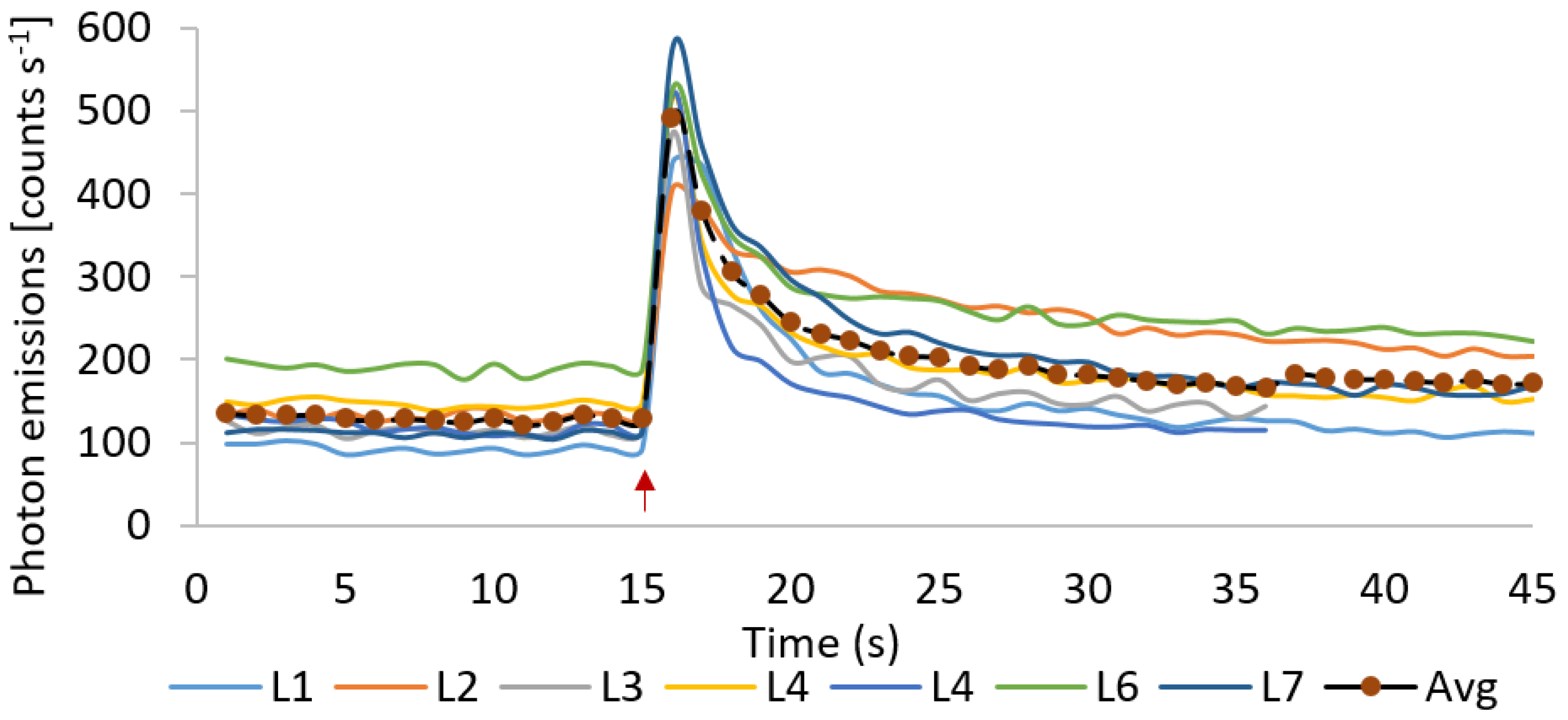
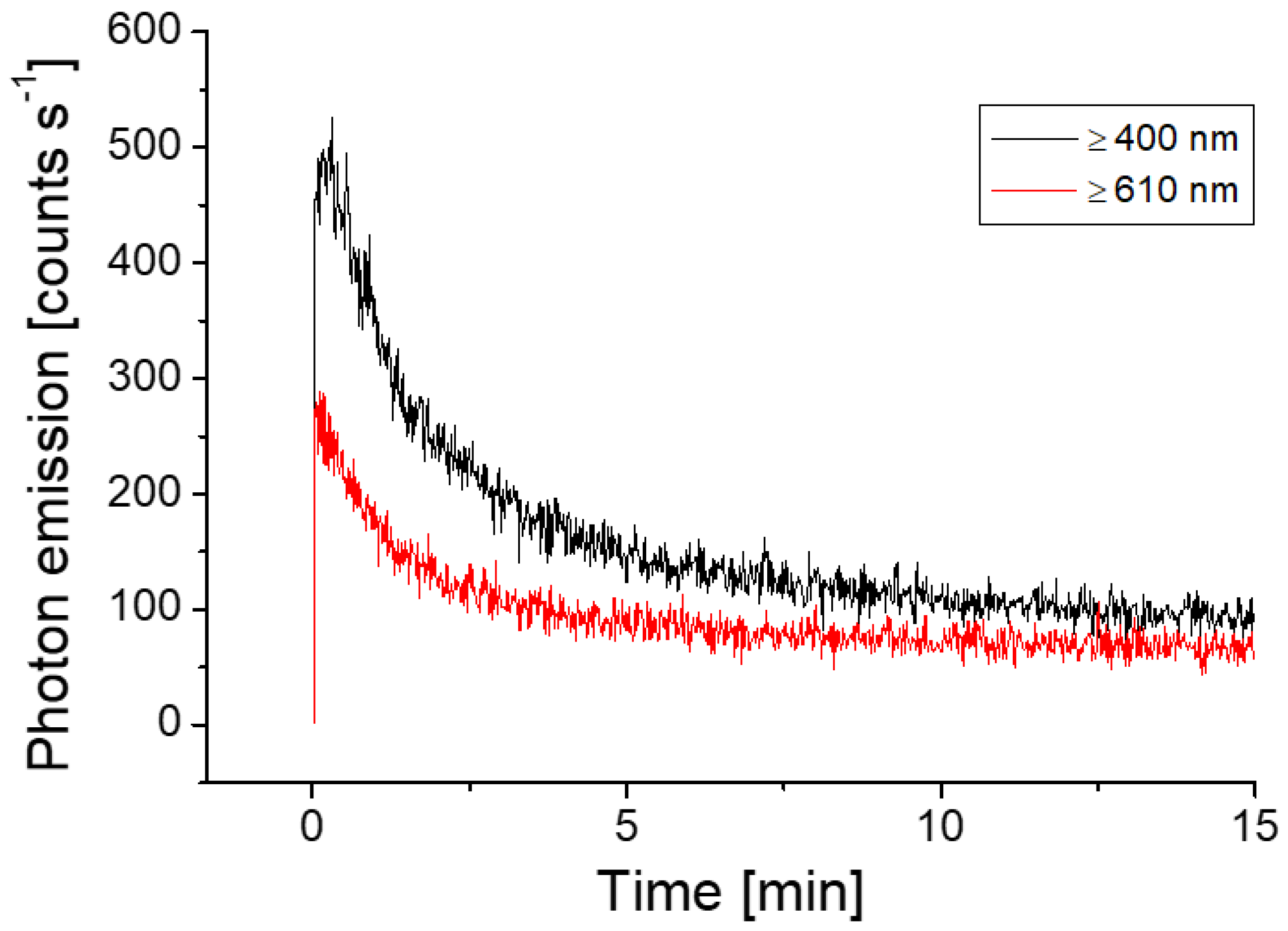
3.2. Delayed Luminescence and Wound-Induced Ultra-Weak Photon Emission
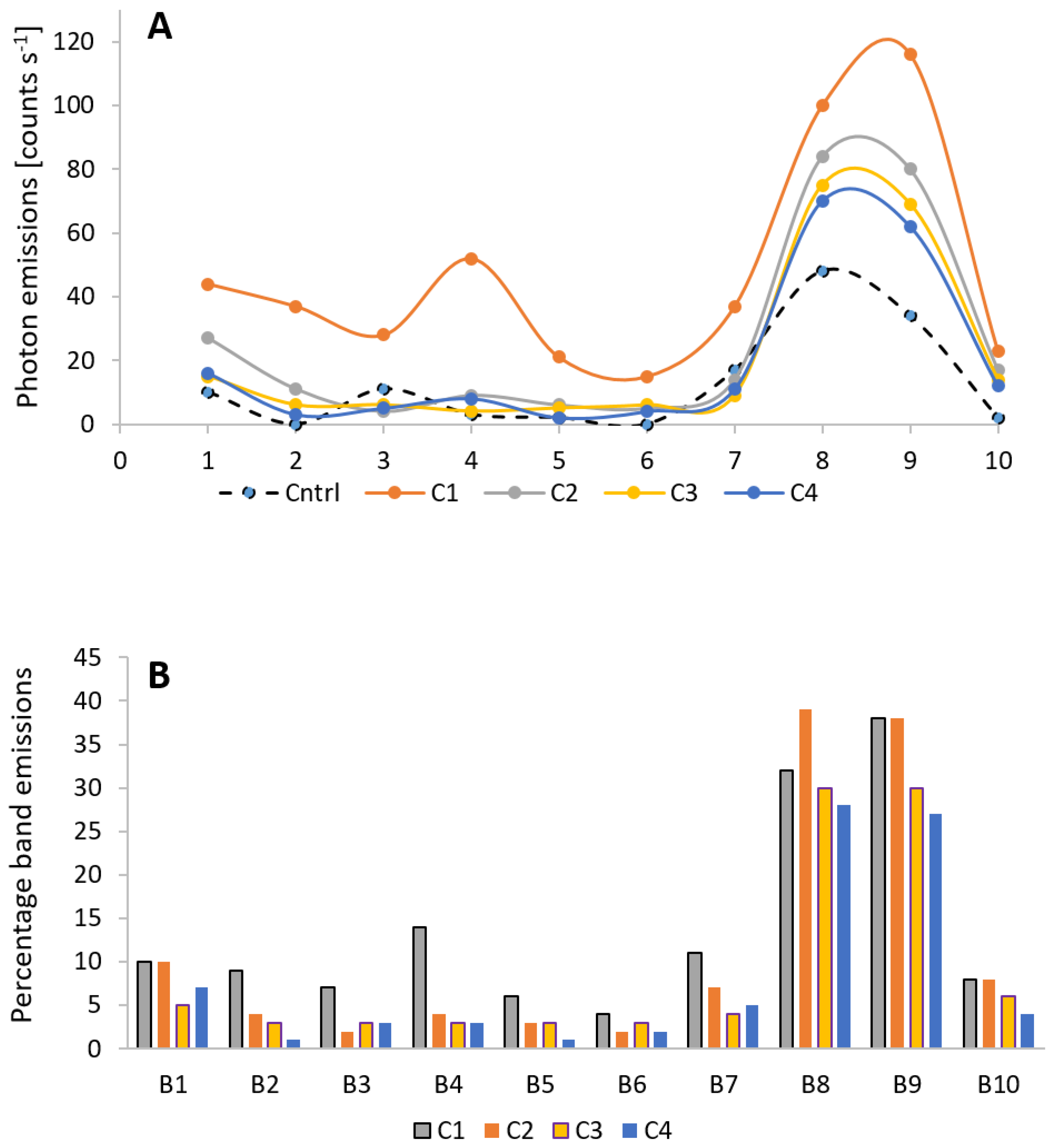
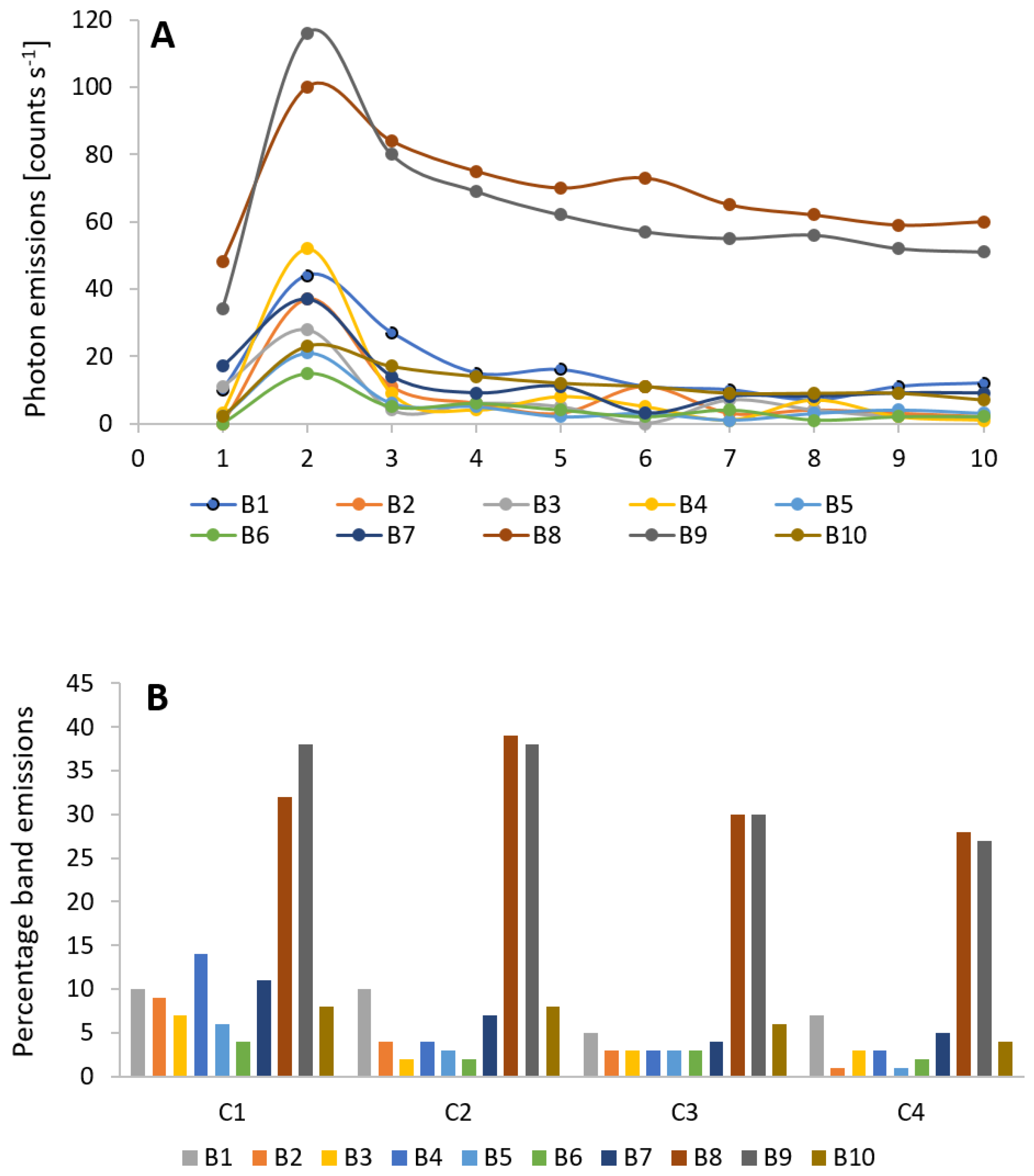
3.3. Wounding and Spectral Distribution of Ultra-Weak Photon Emission
3.4. Oxidative Stress, Electronically Excited Species, and Ultra-Weak Photon Emission
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Usa, M.; Kobayashi, M.; Scott, R.Q.; Maeda, T.; Hiratsuka, R.; Inaba, H. Simultaneous measurement of biophoton emission and biosurface electric potential from germinating soybean (Glycine max). Protoplasma 1989, 149, 64–66. [Google Scholar] [CrossRef]
- Devaraj, B. Biophotons: Ultraweak light emission from living systems. Curr. Opin. Solid State Mater. Sci. 1997, 2, 188–193. [Google Scholar] [CrossRef]
- Kobayashi, M. Modern Technology on Physical Analysis of Biophoton Emission and Its Potential Extracting the Physiological Information. In Fluctuation Phenomena: Disorder and Nonlinearity; World Scientific Publishing Company: Singapore, 2003; pp. 157–187. [Google Scholar]
- Zhao, X.; Pang, J.; Fu, J.; Wang, Y.; Yang, M.; Liu, Y.; Fan, H.; Zhang, L.; Han, J. Spontaneous photon emission: A promising non-invasive diagnostic tool for breast cancer. J. Photochem. Photobiol. B. Biol. 2017, 166, 232–238. [Google Scholar] [CrossRef] [PubMed]
- Cifra, M.; Pospíšil, P. Ultra-weak photon emission from biological samples: Definition, mechanisms, properties, detection and applications. J. Photochem. Photobiol. B. Biol. 2014, 139, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Cifra, M.; Brouder, C.; Nerudová, M.; Kucera, O. Biophotons, coherence and photocount statistics: A critical review. J. Lumin. 2015, 164, 38–51. [Google Scholar] [CrossRef]
- Inagaki, H.; Imaizumi, T.; Wang, G.-X.; Tominaga, T.; Kato, K.; Iyozumi, H.; Nukui, H. Spontaneous ultraweak photon emission from rice (Oryza sativa L.) and paddy weeds treated with a sulfonylurea herbicide. Pestic. Biochem. Physiol. 2007, 89, 158–162. [Google Scholar] [CrossRef]
- Kato, K.; Iyozumi, H.; Kageyama, C.; Inagaki, H.; Yamaguchi, A.; Nukui, H. Application of ultra-weak photon emission measurements in agriculture. J. Photochem. Photobiol. B. Biol. 2014, 139, 54–62. [Google Scholar] [CrossRef]
- Makino, T.; Kato, K.; Lyozumi, H.; Honzawa, H.; Tachiiri, Y.; Hiramatsu, M. Ultraweak Luminescence Generated by Sweet Potato andFusarium oxysporumInteractions Associated with a Defense Response. Photochem. Photobiol. 1996, 64, 953–956. [Google Scholar] [CrossRef]
- Hakamata, T.; Katō, K.; Makino, T.; Yamamoto, S. Weak photon emission from the Japanese black pine inoculated with pine wood nematode. Jpn. J. Phytopathol. 2004, 70, 162–167. [Google Scholar] [CrossRef][Green Version]
- Musumeci, F.; Applegate, L.A.; Privitera, G.; Scordino, A.; Tudisco, S.; Niggli, H.J. Spectral analysis of laser-induced ultraweak delayed luminescence in cultured normal and tumor human cells: Temperature dependence. J. Photochem. Photobiol. B. Biol. 2005, 79, 93–99. [Google Scholar] [CrossRef]
- Nakamura, K.; Hiramatsu, M. Ultra-weak photon emission from human hand: Influence of temperature and oxygen concentration on emission. J. Photochem. Photobiol. B. Biol. 2005, 80, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Cifra, M.; van Wijk, E.; van Wijk, R. Temperature Induced Changes of Spontaneous Photon Emission from Human Hands. In Proceedings of the PIERS, Cambridge, MA, USA, 2–6 July 2008; pp. 907–911. [Google Scholar]
- Chen, W.L.; Xing, D.; Tan, S.; Tang, Y.; He, Y. Imaging of ultra-weak bio-chemiluminescence and singlet oxygen generation in germinating soybean in response to wounding. Luminescence 2003, 18, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Kawabata, R.; Miike, T.; Uefune, M.; Okabe, H.; Takagi, M.; Kai, S. Biophoton Measurement of Herbivore-Induced Plant Responses. Jpn. J. Appl. Èntomol. ZooL. 2004, 48, 289–296. [Google Scholar] [CrossRef]
- Iyozumi, H.; Kato, K.; Kageyama, C.; Inagaki, H.; Yamaguchi, A.; Furuse, K.; Baba, K.; Tsuchiya, H. Plant defense activators potentiate the generation of elicitor-responsive photon emission in rice. Physiol. Mol. Plant Pathol. 2005, 66, 68–74. [Google Scholar] [CrossRef]
- Bertogna, E.; Bezerra, J.; Conforti, E.; Gallep, C.M. Acute stress in seedlings detected by ultra-weak photon emission. J. Photochem. Photobiol. B. Biol. 2013, 118, 74–76. [Google Scholar] [CrossRef]
- Kausar, R.; Hossain, Z.; Makino, T.; Komatsu, S. Characterization of ascorbate peroxidase in soybean under flooding and drought stresses. Mol. Boil. Rep. 2012, 39, 10573–10579. [Google Scholar] [CrossRef]
- Van Breusegem, F.; Vranová, E.; Dat, J.F.; Inzé, D. The role of active oxygen species in plant signal transduction. Plant Sci. 2001, 161, 405–414. [Google Scholar] [CrossRef]
- Prasad, A.; Sedlářová, M.; Kale, R.; Pospíšil, P. Lipoxygenase in singlet oxygen generation as a response to wounding: In vivo imaging in Arabidopsis thaliana. Sci. Rep. 2017, 7, 9831. [Google Scholar] [CrossRef]
- Prasad, A.; Sedlářová, M.; Balukova, A.; Rác, M.; Pospíšil, P. Reactive Oxygen Species as a Response to Wounding: In Vivo Imaging in Arabidopsis thaliana. Front. Plant Sci. 2020, 10. [Google Scholar] [CrossRef]
- Prasad, A.; Pospíšil, P. Two-dimensional imaging of spontaneous ultra-weak photon emission from the human skin: Role of reactive oxygen species. J. Biophotonics 2011, 4, 840–849. [Google Scholar] [CrossRef]
- Prasad, A.; Pospíšil, P. Linoleic Acid-Induced Ultra-Weak Photon Emission from Chlamydomonas reinhardtii as a Tool for Monitoring of Lipid Peroxidation in the Cell Membranes. PLoS ONE 2011, 6, e22345. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, A.; Pospíšil, P. Production of hydrogen peroxide and hydroxyl radical in potato tuber during the necrotrophic phase of hemibiotrophic pathogen Phytophthora infestans infection. J. Photochem. Photobiol. B. Biol. 2012, 117, 202–206. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A.; Pospíšil, P. Towards the two-dimensional imaging of spontaneous ultra-weak photon emission from microbial, plant and animal cells. Sci. Rep. 2013, 3. [Google Scholar] [CrossRef] [PubMed]
- Pospíšil, P.; Prasad, A.; Rác, M. Role of reactive oxygen species in ultra-weak photon emission in biological systems. J. Photochem. Photobiol. B. Biol. 2014, 139, 11–23. [Google Scholar] [CrossRef]
- Rác, M.; Sedlářová, M.; Pospíšil, P. The formation of electronically excited species in the human multiple myeloma cell suspension. Sci. Rep. 2015, 5, 8882. [Google Scholar] [CrossRef]
- Van Wijk, E.P.A.; van Wijk, R.; Cifra, M. Spontaneous ultra-weak photon emission from human hands varies diurnally—Art. No. 66331J. In Biophotonics 2007: Optics in Life Science2007, Proceedings of the SPIE-OSA Biomedical Optics, Munich, Germany, 2007; Popp, J., VonBally, G., Eds.; SPIE: Bellingham, WA, USA, 2007; p. J6331. [Google Scholar]
- Burgos, R.C.R.; Schoeman, J.C.; Van Winden, L.J.; Cervinková, K.; Ramautar, R.; Van Wijk, E.P.A.; Cifra, M.; Berger, R.; Hankemeier, T.; Van Der Greef, J. Ultra-weak photon emission as a dynamic tool for monitoring oxidative stress metabolism. Sci. Rep. 2017, 7, 1229. [Google Scholar] [CrossRef]
- Wijk, R.V.; Wijk, E.P.A.V.; Wiegant, F.A.C.; Ives, J. Free radicals and low-level photon emission in human pathogenesis: State of the art. Indian J. Exp. Biol. 2008, 46, 273–309. [Google Scholar]
- Hossu, M.; Ma, L.; Chen, W. Nonlinear enhancement of spontaneous biophoton emission of sweet potato by silver nanoparticles. J. Photochem. Photobiol. B. Biol. 2010, 99, 44–48. [Google Scholar] [CrossRef]
- Hossu, M.; Ma, L.; Zou, X.; Chen, W. Enhancement of biophoton emission of prostate cancer cells by Ag nanoparticles. Cancer Nanotechnol. 2013, 4, 21–26. [Google Scholar] [CrossRef]
- Rastogi, A.; Pospíšil, P. Effect of exogenous hydrogen peroxide on biophoton emission from radish root cells. Plant Physiol. Biochem. 2010, 48, 117–123. [Google Scholar] [CrossRef]
- Tsuchida, K.; Kobayashi, M. Oxidative stress in human facial skin observed by ultraweak photon emission imaging and its correlation with biophysical properties of skin. Sci. Rep. 2020, 10, 9626. [Google Scholar] [CrossRef] [PubMed]
- Desikan, R. Regulation of the Arabidopsis Transcriptome by Oxidative Stress. Plant Physiol. 2001, 127, 159–172. [Google Scholar] [CrossRef] [PubMed]
- Neill, S.J. Hydrogen peroxide signalling. Curr. Opin. Plant Biol. 2002, 5, 388–395. [Google Scholar] [CrossRef]
- Neill, S.J.; Desikan, R.; Clarke, A.; Hancock, J.T. Nitric oxide is a novel component of abscisic acid signaling in stomatal guard cells. Plant Physiol. 2002, 128, 13–16. [Google Scholar] [CrossRef]
- Dat, J.; Vandenabeele, S.; Van Montagu, M.; Van Breusegem, F.; Vranová, E.; Inzé, D. Dual action of the active oxygen species during plant stress responses. Cell. Mol. Life Sci. 2000, 57, 779–795. [Google Scholar] [CrossRef]
- Mittler, R.; Vanderauwera, S.; Gollery, M.; Van Breusegem, F. Reactive oxygen gene network of plants. Trends Plant Sci. 2004, 9, 490–498. [Google Scholar] [CrossRef]
- Apel, K.; Hirt, H. Reactive oxygen species: Metabolism, Oxidative Stress, and Signal Transduction. Annu. Rev. Plant Biol. 2004, 55, 373–399. [Google Scholar] [CrossRef]
- Foyer, C.H.; Noctor, G. Redox Homeostasis and Antioxidant Signaling: A Metabolic Interface between Stress Perception and Physiological Responses. Plant Cell 2005, 17, 1866–1875. [Google Scholar] [CrossRef]
- Slawinski, J.; Ezzahir, A.; Godlewski, M.; Kwiecinska, T.; Rajfur, Z.; Sitko, D.; Wierzuchowska, D. Stress-induced photon emission from perturbed organisms. Cell. Mol. Life Sci. 1992, 48, 1041–1058. [Google Scholar] [CrossRef]
- Pospíšil, P.; Prasad, A.; Rác, M. Mechanism of the Formation of Electronically Excited Species by Oxidative Metabolic Processes: Role of Reactive Oxygen Species. Biomolecules 2019, 9, 258. [Google Scholar] [CrossRef]
- Van Wijk, R.; Van Wijk, E.P.; Bajpai, R.P. Photocount distribution of photons emitted from three sites of a human body. J. Photochem. Photobiol. B. Biol. 2006, 84, 46–55. [Google Scholar] [CrossRef]
- Denvir, D.J.; Coates, C.G. Electron-multiplying CCD technology: Application to ultrasensitive detection of biomolecules. Int. Symp. Biomed. Opt. 2002, 4626, 502–512. [Google Scholar] [CrossRef]
- Kobayashi, M.; Kikuchi, D.; Okamura, H. Imaging of Ultraweak Spontaneous Photon Emission from Human Body Displaying Diurnal Rhythm. PLoS ONE 2009, 4, e6256. [Google Scholar] [CrossRef] [PubMed]
- Tsuchida, K.; Iwasa, T.; Kobayashi, M. LB1586 Noninvasive imaging of UV-induced oxidative stress in human skin using ultra-weak photon emission. J. Investig. Dermatol. 2018, 138, B20. [Google Scholar] [CrossRef]
- Usui, S.; Tada, M.; Kobayashi, M. Non-invasive visualization of physiological changes of insects during metamorphosis based on biophoton emission imaging. Sci. Rep. 2019, 9, 8576. [Google Scholar] [CrossRef]
- Havaux, M.; Triantaphylides, C.; Genty, B. Autoluminescence imaging: A non-invasive tool for mapping oxidative stress. Trends Plant Sci. 2006, 11, 480–484. [Google Scholar] [CrossRef]
- SaeidFirozeh, H.; Shafiekhani, A.; Cifra, M.; Masoudi, A.A. Endogenous Chemiluminescence from Germinating Arabidopsis Thaliana Seeds. Sci. Rep. 2018, 8, 16231. [Google Scholar] [CrossRef]
- Savatin, D.V.; Gramegna, G.; Modesti, V.; Cervone, F. Wounding in the plant tissue: The defense of a dangerous passage. Front. Plant Sci. 2014, 5, 470. [Google Scholar] [CrossRef]
- Oros, C.; Alves, F. Leaf wound induced ultraweak photon emission is suppressed under anoxic stress: Observations of Spathiphyllum under aerobic and anaerobic conditions using novel in vivo methodology. PLoS ONE 2018, 13, e0198962. [Google Scholar] [CrossRef]
- Flor-Henry, M.; McCabe, T.C.; De Bruxelles, G.L.; Roberts, M.R. Use of a highly sensitive two-dimensional luminescence imaging system to monitor endogenous bioluminescence in plant leaves. BMC Plant Boil. 2004, 4, 19. [Google Scholar] [CrossRef]
- Birtic, S.; Triantaphylidès, C.; Ksas, B.; Genty, B.; Mueller, M.J.; Havaux, M. Using spontaneous photon emission to image lipid oxidation patterns in plant tissues. Plant J. 2011, 67, 1103–1115. [Google Scholar] [CrossRef] [PubMed]
- Havaux, M. Spontaneous and thermoinduced photon emission: New methods to detect and quantify oxidative stress in plants. Trends Plant Sci. 2003, 8, 409–413. [Google Scholar] [CrossRef]
- Makino, T.; Kato, K.; Iyozumi, H.; Aoshima, Y. Biophoton emission and defence systems in plants. In Optical Science and Engineering for the 21st Century; Shen, X., Van Wijk, R., Eds.; Springer: New York, NY, USA, 2005; pp. 205–218. [Google Scholar]
| Band Emission | b1 | b2 | b3 | b4 | b5 |
|---|---|---|---|---|---|
| Band range (nm) | <350 | 350–400 | 400–455 | 455–495 | 495–550 |
| Band emission | b6 | b7 | b8 | b9 | b10 |
| Band range (nm) | 550–610 | 610–645 | 645–695 | 695–780 | >780 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prasad, A.; Gouripeddi, P.; Devireddy, H.R.N.; Ovsii, A.; Rachakonda, D.P.; Wijk, R.V.; Pospíšil, P. Spectral Distribution of Ultra-Weak Photon Emission as a Response to Wounding in Plants: An In Vivo Study. Biology 2020, 9, 139. https://doi.org/10.3390/biology9060139
Prasad A, Gouripeddi P, Devireddy HRN, Ovsii A, Rachakonda DP, Wijk RV, Pospíšil P. Spectral Distribution of Ultra-Weak Photon Emission as a Response to Wounding in Plants: An In Vivo Study. Biology. 2020; 9(6):139. https://doi.org/10.3390/biology9060139
Chicago/Turabian StylePrasad, Ankush, Prabhakar Gouripeddi, Hanumanth Rao Naidu Devireddy, Alina Ovsii, Dattatreya Prabhu Rachakonda, Roeland Van Wijk, and Pavel Pospíšil. 2020. "Spectral Distribution of Ultra-Weak Photon Emission as a Response to Wounding in Plants: An In Vivo Study" Biology 9, no. 6: 139. https://doi.org/10.3390/biology9060139
APA StylePrasad, A., Gouripeddi, P., Devireddy, H. R. N., Ovsii, A., Rachakonda, D. P., Wijk, R. V., & Pospíšil, P. (2020). Spectral Distribution of Ultra-Weak Photon Emission as a Response to Wounding in Plants: An In Vivo Study. Biology, 9(6), 139. https://doi.org/10.3390/biology9060139





