Transcriptome Analysis of Yamame (Oncorhynchus masou) in Normal Conditions after Heat Stress
Abstract
1. Introduction
2. Materials and Methods
2.1. Fish Samples for Sequencing
2.2. Constructing mRNA Libraries and Sequencing
2.3. Data Analysis of mRNAs
2.4. Functional Gene Enrichment Analysis of DEGs
2.5. Data Accessibility
3. Results
3.1. De Novo Assembly of Gill and Adipose Fin Tissues Transcripts
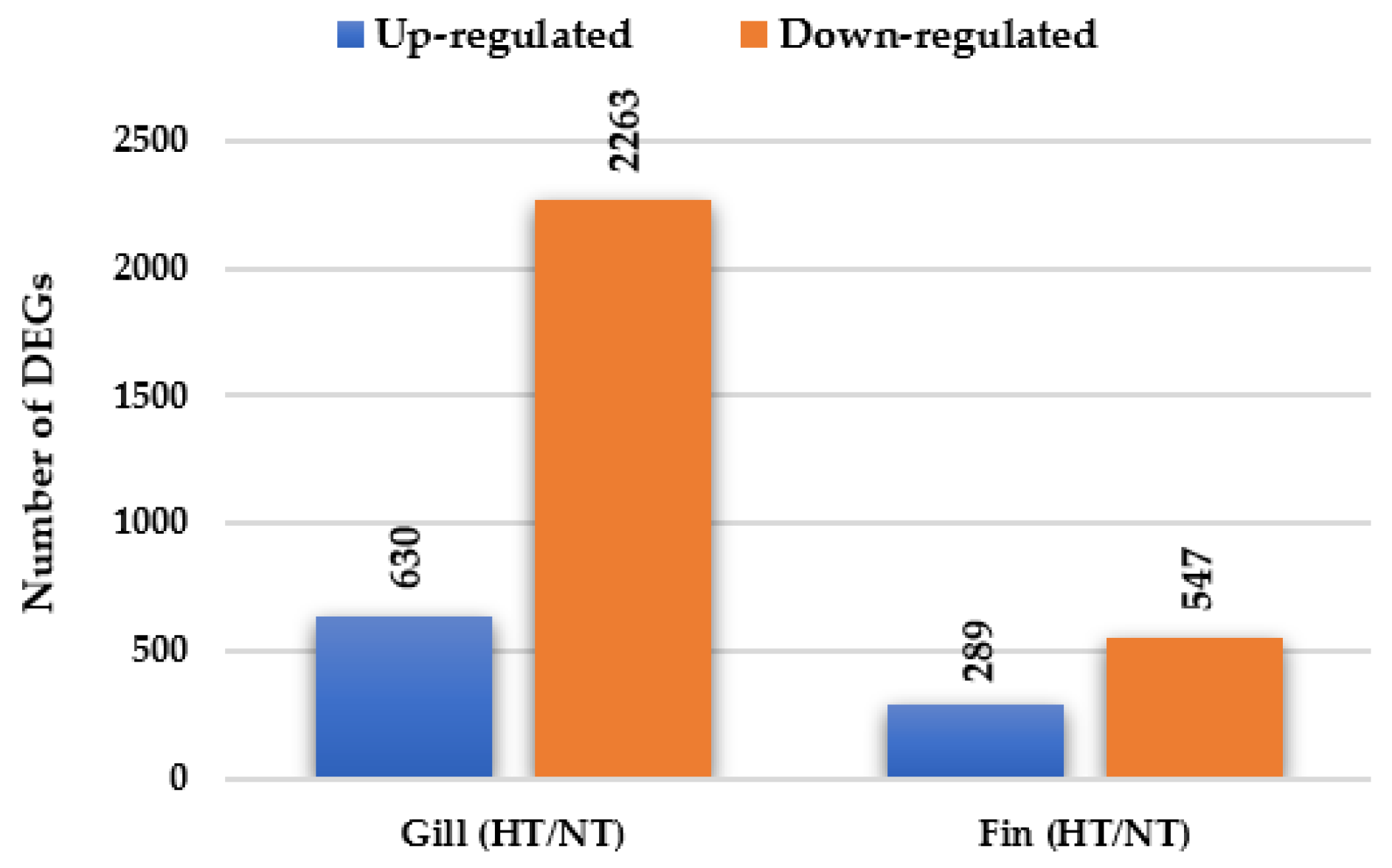
3.2. Gene Expression Profiling
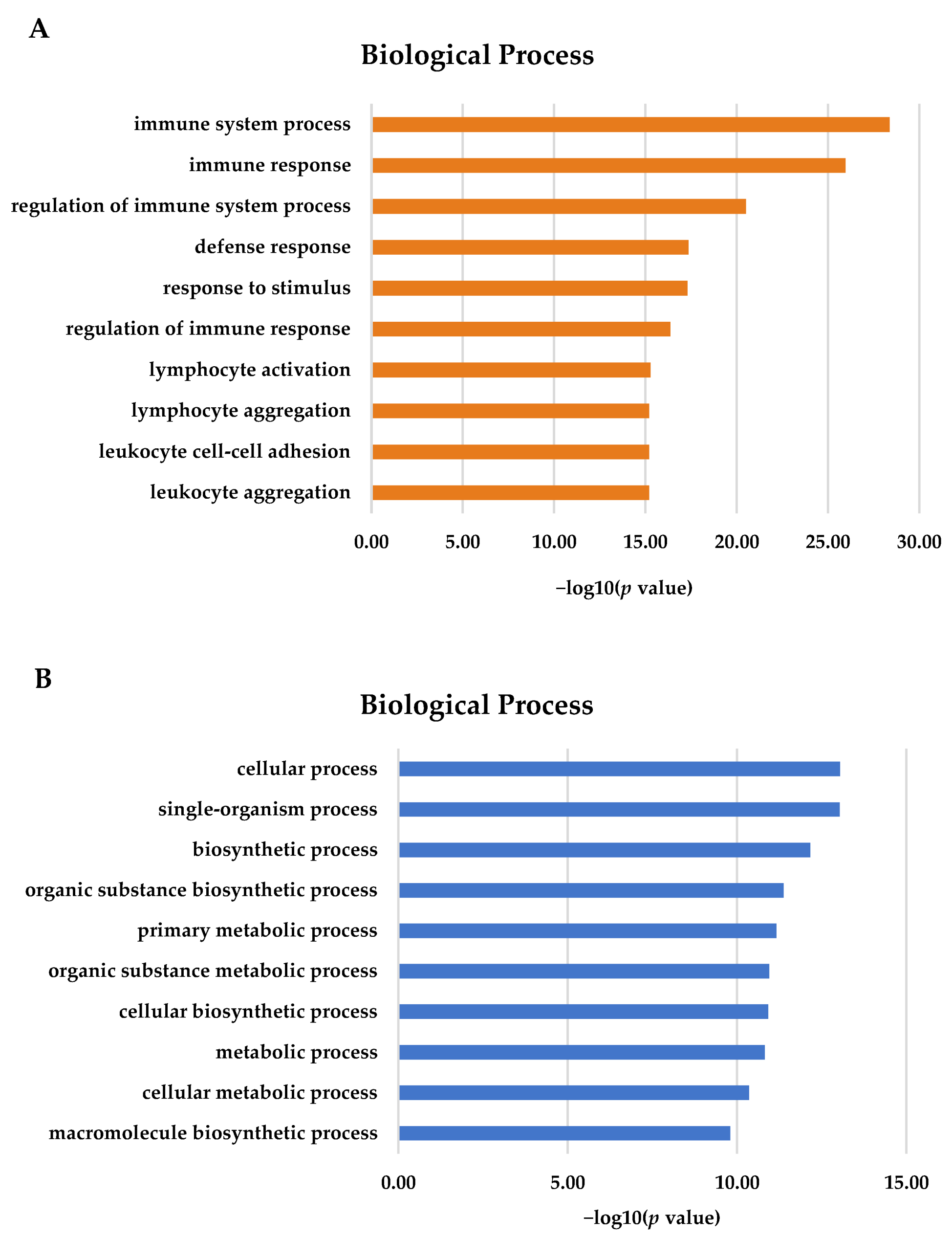
3.3. GO Analysis of DEGs in Gill and Adipose Fin
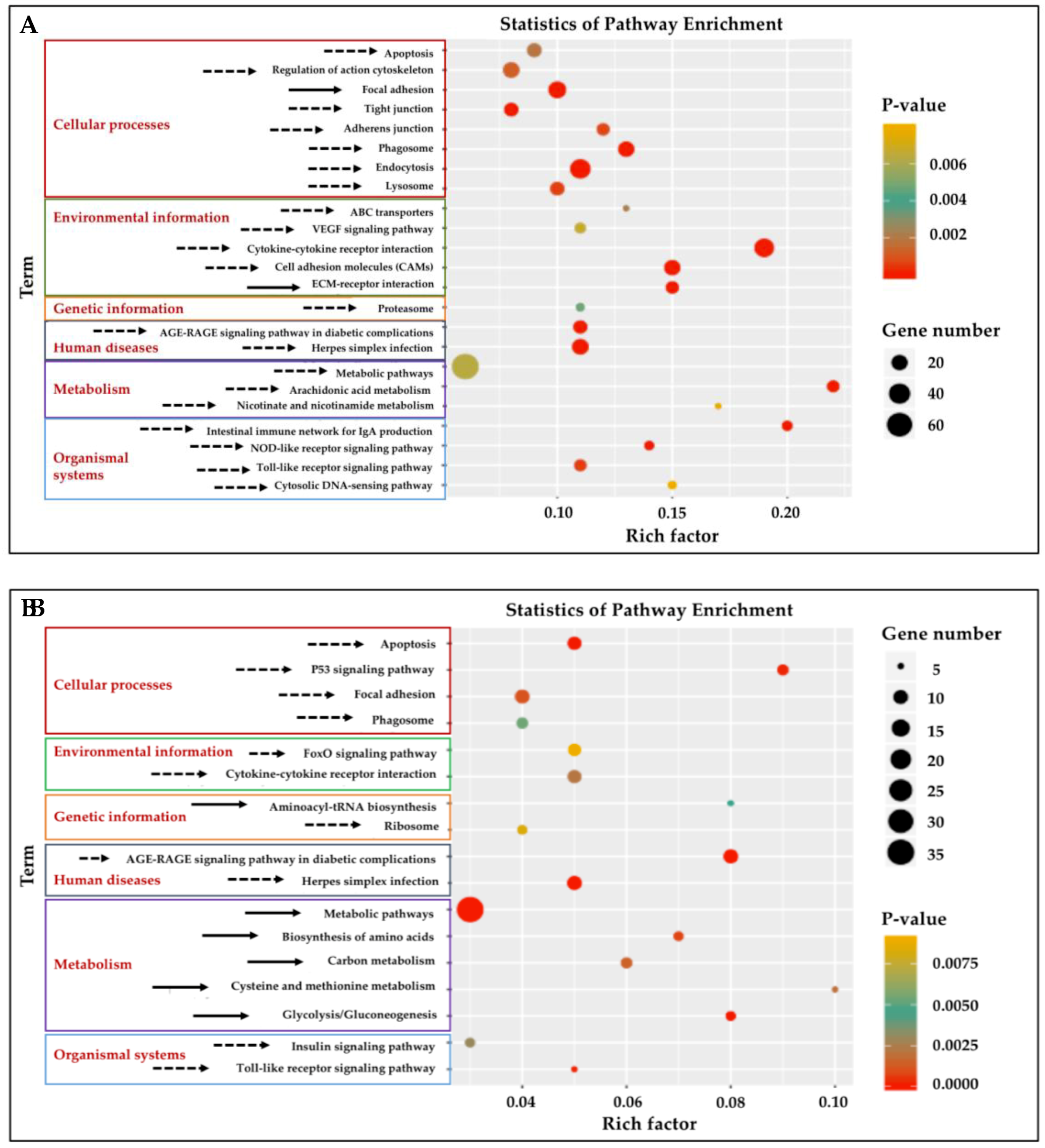
3.4. KEGG Pathway Enrichment Analysis of DEGs in Gill and Adipose Fin
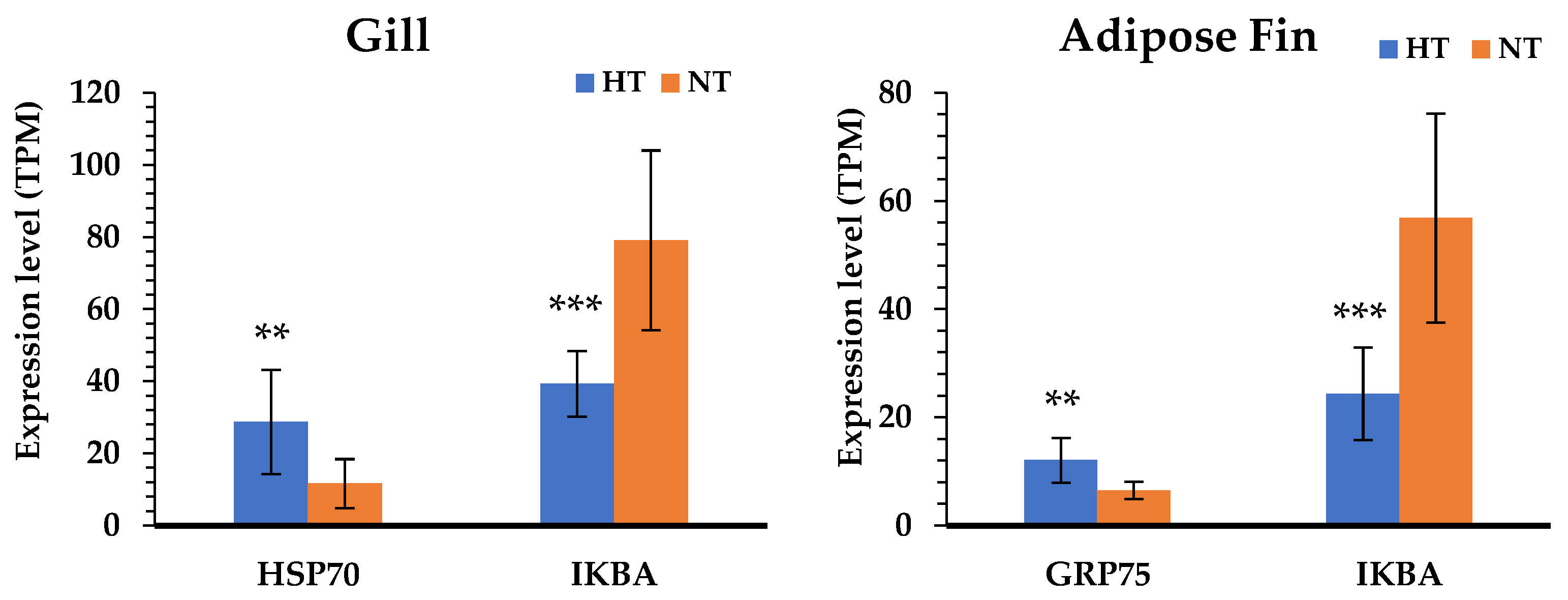
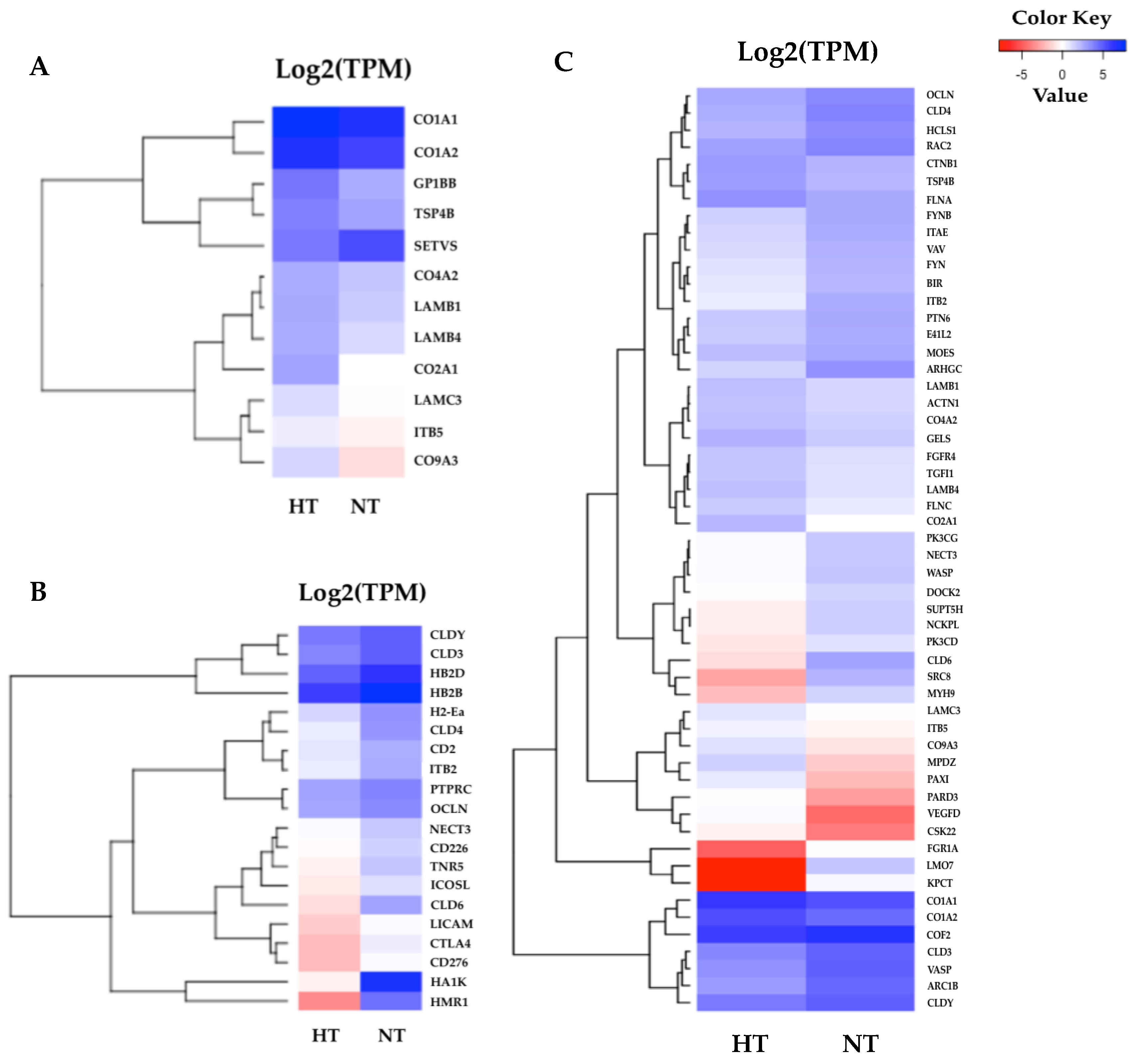
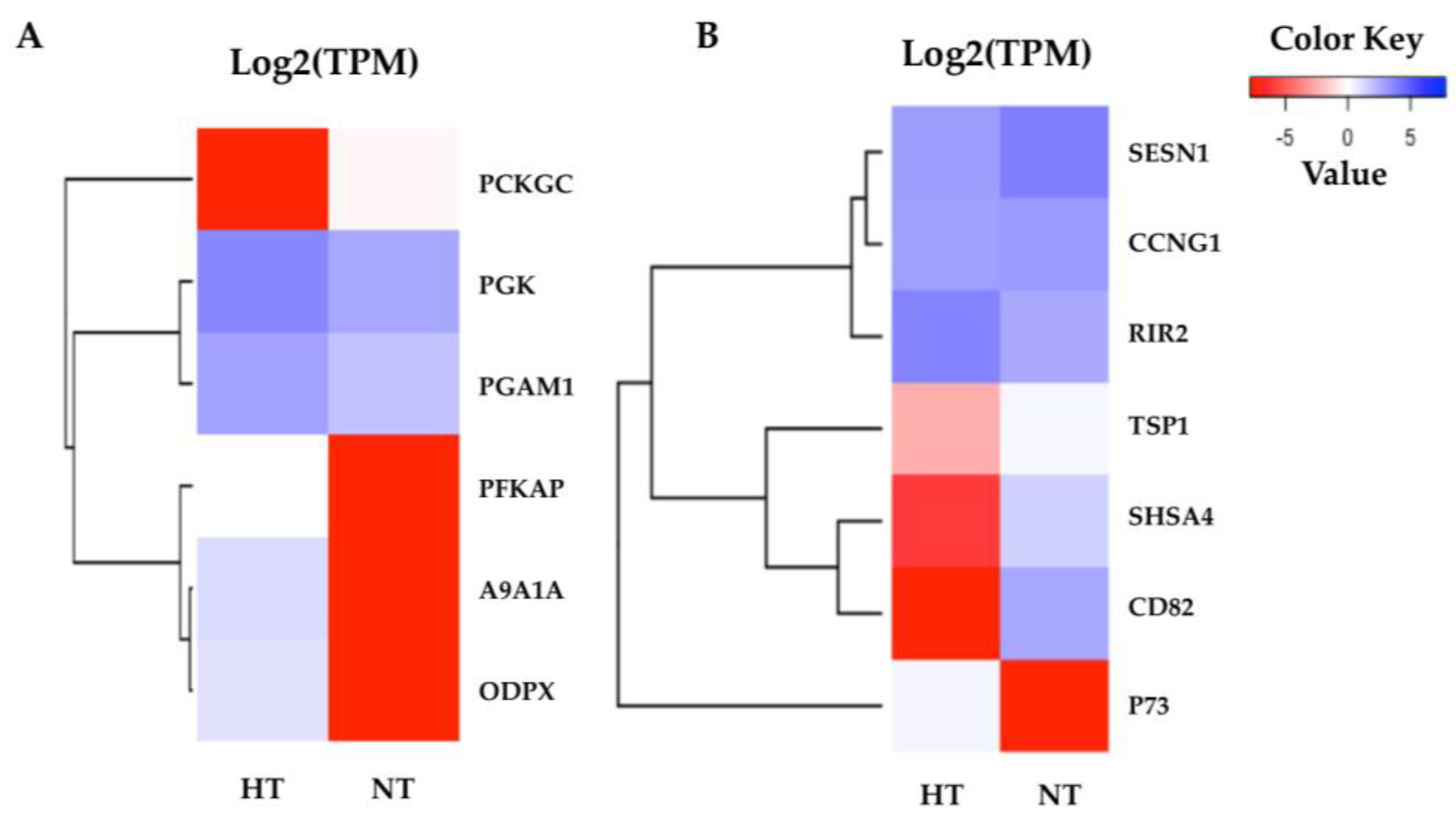
3.5. Specific Transcriptomic Responses in Gill Tissues
3.6. Specific Transcriptomic Responses in Fin Tissues
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wang, C.; Xu, Q.; Bai, Q.; Yin, J.; Jia, Z. Comparison of Growth Performances, Nutritional Composition in Muscle of Diploid and Triploid Masu Salmon (Oncorhynchus masou B., 1856). Turk. J. Fish Aquat. Sci. 2015, 15, 127–135. [Google Scholar] [CrossRef]
- Munakata, A. Migratory Behaviors in Masu Salmon (Oncorhynchus masou) and the Influence of Endocrinological Factors. Aqua BioSci. Monogr. 2012, 5, 29–65. [Google Scholar] [CrossRef]
- Lu, Y.; Wu, Z.; Song, Z.; Xiao, P.; Liu, Y.; Zhang, P.; You, F. Insight into the heat resistance of fish via blood: Effects of heat stress on metabolism, oxidative stress and antioxidation response of olive flounder Paralichthys olivaceus and turbot Scophthalmus maximus. Fish Shellfish Immunol. 2016, 58, 125–135. [Google Scholar] [CrossRef]
- Qiang, J.; Bao, W.J.; Tao, F.Y.; He, J.; Li, X.H.; Xu, P.; Sun, L.Y. The expression profiles of miRNA-mRNA of early response in genetically improved farmed tilapia (Oreochromis niloticus) liver by acute heat stress. Sci. Rep. 2017, 7, 1–15. [Google Scholar] [CrossRef]
- Webster, T.U.; Santos, E.M. Global transcriptomic profiling demonstrates induction of oxidative stress and of compensatory cellular stress responses in brown trout exposed to glyphosate and Roundup. BMC Genom. 2015, 16, 1–14. [Google Scholar] [CrossRef]
- Ruland, J. Return to homeostasis: Downregulation of NF-kB responses. Nat. Immunol. 2011, 12, 709–714. [Google Scholar] [CrossRef]
- Liu, Y.; Ma, D.; Zhao, C.; Xiao, Z.; Xu, S.; Xiao, Y.; Wang, Y.; Liu, Q.; Li, J. The expression pattern of hsp70 plays a critical role in thermal tolerance of marine demersal fish: Multilevel responses of Paralichthys olivaceus and its hybrids (P. olivaceus ♀ x P. dentatus ♂) to chronic and acute stress. Mar. Environ. Res. 2017, 129, 386–395. [Google Scholar] [CrossRef]
- Ineno, T.; Tsuchida, S.; Kanda, M.; Watabe, S. Thermal tolerance of a rainbow trout Oncorhyncus mykiss strain selected by high-temperature breeding. Fish Sci. 2005, 71, 767–775. [Google Scholar] [CrossRef]
- Tan, E.; Wongwarangkana, C.; Kinoshita, S.; Suzuki, Y.; Oshima, K.; Hattori, M.; Ineno, T.; Tamaki, K.; Kera, A.; Muto, K.; et al. Global gene expression analysis of gill tissues from normal and thermally selected strains of rainbow trout. Fish Sci. 2012, 78, 1041–1049. [Google Scholar] [CrossRef]
- Verbeke, P.; Fonager, J.; Clark, B.F.C.; Rattan, S.I.S. Heat shock response and aging: Mechanisms and applications. Cell Biol. Int. 2001, 25, 845–857. [Google Scholar] [CrossRef]
- Finkel, T.; Holbrook, N.J. Oxidants, oxidative stress and the biology of aging. Nature 2000, 408, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Buckley, B.A.; Gracey, A.Y.; Somero, G.N. The cellular response to heat stress in the goby Gillichthys mirabilis: A cDNA microarray and protein-level analysis. J. Exp. Biol. 2006, 209, 2660–2677. [Google Scholar] [CrossRef]
- Tan, E.; Kinoshita, S.; Suzuki, Y.; Ineno, T.; Tamaki, K.; Kera, A.; Muto, K.; Yada, T.; Kitamura, S.; Asakawa, S.; et al. Different gene expression profiles between normal and thermally selected strains of rainbow trout, Oncorhyncus mykiss, as revealed by comprehensive transcriptome analysis. Gene 2016, 576, 637–643. [Google Scholar] [CrossRef]
- Healy, T.M.; Tymchuk, W.E.; Osborne, E.J.; Schulte, P.M. Heat shock response of killifish (Fundulus heteroclitus): Candidate gene and heterologous microarray approaches. Physiol. Genom. 2010, 41, 171–184. [Google Scholar] [CrossRef]
- Liu, S.; Wang, X.; Sun, F.; Zhang, J.; Feng, J.; Liu, H.; Rajendran, K.V.; Sun, L.; Zhang, Y.; Jiang, Y.; et al. RNA-Seq reveals expression signatures of genes involved in oxygen transport, protein synthesis, folding, and degradation in response to heat stress in catfish. Physiol. Genom. 2013, 45, 462–476. [Google Scholar] [CrossRef]
- Rebl, A.; Verleih, M.; Köbis, J.M.; Kühn, C.; Wimmers, K.; Köllner, B.; Goldammer, T. Transcriptome profiling of gill tissue in regionally bred and globally farmed rainbow trout strains reveals different strategies for coping with thermal stress. Mar. Biotechnol. 2013, 15, 445–460. [Google Scholar] [CrossRef] [PubMed]
- Harris, S.L.; Levine, A.J. The p53 pathway: Positive and negative feedback loops. Oncogene 2005, 24, 2899–2908. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Zhang, Z.; Shen, Z.; Zhang, C.; Liu, H. Regional differences in thermal adaptation of a cold-water fish Rhynchocypris oxycephalus revealed by thermal tolerance and transcriptomic responses. Sci. Rep. 2018, 8, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Verleih, M.; Borchel, A.; Krasnov, A.; Rebl, A.; Korytář, T.; Kühn, C.; Goldammer, T. Impact of thermal stress on kidney-specific gene expression in farmed regional and imported rainbow trout. Mar. Biotechnol. 2015, 17, 576–592. [Google Scholar] [CrossRef]
- Ojima, N.; Mekuchi, M.; Ineno, T.; Tamaki, K.; Kera, A.; Kinoshita, S.; Asakawa, S.; Watabe, S. Differential expression of heat-shock proteins in F2 offspring from F1 hybrids produced between thermally selected and normal rainbow trout strains. Fish Sci. 2012, 78, 1051–1057. [Google Scholar] [CrossRef]
- Li, Y.; Huang, J.; Liu, Z.; Zhou, Y.; Xia, B.; Wang, Y.; Kang, Y.; Wang, J. Transcriptome analysis provides insights into hepatic responses to moderate heat stress in the rainbow trout. Gene 2017, 619, 1–9. [Google Scholar] [CrossRef]
- Ineno, T.; Tamaki, K.; Yamada, K.; Kodama, R.; Tsuchida, S.; Tan, E.; Kinoshita, S.; Muto, K.; Yada, T.; Kitamura, S.; et al. Thermal tolerance of a thermally selected strain of rainbow trout Oncorhynchus mykiss and the pedigrees of its F1 and F2 generations indicated by their critical thermal maxima. Fish Sci. 2018, 84, 671–679. [Google Scholar] [CrossRef]
- Huth, T.J.; Place, S.P. Transcriptome wide analyses reveal a sustained cellular stress response in the gill tissue of Trematomus bernacchii after acclimation to multiple stressors. BMC Genom. 2016, 17, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Koppang, E.O.; Fischer, U.; Moore, L.; Tranulis, M.A.; Dijkstra, J.M.; Köllner, B.; Aune, L.; Jirillo, E.; Hordvik, I. Salmonid T cells assemble in the thymus, spleen and in novel interbranchial lymphoid tissue. J. Anat. 2010, 217, 728–739. [Google Scholar] [CrossRef]
- Madeira, C.; Madeira, D.; Diniz, M.S.; Cabral, H.N.; Vinagre, C. Comparing biomarker responses during thermal acclimation: A lethal vs non-lethal approach in a tropical reef clownfish. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2017, 204, 104–112. [Google Scholar] [CrossRef] [PubMed]
- Haas, B.J.; Papanicolaou, A.; Yassour, M.; Grabherr, M.; Blood, P.D.; Bowden, J.; Couger, M.B.; Eccles, D.; Li, B.; Lieber, M.; et al. De novo transcript sequence reconstruction from RNA-seq using the Trinity platform for reference generation and analysis. Nat. Protoc. 2013, 8, 1494–1512. [Google Scholar] [CrossRef]
- Bray, N.L.; Pimentel, H.; Melsted, P.; Pachter, L. Near-optimal probabilistic RNA-seq quantification. Nat. Biotechnol. 2016, 34, 525–527. [Google Scholar] [CrossRef]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Xie, C.; Mao, X.; Huang, J.; Ding, Y.; Wu, J.; Dong, S.; Kong, L.; Gao, G.; Li, C.Y.; Wei, L. KOBAS 2.0: A web server for annotation and identification of enriched pathways and diseases. Nucleic Acids Res. 2011, 39, 316–322. [Google Scholar] [CrossRef] [PubMed]
- Kanehisa, M.; Furumichi, M.; Tanabe, M.; Sato, Y.; Morishima, K. KEGG: New perspectives on genomes, pathways, diseases and drugs. Nucleic Acids Res. 2017, 45, D353–D361. [Google Scholar] [CrossRef]
- Wickham, H. ggplots2: Elegant Graphics for Data Analysis, 2nd ed.; Springer: New York, NY, USA, 2016; ISBN 978-3-319-24275-0. [Google Scholar]
- Warnes, G.R.; Bolker, B.; Bonebakker, L.; Gentleman, R.; Liaw, W.H.A.; Lumley, T.; Maechler, M.; Magnusson, A.; Moeller, S.; Schwart, M.; et al. gplots: Various R Programming Tools for Plotting Data. Available online: https://cran.r-project.org/web/packages/gplots/index.html (accessed on 30 August 2018).
- Logan, C.A.; Somero, G.N. Effects of thermal acclimation on transcriptional responses to acute heat stress in the eurythermal fish Gillichthys mirabilis (Cooper). Am. J. Physiol. Regul. Integr. Comp. Physiol. 2011, 300, R1373–R1383. [Google Scholar] [CrossRef]
- Evans, D.H.; Piermarini, P.M.; Choe, K.P. The multifunctional fish gill: Dominant site of gas exchange, osmoregulation, acid-base regulation, and excretion of nitrogenous waste. Physiol. Rev. 2005, 85, 97–177. [Google Scholar] [CrossRef] [PubMed]
- Ikwegbue, P.C.; Masamba, P.; Oyinloye, B.E.; Kappo, A.P. Roles of heat shock proteins in apoptosis, oxidative stress, human inflammatory diseases, and cancer. Pharmaceuticals 2018, 11, 2. [Google Scholar] [CrossRef]
- Jubran, R.; Kocsis, J.; Garam, N.; Maláti, E.; Gombos, T.; Barabás, L.; Gráf, L.; Prohászka, Z.; Fishelson, Z. Circulating mitochondrial stress 70 protein/mortalin and cytosolic Hsp70 in blood: Risk indicators in colorectal cancer. Int. J. Cancer 2017, 141, 2329–2335. [Google Scholar] [CrossRef]
- Qiukai, K.; Liu, X.; Liu, Y.; Liu, W.; Zuo, J. Over-expression of GRP75 inhibits liver injury induced by oxidative damage. Acta Biochim. Biophys. Sin. 2013, 45, 129–134. [Google Scholar] [CrossRef]
- Kalmar, B.; Greensmith, L. Activation of the heat shock response in a primary cellular model of motoneuron neurodegeneration-evidence for neuroprotective and neurotoxic effects. Cell. Mol. Biol. Lett. 2009, 14, 319–335. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Shibazaki, A.; Ono, R.; Kaisho, T. HSP70 mediates degradation of the p65 subunit of nuclear factor κb to inhibit inflammatory signaling. Sci. Signal 2014, 7, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Tu, Z.; Tang, D.; Zhang, H.; Liu, M.; Wang, K.; Calderwood, S.K.; Xiao, X. The inhibition of LPS-induced production of inflammatory cytokines by HSP70 involves inactivation of the NF-κB pathway but not the MAPK pathways. Shock 2006, 26, 277–284. [Google Scholar] [CrossRef]
- Pei, Y.; Wu, Y.; Qin, Y. Effects of chronic heat stress on the expressions of heat shock proteins 60, 70, 90, A2, and HSC70 in the rabbit testis. Cell Stress Chaperones 2012, 17, 81–87. [Google Scholar] [CrossRef]
- Harms, K.; Nozell, S.; Chen, X. The common and distinct target genes of the p53 family transcription factors. Cell. Mol. Life Sci. 2004, 61, 822–842. [Google Scholar] [CrossRef]
- Sonna, L.A.; Fujita, J.; Gaffin, S.L.; Lilly, C.M. Invited review: Effects of heat and cold stress on mammalian gene expression. J. Appl. Physiol. 2002, 92, 1725–1742. [Google Scholar] [CrossRef] [PubMed]
- Forgati, M.; Kasdalski, P.K.; Herrerias, T.; Zaleski, T.; Machado, C.; Souza, M.; Donatti, L. Effect of heat stress on the renal and branchial carbohydrate metabolism and antioxidant system of Antarctic fish. J. Comp. Physiol. B 2017, 187, 1137–1154. [Google Scholar] [CrossRef]
- Aedo, J.E.; Maldonado, J.; Aballai, V.; Estrada, J.M.; Molina, M.B.; Meneses, C.; Escarate, C.G.; Silva, H.; Molina, A.; Valdés, J.A. mRNA-seq reveals skeleton muscle atrophy in response to handling stress in a marine teleost, the red cusk-eel (Genypterus chilensis). BMC Genom. 2015, 16, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Dokladny, K.; Ye, D.; Kennedy, J.C.; Moseley, P.L.; Ma, T.Y. Cellular and molecular mechanisms of heat stress-induced up-regulation of occluding protein expression. Am. J. Pathol. 2008, 172, 659–670. [Google Scholar] [CrossRef]
- Bouchama, A.; Aziz, M.A.; Mahri, S.A.; Gabere, M.N.; Dlamy, M.A.; Mohamand, S.; Abbad, M.A.; Hussein, M. A model of exposure to extreme environmental heat uncovers the human transcriptome to heat stress. Sci. Rep. 2017, 7, 1–14. [Google Scholar] [CrossRef]
- Komarova, E.A.; Diatchenko, L.; Rokhlin, O.W.; Hill, J.E.; Wang, Z.J.; Krivokrysenko, V.I.; Feinstein, E.; Gudkov, A.V. Stress-induced secretion of growth inhibitors: A novel tumor suppressor function of p53. Oncogen 1998, 17, 1089–1096. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Liang, C.; Hua, Y.; Miao, C.; Zhang, J.; Xu, A.; Zhao, K.; Liu, S.; Tian, Y.; Dong, H.; et al. The metastasis suppressor CD82/KAI1 regulates cell migration and invasion via inhibiting TGF-β1/Smad signaling in renal cell carcinoma. Oncotarget 2017, 8, 51559–51568. [Google Scholar] [CrossRef] [PubMed]
- Sridhar, S.C.; Miranti, C.K. Tetraspanin KAI1/CD82 suppresses invasion by inhibiting integrin-dependent crosstalk with c-Met receptor and Src kinases. Oncogene 2006, 25, 2367–2378. [Google Scholar] [CrossRef] [PubMed]
- Bourdon, J.C.; Renzing, J.; Robertson, P.L.; Fernandes, K.N.; Lane, D.P. Scotin, a novel p53-inducible proapoptotic protein located in the ER and the nuclear membrane. J. Cell Biol. 2002, 158, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Han, H.S.; Kang, G.; Kim, J.S.; Choi, B.H.; Koo, S.H. Regulation of glucose metabolism from a liver-centric perspective. Exp. Mol. Med. 2016, 48, e218. [Google Scholar] [CrossRef]
- Li, X.B.; Gu, J.D.; Zhou, Q.H. Review of aerobic glycolysis and its key enzymes-new targets for lung cancer therapy. Thorac. Cancer 2015, 6, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Lei, Q.Y.; Zhao, S.; Guan, K.L. Regulation of glycolysis and gluconeogenesis by acetylation of PKM and PEPCK. Cold Spring Harb. Symp. Quant. Biol. 2011, 76, 285–289. [Google Scholar] [CrossRef] [PubMed]
| Tissue Samples | Gill | Adipose Fin | ||
|---|---|---|---|---|
| HT | NT | HT | NT | |
| Clean readings | 61,460,394 | 78,088,572 | 71,282,330 | 59,749,778 |
| Clean bases (Gb) | 6.15 | 7.81 | 7.13 | 5.97 |
| Q20 (%) | 97.88 | 97.73 | 97.80 | 97.80 |
| GC content (%) | 48.39 | 48.06 | 48.92 | 48.84 |
| Up-Regulated Genes in Gill | Down-Regulated Genes in Gill | ||
|---|---|---|---|
| Gene Name | log2FC | Gene Name | log2FC |
| Beta-taxilin | 7.82 | Macrophage-expressed gene 1 protein | −25.52 |
| Translocation protein SEC62 | 7.60 | Unknown | −24.65 |
| Tax1-binding protein 1 homolog B | 7.57 | M-protein, striated muscle | −24.06 |
| Protein OS-9 | 7.55 | Unknown | −23.70 |
| Vacuolar protein sorting-associated protein 26A | 7.43 | Transmembrane protein 54 | −23.24 |
| DnaJ homolog subfamily A member 2 | 7.32 | LINE-1 retrotransposable element ORF2 protein | −23.16 |
| CD276 antigen | 7.27 | Nascent polypeptide-associated complex subunit alpha | −22.75 |
| Interferon-induced very large GTPase 1 | 7.26 | Carboxy-Terminal domain RNA polymerase II polypeptide A small phosphatase 2 | −22.54 |
| ADP-ribosylation factor 4 | 7.21 | E3 ubiquitin-protein ligase TRIM39 | −22.49 |
| Protein Mdm4 | 7.15 | C-C motif chemokine 4 | −22.36 |
| Pyruvate dehydrogenase protein X component | 7.09 | Major histocompatibility complex class I-related gene protein | −22.29 |
| Transforming acidic coiled-coil-containing protein 3 | 7.05 | LINE-1 retrotransposable element ORF2 protein | −22.20 |
| Eukaryotic translation initiation factor 3 subunit B | 6.95 | LINE-1 retrotransposable element ORF2 protein | −22.20 |
| Unknown | 6.94 | Complement C1q subcomponent subunit A | −20.66 |
| Insulin-like growth factor 2 mRNA-binding protein 3 | 6.91 | Unknown | −9.90 |
| E3 ubiquitin-protein ligase AMFR | 6.88 | H-2 class I histocompatibility antigen, K-K alpha chain | −9.48 |
| Ribosome-binding protein 1 | 6.84 | Interferon-induced very large GTPase 1 | −9.31 |
| FYVE, RhoGEF and PH domain-containing protein 4 | 6.81 | Ras-related protein Rab-25 | −9.29 |
| Cytochrome b-c1 complex subunit 1 | 6.78 | H-2 class II histocompatibility antigen gamma chain | −9.09 |
| Unknown | 6.75 | Cytochrome c1, heme protein, mitochondrial | −8.98 |
| Up-Regulated Genes in Adipose Fin | Down-Regulated Genes in Adipose Fin | ||
|---|---|---|---|
| Gene Name | log2FC | Gene Name | log2FC |
| Aquaporin-3 | 23.19 | Keratin, type I cytoskeletal 18 | −23.47 |
| Tumor protein p73 | 22.72 | Unknown | −22.65 |
| Nucleolin | 8.54 | Mitogen-activated protein kinase 3 | −22.24 |
| ICOS ligand | 8.00 | Unknown | −22.16 |
| Proliferation marker protein Ki-67 | 7.73 | Nascent polypeptide-associated complex subunit alpha | −12.00 |
| Unknown | 7.59 | Unknown | −10.42 |
| Unknown | 7.47 | Unknown | −10.06 |
| E3 ubiquitin-protein ligase RNF13 | 7.46 | Major histocompatibility complex class I-related gene protein | −9.97 |
| Testin | 7.39 | Nascent polypeptide-associated complex subunit alpha | −9.87 |
| ADP-ribosylation factor 4 | 7.37 | Src substrate protein p85 | −9.65 |
| Olfactomedin-like protein 2B | 7.35 | Macrosialin | −9.06 |
| Septin-2 | 7.16 | A-kinase anchor protein 12 | −8.93 |
| Mitogen-activated protein kinase kinase kinase 5 | 7.10 | EMILIN-2 | −8.86 |
| Aldehyde dehydrogenase family 9 member A1-A | 7.08 | Ras-related protein Rab-25 | −8.82 |
| Unknown | 7.07 | Myocilin | −8.77 |
| Adaptin ear-binding coat-associated protein 2 | 7.04 | Annexin A4 | −8.63 |
| Unknown | 6.95 | Annexin A2-A | −8.58 |
| General transcription factor II-I repeat domain-containing protein 2 | 6.80 | Nesprin-2 | −8.44 |
| Unknown | 6.79 | Unknown | −8.37 |
| Aldehyde dehydrogenase family 9 member A1-A | 6.77 | Cytochrome c1, heme protein, mitochondrial | −8.36 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kraitavin, W.; Yoshitake, K.; Igarashi, Y.; Mitsuyama, S.; Kinoshita, S.; Kambayashi, D.; Watabe, S.; Asakawa, S. Transcriptome Analysis of Yamame (Oncorhynchus masou) in Normal Conditions after Heat Stress. Biology 2019, 8, 21. https://doi.org/10.3390/biology8020021
Kraitavin W, Yoshitake K, Igarashi Y, Mitsuyama S, Kinoshita S, Kambayashi D, Watabe S, Asakawa S. Transcriptome Analysis of Yamame (Oncorhynchus masou) in Normal Conditions after Heat Stress. Biology. 2019; 8(2):21. https://doi.org/10.3390/biology8020021
Chicago/Turabian StyleKraitavin, Waraporn, Kazutoshi Yoshitake, Yoji Igarashi, Susumu Mitsuyama, Shigeharu Kinoshita, Daisuke Kambayashi, Shugo Watabe, and Shuichi Asakawa. 2019. "Transcriptome Analysis of Yamame (Oncorhynchus masou) in Normal Conditions after Heat Stress" Biology 8, no. 2: 21. https://doi.org/10.3390/biology8020021
APA StyleKraitavin, W., Yoshitake, K., Igarashi, Y., Mitsuyama, S., Kinoshita, S., Kambayashi, D., Watabe, S., & Asakawa, S. (2019). Transcriptome Analysis of Yamame (Oncorhynchus masou) in Normal Conditions after Heat Stress. Biology, 8(2), 21. https://doi.org/10.3390/biology8020021





