Telling the Time with a Broken Clock: Quantifying Circadian Disruption in Animal Models
Abstract
1. Circadian Rhythms
2. Measuring Circadian Rhythms
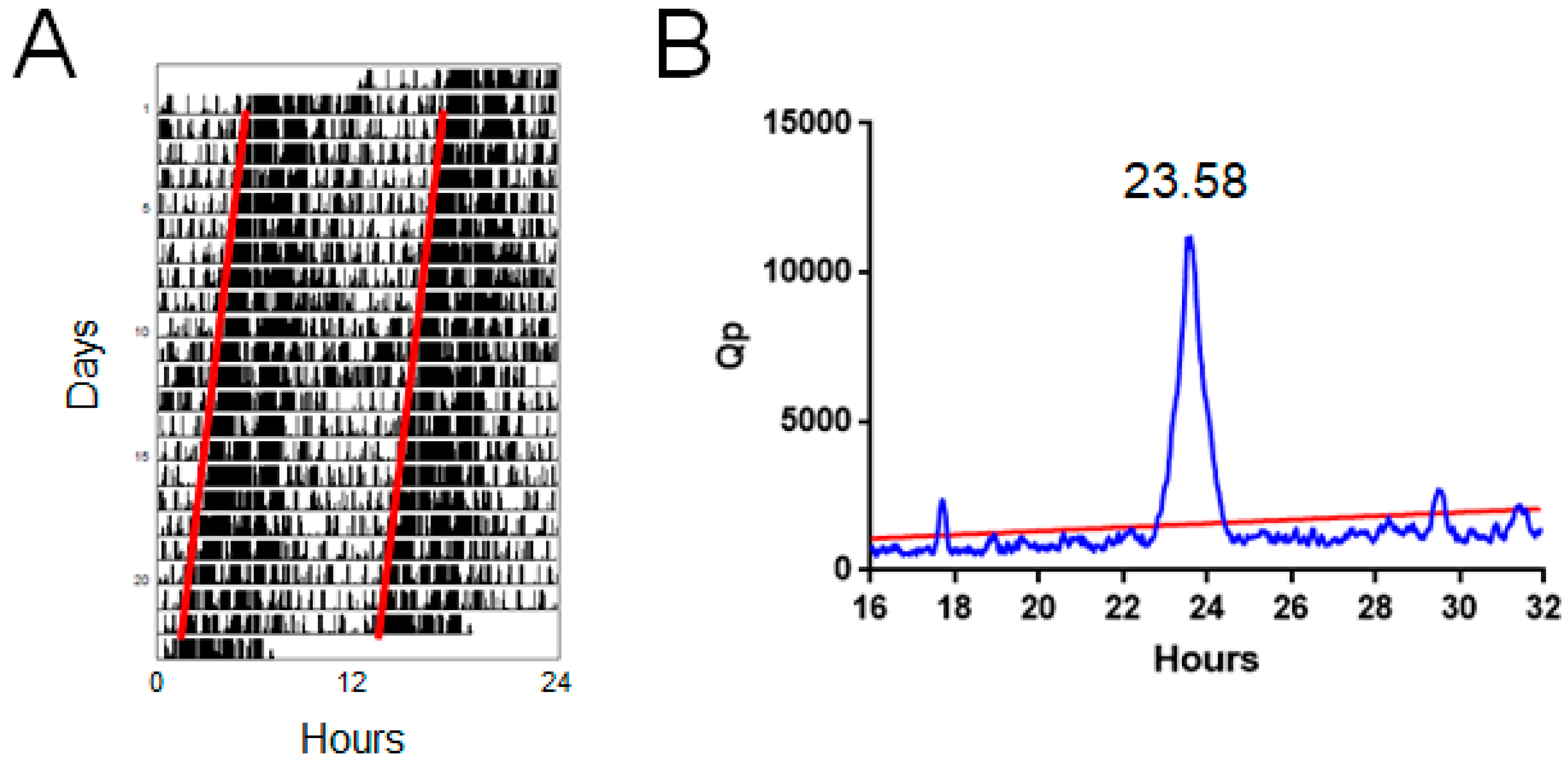
2.1. Enright Periodogram
2.2. Fourier Analysis
2.3. Lomb-Scargle Periodogram
2.4. Activity Onset
3. Circadian Disruption
Forms of Circadian Disruption
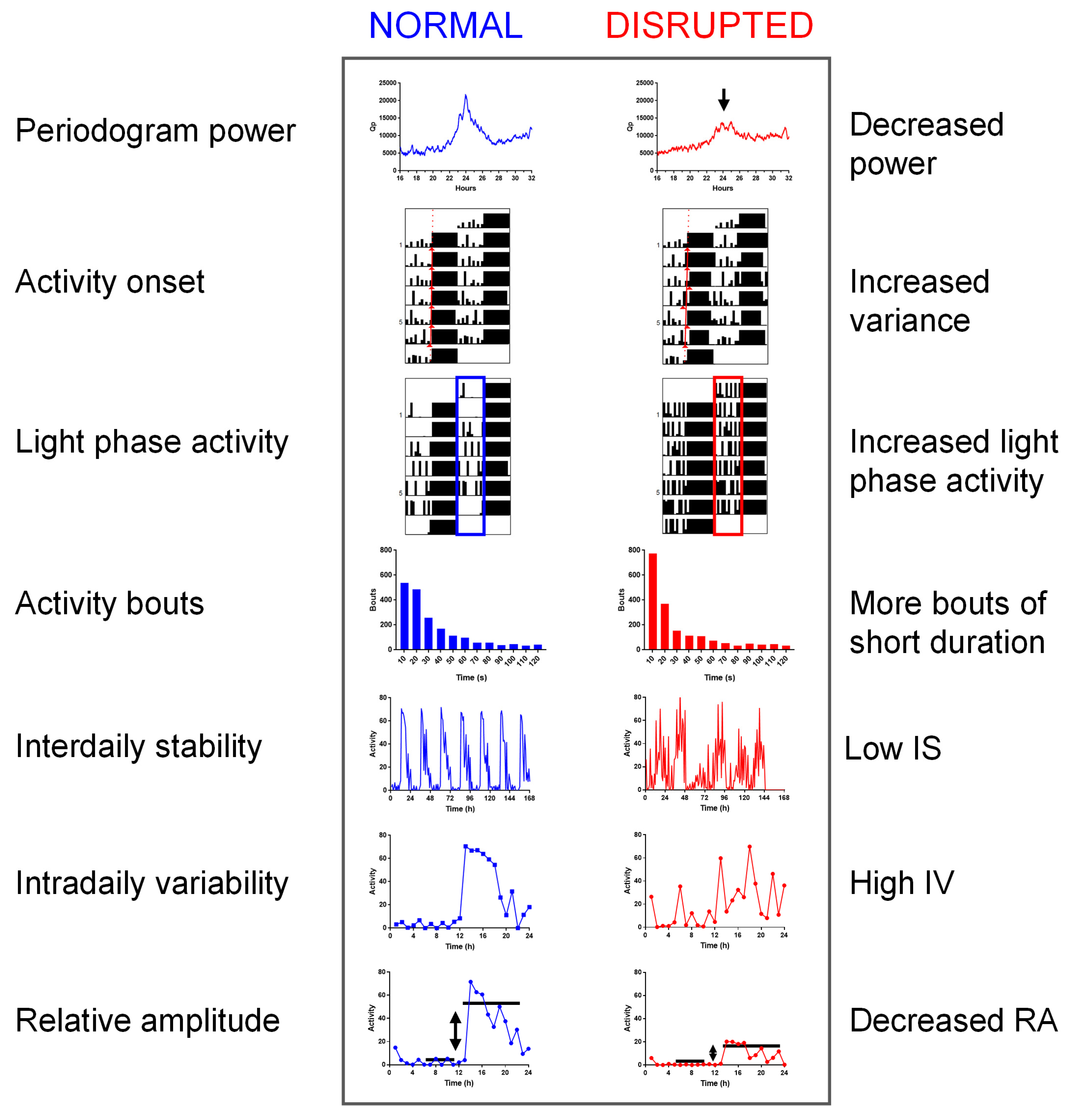
4. Measuring Circadian Disruption
4.1. Visual Inspection of Actograms
4.1.1. Measurement
4.1.2. Strengths and Limitations
4.2. Periodogram Power
4.2.1. Measurement
4.2.2. Strengths and Limitations
4.3. Activity Onset
4.3.1. Measurement
4.3.2. Strengths and Limitations
4.4. Light Phase Activity
4.4.1. Measurement
4.4.2. Strengths and Limitations
4.5. Activity Bouts
4.5.1. Measurement
4.5.2. Strengths and Limitations
4.6. Inter-Daily Stability
4.6.1. Measurement
4.6.2. Strengths and Limitations
4.7. Intra-Daily Variability
4.7.1. Measurement
4.7.2. Strengths and Limitations
4.8. Relative Amplitude
4.8.1. Measurement
4.8.2. Strengths and Limitations
5. Additional Considerations and Alternative Approaches
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Reppert, S.M.; Weaver, D.R. Coordination of circadian timing in mammals. Nature 2002, 418, 935–941. [Google Scholar] [CrossRef] [PubMed]
- Dibner, C.; Schibler, U.; Albrecht, U. The mammalian circadian timing system: Organization and coordination of central and peripheral clocks. Annu. Rev. Physiol. 2010, 72, 517–549. [Google Scholar] [CrossRef] [PubMed]
- Hughes, S.; Jagannath, A.; Rodgers, J.; Hankins, M.W.; Peirson, S.N.; Foster, R.G. Signalling by melanopsin (OPN4) expressing photosensitive retinal ganglion cells. Eye 2016, 30, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Lucas, R.J.; Peirson, S.N.; Berson, D.M.; Brown, T.M.; Cooper, H.M.; Czeisler, C.A.; Figueiro, M.G.; Gamlin, P.D.; Lockley, S.W.; O’Hagan, J.B.; et al. Measuring and using light in the melanopsin age. Trends Neurosci. 2014, 37, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Palumaa, T.; Gilhooley, M.J.; Jagannath, A.; Hankins, M.W.; Hughes, S.; Peirson, S.N. Melanopsin: Photoreceptors, physiology and potential. Curr. Opin. Physiol. 2018, 5, 68–74. [Google Scholar] [CrossRef]
- Hughes, S.; Jagannath, A.; Hankins, M.W.; Foster, R.G.; Peirson, S.N. Photic regulation of clock systems. Methods Enzymol. 2015, 552, 125–143. [Google Scholar]
- Albrecht, U.; Foster, R.G. Placing ocular mutants into a functional context: A chronobiological approach. Methods 2002, 28, 465–477. [Google Scholar] [CrossRef]
- Eckel-Mahan, K.; Sassone-Corsi, P. Phenotyping Circadian Rhythms in Mice. Curr. Protocols Mouse Biol. 2015, 5, 271–281. [Google Scholar] [CrossRef]
- Jud, C.; Schmutz, I.; Hampp, G.; Oster, H.; Albrecht, U. A guideline for analyzing circadian wheel-running behavior in rodents under different lighting conditions. Biol. Proced. Online 2005, 7, 101–116. [Google Scholar] [CrossRef]
- Siepka, S.M.; Takahashi, J.S. Methods to record circadian rhythm wheel running activity in mice. Methods Enzymol. 2005, 393, 230–239. [Google Scholar]
- Peirson, S.N.; Thompson, S.; Hankins, M.W.; Foster, R.G. Mammalian photoentrainment: Results, methods, and approaches. Methods Enzymol. 2005, 393, 697–726. [Google Scholar]
- De la Iglesia, H.O.; Cambras, T.; Schwartz, W.J.; Diez-Noguera, A. Forced desynchronization of dual circadian oscillators within the rat suprachiasmatic nucleus. Curr. Biol. 2004, 14, 796–800. [Google Scholar] [CrossRef]
- Daan, S.; Pittendrigh, C.S. A functional analysis of circadian pacemakers in nocturnal rodents. III Heavy water and constant light: Homeostasis of frequency? J. Comp. Physiol. 1976, 106, 267–290. [Google Scholar] [CrossRef]
- Refinetti, R. Laboratory instrumentation and computing: Comparison of six methods for the determination of the period of circadian rhythms. Physiol. Behav. 1993, 54, 869–875. [Google Scholar] [CrossRef]
- Zielinski, T.; Moore, A.M.; Troup, E.; Halliday, K.J.; Millar, A.J. Strengths and limitations of period estimation methods for circadian data. PLoS ONE 2014, 9, e96462. [Google Scholar] [CrossRef]
- Brown, L.A.; Hasan, S.; Foster, R.G.; Peirson, S.N. COMPASS: Continuous Open Mouse Phenotyping of Activity and Sleep Status. Wellcome Open Res. 2016, 1, 2. [Google Scholar] [CrossRef]
- Enright, J.T. The search for rhythmicity in biological time-series. J. Theor. Biol. 1965, 8, 426–468. [Google Scholar] [CrossRef]
- Sokolove, P.G.; Bushell, W.N. The chi square periodogram: Its utility for analysis of circadian rhythms. J. Theor. Biol. 1978, 72, 131–160. [Google Scholar] [CrossRef]
- Levine, J.D.; Funes, P.; Dowse, H.B.; Hall, J.C. Signal analysis of behavioral and molecular cycles. BMC Neurosci. 2002, 3, 1. [Google Scholar]
- Costa, M.J.; Finkenstadt, B.; Roche, V.; Levi, F.; Gould, P.D.; Foreman, J.; Halliday, K.; Hall, A.; Rand, D.A. Inference on periodicity of circadian time series. Biostatistics 2013, 14, 792–806. [Google Scholar] [CrossRef]
- Herzog, E.D.; Kiss, I.Z.; Mazuski, C. Measuring synchrony in the mammalian central circadian circuit. Methods Enzymol. 2015, 552, 3–22. [Google Scholar] [PubMed]
- Lomb, N.R. Least-squares frequency analysis of unequally spaced data. Astrophys. Space Sci. 1976, 39, 447–462. [Google Scholar] [CrossRef]
- Ruf, T. The Lomb-Scargle periodogram in biological rhythm research: Analysis of incomplete and unequally spaced time-series. Biol. Rhythm Res. 1999, 30, 178–201. [Google Scholar] [CrossRef]
- Van Dongen, H.P.; Olofsen, E.; VanHartevelt, J.H.; Kruyt, E.W. A procedure of multiple period searching in unequally spaced time-series with the Lomb-Scargle method. Biol. Rhythm Res. 1999, 30, 149–177. [Google Scholar] [CrossRef] [PubMed]
- Brainard, G.C.; Hanifin, J.P.; Greeson, J.M.; Byrne, B.; Glickman, G.; Gerner, E.; Rollag, M.D. Action spectrum for melatonin regulation in humans: Evidence for a novel circadian photoreceptor. J. Neurosci. 2001, 21, 6405–6412. [Google Scholar] [CrossRef] [PubMed]
- Hattar, S.; Lucas, R.J.; Mrosovsky, N.; Thompson, S.; Douglas, R.H.; Hankins, M.W.; Lem, J.; Biel, M.; Hofmann, F.; Foster, R.G.; et al. Melanopsin and rod-cone photoreceptive systems account for all major accessory visual functions in mice. Nature 2003, 424, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Lucas, R.J.; Douglas, R.H.; Foster, R.G. Characterization of an ocular photopigment capable of driving pupillary constriction in mice. Nat. Neurosci. 2001, 4, 621–626. [Google Scholar] [CrossRef]
- Thapan, K.; Arendt, J.; Skene, D.J. An action spectrum for melatonin suppression: Evidence for a novel non-rod, non-cone photoreceptor system in humans. J. Physiol. 2001, 535, 261–267. [Google Scholar] [CrossRef]
- Bechtold, D.A.; Gibbs, J.E.; Loudon, A.S. Circadian dysfunction in disease. Trends Pharmacol. Sci. 2010, 31, 191–198. [Google Scholar] [CrossRef]
- Hastings, M.H.; Reddy, A.B.; Maywood, E.S. A clockwork web: Circadian timing in brain and periphery, in health and disease. Nat. Rev. Neurosci. 2003, 4, 649–661. [Google Scholar] [CrossRef]
- Jagannath, A.; Peirson, S.N.; Foster, R.G. Sleep and circadian rhythm disruption in neuropsychiatric illness. Curr. Opin. Neurobiol. 2013, 23, 888–894. [Google Scholar] [CrossRef]
- West, A.C.; Bechtold, D.A. The cost of circadian desynchrony: Evidence, insights and open questions. BioEssays 2015, 37, 777–788. [Google Scholar] [CrossRef]
- Wulff, K.; Gatti, S.; Wettstein, J.G.; Foster, R.G. Sleep and circadian rhythm disruption in psychiatric and neurodegenerative disease. Nature reviews. Neuroscience 2010, 11, 589–599. [Google Scholar]
- Smolensky, M.H.; Hermida, R.C.; Reinberg, A.; Sackett-Lundeen, L.; Portaluppi, F. Circadian disruption: New clinical perspective of disease pathology and basis for chronotherapeutic intervention. Chronobiol. Int. 2016, 33, 1101–1119. [Google Scholar] [CrossRef]
- Banks, G.; Nolan, P.M.; Peirson, S.N. Reciprocal interactions between circadian clocks and aging. Mammalian Genome 2016, 27, 332–340. [Google Scholar] [CrossRef]
- Barnard, A.R.; Nolan, P.M. When clocks go bad: Neurobehavioural consequences of disrupted circadian timing. PLoS Genet. 2008, 4, e1000040. [Google Scholar] [CrossRef]
- Bechtold, D.A.; Loudon, A.S. Hypothalamic clocks and rhythms in feeding behaviour. Trends Neurosci. 2013, 36, 74–82. [Google Scholar] [CrossRef]
- Ruger, M.; Scheer, F.A. Effects of circadian disruption on the cardiometabolic system. Rev. Endocr. Metab. Disord. 2009, 10, 245–260. [Google Scholar] [CrossRef]
- Lamia, K.A. Ticking time bombs: Connections between circadian clocks and cancer. F1000Research 2017, 6, 1910. [Google Scholar] [CrossRef]
- Pritchett, D.; Wulff, K.; Oliver, P.L.; Bannerman, D.M.; Davies, K.E.; Harrison, P.J.; Peirson, S.N.; Foster, R.G. Evaluating the links between schizophrenia and sleep and circadian rhythm disruption. J. Neural Transm. 2012, 119, 1061–1075. [Google Scholar] [CrossRef]
- Schroeder, A.M.; Colwell, C.S. How to fix a broken clock. Trends Pharmacol. Sci. 2013, 34, 605–619. [Google Scholar] [CrossRef]
- Vetter, C. Circadian disruption: What do we actually mean? Eur. J. Neurosci. 2018. [Google Scholar] [CrossRef]
- Erren, T.C.; Reiter, R.J. Defining chronodisruption. J. Pineal Res. 2009, 46, 245–247. [Google Scholar] [CrossRef]
- Wittmann, M.; Dinich, J.; Merrow, M.; Roenneberg, T. Social jetlag: Misalignment of biological and social time. Chronobiol. Int. 2006, 23, 497–509. [Google Scholar] [CrossRef]
- West, A.C.; Smith, L.; Ray, D.W.; Loudon, A.S.I.; Brown, T.M.; Bechtold, D.A. Misalignment with the external light environment drives metabolic and cardiac dysfunction. Nat. Commun. 2017, 8, 417. [Google Scholar] [CrossRef]
- Cambras, T.; Weller, J.R.; Angles-Pujoras, M.; Lee, M.L.; Christopher, A.; Diez-Noguera, A.; Krueger, J.M.; de la Iglesia, H.O. Circadian desynchronization of core body temperature and sleep stages in the rat. Proc. Natl. Acad. Sci. USA 2007, 104, 7634–7639. [Google Scholar] [CrossRef]
- Oliver, P.L.; Sobczyk, M.V.; Maywood, E.S.; Edwards, B.; Lee, S.; Livieratos, A.; Oster, H.; Butler, R.; Godinho, S.I.; Wulff, K.; et al. Disrupted circadian rhythms in a mouse model of schizophrenia. Curr. Biol. 2012, 22, 314–319. [Google Scholar] [CrossRef]
- Pritchett, D.; Jagannath, A.; Brown, L.A.; Tam, S.K.; Hasan, S.; Gatti, S.; Harrison, P.J.; Bannerman, D.M.; Foster, R.G.; Peirson, S.N. Deletion of Metabotropic Glutamate Receptors 2 and 3 (mGlu2 & mGlu3) in Mice Disrupts Sleep and Wheel-Running Activity, and Increases the Sensitivity of the Circadian System to Light. PLoS ONE 2015, 10, e0125523. [Google Scholar]
- Eastman, C.; Rechtschaffen, A. Circadian temperature and wake rhythms of rats exposed to prolonged continuous illumination. Physiol. Behav. 1983, 31, 417–427. [Google Scholar] [CrossRef]
- Refinetti, R. Non-stationary time series and the robustness of circadian rhythms. J. Theor. Biol. 2004, 227, 571–581. [Google Scholar] [CrossRef]
- Schmid, B.; Helfrich-Forster, C.; Yoshii, T. A new ImageJ plug-in "ActogramJ" for chronobiological analyses. J. Biol. Rhythms 2011, 26, 464–467. [Google Scholar] [CrossRef] [PubMed]
- Pittendrigh, C.S.; Daan, S. A functional analysis of circadian pacemakers in nocturnal rodents, I: The stability and lability of spontaneous frequency. J. Comp. Physiol. A 1976, 106, 223–252. [Google Scholar] [CrossRef]
- Altimus, C.M.; Guler, A.D.; Alam, N.M.; Arman, A.C.; Prusky, G.T.; Sampath, A.P.; Hattar, S. Rod photoreceptors drive circadian photoentrainment across a wide range of light intensities. Nat. Neurosci. 2010, 13, 1107–1112. [Google Scholar] [CrossRef] [PubMed]
- Doyle, S.E.; Castrucci, A.M.; McCall, M.; Provencio, I.; Menaker, M. Nonvisual light responses in the Rpe65 knockout mouse: Rod loss restores sensitivity to the melanopsin system. Proc. Natl. Acad. Sci. USA 2006, 103, 10432–10437. [Google Scholar] [CrossRef] [PubMed]
- Butler, M.P.; Silver, R. Divergent photic thresholds in the non-image-forming visual system: Entrainment, masking and pupillary light reflex. Proc. Biol. Sci. 2011, 278, 745–750. [Google Scholar] [CrossRef] [PubMed]
- Peirson, S.N.; Brown, L.A.; Pothecary, C.A.; Benson, L.A.; Fisk, A.S. Light and the laboratory mouse. J. Neurosci. Methods 2018, 300, 26–36. [Google Scholar] [CrossRef]
- Porter, A.J.; Pillidge, K.; Tsai, Y.C.; Dudley, J.A.; Hunt, S.P.; Peirson, S.N.; Brown, L.A.; Stanford, S.C. A lack of functional NK1 receptors explains most, but not all, abnormal behaviours of NK1R-/- mice(1). Genes Brain Behav. 2015, 14, 189–199. [Google Scholar] [CrossRef] [PubMed]
- Pfeffer, M.; Zimmermann, Z.; Gispert, S.; Auburger, G.; Korf, H.W.; von Gall, C. Impaired Photic Entrainment of Spontaneous Locomotor Activity in Mice Overexpressing Human Mutant alpha-Synuclein. Int. J. Mol. Sci. 2018, 19, 1651. [Google Scholar] [CrossRef]
- Loh, D.H.; Jami, S.A.; Flores, R.E.; Truong, D.; Ghiani, C.A.; O’Dell, T.J.; Colwell, C.S. Misaligned feeding impairs memories. eLife 2015, 4, e09460. [Google Scholar] [CrossRef] [PubMed]
- Davies, B.; Brown, L.A.; Cais, O.; Watson, J.; Clayton, A.J.; Chang, V.T.; Biggs, D.; Preece, C.; Hernandez-Pliego, P.; Krohn, J.; et al. A point mutation in the ion conduction pore of AMPA receptor GRIA3 causes dramatically perturbed sleep patterns as well as intellectual disability. Hum. Mol. Genet. 2017, 26, 3869–3882. [Google Scholar] [CrossRef]
- Witting, W.; Kwa, I.H.; Eikelenboom, P.; Mirmiran, M.; Swaab, D.F. Alterations in the circadian rest-activity rhythm in aging and Alzheimer’s disease. Biol. Psychiatry 1990, 27, 563–572. [Google Scholar] [CrossRef]
- Van Someren, E.J.; Hagebeuk, E.E.; Lijzenga, C.; Scheltens, P.; de Rooij, S.E.; Jonker, C.; Pot, A.M.; Mirmiran, M.; Swaab, D.F. Circadian rest-activity rhythm disturbances in Alzheimer’s disease. Biol. Psychiatry 1996, 40, 259–270. [Google Scholar] [CrossRef]
- Van Someren, E.J.; Swaab, D.F.; Colenda, C.C.; Cohen, W.; McCall, W.V.; Rosenquist, P.B. Bright light therapy: Improved sensitivity to its effects on rest-activity rhythms in Alzheimer patients by application of nonparametric methods. Chronobiol. Int. 1999, 16, 505–518. [Google Scholar] [CrossRef] [PubMed]
- Banks, G.; Heise, I.; Starbuck, B.; Osborne, T.; Wisby, L.; Potter, P.; Jackson, I.J.; Foster, R.G.; Peirson, S.N.; Nolan, P.M. Genetic background influences age-related decline in visual and nonvisual retinal responses, circadian rhythms, and sleep. Neurobiol. Aging 2015, 36, 380–393. [Google Scholar] [CrossRef]
- Leise, T.L. Analysis of Nonstationary Time Series for Biological Rhythms Research. J. Biol. Rhythms 2017, 32, 187–194. [Google Scholar] [CrossRef]
- Leise, T.L.; Harrington, M.E. Wavelet-based time series analysis of circadian rhythms. J. Biol. Rhythms 2011, 26, 454–463. [Google Scholar] [CrossRef]
- Leise, T.L.; Indic, P.; Paul, M.J.; Schwartz, W.J. Wavelet meets actogram. J. Biol. Rhythms 2013, 28, 62–68. [Google Scholar] [CrossRef]
- Ortiz-Tudela, E.; Martinez-Nicolas, A.; Campos, M.; Rol, M.A.; Madrid, J.A. A new integrated variable based on thermometry, actimetry and body position (TAP) to evaluate circadian system status in humans. PLoS Comput. Biol. 2010, 6, e1000996. [Google Scholar] [CrossRef]
| Parameter | Mean ± SD | Range | Notes |
|---|---|---|---|
| Periodogram power (QP) 1 | 22,982 ± 5686 | 13,859–35,821 | Chi-square periodogram |
| Activity onset 2 | 3.33 ± 2.22 | 0.00–6.41 | Based on bouts > mean |
| Light phase activity (Light%) | 14% ± 4% | 5–22% | |
| Activity bouts (bouts/day) | 278 ± 63 | 162–420 | Based on sensitive PIR |
| Interdaily stability (IS) | 0.73 ± 0.09 | 0.47–0.88 | |
| Intradaily variability (IV) | 1.18 ± 0.32 | 0.67–1.90 | |
| Relative amplitude (RA) | 0.80 ± 0.07 | 0.66–0.94 | Based on M10 and L5 |
| QP | Onset | Light% | Bouts | IS | IV | RA | |
|---|---|---|---|---|---|---|---|
| QP | 1.00 | −0.46 | −0.69 | −0.45 | 0.81 | −0.81 | 0.74 |
| Onset | 1.00 | 0.60 | 0.33 | −0.31 | 0.31 | −0.47 | |
| Light% | 1.00 | 0.20 | −0.70 | 0.59 | −0.90 | ||
| Bouts | 1.00 | −0.20 | 0.46 | −0.28 | |||
| IS | 1.00 | −0.79 | 0.73 | ||||
| IV | 1.00 | −0.72 | |||||
| RA | 1.00 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brown, L.A.; Fisk, A.S.; Pothecary, C.A.; Peirson, S.N. Telling the Time with a Broken Clock: Quantifying Circadian Disruption in Animal Models. Biology 2019, 8, 18. https://doi.org/10.3390/biology8010018
Brown LA, Fisk AS, Pothecary CA, Peirson SN. Telling the Time with a Broken Clock: Quantifying Circadian Disruption in Animal Models. Biology. 2019; 8(1):18. https://doi.org/10.3390/biology8010018
Chicago/Turabian StyleBrown, Laurence A., Angus S. Fisk, Carina A. Pothecary, and Stuart N. Peirson. 2019. "Telling the Time with a Broken Clock: Quantifying Circadian Disruption in Animal Models" Biology 8, no. 1: 18. https://doi.org/10.3390/biology8010018
APA StyleBrown, L. A., Fisk, A. S., Pothecary, C. A., & Peirson, S. N. (2019). Telling the Time with a Broken Clock: Quantifying Circadian Disruption in Animal Models. Biology, 8(1), 18. https://doi.org/10.3390/biology8010018






