Sensitivity of Yeast Mutants Deficient in Mitochondrial or Vacuolar ABC Transporters to Pathogenesis-Related Protein TcPR-10 of Theobroma cacao
Abstract
1. Introduction
2. Materials and Methods
2.1. Obtaining the TcPR-10 Protein
2.2. Growth Conditions of Yeast S. cerevisiae
2.3. Yeast Survival after TcPR-10p Exposure
2.4. Fluorescence Assay of TcPR-10p Yeast Mutants
2.5. Flow Cytometry
2.6. Analysis of Results
3. Results
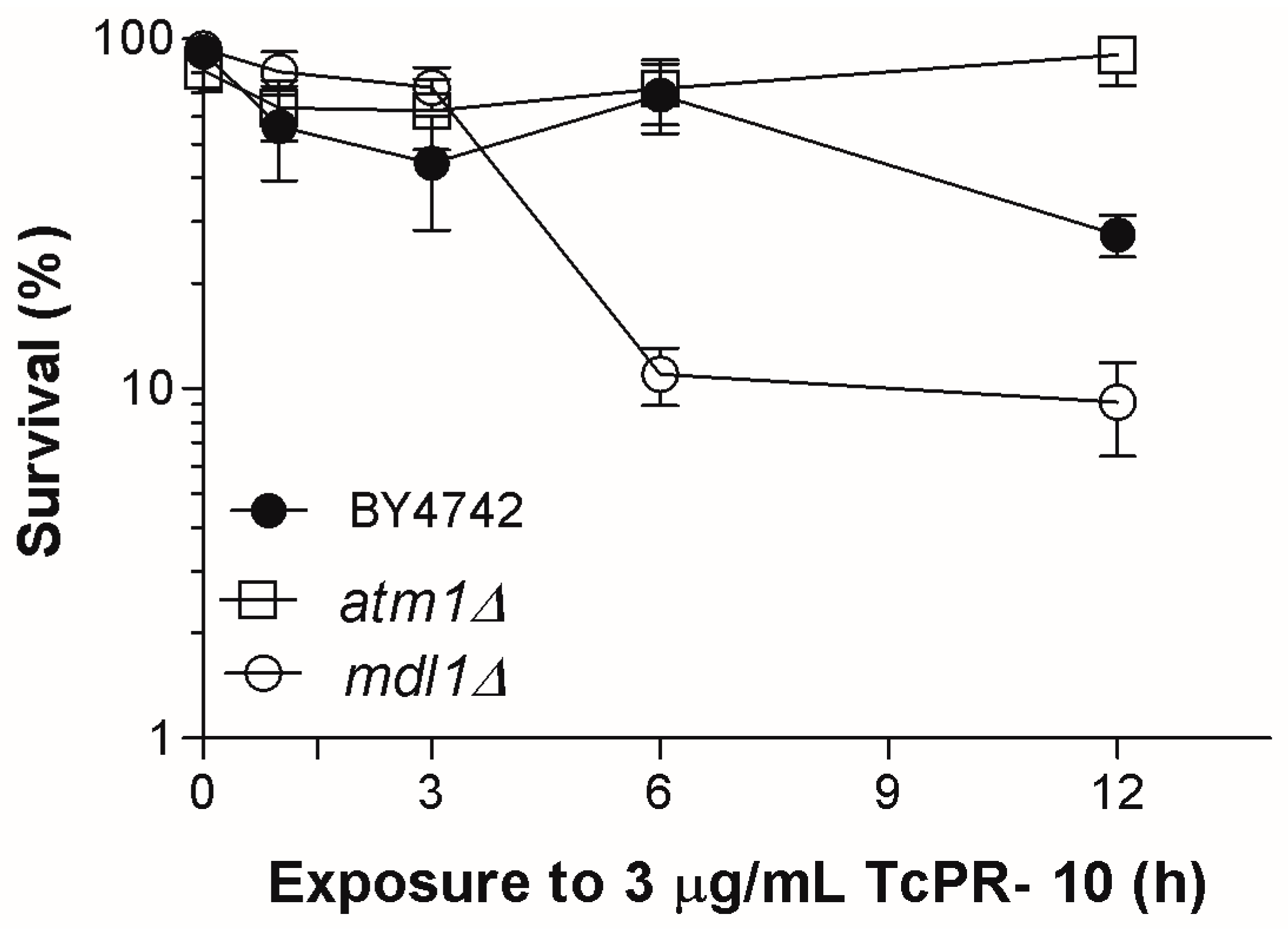
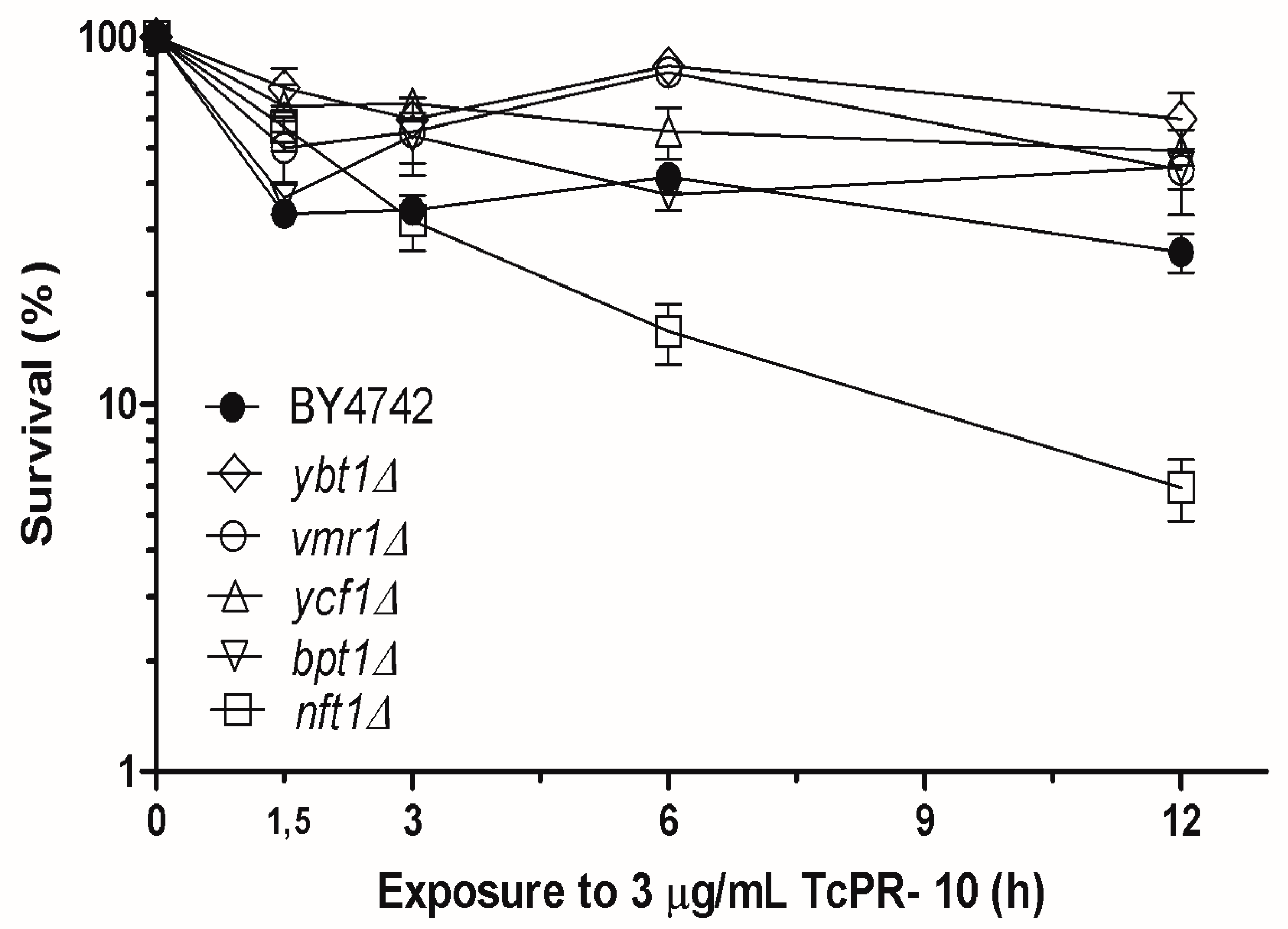
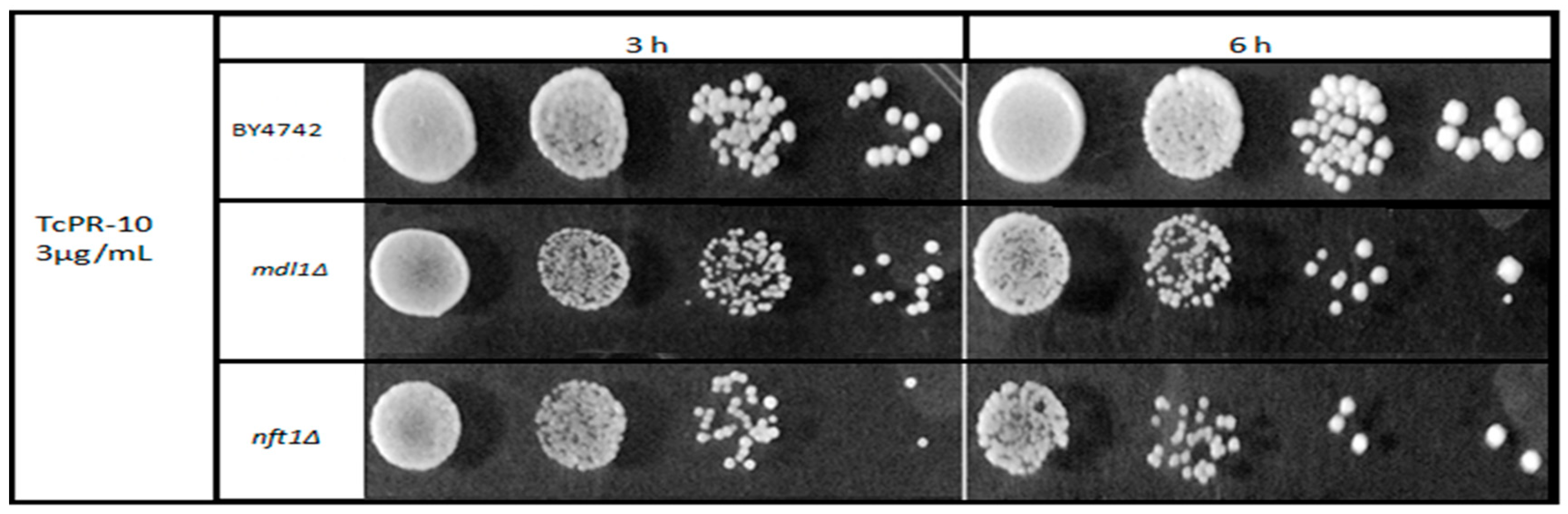
3.1. Yeast Survival after Exposure to TcPR-10p
3.2. Yeast Sensitive Mutants Show Increased ROS Levels Induced by TcPR-10p
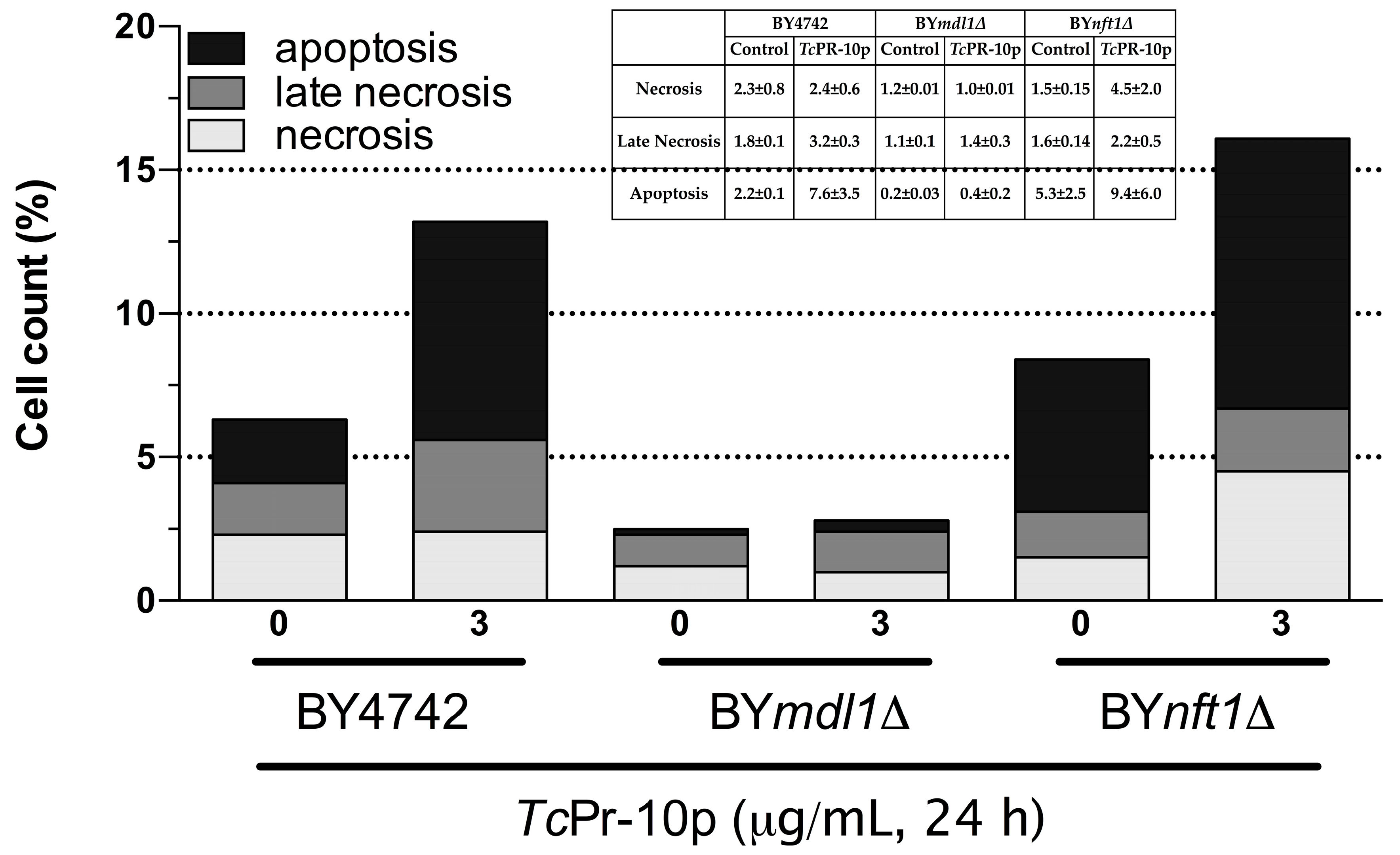
3.3. Flow Cytometry Assay of TcPR-10p Sensitive Yeast Mutants
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Zhao, J.; Davis, L.C.; Verpoorte, R. Elicitor signal transduction leading to production of plant secondary metabolites. Biotechnol. Adv. 2005, 23, 283–333. [Google Scholar] [CrossRef] [PubMed]
- Van Loon, L.; Rep, M.; Pieterse, C. Significance of inducible defense-related proteins in infected plants. Annu. Rev. Phytopathol. 2006, 44, 135–162. [Google Scholar] [CrossRef] [PubMed]
- Sels, J.; Mathys, J.; De Coninck, B.M.; Cammue, B.P.; De Bolle, M.F. Plant pathogenesis-related (PR) proteins: A focus on PR peptides. Plant Physiol. Biochem. 2008, 46, 941–950. [Google Scholar] [CrossRef] [PubMed]
- Doornbos, R.F.; Geraats, B.P.; Kuramae, E.E.; Van Loon, L.C.; Bakker, P.A. Effects of jasmonic acid, ethylene, and salicylic acid signaling on the rhizosphere bacterial community of Arabidopsis thaliana. Mol. Plant Microbe Interact. 2011, 24, 395–407. [Google Scholar] [CrossRef] [PubMed]
- García-Cristobal, J.; García-Villaraco, A.; Ramos, B.; Gutierrez-Mañero, J.; Lucas, J.A. Priming of pathogenesis related-proteins and enzymes related to oxidative stress by plant growth promoting rhizobacteria on rice plants upon abiotic and biotic stress challenge. J. Plant Physiol. 2015, 188, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Karppinen, K.; Derzsó, E.; Jaakola, L.; Hohtola, A. Molecular Cloning and Expression Analysis of hyp-1 Type PR-10 Family Genes in Hypericum perforatum. Front. Plant Sci. 2016, 7, 526. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, P.; Agarwal, P.K. Pathogenesis related-10 proteins are small, structurally similar but with diverse role in stress signaling. Mol. Biol. Rep. 2014, 41, 599–611. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, H.; Michalska, K.; Sikorski, M.; Jaskolski, M. Structural and functional aspects of PR-10 proteins. FEBS J. 2013, 280, 1169–1199. [Google Scholar] [CrossRef] [PubMed]
- Breda, C.; Sallaud, C.; El-Turk, J.; Buffard, D.; De Kozak, I.; Esnault, R.; Kondorosi, A. Defense reaction in Medicago sativa: A gene encoding a class 10 PR protein is expressed in vascular bundles. Mol. Plant Microbe Interact. 1996, 9, 713–719. [Google Scholar] [CrossRef] [PubMed]
- Sikorski, M.M.; Biesiadka, J.; Kasperska, A.E.; Kopcinska, J.; Lotocka, B.; Golinowski, W.; Blegocki, A. Expression of genes encoding PR10 class pathogenesis-related proteins is inhibited in yellow lupine root nodules. Plant Sci. 1999, 149, 125–137. [Google Scholar] [CrossRef]
- Puhringer, H.; Moll, D.; Hoffmann-Sommergruber, K.; Watillon, B.; Katinger, H.; Machado, M.L.D. The promoter of an apple Ypr10 gene, encoding the major allergen Mal d 1, is stress- and pathogen-inducible. Plant Sci. 2000, 152, 35–50. [Google Scholar] [CrossRef]
- Tokuriki, N.; Tawfik, D.S. Protein dynamism and evolvability. Science 2009, 324, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Franco, O.L. Peptide promiscuity: An evolutionary concept for plant defense. FEBS Lett. 2011, 585, 995–1000. [Google Scholar] [CrossRef] [PubMed]
- Moiseyev, G.P.; Beintema, J.J.; Fedoreyeva, L.I.; Yakovlev, G.I. High sequence similarity between a ribonuclease from ginseng calluses and fungus-elicited proteins from parsley indicates that intracellular pathogenesis-related proteins are ribonucleases. Plant 1994, 193, 470–472. [Google Scholar] [CrossRef]
- Pinto, M.P.; Ricardo, C.P. Lupinus albus L. pathogenesis-related proteins that show similarity to PR-10 proteins. Plant Physiol. 1995, 109, 1345–1351. [Google Scholar] [CrossRef] [PubMed]
- Park, C.J.; Kim, K.J.; Shin, R.; Park, J.M.; Shin, Y.C.; Paek, K.H. Pathogenesis-related protein 10 isolated from hot pepper functions as a ribonuclease in an antiviral pathway. Plant J. 2004, 37, 186–198. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Klionsky, D.J. Autophagy, cytosplasm-to-vacuole targeting pathway, and perophagy in yeast and mammalian cells. Annu. Rev. Biochem. 2000, 69, 303–342. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Xu, Y.; Cao, J. Subcellular localization and functional analyses of a PR10 protein gene from Vitis pseudoreticulata in response to Plasmopara viticola infection. Protoplasma 2013, 250, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Gesteira, A.S.; Micheli, F.; Carels, N.; Da Silva, A.C.; Gramacho, K.P.; Schuster, I.; Macêdo, J.N.; Pereira, G.A.; Cascardo, J.C. Comparative analysis of expressed genes from cacao meristems infected by Moniliophthora perniciosa. Ann. Bot. 2007, 100, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.; Kumar, A. The Pathogenesis Related Class 10 proteins in Plant Defense against Biotic and Abiotic Stresses. Adv. Plants Agric. Res. 2015, 3, 00077. [Google Scholar] [CrossRef]
- Pungartnik, C.; Da Silva, A.C.; De Melo, S.A.; Gramacho, K.P.; Mattos Cascardo, J.C.; Brendel, M.; Micheli, F.; Gesteira, A. High-affinity copper transport and Snq2 export permease of Saccharomyces cerevisiae modulate cytotoxicity of PR-10 from Theobroma cacao. Mol. Plant Microbe Interact. 2009, 22, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Silva, F.A.C.; Pirovani, C.P.; Menezes, S.; Pungartnik, C.; Santiago, A.S.; Costa, M.G.C.; Micheli, F.; Gesteira, A.S. Proteomic response of Moniliophthora perniciosa exposed to pathogenesis-related protein-10 from Theobroma cacao. Gen. Mol. Res. 2013, 12, 4855–4868. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Kogan, M.; Knight, S.S.B.; Pain, D.; Dancis, A. Yeast Mitochondrial Protein, Nfs1p, Coordinately Regulates Iron-Sulfur Cluster Proteins, Cellular Iron Uptake, and Iron Distribution. J. Biol. Chem. 1999, 274, 33025–33034. [Google Scholar] [CrossRef] [PubMed]
- Young, L.; Leonhard, K.; Tatsuta, T.; Trowsdale, J.; Langer, T. Role of the ABC transporter Mdl1 in peptide export from mitochondria. Science 2011, 291, 2135–2138. [Google Scholar] [CrossRef] [PubMed]
- Klionsky, D.J.; Baehrecke, E.H.; Brumell, J.H.; Chu, C.T.; Codogno, P.; Cuervo, A.M.; Debnath, J.; Deretic, V.; Elazar, Z.; Eskelinen, E.L.; et al. A comprehensive glossary of autophagy-related molecules and processes (2nd edition). Autophagy 2011, 7, 1273–1294. [Google Scholar] [CrossRef] [PubMed]
- Burke, D.; Dawson, D.; Stearns, T. Methods in Yeast Genetics: A Cold Spring Harbor Laboratory Course Manual; CSHL Press: New York, NY, USA, 2000. [Google Scholar]
- Brendel, M.; Haynes, R.H. Interactions among genes controlling sensitivity to radiation and alkylation in yeast. Mol. Gen. Genet. MGG 1973, 125, 197–216. [Google Scholar] [CrossRef] [PubMed]
- Brendel, M.; Khan, N.; Haynes, R. Common steps in the repair of alkylation and radiation damage in yeast. Mol. Gen. Genet. MGG 1970, 106, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Bradner, J.; Nevalainen, K. Metabolic activity in filamentous fungi can be analysed by flow cytometry. J. Microbial. Meth. 2003, 54, 193–201. [Google Scholar] [CrossRef]
- Koistinen, K.M.; Soininen, P.; Venäläinen, T.A.; Häyrinen, J.; Laatikainen, R.; Peräkylä, M.; Tervahauta, A.I.; Kärenlampi, S.O. Birch PR-10c interacts with several biologically important ligands. Phytochemistry 2005, 66, 2524–2533. [Google Scholar] [CrossRef] [PubMed]
- Kundu, S.; Roy, D. Structural study of biologically significant ligands with major birch pollen allergen Betv1 by docking and molecular dynamics simulation. Bioinformation 2010, 4, 326–330. [Google Scholar] [CrossRef] [PubMed]
- Fister, A.S.; Mejia, L.C.; Zhang, Y.; Herre, E.A.; Maximova, S.N.; Guiltinan, M.J. Theobroma cacao L. pathogenesis-related gene tandem array members show diverse expression dynamics in response to pathogen colonization. BMC Genom. 2016, 17, 363. [Google Scholar] [CrossRef]
- Hwang, J.; Song, W.; Hong, D.; Ko, D.; Yamaoka, Y.; Jang, S.; Yim, S.; Lee, H.; Khare, D.; Kim, K.; et al. Plant ABC Transporters Enable Many Unique Aspects of a Terrestrial Plant’s Lifestyle. Mol. Plant. 2016, 9, 338–355. [Google Scholar] [CrossRef] [PubMed]
- Matuo, R.; Sousa, F.G.; Soares, D.G.; Bonatto, D.; Saffi, J.; Escargueil, A.E.; Larsen, A.K.; Henriques, J.A.P. Saccharomyces cerevisiae as a model system to study the response to anticancer agents. Cancer Chemoth. Pharm. 2012, 70, 491–502. [Google Scholar] [CrossRef] [PubMed]
- Theresia, A.S.; Belinda, F.; Chitra, A.S.; Carpenter, E.P.; Vasundara, S.; Van Veen, H.; Balk, J. Structures and functions of mitochondrial ABC transporters. Biochem. Soc. Trans. 2015, 43, 943–951. [Google Scholar] [CrossRef]
- Khan, A.A.; Quigley, J.G. Control of intracellular heme levels: Heme transporters and Heme oxygenases. Biochim. Biophys. Acta 2011, 1813, 668–682. [Google Scholar] [CrossRef] [PubMed]
- Chloupkova, M.; Lebard, L.S.; Koeller, D.M. MDL1 is a high copy suppressor of ATM1: Evidence for a role in resistance to oxidative stress. J. Mol. Biol. 2003, 331, 155–165. [Google Scholar] [CrossRef]
- Liesa, M.; Qiu, W.; Shirihai, O.S. Mitochondrial ABC transporters function: The role of ABCB10 (ABC-me) as a novel player in cellular handling of reactive oxygen species. Biochim. Biophys. Acta 2012, 1823, 1945–1957. [Google Scholar] [CrossRef] [PubMed]
- Mroczek, S.; Kufel, J. Apoptotic signals induce specific degradation of ribosomal RNA in yeast. Nucleic Acids Res. 2008, 36, 2874–2888. [Google Scholar] [CrossRef] [PubMed]
- Yorimitsu, T.; Zaman, S.; Broach, J.R.; Klionsky, D.J. Protein Kinase A and Sch9 Cooperatively Regulate Induction of Autophagy in Saccharomyces cerevisiae. Mol. Biol. Cell 2007, 18, 4180–4189. [Google Scholar] [CrossRef] [PubMed]
- Wawrzycka, D.; Sobczak, I.; Bartosz, G.; Bocer, T.; Ułaszewski, S.; Goffeau, A. Vmr1p is a novel vacuolar multidrug resistance ABC transporter in Saccharomyces cerevisiae. FEMS Yeast Res. 2010, 10, 828–838. [Google Scholar] [CrossRef] [PubMed]
- Adamis, P.D.B.; Panek, A.D.; Eleutherio, E.C.A. Vacuolar compartmentation of the cadmium-glutathione complex protects Saccharomyces cerevisiae from mutagenesis. Toxicol. Lett. 2007, 173, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Paumi, C.M.; Pickin, K.A.; Jarrar, R.; Herren, C.K.; Cowley, S.T. Ycf1p attenuates basal level oxidative stress response in Saccharomyces cerevisiae. FEBS Lett. 2012, 586, 847–853. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Qi, H.; Taylor, R.; Xu, W.; Liu, L.F.; Jin, S.V. The Role of Autophagy in Mitochondria Maintenance: Characterization of Mitochondrial Functions in Autophagy-Deficient S. cerevisiae Strains. Autophagy 2007, 3, 337–346. [Google Scholar] [CrossRef] [PubMed]
- Yan, Q.; Qi, X.; Jiang, Z.; Yang, S.; Han, L. Characterization of a pathogenesis-related class 10 protein (PR-10) from Astragalus mongholicus with ribonuclease activity. Plant Physiol. Biochem. 2008, 46, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Madeo, F.; Herker, E.; Wissing, S.; Jungwirth, H.; Eisenberg, T.; Fröhlich, K.U. Apoptosis in yeast. Curr. Opin. Microbiol. 2004, 7, 655–660. [Google Scholar] [CrossRef] [PubMed]
- Fabrizio, P.; Battistella, L.; Vardavas, R.; Gattazzo, C.; Liou, L.L.; Diaspro, A.; Dossen, J.W.; Gralla, E.B.; Longo, V.D. Superoxide is a mediator of an altruistic aging program in Saccharomyces cerevisiae. J. Cell. Biol. 2004, 166, 1055–1067. [Google Scholar] [CrossRef] [PubMed]
- Desagher, S.; Martinou, J.C. Mitochondria as the central control point of apoptosis. Trends Cell Biol. 2000, 10, 369–376. [Google Scholar] [CrossRef]
- Kroemer, G.; Reed, J.C. Mitochondrial control of cell death. Nat Med. 2000, 6, 513–516. [Google Scholar] [CrossRef] [PubMed]
- Basso, T.S.; Vita-Santos, E.; Marisco, G.; Pungartnik, C.; Brendel, M. Changes in cellular infrastructure after induced endoplasmic reticulum stress in Moniliophthora perniciosa. Mycologia 2016, 108l, 869–881. [Google Scholar] [CrossRef] [PubMed]

| Strains * | Genotype | Name Description/Cellular Component ** |
|---|---|---|
| BY4742 (WT) | MATα his3∆1 leu2∆0 lys2∆0 ura3∆0 | Wild type for ABC transporters |
| BYmdl1Δ | Same as WT, YLR188w deleted | Multi-Drug resistance-Like/Mitochondrial inner membrane half-type ABC transporter; mediates export of peptides generated upon proteolysis of mitochondrial proteins; plays a role in the regulation of cellular resistance to oxidative stress |
| BYatm1Δ | Same as WT, YMR301c deleted | ABC Transporter, Mitochondria/Mitochondrial inner membrane ATP-binding cassette (ABC) transporter; exports mitochondrially synthesized precursors of iron-sulfur (Fe/S) clusters to the cytosol |
| BYybt1Δ | Same as WT, YLL048c deleted | Yeast Bile Transporter/Transporter of the ATP-binding cassette (ABC) family; negative regulator of vacuole fusion |
| BYnft1Δ | Same as WT, YKR103w deleted | New Full-length MRP-type Transporter/Transporter of the ATP-binding cassette (ABC) vacuole; multidrug resistance-associated protein |
| BYyct1Δ | Same as WT, YLL055w deleted | Yeast Cysteine Transporter/Transporter of the ATP-binding cassette (ABC) vacuole; High-affinity cysteine-specific transporter |
| BYbpt1Δ | Same as WT, YLL015w deleted | Bile Pigment Transporter/ABC type transmembrane transporter of MRP/CFTR family; found in vacuolar membrane, involved in the transport of unconjugated bilirubin and in heavy metal detoxification via glutathione conjugates, along with Ycf1p |
| BYvmr1Δ | Same as WT, YHL035c deleted | Vacuolar Multidrug Resistance/Vacuolar membrane protein; involved in multiple drug resistance and metal sensitivity; ATP-binding cassette (ABC) family member involved in drug transport |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barreto, L.R.; Barreto, T.; Melo, S.; Pungartnik, C.; Brendel, M. Sensitivity of Yeast Mutants Deficient in Mitochondrial or Vacuolar ABC Transporters to Pathogenesis-Related Protein TcPR-10 of Theobroma cacao. Biology 2018, 7, 35. https://doi.org/10.3390/biology7020035
Barreto LR, Barreto T, Melo S, Pungartnik C, Brendel M. Sensitivity of Yeast Mutants Deficient in Mitochondrial or Vacuolar ABC Transporters to Pathogenesis-Related Protein TcPR-10 of Theobroma cacao. Biology. 2018; 7(2):35. https://doi.org/10.3390/biology7020035
Chicago/Turabian StyleBarreto, Louise R., Thayná Barreto, Sonia Melo, Cristina Pungartnik, and Martin Brendel. 2018. "Sensitivity of Yeast Mutants Deficient in Mitochondrial or Vacuolar ABC Transporters to Pathogenesis-Related Protein TcPR-10 of Theobroma cacao" Biology 7, no. 2: 35. https://doi.org/10.3390/biology7020035
APA StyleBarreto, L. R., Barreto, T., Melo, S., Pungartnik, C., & Brendel, M. (2018). Sensitivity of Yeast Mutants Deficient in Mitochondrial or Vacuolar ABC Transporters to Pathogenesis-Related Protein TcPR-10 of Theobroma cacao. Biology, 7(2), 35. https://doi.org/10.3390/biology7020035





