Raising an Eye at Facial Muscle Morphology in Canids
Abstract
Simple Summary
Abstract
1. Introduction
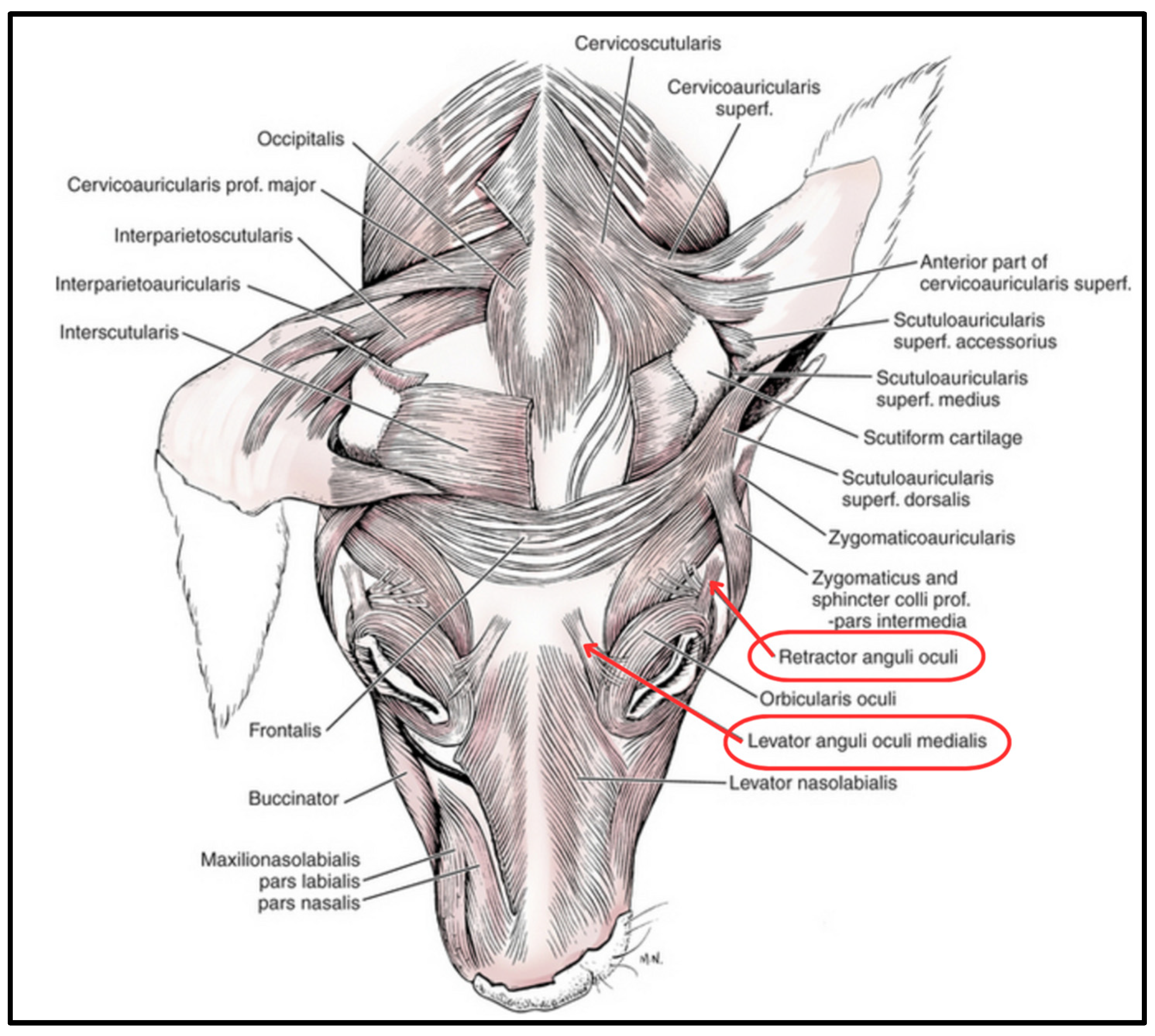
2. Materials and Methods
2.1. Ethical Considerations
2.2. Anatomical Specimens and Experimental Procedure
3. Results
4. Discussion
5. Conclusions
- Analyses of neural differences as they relate to behavioral differences acquired during domestication. In particular, are dogs’ brains now “wired” differently from wolves or other canids to recognize facial expressions in humans (and perhaps less so in conspecifics), and to instinctively make similar expressions to communicate with humans on whom they are dependent [48,78]?
- Analyses of the impact of superficial phenotypic features of the face—such as pigmentation, markings, and patterning—on interspecies communication, specifically between dogs and humans. Previous work suggests that the diversity of facial appearance (e.g., superficial facial features) is significant in conspecific communication in highly social species of primates and canids [11,79,80], and perhaps even more meaningful than subsurface muscle movements.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kaminski, J.; Waller, B.M.; Diogo, R.; Hartstone-Rose, A.; Burrows, A.M. Evolution of Facial Muscle Anatomy in Dogs. Proc. Natl. Acad. Sci. USA 2019, 116, 14677–14681. [Google Scholar] [CrossRef] [PubMed]
- Carr, L.; Iacoboni, M.; Dubeau, M.-C.; Mazziotta, J.C.; Lenzi, G.L. Neural Mechanisms of Empathy in Humans: A Relay from Neural Systems for Imitation to Limbic Areas. Proc. Natl. Acad. Sci. USA 2003, 100, 5497–5502. [Google Scholar] [CrossRef] [PubMed]
- Goubert, L.; Craig, K.D.; Vervoort, T.; Morley, S.; Sullivan, M.J.L.; de Williams, C.A.C.; Cano, A.; Crombez, G. Facing Others in Pain: The Effects of Empathy. Pain 2005, 118, 285–288. [Google Scholar] [CrossRef] [PubMed]
- Jack, R.E.; Schyns, P.G. The Human Face as a Dynamic Tool for Social Communication. Curr. Biol. 2015, 25, R621–R634. [Google Scholar] [CrossRef] [PubMed]
- Kanwisher, N.; Yovel, G. The Fusiform Face Area: A Cortical Region Specialized for the Perception of Faces. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2006, 361, 2109–2128. [Google Scholar] [CrossRef]
- Hare, B.; Tomasello, M. Human-like Social Skills in Dogs? Trends Cogn. Sci. 2005, 9, 439–444. [Google Scholar] [CrossRef] [PubMed]
- Kaminski, J.; Nitzschner, M. Do Dogs Get the Point? A Review of Dog–Human Communication Ability. Learn. Motiv. 2013, 44, 294–302. [Google Scholar] [CrossRef]
- Salomons, H.; Smith, K.C.M.; Callahan-Beckel, M.; Callahan, M.; Levy, K.; Kennedy, B.S.; Bray, E.E.; Gnanadesikan, G.E.; Horschler, D.J.; Gruen, M.; et al. Cooperative Communication with Humans Evolved to Emerge Early in Domestic Dogs. Curr. Biol. 2021, 31, 3137–3144.e11. [Google Scholar] [CrossRef] [PubMed]
- Savalli, C.; Resende, B.; Gaunet, F. Eye Contact Is Crucial for Referential Communication in Pet Dogs. PLoS ONE 2016, 11, e0162161. [Google Scholar] [CrossRef]
- Sommese, A.; Miklósi, Á.; Pogány, Á.; Temesi, A.; Dror, S.; Fugazza, C. An Exploratory Analysis of Head-Tilting in Dogs. Anim. Cogn. 2022, 25, 701–705. [Google Scholar] [CrossRef]
- Sexton, C.L.; Buckley, C.; Lieberfarb, J.; Subiaul, F.; Hecht, E.E.; Bradley, B.J. What Is Written on a Dog’s Face? Evaluating the Impact of Facial Phenotypes on Communication between Humans and Canines. Animals 2023, 13, 2385. [Google Scholar] [CrossRef] [PubMed]
- Kaminski, J.; Marshall-Pescini, S. The Social Dog: Behavior and Cognition; Elsevier: Amsterdam, The Netherlands, 2014; ISBN 9780124079311. [Google Scholar]
- Scott, J.P.; Fuller, J.L. Genetics and the Social Behavior of the Dog; University of Chicago Press: Chicago, IL, USA, 2012; ISBN 9780226190822. [Google Scholar]
- Siniscalchi, M.; d’Ingeo, S.; Minunno, M.; Quaranta, A. Communication in Dogs. Animals 2018, 8, 131. [Google Scholar] [CrossRef] [PubMed]
- Leopold, D.A.; Rhodes, G. A Comparative View of Face Perception. J. Comp. Psychol. 2010, 124, 233–251. [Google Scholar] [CrossRef] [PubMed]
- Kret, M.E.; Prochazkova, E.; Sterck, E.H.M.; Clay, Z. Emotional Expressions in Human and Non-Human Great Apes. Neurosci. Biobehav. Rev. 2020, 115, 378–395. [Google Scholar] [CrossRef]
- Steiper, M.E.; Grube, N.T.; Gagnon, C.M. Elevated Diversity in Loci Linked to Facial Morphology Is Consistent with the Hypothesis That Individual Facial Recognition Is Important across Hominoids. Am. J. Phys. Anthropol. 2021, 174, 785–791. [Google Scholar] [CrossRef]
- Palagi, E.; Mancini, G. Playing with the Face: Playful Facial “Chattering” and Signal Modulation in a Monkey Species (Theropithecus Gelada). J. Comp. Psychol. 2011, 125, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Santana, S.E.; Alfaro, J.L.; Noonan, A.; Alfaro, M.E. Adaptive Response to Sociality and Ecology Drives the Diversification of Facial Colour Patterns in Catarrhines. Nat. Commun. 2013, 4, 2765. [Google Scholar] [CrossRef] [PubMed]
- Waller, B.M.; Kavanagh, E.; Micheletta, J.; Clark, P.R.; Whitehouse, J. The Face Is Central to Primate Multicomponent Signals. Int. J. Primatol. 2022. [Google Scholar] [CrossRef]
- Davila Ross, M.; Menzler, S.; Zimmermann, E. Rapid Facial Mimicry in Orangutan Play. Biol. Lett. 2008, 4, 27–30. [Google Scholar] [CrossRef]
- Knolle, F.; Goncalves, R.P.; Morton, A.J. Sheep Recognize Familiar and Unfamiliar Human Faces from Two-Dimensional Images. R Soc Open Sci 2017, 4, 171228. [Google Scholar] [CrossRef]
- Marzluff, J.M.; Miyaoka, R.; Minoshima, S.; Cross, D.J. Brain Imaging Reveals Neuronal Circuitry Underlying the Crow’s Perception of Human Faces. Proc. Natl. Acad. Sci. USA 2012, 109, 15912–15917. [Google Scholar] [CrossRef] [PubMed]
- Taylor, D.; Hartmann, D.; Dezecache, G.; Te Wong, S.; Davila-Ross, M. Facial Complexity in Sun Bears: Exact Facial Mimicry and Social Sensitivity. Sci. Rep. 2019, 9, 4961. [Google Scholar] [CrossRef] [PubMed]
- Parr, L.A.; Waller, B.M.; Fugate, J. Emotional Communication in Primates: Implications for Neurobiology. Curr. Opin. Neurobiol. 2005, 15, 716–720. [Google Scholar] [CrossRef] [PubMed]
- Bloom, T.; Friedman, H. Classifying Dogs’ (Canis Familiaris) Facial Expressions from Photographs. Behav. Process. 2013, 96, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Fox, M.W. A Comparative Study of the Development of Facial Expressions in Canids; Wolf, Coyote and Foxes. Behaviour 1970, 36, 49–73. [Google Scholar] [CrossRef]
- Waller, B.M.; Cray, J.J.; Burrows, A.M. Selection for Universal Facial Emotion. Emotion 2008, 8, 435–439. [Google Scholar] [CrossRef]
- Cattaneo, L.; Pavesi, G. The Facial Motor System. Neurosci. Biobehav. Rev. 2014, 38, 135–159. [Google Scholar] [CrossRef]
- Burrows, A.M. The Facial Expression Musculature in Primates and Its Evolutionary Significance. Bioessays 2008, 30, 212–225. [Google Scholar] [CrossRef] [PubMed]
- Powell, V.; Esteve-Altava, B.; Molnar, J.; Villmoare, B.; Pettit, A.; Diogo, R. Primate Modularity and Evolution: First Anatomical Network Analysis of Primate Head and Neck Musculoskeletal System. Sci. Rep. 2018, 8, 2341. [Google Scholar] [CrossRef]
- Albuquerque, N.; Guo, K.; Wilkinson, A.; Savalli, C.; Otta, E.; Mills, D. Dogs Recognize Dog and Human Emotions. Biol. Lett. 2016, 12, 20150883. [Google Scholar] [CrossRef]
- Bekoff, M. The Emotional Lives of Animals: A Leading Scientist Explores Animal Joy, Sorrow, and Empathy—And Why They Matter; New World Library: Novato, CA, USA, 2010; ISBN 9781577313489. [Google Scholar]
- Safina, C. Beyond Words. In Farming, Food and Nature; Series: Earthscan food and agriculture; Routledge: New York, NY, USA, 2018; pp. 75–85. ISBN 9781351011013. [Google Scholar]
- Kaminski, J.; Hynds, J.; Morris, P.; Waller, B.M. Human Attention Affects Facial Expressions in Domestic Dogs. Sci. Rep. 2017, 7, 12914. [Google Scholar] [CrossRef] [PubMed]
- DogFACS: The Dog Facial Action Coding System. Available online: https://animalfacs.com/dogfacs_new (accessed on 17 November 2017).
- Burrows, A.; Diogo, R.; Waller, B.; Kaminski, J. Variation of Facial Musculature between Wolves and Domestic Dogs: Evolutionary Divergence in Facial Movement. FASEB J. 2018, 31, 577.3. [Google Scholar] [CrossRef]
- Ghazanfar, A.A.; Kelly, L.M.; Takahashi, D.Y.; Winters, S.; Terrett, R.; Higham, J.P. Domestication Phenotype Linked to Vocal Behavior in Marmoset Monkeys. Curr. Biol. 2020, 30, 5026–5032.e3. [Google Scholar] [CrossRef] [PubMed]
- San-Jose, L.M.; Roulin, A. On the Potential Role of the Neural Crest Cells in Integrating Pigmentation into Behavioral and Physiological Syndromes. Front. Ecol. Evol. 2020, 8, 278. [Google Scholar] [CrossRef]
- Frantz, L.A.F.; Mullin, V.E.; Pionnier-Capitan, M.; Lebrasseur, O.; Ollivier, M.; Perri, A.; Linderholm, A.; Mattiangeli, V.; Teasdale, M.D.; Dimopoulos, E.A.; et al. Genomic and Archaeological Evidence Suggest a Dual Origin of Domestic Dogs. Science 2016, 352, 1228–1231. [Google Scholar] [CrossRef]
- Larson, G.; Karlsson, E.K.; Perri, A.; Webster, M.T.; Ho, S.Y.W.; Peters, J.; Stahl, P.W.; Piper, P.J.; Lingaas, F.; Fredholm, M.; et al. Rethinking Dog Domestication by Integrating Genetics, Archeology, and Biogeography. Proc. Natl. Acad. Sci. USA 2012, 109, 8878–8883. [Google Scholar] [CrossRef] [PubMed]
- Savolainen, P.; Zhang, Y.-P.; Luo, J.; Lundeberg, J.; Leitner, T. Genetic Evidence for an East Asian Origin of Domestic Dogs. Science 2002, 298, 1610–1613. [Google Scholar] [CrossRef]
- Vilà, C.; Savolainen, P.; Maldonado, J.E.; Amorim, I.R.; Rice, J.E.; Honeycutt, R.L.; Crandall, K.A.; Lundeberg, J.; Wayne, R.K. Multiple and Ancient Origins of the Domestic Dog. Science 1997, 276, 1687–1689. [Google Scholar] [CrossRef]
- Caragiulo, A.; Gaughran, S.J.; Duncan, N.; Nagy, C.; Weckel, M.; vonHoldt, B.M. Coyotes in New York City Carry Variable Genomic Dog Ancestry and Influence Their Interactions with Humans. Genes 2022, 13, 1661. [Google Scholar] [CrossRef]
- vonHoldt, B.M.; Brzeski, K.E.; Aardema, M.L.; Schell, C.J.; Rutledge, L.Y.; Fain, S.R.; Shutt, A.C.; Linderholm, A.; Murphy, W.J. Persistence and Expansion of Cryptic Endangered Red Wolf Genomic Ancestry along the American Gulf Coast. Mol. Ecol. 2022, 31, 5440–5454. [Google Scholar] [CrossRef]
- Wrangham, R.W. Hypotheses for the Evolution of Reduced Reactive Aggression in the Context of Human Self-Domestication. Front. Psychol. 2019, 10, 1914. [Google Scholar] [CrossRef] [PubMed]
- Range, F.; Marshall-Pescini, S. Comparing Wolves and Dogs: Current Status and Implications for Human “Self-Domestication”. Trends Cogn. Sci. 2022, 26, 337–349. [Google Scholar] [CrossRef] [PubMed]
- Bray, E.E.; Gnanadesikan, G.E.; Horschler, D.J.; Levy, K.M.; Kennedy, B.S.; Famula, T.R.; MacLean, E.L. Early-Emerging and Highly Heritable Sensitivity to Human Communication in Dogs. Curr. Biol. 2021, 31, 3132–3136.e5. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.E.; Evans, H.E. Miller’s Anatomy of the Dog; W.B. Saunders: Philadelphia, PA, USA, 1993. [Google Scholar]
- Diogo, R.; Abdala, V.; Lonergan, N.; Wood, B.A. From Fish to Modern Humans—Comparative Anatomy, Homologies and Evolution of the Head and Neck Musculature. J. Anat. 2008, 213, 391–424. [Google Scholar] [CrossRef] [PubMed]
- Tedford, R.H.; Wang, X.; Taylor, B.E. Phylogenetic Systematics of the North American Fossil Caninae (Carnivora: Canidae). Bull. Am. Mus. Nat. Hist. 2009, 2009, 1–218. [Google Scholar] [CrossRef]
- Zhu, Q.; Hillmann, D.J.; Henk, W.G. Observations on the Muscles of the Eye of the Bowhead Whale, Balaena Mysticetus. Anat. Rec. 2000, 259, 189–204. [Google Scholar] [CrossRef]
- Bremhorst, A.; Mills, D.S.; Stolzlechner, L.; Würbel, H.; Riemer, S. “Puppy Dog Eyes” Are Associated With Eye Movements, Not Communication. Front. Psychol. 2021, 12, 568935. [Google Scholar] [CrossRef] [PubMed]
- American Kennel Club. The New Complete Dog Book. In Official Breed Standards and Profiles for Over 200 Breeds, 23rd ed.; CompanionHouse Books: Fox Chapel, PA, USA, 2023; ISBN 978-1621871972. [Google Scholar]
- Burrows, A.M.; Kaminski, J.; Waller, B.M.; Omstead, K.M.; Rogers-Vizena, C.; Mendelson, B. Dog Faces Exhibit Anatomical Differences in Comparison to Other Domestic Animals. Anat. Rec. 2021, 304, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Ostrander, E.A.; Wayne, R.K. The Canine Genome. Genome Res. 2005, 15, 1706–1716. [Google Scholar] [CrossRef] [PubMed]
- Schatz, K.Z.; Engelke, E.; Pfarrer, C. Comparative Morphometric Study of the Mimic Facial Muscles of Brachycephalic and Dolichocephalic Dogs. Anat. Histol. Embryol. 2021, 50, 863–875. [Google Scholar] [CrossRef]
- Gompper, M.E. Top Carnivores in the Suburbs? Ecological and Conservation Issues Raised by Colonization of North-Eastern North America by Coyotes: The Expansion of the Coyote’s Geographical Range May Broadly Influence Community Structure, and Rising Coyote Densities in the Suburbs May Alter How the General Public Views Wildlife. Bioscience 2002, 52, 185–190. [Google Scholar]
- Gehrt, S.D. Ecology of Coyotes in Urban Landscapes; University of Nebraska: Lincoln, NE, USA, 2007. [Google Scholar]
- Gese, E.M.; Morey, P.S.; Gehrt, S.D. Influence of the Urban Matrix on Space Use of Coyotes in the Chicago Metropolitan Area. J. Ethol. 2012, 30, 413–425. [Google Scholar] [CrossRef]
- Grubbs, S.E.; Krausman, P.R. Use of Urban Landscape by Coyotes. Southwest. Nat. 2009, 54, 1–12. [Google Scholar] [CrossRef]
- Breck, S.W.; Poessel, S.A.; Mahoney, P.; Young, J.K. The Intrepid Urban Coyote: A Comparison of Bold and Exploratory Behavior in Coyotes from Urban and Rural Environments. Sci. Rep. 2019, 9, 2104. [Google Scholar] [CrossRef] [PubMed]
- Schell, C.J.; Young, J.K.; Lonsdorf, E.V.; Santymire, R.M.; Mateo, J.M. Parental Habituation to Human Disturbance over Time Reduces Fear of Humans in Coyote Offspring. Ecol. Evol. 2018, 8, 12965–12980. [Google Scholar] [CrossRef] [PubMed]
- Monzón, J.; Kays, R.; Dykhuizen, D.E. Assessment of Coyote-Wolf-Dog Admixture Using Ancestry-Informative Diagnostic SNPs. Mol. Ecol. 2014, 23, 182–197. [Google Scholar] [CrossRef] [PubMed]
- vonHoldt, B.M.; Kays, R.; Pollinger, J.P.; Wayne, R.K. Admixture Mapping Identifies Introgressed Genomic Regions in North American Canids. Mol. Ecol. 2016, 25, 2443–2453. [Google Scholar] [CrossRef] [PubMed]
- Wheeldon, T.J.; Rutledge, L.Y.; Patterson, B.R.; White, B.N.; Wilson, P.J. Y-Chromosome Evidence Supports Asymmetric Dog Introgression into Eastern Coyotes. Ecol. Evol. 2013, 3, 3005–3020. [Google Scholar] [CrossRef] [PubMed]
- Bekoff, M.; Diamond, J.; Mitton, J.B. Life-History Patterns and Sociality in Canids: Body Size, Reproduction, and Behavior. Oecologia 1981, 50, 386–390. [Google Scholar] [CrossRef]
- Kleiman, D.G. Canid Mating Systems, Social Behavior, Parental Care and Ontogeny: Are They Flexible? Behav. Genet. 2011, 41, 803–809. [Google Scholar] [CrossRef]
- Moehlman, P.D. Intraspecific Variation in Canid Social Systems. In Carnivore Behavior, Ecology, and Evolution; Gittleman, J.L., Ed.; Springer US: Boston, MA, USA, 1989; pp. 143–163. ISBN 9781475747164. [Google Scholar]
- Hare, B.; Plyusnina, I.; Ignacio, N.; Schepina, O.; Stepika, A.; Wrangham, R.; Trut, L. Social Cognitive Evolution in Captive Foxes Is a Correlated By-Product of Experimental Domestication. Curr. Biol. 2005, 15, 226–230. [Google Scholar] [CrossRef] [PubMed]
- Kukekova, A.V.; Trut, L.N.; Acland, G.M. Genetics of Domesticated Behavior in Dogs and Foxes. In Genetics and the Behavior of Domestic Animals; Elsevier: Amsterdam, The Netherlands, 2022; pp. 275–323. ISBN 9780323857529. [Google Scholar]
- Trut, L.; Oskina, I.; Kharlamova, A. Animal Evolution during Domestication: The Domesticated Fox as a Model. Bioessays 2009, 31, 349–360. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, D.; Sau, S.; Das, J.; Bhadra, A. Free-Ranging Dogs Prefer Petting over Food in Repeated Interactions with Unfamiliar Humans. J. Exp. Biol. 2017, 220, 4654–4660. [Google Scholar] [CrossRef] [PubMed]
- Sen Majumder, S.; Paul, M.; Sau, S.; Bhadra, A. Denning Habits of Free-Ranging Dogs Reveal Preference for Human Proximity. Sci. Rep. 2016, 6, 32014. [Google Scholar] [CrossRef] [PubMed]
- Borg, B.L.; Brainerd, S.M.; Meier, T.J.; Prugh, L.R. Impacts of Breeder Loss on Social Structure, Reproduction and Population Growth in a Social Canid. J. Anim. Ecol. 2015, 84, 177–187. [Google Scholar] [CrossRef] [PubMed]
- Marshall-Pescini, S.; Cafazzo, S.; Virányi, Z.; Range, F. Integrating Social Ecology in Explanations of Wolf–Dog Behavioral Differences. Curr. Opin. Behav. Sci. 2017, 16, 80–86. [Google Scholar] [CrossRef]
- Mech, L.D. Alpha Status, Dominance, and Division of Labor in Wolf Packs. Can. J. Zool. 1999, 77, 1196–1203. [Google Scholar] [CrossRef]
- Kujala, M.V.; Kauppi, J.-P.; Törnqvist, H.; Helle, L.; Vainio, O.; Kujala, J.; Parkkonen, L. Time-Resolved Classification of Dog Brain Signals Reveals Early Processing of Faces, Species and Emotion. Sci. Rep. 2020, 10, 19846. [Google Scholar] [CrossRef]
- Santana, S.E.; Dobson, S.D.; Diogo, R. Plain Faces Are More Expressive: Comparative Study of Facial Colour, Mobility and Musculature in Primates. Biol. Lett. 2014, 10, 20140275. [Google Scholar] [CrossRef]
- Ueda, S.; Kumagai, G.; Otaki, Y.; Yamaguchi, S.; Kohshima, S. A Comparison of Facial Color Pattern and Gazing Behavior in Canid Species Suggests Gaze Communication in Gray Wolves (Canis Lupus). PLoS ONE 2014, 9, e98217. [Google Scholar] [CrossRef]
| Specimen | Species | ~Age/Sex | LAOM Present? Y/N | RAOL Present? Y/N | Social/Reproductive Behavior |
|---|---|---|---|---|---|
| N/A | Gray wolf (C. lupus) | --- | Burrows et al., 2018 [37] describe wolf sample (N = 4) in which presence and size of both muscles varied. | Social/cooperative breeders | |
| 1 | Dog (C. familiaris) | Adult/M | Y (robust) | Y (robust) | Social/no cooperative breeding |
| 2 | Dog (C. familiaris) | Adult/M | Y (robust) | Y (robust) | Social/no cooperative breeding |
| 3 | Coyote (C. latrans) | Young Adult?/F | Y (gracile) | Y (gracile) | Social/communal—den sharing |
| 4 | Coyote (C. latrans) | Adult/F | Y (gracile) | Y | Social/communal—den sharing |
| 5 | Coyote (C. latrans) | Adult/M | Y (robust) | Y (gracile) | Social/communal—den sharing |
| 6 | Arctic fox (V. lagopus) | unk/unk | N | undetermined | Monogamous pairs/some den sharing |
| 7 | Red fox (V. v. fulvus) | Adult/F | N | n (orbicularis?) | Social/polygynandry |
| 8 | Red fox (V. v. fulvus) | Juvenile/M | N | n (orbicularis?) | Social/polygynandry |
| 9 | Gray fox (U. cinereoargenteus) | Adult/F | N | N | Solitary/seasonal monogamy/ not cooperative |
| 10 | Gray fox (U. cinereoargenteus) | Adult/M | N | N | Solitary/seasonal monogamy/ not cooperative |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sexton, C.L.; Diogo, R.; Subiaul, F.; Bradley, B.J. Raising an Eye at Facial Muscle Morphology in Canids. Biology 2024, 13, 290. https://doi.org/10.3390/biology13050290
Sexton CL, Diogo R, Subiaul F, Bradley BJ. Raising an Eye at Facial Muscle Morphology in Canids. Biology. 2024; 13(5):290. https://doi.org/10.3390/biology13050290
Chicago/Turabian StyleSexton, Courtney L., Rui Diogo, Francys Subiaul, and Brenda J. Bradley. 2024. "Raising an Eye at Facial Muscle Morphology in Canids" Biology 13, no. 5: 290. https://doi.org/10.3390/biology13050290
APA StyleSexton, C. L., Diogo, R., Subiaul, F., & Bradley, B. J. (2024). Raising an Eye at Facial Muscle Morphology in Canids. Biology, 13(5), 290. https://doi.org/10.3390/biology13050290








