The Impact of Cytomegalovirus Infection on Natural Killer and CD8+ T Cell Phenotype in Multiple Sclerosis
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. CMV Serology
2.3. Detection of CMV DNA by Real-Time PCR
2.4. Isolation of Peripheral Blood Mononuclear Cells
2.5. Flow Cytometry Antibody Staining
2.6. Flow Cytometry Data Analysis
2.7. Statistical Analysis
3. Results
3.1. Study Population
3.2. Detection of Anti-CMV IgG Antibodies and CMV DNA in MS Patients
3.3. Increased NKG2C Expression Levels in MS Patients Compared to Healthy Donors
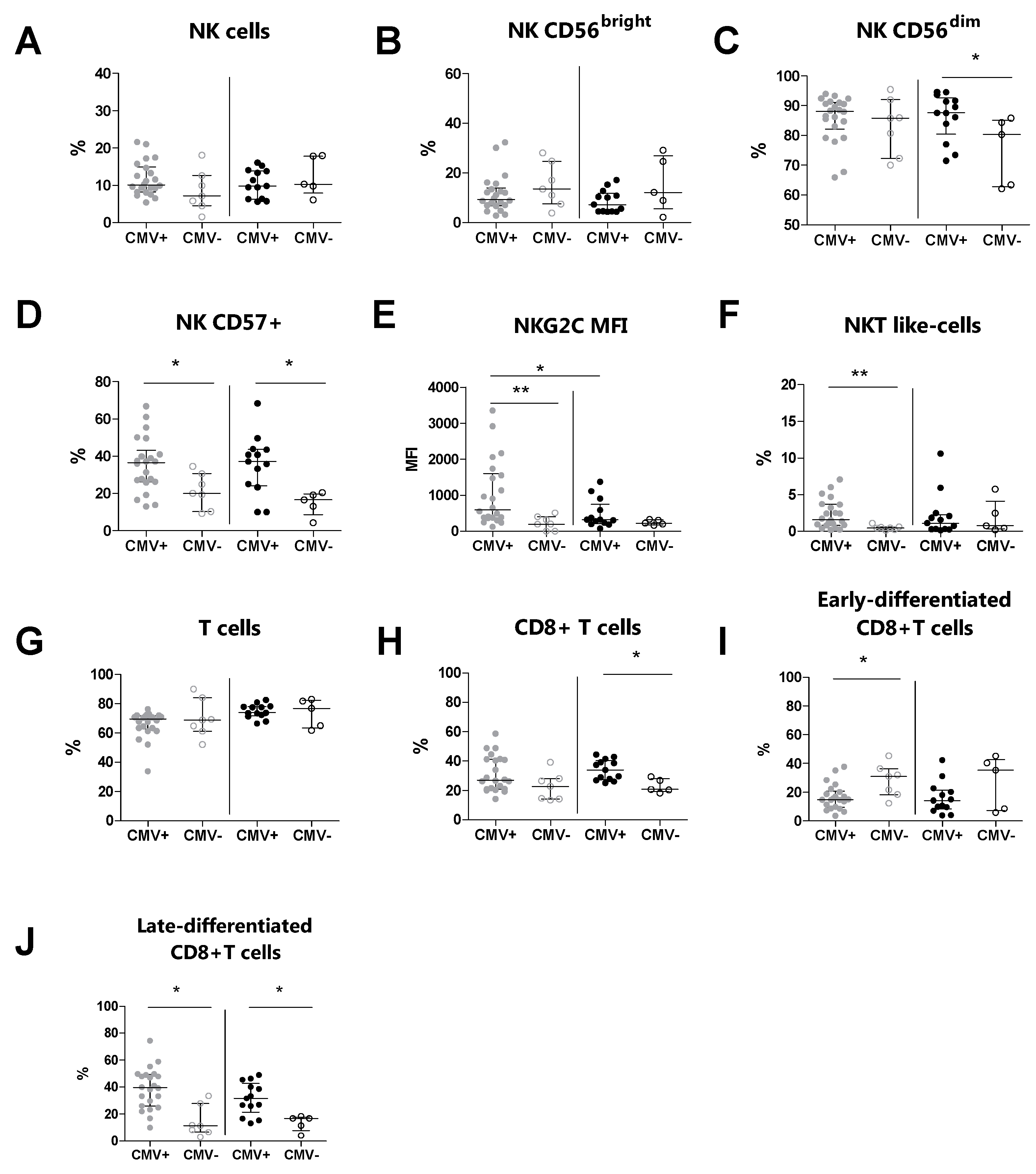
3.4. The CMV Serostatus Influences the Peripheral Blood Phenotype in MS Patients
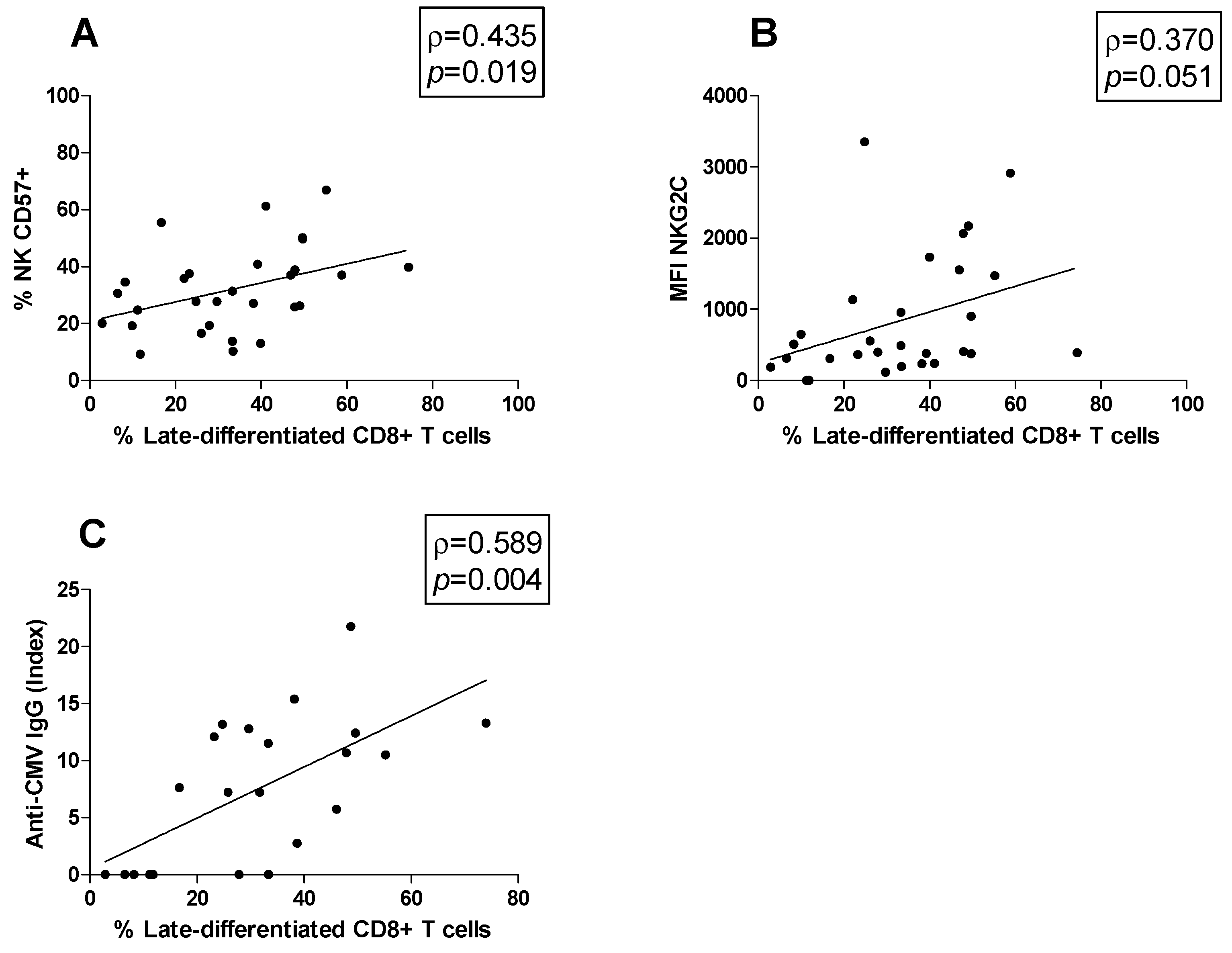
3.5. Correlations
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Compston, A.; Coles, A. Multiple Sclerosis. Lancet 2008, 372, 1502–1517. [Google Scholar] [CrossRef] [PubMed]
- Noseworthy, J.H.; Lucchinetti, C.; Rodriguez, M.; Weinshenker, B.G. Multiple Sclerosis. N. Engl. J. Med. 2000, 343, 938–952. [Google Scholar] [CrossRef]
- Cavallo, S. Immune-Mediated Genesis of Multiple Sclerosis. J. Transl. Autoimmun. 2020, 3, 100039. [Google Scholar] [CrossRef] [PubMed]
- Tarlinton, R.E.; Martynova, E.; Rizvanov, A.A.; Khaiboullina, S.; Verma, S. Role of Viruses in the Pathogenesis of Multiple Sclerosis. Viruses 2020, 12, 643. [Google Scholar] [CrossRef]
- Mechelli, R.; Romano, C.; Reniè, R.; Manfrè, G.; Buscarinu, M.C.; Romano, S.; Marrone, A.; Bigi, R.; Bellucci, G.; Ballerini, C.; et al. Viruses and Neuroinflammation in Multiple Sclerosis. Neuroimmunol. Neuroinflamm. 2021, 8, 269. [Google Scholar] [CrossRef]
- Bjornevik, K.; Cortese, M.; Healy, B.C.; Kuhle, J.; Mina, M.J.; Leng, Y.; Elledge, S.J.; Niebuhr, D.W.; Scher, A.I.; Munger, K.L.; et al. Longitudinal Analysis Reveals High Prevalence of Epstein-Barr Virus Associated with Multiple Sclerosis. Science 2022, 375, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Vanheusden, M.; Stinissen, P.; ’t Hart, B.A.; Hellings, N. Cytomegalovirus: A Culprit or Protector in Multiple Sclerosis? Trends Mol. Med. 2015, 21, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Alari-Pahissa, E.; Moreira, A.; Zabalza, A.; Alvarez-Lafuente, R.; Munteis, E.; Vera, A.; Arroyo, R.; Alvarez-Cermeño, J.C.; Villar, L.M.; López-Botet, M.; et al. Low Cytomegalovirus Seroprevalence in Early Multiple Sclerosis: A Case for the “Hygiene Hypothesis”? Eur. J. Neurol. 2018, 25, 925–933. [Google Scholar] [CrossRef]
- Vanheusden, M.; Broux, B.; Welten, S.P.M.; Peeters, L.M.; Panagioti, E.; Van Wijmeersch, B.; Somers, V.; Stinissen, P.; Arens, R.; Hellings, N. Cytomegalovirus Infection Exacerbates Autoimmune Mediated Neuroinflammation. Sci. Rep. 2017, 7, 663. [Google Scholar] [CrossRef]
- Sanadgol, N.; Ramroodi, N.; Ahmadi, G.A.; Komijani, M.; Moghtaderi, A.; Bouzari, M.; Rezaei, M.; Kardi, M.T.; Dabiri, S.; Moradi, M.; et al. Prevalence of Cytomegalovirus Infection and Its Role in Total Immunoglobulin Pattern in Iranian Patients with Different Subtypes of Multiple Sclerosis. New Microbiol. 2011, 34, 263–274. [Google Scholar]
- Cannon, M.J.; Schmid, D.S.; Hyde, T.B. Review of Cytomegalovirus Seroprevalence and Demographic Characteristics Associated with Infection. Rev. Med. Virol. 2010, 20, 202–213. [Google Scholar] [CrossRef] [PubMed]
- Kothari, A.; Ramachandran, V.G.; Gupta, P.; Singh, B.; Talwar, V. Seroprevalence of Cytomegalovirus among Voluntary Blood Donors in Delhi, India. J. Health Popul. Nutr. 2002, 20, 348–351. [Google Scholar] [PubMed]
- Mendelson, M.; Monard, S.; Sissons, P.; Sinclair, J. Detection of Endogenous Human Cytomegalovirus in CD34+ Bone Marrow Progenitors. J. Gen. Virol. 1996, 77 Pt 12, 3099–3102. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Sejas, N.; Campos, C.; Hassouneh, F.; Sanchez-Correa, B.; Tarazona, R.; Pera, A.; Solana, R. Effect of CMV and Aging on the Differential Expression of CD300a, CD161, T-Bet, and Eomes on NK Cell Subsets. Front. Immunol. 2016, 7, 476. [Google Scholar] [CrossRef]
- Sester, M.; Sester, U.; Gärtner, B.C.; Girndt, M.; Meyerhans, A.; Köhler, H. Dominance of Virus-Specific CD8 T Cells in Human Primary Cytomegalovirus Infection. J. Am. Soc. Nephrol. 2002, 13, 2577–2584. [Google Scholar] [CrossRef] [PubMed]
- Gumá, M.; Budt, M.; Sáez, A.; Brckalo, T.; Hengel, H.; Angulo, A.; López-Botet, M. Expansion of CD94/NKG2C+ NK Cells in Response to Human Cytomegalovirus-Infected Fibroblasts. Blood 2006, 107, 3624–3631. [Google Scholar] [CrossRef] [PubMed]
- Wikby, A.; Johansson, B.; Olsson, J.; Löfgren, S.; Nilsson, B.O.; Ferguson, F. Expansions of Peripheral Blood CD8 T-Lymphocyte Subpopulations and an Association with Cytomegalovirus Seropositivity in the Elderly: The Swedish NONA Immune Study. Exp. Gerontol. 2002, 37, 445–453. [Google Scholar] [CrossRef]
- Solana, R.; Tarazona, R.; Aiello, A.E.; Akbar, A.N.; Appay, V.; Beswick, M.; Bosch, J.A.; Campos, C.; Cantisán, S.; Cicin-Sain, L.; et al. CMV and Immunosenescence: From Basics to Clinics. Immun. Ageing 2012, 9, 23. [Google Scholar] [CrossRef]
- Luetke-Eversloh, M.; Hammer, Q.; Durek, P.; Nordström, K.; Gasparoni, G.; Pink, M.; Hamann, A.; Walter, J.; Chang, H.-D.; Dong, J.; et al. Human Cytomegalovirus Drives Epigenetic Imprinting of the IFNG Locus in NKG2Chi Natural Killer Cells. PLoS Pathog. 2014, 10, e1004441. [Google Scholar] [CrossRef]
- Muntasell, A.; Pupuleku, A.; Cisneros, E.; Vera, A.; Moraru, M.; Vilches, C.; López-Botet, M. Relationship of NKG2C Copy Number with the Distribution of Distinct Cytomegalovirus-Induced Adaptive NK Cell Subsets. J. Immunol. 2016, 196, 3818–3827. [Google Scholar] [CrossRef]
- Gumá, M.; Angulo, A.; Vilches, C.; Gómez-Lozano, N.; Malats, N.; López-Botet, M. Imprint of Human Cytomegalovirus Infection on the NK Cell Receptor Repertoire. Blood 2004, 104, 3664–3671. [Google Scholar] [CrossRef] [PubMed]
- Kastrukoff, L.F.; Lau, A.; Wee, R.; Zecchini, D.; White, R.; Paty, D.W. Clinical Relapses of Multiple Sclerosis Are Associated with “novel” Valleys in Natural Killer Cell Functional Activity. J. Neuroimmunol. 2003, 145, 103–114. [Google Scholar] [CrossRef] [PubMed]
- Infante-Duarte, C.; Weber, A.; Krätzschmar, J.; Prozorovski, T.; Pikol, S.; Hamann, I.; Bellmann-Strobl, J.; Aktas, O.; Dörr, J.; Wuerfel, J.; et al. Frequency of Blood CX3CR1-Positive Natural Killer Cells Correlates with Disease Activity in Multiple Sclerosis Patients. FASEB J. 2005, 19, 1902–1904. [Google Scholar] [CrossRef] [PubMed]
- Zaguia, F.; Saikali, P.; Ludwin, S.; Newcombe, J.; Beauseigle, D.; McCrea, E.; Duquette, P.; Prat, A.; Antel, J.P.; Arbour, N. Cytotoxic NKG2C+ CD4 T Cells Target Oligodendrocytes in Multiple Sclerosis. J. Immunol. 2013, 190, 2510–2518. [Google Scholar] [CrossRef] [PubMed]
- van den Berg, S.P.H.; Pardieck, I.N.; Lanfermeijer, J.; Sauce, D.; Klenerman, P.; van Baarle, D.; Arens, R. The Hallmarks of CMV-Specific CD8 T-Cell Differentiation. Med. Microbiol. Immunol. 2019, 208, 365–373. [Google Scholar] [CrossRef] [PubMed]
- Parish, S.T.; Wu, J.E.; Effros, R.B. Sustained CD28 Expression Delays Multiple Features of Replicative Senescence in Human CD8 T Lymphocytes. J. Clin. Immunol. 2010, 30, 798–805. [Google Scholar] [CrossRef] [PubMed]
- Weng, N.-P.; Akbar, A.N.; Goronzy, J. CD28− T Cells: Their Role in the Age-Associated Decline of Immune Function. Trends Immunol. 2009, 30, 306–312. [Google Scholar] [CrossRef] [PubMed]
- Mikulkova, Z.; Praksova, P.; Stourac, P.; Bednarik, J.; Strajtova, L.; Pacasova, R.; Belobradkova, J.; Dite, P.; Michalek, J. Numerical Defects in CD8+CD28− T-Suppressor Lymphocyte Population in Patients with Type 1 Diabetes Mellitus and Multiple Sclerosis. Cell Immunol. 2010, 262, 75–79. [Google Scholar] [CrossRef]
- Kurtzke, J.F. Rating Neurologic Impairment in Multiple Sclerosis: An Expanded Disability Status Scale (EDSS). Neurology 1983, 33, 1444–1452. [Google Scholar] [CrossRef]
- Zingaropoli, M.A.; Iannetta, M.; Pontecorvo, S.; Anzivino, E.; Prezioso, C.; Rodio, D.M.; Morreale, M.; D’Abramo, A.; Oliva, A.; Lichtner, M.; et al. JC Virus-DNA Detection Is Associated with CD8 Effector Accumulation in Peripheral Blood of Patients with Multiple Sclerosis under Natalizumab Treatment, Independently from JC Virus Serostatus. BioMed Res. Int. 2018, 2018, 5297980. [Google Scholar] [CrossRef]
- Zingaropoli, M.A.; Parente, A.; Kertusha, B.; Campagna, R.; Tieghi, T.; Garattini, S.; Marocco, R.; Carraro, A.; Tortellini, E.; Guardiani, M.; et al. Longitudinal Virological and Immunological Profile in a Case of Human Monkeypox Infection. Open Forum Infect. Dis. 2022, 9, ofac569. [Google Scholar] [CrossRef]
- Olival, G.S.D.; Lima, B.M.; Sumita, L.M.; Serafim, V.; Fink, M.C.; Nali, L.H.; Romano, C.M.; Thomaz, R.B.; Cavenaghi, V.B.; Tilbery, C.P.; et al. Multiple Sclerosis and Herpesvirus Interaction. Arq. Neuropsiquiatr. 2013, 71, 727–730. [Google Scholar] [CrossRef]
- Horakova, D.; Zivadinov, R.; Weinstock-Guttman, B.; Havrdova, E.; Qu, J.; Tamaño-Blanco, M.; Badgett, D.; Tyblova, M.; Bergsland, N.; Hussein, S.; et al. Environmental Factors Associated with Disease Progression after the First Demyelinating Event: Results from the Multi-Center SET Study. PLoS ONE 2013, 8, e53996. [Google Scholar] [CrossRef] [PubMed]
- Smyk, D.S.; Alexander, A.K.; Walker, M.; Walker, M. Acute Disseminated Encephalomyelitis Progressing to Multiple Sclerosis: Are Infectious Triggers Involved? Immunol. Res. 2014, 60, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Laroni, A.; Armentani, E.; Kerlero de Rosbo, N.; Ivaldi, F.; Marcenaro, E.; Sivori, S.; Gandhi, R.; Weiner, H.L.; Moretta, A.; Mancardi, G.L.; et al. Dysregulation of Regulatory CD56bright NK Cells/T Cells Interactions in Multiple Sclerosis. J. Autoimmun. 2016, 72, 8–18. [Google Scholar] [CrossRef]
- Canto-Gomes, J.; Da Silva-Ferreira, S.; Silva, C.S.; Boleixa, D.; Martins da Silva, A.; González-Suárez, I.; Cerqueira, J.J.; Correia-Neves, M.; Nobrega, C. People with Primary Progressive Multiple Sclerosis Have a Lower Number of Central Memory T Cells and HLA-DR+ Tregs. Cells 2023, 12, 439. [Google Scholar] [CrossRef] [PubMed]
- van Langelaar, J.; Rijvers, L.; Smolders, J.; van Luijn, M.M. B and T Cells Driving Multiple Sclerosis: Identity, Mechanisms and Potential Triggers. Front. Immunol. 2020, 11, 760. [Google Scholar] [CrossRef] [PubMed]
- Filaci, G.; Bacchetta, R.; Zanetti, M. Is There a Role for NK Cells in the Pathogenesis of Multiple Sclerosis? A Case Study. Hum. Immunol. 1999, 60, 231–238. [Google Scholar] [CrossRef] [PubMed]
- Long, E.O.; Kim, H.S.; Liu, D.; Peterson, M.E.; Rajagopalan, S. Controlling NK Cell Responses: Integration of Signals for Activation and Inhibition. Annu. Rev. Immunol. 2013, 31, 227–258. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, R.; Laroni, A.; Weiner, H.L. Role of the Innate Immune System in the Pathogenesis of Multiple Sclerosis. J. Neuroimmunol. 2010, 221, 7–14. [Google Scholar] [CrossRef]
- Dendrou, C.A.; Petersen, J.; Rossjohn, J.; Fugger, L. HLA Variation and Disease. Nat. Rev. Immunol. 2018, 18, 325–339. [Google Scholar] [CrossRef] [PubMed]
- Kaur, G.; Trowsdale, J.; Fugger, L. Natural Killer Cells and Their Receptors in Multiple Sclerosis. Brain 2013, 136, 2657–2676. [Google Scholar] [CrossRef] [PubMed]
- Lanier, L.L. Evolutionary Struggles between NK Cells and Viruses. Nat. Rev. Immunol. 2008, 8, 259–268. [Google Scholar] [CrossRef] [PubMed]
- Durrenberger, P.F.; Webb, L.V.; Sim, M.J.W.; Nicholas, R.S.; Altmann, D.M.; Boyton, R.J. Increased HLA-E Expression in White Matter Lesions in Multiple Sclerosis. Immunology 2012, 137, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Höftberger, R.; Aboul-Enein, F.; Brueck, W.; Lucchinetti, C.; Rodriguez, M.; Schmidbauer, M.; Jellinger, K.; Lassmann, H. Expression of Major Histocompatibility Complex Class I Molecules on the Different Cell Types in Multiple Sclerosis Lesions. Brain Pathol. 2004, 14, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Abel, A.M.; Yang, C.; Thakar, M.S.; Malarkannan, S. Natural Killer Cells: Development, Maturation, and Clinical Utilization. Front. Immunol. 2018, 9, 1869. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, C.M.; White, M.J.; Goodier, M.R.; Riley, E.M. Functional Significance of CD57 Expression on Human NK Cells and Relevance to Disease. Front. Immunol. 2013, 4, 422. [Google Scholar] [CrossRef] [PubMed]
- Foley, B.; Cooley, S.; Verneris, M.R.; Pitt, M.; Curtsinger, J.; Luo, X.; Lopez-Vergès, S.; Lanier, L.L.; Weisdorf, D.; Miller, J.S. Cytomegalovirus Reactivation after Allogeneic Transplantation Promotes a Lasting Increase in Educated NKG2C+ Natural Killer Cells with Potent Function. Blood 2012, 119, 2665–2674. [Google Scholar] [CrossRef]
- Strioga, M.; Pasukoniene, V.; Characiejus, D. CD8+ CD28- and CD8+ CD57+ T Cells and Their Role in Health and Disease. Immunology 2011, 134, 17–32. [Google Scholar] [CrossRef]
- Akbar, A.N.; Henson, S.M.; Lanna, A. Senescence of T Lymphocytes: Implications for Enhancing Human Immunity. Trends Immunol. 2016, 37, 866–876. [Google Scholar] [CrossRef]
- Iglesias-Escudero, M.; Moro-García, M.A.; Marcos-Fernández, R.; García-Torre, A.; Álvarez-Argüelles, M.E.; Suárez-Fernández, M.L.; Martínez-Camblor, P.; Rodríguez, M.; Alonso-Arias, R. Levels of Anti-CMV Antibodies Are Modulated by the Frequency and Intensity of Virus Reactivations in Kidney Transplant Patients. PLoS ONE 2018, 13, e0194789. [Google Scholar] [CrossRef] [PubMed]
- Krijgsman, D.; Hokland, M.; Kuppen, P.J.K. The Role of Natural Killer T Cells in Cancer-A Phenotypical and Functional Approach. Front. Immunol. 2018, 9, 367. [Google Scholar] [CrossRef] [PubMed]
- Johnson, T.A.; Evans, B.L.; Durafourt, B.A.; Blain, M.; Lapierre, Y.; Bar-Or, A.; Antel, J.P. Reduction of the Peripheral Blood CD56bright NK Lymphocyte Subset in FTY720-Treated Multiple Sclerosis Patients. J. Immunol. 2011, 187, 570–579. [Google Scholar] [CrossRef] [PubMed]
- Thanapati, S.; Das, R.; Tripathy, A.S. Phenotypic and Functional Analyses of NK and NKT-like Populations during the Early Stages of Chikungunya Infection. Front. Microbiol. 2015, 6, 895. [Google Scholar] [CrossRef]
- Ho, L.-P.; Denney, L.; Luhn, K.; Teoh, D.; Clelland, C.; McMichael, A.J. Activation of Invariant NKT Cells Enhances the Innate Immune Response and Improves the Disease Course in Influenza A Virus Infection. Eur. J. Immunol. 2008, 38, 1913–1922. [Google Scholar] [CrossRef] [PubMed]
- van Dommelen, S.L.H.; Tabarias, H.A.; Smyth, M.J.; Degli-Esposti, M.A. Activation of Natural Killer (NK) T Cells during Murine Cytomegalovirus Infection Enhances the Antiviral Response Mediated by NK Cells. J. Virol. 2003, 77, 1877–1884. [Google Scholar] [CrossRef] [PubMed]
- McKay, F.C.; Swain, L.I.; Schibeci, S.D.; Rubio, J.P.; Kilpatrick, T.J.; Heard, R.N.; Stewart, G.J.; Booth, D.R. CD127 Immunophenotyping Suggests Altered CD4+ T Cell Regulation in Primary Progressive Multiple Sclerosis. J. Autoimmun. 2008, 31, 52–58. [Google Scholar] [CrossRef]


| MS | HDs | |
|---|---|---|
| Characteristics | ||
| Total | 74 | 18 |
| Gender, n (%) | ||
| Male, n (%) | 37 (50) | 7 (39) |
| Female, n (%) | 37 (50) | 11 (61) |
| Age, median (IQR) | 51 (43–58) | 52 (38–59) |
| Disease duration, median (IQR) | 11 (6–19) | - |
| EDSS, median (IQR) | 5.0 (3.0–6.0) | - |
| CMV serostatus, n (%) | ||
| IgG+/IgM−, n (%) | 51 (69) | 13 (73) |
| IgG−/IgM−, n (%) | 23 (31) | 5 (27) |
| IgG+/IgM+, n (%) | 0 | 0 |
| CMV DNA, n (%) | ||
| Positive, n (%) | 12 (16) | 0 |
| Negative, n (%) | 62 (84) | 0 |
| Anti CMV-IgG | p a | CMV DNA | p b | |||
|---|---|---|---|---|---|---|
| Positive | Negative | Positive | Negative | |||
| Total MS, n (%) | 51 (69) | 23 (31) | - | 12 (16) | 62 (84) | - |
| Male, n (%) | 26 (70) | 11 (30) | - | 6 (16) | 31 (84) | - |
| Female, n (%) | 25 (67) | 12 (33) | - | 6 (16) | 31 (84) | - |
| Age, median, (IQR) | 52 (46–59) | 44 (35–54) | 0.02 | 53 (41–56) | 50 (43–58) | 0.91 |
| Disease duration, median (IQR) | 11 (6–18) | 8 (3–19) | 0.32 | 11 (8–24) | 11 (5–18) | 0.28 |
| EDSS, median (IQR) | 5.0 (3.5–6.0) | 5.0 (1.0–6.0) | 0.25 | 5.5 (2.0–6.5) | 5.0 (3.0–6.0) | 0.77 |
| MS (n = 29) | HDs (n = 18) | p a | |
|---|---|---|---|
| NK cells | 9.9 (7.4–13.9) | 10.1 (6.3–14.3) | 0.818 |
| CD56bright cells | 11.0 (7.0–15.9) | 9.5 (4.4–13.4) | 0.352 |
| CD56dim cells | 86.5 (79.9–91.2) | 85.9 (76.1–91.5) | 0.614 |
| CD57+ cells | 30.6 (19.7–39.3) | 29.1 (15.7–41.4) | 0.662 |
| NKG2C (MFI) | 409 (274–1304) | 318 (195.5–424) | 0.041 |
| NKT-like cells | 1.1 (0.5–2.9) | 1.0 (0.3–2.5) | 0.661 |
| T cells | 68.9 (62.3–71.3) | 75.3 (70.4–78.9) | 0.001 |
| CD8+ cells | 26.5 (20.7–40.7) | 29.2 (25.8–38.7) | 0.376 |
| Early-differentiated CD8+ cells | 16.8 (11.8–26.9) | 14.5 (8.01–32.1) | 0.592 |
| Late-differentiated CD8+ cells | 33.3 (19.4–47.9) | 26.3 (16.3–39.0) | 0.208 |
| MS (n = 29) | HDs (n = 18) | |||||
|---|---|---|---|---|---|---|
| CMV+ (n = 22) | CMV− (n = 7) | p a | CMV+ (n = 13) | CMV− (n = 5) | p b | |
| NK cells | 10.1 (8.2–14.9) | 7.2 (4.5–14.3) | 0.153 | 9.8 (6.3–13.9) | 10.3 (7.9–17.9) | 0.430 |
| CD56bright cells | 9.3 (6.8–13.9) | 13.5 (7.5–24.7) | 0.320 | 7.2 (4.4–11.8) | 12.10 (5.5–26.9) | 0.324 |
| CD56dim cells | 88.1 (82.1–91.1) | 85.8 (72.3–92) | 0.558 | 87.6 (80.5–92.6) | 80.3 (62.7–85.1) | 0.038 |
| CD57+ cells | 36.5 (26.2–43.1) | 20.0 (10.3–30.1) | 0.020 | 37.1 (24.2–43.6) | 16.6 (8.6–19.8) | 0.018 |
| NKG2C (MFI) | 600.5 (375.5–1597) | 197 (0–399) | 0.008 | 320 (210–750) | 224 (176–321) | 0.278 |
| NKT-like cells | 1.6 (0.8–3.7) | 0.5 (0.2–0.6) | 0.008 | 1.1 (0.3–2.3) | 0.8 (0.3–4.1) | 1.000 |
| T cells (%) | 69.5 (62.8–71.7) | 68.9 (61.2–84.1) | 0.939 | 73.8 (71.9–78.1) | 76.8 (63.4–82.4) | 1.000 |
| CD3+CD8+ cells | 27.0 (21.1–41.3) | 22.8 (14.2–28.1) | 0.101 | 34.0 (27.4–40.3) | 20.7 (19.3–28.1) | 0.014 |
| Early-differentiated CD8+ cells | 14.7 (9.43–20.6) | 30.1 (18.2–36.3) | 0.023 | 14.1 (8.2–21.4) | 35.2 (7.1–42.7) | 0.375 |
| Late-differentiated CD8+ cells | 39.5 (25.8–49.2) | 11.2 (6.5–27.9) | 0.002 | 31.6 (21.3–42.7) | 16.7 (7.6–17.6) | 0.023 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perri, V.; Zingaropoli, M.A.; Pasculli, P.; Ciccone, F.; Tartaglia, M.; Baione, V.; Malimpensa, L.; Ferrazzano, G.; Mastroianni, C.M.; Conte, A.; et al. The Impact of Cytomegalovirus Infection on Natural Killer and CD8+ T Cell Phenotype in Multiple Sclerosis. Biology 2024, 13, 154. https://doi.org/10.3390/biology13030154
Perri V, Zingaropoli MA, Pasculli P, Ciccone F, Tartaglia M, Baione V, Malimpensa L, Ferrazzano G, Mastroianni CM, Conte A, et al. The Impact of Cytomegalovirus Infection on Natural Killer and CD8+ T Cell Phenotype in Multiple Sclerosis. Biology. 2024; 13(3):154. https://doi.org/10.3390/biology13030154
Chicago/Turabian StylePerri, Valentina, Maria Antonella Zingaropoli, Patrizia Pasculli, Federica Ciccone, Matteo Tartaglia, Viola Baione, Leonardo Malimpensa, Gina Ferrazzano, Claudio Maria Mastroianni, Antonella Conte, and et al. 2024. "The Impact of Cytomegalovirus Infection on Natural Killer and CD8+ T Cell Phenotype in Multiple Sclerosis" Biology 13, no. 3: 154. https://doi.org/10.3390/biology13030154
APA StylePerri, V., Zingaropoli, M. A., Pasculli, P., Ciccone, F., Tartaglia, M., Baione, V., Malimpensa, L., Ferrazzano, G., Mastroianni, C. M., Conte, A., & Ciardi, M. R. (2024). The Impact of Cytomegalovirus Infection on Natural Killer and CD8+ T Cell Phenotype in Multiple Sclerosis. Biology, 13(3), 154. https://doi.org/10.3390/biology13030154







