Effects of Noise Exposure and Ageing on Anxiety and Social Behaviour in Zebrafish
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Test Animals and Husbandry
2.2. Noise Treatments
2.3. Behavioural Tests
2.4. Novel Tank Diving (NTD) Test
2.5. Mirror/Conspecific Interaction Test
2.6. Shoaling Test
2.7. Data Analysis
3. Results
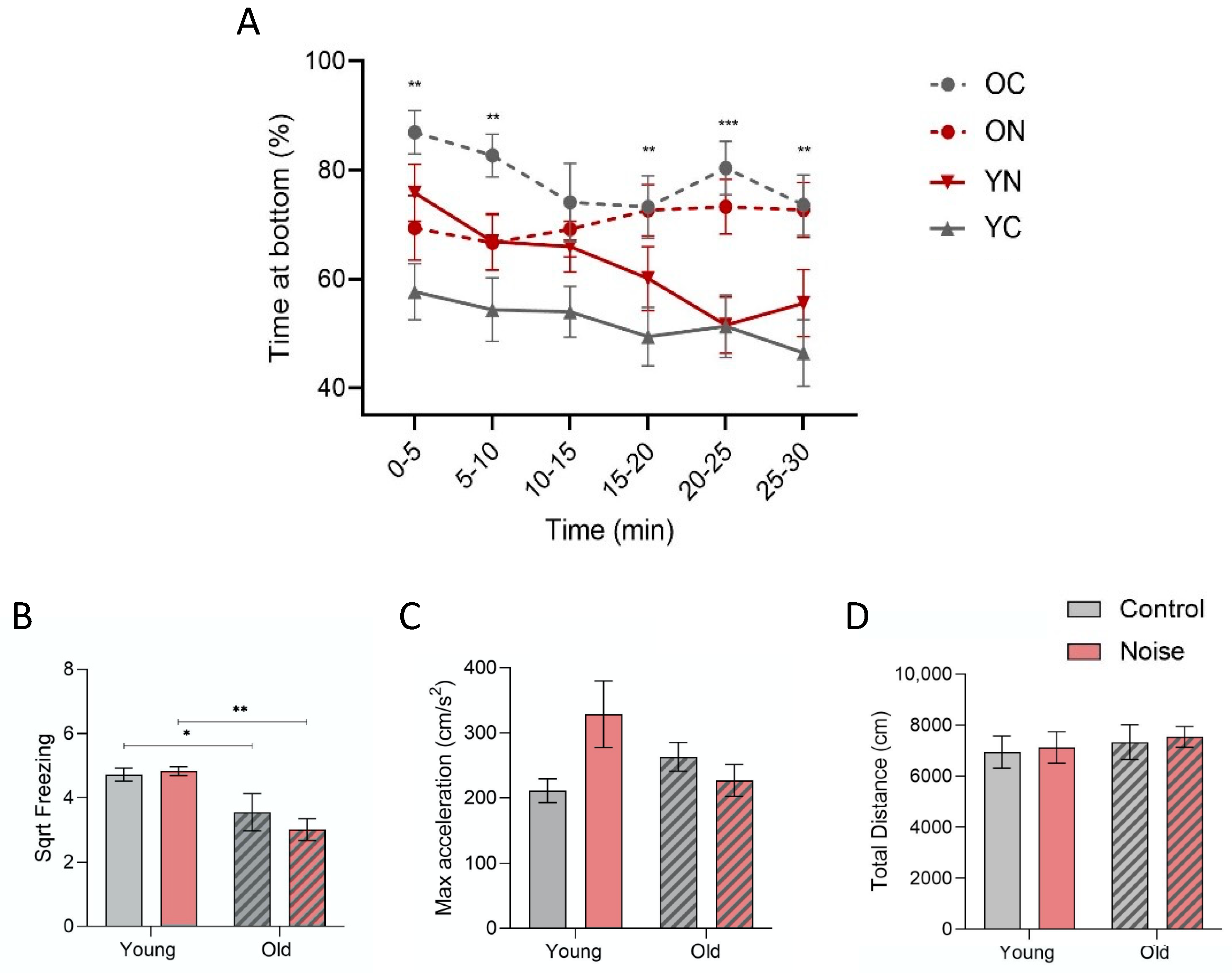
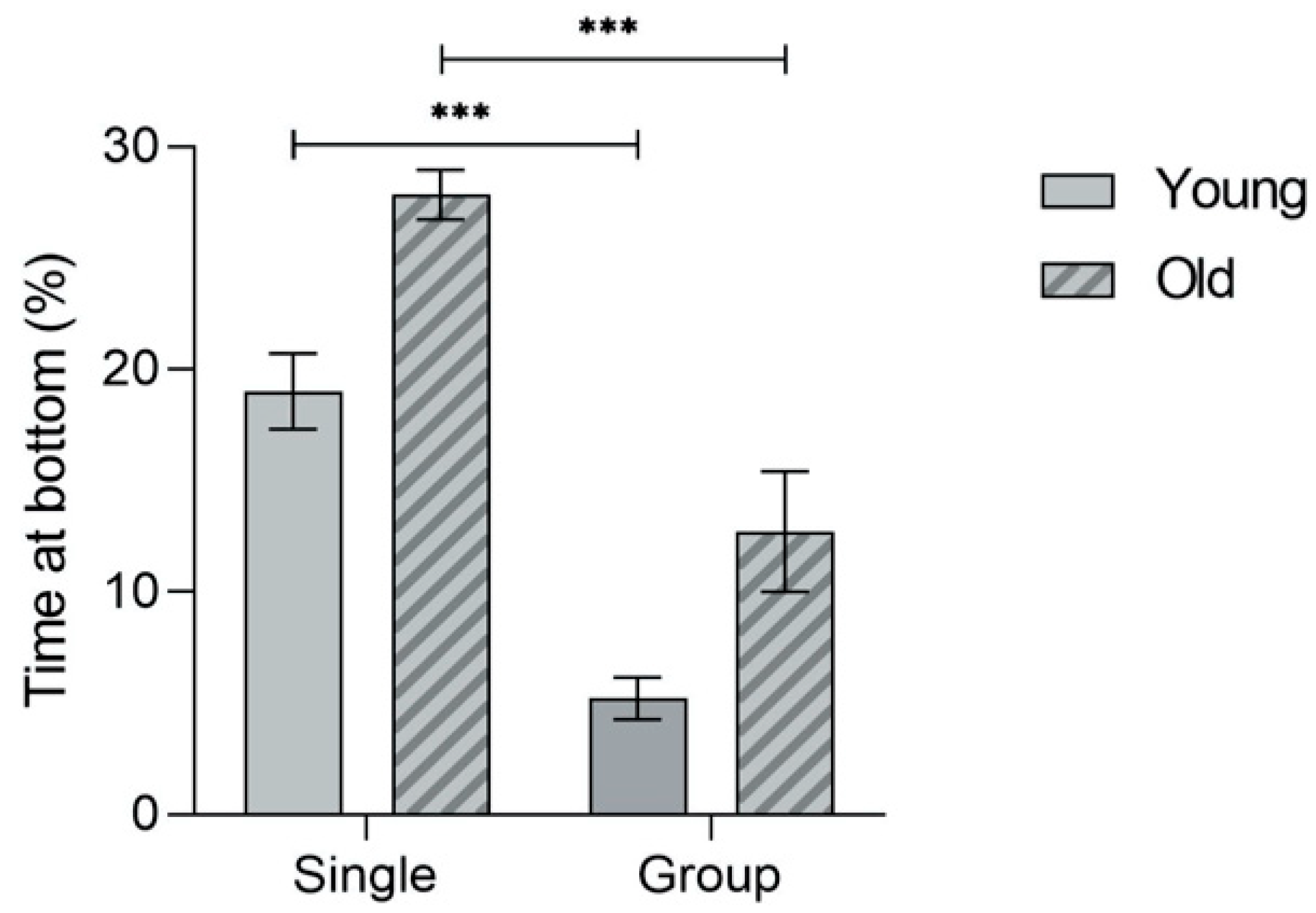
3.1. Anxiety-Related Novel Tank Diving
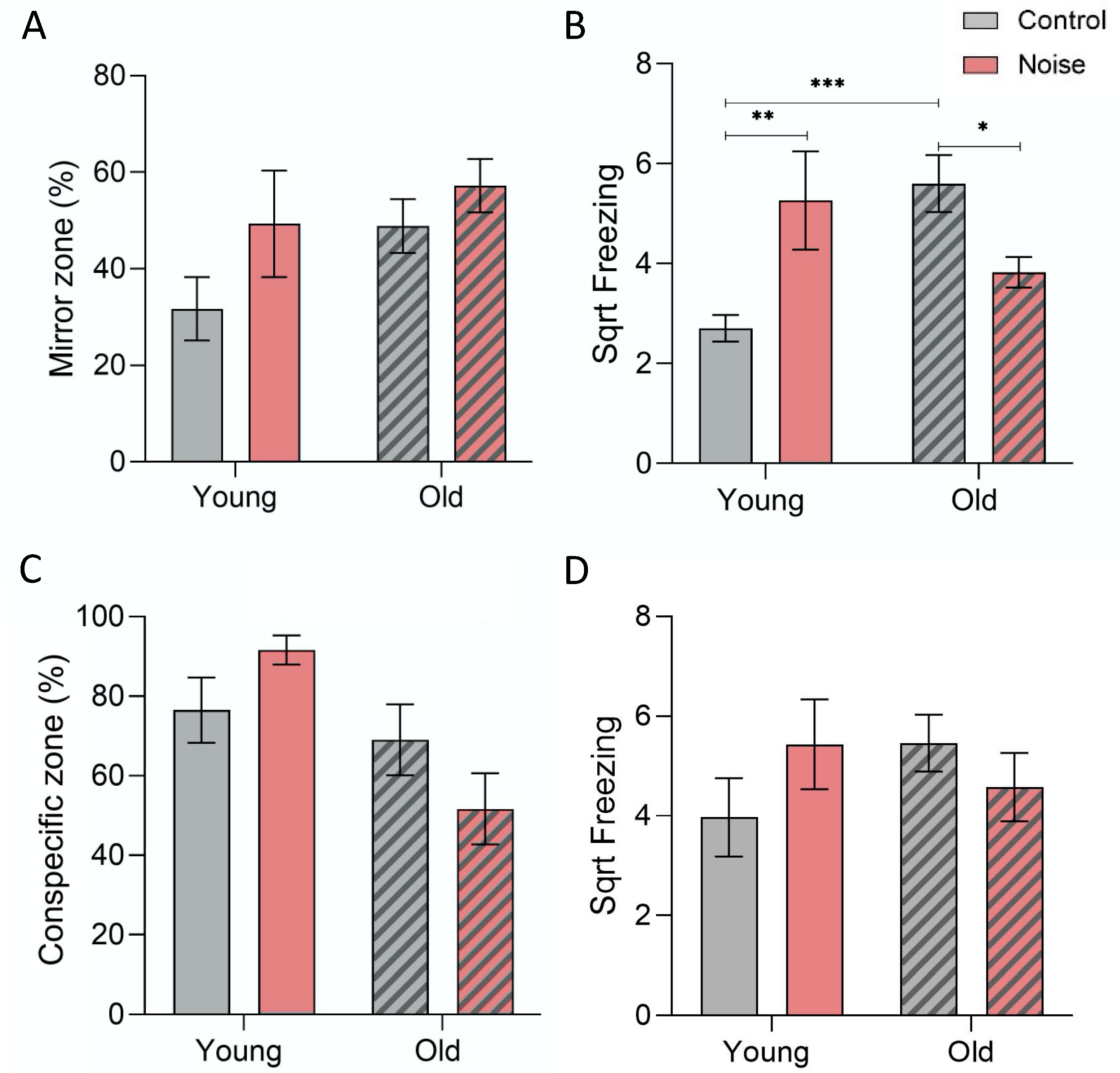
3.2. Mirror/Conspecific Interaction
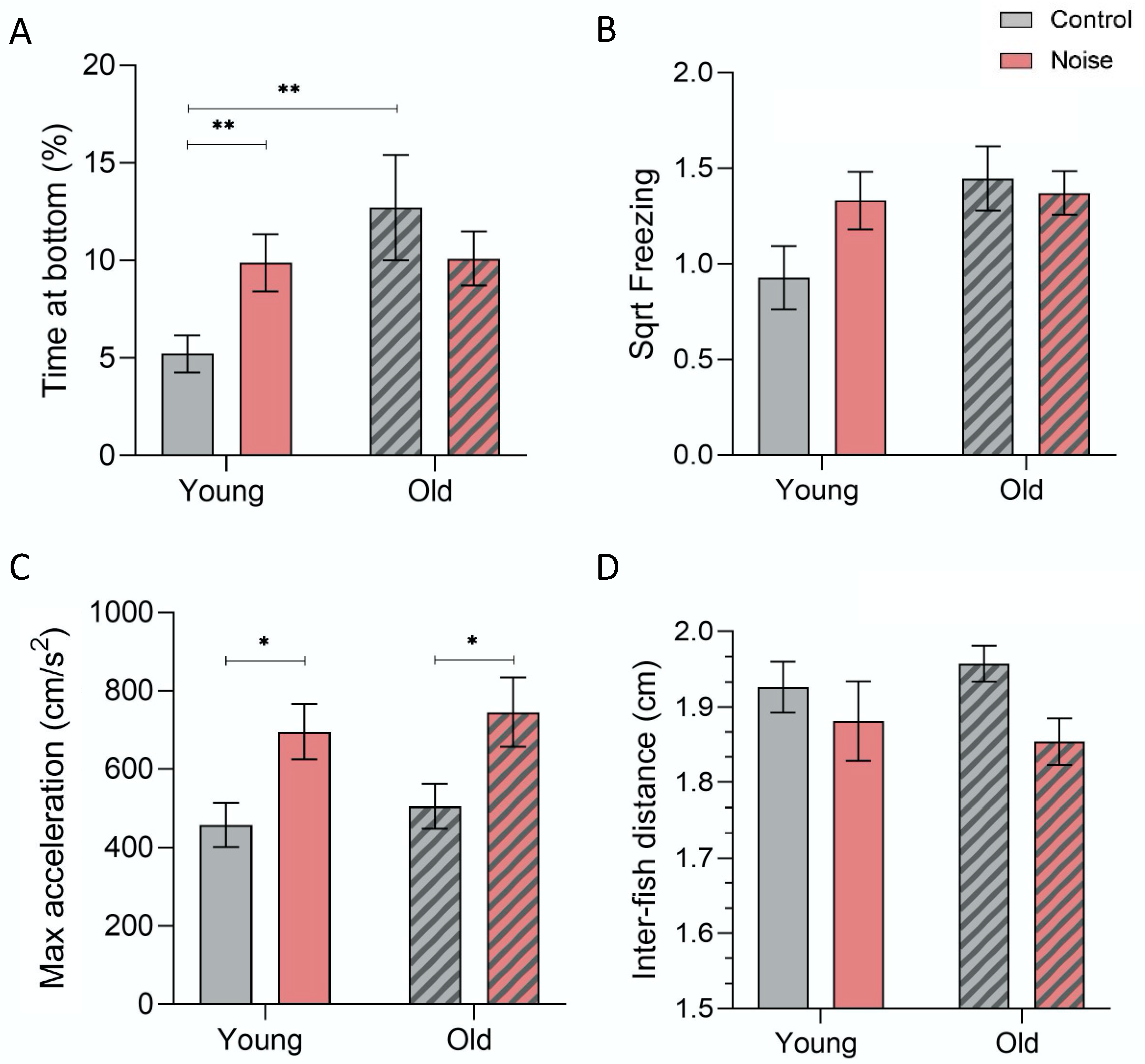
3.3. Shoaling Behaviour
4. Discussion
4.1. Effects of Noise on Anxiety and Social Behaviour
4.2. Effects of Ageing on Anxiety and Social Behaviour
4.3. Interaction between Ageing and Noise Exposure Effects
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Merilä, J.; Hendry, A.P. Climate change, adaptation, and phenotypic plasticity: The problem and the evidence. Evol. Appl. 2014, 7, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Radchuk, V.; Reed, T.; Teplitsky, C.; van de Pol, M.; Charmantier, A.; Hassall, C.; Adamík, P.; Adriaensen, F.; Ahola, M.P.; Arcese, P.; et al. Adaptive responses of animals to climate change are most likely insufficient. Nat. Commun. 2019, 10, 3109. [Google Scholar] [CrossRef] [PubMed]
- Ventriglio, A.; Bellomo, A.; di Gioia, I.; Di Sabatino, D.; Favale, D.; De Berardis, D.; Cianconi, P. Environmental pollution and mental health: A narrative review of literature. CNS Spectr. 2021, 26, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Münzel, T.; Daiber, A.; Steven, S.; Tran, L.P.; Ullmann, E.; Kossmann, S.; Schmidt, F.P.; Oelze, M.; Xia, N.; Li, H.; et al. Effects of noise on vascular function, oxidative stress, and inflammation: Mechanistic insight from studies in mice. Eur. Heart J. 2017, 38, 2838–2849. [Google Scholar] [CrossRef]
- Koczorowski, M.; Bernard, N.; Mauny, F.; Chagué, F.; Pujol, S.; Maza, M.; Cottin, Y.; Zeller, M.; ENVI-MI Study Group. Environmental noise exposure is associated with atherothrombotic risk. Sci. Rep. 2022, 12, 3151. [Google Scholar] [CrossRef] [PubMed]
- Röösli, M.; Mohler, E.; Frei, P.; Vienneau, D. Noise-related sleep disturbances: Does gender matter? Noise Health 2014, 16, 197–204. [Google Scholar] [CrossRef]
- Cui, B.; Gai, Z.; She, X.; Wang, R.; Xi, Z. Effects of chronic noise on glucose metabolism and gut microbiota-host inflammatory homeostasis in rats. Sci. Rep. 2016, 6, 36693. [Google Scholar] [CrossRef]
- Stansfeld, S.A.; Berglund, B.; Clark, C.; Lopez-Barrio, I.; Fischer, P.; Ohrström, E.; Haines, M.M.; Head, J.; Hygge, S.; van Kamp, I.; et al. Aircraft and road traffic noise and children’s cognition and health: A cross-national study. Lancet 2005, 365, 1942–1949. [Google Scholar] [CrossRef]
- Kight, C.R.; Swaddle, J.P. How and why environmental noise impacts animals: An integrative, mechanistic review. Ecol. Lett. 2011, 14, 1052–1061. [Google Scholar] [CrossRef]
- Clark, C.; Paunovic, K. WHO Environmental Noise Guidelines for the European Region: A Systematic Review on Environmental Noise and Quality of Life, Wellbeing and Mental Health. IJERPH 2018, 15, 2400. [Google Scholar] [CrossRef]
- Beutel, M.E.; Jünger, C.; Klein, E.M.; Wild, P.; Lackner, K.; Blettner, M.; Binder, H.; Michal, M.; Wiltink, J.; Brähler, E.; et al. Noise Annoyance Is Associated with Depression and Anxiety in the General Population- The Contribution of Aircraft Noise. PLoS ONE 2016, 11, e0155357. [Google Scholar] [CrossRef] [PubMed]
- Erbe, C.; Dent, M.L.; Gannon, W.L.; McCauley, R.D.; Römer, H.; Southall, B.L.; Stansbury, A.L.; Stoeger, A.S.; Thomas, J.A. The Effects of Noise on Animals. In Exploring Animal Behavior through Sound; Erbe, C., Thomas, J.A., Eds.; Springer International Publishing: Cham, Switzerland, 2022; Volume 1, pp. 459–506. [Google Scholar]
- Shannon, G.; McKenna, M.F.; Angeloni, L.M.; Crooks, K.R.; Fristrup, K.M.; Brown, E.; Warner, K.A.; Nelson, M.D.; White, C.; Briggs, J.; et al. A synthesis of two decades of research documenting the effects of noise on wildlife. Biol. Rev. Camb. Philos. Soc. 2016, 91, 982–1005. [Google Scholar] [CrossRef] [PubMed]
- Duarte, C.M.; Chapuis, L.; Collin, S.P.; Costa, D.P.; Devassy, R.P.; Eguiluz, V.M.; Erbe, C.; Gordon, T.A.C.; Halpern, B.S.; Harding, H.R.; et al. The soundscape of the Anthropocene ocean. Science 2021, 371, eaba4658. [Google Scholar] [CrossRef] [PubMed]
- Braga Goncalves, I.; Richmond, E.; Harding, H.R.; Radford, A.N. Impacts of additional noise on the social interactions of a cooperatively breeding fish. R. Soc. Open Sci. 2021, 8, 210982. [Google Scholar] [CrossRef]
- Herbert-Read, J.E.; Kremer, L.; Bruintjes, R.; Radford, A.N.; Ioannou, C.C. Anthropogenic noise pollution from pile-driving disrupts the structure and dynamics of fish shoals. Proc. Biol. Sci. 2017, 284, 20171627. [Google Scholar] [CrossRef]
- Tidau, S.; Briffa, M. Anthropogenic noise pollution reverses grouping behaviour in hermit crabs. Anim. Behav. 2019, 151, 113–120. [Google Scholar] [CrossRef]
- Butler, J.M.; Maruska, K.P. Underwater noise impairs social communication during aggressive and reproductive encounters. Anim. Behav. 2020, 164, 9–23. [Google Scholar] [CrossRef]
- Henríquez Martínez, A.; Ávila, L.C.; Pulido, M.A.; Ardila, Y.A.; Akle, V.; Bloch, N.I. Age-Dependent Effects of Chronic Stress on Zebrafish Behavior and Regeneration. Front. Physiol. 2022, 13, 856778. [Google Scholar] [CrossRef]
- Burke, K.; Screven, L.A.; Kobrina, A.; Charlton, P.E.; Schrode, K.; Villavisanis, D.F.; Dent, M.L.; Lauer, A.M. Effects of Noise Exposure and Aging on Behavioral Tone Detection in Quiet and Noise by Mice. eNeuro 2022, 9, 1–15. [Google Scholar] [CrossRef]
- López-Otín, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. Hallmarks of aging: An expanding universe. Cell 2023, 186, 243–278. [Google Scholar] [CrossRef]
- Schmiedt, R.A. The Physiology of Cochlear Presbycusis. In The Aging Auditory System. Springer Handbook of Auditory Research; Gordon-Salant, S., Frisina, R., Popper, A., Fay, R., Eds.; Springer: New York, NY, USA, 2010; Volume 34. [Google Scholar] [CrossRef]
- Kujawa, S.G.; Liberman, M.C. Synaptopathy in the noise-exposed and aging cochlea: Primary neural degeneration in acquired sensorineural hearing loss. Hear. Res. 2015, 330, 191–199. [Google Scholar] [CrossRef]
- Kujawa, S.G.; Liberman, M.C. Translating animal models to human therapeutics in noise-induced and age-related hearing loss. Hear. Res. 2019, 377, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Shehabi, A.M.; Prendergast, G.; Plack, C.J. The Relative and Combined Effects of Noise Exposure and Aging on Auditory Peripheral Neural Deafferentation: A Narrative Review. Front. Aging Neurosci. 2022, 14, 877588. [Google Scholar] [CrossRef] [PubMed]
- Sergeyenko, Y.; Lall, K.; Liberman, M.C.; Kujawa, S.G. Age-related cochlear synaptopathy: An early-onset contributor to auditory functional decline. J. Neurosci. 2013, 33, 13686–13694. [Google Scholar] [CrossRef]
- Balogová, Z.; Popelář, J.; Chiumenti, F.; Chumak, T.; Burianová, J.S.; Rybalko, N.; Syka, J. Age-Related Differences in Hearing Function and Cochlear Morphology between Male and Female Fischer 344 Rats. Front. Aging Neurosci. 2017, 9, 428. [Google Scholar] [CrossRef] [PubMed]
- Cai, R.; Montgomery, S.C.; Graves, K.A.; Caspary, D.M.; Cox, B.C. The FBN rat model of aging: Investigation of ABR waveforms and ribbon synapse changes. Neurobiol. Aging 2017, 62, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Fuentes-Santamaría, V.; Alvarado, J.C.; Mellado, S.; Melgar-Rojas, P.; Gabaldón-Ull, M.C.; Cabanes-Sanchis, J.J.; Juiz, J.M. Age-Related Inflammation and Oxidative Stress in the Cochlea Are Exacerbated by Long-Term, Short-Duration Noise Stimulation. Front. Aging Neurosci. 2022, 14, 853320. [Google Scholar] [CrossRef]
- Kalueff, A.V.; Stewart, A.M.; Gerlai, R. Zebrafish as an emerging model for studying complex brain disorders. Trends Pharmacol. Sci. 2014, 35, 63–75. [Google Scholar] [CrossRef]
- de Abreu, M.S.; Demin, K.A.; Giacomini, A.C.V.V.; Amstislavskaya, T.G.; Strekalova, T.; Maslov, G.O.; Kositsin, Y.; Petersen, E.V.; Kalueff, A.V. Understanding how stress responses and stress-related behaviors have evolved in zebrafish and mammals. Neurobiol. Stress. 2021, 15, 100405. [Google Scholar] [CrossRef]
- Lara, R.A.; Vasconcelos, R.O. Impact of noise on development, physiological stress and behavioural patterns in larval zebrafish. Sci. Rep. 2021, 11, 6615. [Google Scholar] [CrossRef]
- Popper, A.N.; Sisneros, J.A. The Sound World of Zebrafish: A Critical Review of Hearing Assessment. Zebrafish 2022, 19, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Henriques, C.M.; Ferreira, M.G. Consequences of telomere shortening during lifespan. Curr. Opin. Cell Biol. 2012, 24, 804–808. [Google Scholar] [CrossRef] [PubMed]
- Carneiro, M.C.; de Castro, I.P.; Ferreira, M.G. Telomeres in aging and disease: Lessons from zebrafish. Dis. Model. Mech. 2016, 9, 737–748. [Google Scholar] [CrossRef] [PubMed]
- Cayuela, M.L.; Claes, K.B.M.; Ferreira, M.G.; Henriques, C.M.; van Eeden, F.; Varga, M.; Vierstraete, J.; Mione, M.C. The Zebrafish as an Emerging Model to Study DNA Damage in Aging, Cancer and Other Diseases. Front. Cell Dev. Biol. 2018, 6, 178. [Google Scholar] [CrossRef]
- Espigares, F.; Abad-Tortosa, D.; Varela, S.A.M.; Ferreira, M.G.; Oliveira, R.F. Short telomeres drive pessimistic judgement bias in zebrafish. Biol. Lett. 2021, 17, 20200745. [Google Scholar] [CrossRef]
- Breitzler, L.; Lau, I.H.; Fonseca, P.J.; Vasconcelos, R.O. Noise-induced hearing loss in zebrafish: Investigating structural and functional inner ear damage and recovery. Hear. Res. 2020, 391, 107952. [Google Scholar] [CrossRef]
- Lara, R.A.; Breitzler, L.; Lau, I.H.; Gordillo-Martinez, F.; Chen, F.; Fonseca, P.J.; Bass, A.H.; Vasconcelos, R.O. Noise-induced hearing loss correlates with inner ear hair cell decrease in larval zebrafish. J. Exp. Biol. 2022, 225, jeb243743. [Google Scholar] [CrossRef]
- Wong, M.I.; Lau, I.H.; Gordillo-Martinez, F.; Vasconcelos, R.O. The effect of time regime in noise exposure on the auditory system and behavioural stress in the zebrafish. Sci. Rep. 2022, 12, 15353. [Google Scholar] [CrossRef]
- Lara, R.A.; Vasconcelos, R.O. Characterization of the Natural Soundscape of Zebrafish and Comparison with the Captive Noise Conditions. Zebrafish 2019, 16, 152–164. [Google Scholar] [CrossRef]
- Audira, G.; Sampurna, B.P.; Juniardi, S.; Liang, S.-T.; Lai, Y.-H.; Hsiao, C.-D. A Versatile Setup for Measuring Multiple Behavior Endpoints in Zebrafish. Inventions 2018, 3, 75. [Google Scholar] [CrossRef]
- Cachat, J.; Stewart, A.; Grossman, L.; Gaikwad, S.; Kadri, F.; Chung, K.M.; Wu, N.; Wong, K.; Roy, S.; Suciu, C.; et al. Measuring behavioral and endocrine responses to novelty stress in adult zebrafish. Nat. Protoc. 2010, 5, 1786–1799. [Google Scholar] [CrossRef]
- Moretz, J.A.; Martins, E.P.; Robison, B.D. The effects of early and adult social environment on zebrafish (Danio rerio) behavior. Environ. Biol. Fishes 2007, 80, 91–101. [Google Scholar] [CrossRef]
- Teles, M.C.; Oliveira, R.F. Quantifying Aggressive Behavior in Zebrafish. Methods Mol. Biol. 2016, 1451, 293–305. [Google Scholar] [CrossRef] [PubMed]
- Engeszer, R.E.; Ryan, M.J.; Parichy, D.M. Learned social preference in zebrafish. Curr. Biol. 2004, 14, 881–884. [Google Scholar] [CrossRef]
- Pham, M.; Raymond, J.; Hester, J.; Kyzar, E.J.; Gaikwad, S.; Bruce, I.; Fryar, C.M.; Chanin, S.; Enriquez, J.; Bagawandoss, S.; et al. Assessing Social Behavior Phenotypes in Adult Zebrafish: Shoaling, Social Preference, and Mirror Biting Tests. In Zebrafish Protocols for Neurobehavioral Research; Kalueff, A.V., Stewart, A.M., Eds.; Humana Press: Totowa, NJ, USA, 2012; pp. 231–246. [Google Scholar] [CrossRef]
- Buske, C.; Gerlai, R. Shoaling develops with age in Zebrafish (Danio rerio). Prog. Neuropsychopharmacol. Biol. Psychiatry 2011, 35, 1409–1415. [Google Scholar] [CrossRef]
- Haghani, S.; Karia, M.; Cheng, R.K.; Mathuru, A.S. An automated assay system to study novel tank induced anxiety. Front. Behav. Neurosci. 2019, 13, 180. [Google Scholar] [CrossRef]
- Prut, L.; Belzung, C. The open field as a paradigm to measure the effects of drugs on anxiety-like behaviors: A review. Eur. J. Pharmacol. 2003, 463, 3–33. [Google Scholar] [CrossRef]
- Tran, S.; Gerlai, T.R. The novel tank test: Handling stress and the context specific psychopharmacology of anxiety. Curr. Psychopharmacol. 2016, 5, 169–179. [Google Scholar] [CrossRef]
- Gusso, D.; Altenhofen, S.; Fritsch, P.M.; Rübensam, G.; Bonan, C.D. Oxytetracycline induces anxiety-like behavior in adult zebrafish. Toxicol. Appl. Pharmacol. 2021, 426, 115616. [Google Scholar] [CrossRef]
- Pagnussat, N.; Piato, A.L.; Schaefer, I.C.; Blank, M.; Tamborski, A.R.; Guerim, L.D.; Bonan, C.D.; Vianna, M.R.; Lara, D.R. One for all and all for one: The importance of shoaling on behavioral and stress responses in zebrafish. Zebrafish 2013, 10, 338–342. [Google Scholar] [CrossRef]
- Krause, J.; Ruxton, G.D. Living in Groups; Oxford University Press: Oxford, UK, 2002. [Google Scholar]
- Schmidel, A.J.; Assmann, K.L.; Werlang, C.C.; Bertoncello, K.T.; Francescon, F.; Rambo, C.L.; Beltrame, G.M.; Calegari, D.; Batista, C.B.; Blaser, R.E.; et al. Subchronic atrazine exposure changes defensive behaviour profile and disrupts brain acetylcholinesterase activity of zebrafish. Neurotoxicol. Teratol. 2014, 44, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Bruintjes, R.; Radford, A.N. Context-dependent impacts of anthropogenic noise on individual and social behaviour in a cooperatively breeding fish. Anim. Behav. 2013, 85, 1343–1349. [Google Scholar] [CrossRef]
- Currie, H.A.L.; White, P.R.; Leighton, T.G.; Kemp, P.S. Group behavior and tolerance of Eurasian minnow (Phoxinus phoxinus) in response to tones of differing pulse repetition rate. J. Acoust. Soc. Am. 2020, 147, 1709–1718. [Google Scholar] [CrossRef]
- Short, M.; White, P.R.; Leighton, T.G.; Kemp, P.S. Influence of acoustics on the collective behaviour of a shoaling freshwater fish. Fresh Biol. 2020, 65, 2186–2195. [Google Scholar] [CrossRef]
- Kovacs, G.G.; Adle-Biassette, H.; Milenkovic, I.; Cipriani, S.; van Scheppingen, J.; Aronica, E. Linking pathways in the developing and aging brain with neurodegeneration. Neuroscience 2014, 269, 152–172. [Google Scholar] [CrossRef] [PubMed]
- Vardy, Y. The Alzheimer conundrum: Entanglements of dementia and aging. Glob. Public Health 2015, 10, 132–133. [Google Scholar] [CrossRef] [PubMed]
- Van Houcke, J.; De Groef, L.; Dekeyster, E.; Moons, L. The zebrafish as a gerontology model in nervous system aging, disease, and repair. Ageing Res. Rev. 2015, 24 Pt. B, 358–368. [Google Scholar] [CrossRef]
- Philpott, C.; Donack, C.J.; Cousin, M.A.; Pierret, C. Reducing the noise in behavioral assays: Sex and age in adult zebrafish locomotion. Zebrafish 2012, 9, 191–194. [Google Scholar] [CrossRef]
- Gilbert, M.J.; Zerulla, T.C.; Tierney, K.B. Zebrafish (Danio rerio) as a model for the study of aging and exercise: Physical ability and trainability decrease with age. Exp. Gerontol. 2014, 50, 106–113. [Google Scholar] [CrossRef]
- Rosa, C.E.; Kuradomi, R.Y.; Almeida, D.V.; Lannes, C.F.; Figueiredo Mde, A.; Dytz, A.G.; Fonseca, D.B.; Marins, L.F. GH overexpression modifies muscle expression of anti-oxidant enzymes and increases spinal curvature of old zebrafish. Exp. Gerontol. 2010, 45, 449–456. [Google Scholar] [CrossRef]
- Kacprzak, V.; Patel, N.A.; Riley, E.; Yu, L.; Yeh, J.J.; Zhdanova, I.V. Dopaminergic control of anxiety in young and aged zebrafish. Pharmacol. Biochem. Behav. 2017, 157, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.R.; Torres-Pérez, J.V.; Miletto Petrazzini, M.E.; Riley, R.; Brennan, C.H. Stress reactivity elicits a tissue-specific reduction in telomere length in aging zebrafish (Danio rerio). Sci. Rep. 2021, 11, 339. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Tucci, V.; Kishi, S.; Zhdanova, I.V. Cognitive aging in zebrafish. PLoS ONE 2006, 1, e14. [Google Scholar] [CrossRef] [PubMed]
- Shoji, H.; Takao, K.; Hattori, S.; Miyakawa, T. Age-related Changes in Behavior in C57BL/6J Mice from Young Adulthood to Middle Age. Mol. Brain 2016, 9, 11. [Google Scholar] [CrossRef]
- Soravia, C.; Ashton, B.J.; Thornton, A.; Ridley, A.R. General cognitive performance declines with female age and is negatively related to fledging success in a wild bird. Proc. Biol. Sci. 2022, 289, 20221748. [Google Scholar] [CrossRef]
- Ebner, N.C.; Fischer, H.K. Emotion and Aging: Evidence from Brain and Behavior. Front. Psychol. 2014, 5, 996. [Google Scholar] [CrossRef]
- Novais, A.; Monteiro, S.; Roque, S.; Correia-Neves, M.; Sousa, N. How Age, Sex and Genotype Shape the Stress Response. Neurobiol. Stress 2017, 6, 44–56. [Google Scholar] [CrossRef]
- Zapater-Fajarí, M.; Crespo-Sanmiguel, I.; Pulopulos, M.M.; Hidalgo, V.; Salvador, A. Resilience and Psychobiological Response to Stress in Older People: The Mediating Role of Coping Strategies. Front. Aging Neurosci. 2021, 13, 632141. [Google Scholar] [CrossRef]
- de Miguel, Z.; Haditsch, U.; Palmer, T.D.; Azpiroz, A.; Sapolsky, R.M. Adult-generated Neurons Born during Chronic Social Stress Are Uniquely Adapted to Respond to Subsequent Chronic Social Stress. Mol. Psychiatry 2018, 24, 1178–1188. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vasconcelos, R.O.; Gordillo-Martinez, F.; Ramos, A.; Lau, I.H. Effects of Noise Exposure and Ageing on Anxiety and Social Behaviour in Zebrafish. Biology 2023, 12, 1165. https://doi.org/10.3390/biology12091165
Vasconcelos RO, Gordillo-Martinez F, Ramos A, Lau IH. Effects of Noise Exposure and Ageing on Anxiety and Social Behaviour in Zebrafish. Biology. 2023; 12(9):1165. https://doi.org/10.3390/biology12091165
Chicago/Turabian StyleVasconcelos, Raquel O., Flora Gordillo-Martinez, Andreia Ramos, and Ieng Hou Lau. 2023. "Effects of Noise Exposure and Ageing on Anxiety and Social Behaviour in Zebrafish" Biology 12, no. 9: 1165. https://doi.org/10.3390/biology12091165
APA StyleVasconcelos, R. O., Gordillo-Martinez, F., Ramos, A., & Lau, I. H. (2023). Effects of Noise Exposure and Ageing on Anxiety and Social Behaviour in Zebrafish. Biology, 12(9), 1165. https://doi.org/10.3390/biology12091165




