Blood Metabolites and Faecal Microbial Communities in Nonpregnant and Early Gestation Ewes in Highly Cold Areas
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
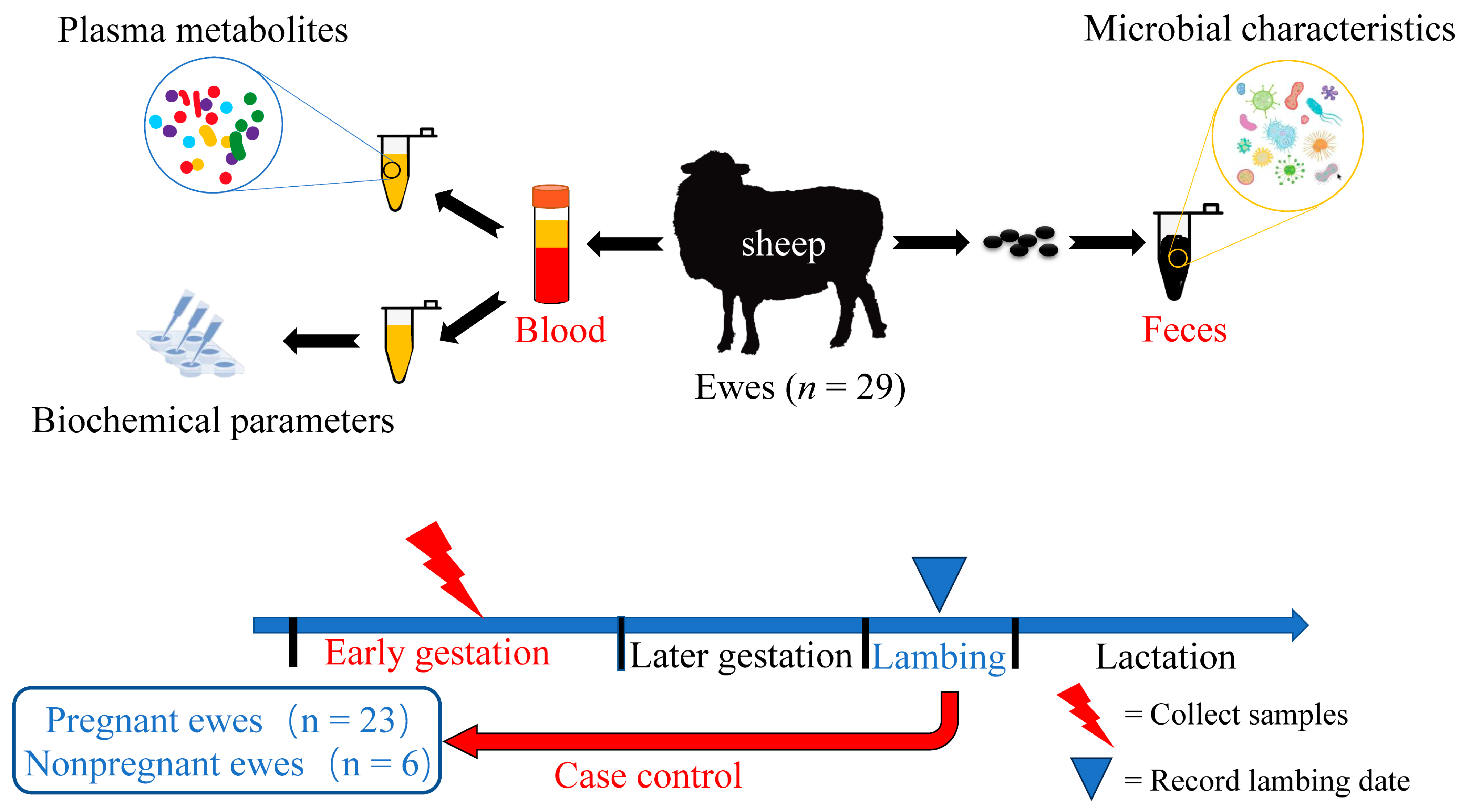
2.1. Animals
2.2. Sample Collection
2.2.1. Blood Sample Collection
2.2.2. Faecal Sample Collection
2.3. Measurement of Blood Biochemical Parameters, Mineral and Hormonal Concentrations, and Immune Parameters
2.4. Metabolomics Profiling of Sheep Plasma
2.4.1. Plasma Metabolite Extraction
2.4.2. Ultra-Performance Liquid Chromatography (UPLC)–Mass Spectrometry (MS) Experiments
2.4.3. Metabolome Data Analysis
2.5. 16S rDNA Amplicon Sequencing and Analysis
2.6. Statistical Analyses
3. Results
3.1. Plasma Biochemical Parameters, Mineral and Hormonal Concentrations, and Immune Parameters
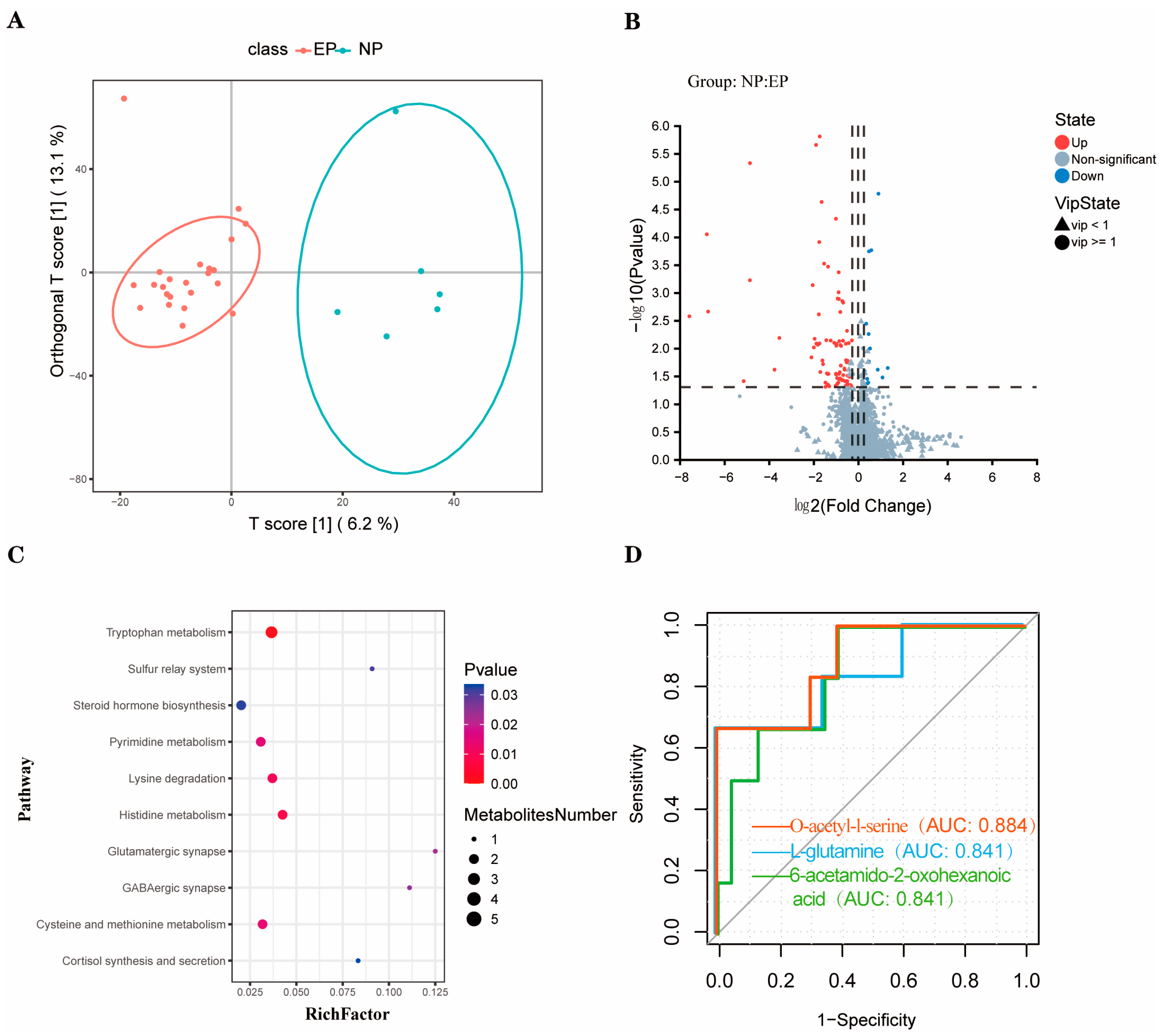
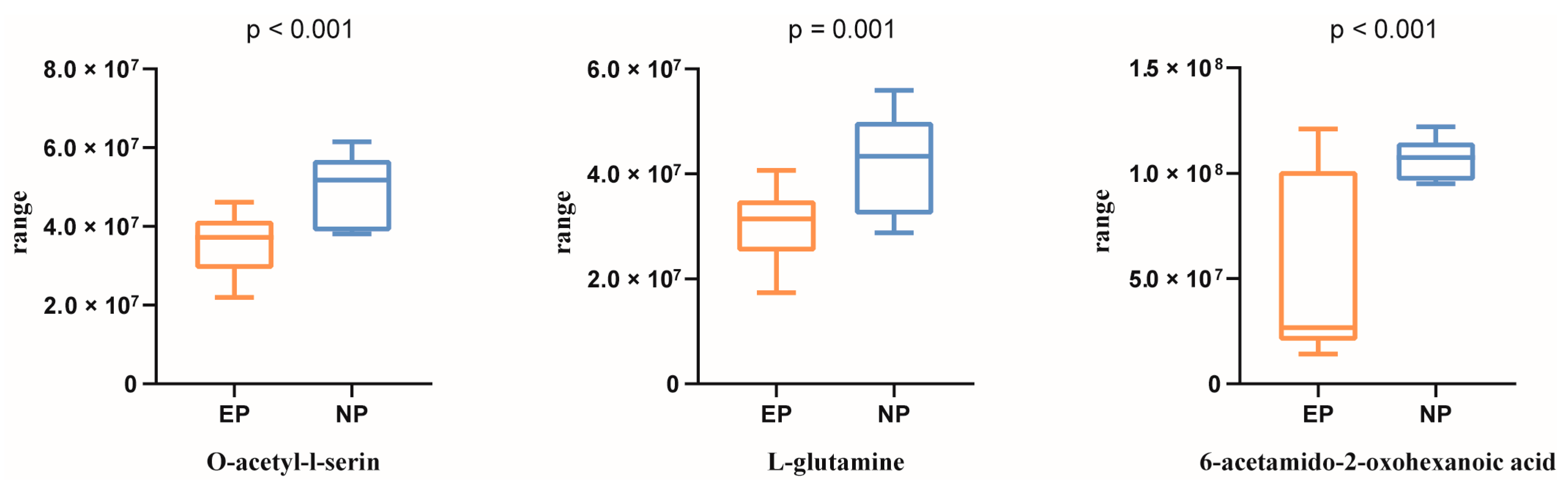
3.2. Metabolomic Profiling
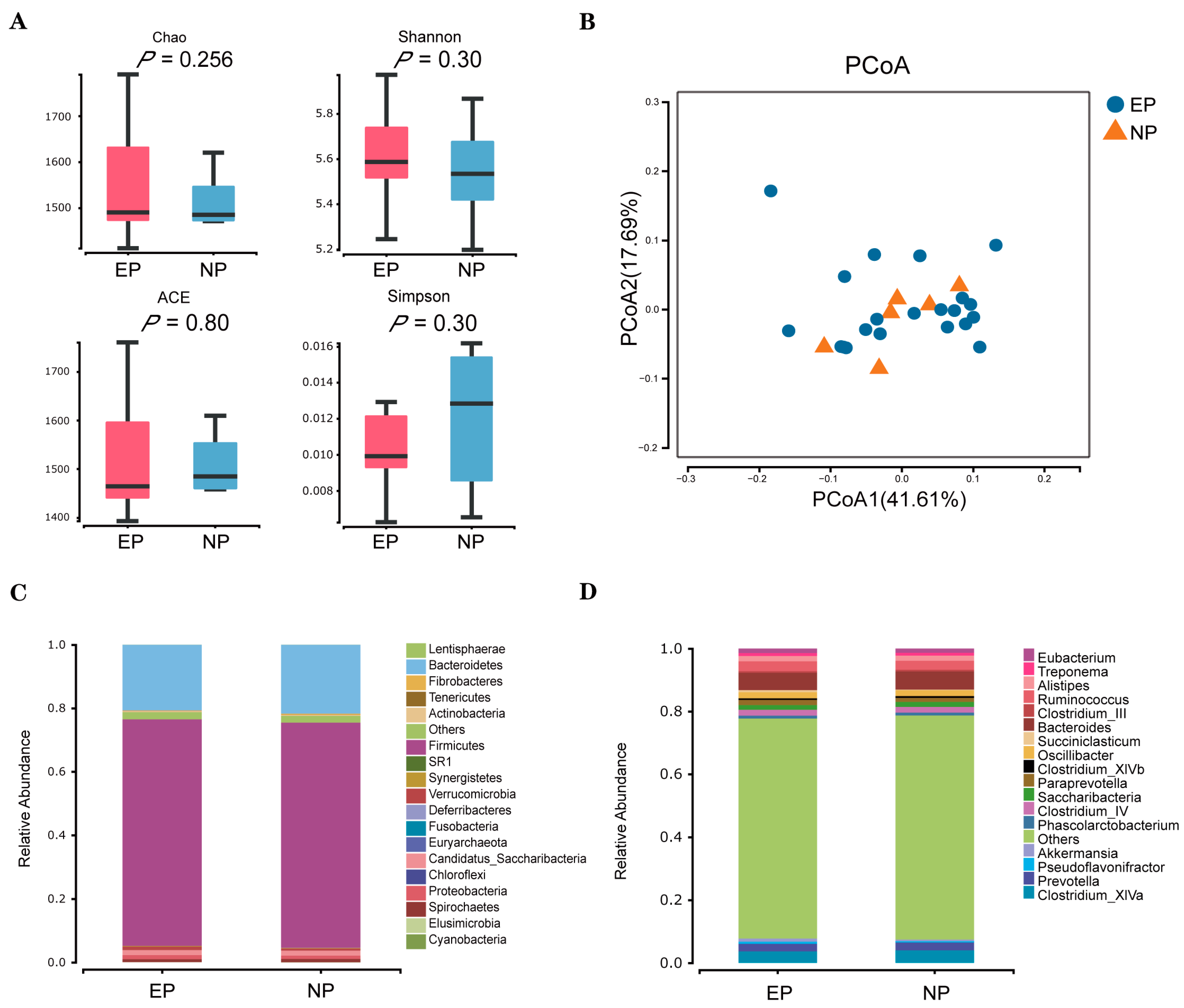
3.3. Faecal Microbial Diversity
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Goldansaz, S.A.; Markus, S.; Plastow, G.; Wishart, D.S. Predictive blood biomarkers of sheep pregnancy and litter size. Sci. Rep. 2022, 12, 10307. [Google Scholar] [CrossRef]
- Dunlop, A.L.; Mulle, J.G.; Ferranti, E.P.; Edwards, S.; Dunn, A.B.; Corwin, E.J. Maternal Microbiome and Pregnancy Outcomes That Impact Infant Health. Adv. Neonat. Care 2015, 15, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Santarosa, B.P.; Ferreira, D.O.L.; Hooper, H.B.; Sinzato, Y.K.; Damasceno, D.C.; Polizel, D.M.; Fioratti, E.G.; Dos Santos, V.H.; da Silva, A.A.; Gonçalves, R.C. Endocrine-metabolic adaptations in Dorper ewes: Comparison between single and twin pregnancies during gestation, parturition, and postpartum. Trop. Anim. Health Prod. 2022, 54, 307. [Google Scholar] [CrossRef] [PubMed]
- Anwar, M.; Riaz, A.; Ullah, N.; Rafiq, M. Use of ultrasonography for pregnancy diagnosis in balkhi sheep. Pak. Vet. J. 2008, 28, 144–146. [Google Scholar]
- Brzozowska, A.; Stankiewicz, T.; Blaszczyk, B.; Chundekkad, P.; Udala, J.; Wojtasiak, N. Ultrasound parameters of early pregnancy and Doppler indices of blood vessels in the placenta and umbilical cord throughout the pregnancy period in sheep. BMC Vet. Res. 2022, 18, 326. [Google Scholar] [CrossRef]
- Shahin, M.; Friedrich, M.; Gauly, M.; Beckers, J.F.; Holtz, W. Pregnancy-associated glycoprotein (PAG) pattern and pregnancy detection in Boer goats using an ELISA with different antisera. Small Rumin. Res. 2013, 113, 141–144. [Google Scholar] [CrossRef]
- Ding, H.; Pang, L.; Wang, J.; Liu, Y.; Wang, Y.; Feng, T. Analysis of Fecal Microbiota and Blood Metabolome Characteristics of Non-Pregnant Ewes. CJAN 2022, 34, 3107–3121. [Google Scholar]
- Al-Samawi, K.; Al-Hassan, M.; Migdadi, H.; Ammar, M.; Alghamdi, S. Interferon-stimulated Gene 15 and Interferon-1 Stimulated Gene 17 Messenger RNA-based Detection of Early Pregnancy in Aardi Goats in Saudi Arabia. Pak. J. Zool. 2021, 53, 1005–1013. [Google Scholar] [CrossRef]
- Hitit, M.; Kose, M.; Kaya, M.S.; Kirbas, M.; Dursun, S.; Alak, I.; Atli, M.O. Circulating miRNAs in maternal plasma as potential biomarkers of early pregnancy in sheep. Front. Genet. 2022, 13, 929477. [Google Scholar] [CrossRef]
- Wang, D.; Cheng, X.; Fang, H.; Ren, Y.; Li, X.; Ren, W.; Xue, B.; Yang, C. Effect of cold stress on ovarian & uterine microcirculation in rats and the role of endothelin system. Reprod. Biol. Endocrin. 2020, 18, 29. [Google Scholar] [CrossRef]
- Nabenishi, H.; Yamazaki, A. Impaired reproduction in Japanese Black cattle under cold environmental conditions. Reprod. Domest. Anim. 2017, 52, 371–375. [Google Scholar] [CrossRef] [PubMed]
- Goldansaz, S.A.; Guo, A.C.; Sajed, T.; Steele, M.A.; Plastow, G.S.; Wishart, D.S. Livestock metabolomics and the livestock metabolome: A systematic review. PLoS ONE 2017, 12, e0177675. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. UPARSE: Highly accurate OTU sequences from microbial amplicon reads. Nat Methods 2013, 10, 996–998. [Google Scholar] [CrossRef] [PubMed]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Peña, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat Methods 2010, 7, 335–336. [Google Scholar] [CrossRef] [PubMed]
- Al-Rukibat, R.; Ismail, Z.; Al-Zghoul, M.B.; Hananeh, W. Establishment of reference intervals of selected blood biochemical parameters in Shami goats. Vet. Clin. Pathol. 2020, 49, 665–668. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; Liu, J.; Yuan, E.; Li, Y.; Wang, Q.; Jia, L.; Wang, L.; Su, Y. Gestational age-specific reference intervals for 15 biochemical measurands during normal pregnancy in China. Ann. Clin. Biochem. 2018, 55, 446–452. [Google Scholar] [CrossRef]
- Vojta, A.; Shek-Vugrovečki, A.; Radin, L.; Efendić, M.; Pejaković, J.; Šimpraga, M. Hematological and biochemical reference intervals in Dalmatian pramenka sheep estimated from reduced sample size by bootstrap resampling. Vet. Arhiv. 2011, 81, 25–33. [Google Scholar]
- La Du, B.N. Butyrylcholinesterase variants and the new methods of molecular biology. Acta Anaesthesiol. Scand. 1995, 39, 139–141. [Google Scholar] [CrossRef]
- Kovacs, C.S. Bone development and mineral homeostasis in the fetus and neonate: Roles of the calciotropic and phosphotropic hormones. Physiol. Rev. 2014, 94, 1143–1218. [Google Scholar] [CrossRef]
- Fanni, D.; Gerosa, C.; Nurchi, V.M.; Manchia, M.; Saba, L.; Coghe, F.; Crisponi, G.; Gibo, Y.; Van Eyken, P.; Fanos, V.; et al. The Role of Magnesium in Pregnancy and in Fetal Programming of Adult Diseases. Biol. Trace Elem. Res. 2021, 199, 3647–3657. [Google Scholar] [CrossRef]
- Dalton, L.M.; Fhloinn, D.M.N.; Gaydadzhieva, G.T.; Mazurkiewicz, O.M.; Leeson, H.; Wright, C.P. Magnesium in pregnancy. Nutr. Rev. 2016, 74, 549–557. [Google Scholar] [CrossRef] [PubMed]
- Grzeszczak, K.; Kwiatkowski, S.; Kosik-Bogacka, D. The Role of Fe, Zn, and Cu in Pregnancy. Biomolecules 2020, 10, 1176. [Google Scholar] [CrossRef]
- Harrison-Findik, D.D. Gender-related variations in iron metabolism and liver diseases. World J. Hepatol. 2010, 2, 302–310. [Google Scholar] [CrossRef]
- Khanwalker, M.; Johns, J.; Honikel, M.M.; Smith, V.; Maxwell, S.; Santhanaraman, S.; La Belle, J. Electrochemical Detection of Fertility Hormones. Crit. Rev. Biom. Eng. 2019, 47, 235–247. [Google Scholar] [CrossRef] [PubMed]
- Beasley, A.M.; Kahn, L.P.; Windon, R.G. The periparturient relaxation of immunity in Merino ewes infected with Trichostrongylus colubriformis: Endocrine and body compositional responses. Vet. Parasitol. 2010, 168, 51–59. [Google Scholar] [CrossRef]
- Marceau, K.; Rolan, E.; Robertson, O.; Wang, W.; Shirtcliff, E.A. Within-person changes of cortisol, dehydroepiandrosterone, testosterone, estradiol, and progesterone in hair across pregnancy, with comparison to a non-pregnant reference group. Compr. Psychoneuroendocrinol. 2021, 5, 100024. [Google Scholar] [CrossRef]
- Luyckx, A.S.; Gerard, J.; Gaspard, U.; Lefebvre, P.J. Plasma glucagon levels in normal women during pregnancy. Diabetologia 1975, 11, 549–554. [Google Scholar] [CrossRef]
- Saudek, C.D.; Finkowski, M.; Knopp, R.H. Plasma glucagon and insulin in rat pregnancy. Roles in glucose homeostasis. J. Clin. Investig. 1975, 55, 180–187. [Google Scholar] [CrossRef]
- Qiao, L.; Saget, S.; Lu, C.; Zang, T.; Dzyuba, B.; Hay, W.W., Jr.; Shao, J. The Essential Role of Pancreatic α-Cells in Maternal Metabolic Adaptation to Pregnancy. Diabetes 2022, 71, 978–988. [Google Scholar] [CrossRef]
- Feng, L.; Qi, Q.; Wang, P.; Chen, H.; Chen, Z.; Meng, Z.; Liu, L. Serum levels of IL-6, IL-8, and IL-10 are indicators of prognosis in pancreatic cancer. J. Int. Med. Res. 2018, 46, 5228–5236. [Google Scholar] [CrossRef]
- Luppi, P. How immune mechanisms are affected by pregnancy. Vaccine 2003, 21, 3352–3357. [Google Scholar] [CrossRef] [PubMed]
- Amoudruz, P.; Minang, J.T.; Sundström, Y.; Nilsson, C.; Lilja, G.; Troye-Blomberg, M.; Sverremark-Ekström, E. Pregnancy, but not the allergic status, influences spontaneous and induced interleukin-1beta (IL-1beta), IL-6, IL-10 and IL-12 responses. Immunology 2006, 119, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Griffith, O.W.; Chavan, A.R.; Protopapas, S.; Maziarz, J.; Romero, R.; Wagner, G.P. Embryo implantation evolved from an ancestral inflammatory attachment reaction. Proc. Natl. Acad. Sci. USA 2017, 114, E6566–E6575. [Google Scholar] [CrossRef]
- Guo, Y.S.; Tao, J.Z. Metabolomics and pathway analyses to characterize metabolic alterations in pregnant dairy cows on D 17 and D 45 after AI. Sci. Rep. 2018, 8, 5973. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Liu, H.; Bai, M.; Gao, J.; Wu, X.; Yin, Y. Redox Properties of Tryptophan Metabolism and the Concept of Tryptophan Use in Pregnancy. Int. J. Mol. Sci. 2017, 18, 1595. [Google Scholar] [CrossRef]
- Sarangi, A.; Ghosh, M.; Sangwan, S.; Kumar, R.; Balhara, S.; Phulia, S.K.; Sharma, R.K.; Sahu, S.; Kumar, S.; Mohanty, A.K.; et al. Exploration of urinary metabolite dynamicity for early detection of pregnancy in water buffaloes. Sci. Rep. 2022, 12, 16295. [Google Scholar] [CrossRef]
- Di Giulio, A.M.; Carelli, S.; Castoldi, R.E.; Gorio, A.; Taricco, E.; Cetin, I. Plasma amino acid concentrations throughout normal pregnancy and early stages of intrauterine growth restricted pregnancy. J. Matern.-Fetal Neonatal Med. 2004, 15, 356–362. [Google Scholar] [CrossRef]
- Ortega, P.; Castejon, H.V.; Argotte, M.G.; Gomez, G.; Bohorquez, L.; Urrieta, J.R. Plasma amino acids profile of healthy pregnant adolescent girls in Maracaibo, Venezuela. Arch. Latinoam. Nutr. 2003, 53, 157–164. [Google Scholar]
- DiGiulio, D.B.; Callahan, B.J.; McMurdie, P.J.; Costello, E.K.; Lyell, D.J.; Robaczewska, A.; Sun, C.L.; Goltsman, D.S.; Wong, R.J.; Shaw, G.; et al. Temporal and spatial variation of the human microbiota during pregnancy. Proc. Natl. Acad. Sci. USA 2015, 112, 11060–11065. [Google Scholar] [CrossRef]
- Komiya, S.; Naito, Y.; Okada, H.; Matsuo, Y.; Hirota, K.; Takagi, T.; Mizushima, K.; Inoue, R.; Abe, A.; Morimoto, Y. Characterizing the gut microbiota in females with infertility and preliminary results of a water-soluble dietary fiber intervention study. J. Clin. Biochem. Nutr. 2020, 67, 105–111. [Google Scholar] [CrossRef]
- Koester, L.R.; Petry, A.L.; Youngs, C.R.; Schmitz-Esser, S. Ewe Vaginal Microbiota: Associations with Pregnancy Outcome and Changes during Gestation. Front. Microbiol. 2021, 12, 745884. [Google Scholar] [CrossRef]
- Smoliński, J.; Szeligowska, N.; Cholewińska, P.; Czyż, K.; Janczak, M. Levels of Main Bacterial Phyla in the Gastrointestinal Tract of Sheep Depending on Parity and Age. Animals 2021, 11, 2203. [Google Scholar] [CrossRef] [PubMed]
- Cholewińska, P.; Górniak, W.; Wojnarowski, K. Impact of selected environmental factors on microbiome of the digestive tract of ruminants. BMC Vet. Res. 2021, 17, 25. [Google Scholar] [CrossRef] [PubMed]
- Blanco-Penedo, I.; Cantalapiedra, J.; Llonch, P. Climate change impacts on animal welfare in livestock systems. ITEA-Inf. Técnica Económica Agrar. 2020, 116, 424–443. [Google Scholar] [CrossRef]
- Huang, X.; Mi, J.; Denman, S.E.; Basangwangdui; Pingcuozhandui; Zhang, Q.; Long, R.; McSweeney, C.S. Changes in rumen microbial community composition in yak in response to seasonal variations. J. Appl. Microbiol. 2022, 132, 1652–1665. [Google Scholar] [CrossRef]
- Zhang, L.; Xue, J.; Wang, Q.; Lv, W.; Mi, H.; Liu, Y.; Yang, L. Changes in expression of ISG15, progesterone receptor and progesterone-induced blocking factor in ovine thymus during early pregnancy. Theriogenology 2018, 121, 153–159. [Google Scholar] [CrossRef]
- Kiyma, Z.; Kose, M.; Atli, M.O.; Ozel, C.; Hitit, M.; Sen, G.; Kaya, M.; Kaya, M.S.; Kurar, E.; Kayis, S.A.; et al. Investigation of interferon-tau stimulated genes (ISGs) simultaneously in the endometrium, corpus luteum (CL) and peripheral blood leukocytes (PBLs) in the preluteolytic stage of early pregnancy in ewes. Small Ruminant Res. 2016, 140, 1–6. [Google Scholar] [CrossRef]
- Koch, J.M.; Ramadoss, J.; Magness, R.R. Proteomic Profile of Uterine Luminal Fluid from Early Pregnant Ewes. J. Proteome Res. 2010, 9, 3878–3885. [Google Scholar] [CrossRef]
- Khan, D.; Khan, H.; Ahmad, N.; Tunio, M.T.; Tahir, M.; Khan, M.S.; Khan, R.U. Early Pregnancy Diagnosis using Pregnancy-Associated Glycoproteins in the Serum of Pregnant Ruminants. Pak. J. Zool. 2020, 52, 785–788. [Google Scholar] [CrossRef]
- Karen, A.; Beckers, J.F.; Sulon, J.; de Sousa, N.M.; Szabados, K.; Reczigel, J.; Szenci, O. Early pregnancy diagnosis in sheep by progesterone and pregnancy-associated glycoprotein tests. Theriogenology 2003, 59, 1941–1948. [Google Scholar] [CrossRef]
- Elango, R. Methionine Nutrition and Metabolism: Insights from Animal Studies to Inform Human Nutrition. J. Nutr. 2020, 150, 2518S–2523S. [Google Scholar] [CrossRef] [PubMed]
- Dasarathy, J.; Gruca, L.L.; Bennett, C.; Parimi, P.S.; Duenas, C.; Marczewski, S.; Fierro, J.L.; Kalhan, S.C. Methionine metabolism in human pregnancy. Am. J. Clin. Nutr. 2010, 91, 357–365. [Google Scholar] [CrossRef] [PubMed]
- Anderson, P.M.; Lalla, R.V. Glutamine for Amelioration of Radiation and Chemotherapy Associated Mucositis during Cancer Therapy. Nutrients 2020, 12, 1675. [Google Scholar] [CrossRef] [PubMed]
- Cruzat, V.; Rogero, M.M.; Keane, K.N.; Curi, R.; Newsholme, P. Glutamine: Metabolism and Immune Function, Supplementation and Clinical Translation. Nutrients 2018, 10, 1564. [Google Scholar] [CrossRef]
- Cuesta, A.; Taunton, J. Lysine-Targeted Inhibitors and Chemoproteomic Probes. Annu. Rev. Biochem. 2019, 88, 365–381. [Google Scholar] [CrossRef]
| Parameters | NP (n = 23) | EP (n = 6) | SEM | p |
|---|---|---|---|---|
| TP (g/L) | 83.9 | 83.4 | 3.13 | 0.891 |
| ALB (g/L) | 38.2 | 37.1 | 1.36 | 0.437 |
| BUN (mmol/L) | 7.62 | 6.81 | 0.60 | 0.192 |
| ALT (U/L) | 16.4 | 11.9 | 2.77 | 0.115 |
| AST (U/L) | 120 | 98.5 | 9.13 | 0.027 |
| ALP (U/L) | 114 | 130 | 27.9 | 0.570 |
| GGT (U/L) | 57.0 | 50.7 | 5.25 | 0.242 |
| CHE (mmol/L) | 240 | 172 | 13.2 | <0.001 |
| DBIL (U/L) | 0.80 | 0.85 | 0.23 | 0.838 |
| TBIL (mmol/L) | 1.88 | 2.62 | 0.51 | 0.160 |
| TG (mmol/L) | 0.27 | 0.18 | 0.04 | 0.031 |
| CHOL (mmol/L) | 1.75 | 1.71 | 0.11 | 0.711 |
| LDL-C (μmol/L) | 0.67 | 0.71 | 0.08 | 0.617 |
| HDL-C (mmol/L) | 1.04 | 0.99 | 0.05 | 0.415 |
| FFA (μmol/L) | 150 | 184 | 42.9 | 0.432 |
| BHB (mmol/L) | 0.75 | 0.72 | 0.08 | 0.735 |
| GLU (mmol/L) | 4.25 | 4.06 | 0.44 | 0.665 |
| AMS (mmol/L) | 51.0 | 24.5 | 35.0 | 0.482 |
| LAC (μmol/L) | 9.12 | 9.63 | 1.74 | 0.769 |
| LDH (U/L) | 542 | 449 | 31.7 | 0.007 |
| CRE (μmol/L) | 79.8 | 87.2 | 6.71 | 0.283 |
| Parameters | NP (n = 23) | EP (n = 6) | SEM | p |
|---|---|---|---|---|
| Mg (μmol/L) | 1.11 | 0.90 | 0.06 | 0.003 |
| Fe (mmol/L) | 29.3 | 24.6 | 2.06 | 0.032 |
| Ca (mmol/L) | 2.50 | 2.37 | 0.10 | 0.220 |
| P (U/L) | 1.61 | 1.82 | 0.20 | 0.291 |
| Parameters | NP (n = 23) | EP (n = 6) | SEM | p |
|---|---|---|---|---|
| INS (μIU/mL) | 3.43 | 2.99 | 0.35 | 0.263 |
| GC (pg/mL) | 1076 | 1575 | 139 | 0.002 |
| LH (mIU/mL) | 4.60 | 1.15 | 2.79 | 0.268 |
| FSH (mIU/mL) | 0.26 | 0.25 | 0.21 | 0.940 |
| P4 (ng/mL) | 2.19 | 4.01 | 1.30 | 0.097 |
| E2 (pg/mL) | 204 | 527 | 115 | 0.012 |
| Parameters | NP (n = 23) | EP (n = 6) | SEM | p |
|---|---|---|---|---|
| IL-1β (pg/mL) | 83.2 | 122 | 83.0 | 0.643 |
| IL-6 (pg/mL) | 21.2 | 64.6 | 10.2 | <0.001 |
| TNF-α (pg/mL) | 8.15 | 7.88 | 0.29 | 0.348 |
| IgG (g/L) | 11.3 | 13.3 | 2.90 | 0.511 |
| IgA (g/L) | 1.20 | 0.92 | 0.21 | 0.195 |
| IgM (g/L) | 1.35 | 1.45 | 0.27 | 0.719 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, Z.; Yang, Y.; Wang, B.; Gebeyew, K.; Tang, S.; Han, X.; He, Z.; Tan, Z. Blood Metabolites and Faecal Microbial Communities in Nonpregnant and Early Gestation Ewes in Highly Cold Areas. Biology 2023, 12, 1436. https://doi.org/10.3390/biology12111436
Wu Z, Yang Y, Wang B, Gebeyew K, Tang S, Han X, He Z, Tan Z. Blood Metabolites and Faecal Microbial Communities in Nonpregnant and Early Gestation Ewes in Highly Cold Areas. Biology. 2023; 12(11):1436. https://doi.org/10.3390/biology12111436
Chicago/Turabian StyleWu, Zhiwu, Yanyan Yang, Biao Wang, Kefyalew Gebeyew, Shaoxun Tang, Xuefeng Han, Zhixiong He, and Zhiliang Tan. 2023. "Blood Metabolites and Faecal Microbial Communities in Nonpregnant and Early Gestation Ewes in Highly Cold Areas" Biology 12, no. 11: 1436. https://doi.org/10.3390/biology12111436
APA StyleWu, Z., Yang, Y., Wang, B., Gebeyew, K., Tang, S., Han, X., He, Z., & Tan, Z. (2023). Blood Metabolites and Faecal Microbial Communities in Nonpregnant and Early Gestation Ewes in Highly Cold Areas. Biology, 12(11), 1436. https://doi.org/10.3390/biology12111436






