Individual Immune Response to SARS-CoV-2 Infection—The Role of Seasonal Coronaviruses and Human Leukocyte Antigen
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Cohort
2.1.1. Natural Infection Phase—Sample
2.1.2. Immunization—Sample
2.2. Longitudinal Antibody Data
2.3. Multiplex Bead-Based Immunoassay
2.4. Dichotomization into High and Low Responders with Regard to SARS-CoV-2 Infection
2.5. HLA Typing with Next-Generation Sequencing (NGS)
2.6. Statistical Analyses
2.7. Ethical Approval
3. Results
3.1. Humoral Response against sCoVs, MERS-CoV, SARS-CoV and SARS-CoV-2 in the Natural Infection Phase without Vaccinations
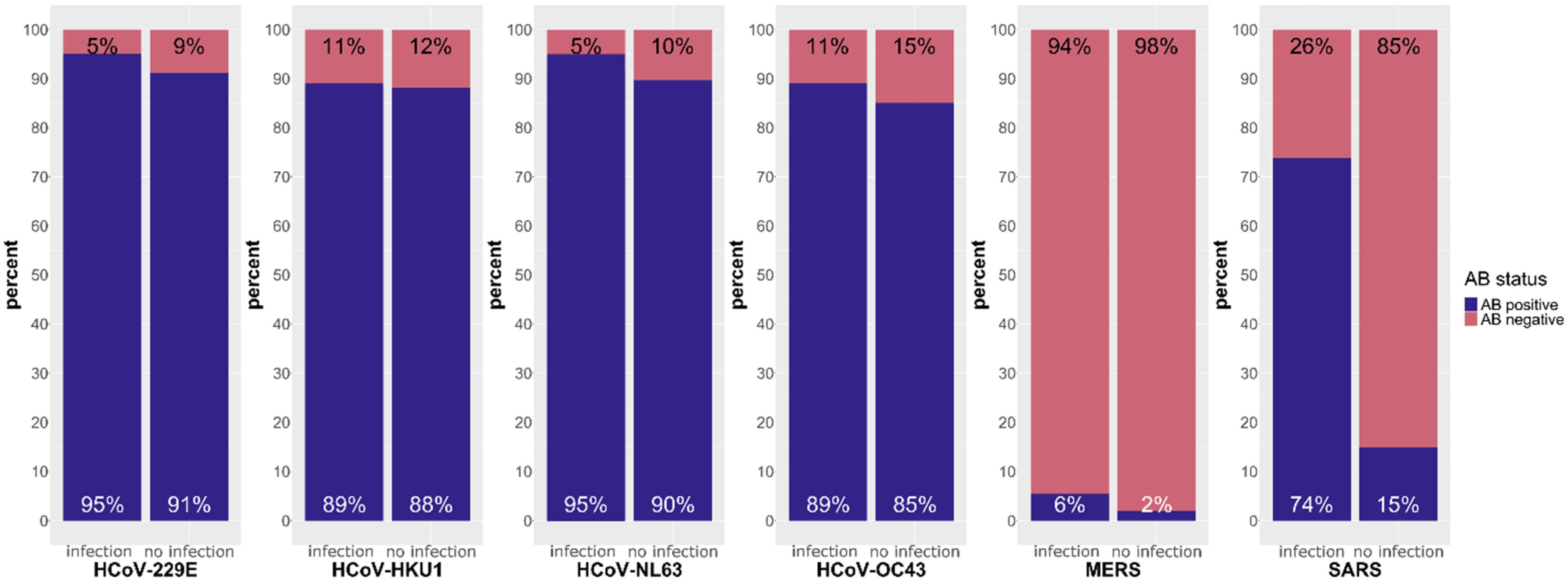
3.1.1. Responses against sCoVs, MERS-CoV and SARS-CoV
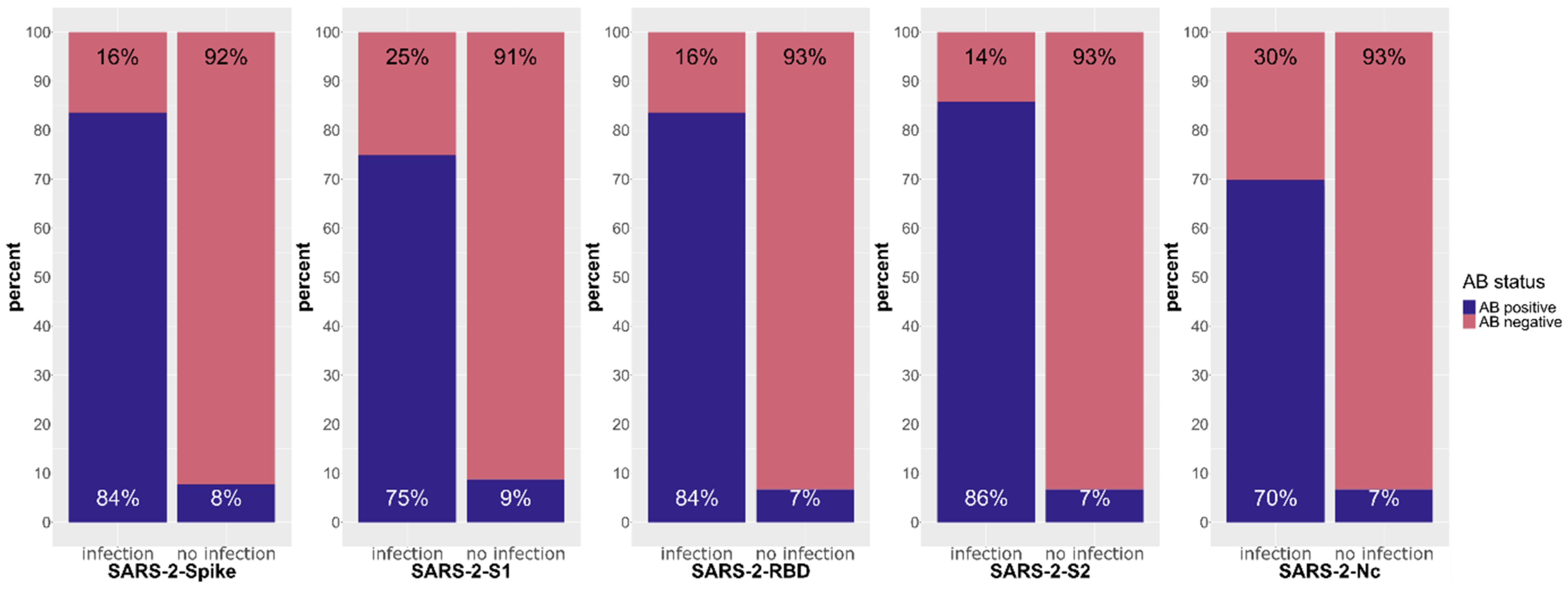
3.1.2. Response against SARS-CoV-2
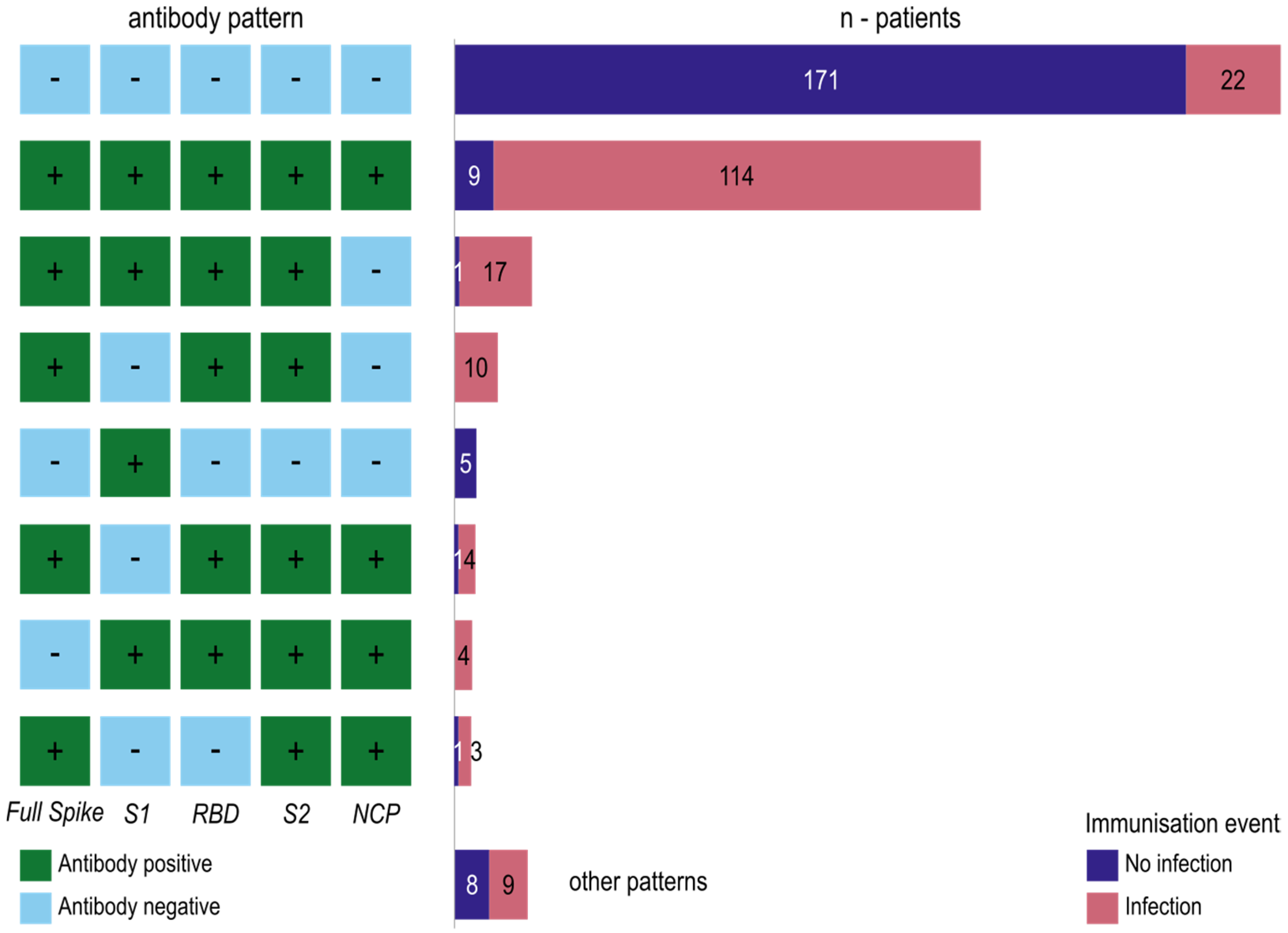
3.1.3. Antibody Formation against the SARS-CoV-2 Domains Reveals Immuno-Reactive Patterns
3.1.4. Correlation Analysis between Antibody Levels against Seasonal Coronaviruses, MERS-CoV, SARS-CoV and SARS-CoV-2
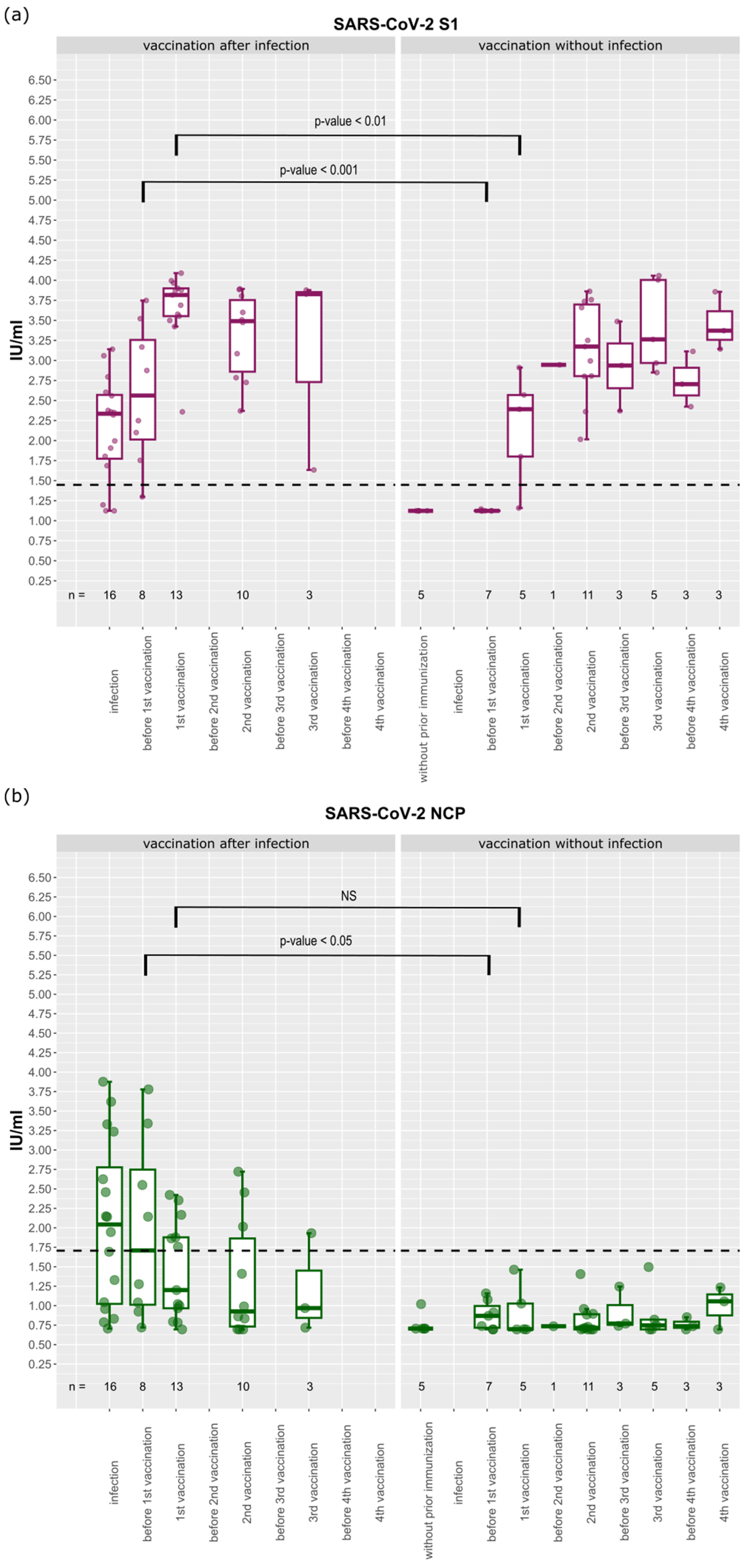
3.2. Humoral Response towards SARS-CoV-2 after the First Immunization Event
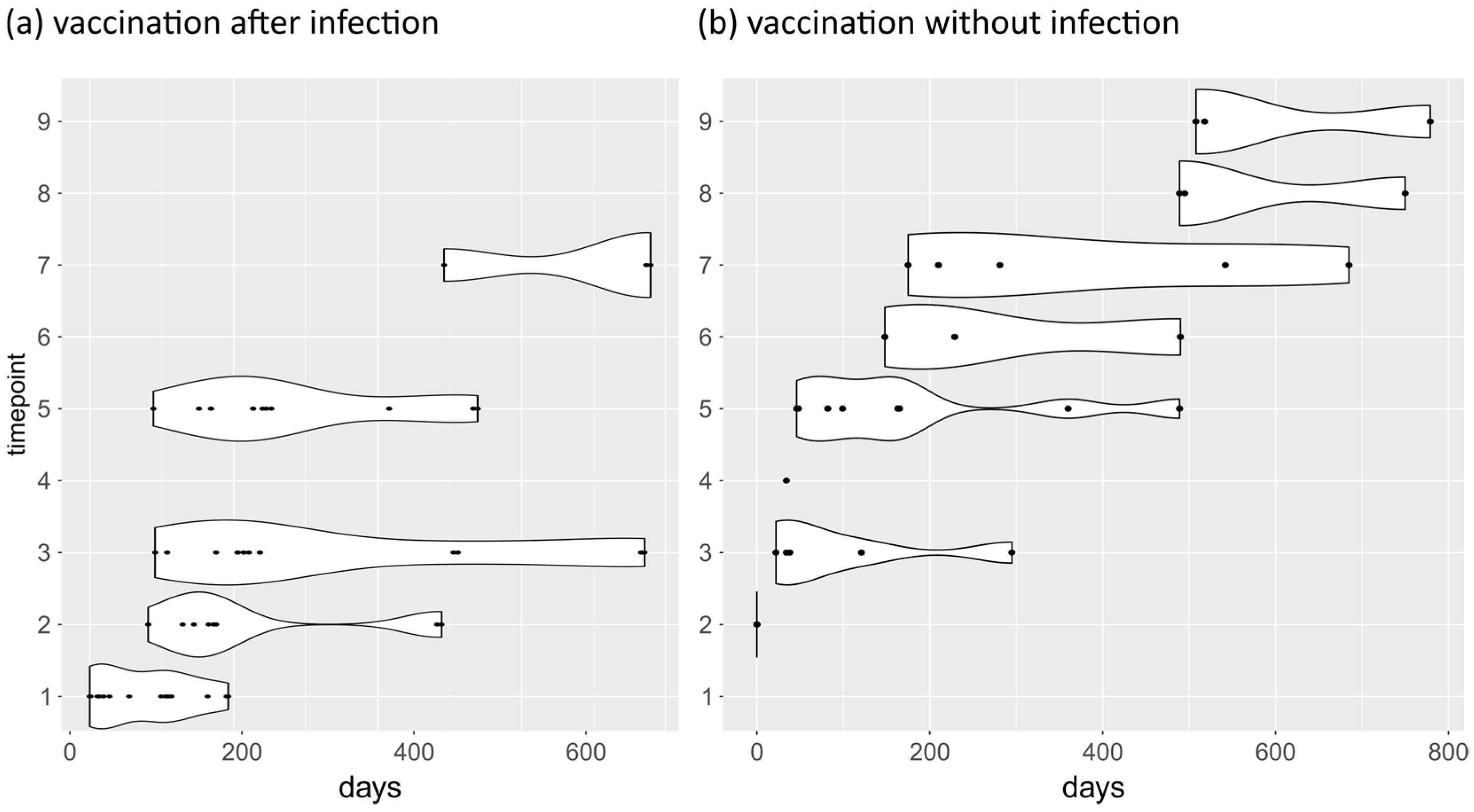
3.3. Longitudinal Analysis of Antibody Levels against SARS-CoV-2
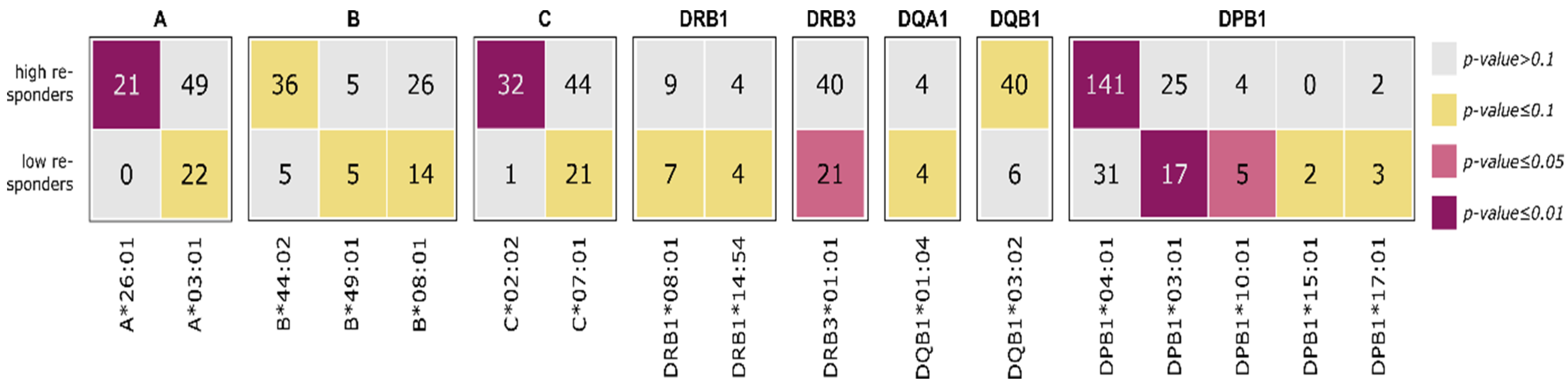
3.4. HLA Enrichment Analysis of Low and High Responders
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO Coronavirus (COVID-19) Dashboard|WHO Coronavirus (COVID-19) Dashboard with Vaccination Data. Available online: https://covid19.who.int/ (accessed on 24 September 2023).
- Merad, M.; Blish, C.A.; Sallusto, F.; Iwasaki, A. The immunology and immunopathology of COVID-19. Science 2022, 375, 1122–1127. [Google Scholar] [CrossRef] [PubMed]
- Qi, H.; Liu, B.; Wang, X.; Zhang, L. The humoral response and antibodies against SARS-CoV-2 infection. Nat. Immunol. 2022, 23, 1008–1020. [Google Scholar] [CrossRef] [PubMed]
- Galipeau, Y.; Greig, M.; Liu, G.; Driedger, M.; Langlois, M.-A. Humoral Responses and Serological Assays in SARS-CoV-2 Infections. Front. Immunol. 2020, 11, 610688. [Google Scholar] [CrossRef] [PubMed]
- Edridge, A.W.D.; Kaczorowska, J.; Hoste, A.C.R.; Bakker, M.; Klein, M.; Loens, K.; Jebbink, M.F.; Matser, A.; Kinsella, C.M.; Rueda, P.; et al. Seasonal coronavirus protective immunity is short-lasting. Nat. Med. 2020, 26, 1691–1693. [Google Scholar] [CrossRef] [PubMed]
- Hicks, J.; Klumpp-Thomas, C.; Kalish, H.; Shunmugavel, A.; Mehalko, J.; Denson, J.-P.; Snead, K.R.; Drew, M.; Corbett, K.S.; Graham, B.S.; et al. Serologic Cross-Reactivity of SARS-CoV-2 with Endemic and Seasonal Betacoronaviruses. J. Clin. Immunol. 2021, 41, 906–913. [Google Scholar] [CrossRef]
- Fischer, J.C.; Balz, V.; Jazmati, D.; Bölke, E.; Freise, N.F.; Keitel, V.; Feldt, T.; Jensen, B.-E.O.; Bode, J.; Lüdde, T.; et al. Prognostic markers for the clinical course in the blood of patients with SARS-CoV-2 infection. Eur. J. Med. Res. 2022, 27, 255. [Google Scholar] [CrossRef]
- Lehmann, C.; Loeffler-Wirth, H.; Balz, V.; Enczmann, J.; Landgraf, R.; Lakowa, N.; Gruenewald, T.; Fischer, J.C.; Doxiadis, I. Immunogenetic Predisposition to SARS-CoV-2 Infection. Biology 2022, 12, 37. [Google Scholar] [CrossRef]
- Augusto, D.G.; Murdolo, L.D.; Chatzileontiadou, D.S.M.; Sabatino, J.J.; Yusufali, T.; Peyser, N.D.; Butcher, X.; Kizer, K.; Guthrie, K.; Murray, V.W.; et al. A common allele of HLA is associated with asymptomatic SARS-CoV-2 infection. Nature 2023, 620, 128–136. [Google Scholar] [CrossRef]
- Wolday, D.; Fung, C.Y.J.; Morgan, G.; Casalino, S.; Frangione, E.; Taher, J.; Lerner-Ellis, J.P. HLA Variation and SARS-CoV-2 Specific Antibody Response. Viruses 2023, 15, 906. [Google Scholar] [CrossRef]
- Dendrou, C.A.; Petersen, J.; Rossjohn, J.; Fugger, L. HLA variation and disease. Nat. Rev. Immunol. 2018, 18, 325–339. [Google Scholar] [CrossRef]
- Gerhards, C.; Thiaucourt, M.; Kittel, M.; Becker, C.; Ast, V.; Hetjens, M.; Neumaier, M.; Haselmann, V. Longitudinal assessment of anti-SARS-CoV-2 antibody dynamics and clinical features following convalescence from a COVID-19 infection. Int. J. Infect. Dis. 2021, 107, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Wright, P.F.; Prevost-Reilly, A.C.; Natarajan, H.; Brickley, E.B.; Connor, R.I.; Wieland-Alter, W.F.; Miele, A.S.; Weiner, J.A.; Nerenz, R.D.; Ackerman, M.E. Longitudinal Systemic and Mucosal Immune Responses to SARS-CoV-2 Infection. J. Infect. Dis. 2022, 226, 1204–1214. [Google Scholar] [CrossRef]
- Vo, H.T.M.; Maestri, A.; Auerswald, H.; Sorn, S.; Lay, S.; Seng, H.; Sann, S.; Ya, N.; Pean, P.; Dussart, P.; et al. Robust and Functional Immune Memory up to 9 Months After SARS-CoV-2 Infection: A Southeast Asian Longitudinal Cohort. Front. Immunol. 2022, 13, 817905. [Google Scholar] [CrossRef] [PubMed]
- Loesche, M.; Karlson, E.W.; Talabi, O.; Zhou, G.; Boutin, N.; Atchley, R.; Loevinsohn, G.; Chang, J.B.P.; Hasdianda, M.A.; Okenla, A.; et al. Longitudinal SARS-CoV-2 Nucleocapsid Antibody Kinetics, Seroreversion, and Implications for Seroepidemiologic Studies. Emerg. Infect. Dis. 2022, 28, 1859–1862. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.S.; Rowland-Jones, S.; Gea-Mallorquí, E. Will SARS-CoV-2 Infection Elicit Long-Lasting Protective or Sterilising Immunity? Implications for Vaccine Strategies (2020). Front. Immunol. 2020, 11, 571481. [Google Scholar] [CrossRef]
- Anderson, M.; Stec, M.; Gosha, A.; Mohammad, T.; Boler, M.; Tojo Suarez, R.; Behun, D.; Landay, A.; Cloherty, G.; Moy, J. Longitudinal Severe Acute Respiratory Syndrome Coronavirus 2 Vaccine Antibody Responses and Identification of Vaccine Breakthrough Infections among Healthcare Workers Using Nucleocapsid Immunoglobulin G. J. Infect. Dis. 2022, 226, 1934–1942. [Google Scholar] [CrossRef]
- Khoury, J.; Najjar-Debbiny, R.; Hanna, A.; Jabbour, A.; Abu Ahmad, Y.; Saffuri, A.; Abu-Sinni, M.; Shkeiri, R.; Elemy, A.; Hakim, F. COVID-19 vaccine—Long term immune decline and breakthrough infections. Vaccine 2021, 39, 6984–6989. [Google Scholar] [CrossRef]
- Wedia. COVID-19 Vaccinations Start in Germany Today: What You Need to Know. Available online: https://www.iamexpat.de/expat-info/german-expat-news/covid-19-vaccinations-start-germany-today-what-you-need-know (accessed on 19 August 2023).
- Müller, L.; Kannenberg, J.; Biemann, R.; Hönemann, M.; Ackermann, G.; Jassoy, C. Comparison of the measured values of quantitative SARS-CoV-2 spike antibody assays. J. Clin. Virol. 2022, 155, 105269. [Google Scholar] [CrossRef]
- Kannenberg, J.; Schnurra, C.; Reiners, N.; Henschler, R.; Buhmann, R.; Kaiser, T.; Biemann, R.; Hönemann, M.; Ackermann, G.; Trawinski, H.; et al. Sensitivity of SARS-CoV-2 antibody tests with late convalescent sera. J. Clin. Virol. Plus 2021, 1, 100038. [Google Scholar] [CrossRef]
- Luo, J.; Brakel, A.; Krizsan, A.; Ludwig, T.; Mötzing, M.; Volke, D.; Lakowa, N.; Grünewald, T.; Lehmann, C.; Wolf, J.; et al. Sensitive and specific serological ELISA for the detection of SARS-CoV-2 infections. Virol. J. 2022, 19, 50. [Google Scholar] [CrossRef]
- Bray, R.A.; Lee, J.-H.; Brescia, P.; Kumar, D.; Nong, T.; Shih, R.; Woodle, E.S.; Maltzman, J.S.; Gebel, H.M. Development and Validation of a Multiplex, Bead-based Assay to Detect Antibodies Directed Against SARS-CoV-2 Proteins. Transplantation 2021, 105, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Hober, S.; Hellström, C.; Olofsson, J.; Andersson, E.; Bergström, S.; Jernbom Falk, A.; Bayati, S.; Mravinacova, S.; Sjöberg, R.; Yousef, J.; et al. Systematic evaluation of SARS-CoV-2 antigens enables a highly specific and sensitive multiplex serological COVID-19 assay. Clin. Transl. Immunol. 2021, 10, e1312. [Google Scholar] [CrossRef] [PubMed]
- Mariën, J.; Ceulemans, A.; Michiels, J.; Heyndrickx, L.; Kerkhof, K.; Foque, N.; Widdowson, M.-A.; Mortgat, L.; Duysburgh, E.; Desombere, I.; et al. Evaluating SARS-CoV-2 spike and nucleocapsid proteins as targets for antibody detection in severe and mild COVID-19 cases using a Luminex bead-based assay. J. Virol. Methods 2021, 288, 114025. [Google Scholar] [CrossRef] [PubMed]
- Huang, A.T.; Garcia-Carreras, B.; Hitchings, M.D.T.; Yang, B.; Katzelnick, L.C.; Rattigan, S.M.; Borgert, B.A.; Moreno, C.A.; Solomon, B.D.; Trimmer-Smith, L.; et al. A systematic review of antibody mediated immunity to coronaviruses: Kinetics, correlates of protection, and association with severity. Nat. Commun. 2020, 11, 4704. [Google Scholar] [CrossRef]
- Grant, R.; Malik, M.R.; Elkholy, A.; van Kerkhove, M.D. A Review of Asymptomatic and Subclinical Middle East Respiratory Syndrome Coronavirus Infections. Epidemiol. Rev. 2019, 41, 69–81. [Google Scholar] [CrossRef]
- LEUNG, G.M.; LIM, W.W.; Ho, L.-M.; Lam, T.-H.; GHANI, A.C.; DONNELLY, C.A.; FRASER, C.; RILEY, S.; FERGUSON, N.M.; ANDERSON, R.M.; et al. Seroprevalence of IgG antibodies to SARS-coronavirus in asymptomatic or subclinical population groups. Epidemiol. Infect. 2006, 134, 211–221. [Google Scholar] [CrossRef]
- Ng, K.W.; Faulkner, N.; Cornish, G.H.; Rosa, A.; Harvey, R.; Hussain, S.; Ulferts, R.; Earl, C.; Wrobel, A.G.; Benton, D.J.; et al. Preexisting and de novo humoral immunity to SARS-CoV-2 in humans. Science 2020, 370, 1339–1343. [Google Scholar] [CrossRef]
- Aydillo, T.; Rombauts, A.; Stadlbauer, D.; Aslam, S.; Abelenda-Alonso, G.; Escalera, A.; Amanat, F.; Jiang, K.; Krammer, F.; Carratala, J.; et al. Immunological imprinting of the antibody response in COVID-19 patients. Nat. Commun. 2021, 12, 3781. [Google Scholar] [CrossRef]
- Dugas, M.; Grote-Westrick, T.; Vollenberg, R.; Lorentzen, E.; Brix, T.; Schmidt, H.; Tepasse, P.-R.; Kühn, J. Less severe course of COVID-19 is associated with elevated levels of antibodies against seasonal human coronaviruses OC43 and HKU1 (HCoV OC43, HCoV HKU1). Int. J. Infect. Dis. 2021, 105, 304–306. [Google Scholar] [CrossRef]
- Lin, C.-Y.; Wolf, J.; Brice, D.C.; Sun, Y.; Locke, M.; Cherry, S.; Castellaw, A.H.; Wehenkel, M.; Crawford, J.C.; Zarnitsyna, V.I.; et al. Pre-existing humoral immunity to human common cold coronaviruses negatively impacts the protective SARS-CoV-2 antibody response. Cell Host Microbe 2022, 30, 83–96.e4. [Google Scholar] [CrossRef]
- Shrock, E.; Fujimura, E.; Kula, T.; Timms, R.T.; Lee, I.-H.; Leng, Y.; Robinson, M.L.; Sie, B.M.; Li, M.Z.; Chen, Y.; et al. Viral epitope profiling of COVID-19 patients reveals cross-reactivity and correlates of severity. Science 2020, 370, eabd4250. [Google Scholar] [CrossRef]
- Tavasolian, F.; Rashidi, M.; Hatam, G.R.; Jeddi, M.; Hosseini, A.Z.; Mosawi, S.H.; Abdollahi, E.; Inman, R.D. HLA, Immune Response, and Susceptibility to COVID-19. Front. Immunol. 2020, 11, 601886. [Google Scholar] [CrossRef]
- Aguilar-Bretones, M.; Westerhuis, B.M.; Raadsen, M.P.; de Bruin, E.; Chandler, F.D.; Okba, N.M.; Haagmans, B.L.; Langerak, T.; Endeman, H.; van den Akker, J.P.; et al. Seasonal coronavirus-specific B cells with limited SARS-CoV-2 cross-reactivity dominate the IgG response in severe COVID-19. J. Clin. Investig. 2021, 131, e150613. [Google Scholar] [CrossRef]
- Castro Dopico, X.; Ols, S.; Loré, K.; Karlsson Hedestam, G.B. Immunity to SARS-CoV-2 induced by infection or vaccination. J. Intern. Med. 2022, 291, 32–50. [Google Scholar] [CrossRef]
- Song, G.; He, W.; Callaghan, S.; Anzanello, F.; Huang, D.; Ricketts, J.; Torres, J.L.; Beutler, N.; Peng, L.; Vargas, S.; et al. Cross-reactive serum and memory B-cell responses to spike protein in SARS-CoV-2 and endemic coronavirus infection. Nat. Commun. 2021, 12, 2938. [Google Scholar] [CrossRef]
- Tong, P.-B.-V.; Lin, L.-Y.; Tran, T.H. Coronaviruses pandemics: Can neutralizing antibodies help? Life Sci. 2020, 255, 117836. [Google Scholar] [CrossRef]
- Vogelzang, E.H.; Loeff, F.C.; Derksen, N.I.L.; Kruithof, S.; Ooijevaar-de Heer, P.; van Mierlo, G.; Linty, F.; Mok, J.Y.; van Esch, W.; de Bruin, S.; et al. Development of a SARS-CoV-2 Total Antibody Assay and the Dynamics of Antibody Response over Time in Hospitalized and Nonhospitalized Patients with COVID-19. J. Immunol. 2020, 205, 3491–3499. [Google Scholar] [CrossRef] [PubMed]
- Long, Q.-X.; Liu, B.-Z.; Deng, H.-J.; Wu, G.-C.; Deng, K.; Chen, Y.-K.; Liao, P.; Qiu, J.-F.; Lin, Y.; Cai, X.-F.; et al. Antibody responses to SARS-CoV-2 in patients with COVID-19. Nat. Med. 2020, 26, 845–848. [Google Scholar] [CrossRef] [PubMed]
- Siracusano, G.; Brombin, C.; Pastori, C.; Cugnata, F.; Noviello, M.; Tassi, E.; Princi, D.; Cantoni, D.; Malnati, M.S.; Maugeri, N.; et al. Profiling Antibody Response Patterns in COVID-19: Spike S1-Reactive IgA Signature in the Evolution of SARS-CoV-2 Infection. Front. Immunol. 2021, 12, 772239. [Google Scholar] [CrossRef] [PubMed]
- Demonbreun, A.R.; McDade, T.W.; Pesce, L.; Vaught, L.A.; Reiser, N.L.; Bogdanovic, E.; Velez, M.P.; Hsieh, R.R.; Simons, L.M.; Saber, R.; et al. Patterns and persistence of SARS-CoV-2 IgG antibodies in Chicago to monitor COVID-19 exposure. JCI Insight 2021, 6, e146148. [Google Scholar] [CrossRef] [PubMed]
- Comirnaty: EPAR—Product Information. Available online: https://www.ema.europa.eu/en/documents/product-information/comirnaty-epar-product-information_en.pdf (accessed on 24 September 2023).
- Spikevax (Previously COVID-19 Vaccine Moderna): EPAR—Product Information. Available online: https://www.ema.europa.eu/en/documents/product-information/spikevax-previously-covid-19-vaccine-moderna-epar-product-information_en.pdf (accessed on 24 September 2023).
- Vaxzevria (Previously COVID-19 Vaccine AstraZeneca): EPAR—Product Information. Available online: https://www.ema.europa.eu/en/documents/product-information/vaxzevria-previously-covid-19-vaccine-astrazeneca-epar-product-information_en.pdf (accessed on 24 September 2023).
- Kannenberg, J.; Trawinski, H.; Henschler, R.; Buhmann, R.; Hönemann, M.; Jassoy, C. Antibody Course and Memory B-Cell Response in the First Year after Severe Acute Respiratory Syndrome Coronavirus 2 Infection. J. Infect. Dis. 2022, 226, 664–672. [Google Scholar] [CrossRef] [PubMed]
- Augusto, D.G.; Hollenbach, J.A. HLA variation and antigen presentation in COVID-19 and SARS-CoV-2 infection. Curr. Opin. Immunol. 2022, 76, 102178. [Google Scholar] [CrossRef] [PubMed]
- Langton, D.J.; Bourke, S.C.; Lie, B.A.; Reiff, G.; Natu, S.; Darlay, R.; Burn, J.; Echevarria, C. The influence of HLA genotype on the severity of COVID-19 infection. HLA 2021, 98, 14–22. [Google Scholar] [CrossRef] [PubMed]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rottmayer, K.; Loeffler-Wirth, H.; Gruenewald, T.; Doxiadis, I.; Lehmann, C. Individual Immune Response to SARS-CoV-2 Infection—The Role of Seasonal Coronaviruses and Human Leukocyte Antigen. Biology 2023, 12, 1293. https://doi.org/10.3390/biology12101293
Rottmayer K, Loeffler-Wirth H, Gruenewald T, Doxiadis I, Lehmann C. Individual Immune Response to SARS-CoV-2 Infection—The Role of Seasonal Coronaviruses and Human Leukocyte Antigen. Biology. 2023; 12(10):1293. https://doi.org/10.3390/biology12101293
Chicago/Turabian StyleRottmayer, Karla, Henry Loeffler-Wirth, Thomas Gruenewald, Ilias Doxiadis, and Claudia Lehmann. 2023. "Individual Immune Response to SARS-CoV-2 Infection—The Role of Seasonal Coronaviruses and Human Leukocyte Antigen" Biology 12, no. 10: 1293. https://doi.org/10.3390/biology12101293
APA StyleRottmayer, K., Loeffler-Wirth, H., Gruenewald, T., Doxiadis, I., & Lehmann, C. (2023). Individual Immune Response to SARS-CoV-2 Infection—The Role of Seasonal Coronaviruses and Human Leukocyte Antigen. Biology, 12(10), 1293. https://doi.org/10.3390/biology12101293





