The Interaction between Mushroom Polysaccharides and Gut Microbiota and Their Effect on Human Health: A Review
Abstract
Simple Summary
Abstract
1. Introduction
2. Effects of the Structural Characteristics of Mushroom Polysaccharides on Gut Microbiota Diversity
2.1. Molecular Weight
2.2. Monosaccharide Composition
2.3. Glycosidic Bonds
3. Metabolism of Polysaccharides by Gut Microbiota
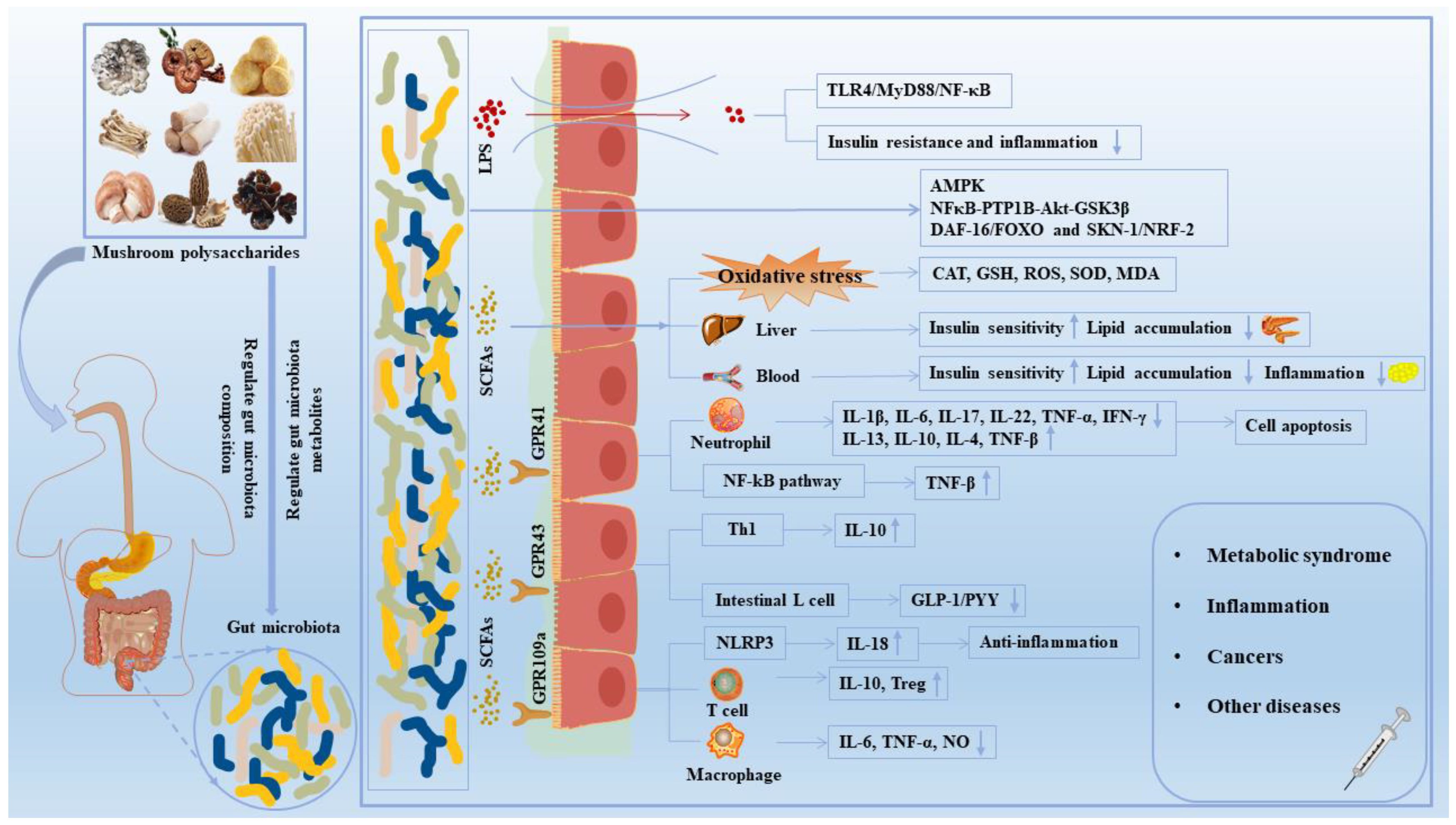
4. Effects of Mushroom Polysaccharides on Gut Microbe-Mediated Diseases
4.1. Improvement of Lipid and Glucose Metabolism Disorders
4.1.1. T2DM
| Disease | Mushroom | Model | Gut Microbiota Regulation | Effects on Hosts & Functional Mechanisms | Ref. |
|---|---|---|---|---|---|
| T2DM | Grifola frondosa | STZ-induced KM mice | Alistipes↑ Streptococcus, Enterococcus, Staphylococcus and Aerococcus↓ | Reduced the serum levels of FBG, OGT, TC, TG and LDL-C, and decreased the hepatic levels of TC, TG and FFA; increased mRNA expression of CYP7A1 and BSEP. | [20] |
| T2DM | Ganoderma lucidum | STZ-induced mice | Blautia, Dehalobacterium, Parabacteroides and Bacteroides↑ Aerococcus, Ruminococcus, Corynebactrium and Proteus↓ | Decreased the levels of fasting blood glucose and insulin; restored the amino acids metabolism, carbohydrates metabolism, inflammatory substances metabolism. | [51] |
| Hyperlipidemia | Grifola frondosa | HFD-induced Wistar rats | Helicobater, Intestinimonas, Parasutterella, Ruminococcus and Flavonifracter↑ Clostridium-XVIII, Butyricicoccus and Turicibacter↓ | Through decreasing the serum TG, TC, and FFA levels, and increasing the serum HDL-C level; increased the mRNA levels of BSEP, CYP7A1, Acox1 and hepatic GS. | [59] |
| T2DM | Grifola frondosa | STZ-induced ICR mice | Porphyromonas gingivalis, Akkermansia muciniphila, Lactobacillus acidophilus, Bacteroides acidifaciens↑ Firmicutes/Bacteroidetes ratio and Proteobacteria↓ | Decreased the fasting blood glucose level, improved oral glucose tolerance, alleviated insulin resistance; activated IRS1, PI3K, and GLUT4, inhibited JNK1/2; regulated the IRS1/PI3K and JNK signaling. | [60] |
| T2DM | Ganoderma lucidum | HFD-induced and STZ-induced KM mice | Ruminococcaceae, Prevotellaceae and Peptococcaceae↑ Lachnospiraceae, Desulfovibrionaceae and Lactobacillaceae↓ | Repaired islet cells and increased insulin secretion, improved insulin resistance, and improved carbohydrate metabolism, amino acid metabolism and lipid metabolism. | [101] |
| T2DM | Ganoderma lucidum | STZ-induced SD rats | Lactobacillus↑ Proteobacteria↓ | Promoted the expression of GS2, GYG1, Insig1, Insig2, ACC; elevated the level of HDL-C and reduced levels of TC and TG. | [102] |
| T2DM | Grifola frondosa | STZ-induced KM mice | Alistipes↑ Streptococcus, Enterococcus, Staphylococcus and Aerococcus↓ | Improved abnormal serum biochemical indicators TG, TC, LDL-C and glucose, inhibited lipid accumulation and steatosis; downregulated CD36 and SREBP-1C, upregulated CYP7A1. | [103] |
| Obesity | Ganoderma lucidum | HFD-induced C57BL/6J mice | Akkermansia, Bifidobacterium, Turicibacter, Parabacteroides↑ Blautia, Rikenella, Ruminiclostridium_UGC-009 and Lachnospiraceae↓ | Inhibited fat accumulation and body weight, hyperlipidemia; reduced LPS level; decreased levels of TNF-α and IL-1β; increased acetate and butyrate production; inhibited LPS/TLR4/NF-κB signaling pathway. | [107] |
| T2DM | Grifola frondosa | STZ-induced ICR mice | Lactobacillus, Desulfovibrio, Helicobacter, Lactobacillus and Bacteroides↑ Verrucomicrobia, Ruminococcus and Prevotella↓ | Improved the composition of gut microbiota and promoted the proliferation of beneficial bacteria. | [113] |
| Obesity | Agrocybe cylindracea | HFD-induced C57BL/6J mice | Bacteroides, Parabacteroides, Butyricimonas and Dubosiella↑ Desulfovibrio and Oscillibacter↓ | Reduced the levels of obesity-related TNF-α and IL-6, reduced fasting glucose and insulin levels. | [114] |
| T2DM | Sanghuang- porous vaninii | high-fat and high-sucrose ICR mice | Akkermansia, Dubosiella, Bacteroides and Parabacteroides↑ Lactobacillus, Flavonifractor, Odoribacter and Desulfovibrio↓ | Improved body weight, glycolipid metabolism, and inflammation-related parameters; ameliorated pancreas and jejunum injuries; enriched insulin signaling pathway and PI3K-Akt signaling pathway. | [115] |
| Obesity | Grifola frondosa | HFD-induced C57BL/6JNju mice | Mucispirillum, Bilophila and Dehalobacterium, Sutterella↑ Coprococcus and Ruminococcus↓ | Controlled the body weight, blood glucose and related organ indices, counteracted hyperlipidemia and IR triggered; regulated AST and ALT; down-regulated TLR4/NF-κB signaling. | [116] |
| NAFLD | Lentinan | HFD-induced C57BL/6J mice | Bifidobacterium, Streptococcaceae and Enterococcaceae genus, Streptococcus, Enterococcus, Ruminococcaceae↑ Helicobacteraceae and Helicobacter↓ | Restored intestinal redox balance, and reduced serum LPS; altered inflammation-insulin (NFκB-PTP1B- Akt-GSK3β) signaling molecules. | [117] |
| NAFLD | Grifola frondosa | HFD-induced Wistar rats | Bacteroides, Bifidobacterium, Blautia, Coprococcus, Phascolarctobacterium, Prevotella↑ Alistipes, Flavonifractor, Paraprevotella and Oscillibacter↓ | Modulated the expression of specific gene related to lipid synthesis and conversion, CYP4A1, ACC, TNF-α, SOCS2 and CYP7A1; reduced hepatocyte steatosis and liver cell injury. | [118] |
| Hyperlipidemia | Auricularia auricular | HFD-induced SD rats | Firmicutes, Roseburia, Flavonifractor and Clostridium IV↑ Bacteroidetes↓ | Reduced the levels of TC and LDL-C; induced the significant growth of SCFA-producing bacteria and the accumulation of SCFAs concentrations. | [119] |
| Obesity | Pleurotus eryngiion | HFD-induced C57BL/6J mice | Lactococcus↑ Roseburia↓ | Suppressed fat accumulation; decreased LDL-C; increased fecal bile acids; increased the concentration of SCFAs. | [120] |
| T2DM | G. frondosa | HFD-induced SD rats | Bacteroidetes/Firmicutes, Lactobacillus and Turicibacter↑ Prevotella and Bifidobacterium↓ | Decreased the expression levels of TNF-α, IL-1β and IL-6; alleviated inflammation by the TLR4/MyD88/NF-κB pathway. | [121] |
| Obesity | Ganoderma lucidum | HFD-induced C57BL/6 mice | Bacteroides spp., Anaerotruncus colihominis, Clostridium↑ Enterococcus spp., Lactococcus lactis and Oscillibacter valericigenes↓ | Improved gut barrier integrity, reduced endotoxemia, decreased TLR4 signal and inflammation; reduced the number of macrophages. | [122] |
| Hypercholesterolemia | Ganoderma lucidum | HCD-induced C57BL/6 mice | Faecalibacterium prausnitzii, Lactobacillus and Prevotella↑ Bacteroides acidifaciens, Mucispirillum schaedleri and Parabacteroides distasonis↓ | Prevented FA synthesis and accumulation through down-modulating genes involved in lipogenesis, elongation and desaturation; activation of PPARs, fatty acid oxidation and bile acid conversion. | [123] |
| Hyperlipidemia | Ganoderma lucidum | HFD-induced Syrian golden hamsters | Ruminococcus, Oscillibacter, Bifidobacterium, Prevotella and Alistipes↑ Desulfovibrio, Clostridium↓ | Alleviated the serum levels of TG, TC, and LDL-C; decreased the serum levels of AST; increased beneficial bacteria and reduced harmful bacteria. | [124] |
4.1.2. Obesity
4.1.3. NAFLD
4.2. Immunoregulation Effects
4.3. Antitumor Effects
4.4. Other Beneficial Effects
5. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Liu, C.; Zhou, N.; Du, M.X.; Sun, Y.T.; Wang, K.; Wang, Y.J.; Li, D.H.; Yu, H.Y.; Song, Y.Q.; Bai, B.B.; et al. The Mouse Gut Microbial Biobank expands the coverage of cultured bacteria. Nat. Commun. 2020, 11, 79. [Google Scholar] [CrossRef] [PubMed]
- Arumugam, M.; Raes, J.; Pelletier, E.; Le Paslier, D.; Yamada, T.; Mende, D.R.; Fernandes, G.R.; Tap, J.; Bruls, T.; Batto, J.M.; et al. Enterotypes of the human gut microbiome. Nature 2011, 473, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Eckburg, P.B.; Bik, E.M.; Bernstein, C.N.; Purdom, E.; Dethlefsen, L.; Sargent, M.; Gill, S.R.; Nelson, K.E.; Relman, D.A. Diversity of the human intestinal microbial flora. Science 2005, 308, 1635–1638. [Google Scholar] [CrossRef] [PubMed]
- Leeming, E.R.; Johnson, A.J.; Spector, T.D.; Le Roy, C.I. Effect of Diet on the Gut Microbiota: Rethinking Intervention Duration. Nutrients 2019, 11, 2862. [Google Scholar] [CrossRef] [PubMed]
- Sonnenburg, J.L.; Backhed, F. Diet-microbiota interactions as moderators of human metabolism. Nature 2016, 535, 56–64. [Google Scholar] [CrossRef]
- Levy, M.; Blacher, E.; Elinav, E. Microbiome, metabolites and host immunity. Curr. Opin. Microbiol. 2017, 35, 8–15. [Google Scholar] [CrossRef]
- Schwalm, N.D.; Groisman, E.A. Navigating the Gut Buffet: Control of Polysaccharide Utilization in Bacteroides spp. Trends Microbiol. 2017, 25, 1005–1015. [Google Scholar] [CrossRef]
- Zhu, Y.; Bai, J.; Zhang, Y.; Xiao, X.; Dong, Y. Effects of bitter melon (Momordica charantia L.) on the gut microbiota in high fat diet and low dose streptozocin-induced rats. Int. J. Food Sci. Nutr. 2016, 67, 686–695. [Google Scholar] [CrossRef]
- Lavelle, A.; Sokol, H. Gut microbiota-derived metabolites as key actors in inflammatory bowel disease. Nat. Rev. Gastro Hepat. 2020, 17, 223–237. [Google Scholar] [CrossRef]
- Canfora, E.E.; Meex, R.C.R.; Venema, K.; Blaak, E.E. Gut microbial metabolites in obesity, NAFLD and T2DM. Nat. Rev. Endocrinol. 2019, 15, 261–273. [Google Scholar] [CrossRef]
- Li, H.Y.; Xie, W.C.; Sun, H.H.; Cao, K.W.; Yang, X.H. Effect of the structural characterization of the fungal polysaccharides on their immunomodulatory activity. Int. J. Biol. Macromol. 2020, 164, 3603–3610. [Google Scholar] [CrossRef]
- Seedevi, P.; Ganesan, A.R.; Mohan, K.; Raguraman, V.; Sivakumar, M.; Sivasankar, P.; Loganathan, S.; Rajamalar, P.; Vairamani, S.; Shanmugam, A.; et al. Chemical structure and biological properties of a polysaccharide isolated from Pleurotus sajor-caju. RSC Adv. 2019, 9, 20472–20482. [Google Scholar] [CrossRef]
- Jayachandran, M.; Xiao, J.B.; Xu, B.J. A Critical Review on Health Promoting Benefits of Edible Mushrooms through Gut Microbiota. Int. J. Mol. Sci. 2017, 18, 1934. [Google Scholar] [CrossRef]
- Li, L.Y.; Guo, Y.N.; Huang, Q.; Shi, X.J.; Liu, Q.Q.; Wang, F.; Liu, Q.F.; Yu, K.; Wang, Z. GPP (composition of Ganoderma lucidum polysaccharides and Polyporus umbellatus polysaccharides) protects against DSS-induced murine colitis by enhancing immune function and regulating intestinal flora. Food Sci. Hum. Wellness 2022, 11, 795–805. [Google Scholar] [CrossRef]
- Zhang, S.S.; Liu, X.Q.; Yan, L.H.; Zhang, Q.W.; Zhu, J.J.; Huang, N.; Wang, Z.M. Chemical Compositions and Antioxidant Activities of Polysaccharides from the Sporophores and Cultured Products of Armillaria mellea. Molecules 2015, 20, 5680–5697. [Google Scholar] [CrossRef]
- Cao, J.P.; Tang, D.D.; Wang, Y.; Li, X.; Hong, L.; Sun, C.D. Characteristics and immune-enhancing activity of pectic polysaccharides from sweet cherry (Prunus avium). Food Chem. 2018, 254, 47–54. [Google Scholar] [CrossRef]
- Chu, Q.; Zhang, Y.R.; Chen, W.; Jia, R.Y.; Yu, X.; Wang, Y.X.; Li, Y.L.; Liu, Y.Y.; Ye, X.; Yu, L.S.; et al. Apios americana Medik flowers polysaccharide (AFP) alleviate Cyclophosphamide-induced immunosuppression in ICR mice. Int. J. Biol. Macromol. 2020, 144, 829–836. [Google Scholar] [CrossRef]
- Cao, Q.Z.; Lin, Z.B. Antitumor and anti-angiogenic activity of Ganoderma lucidum polysaccharides peptide. Acta Pharmacol. Sin. 2004, 25, 833–838. [Google Scholar]
- Liang, Z.E.N.; Yuan, Z.H.; Li, G.Y.; Fu, F.H.; Shan, Y. Hypolipidemic, Antioxidant, and Antiapoptotic Effects of Polysaccharides Extracted from Reishi Mushroom, Ganoderma lucidum (Leysser: Fr) Karst, in Mice Fed a High-Fat Diet. J. Med. Food 2018, 21, 1218–1227. [Google Scholar] [CrossRef]
- Guo, W.L.; Deng, J.C.; Pan, Y.Y.; Xu, J.X.; Hong, J.L.; Shi, F.F.; Liu, G.L.; Qian, M.; Bai, W.D.; Zhang, W.; et al. Hypoglycemic and hypolipidemic activities of Grifola frondosa polysaccharides and their relationships with the modulation of intestinal microflora in diabetic mice induced by high-fat diet and streptozotocin. Int. J. Biol. Macromol. 2020, 153, 1231–1240. [Google Scholar] [CrossRef]
- Chen, X.P.; Chen, Y.; Li, S.B.; Chen, Y.G.; Lan, J.Y.; Liu, L.P. Free radical scavenging of Ganoderma lucidum polysaccharides and its effect on antioxidant enzymes and immunity activities in cervical carcinoma rats. Carbohydr. Polym. 2009, 77, 389–393. [Google Scholar]
- Guo, Y.J.; Deng, G.F.; Xu, X.R.; Wu, S.; Li, S.; Xia, E.Q.; Li, F.; Chen, F.; Ling, W.H.; Li, H.B.; et al. Antioxidant capacities, phenolic compounds and polysaccharide contents of 49 edible macro-fungi. Food Funct. 2012, 3, 1195–1205. [Google Scholar] [CrossRef] [PubMed]
- Mustafa, F.; Chopra, H.; Baig, A.A.; Avula, S.K.; Kumari, S.; Mohanta, T.K.; Saravanan, M.; Mishra, A.K.; Sharma, N.; Mohanta, Y.K.; et al. Edible Mushrooms as Novel Myco-Therapeutics: Effects on Lipid Level, Obesity, and BMI. J. Fungi 2022, 8, 211. [Google Scholar] [CrossRef] [PubMed]
- Huo, W.Y.; Qi, P.; Cui, L.J.; Zhang, L.G.; Dai, L.; Liu, Y.; Hu, S.Y.; Feng, Z.P.; Qiao, T.; Li, J.Z.; et al. Polysaccharide from wild morels alters the spatial structure of gut microbiota and the production of short-chain fatty acids in mice. Biosci. Microbiota Food Health 2020, 39, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Teng, C.Y.; Yu, S.M.; Wang, X.; Liang, J.S.; Bai, X.; Dong, L.Y.; Song, T.; Yu, M.; Qu, J.J.; et al. Inonotus obliquus polysaccharide regulates gut microbiota of chronic pancreatitis in mice. AMB Express 2017, 7, 39. [Google Scholar] [CrossRef]
- Zhao, R.Q.; Hu, Q.H.; Ma, G.X.; Su, A.X.; Xie, M.H.; Li, X.F.; Chen, G.T.; Zhao, L.Y. Effects of Flammulina velutipes polysaccharide on immune response and intestinal microbiota in mice. J. Funct. Foods 2019, 56, 255–264. [Google Scholar] [CrossRef]
- Xu, X.F.; Zhang, X.W. Lentinula edodes-Derived Polysaccharide Alters the Spatial Structure of Gut Microbiota in Mice. PLoS ONE 2015, 10, e0115037. [Google Scholar] [CrossRef]
- Song, X.J.; Feng, Z.F.; Tan, J.B.; Wang, Z.Y.; Zhu, W. Dietary administration of Pleurotus ostreatus polysaccharides (POPS) modulates the non-specific immune response and gut microbiota diversity of Apostichopus japonicus. Aquac. Rep. 2021, 19, 100578. [Google Scholar] [CrossRef]
- Xie, J.L.; Liu, Y.X.; Chen, B.H.; Zhang, G.W.; Ou, S.Y.; Luo, J.M.; Peng, X.C. Ganoderma lucidum polysaccharide improves rat DSS-induced colitis by altering cecal microbiota and gene expression of colonic epithelial cells. Food Nutr. Res. 2019, 63, 1559. [Google Scholar] [CrossRef]
- Zhang, S.; Lei, L.; Zhou, Y.; Ye, F.Y.; Zhao, G.H. Roles of mushroom polysaccharides in chronic disease management. J. Integr. Agric. 2022, 21, 1839–1866. [Google Scholar] [CrossRef]
- Li, Q.; Zhang, F.; Chen, G.; Chen, Y.; Zhang, W.; Mao, G.; Zhao, T.; Zhang, M.; Yang, L.; Wu, X.; et al. Purification, characterization and immunomodulatory activity of a novel polysaccharide from Grifola frondosa. Int. J. Biol. Macromol. 2018, 111, 1293–1303. [Google Scholar] [CrossRef]
- Wang, M.; Gao, Y.; Xu, D.; Gao, Q. A polysaccharide from cultured mycelium of Hericium erinaceus and its anti-chronic atrophic gastritis activity. Int. J. Biol. Macromol. 2015, 81, 656–661. [Google Scholar] [CrossRef]
- Li, Y.Q.; Fang, L.; Zhang, K.C. Structure and bioactivities of a galactose rich extracellular polysaccharide from submergedly cultured Ganoderma lucidum. Carbohydr. Polym. 2007, 68, 323–328. [Google Scholar] [CrossRef]
- Xu, D.D.; Wang, H.Y.; Zheng, W.; Gao, Y.; Wang, M.X.; Zhang, Y.Q.; Gao, Q.P. Charaterization and immunomodulatory activities of polysaccharide isolated from Pleurotus eryngii. Int. J. Biol. Macromol. 2016, 92, 30–36. [Google Scholar] [CrossRef]
- Zhang, Y.Y.; Xu, X.J.; Zhang, L. Gel formation and low-temperature intramolecular conformation transition of a triple-helical polysaccharide lentinan in water. Biopolymers 2008, 89, 852–861. [Google Scholar] [CrossRef]
- Xu, X.F.; Yan, H.D.; Zhang, X.W. Structure and Immuno-Stimulating Activities of a New Heteropolysaccharide from Lentinula edodes. J. Agric. Food Chem. 2012, 60, 11560–11566. [Google Scholar] [CrossRef]
- Xu, X.F.; Yang, J.G.; Ning, Z.X.; Zhang, X.W. Lentinula edodes-derived polysaccharide rejuvenates mice in terms of immune responses and gut microbiota. Food Funct. 2015, 6, 2653–2663. [Google Scholar] [CrossRef]
- Zhou, R.; Wang, Y.T.; Li, C.; Jia, S.T.; Shi, Y.A.; Tang, Y.F.; Li, Y.Q. A preliminary study on preparation, characterization, and prebiotic activity of a polysaccharide from the edible mushroom Ramaria flava. J. Food Biochem. 2022, 46, e14371. [Google Scholar] [CrossRef]
- Tian, B.M.; Geng, Y.; Xu, T.R.; Zou, X.G.; Mao, R.L.; Pi, X.G.; Wu, W.C.; Huang, L.S.; Yang, K.; Zeng, X.X.; et al. Digestive Characteristics of Hericium erinaceus Polysaccharides and Their Positive Effects on Fecal Microbiota of Male and Female Volunteers During in vitro Fermentation. Front. Nutr. 2022, 9, 858585. [Google Scholar] [CrossRef]
- Abdureyim, Z.; Wang, L.; Tao, Q.; Xu, J.; Yimiti, D.; Gao, Q. Bachu Mushroom Polysaccharide Alleviates Colonic Injury by Modulating the Gut Microbiota. Comput. Math. Methods Med. 2022, 2022, 1353724. [Google Scholar] [CrossRef]
- Pan, W.; Jiang, P.F.; Zhao, J.X.; Shi, H.L.; Zhang, P.; Yang, X.Y.; Biazik, J.; Hu, M.M.; Hua, H.; Ge, X.; et al. β-Glucan from Lentinula edodes prevents cognitive impairments in high-fat diet-induced obese mice: Involvement of colon-brain axis. J. Transl. Med. 2021, 19, 54. [Google Scholar] [CrossRef] [PubMed]
- Rehman, A.U.; Siddiqui, N.Z.; Farooqui, N.A.; Alam, G.; Gul, A.; Ahmad, B.; Asim, M.; Khan, A.I.; Xin, Y.; Zexu, W.; et al. Morchella esculenta mushroom polysaccharide attenuates diabetes and modulates intestinal permeability and gut microbiota in a type 2 diabetic mice model. Front. Nutr. 2022, 9, 984695. [Google Scholar] [CrossRef] [PubMed]
- Ma, G.X.; Xu, Q.; Du, H.J.; Kimatu, B.M.; Su, A.X.; Yang, W.J.; Hu, Q.H.; Xiao, H. Characterization of polysaccharide from Pleurotus eryngii during simulated gastrointestinal digestion and fermentation. Food Chem. 2022, 370, 131303. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.Y.; Hu, B.; Liu, C.; Hua, H.Y.; Guo, Y.H.; Cheng, Y.L.; Yao, W.R.; Qian, H. Comprehensive analysis of Sparassis crispa polysaccharide characteristics during the in vitro digestion and fermentation model. Food Res. Int. 2022, 154, 111005. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.Y.; Hu, B.; Han, M.; Guo, Y.H.; Cheng, Y.L.; Qian, H. Purification, structural characterization and neuroprotective effect of a neutral polysaccharide from Sparassis crispa. Int. J. Biol. Macromol. 2022, 201, 389–399. [Google Scholar] [CrossRef]
- Xu, Y.Y.; Xie, L.Y.; Zhang, Z.Y.; Zhang, W.W.; Tang, J.; He, X.L.; Zhou, J.; Peng, W.H. Tremella fuciformis Polysaccharides Inhibited Colonic Inflammation in Dextran Sulfate Sodium-Treated Mice via Foxp3+T Cells, Gut Microbiota, and Bacterial Metabolites. Front. Immunol. 2021, 12, 648162. [Google Scholar] [CrossRef]
- Wang, T.; Han, J.J.; Dai, H.Q.; Sun, J.Z.; Ren, J.W.; Wang, W.Z.; Qiao, S.S.; Liu, C.; Sun, L.; Liu, S.J.; et al. Polysaccharides from Lyophyllum decastes reduce obesity by altering gut microbiota and increasing energy expenditure. Carbohydr. Polym. 2022, 295, 119862. [Google Scholar] [CrossRef]
- Yang, L.; Kang, X.C.; Dong, W.J.; Wang, L.; Liu, S.F.; Zhong, X.H.; Liu, D.B. Prebiotic properties of Ganoderma lucidum polysaccharides with special enrichment of Bacteroides ovatus and B. uniformis in vitro. J. Funct. Foods 2022, 92, 105069. [Google Scholar] [CrossRef]
- Pan, M.C.; Kong, F.G.; Xing, L.; Yao, L.; Li, Y.; Liu, Y.; Li, C.T.; Li, L.Z. The Structural Characterization and Immunomodulatory Activity of Polysaccharides from Pleurotus abieticola Fruiting Bodies. Nutrients 2022, 14, 4410. [Google Scholar] [CrossRef]
- Huo, W.Y.; Feng, Z.P.; Hu, S.Y.; Cui, L.J.; Qiao, T.; Dai, L.; Qi, P.; Zhang, L.G.; Liu, Y.; Li, J.Z.; et al. Effects of polysaccharides from wild morels on immune response and gut microbiota composition in non-treated and cyclophosphamide-treated mice. Food Funct. 2020, 11, 4291–4303. [Google Scholar] [CrossRef]
- Chen, M.Y.; Xiao, D.; Liu, W.; Song, Y.F.; Zou, B.R.; Li, L.; Li, P.; Cai, Y.; Liu, D.L.; Liao, Q.F.; et al. Intake of Ganoderma lucidum polysaccharides reverses the disturbed gut microbiota and metabolism in type 2 diabetic rats. Int. J. Biol. Macromol. 2020, 155, 890–902. [Google Scholar] [CrossRef]
- Zhao, D.; Dai, W.; Tao, H.; Zhuang, W.; Qu, M.; Chang, Y.N. Polysaccharide isolated from Auricularia auricular-judae (Bull.) prevents dextran sulfate sodium-induced colitis in mice through modulating the composition of the gut microbiota. J. Food Sci. 2020, 85, 2943–2951. [Google Scholar] [CrossRef]
- Kanwal, S.; Aliya, S.; Xin, Y. Anti-Obesity Effect of Dictyophora indusiata Mushroom Polysaccharide (DIP) in High Fat Diet-Induced Obesity via Regulating Inflammatory Cascades and Intestinal Microbiome. Front. Endocrinol. 2020, 11, 558874. [Google Scholar] [CrossRef]
- Zhang, R.; Yuan, S.; Ye, J.; Wang, X.; Zhang, X.; Shen, J.; Yuan, M.; Liao, W. Polysaccharide from Flammuliana velutipes improves colitis via regulation of colonic microbial dysbiosis and inflammatory responses. Int. J. Biol. Macromol. 2020, 149, 1252–1261. [Google Scholar] [CrossRef]
- Xue, Z.; Ma, Q.; Chen, Y.; Lu, Y.; Wang, Y.; Jia, Y.; Zhang, M.; Chen, H. Structure characterization of soluble dietary fiber fractions from mushroom Lentinula edodes (Berk.) Pegler and the effects on fermentation and human gut microbiota in vitro. Food Res. Int. 2020, 129, 108870. [Google Scholar] [CrossRef]
- Zhao, R.Q.; Fang, D.G.; Ji, Y.; Chen, X.; Ma, G.X.; Su, A.X.; Xie, M.H.; Zhao, L.Y.; Hu, Q.H. In vitro and in vivo functional characterization of an immune activation Flammulina velutipes polysaccharide based on gut microbiota regulation. Food Agric. Immunol. 2020, 31, 667–686. [Google Scholar] [CrossRef]
- Kanwal, S.; Joseph, T.P.; Aliya, S.; Song, S.Y.; Saleem, M.Z.; Nisar, M.A.; Wang, Y.; Meyiah, A.; Ma, Y.F.; Xin, Y.; et al. Attenuation of DSS induced colitis by Dictyophora indusiata polysaccharide (DIP) via modulation of gut microbiota and inflammatory related signaling pathways. J. Funct. Foods 2020, 64, 103641. [Google Scholar] [CrossRef]
- Zhao, R.Q.; Cheng, N.H.; Nakata, P.A.; Zhao, L.Y.; Hu, Q.H. Consumption of polysaccharides from Auricularia auricular modulates the intestinal microbiota in mice. Food Res. Int. 2019, 123, 383–392. [Google Scholar] [CrossRef]
- Li, L.; Guo, W.L.; Zhang, W.; Xu, J.X.; Qian, M.; Bai, W.D.; Zhang, Y.Y.; Rao, P.F.; Ni, L.; Lv, X.C. Grifola frondosa polysaccharides ameliorate lipid metabolic disorders and gut microbiota dysbiosis in high-fat diet fed rats. Food Funct. 2019, 10, 2560–2572. [Google Scholar] [CrossRef]
- Chen, Y.Q.; Liu, D.; Wang, D.Y.; Lai, S.S.; Zhong, R.T.; Liu, Y.Y.; Yang, C.F.; Liu, B.; Sarker, M.R.; Zhao, C.; et al. Hypoglycemic activity and gut microbiota regulation of a novel polysaccharide from Grifola frondosa in type 2 diabetic mice. Food Chem. Toxicol. 2019, 126, 295–302. [Google Scholar] [CrossRef]
- Kanwal, S.; Joseph, T.P.; Owusu, L.; Ren, X.M.; Li, M.Q.; Xin, Y. A Polysaccharide Isolated from Dictyophora indusiata Promotes Recovery from Antibiotic-Driven Intestinal Dysbiosis and Improves Gut Epithelial Barrier Function in a Mouse Model. Nutrients 2018, 10, 1003. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.L.; Geng, Y.; Du, Y.; Li, W.; Lu, Z.M.; Xu, H.Y.; Xu, G.H.; Shi, J.S.; Xu, Z.H. Polysaccharide of Hericium erinaceus attenuates colitis in C57BL/6 mice via regulation of oxidative stress, inflammation-related signaling pathways and modulating the composition of the gut microbiota. J. Nutr. Biochem. 2018, 57, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Mallard, B.; Leach, D.N.; Wohlmuth, H.; Tiralongo, J. Synergistic immuno-modulatory activity in human macrophages of a medicinal mushroom formulation consisting of Reishi, Shiitake and Maitake. PLoS ONE 2019, 14, e0224740. [Google Scholar] [CrossRef] [PubMed]
- Friedman, M. Mushroom Polysaccharides: Chemistry and Antiobesity, Antidiabetes, Anticancer, and Antibiotic Properties in Cells, Rodents, and Humans. Foods 2016, 5, 80. [Google Scholar] [CrossRef] [PubMed]
- Cerletti, C.; Esposito, S.; Iacoviello, L. Edible Mushrooms and Beta-Glucans: Impact on Human Health. Nutrients 2021, 13, 2195. [Google Scholar] [CrossRef]
- Ranjbar, R.; Vahdati, S.N.; Tavakoli, S.; Khodaie, R.; Behboudi, H. Immunomodulatory roles of microbiota-derived short-chain fatty acids in bacterial infections. Biomed. Pharmacother. 2021, 141, 111817. [Google Scholar] [CrossRef]
- Jayachandran, M.; Chen, J.L.; Chung, S.S.M.; Xu, B.J. A critical review on the impacts of beta-glucans on gut microbiota and human health. J. Nutr. Biochem. 2018, 61, 101–110. [Google Scholar] [CrossRef]
- Saxami, G.; Kerezoudi, E.N.; Mitsou, E.K.; Koutrotsios, G.; Zervakis, G.I.; Pletsa, V.; Kyriacou, A. Fermentation Supernatants of Pleurotus eryngii Mushroom Ameliorate Intestinal Epithelial Barrier Dysfunction in Lipopolysaccharide-Induced Caco-2 Cells via Upregulation of Tight Junctions. Microorganisms 2021, 9, 2071. [Google Scholar] [CrossRef]
- Wang, G.L.; Zhang, Y.L.; Zhang, R.G.; Pan, J.L.; Qi, D.F.; Wang, J.; Yang, X.Y. The protective effects of walnut green husk polysaccharide on liver injury, vascular endothelial dysfunction and disorder of gut microbiota in high fructose-induced mice. Int. J. Biol. Macromol. 2020, 162, 92–106. [Google Scholar] [CrossRef]
- Luo, J.M.; Zhang, C.; Liu, R.; Gao, L.J.; Ou, S.Y.; Liu, L.; Peng, X.C. Ganoderma lucidum polysaccharide alleviating colorectal cancer by alteration of special gut bacteria and regulation of gene expression of colonic epithelial cells. J. Funct. Foods 2018, 47, 127–135. [Google Scholar] [CrossRef]
- Sun, S.S.; Wang, K.; Ma, K.; Bao, L.; Liu, H.W. An insoluble polysaccharide from the sclerotium of Poria cocos improves hyperglycemia, hyperlipidemia and hepatic steatosis in ob/ob mice via modulation of gut microbiota. Chin. J. Nat. Med. 2019, 17, 3–14. [Google Scholar] [CrossRef]
- He, X.R.; Wang, X.X.; Fang, J.C.; Chang, Y.; Ning, N.; Guo, H.; Huang, L.H.; Huang, X.Q.; Zhao, Z.F. Polysaccharides in Grifola frondosa mushroom and their health promoting properties: A review. Int. J. Biol. Macromol. 2017, 101, 910–921. [Google Scholar] [CrossRef]
- Han, X.Q.; Yue, G.L.; Yue, R.Q.; Dong, C.X.; Chan, C.L.; Ko, C.H.; Cheung, W.S.; Luo, K.W.; Dai, H.; Wong, C.K.; et al. Structure Elucidation and Immunomodulatory Activity of A Beta Glucan from the Fruiting Bodies of Ganoderma Sinense. PLoS ONE 2014, 9, 100380. [Google Scholar] [CrossRef]
- Gao, X.; Qi, J.Y.; Ho, C.T.; Li, B.; Mu, J.J.; Zhang, Y.T.; Hu, H.P.; Mo, W.P.; Chen, Z.Z.; Xie, Y.Z.; et al. Structural characterization and immunomodulatory activity of a water-soluble polysaccharide from Ganoderma leucocontextum fruiting bodies. Carbohydr. Polym. 2020, 249, 116874. [Google Scholar] [CrossRef]
- Xu, X.F.; Yan, H.D.; Tang, J.; Chen, J.; Zhang, X.W. Polysaccharides in Lentinus edodes: Isolation, Structure, Immunomodulating Activity and Future Prospective. Crit. Rev. Food Sci. Nutr. 2014, 54, 474–487. [Google Scholar] [CrossRef]
- Su, A.X.; Ma, G.X.; Xie, M.H.; Ji, Y.; Li, X.F.; Zhao, L.Y.; Hu, Q.H. Characteristic of polysaccharides from Flammulina velutipes in vitro digestion under salivary, simulated gastric and small intestinal conditions and fermentation by human gut microbiota. J. Food Sci. Technol. 2019, 54, 2277–2287. [Google Scholar] [CrossRef]
- Cantarel, B.L.; Lombard, V.; Henrissat, B. Complex Carbohydrate Utilization by the Healthy Human Microbiome. PLoS ONE 2012, 7, e28742. [Google Scholar] [CrossRef]
- Salyers, A.A.; Vercellotti, J.R.; West, S.E.; Wilkins, T.D. Fermentation of mucin and plant polysaccharides by strains of Bacteroides from the human colon. Appl. Environ. Microbiol. 1977, 33, 319–322. [Google Scholar] [CrossRef]
- Su, A.X.; Yang, W.J.; Zhao, L.Y.; Pei, F.; Yuan, B.; Zhong, L.; Ma, G.X.; Hu, Q.H. Flammulina velutipes polysaccharides improve scopolamine-induced learning and memory impairment in mice by modulating gut microbiota composition. Food Funct. 2018, 9, 1424–1432. [Google Scholar] [CrossRef]
- Ravcheev, D.A.; Godzik, A.; Osterman, A.L.; Rodionov, D.A. Polysaccharides utilization in human gut bacterium Bacteroides thetaiotaomicron: Comparative genomics reconstruction of metabolic and regulatory networks. BMC Genom. 2013, 14, 873. [Google Scholar] [CrossRef]
- Wardman, J.F.; Bains, R.K.; Rahfeld, P.; Withers, S.G. Carbohydrate-active enzymes (CAZymes) in the gut microbiome. Nat. Rev. Microbiol. 2022, 20, 542–556. [Google Scholar] [CrossRef]
- White, B.A.; Lamed, R.; Bayer, E.A.; Flint, H.J. Biomass utilization by gut microbiomes. Annu. Rev. Microbiol. 2014, 68, 279–296. [Google Scholar] [CrossRef] [PubMed]
- Lombard, V.; Bernard, T.; Rancurel, C.; Brumer, H.; Coutinho, P.M.; Henrissat, B. A hierarchical classification of polysaccharide lyases for glycogenomics. Biochem. J. 2010, 432, 437–444. [Google Scholar] [CrossRef] [PubMed]
- Biely, P. Microbial carbohydrate esterases deacetylating plant polysaccharides. Biotechnol. Adv. 2012, 30, 1575–1588. [Google Scholar] [CrossRef] [PubMed]
- El Kaoutari, A.; Armougom, F.; Gordon, J.I.; Raoult, D.; Henrissat, B. The abundance and variety of carbohydrate-active enzymes in the human gut microbiota. Nat. Rev. Microbiol. 2013, 11, 497–504. [Google Scholar] [CrossRef]
- Rakoff-Nahoum, S.; Foster, K.R.; Comstock, L.E. The evolution of cooperation within the gut microbiota. Nature 2016, 533, 255–259. [Google Scholar] [CrossRef]
- Yonit, B.D.; Dassa, B.; Borovok, I.; Lamed, R.; Koropatkin, N.M.; Martens, E.C.; White, B.A.; Bernalier-Donadille, A.; Duncan, S.H.; Flint, H.J.; et al. Ruminococcal cellulosome systems from rumen to human. Environ. Microbiol. 2015, 17, 3407–3426. [Google Scholar]
- Cockburn, D.W.; Orlovsky, N.I.; Foley, M.H.; Kwiatkowski, K.J.; Bahr, C.M.; Maynard, M.; Demeler, B.; Koropatkin, N.M. Molecular details of a starch utilization pathway in the human gut symbiont Eubacterium rectale. Mol. Microbiol. 2015, 95, 209–230. [Google Scholar] [CrossRef]
- Singh, R.P. Glycan utilisation system in Bacteroides and Bifidobacteria and their roles in gut stability and health. Appl. Microbiol. Biotechnol. 2019, 103, 7287–7315. [Google Scholar] [CrossRef]
- Turroni, F.; Milani, C.; Duranti, S.; Mahony, J.; van Sinderen, D.; Ventura, M. Glycan Utilization and Cross-Feeding Activities by Bifidobacteria. Trends Microbiol. 2018, 26, 339–350. [Google Scholar] [CrossRef]
- Zhang, T.H.; Yang, Y.; Liang, Y.; Jiao, X.; Zhao, C.H. Beneficial Effect of Intestinal Fermentation of Natural Polysaccharides. Nutrients 2018, 10, 1055. [Google Scholar] [CrossRef]
- Fierobe, H.P.; Bayer, E.A.; Tardif, C.; Czjzek, M.; Mechaly, A.; Belaich, A.; Lamed, R.; Shoham, Y.; Belaich, J.P. Degradation of cellulose substrates by cellulosome chimeras-Substrate targeting versus proximity of enzyme components. J. Biol. Chem. 2002, 277, 49621–49630. [Google Scholar] [CrossRef]
- Wang, Q.; Wu, X.Y.; Shi, F.L.; Liu, Y. Comparison of antidiabetic effects of saponins and polysaccharides from Momordica charantia L. in STZ-induced type 2 diabetic mice. Biomed. Pharmacother. 2019, 109, 744–750. [Google Scholar] [CrossRef]
- Songer, T.J.; Zimmet, P.Z. Epidemiology of type II diabetes: An international perspective. Pharmacoeconomics 1995, 8, 1–11. [Google Scholar] [CrossRef]
- Nasr, N.E.; Sadek, K.M. Role and mechanism(s) of incretin-dependent therapies for treating diabetes mellitus. Environ. Sci. Pollut. Res. 2022, 29, 18408–18422. [Google Scholar] [CrossRef]
- Khursheed, R.; Singh, S.K.; Wadhwa, S.; Gulati, M.; Awasthi, A. Therapeutic potential of mushrooms in diabetes mellitus: Role of polysaccharides. Int. J. Biol. Macromol. 2020, 164, 1194–1205. [Google Scholar] [CrossRef]
- Depommier, C.; Everard, A.; Druart, C.; Plovier, H.; van Hul, M.; Vieira-Silva, S.; Falony, G.; Raes, J.; Maiter, D.; Delzenne, N.M.; et al. Supplementation with Akkermansia muciniphila in overweight and obese human volunteers: A proof-of-concept exploratory study. Nat. Med. 2019, 25, 1096–1103. [Google Scholar] [CrossRef]
- Yang, R.; Li, Y.D.; Mehmood, S.; Yan, C.C.; Huang, Y.Z.; Cai, J.J.; Ji, J.Q.; Pan, W.J.; Zhang, W.N.; Chen, Y.; et al. Polysaccharides from Armillariella tabescens mycelia ameliorate renal damage in type 2 diabetic mice. Int. J. Biol. Macromol. 2020, 162, 1682–1691. [Google Scholar] [CrossRef]
- Yi, H.B.; Wang, L.; Xiong, Y.X.; Wang, Z.L.; Qiu, Y.Q.; Wen, X.L.; Jiang, Z.Y.; Yang, X.F.; Ma, X.Y. Lactobacillus reuteri LR1 Improved Expression of Genes of Tight Junction Proteins via the MLCK Pathway in IPEC-1 Cells during Infection with Enterotoxigenic Escherichia coli K88. Mediat. Inflamm. 2018, 2018, 6434910. [Google Scholar] [CrossRef]
- Jang, Y.J.; Kim, W.K.; Han, D.H.; Lee, K.; Ko, G. Lactobacillus fermentum species ameliorate dextran sulfate sodium-induced colitis by regulating the immune response and altering gut microbiota. Gut Microbes 2019, 10, 696–711. [Google Scholar] [CrossRef]
- Shao, W.M.; Xiao, C.; Yong, T.Q.; Zhang, Y.F.; Hu, H.P.; Xie, T.; Liu, R.J.; Huang, L.H.; Li, X.M.; Xie, Y.Z.; et al. A polysaccharide isolated from Ganoderma lucidum ameliorates hyperglycemia through modulating gut microbiota in type 2 diabetic mice. Int. J. Biol. Macromol. 2022, 197, 23–38. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.M.; Zhang, N.; Zhou, W.; Zhou, Z.K.; Bai, Y.; Strappe, P.; Blanchard, C. Manipulations of glucose/lipid metabolism and gut microbiota of resistant starch encapsulated Ganoderma lucidum spores in T2DM rats. Food Sci. Biotechnol. 2021, 30, 755–764. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.L.; Chen, M.; Pan, W.L.; Zhang, Q.; Xu, J.X.; Lin, Y.C.; Li, L.; Liu, B.; Bai, W.D.; Zhang, Y.Y.; et al. Hypoglycemic and hypolipidemic mechanism of organic chromium derived from chelation of Grifola frondosa polysaccharide-chromium (III) and its modulation of intestinal microflora in high fat-diet and STZ-induced diabetic mice. Int. J. Biol. Macromol. 2020, 145, 1208–1218. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, T.; Mori, K.; Ouchi, K.; Kushida, M.; Tsuduki, T. Effects of Dietary Intake of Japanese Mushrooms on Visceral Fat Accumulation and Gut Microbiota in Mice. Nutrients 2018, 10, 610. [Google Scholar] [CrossRef] [PubMed]
- Kilner, J.; Corfe, B.M.; McAuley, M.T.; Wilkinson, S.J. A deterministic oscillatory model of microtubule growth and shrinkage for differential actions of short chain fatty acids. Mol. BioSystems 2016, 12, 93–101. [Google Scholar] [CrossRef]
- Liu, Y.M.; Liu, W.; Li, J.; Tang, S.; Wang, M.J.; Huang, W.H.; Yao, W.B.; Gao, X.D. A polysaccharide extracted from Astragalus membranaceus residue improves cognitive dysfunction by altering gut microbiota in diabetic mice. Carbohydr. Polym. 2019, 205, 500–512. [Google Scholar] [CrossRef]
- Sang, T.T.; Guo, C.J.; Guo, D.D.; Wu, J.J.; Wang, Y.J.; Wang, Y.; Chen, J.J.; Chen, C.J.; Wu, K.K.; Na, K.; et al. Suppression of obesity and inflammation by polysaccharide from sporoderm-broken spore of Ganoderma lucidum via gut microbiota regulation. Carbohydr. Polym. 2021, 256, 117594. [Google Scholar] [CrossRef]
- Waddell, I.S.; Orfila, C. Dietary fiber in the prevention of obesity and obesity-related chronic diseases: From epidemiological evidence to potential molecular mechanisms. Crit. Rev. Food Sci. Nutr. 2022, 26, 1–16. [Google Scholar] [CrossRef]
- Feng, H.; Zhang, S.J.; Wan, J.M.F.; Gui, L.F.; Ruan, M.C.; Li, N.; Zhang, H.Y.; Liu, Z.G.; Wang, H.L. Polysaccharides extracted from Phellinus linteus ameliorate high-fat high-fructose diet induced insulin resistance in mice. Carbohydr. Polym. 2018, 200, 144–153. [Google Scholar] [CrossRef]
- Liu, N.N.; Chen, X.F.; Song, J.N.; Chen, M.Y.; Gong, P.; Jia, W.; Li, G.L. Hypoglycemic effects of Auricularia auricula polysaccharides on high fat diet and streptozotocin-induced diabetic mice using metabolomics analysis. Food Funct. 2021, 12, 9994–10007. [Google Scholar] [CrossRef]
- Xu, N.; Zhou, Y.J.; Lu, X.Y.; Chang, Y.N. Auricularia auricula-judae (Bull.) polysaccharides improve type 2 diabetes in HFD/STZ-induced mice by regulating the AKT/AMPK signaling pathways and the gut microbiota. J. Food Sci. 2021, 86, 5479–5494. [Google Scholar] [CrossRef]
- Wang, C.E.; Zeng, F.; Liu, Y.L.; Pan, Y.Y.; Xu, J.X.; Ge, X.D.; Zheng, H.P.; Pang, J.; Liu, B.; Huang, Y.; et al. Coumarin-rich Grifola frondosa ethanol extract alleviate lipid metabolism disorders and modulates intestinal flora compositions of high-fat diet rats. J. Funct. Foods 2021, 85, 104649. [Google Scholar] [CrossRef]
- Gao, X.X.; Liu, D.; Gao, L.Y.; Ouyang, Y.Z.; Wen, Y.X.; Ai, C.; Chen, Y.Q.; Zhao, C. Health benefits of Grifola frondosa polysaccharide on intestinal microbiota in type 2 diabetic mice. Food Sci. Hum. Wellness 2022, 11, 68–73. [Google Scholar] [CrossRef]
- Zhu, Z.J.; Huang, R.; Huang, A.H.; Wang, J.; Liu, W.; Wu, S.J.; Chen, M.F.; Chen, M.T.; Xie, Y.Z.; Jiao, C.W.; et al. Polysaccharide from Agrocybe cylindracea prevents diet-induced obesity through inhibiting inflammation mediated by gut microbiota and associated metabolites. Int. J. Biol. Macromol. 2022, 209, 1430–1438. [Google Scholar] [CrossRef]
- Huang, Z.R.; Zhao, L.Y.; Zhu, F.R.; Liu, Y.; Xiao, J.Y.; Chen, Z.C.; Lv, X.C.; Huang, Y.; Liu, B. Anti-Diabetic Effects of Ethanol Extract from Sanghuangporous vaninii in High-Fat/Sucrose Diet and Streptozotocin-Induced Diabetic Mice by Modulating Gut Microbiota. Foods 2022, 11, 974. [Google Scholar] [CrossRef]
- Jiang, X.; Hao, J.; Liu, Z.J.; Ma, X.T.; Feng, Y.X.; Teng, L.R.; Li, Y.; Wang, D. Anti-obesity effects of Grifola frondosa through the modulation of lipid metabolism via ceramide in mice fed a high-fat diet. Food Funct. 2021, 12, 6725–6739. [Google Scholar] [CrossRef]
- Yang, X.Y.; Zheng, M.X.; Zhou, M.L.; Zhou, L.M.; Ge, X.; Pang, N.; Li, H.C.; Li, X.Y.; Li, M.D.; Zhang, J.; et al. Lentinan Supplementation Protects the Gut-Liver Axis and Prevents Steatohepatitis: The Role of Gut Microbiota Involved. Front. Nutr. 2022, 8, 803691. [Google Scholar] [CrossRef]
- Li, X.; Zeng, F.; Huang, Y.F.; Liu, B. The Positive Effects of Grifola frondosa Heteropolysaccharide on NAFLD and Regulation of the Gut Microbiota. Int. J. Mol. Sci. 2019, 20, 5302. [Google Scholar] [CrossRef]
- Zhang, T.T.; Zhao, W.Y.; Xie, B.Z.; Liu, H. Effects of Auricularia auricula and its polysaccharide on diet-induced hyperlipidemia rats by modulating gut microbiota. J. Funct. Foods 2020, 72, 104038. [Google Scholar] [CrossRef]
- Nakahara, D.; Nan, C.; Mori, K.; Hanayama, M.; Kikuchi, H.; Hirai, S.; Egashira, Y. Effect of mushroom polysaccharides from Pleurotus eryngiion obesity and gut microbiota in mice fed a high-fat diet. Eur. J. Nutr. 2020, 59, 3231–3244. [Google Scholar] [CrossRef]
- Xiao, C.; Jiao, C.W.; Xie, Y.Z.; Ye, L.H.; Li, Q.Q.; Wu, Q.P. Grifola frondosa GF5000 improves insulin resistance by modulation the composition of gut microbiota in diabetic rats. J. Funct. Foods 2021, 77, 104313. [Google Scholar] [CrossRef]
- Chang, C.J.; Lin, C.S.; Lu, C.C.; Martel, J.; Ko, Y.F.; Ojcius, D.M.; Tseng, S.F.; Wu, T.R.; Chen, Y.Y.M.; Young, J.D.; et al. Ganoderma lucidum reduces obesity in mice by modulating the composition of the gut microbiota. Nat. Commun. 2015, 6, 7489. [Google Scholar] [CrossRef] [PubMed]
- Romero-Cordoba, S.L.; Salido-Guadarrama, I.; Meneses, M.E.; Cosentino, G.; Iorio, M.V.; Tagliabue, E.; Torres, N.; Sanchez-Tapia, M.; Bonilla, M.; Castillo, I.; et al. Mexican Ganoderma Lucidum Extracts Decrease Lipogenesis Modulating Transcriptional Metabolic Networks and Gut Microbiota in C57BL/6 Mice Fed with a High-Cholesterol Diet. Nutrients 2021, 13, 38. [Google Scholar] [CrossRef] [PubMed]
- Tong, A.J.; Hu, R.K.; Wu, L.X.; Lv, X.C.; Li, X.; Zhao, L.N.; Liu, B. Ganoderma polysaccharide and chitosan synergistically ameliorate lipid metabolic disorders and modulate gut microbiota composition in high fat diet-fed golden hamsters. J. Food Biochem. 2020, 44, e13109. [Google Scholar] [CrossRef] [PubMed]
- Pung, H.C.; Lin, W.S.; Lo, Y.C.; Hsu, C.C.; Ho, C.T.; Pan, M.H. Ulva prolifera polysaccharide exerts anti-obesity effects via upregulation of adiponectin expression and gut microbiota modulation in high-fat diet-fed C57BL/6 mice. J. Food Drug Anal. 2022, 30, 46–61. [Google Scholar] [CrossRef]
- Gurevich-Panigrahi, T.; Panigrahi, S.; Wiechec, E.; Los, M. Obesity: Pathophysiology and Clinical Management. Curr. Med. Chem. 2009, 16, 506–521. [Google Scholar] [CrossRef]
- Hale, C.; Veniant, M.M. Growth differentiation factor 15 as a potential therapeutic for treating obesity. Mol. Metab. 2021, 46, 101117. [Google Scholar] [CrossRef]
- Ridaura, V.K.; Faith, J.J.; Rey, F.E.; Cheng, J.Y.; Duncan, A.E.; Kau, A.L.; Griffin, N.W.; Lombard, V.; Henrissat, B.; Bain, J.R.; et al. Gut Microbiota from Twins Discordant for Obesity Modulate Metabolism in Mice. Science 2013, 341, 1241214. [Google Scholar] [CrossRef]
- Brun, P.; Castagliuolo, I.; di Leo, V.; Buda, A.; Pinzani, M.; Palu, G.; Martines, D. Increased intestinal permeability in obese mice: New evidence in the pathogenesis of nonalcoholic steatohepatitis. Am. J. Physiol. Gastrointest. Liver Physiol. 2007, 292, G518–G525. [Google Scholar] [CrossRef]
- De Vadder, F.; Kovatcheva-Datchary, P.; Goncalves, D.; Vinera, J.; Zitoun, C.; Duchampt, A.; Backhed, F.; Mithieux, G. Microbiota-Generated Metabolites Promote Metabolic Benefits via Gut-Brain Neural Circuits. Cell 2014, 156, 84–96. [Google Scholar] [CrossRef]
- Li, R.L.; Xue, Z.H.; Li, S.Q.; Zhou, J.N.; Liu, J.Y.; Zhang, M.; Panichayupakaranant, P.; Chen, H.X. Mulberry leaf polysaccharides ameliorate obesity through activation of brown adipose tissue and modulation of the gut microbiota in high-fat diet fed mice. Food Funct. 2022, 13, 561–573. [Google Scholar] [CrossRef]
- Den Besten, G.; Bleeker, A.; Gerding, A.; van Eunen, K.; Havinga, R.; van Dijk, T.H.; Oosterveer, M.H.; Jonker, J.W.; Groen, A.K.; Reijngoud, D.J.; et al. Short-Chain Fatty Acids Protect Against High-Fat Diet-Induced Obesity via a PPAR-Dependent Switch from Lipogenesis to Fat Oxidation. Diabetes 2015, 64, 2398–2408. [Google Scholar] [CrossRef]
- Liu, H.; Wang, J.; He, T.; Becker, S.; Zhang, G.L.; Li, D.F.; Ma, X. Butyrate: A Double-Edged Sword for Health? Adv. Nutr. 2018, 9, 21–29. [Google Scholar] [CrossRef]
- Mfopa, A.; Mediesse, F.K.; Mvongo, C.; Nkoubatchoundjwen, S.; Lum, A.A.; Sobngwi, E.; Kamgang, R.; Boudjeko, T. Antidyslipidemic Potential of Water-Soluble Polysaccharides of Ganoderma applanatum in MACAPOS-2-Induced Obese Rats. Evid. Based Complement. Altern. Med. 2021, 18, 2452057. [Google Scholar] [CrossRef]
- Ma, G.X.; Kimatu, B.M.; Zhao, L.Y.; Yang, W.J.; Pei, F.; Hu, Q.H. In vivo fermentation of a Pleurotus eryngii polysaccharide and its effects on fecal microbiota composition and immune response. Food Funct. 2017, 8, 1810–1821. [Google Scholar] [CrossRef]
- Aranaz, P.; Pena, A.; Vettorazzi, A.; Fabra, M.J.; Martinez-Abad, A.; Lopez-Rubio, A.; Pera, J.; Parlade, J.; Castellari, M.; Milagro, F.I.; et al. Grifola frondosa (Maitake) Extract Reduces Fat Accumulation and Improves Health Span in C. elegans through the DAF-16/FOXO and SKN-1/NRF2 Signalling Pathways. Nutrients 2021, 13, 3968. [Google Scholar] [CrossRef]
- Cao, Y.X.; Deng, B.H.; Zhang, S.H.; Gao, H.M.; Song, P.K.; Zhang, J.X.; Zhao, J.X. Astragalus polysaccharide regulates brown adipogenic differentiation through miR-1258-5p-modulated cut-like homeobox 1 expression. Acta Biochim. Biophys. Sin. 2021, 53, 1713–1722. [Google Scholar] [CrossRef]
- Bai, J.; Li, J.; Pan, R.R.; Zhu, Y.; Xiao, X.; Li, Y.; Li, C.T. Polysaccharides from Volvariella volvacea inhibit fat accumulation in C. elegans dependent on the aak-2/nhr-49-mediated pathway. J. Food Biochem. 2021, 45, e13912. [Google Scholar] [CrossRef]
- Ferro, D.; Baratta, F.; Pastori, D.; Cocomello, N.; Colantoni, A.; Angelico, F.; Del Ben, M. New Insights into the Pathogenesis of Non-Alcoholic Fatty Liver Disease: Gut-Derived Lipopolysaccharides and Oxidative Stress. Nutrients 2020, 12, 2762. [Google Scholar] [CrossRef]
- Abu-Shanab, A.; Quigley, E.M.M. The role of the gut microbiota in nonalcoholic fatty liver disease. Nat. Rev. Gastroenterol. Hepatol. 2010, 7, 691–701. [Google Scholar] [CrossRef]
- Ghetti, F.D.; Oliveira, D.G.; de Oliveira, J.M.; de Castro Ferreira, L.E.V.V.; Cesar, D.E.; Moreira, A.P.B. Influence of gut microbiota on the development and progression of nonalcoholic steatohepatitis. Eur. J. Nutr. 2018, 57, 861–876. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.L.; He, J.; Zheng, P.; Mao, X.B.; Huang, Z.Q.; Yan, H.; Luo, Y.H.; Yu, J.; Luo, J.Q.; Yu, B.; et al. Prebiotic inulin as a treatment of obesity related nonalcoholic fatty liver disease through gut microbiota: A critical review. Crit. Rev. Food Sci. 2021, 22, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Hess, J.; Wang, Q.; Gould, T.; Slavin, J. Impact of Agaricus bisporus Mushroom Consumption on Gut Health Markers in Healthy Adults. Nutrients 2018, 10, 1402. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.R.; Lin, C.S.; Chang, C.J.; Lin, T.L.; Martel, J.; Ko, Y.F.; Ojcius, D.M.; Lu, C.C.; Young, J.D.; Lai, H.C.; et al. Gut commensal Parabacteroides goldsteinii plays a predominant role in the anti-obesity effects of polysaccharides isolated from Hirsutella sinensis. Gut 2019, 68, 248–262. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.Y.; Kang, S.; Hua, C.S.; Ting, Z.; Park, S. Synbiotic effects of beta-glucans from cauliflower mushroom and Lactobacillus fermentum on metabolic changes and gut microbiome in estrogen-deficient rats. Genes Nutr. 2017, 12, 31. [Google Scholar] [CrossRef]
- Fontes, A.; Alemany-Pages, M.; Oliveira, P.J.; Ramalho-Santos, J.; Zischka, H.; Azul, A.M. Antioxidant Versus Pro-Apoptotic Effects of Mushroom-Enriched Diets on Mitochondria in Liver Disease. Int. J. Mol. Sci. 2019, 20, 3987. [Google Scholar] [CrossRef]
- Hu, B.F.; Yang, H.B.; Chen, G.M.; Sun, X.J.; Zou, X.J.; Ma, J.; Yao, X.W.; Liang, Q.; Liu, H.T. Structural characterization and preventive effect on non-alcoholic fatty liver disease of oligosaccharides from Bletilla striata. Food Funct. 2022, 13, 4757–4769. [Google Scholar] [CrossRef]
- Cao, Y.N.; Yue, S.S.; Wang, A.Y.; Xu, L.; Hu, Y.T.; Qiao, X.; Wu, T.Y.; Ye, M.; Wu, Y.C.; Qi, R.; et al. Antrodia cinnamomea and its compound dehydroeburicoic acid attenuate nonalcoholic fatty liver disease by upregulating ALDH2 activity. J. Ethnopharmacol. 2022, 292, 115146. [Google Scholar] [CrossRef]
- Wu, X.F.; Wu, X.X.; Guo, W.J.; Luo, Q.; Gu, Y.H.; Shen, Y.; Tan, R.X.; Sun, Y.; Xu, Q.; Cerebroside, D.; et al. A glycoceramide compound, improves experimental colitis in mice with multiple targets against activated T lymphocytes. Toxicol. Appl. Pharm. 2012, 263, 296–302. [Google Scholar] [CrossRef]
- Rashvand, S.; Behrooz, M.; Samsamikor, M.; Jacobson, K.; Hekmatdoost, A. Dietary patterns and risk of ulcerative colitis: A case—Control study. J. Hum. Nutr. Diet. 2018, 31, 408–412. [Google Scholar] [CrossRef]
- Pereira, C.; Gracio, D.; Teixeira, J.P.; Magro, F. Oxidative Stress and DNA Damage: Implications in Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2015, 21, 2403–2417. [Google Scholar] [CrossRef]
- Wasilewski, A.; Zielinska, M.; Storr, M.; Fichna, J. Beneficial Effects of Probiotics, Prebiotics, Synbiotics, and Psychobiotics in Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2015, 21, 1674–1682. [Google Scholar] [CrossRef]
- De Waal, G.M.; de Villiers, W.J.S.; Forgan, T.; Roberts, T.; Pretorius, E. Colorectal cancer is associated with increased circulating lipopolysaccharide, inflammation and hypercoagulability. Sci. Rep. 2020, 10, 8777. [Google Scholar] [CrossRef]
- Zhang, Y.Z.; Chen, D.; Wang, F.; Li, X.W.; Xue, X.M.; Jiang, M.Z.; Xu, B.; Chu, Y.; Wang, W.J.; Wu, K.C.; et al. Comparison of the efficiency of different enemas on patients with distal ulcerative colitis. Cell Prolif. 2019, 52, e12559. [Google Scholar] [CrossRef]
- Park, J.S.; Choi, J.; Jhun, J.; Kwon, J.Y.; Lee, B.I.; Yang, C.W.; Park, S.H.; Cho, M.L. Lactobacillus acidophilus Improves Intestinal Inflammation in an Acute Colitis Mouse Model by Regulation of Th17 and Treg Cell Balance and Fibrosis Development. J. Med. Food 2018, 21, 215–224. [Google Scholar] [CrossRef]
- Chen, Y.; Jin, Y.; Stanton, C.; Ross, R.P.; Zhao, J.X.; Zhan, H.; Yang, B.; Chen, W. Alleviation effects of Bifidobacterium breve on DSS-induced colitis depends on intestinal tract barrier maintenance and gut microbiota modulation. Eur. J. Nutr. 2021, 60, 369–387. [Google Scholar] [CrossRef]
- Hoffmann, T.W.; Pham, H.P.; Bridonneau, C.; Aubry, C.; Lamas, B.; Martin-Gallausiaux, C.; Moroldo, M.; Rainteau, D.; Lapaque, N.; Six, A.; et al. Microorganisms linked to inflammatory bowel disease-associated dysbiosis differentially impact host physiology in gnotobiotic mice. ISME J. 2016, 10, 460–477. [Google Scholar] [CrossRef]
- Hanaoka, R.; Ueno, Y.; Tanaka, S.; Nagai, K.; Onitake, T.; Yoshioka, K.; Chayama, K. The Water-Soluble Extract from Cultured Medium of Ganoderma lucidum (Reishi) Mycelia (Designated as MAK) Ameliorates Murine Colitis Induced by Trinitrobenzene Sulphonic Acid. Scand. J. Immunol. 2011, 74, 454–462. [Google Scholar] [CrossRef]
- Li, M.Y.; Yu, L.L.; Zhai, Q.X.; Liu, B.S.; Zhao, J.X.; Zhang, H.; Chen, W.; Tian, F.W. Ganoderma applanatum polysaccharides and ethanol extracts promote the recovery of colitis through intestinal barrier protection and gut microbiota modulations. Food Funct. 2022, 13, 688–701. [Google Scholar] [CrossRef]
- Li, K.K.; Zhuo, C.; Teng, C.Y.; Yu, S.M.; Wang, X.; Hu, Y.; Ren, G.M.; Yu, M.; Qu, J.J. Effects of Ganoderma lucidum polysaccharides on chronic pancreatitis and intestinal microbiota in mice. Int. J. Biol. Macromol. 2016, 93, 904–912. [Google Scholar] [CrossRef]
- Guo, C.L.; Guo, D.D.; Fang, L.; Sang, T.T.; Wu, J.J.; Guo, C.J.; Wang, Y.J.; Wang, Y.; Chen, C.J.; Chen, J.J.; et al. Ganoderma lucidum polysaccharide modulates gut microbiota and immune cell function to inhibit inflammation and tumorigenesis in colon. Carbohydr. Polym. 2021, 267, 118231. [Google Scholar] [CrossRef]
- Ren, G.; Xu, L.; Lu, T.; Zhang, Y.; Wang, Y.; Yin, J. Protective effects of lentinan on lipopolysaccharide induced inflammatory response in intestine of juvenile taimen (Hucho taimen, Pallas). Int. J. Biol. Macromol. 2019, 121, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.L.; Yang, X.; Zheng, C.Q.; Yang, J.; Tang, X.C.; Chen, J.; Shuai, O.; Xie, Y.Z. Extracts from Hericium erinaceus relieve inflammatory bowel disease by regulating immunity and gut microbiota. Oncotarget 2017, 8, 85838–85857. [Google Scholar]
- Tsopmejio, I.S.N.; Ding, M.; Wei, J.L.; Zhao, C.; Jiang, Y.; Li, Y.T.; Song, H. Auricularia polytricha and Flammulina velutipes ameliorate inflammation and modulate the gut microbiota via regulation of NF-kappa B and Keap1/Nrf2 signaling pathways on DSS-induced inflammatory bowel disease. Food Biosci. 2022, 47, 101426. [Google Scholar] [CrossRef]
- Lu, C.L.; Li, H.X.; Zhu, X.Y.; Luo, Z.S.; Rao, S.Q.; Yang, Z.Q. Regulatory effect of intracellular polysaccharides from Antrodia cinnamomea on the intestinal microbiota of mice with antibiotic-associated diarrhea. Qual. Assur. Saf. Crops Foods 2022, 14, 124–134. [Google Scholar] [CrossRef]
- Hu, Q.; Yuan, B.; Wu, X.; Du, H.; Gu, M.; Han, Y.; Yang, W.; Song, M.; Xiao, H. Dietary Intake of Pleurotus eryngii Ameliorated Dextran-Sodium-Sulfate-Induced Colitis in Mice. Mol. Nutr. Food Res. 2019, 63, 1801265. [Google Scholar] [CrossRef]
- Hasnat, M.A.; Pervin, M.; Cha, K.M.; Kim, S.K.; Lim, B.O. Anti-inflammatory activity on mice of extract of Ganoderma lucidum grown on rice via modulation of MAPK and NF-kappaB pathways. Phytochemistry 2015, 114, 125–136. [Google Scholar] [CrossRef]
- Lin, M.; Dong, L.; Chen, Q.; Xu, H.; Han, X.; Luo, R.; Pu, X.; Qi, S.; Nie, W.; Ma, M.; et al. Lentinan-Based Oral Nanoparticle Loaded Budesonide with Macrophage-Targeting Ability for Treatment of Ulcerative Colitis. Front. Bioeng. Biotechnol. 2021, 9, 702173. [Google Scholar] [CrossRef]
- Yin, C.M.; Noratto, G.D.; Fan, X.Z.; Chen, Z.Y.; Yao, F.; Shi, D.F.; Gao, H. The Impact of Mushroom Polysaccharides on Gut Microbiota and Its Beneficial Effects to Host: A Review. Carbohydr. Polym. 2020, 250, 116942. [Google Scholar] [CrossRef]
- Smith, P.M.; Howitt, M.R.; Panikov, N.; Michaud, M.; Gallini, C.A.; Bohlooly, Y.M.; Glickman, J.N.; Garrett, W.S. The microbial metabolites, short-chain fatty acids, regulate colonic Treg cell homeostasis. Science 2013, 341, 569–573. [Google Scholar] [CrossRef]
- Shao, S.; Wang, D.; Zheng, W.; Li, X.; Zhang, H.; Zhao, D.; Wang, M. A unique polysaccharide from Hericium erinaceus mycelium ameliorates acetic acid-induced ulcerative colitis rats by modulating the composition of the gut microbiota, short chain fatty acids levels and GPR41/43 respectors. Int. Immunopharmacol. 2019, 71, 411–422. [Google Scholar] [CrossRef]
- Grigsby, B.; Rodriguez-Rilo, H.; Khan, K. Antioxidants and Chronic Pancreatitis: Theory of Oxidative Stress and Trials of Antioxidant Therapy. Digest. Dis. Sci. 2012, 57, 835–841. [Google Scholar] [CrossRef]
- Ren, G.M.; Yu, M.; Li, K.K.; Hu, Y.; Wang, Y.; Xu, X.H.; Qu, J.J. Seleno-lentinan prevents chronic pancreatitis development and modulates gut microbiota in mice. J. Funct. Foods 2016, 22, 177–188. [Google Scholar] [CrossRef]
- Jin, M.L.; Zhu, Y.M.; Shao, D.Y.; Zhao, K.; Xu, C.L.; Li, Q.; Yang, H.; Huang, Q.S.; Shi, J.L. Effects of polysaccharide from mycelia of Ganoderma lucidum on intestinal barrier functions of rats. Int. J. Biol. Macromol. 2017, 94, 1–9. [Google Scholar] [CrossRef]
- Liu, J.S.; Liu, L.; Zhang, G.W.; Peng, X.C. Poria cocos polysaccharides attenuate chronic nonbacterial prostatitis by targeting the gut microbiota: Comparative study of Poria cocos polysaccharides and finasteride in treating chronic prostatitis. Int. J. Biol. Macromol. 2021, 189, 346–355. [Google Scholar] [CrossRef]
- Zhang, S.Y.; Lu, S.M.; Li, Z.F. Extrahepatic factors in hepatic immune regulation. Front. Immunol. 2022, 13, 941721. [Google Scholar] [CrossRef]
- Huang, Q.X.; Gao, S.; Yao, Y.; Wang, Y.S.; Li, J.; Chen, J.J.; Guo, C.; Zhao, D.Q.; Li, X.Y. Innate immunity and immunotherapy for hemorrhagic shock. Front. Immunol. 2022, 13, 918380. [Google Scholar] [CrossRef]
- Wyatt, J.; Muller, M.M.; Tavassoli, M. Cancer Treatment Goes Viral: Using Viral Proteins to Induce Tumour-Specific Cell Death. Cancers 2019, 11, 1975. [Google Scholar] [CrossRef]
- Isik, A.; Soyturk, M.; Suleyman, S.; Firat, D.; Peker, K.; Yilmaz, I.; Celebi, F. Correlation of Bowel Wall Thickening Seen Using Computerized Tomography with Colonoscopies: A Preliminary Study. Surg. Laparosc. Endosc. Percutaneous Tech. 2017, 27, 154–157. [Google Scholar] [CrossRef]
- Castellarin, M.; Warren, R.L.; Freeman, J.D.; Dreolini, L.; Krzywinski, M.; Strauss, J.; Barnes, R.; Watson, P.; Allen-Vercoe, E.; Moore, R.A.; et al. Fusobacterium nucleatum infection is prevalent in human colorectal carcinoma. Genome Res. 2012, 22, 299–306. [Google Scholar] [CrossRef]
- Wang, F.; Meng, W.B.; Wang, B.Y.; Qiao, L. Helicobacter pylori-induced gastric inflammation and gastric cancer. Cancer Lett. 2014, 345, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Mira-Pascual, L.; Cabrera-Rubio, R.; Ocon, S.; Costales, P.; Parra, A.; Suarez, A.; Moris, F.; Rodrigo, L.; Mira, A.; Collado, M.C.; et al. Microbial mucosal colonic shifts associated with the development of colorectal cancer reveal the presence of different bacterial and archaeal biomarkers. J. Gastroenterol. 2015, 50, 167–179. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.C.; Lin, W.C.; Kong, M.S.; Shi, H.N.; Walker, W.A.; Lin, C.Y.; Huang, C.T.; Lin, Y.C.; Jung, S.M.; Lin, T.Y.; et al. Oral inoculation of probiotics Lactobacillus acidophilus NCFM suppresses tumour growth both in segmental orthotopic colon cancer and extra-intestinal tissue. Br. J. Nutr. 2012, 107, 1623–1634. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, L.; Delgado, S.; Ruas-Madiedo, P.; Sanchez, B.; Margolles, A. Bifidobacteria and Their Molecular Communication with the Immune System. Front. Microbiol. 2017, 8, 2345. [Google Scholar] [CrossRef] [PubMed]
- Na, K.; Li, K.; Sang, T.; Wu, K.; Wang, Y.; Wang, X. Anticarcinogenic effects of water extract of sporoderm-broken spores of Ganoderma lucidum on colorectal cancer in vitro and in vivo. Int. J. Oncol. 2017, 50, 1541–1554. [Google Scholar] [CrossRef]
- Su, J.Y.; Su, L.; Li, D.; Shuai, O.; Zhang, Y.F.; Liang, H.J.; Jiao, C.W.; Xu, Z.C.; Lai, Y.; Xie, Y.Z.; et al. Antitumor Activity of Extract from the Sporoderm-Breaking Spore of Ganoderma lucidum: Restoration on Exhausted Cytotoxic T Cell with Gut Microbiota Remodeling. Front. Immunol. 2018, 9, 1765. [Google Scholar] [CrossRef]
- Su, J.Y.; Li, D.; Chen, Q.J.; Li, M.X.; Su, L.; Luo, T.; Liang, D.L.; Lai, G.X.; Shuai, O.; Jiao, C.W.; et al. Anti-breast Cancer Enhancement of a Polysaccharide from Spore of Ganoderma lucidum With Paclitaxel: Suppression on Tumor Metabolism with Gut Microbiota Reshaping. Front. Microbiol. 2019, 10, 3099. [Google Scholar] [CrossRef]
- Khan, I.; Huang, G.X.; Li, X.A.; Liao, W.L.; Leong, W.K.; Xia, W.R.; Bian, X.Q.; Wu, J.L.; Hsiao, W.L.W. Mushroom polysaccharides and jiaogulan saponins exert cancer preventive effects by shaping the gut microbiota and microenvironment in Apc(Min/+) mice. Pharmacol. Res. 2019, 148, 104448. [Google Scholar] [CrossRef]
- Wang, C.H.; Yang, S.X.; Gao, L.; Wang, L.L.; Cao, L. Carboxymethyl pachyman (CMP) reduces intestinal mucositis and regulates the intestinal microflora in 5-fluorouracil-treated CT26 tumour-bearing mice. Food Funct. 2018, 9, 2695–2704. [Google Scholar] [CrossRef]
- Eslami, M.; Yousefi, B.; Kokhaei, P.; Hemati, M.; Nejad, Z.R.; Arabkari, V.; Namdar, A. Importance of probiotics in the prevention and treatment of colorectal cancer. J. Cell Physiol. 2019, 234, 17127–17143. [Google Scholar] [CrossRef]
- Tannock, G.W.; Liu, Y.F. Guided dietary fibre intake as a means of directing short-chain fatty acid production by the gut microbiota. J. R. Soc. N. Z. 2020, 50, 434–455. [Google Scholar] [CrossRef]
- Raman, M.; Ambalam, P.; Kondepudi, K.K.; Pithva, S.; Kothari, C.; Patel, A.T.; Purama, R.K.; Dave, J.M.; Vyas, B.R.M. Potential of probiotics, prebiotics and synbiotics for management of colorectal cancer. Gut Microbes 2013, 4, 181–192. [Google Scholar] [CrossRef]
- Kasubuchi, M.; Hasegawa, S.; Hiramatsu, T.; Ichimura, A.; Kimura, I. Dietary Gut Microbial Metabolites, Short-chain Fatty Acids, and Host Metabolic Regulation. Nutrients 2015, 7, 2839–2849. [Google Scholar] [CrossRef]
- Li, L.F.; Liu, H.B.; Zhang, Q.W.; Li, Z.P.; Wong, T.L.; Fung, H.Y.; Zhang, J.X.; Bai, S.P.; Lu, A.P.; Han, Q.B. Comprehensive comparison of polysaccharides from Ganoderma lucidum and G-sinense: Chemical, antitumor, immunomodulating and gut-microbiota modulatory properties. Sci. Rep. 2018, 8, 6172. [Google Scholar] [CrossRef]
- Morais, L.H.; Schreiber, H.L.; Mazmanian, S.K. The gut microbiota-brain axis in behaviour and brain disorders. Nat. Rev. Microbiol. 2021, 19, 241–255. [Google Scholar] [CrossRef]
- Lin, C.X.; Zhao, S.; Zhu, Y.L.; Fan, Z.Q.; Wang, J.; Zhang, B.R.; Chen, Y.X. Microbiota-gut-brain axis and toll-like receptors in Alzheimer’s disease. Comput. Struct. Biotechnol. J. 2019, 17, 1309–1317. [Google Scholar] [CrossRef]
- Vogt, N.M.; Kerby, R.L.; Dill-McFarland, K.A.; Harding, S.J.; Merluzzi, A.P.; Johnson, S.C.; Carlsson, C.M.; Asthana, S.; Zetterberg, H.; Blennow, K.; et al. Gut microbiome alterations in Alzheimer’s disease. Sci. Rep. 2017, 7, 13537. [Google Scholar] [CrossRef]
- Zhang, W.; Guo, Y.; Cheng, Y.; Yao, W.; Qian, H. Neuroprotective effects of polysaccharide from Sparassis crispa on Alzheimer’s disease-like mice: Involvement of microbiota-gut-brain axis. Int. J. Biol. Macromol. 2022, 225, 974–986. [Google Scholar] [CrossRef]
- Gao, Y.; Li, B.; Liu, H.; Tian, Y.; Gu, C.; Du, X.; Bu, R.; Gao, J.; Liu, Y.; Li, G.; et al. Cistanche deserticola polysaccharides alleviate cognitive decline in aging model mice by restoring the gut microbiota-brain axis. Aging 2021, 13, 15320–15335. [Google Scholar] [CrossRef]
- Newgard, C.B.; Sharpless, N.E. Coming of age: Molecular drivers of aging and therapeutic opportunities. J. Clin. Investig. 2013, 123, 946–950. [Google Scholar] [CrossRef]
- Muturi, E.J.; Ramirez, J.L.; Rooney, A.P.; Kim, C.H. Comparative analysis of gut microbiota of mosquito communities in central Illinois. PLoS Negl. Trop. Dis. 2017, 11, e0005377. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.P.; Chen, X.; Zhang, Y.F.; Liu, X.; Wang, C.Y.; Teng, L.S.; Wang, D. Protective roles of Amanita caesarea polysaccharides against Alzheimer’s disease via Nrf2 pathway. Int. J. Biol. Macromol. 2019, 121, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.R.; Xing, S.L.; Chen, C.; Shen, D.Z.; Chen, J.L. Codonopsis pilosula Polysaccharides Alleviate Abeta (1-40)-Induced PC12 Cells Energy Dysmetabolism via CD38/NAD+ Signaling Pathway. Curr. Alzheimer Res. 2021, 18, 208–221. [Google Scholar] [PubMed]
- An, S.; Lu, W.; Zhang, Y.; Yuan, Q.; Wang, D. Pharmacological Basis for Use of Armillaria mellea Polysaccharides in Alzheimer’s Disease: Antiapoptosis and Antioxidation. Oxidative Med. Cell Longev. 2017, 2017, 4184562. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Wang, P.; Chen, Z.; Sui, X.; Xie, X.; Zhang, J. Alteration of the fecal microbiota in North-Eastern Han Chinese population with sporadic Parkinson’s disease. Neurosci. Lett. 2019, 707, 134297. [Google Scholar] [CrossRef]
- Qian, Y.; Yang, X.; Xu, S.; Wu, C.; Song, Y.; Qin, N.; Chen, S.D.; Xiao, Q. Alteration of the fecal microbiota in Chinese patients with Parkinson’s disease. Brain Behav. Immun. 2018, 70, 194–202. [Google Scholar] [CrossRef]
- Aho, V.T.E.; Pereira, P.A.B.; Voutilainen, S.; Paulin, L.; Pekkonen, E.; Auvinen, P.; Scheperjans, F. Gut microbiota in Parkinson’s disease: Temporal stability and relations to disease progression. EBioMedicine 2019, 44, 691–707. [Google Scholar] [CrossRef]
- Bedarf, J.R.; Hildebrand, F.; Coelho, L.P.; Sunagawa, S.; Bahram, M.; Goeser, F.; Bork, P.; Wullner, U. Functional implications of microbial and viral gut metagenome changes in early stage L-DOPA-naive Parkinson’s disease patients. Genome Med. 2017, 9, 39. [Google Scholar] [CrossRef]
- Claudino dos Santos, J.C.; Lima, M.P.P.; Brito, G.A.C.; Viana, G.S.B. Role of enteric glia and microbiota-gut-brain axis in parkinson disease pathogenesis. Ageing Res. Rev. 2022, 84, 101812. [Google Scholar] [CrossRef]
- Castelli, V.; d’Angelo, M.; Lombardi, F.; Alfonsetti, M.; Antonosante, A.; Catanesi, M.; Benedetti, E.; Palumbo, P.; Cifone, M.G.; Giordano, A.; et al. Effects of the probiotic formulation SLAB51 in in vitro and in vivo Parkinson’s disease models. Aging 2020, 12, 4641–4659. [Google Scholar] [CrossRef]
- Shen, T.; Yue, Y.M.; He, T.T.; Huang, C.; Qu, B.Y.; Lv, W.; Lai, H.Y. The Association Between the Gut Microbiota and Parkinson’s Disease, a Meta-Analysis. Front. Aging Neurosci. 2021, 13, 636545. [Google Scholar] [CrossRef] [PubMed]
- Ren, Z.L.; Wang, C.D.; Wang, T.; Ding, H.; Zhou, M.; Yang, N.; Liu, Y.Y.; Chan, P. Ganoderma lucidum extract ameliorates MPTP-induced parkinsonism and protects dopaminergic neurons from oxidative stress via regulating mitochondrial function, autophagy, and apoptosis. Acta Pharmacol. Sin. 2019, 40, 441–450. [Google Scholar] [CrossRef] [PubMed]
- Badshah, S.L.; Riaz, A.; Muhammad, A.; Cayan, G.T.; Cayan, F.; Duru, M.E.; Ahmad, N.; Emwas, A.H.; Jaremko, M. Isolation, Characterization, and Medicinal Potential of Polysaccharides of Morchella esculenta. Molecules 2021, 26, 1459. [Google Scholar] [CrossRef] [PubMed]
- Hebuterne, X. Gut changes attributed to ageing: Effects on intestinal microflora. Curr. Opin. Clin. Nutr. Metab. Care 2003, 6, 49–54. [Google Scholar] [CrossRef]
- Biagi, E.; Nylund, L.; Candela, M.; Ostan, R.; Bucci, L.; Pini, E.; Nikkila, J.; Monti, D.; Satokari, R.; Franceschi, C.; et al. Through Ageing, and Beyond: Gut Microbiota and Inflammatory Status in Seniors and Centenarians. PLoS ONE 2010, 5, e10667. [Google Scholar] [CrossRef]
- Biagi, E.; Franceschi, C.; Rampelli, S.; Severgnini, M.; Ostan, R.; Turroni, S.; Consolandi, C.; Quercia, S.; Scurti, M.; Monti, D.; et al. Gut Microbiota and Extreme Longevity. Curr. Biol. 2016, 26, 1480–1485. [Google Scholar] [CrossRef]
- Roberts, C.K.; Barnard, R.J. Effects of exercise and diet on chronic disease. J. Appl. Physiol. 2005, 98, 3–30. [Google Scholar] [CrossRef]
- Zhong, L.; Ma, N.; Zheng, H.H.; Ma, G.X.; Zhao, L.Y.; Hu, Q.H. Tuber indicum polysaccharide relieves fatigue by regulating gut microbiota in mice. J. Funct. Foods 2019, 63, 103580. [Google Scholar] [CrossRef]

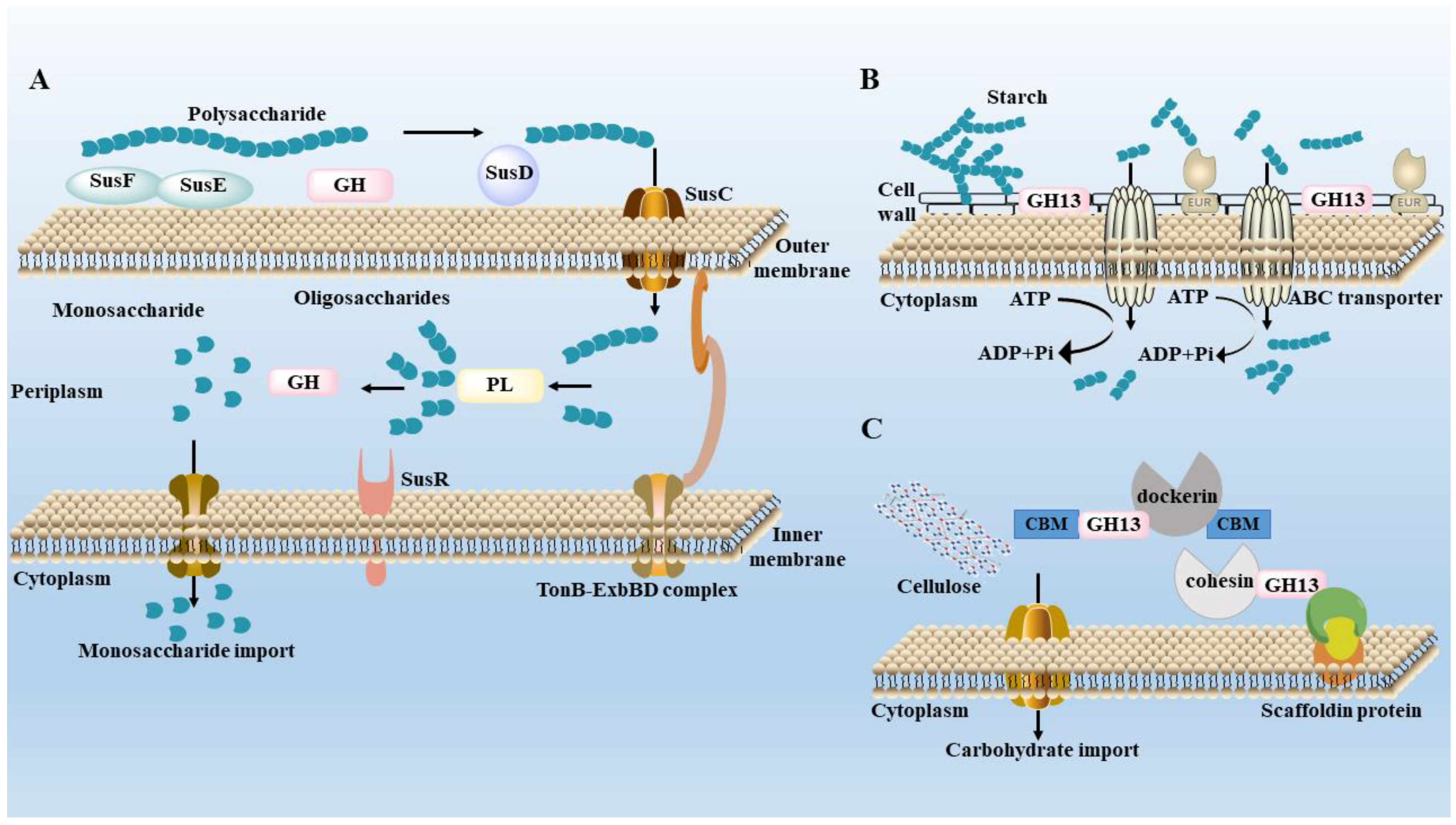
| Source | Fractions and Structural Features | Gut Microbiota Modulation | Ref. |
|---|---|---|---|
| G. frondosa | GFP: 1.82 × 104 Da; Man/Rha/GlcA/GalA/Glc/Gal/Fuc=25.49/5.18/9.49/7.30/27.59/15.02/9.92 | Alistipes↑ Streptococcus, Enterococcus, Staphylococcus and Aerococcus↓ | [20] |
| Inonotus obliquus | IOP: 3.25 × 104 Da; Man/Rha/Glc/Gal/Xyl/Ara=9.80/13.60/29.10/20.50/21.60/5.40 | Bacteroidetes, Prevotella and Lactobacillus↑ Alistipes, Incertae_Sedis, Helicobacter, Parabacteroides and Rikenella↓ | [25] |
| L. edodes | L2: 26 kDa; Glc/Gal/Ara=87.50/9.60/2.80 | Proteobacteria, Bacteroides acidifaciens, Alistipes and Helicobacter suncus↑ Bacilli, Betaproteobacteria, Firmicutes/Bacteroidetes, Lactobacillaceae and Alcaligenaceae↓ | [27,36,37] |
| Ramaria flava | DRFP: 1.02 × 105 Da; Glc/Gal/Man/Fuc/Xyl/Rha/Ara/GlcA=40.61/26.97/17.72/7.78/6.31/0.11/0.06/0.44 | Lactobacillus rhamnosus↑ | [38] |
| H. erinaceus | HEP-30: 8.23 × 105 Da; Fuc/Man/Glc/Gal=0.30/1.30/9.80/0.30; HEP-50: 1.67 × 104 Da; Fuc/Man/Glc/Gal=1.70/0.50/10.60/10.40; HEP-70: 4.77 × 103 Da; Fuc/Man/Glc/Gal=1.20/1.30/23.70/0.30 | Bifidobacterium, Faecalibacterium, Blautia, Butyricicoccus and Lactobacillus↑ Escherichia-Shigella, Klebsiella and Enterobacter↓ | [39] |
| Helvella leucopus | p-HLP: 3.91 × 109 Da; Man/Glc/Rha/Gal=43.68/38.16/9.34/4.35 | Verrumicrobiota, Lactobacillus and Proteobacteria↑ Lachnospiraceae genera, Lachonospiraceae_NK4A136_group and Lachnospiraceae_unclassified↓ | [40] |
| L. edodes | (1, 3)/(1, 6)-β-glucan | Clostridiales, class Clostridia, family Lachnospiraceae and family Ruminococcaceae↑ | [41] |
| Morchella esculenta | MEP: Man/Glc/Gal/Ara=1.00/14.10/0.61/1.56 | Lactobacillus and Firmicutes↑ Actinobacteria, Corynebacterium, Bacteroides and Facklamia↓ | [42] |
| P. eryngii | PEP: Man/Rib/Glc/Gal/Ara/Fuc=9.43/0.43/78.32/8.47/3.05/0.30 | Firmicutes↑ Proteobacteria and Bacteroidetes↓ | [43] |
| Sparassis crispa | SCP-1: 1.36 × 104 Da; Glu/Gal/Fuc/Man=52.10/31.10/15.04/1.76; MC: (1→6)-α-D-Galp, (1→6)-β-D-Glcp, (1→3)-β-D-Glcp, (1→2,6)-α-D-Galp and (1→3,6)-β-D-Glcp; SC: (1→6)-2-OMe-α-D-Galp, (1→4)-β-D-Glcp, (1→3)-β-D-Glcp | Prevotella 9, Dialister, Megamonas and Megasphaera ↑ Escherichia/Shigella↓ | [44,45] |
| Tremella fuciformis | TPs: 2.89 × 105 Da; Man/Rib/Rha/GlcA/Glc/Gal/Xyl/Ara/Fuc=43.68/0.19/0.21/12.93/1.00/0.79/14.40/0.89/11.25; MC: (1→3)-α-D-Manp; SC: D-Xyl, D-Man, L-Fuc, D-GlcA | Firmicutes/Bacteroidetes, Lactobacillaceae and Lactobacillus↑ Ruminococcaceae and Helicobacter↓ | [46] |
| Lyophyllum decastes | LDP1-1: 5.02 × 105 Da; Man/Glc/Gal/Fuc=1.00/2.38/2.58/0.73; DB: 45.90%; 1,3-Fucp, T-Galp, 1,4-Glup, 1,6-Glup, 1,6-Galp, 1,2,6-Manp LDP1-2: 1.13 × 106 Da; Man/Glc/Gal/Fuc=1.00/2.33/2.51/0.78; DB: 43.51%; 1,3-Fucp, T-Galp, 1,4-Glup, 1,6-Glup, 1,6-Galp, 1,2,6-Manp | Bacteroides intestinalis and Lactobacillus johnsonii↑ | [47] |
| G. lucidum | GLP: 1.33 × 105 Da; Glc/Gal/Man/Fuc/Xyl/GlcA=58.97/17.54/8.63/2.79/2.02/6.77 | Bacteroidetes/Firmicutes, Bacteroides ovatus and Bacteroides uniformis↑ | [48] |
| Pleurotus abieticola | PAPS1: 1.72 × 104 Da; Fuc/Gal/Glc/Man=1.73/49.66/12.00/36.60; MC: →2,6)-α-D-Galp-(1→, →6)-α-D-Galp-(1→ and →3)-β-D-Glcp-(1→; SC: β-D-Manp-(1→ and β-D-Manp-(1→6)-α-D-Galp-(1→ | Prevotella, Alistipes, Coprococcus and Oscillospira↑ | [49] |
| Wild morels | MP: 3.97 × 106 Da; Man/Glc/Gal/Rha=43.15/19.56/ 20.25 /1.00 | Lachnospiraceae, Ruminococcaceae and Erysipelotrichaceae↑ Lactobacillus↓ | [50] |
| G. lucidum | GLP: 1.37 × 104 Da; Man/Glc/Gal/Rha/Ara=3.16/16.17/3.74/1.65/1.00 | Parabacteroides, Adlecreuzia, Rothia and Bacteroides↑ Proteus, Corynebacterium, Proteus, Ruminococcus and Coprococcus↓ | [51] |
| Auricularia auricular- judae (Bull.) | AAP: Rha/Man/Glc=1.46/2.34/0.63 | Ruminococcus↓ | [52] |
| Dictyophora indusiata | DIP: Glc/Man/Gal=59.84/23.55/12.95 | Firmicutes, Clostridia and Bacilli↓ | [53] |
| Flammuliana velutipes | FVP1: 5.48 × 104 Da; Glc/Man/Gal=56.20/29.70/14.10 | Lachnospiraceae, Bacteroidales family S24-7 and Firmicutes/Bacteroidetes↑ | [54] |
| L. edodes | LESDF-3: β-D-Arap-(1→, →3)-α-D-Galp-(1→, →3,6)-α-D-Manp-(1→, →4)-β-D-Xylp-(1→, and →2,4)-α-D-Glcp-(1→ | Parasutterella, Bacteroides, Parabacteroides and Lachnospira↑ | [55] |
| F. velutipes | FVP: 7.47 × 106 Da (48.09%) and 1.51 × 104 Da (51.91%); Man/Glc/Xyl/Ara/Fuc | Allobaculum, Lactobacillus, Alloprevotella, Akkermansia and Bifidobacterium↑ | [56] |
| Dictyophora indusiata | DIP: Glc/Man/Gal=59.84/23.55/12.95 | Lactobacillus↑ Gammaproteobacteria↓ | [57] |
| Auricularia auricular | AAP: 3.65 × 105 Da; GlcA/Glc/Xyl/Ara/Fuc=7.60/44.20/7.00/35.80/4.50 | Bacteroidetes and Porphyromonadaceae↑ Firmicutes, Lachnospiraceae, Rikenellaceae and Ruminococcaceae↓ | [58] |
| G. frondosa | GFP: 1.82 × 104 Da | Helicobater, Intestinimonas, Barnesiella, Defluviitalea, Ruminococcus, Flavonifractor and Paraprevotella↑ Clostridium-XVIII, Butyricicoccus and Turicibacter↓ | [59] |
| G. frondosa | GFP-N: 1.26 × 107 Da; Ara/Man/Glc=3.79/1.00/49.70; →2,6)-α-D-Manp-(1→4, α-L-Araf-C1→, and →3,6)-β-D-Glcp-(1→ | Akkermansia, Lactobacillus and Turicibacter↑ | [60] |
| Dictyophora indusiata | DIP: Glc/Man/Gal=59.84/23.55/12.95 | Lactobacillaceae and Ruminococaceae↑ Enterococcus, Bacteroides and Proteobacteria↓ | [61] |
| H. erinaceus | HECP: 8.67 × 104 Da; Glc/Gal/Ara/Xyl/Rha/Man=76.71/14.26/4.04/2.57/1.32/1.14 | Verrucomicrobia and Actinobacteria↑ Bacteroidetes↓ | [62] |
| Disease | Mushroom | Model | Gut Microbiota Regulation | Effects on Hosts & Functional Mechanisms | Ref. |
|---|---|---|---|---|---|
| Chronic pancreatitis | Inonotus obliquus | DDC-induced ICR mice | Bacteroidetes↑ Firmicutes↓ | Increased GSH-PX and TAOC levels, and decreased TNF-α, TGF-β, lipase and trypsin levels; reduced the gut microbiota diversity and richness. | [25] |
| Colitis | G. lucidum | DSS-induced Wistar rats | Ruminococcus_1, Pasteurella, Fusicatenibacter, Enterorhabdus, Marvinbryantia, Erysipelatoclostridium and Anaerofilum↑ | Regulated 11 genes, including six upregulated (Ccl5, Cd3e, Cd8a, Il21r, Lck, and Trbv) and five downregulated (Ccl3, Gro, Il11, Mhc2, and Ptgs) genes; lowered the DAI. | [29] |
| Colonic injury | Helvella leucopus | DSS-induced C57BL/6 mice | Verrumicrobiota, Lactobacillus and Proteobacteria↑ Lachnospiraceae_NK4A136_group and Lachnospiraceae_unclassified↓ | Downregulated IL-6, IL-1β, TNF-α and COX-2, iNOS; upregulated IL-10. | [40] |
| Colitis | Flammuliana velutipe | DSS-induced SD rats | Ruminal butyrivibrios, Roseburia and Bacteroidales family S24-7↑ | Down-regulated inflammatory signal pathways of TLR4\NF-κB; promoted the SCFAs. | [54] |
| Colitis | Dictyophora indusiata | DSS-induced BALB/c mice | Lactobacillus↑ Gammaproteobacteria, Proteobacteria, Bacteroides and Enterobacteriaceae↓ | Dephosphorylated NF-κB and MAPK; inhibited the level of iNOS, COX-2; reduced the level of TNF-α and IL-6. | [57] |
| Chronic pancreatitis | G. lucidum | DDC-induced ICR mice | Lactobacillales, Lachnospiraceae and Roseburia↑ Prevotella, S24-7, Bacteroides and Helicobacter↓ | Decreased lipase, AMS, IFN-γ and TNF-α level, increased SOD and TOAC; altered the composition and diversity of intestinal microbiota. | [160] |
| Colitis | G. lucidum | AOM/DSS- induced mice | Bacteroidetes/Firmicutes, Lactobacillus and Bifidobacterium↑ Oscillibacter, Desulfovibrio, Alistipes and Lachnoclostridium↓ | Downregulated IL-1β, iNOS, and COX-2 expressions; improved gut barrier function; inhibited TLR4/MyD88/NF-κB signaling. | [161] |
| Colitis | Lentinan | LPS-induced Juvenile taimen | Firmicutes, Cyanobacteria, Actinobacteria, Lactobacillus, Bacteroides and Brevinema↑ Proteobacteria, Myroides, Klebsiella, Raoultella and Fusobacteria↓ | Increased the activities of SOD, GSH-Px and CAT, and inhibited the lipid peroxidation; increased the expression levels of TGF-β, TNF-α, IL1β, IL6 and IL8; modified intestinal microbiota. | [162] |
| Colitis | H. erinaceus | TNBS-induced SD rats | Bacteroides, Bifidobacterium, Desulfovibrio and Lactobacillus↑ Corynebacterium, Staphylococcus, Ruminococcus and Dorea↓ | Improved the levels of IL-2, IL-8, IL-10, TNF-γ, TNF-α, VGEF and M-CSF; improved the expression of NF-κB p65, TNF-α, and IL-10. | [163] |
| Colitis | Auricularia polytricha and F. velutipes | DSS-induced ICR mice | Ruminococcaceae, Lachnospiraceae and Prevotellaceae↑ | Improved the unbalanced Th1/Th2 and Th17/Treg ratio; inhibited the NF-κB and MAPK/ERK1/2 signaling pathways and stimulated the Keap1/Nrf2 signaling pathways. | [164] |
| Antibiotic-associated diarrhea | Antrodia cinnamomea | ICR mice | Lachnospiraceae_NK4A136_group, Osllospiraceae and Lachnospiraceae↑ Enterococcus↓ | Reduced the level of TNF-α and IL-6. | [165] |
| Colitis | P. eryngii | DSS-induced ICR mice | Adlercreutzia, Akkermanisa, Lactobacillu, Anaerostipes and Allobaculum↑ Turicibacter, Dorea, rc4-4, Bacteroides and Prvotella↓ | Reduced the level of IL-1β and IL-17. | [166] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, J.; Hu, Y.; Qian, C.; Hussain, M.; Liu, S.; Zhang, A.; He, R.; Sun, P. The Interaction between Mushroom Polysaccharides and Gut Microbiota and Their Effect on Human Health: A Review. Biology 2023, 12, 122. https://doi.org/10.3390/biology12010122
Zhao J, Hu Y, Qian C, Hussain M, Liu S, Zhang A, He R, Sun P. The Interaction between Mushroom Polysaccharides and Gut Microbiota and Their Effect on Human Health: A Review. Biology. 2023; 12(1):122. https://doi.org/10.3390/biology12010122
Chicago/Turabian StyleZhao, Jiahui, Yixin Hu, Chao Qian, Muhammad Hussain, Shizhu Liu, Anqiang Zhang, Rongjun He, and Peilong Sun. 2023. "The Interaction between Mushroom Polysaccharides and Gut Microbiota and Their Effect on Human Health: A Review" Biology 12, no. 1: 122. https://doi.org/10.3390/biology12010122
APA StyleZhao, J., Hu, Y., Qian, C., Hussain, M., Liu, S., Zhang, A., He, R., & Sun, P. (2023). The Interaction between Mushroom Polysaccharides and Gut Microbiota and Their Effect on Human Health: A Review. Biology, 12(1), 122. https://doi.org/10.3390/biology12010122






