The Role of p16/Ki-67 Immunostaining, hTERC Amplification and Fibronectin in Predicting Cervical Cancer Progression: A Systematic Review
Abstract
Simple Summary
Abstract
1. Introduction
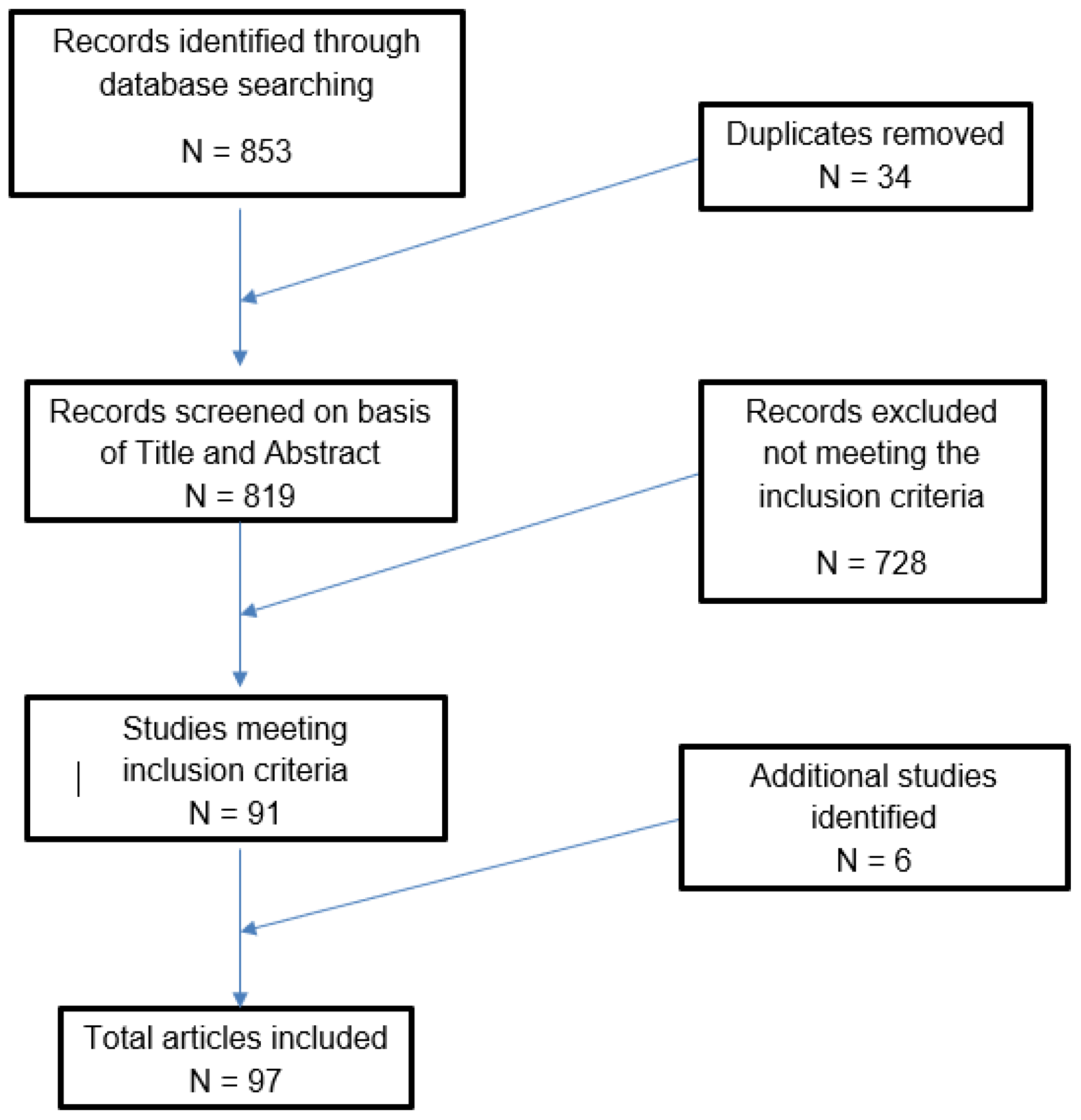
2. Materials and Methods
2.1. Study Selection
2.2. Data Extraction
2.3. Inclusion Criteria
2.4. Exclusion Criteria
2.5. Data Synthesis and Statistical Analysis
2.6. Limitations
3. Results
- p16 staining
| Reference, Year | Number of Biopsy Specimens | HPV Detection and Correlation with Biopsy Results | p16 Positive IHC and Correlation with Biopsy Results |
|---|---|---|---|
| Alhamlan et al., 2021 [44] | 315, of which: | 96/315, of which: | 111/212, of which: |
| 82 NNL | 6/82 NNL | 2/54 NNL | |
| 54 CIN1 | 6/54 CIN1 | 9/37 CIN1 | |
| 16 CIN2 | 3/16 CIN2 | 7/10 CIN2 | |
| 45 CIN3 | 17/45 CIN3 | 20/25 CIN3 | |
| 118 CC | 64/118 CC | 73/84 CC | |
| Castle et al., 2019 [6] | 4010, of which: 283 NNL | 3172/4010 | 2520/4010, of which: |
| 934 CIN1 | 59/283 NNL | 21/283 NNL | |
| 1512 CIN2 | 507/934 CIN1 | 248/934 CIN1 | |
| 1208 CIN3 | 1386/1512 CIN2 | 1087/1512 CIN2 | |
| 73 CC | 1154/1208 CIN3 | 1095/1208 CIN3 | |
| 66/73 CC, of which: | 69/73 CC | ||
| 1283/3172 HPV16: | |||
| 9/283 NNL | |||
| 67/934 CIN1 | |||
| 506/1512 CIN2 | |||
| 658/1208 CIN3 | |||
| 43/73 CC | |||
| 242/3172 HPV 18/45: | |||
| 7/283 NNL | |||
| 49/934 CIN1 | |||
| 111/1512 CIN2 | |||
| 65/1208 CIN3 | |||
| 10/73 CC | |||
| 1357/3172 OHR-HPV: | |||
| 28/283 NNL | |||
| 270/934 CIN1 | |||
| 659/1512 CIN2 | |||
| 390/1208 CIN3 | |||
| 10/73 CC | |||
| 213/3172 IR-HPV: | |||
| 11/283 NNL | |||
| 82/934 CIN1 | |||
| 85/1512 CIN2 | |||
| 34/1208 CIN3 | |||
| 2/73 CC | |||
| 76/3172 LR-HPV: | |||
| 4/283 NNL | |||
| 39/934 CIN1 | |||
| 25/1512 CIN2 | |||
| 7/1208 CIN3 | |||
| 1/73 CC | |||
| Haltas et al., 2012 [48] | 64, of which: | N/A | 37/64, of which: |
| 8 NNL | 0/8 NNL | ||
| 26 CIN1 | 12/26 CIN1 | ||
| 19 CIN2 | 15/19 CIN2 | ||
| 8 CIN3 | 7/8 CIN3 | ||
| 3 CC | 3/3 CC | ||
| Huang et al., 2011 [33] | 272, of which: | 170/272, of which: (HR-HPV) | 153/272, of which: |
| 82 NNL | 19/82 NNL | 14/82 NNL | |
| 94 CIN1 | 63/94 CIN1 | 47/94 CIN1 | |
| 41 CIN2 | 35/41 CIN2 | 37/41 CIN2 | |
| 28 CIN3 | 27/28 CIN3 | 28/28 CIN3 | |
| 27 CC | 26/27 CC | 27/27 CC | |
| Indarti et al., 2013 [34] | 30, of which: | 14/30, of which: | 17/30 |
| 11 CIN1 | 0/11 CIN1 | 0/11 CIN1 | |
| 9 CIN2 | 5/9 CIN2 | 7/9 CIN2 | |
| 10 CIN3 | 9/10 CIN3 | 10/10 CIN3 | |
| Liao et al., 2013 [35] | 463, of which: | 248/463 | 160/463 |
| 187 NNL | 29/187 NNL | 5/187 NNL | |
| 171 CIN1 | 124/171 CIN1 | 73/171 CIN1 | |
| 53 CIN2 | 45/53 CIN2 | 40/53 CIN2 | |
| 49 CIN3 | 47/43 CIN3 | 39/49 CIN3 | |
| 3 CC | 3/3 CC | 3/3 CC | |
| Ma et al., 2011 [36] | 131, of which: | 88/131 HR-HPV, of which: | 49/131 |
| 79 NNL | 43/79 NNL | 10/79 NNL | |
| 26 CIN1 | 21/26 CIN1 | 16/26 CIN1 | |
| 23 CIN2/3 | 21/23 CIN2/3 | 20/23 CIN2/3 | |
| 3 CC | 3/3 CC | 3/3 CC | |
| Pabuccu et al., 2017 [49] | 27, of which: | N/A | 13/27 |
| 14 NNL | 1/14 NNL | ||
| 5 CIN1 | 5/5 CIN1 | ||
| 8 CIN2/3 | 7/8 CIN2/3 | ||
| Pacchiarotti et al., 2014 [50] | 577, of which: | N/A | 193/577, of which: |
| 312 NNL | 6/312 NNL | ||
| 159 CIN1 | 91/159 CIN1 | ||
| 39 CIN2 | 36/39 CIN2 | ||
| 58 CIN3 | 53/58 CIN3 | ||
| 9 CC | 7/9 CC | ||
| Sarma et al., 2017 [51] | 110, of which: | N/A | 60/110, of which: |
| 25 NNL | 2/25 NNL | ||
| 25 CIN1 | 8/25 CIN1 | ||
| 21 CIN2 | 11/21 CIN2 | ||
| 12 CIN3 | 12/12 CIN3 | ||
| 27 CC | 27/27 CC | ||
| Tsoumpou et al., 2011 [52] | 126, of which: | 64/126, of which: | 28/126, of which: |
| 12 NNL | 28/78 NNL/CIN1 | 8/78 NNL/CIN1 | |
| 66 CIN1 | 36/48 CIN2/3 | 20/48 CIN2/3 | |
| 36 CIN2 | |||
| 12 CIN3 | |||
| Valasoulis et al., 2013 [37] | 200, of which: | 133/200 HPV: | 53/200, of which: |
| 23 NNL | 6/23 NNL | 2/23 NNL | |
| 79 CIN1 | 41/79 CIN1 | 12/79 CIN1 | |
| 50 CIN2 | 41/50 CIN2 | 17/50 CIN2 | |
| 48 CIN3 | 45/48 CIN3 | 22/48 CIN3 | |
| 118/200 HR-HPV: | |||
| 5/23 NNL | |||
| 30/79 CIN1 | |||
| 38/50 CIN2 | |||
| 45/48 CIN3 | |||
| 60/200 HPV16/18: | |||
| 0/23 NNL | |||
| 14/79 CIN1 | |||
| 17/50 CIN2 | |||
| 29/48 CIN3 | |||
| van Baars et al., 2015 [39] | 104, of which: | 90/104, of which: | 76/104, of which: |
| 25 NNL | 13/25 NNL | 0/25 NNL | |
| 11 CIN1 | 11/11 CIN1 | 8/11 CIN1 | |
| 23 CIN2 | 23/23 CIN2 | 23/23 CIN2 | |
| 45 CIN3 | 43/45 CIN3 | 45/45 CIN3 |
- p16/Ki-67 DS
| Reference, Year | Number of Biopsy Specimens | HPV Detection and Correlation with Biopsy Results | p16/Ki67 Positive IHC and Correlation with Biopsy Results |
|---|---|---|---|
| Celewicz et al., 2018 [53] | 43, of which: | NA | 30/43, of which: |
| 17 NNL | 9/17 NNL | ||
| 5 CIN1 | 2/5 CIN1 | ||
| 10 CIN2 | 9/10 CIN2 | ||
| 8 CIN3 | 7/8 CIN3 | ||
| 3 CC | 3/3 CC | ||
| Diouf et al., 2020 [54] | 69, of which: | 30/38, of which: | 32/46, of which: |
| 30 NNL | 1/7 NNL | 1/7 NNL | |
| 14 CIN1 | 4/6 CIN1 | 6/14 CIN1 | |
| 3 CIN2 | 6/6 CIN2/3 | 6/6 CIN2/3 | |
| 3 CIN3 | 19/19 CC | 19/19 CC | |
| 19 CC | |||
| Donà et al., 2012 [64] | 113, of which: | 95/107 | 62/107, of which: |
| 14 NNL | 5/13 NNL | 0/13 NNL | |
| 35 CIN1 | 31/33 CIN1 | 13/33 CIN1 | |
| 24 CIN2 | 23/24 CIN2 | 17/24 CIN2 | |
| 37 CIN3 | 36/37 CIN3/CC | 32/37 CIN3/CC | |
| 3 CC | |||
| 84/107 HR-HPV | |||
| 3/13 NNL | |||
| 25/33 CIN1 | |||
| 20/24 CIN2 | |||
| 36/37 CIN3/CC | |||
| 11/107 O-HPV | |||
| 2/13 NNL | |||
| 6/33 CIN1 | |||
| 3/24 CIN2 | |||
| 0/37 CIN3/CC | |||
| El-Zein et al., 2020 [55] | 492, of which: | 321/492, of which: | 279/492, of which: |
| 134 NNL | 47/134 NNL | 41/134 NNL | |
| 130 CIN1 | 69/130 CIN1 | 54/130 CIN1 | |
| 99 CIN2 | 86/99 CIN2 | 72/99 CIN2 | |
| 121 CIN3 | 111/121 CIN3 | 105/121 CIN3 | |
| 8 CC | 8/8 CC | 7/8 CC | |
| 119/492 HPV16: | |||
| 7/134 NNL | |||
| 17/130 CIN | |||
| 37/99 CIN2 | |||
| 55/121 CIN3 | |||
| 3/8 CC | |||
| 26/492 HPV18: | |||
| 6/134 NNL | |||
| 4/130 CIN1 | |||
| 5/99 CIN2 | |||
| 5/121 CIN3 | |||
| 6/8 CC | |||
| 139/492 HPV16/18: | |||
| 12/134 NNL | |||
| 20/130 CIN1 | |||
| 41/99 CIN2 | |||
| 58/121 CIN3 | |||
| 8/8 CC | |||
| 235/492 OHR-HPV: | |||
| 41/134 NNL | |||
| 63/130 CIN1 | |||
| 58/99 CIN2 | |||
| 70/121 CIN3 | |||
| 3/8 CC | |||
| 321/492 ANY HR-HPV: | |||
| 47/134 NNL | |||
| 69/130 CIN1 | |||
| 86/99 CIN2 | |||
| 111/121 CIN3 | |||
| 8/8 CC | |||
| Frega et al., 2019 [56] | 78, of which: | 73/78, of which: | 74/78, of which: |
| 53 CIN2 | 50/53 CIN2 | 50/53 CIN2 | |
| 25 CIN3 | 23/25 CIN3 | 24/25 CIN3 | |
| Liu et al., 2020 [65] | 305, of which: | N/A | 165/305, of which: |
| 90 NNL | 3/90 NNL | ||
| 48 CIN1 | 8/48 CIN1 | ||
| 35 CIN2 | 26/35 CIN2 | ||
| 117 CIN3 | 113/117 CIN3 | ||
| 15 ICC | 15/15 CC | ||
| Ngugi et al., 2015 [57] | 22, of which: | 21/22 HR-HPV, of which: | 8/22, of which: |
| 12 NNL | 11/12 NNL | 1/12 NNL | |
| 2 CIN1 | 2/2 CIN1 | 0/2 CIN1 | |
| 2 CIN2 | 2/2 CIN2 | 1/2 CIN2 | |
| 6 CIN3 | 6/6 CIN3 | 6/6 CIN3 | |
| Waldstrøm et al., 2013 [59] | 226, of which: | 174/226, of which: | 154/226, of which: |
| 42 NNL | 28/42 NNL | 23/42 NNL | |
| 97 CIN1 | 66/97 CIN1 | 54/97 CIN1 | |
| 41 CIN2 | 36/41 CIN2 | 33/41 CIN2 | |
| 45 CIN3 | 43/45 CIN3 | 43/45 CIN3 | |
| 1 CC | 1/1 CC | 1/1 CC | |
| Wentzensen et al., 2012 [32] | 623, of which: | 171/623 HPV16, of which: | 371/623, of which: |
| 137 NNL | 24/137 NNL | 42/137 NNL | |
| 228 CIN1 | 31/228 CIN1 | 106/228 CIN1 | |
| 169 CIN2 | 60/169 CIN2 | 140/169 CIN2 | |
| 83 CIN3 | 53/83 CIN3 | 77/83 CIN3 | |
| 6 CC | 3/6 CC | 6/6 CC | |
| Yu et al., 2016 [60] | 1290, of which: | 463/1290, of which: | 427/1290, of which: |
| 996 NNL | 204/996 NNL | 183/996 NNL | |
| 63 CIN1 | 41/63 CIN1 | 34/63 CIN1 | |
| 42 CIN2 | 40/42 CIN2 | 34/42 CIN2 | |
| 119 CIN3 | 111/119 CIN3 | 111/119 CIN3 | |
| 70 CC | 67/70 CC | 65/70 CC | |
| Yu et al., 2016 [61] | 701, of which: | 173/701, of which: | 149/701, of which: |
| 640 NNL | 126/640 NNL | 111/640 NNL | |
| 46 CIN1 | 32/46 CIN1 | 26/46 CIN1 | |
| 11 CIN2 | 11/11 CIN2 | 8/11 CIN2 | |
| 4 CIN3 | 4/4 CIN3 | 4/4 CIN3 | |
| Zhang et al., 2019 [62] | 537, of which: | 294/537, of which: | 234/537, of which: |
| 298 NNL | 76/298 NNL | 39/298 NNL | |
| 29 CIN | 18/29 CIN | 10/29 CIN | |
| 49 CIN2 | 45/49 CIN2 | 38/49 CIN2 | |
| 111 CIN3 | 106/111 CIN3 | 99/111 CIN3 | |
| 50 CC | 49/50 CC | 48/50 CC | |
| 168/537 HPV16/18 | |||
| 23/298 NNL | |||
| 8/29 CIN | |||
| 16/49 CIN2 | |||
| 80/111 CIN3 | |||
| 41/50 CC | |||
| 168/537 O-HPV | |||
| 59/298 NNL | |||
| 10/29 CIN | |||
| 34/49 CIN2 | |||
| 50/111 CIN3 | |||
| 15/50 CC | |||
| Zhu et al., 2019 [63] | 300, of which: | 256/300, of which: | 96/300, of which: |
| 138 NNL | 103/138 NILM | 3/138 NILM | |
| 108 CIN1 | 100/108 CIN1 | 40/108 CIN1 | |
| 29 CIN2 | 28/29 CIN2 | 28/29 CIN2 | |
| 22 CIN3 | 22/22 CIN3 | 22/22 CIN3 | |
| 3 CC | 3/3 CC | 3/3 CC |
- Ki-67 staining
| Reference Year | Number of Biopsy Specimens | HPV Detection and Correlation with Biopsy Results | p16 Positive IHC and Correlation with Biopsy Results | KI-67 Positive IHC | DS Positive IHC and Correlation with Biopsy Results |
|---|---|---|---|---|---|
| and Correlation with Biopsy Results | |||||
| Chang et al., 2014 [40] | 143, of which: | 70/143, of which: | 31/141, of which: | 29/124 of which: | NA |
| 77 NNL | 23/77 NNL | 5/75 NNL | 2 /69 NNL | ||
| 33 CIN | 21/33 CIN1 | 3/33 CIN1 | 2/27 CIN1 | ||
| 6 CIN2 | 4/6 CIN2 | 4/6 CIN2 | 4/5 CIN2 | ||
| 22 CIN3 | 18/22 CIN3 | 15/21 CIN3 | 17/19 CIN3 | ||
| 5 CC | 4/5 CC | 4/5 CC | 4/4 CC | ||
| Gatta et al., 2011 [67] | 72, of which: | 9/72, of which: | 41/72, of which: | N/A | NA |
| 10 NNL (controls) | 0/10 NNL | 0/10 NNL | |||
| 32 CIN1 | 8/32 CIN1 | 11/32 CIN1 | |||
| 10 CIN2 | 1/10 CIN2 | 10/10 CIN2 | |||
| 10 CIN3 | 0/10 CIN3 | 10/10 CIN3 | |||
| 10 CC | 0/10 CC | 10/10 CC | |||
| Jackson et al., 2012 [43] | 97, of which: | 17/36, of which: | 14/97, of which: | 25//97, of which: | 13/97, of which: |
| 39 NNL | 4/9 NNL | 1/39 NNL1 | 4/39 NNL | 1/39 NNL | |
| 46 CIN1 | 10/24 CIN1 | 5/46 CIN1 | 11/46 CIN1 | 4/46 CIN1 | |
| 12 CIN2/3 | 3/3 CIN2/3 | 8/12 CIN2/3 | 10/12 CIN2/3 | 8/12 CIN2/3 | |
| Koo et al., 2013 [41] | 70, of which: | 36/70 HR-HPV: | 50/70, of which: | 48/70, of which: | 43/70, of which: |
| 27 NNL | 9/27 NNL | 15/27 NNL | 16/27 NNL | 4/27 NNL | |
| 6 CIN1 | 2/6 CIN1 | 2/6 CIN1 | 2/6 CIN1 | 4/6 CIN1 | |
| 20 CIN2 | 14/20 CIN2 | 16/20 CIN2 | 14/20 CIN2 | 18/20 CIN2 | |
| 17 CIN3 | 11/17 CIN3 | 17/17 CIN3 | 16/17 CIN3 | 17/17 CIN3 | |
| of which: | |||||
| 18/36 HPV 16/18: | |||||
| 3/9 NNL | |||||
| 0/2 CIN1 | |||||
| 7/14 CIN2 | |||||
| 8/11 CIN3 | |||||
| Li et al., 2019 [42] | 350, of which: | 271/350, of which: | 197/350, of which: | 276/350, of which: | 185/350 |
| 84 NNL | 49/84 NNL | ||||
| 77 CIN1 | 50/77 CIN1 | 9/84 NNL | 41/84 NNL | 8/84 NNL | |
| 68 CIN2 | 56/68 CIN2 | 22/77 CIN1 | 56/77 CIN1 | 17/77 CIN1 | |
| 89 CIN3 | 87/89 CIN3 | 55/68 CIN2 | 60/68 CIN2 | 50/68 CIN2 | |
| 32 CC | 29/32 CC | 80/89 CIN3 | 87/89 CIN3 | 79/89 CIN3 | |
| 31/32 CC | 32/32 CC | 31/32 CC | |||
| 271/350, of which: | |||||
| 141/350 HPV16 | |||||
| 16/350 HPV 18 | |||||
| 16/350 HPV 31 | |||||
| 21/350 HPV 33 | |||||
| 13/350 HPV 35 | |||||
| 13/350 HPV 39 | |||||
| 3/350 HPV 45 | |||||
| 16/350 HPV 51 | |||||
| 56/350 HPV 52 | |||||
| 10/350 HPV 56 | |||||
| 61/350 HPV 58 | |||||
| 8/350 HPV 59 | |||||
| 11/350 HPV 68 | |||||
| Toll et al., 2014 [66] | 13, of which: | 8/13, of which: | 10/13, of which: | 6/13, of which: | 5/13, of which: |
| 6 NNL | 2/6 NNL | 4/6 NNL | 3/6 NNL | 2/6 NNL | |
| 2 CIN1 | 2/2 CIN1 | 1/2 CIN1 | 0/2 CIN1 | 0/2 CIN1 | |
| 2 CIN1/2 | 1/2 CIN1/2 | 2/2 CIN1/2 | 1/2 CIN1/2 | 1/2 CIN1/2 | |
| 3 CIN3 | 3/3 CIN3 | 3/3 CIN3 | 2/3 CIN3 | 2/3 CIN3 |
- Telomerase
| Reference, Year | Number of Biopsy Specimens | HPV Detection and Correlation with Biopsy Results | hTERC up-Regulation and Correlation with Biopsy Results |
|---|---|---|---|
| Chen et al., 2012 [68] | 243, of which: | 158/243, of which: | 55/243, of which: |
| NNL = 164 | NNL = 84/164 | NNL = 15/164 | |
| CIN1 = 29 | CIN1 = 24/29 | CIN1 = 5/29 | |
| CIN2 = 21 | CIN2 = 21/21 | CIN2 = 6/21 | |
| CIN3 = 22 | CIN3 = 22/22 | CIN3 = 22/22 | |
| CC = 7 | CC = 7/7 | CC = 7/7 | |
| He et al., 2012 [69] | 175, of which: | N/A | 86/175, of which: |
| NNL = 24 | NNL = 0/24 | ||
| CIN1 = 34 | CIN1 = 5/34 | ||
| CIN2 = 36 | CIN2 = 18/36 | ||
| CIN3 = 33 | CIN3 = 23/33 | ||
| CC = 48 | CC = 40/48 | ||
| He et al., 2020 [70] | 135, of which: | 97/135 | 109/135 |
| CIN 1/2 = 65 | CIN1/2 = 32/65 | CIN 1/2 = 41/65 | |
| CIN3 = 39 | CIN3 = 35/39 | CIN3 = 37/39 | |
| CC = 31 | CC = 30/31 | CC = 31/31 | |
| Ji et al., 2019 [71] | 213, of which: | 103/213 | 64/213, of which: |
| NNL = 159 | 75 HR, 28 LR, of which: | NNL = 29/159 | |
| CIN1 = 31 | NNL = 41 HR, 25 LR/159 | CIN1 = 18/31 | |
| CIN2 = 14 | CIN1 = 16 HR, 2 LR/31 | CIN2 = 9/14 | |
| CIN3 = 7 | CIN2 = 10 HR, 1 LR/14 | CIN3 = 6/7 | |
| CC = 2 | CIN3 = 6 HR/7 | CC = 2/2 | |
| CC = 2 HR/2 | |||
| Jiang et al., 2010 [72] | 6726, of which: | 1752/2313, of which: | 3250/6726, of which: |
| NNL = 1257 | NNL = 156/385 | NNL = 124/1257 | |
| CIN1 = 2054 | CIN1 = 560/794 | CIN1 = 428/2054 | |
| CIN2 = 1387 | CIN2 = 406/461 | CIN2 = 952/1387 | |
| CIN3 = 1410 | CIN3 = 490/522 | CIN3 = 1162/1410 | |
| CC = 618 | CC = 140/151 | CC = 584/618 | |
| Jin et al., 2011 [73] | 130, of which: | N/A | 46/130, of which: |
| NNL = 52 | NNL = 2/52 | ||
| CIN1 = 33 | CIN1 = 6/33 | ||
| CIN2 = 9 | CIN2 = 6/9 | ||
| CIN3 = 26 | CIN3 = 22/26 | ||
| CC = 10 | CC = 10/10 | ||
| Koeneman et al., 2019 [83] | 19, of which: | 19/19 | 15/19, of which: |
| CIN2 = 3 | CIN 2 = 3/3 | ||
| CIN3 = 16 | CIN 3 = 12/16 | ||
| Kudela et al., 2018 [74] | 111, of which: | 90/111, of which: | 58/111, of which: |
| NNL = 27 | NNL = 14/27 | NNL = 1/27 | |
| CIN1 = 15 | CIN1 = 7/15 | CIN1 = 4/15 | |
| CIN2 = 24 | CIN2 = 24/24 | CIN2 = 11/24 | |
| CIN3 = 25 | CIN3/CIS = 25/25 | CIN3 = 21/25 | |
| CC = 20 | CC = 20/20 | CC = 20/20 | |
| Kuglik et al., 2015 [75] | 74, of which: | 64/74, of which: | 23/74, of which: |
| NNL = 12 | NNL = 10/12 | NNL = 3/12 | |
| CIN1 = 6 | CIN1 = 3/6 | CIN1 = 1/6 | |
| CIN2 = 6 | CIN2 = 3/6 | CIN2 = 3/6 | |
| CIN3 = 12 | CIN3 = 10/12 | CIN3 = 7/12 | |
| CC = 38 | CC = 34/38 | CC = 33/38 | |
| Li et al., 2014 [77] | 171, of which: | N/A | 67/171, of which: |
| NNL = 64 | NNL = 6/64 | ||
| CIN1 = 26 | CIN1 = 6/26 | ||
| CIN2 = 29 | CIN2 = 15/29 | ||
| CIN3 = 36 | CIN3 = 26/36 | ||
| CC = 16 | CC = 14/16 | ||
| Liu et al., 2012 [23] | 114, of which: | 77/114 | 51/114 |
| NNL = 27 | NNL = 0/26 | ||
| CIN1 = 26 | CIN1 = 4/19 | ||
| CIN2 = 16 | CIN2 = 6/12 | ||
| CIN3 = 24 | CIN 3 = 22/27 | ||
| CC = 21 | CC = 19/19 | ||
| Liu et al., 2019 [24] | 150, of which: | 108/150, of which: | 64/150 |
| NNL = 32 | NNL = 10/32 | NNL = 4/32 | |
| CIN1 = 38 | CIN1 = 25/38 | CIN1 = 13/38 | |
| CIN2/3 = 66 | CIN2/3 = 60/66 | CIN2/3 = 35/66 | |
| CC = 14 | CC = 13/14 | CC = 12/14 | |
| Xiang et al., 2012 [21] | 92, of which: | N/A | 62/92 |
| NNL = 20 | NNL = 0/20 | ||
| CIN3 = 14 | CIN3 = 12/14 | ||
| CC = 58 | CC = 50/58 | ||
| Yin et al., 2012 [78] | 166, of which: | N/A | 101/166 |
| NNL = 40 | NNL = 0/40 | ||
| CIN1 = 27 | CIN1 = 12/27 | ||
| CIN2/3 = 54 | CIN2/3 = 46/54 | ||
| CC = 45 | CC = 43/45 | ||
| Zappacosta et al., 2015 [25] | 54, of which: | 52/54 | 20/54, of which: |
| NNL = 8 | NNL = 0/8 | ||
| CIN1 = 26 | CIN1 = 6/26 | ||
| CIN2 = 9 | CIN2 = 6/9 | ||
| CIN3 = 11 | CIN3 = 8/11 | ||
| Zheng et al., 2013 [79] | 373, of which: | 267/373, of which: | 192/373, of which: |
| NNL = 89 | NNL = 26/89 | NNL = 0/89 | |
| CIN1 = 36 | CIN1 = 19/36 | CIN1 = 5/36 | |
| CIN2 = 43 | CIN2 = 32/43 | CIN2 = 18/43 | |
| CIN3 = 129 | CIN3 = 119/129 | CIN3 = 101/129 | |
| CC = 76 | CC = 71/76 | CC = 68/76 | |
| Zhu et al., 2018 [80] | 138, of which: | 85/138, of which: | 74/138, of which: |
| NNL = 23 | NNL = 4/23 | NNL = 2/23 | |
| CIN1 = 42 | CIN1 = 16/42 | CIN1 = 13/42 | |
| CIN2 = 20 | CIN2 = 14/20 | CIN2 = 11/20 | |
| CIN3 = 28 | CIN3 = 26/28 | CIN3 = 23/28 | |
| CC = 25 | CC = 25/25 | CC = 25/25 |
- Fibronectin
4. Discussion
- P16/Ki-67 staining
- Telomerase
- Fibronectin
5. Challenges and Future Scope
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schiffman, M.; Wentzensen, N.; Wacholder, S.; Kinney, W.; Gage, J.C.; Castle, P.E. Human papillomavirus testing in the prevention of cervical cancer. J. Natl. Cancer Inst. 2011, 103, 368–383. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. International Agency for Research on Cancer IARC Monographs on the Evaluation of Carcinogenic Risks to Humans Volume 90 Human Papillomaviruses; World Health Organization: Geneva, Switzerland, 2007; Volume 90. [Google Scholar]
- Wentzensen, N.; Schiffman, M.; Palmer, T.; Arbyn, M. Triage of HPV positive women in cervical cancer screening. J. Clin. Virol. 2016, 76, S49–S55. [Google Scholar] [CrossRef] [PubMed]
- Gheit, T. Mucosal and Cutaneous Human Papillomavirus Infections and Cancer Biology. Front. Oncol. 2019, 9, 355. [Google Scholar] [CrossRef] [PubMed]
- De Sanjose, S.; Quint, W.G.; Alemany, L.; Geraets, D.T.; Klaustermeier, J.E.; Lloveras, B.; Tous, S.; Felix, A.; Bravo, L.E.; Shin, H.-R.; et al. Human papillomavirus genotype attribution in invasive cervical cancer: A retrospective cross-sectional worldwide study. Lancet Oncol. 2010, 11, 1048–1056. [Google Scholar] [CrossRef]
- Castle, P.E.; Adcock, R.; Cuzick, J.; Wentzensen, N.; Torrez-Martinez, N.E.; Torres, S.M.; Stoler, M.H.; Ronnett, B.M.; Joste, N.E.; Darragh, T.M.; et al. Relationships of p16 Immunohistochemistry and Other Biomarkers with Diagnoses of Cervical Abnormalities: Implications for LAST Terminology. Arch. Pathol. Lab. Med. 2020, 144, 725–734. [Google Scholar] [CrossRef]
- The Global Cancer Observatory. Epidemiology of Cancer in Romania 2020; International Agency for Research on Cancer: Lyon, France, 2020; Volume 938, pp. 2020–2021. [Google Scholar]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Kyrgiou, M.; Arbyn, M.; Bergeron, C.; Bosch, F.X.; Dillner, J.; Jit, M.; Kim, J.; Poljak, M.; Nieminen, P.; Sasieni, P.; et al. Cervical screening: ESGO-EFC position paper of the European Society of Gynaecologic Oncology (ESGO) and the European Federation of Colposcopy (EFC). Br. J. Cancer 2020, 123, 510–517. [Google Scholar] [CrossRef]
- Moyer, V.A. Screening for Cervical Cancer: U.S. Preventive Services Task Force Recommendation Statement. Ann. Intern. Med. 2012, 156, 880–891. [Google Scholar] [CrossRef]
- Melnikow, J.; Henderson, J.T.; Burda, B.U.; Senger, C.A.; Durbin, S.; Soulsby, M.A. Introduction. Agency for Healthcare Research and Quality (US). 2018. Available online: https://www.ncbi.nlm.nih.gov/books/NBK526308/ (accessed on 2 May 2022).
- WHO. ‘Best Buys’ and Other Recommended Interventions for the Prevention and Control of Noncommunicable Diseases; “The Updated Appendix 3 of the WHO Global NCD Action Plan 2013–2020. WHO Glob NCD Action Plan 2013–2020; World Health Organization: Geneva, Switzerland, 2017; pp. 65–70. [Google Scholar]
- Olivas, A.D.; Barroeta, J.E.; Lastra, R.R. Role of Ancillary Techniques in Gynecologic Cytopathology Specimens. Acta Cytol. 2020, 64, 63–70. [Google Scholar] [CrossRef]
- Screening for Cervical Cancer: A Systematic Evidence Review for the U.S. Preventive Services Task Force. Available online: https://pubmed.ncbi.nlm.nih.gov/22132428/ (accessed on 3 May 2022).
- Phaliwong, P.; Pariyawateekul, P.; Khuakoonratt, N.; Sirichai, W.; Bhamarapravatana, K.; Suwannarurk, K. Cervical Cancer Detection between Conventional and Liquid Based Cervical Cytology: A 6-Year Experience in Northern Bangkok Thailand. Asian Pac. J. Cancer Prev. 2018, 19, 1331–1336. [Google Scholar] [CrossRef]
- The 2001 Bethesda System. World Health Organization. 2014. Available online: https://www.ncbi.nlm.nih.gov/books/NBK269610/ (accessed on 2 May 2022).
- Nayar, R.; Wilbur, D.C. (Eds.) The Bethesda System for Reporting Cervical Cytology: Definitions, Criteria, and Explanatory Notes; Springer International Publishing: Cham, Switzerland, 2015. [Google Scholar] [CrossRef]
- Arbyn, M.; Roelens, J.; Simoens, C.; Buntinx, F.; Paraskevaidis, E.; Martin-Hirsch, P.P.; Prendiville, W.J. Human papillomavirus testing versus repeat cytology for triage of minor cytological cervical lesions. Cochrane Database Syst. Rev. 2013, CD008054. [Google Scholar] [CrossRef]
- Jenkins, T.M.; Shojaei, H.; Song, S.J.; Schwartz, L.E. Role of Ancillary Techniques in Cervical Biopsy and Endocervical Curettage Specimens as Follow-Up to Papanicolaou Test Results Indicating a Diagnosis of Atypical Squamous Cells, Cannot Exclude High-Grade Squamous Intraepithelial Lesion, or High-Grade Squamous Intraepithelial Lesion. Acta Cytol. 2020, 64, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Fei, L.; Liu, X.; Pi, X.; Wang, L.; Chen, S. Application of p16/Ki-67 dual-staining cytology in cervical cancers. J Cancer 2019, 10, 2654–2660. [Google Scholar] [CrossRef] [PubMed]
- Xiang, L.; Yang, H.; Li, J.; Wu, X.; Zhou, X. Different amplification patterns of the human telomerase RNA gene in invasive cervical carcinomas and cervical intraepithelial neoplasia grade III. Diagn. Cytopathol. 2012, 40, 849–855. [Google Scholar] [CrossRef] [PubMed]
- Castro-Duque, A.F.; Loango-Chamorro, N.; Ruiz-Hoyos, B.M.; Landázuri, P. Telomerase activity associated with progression of cervical lesions in a group of Colombian patients. Rev. Bras. Ginecol. Obstet. 2015, 37, 559–564. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Liu, H.; Liu, S.; Wang, H.; Xie, X.; Youcheng, Z.; Zhang, X.; Zhang, Y. Genomic amplification of the human telomerase gene (hTERC) associated with human papillomavirus is related to the progression of uterine cervical dysplasia to invasive cancer. Diagn. Pathol. 2012, 7, 147. [Google Scholar] [CrossRef]
- Liu, Y.; Fan, P.; Yang, Y.; Xu, C.; Huang, Y.; Li, D.; Qing, Q.; Sun, C.; Zhou, H. Human papillomavirus and human telomerase RNA component gene in cervical cancer progression. Sci. Rep. 2019, 9, 15926. [Google Scholar] [CrossRef]
- Zappacosta, R.; Ianieri, M.M.; Buca, D.; Repetti, E.; Ricciardulli, A.; Liberati, M. Clinical Role of the Detection of Human Telomerase RNA Component Gene Amplification by Fluorescence in situ Hybridization on Liquid-Based Cervical Samples: Comparison with Human Papillomavirus-DNA Testing and Histopathology. Acta Cytol. 2015, 59, 345–354. [Google Scholar] [CrossRef]
- Bin, H.; Ruifang, W.; Ruizhen, L.; Zhihong, L.; Juan, L.; Chun, W.; Zhou, Z.; Leiming, W. Detention of HPV L1 Capsid Protein and hTERC Gene in Screening of Cervical Cancer. Iran. J. Basic Med. Sci. 2013, 16, 797–802. [Google Scholar]
- Dou, L.; Zhang, X. Upregulation of CCT3 promotes cervical cancer progression through FN1. Mol. Med. Rep. 2021, 24, 856. [Google Scholar] [CrossRef]
- Kim, H.; Park, J.; Kim, Y.; Sohn, A.; Yeo, I.; Yu, S.J.; Yoon, J.-H.; Park, T.; Kim, Y. Serum fibronectin distinguishes the early stages of hepatocellular carcinoma. Sci. Rep. 2017, 7, 9449. [Google Scholar] [CrossRef] [PubMed]
- Waalkes, S.; Atschekzei, F.; Kramer, M.W.; Hennenlotter, J.; Vetter, G.; Becker, J.U.; Stenzl, A.; Merseburger, A.S.; Schrader, A.J.; Kuczyk, M.A.; et al. Fibronectin 1 mRNA expression correlates with advanced disease in renal cancer. BMC Cancer 2010, 10, 503. [Google Scholar] [CrossRef] [PubMed]
- Goldhirsch, A.; Ingle, J.N.; Gelber, R.D.; Coates, A.S.; Thürlimann, B.; Senn, H.J. Thresholds for therapies: Highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2009. Ann. Oncol. 2009, 20, 1319–1329. [Google Scholar] [CrossRef] [PubMed]
- Goldhirsch, A.; Wood, W.C.; Coates, A.S.; Gelber, R.D.; Thürlimann, B.; Senn, H.J. Strategies for subtypes—Dealing with the diversity of breast cancer: Highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2011. Ann. Oncol. 2011, 22, 1736–1747. [Google Scholar] [CrossRef] [PubMed]
- Wentzensen, N.; Schwartz, L.; Zuna, R.E.; Smith, K.; Mathews, C.; Gold, M.A.; Allen, R.A.; Zhang, R.; Dunn, S.T.; Walker, J.L.; et al. Performance of p16/Ki-67 immunostaining to detect cervical cancer precursors in a colposcopy referral population. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2012, 18, 4154–4162. [Google Scholar] [CrossRef]
- Huang, M.Z.; Huang, S.; Li, D.Q.; Nie, X.M.; Li, H.B.; Jiang, X.M. The study of the combination detection of HPV-DNA and p16INK4a in cervical lesions. Med. Oncol. 2011, 28 (Suppl. 1), 547–552. [Google Scholar] [CrossRef]
- Indarti, J.; Fernando, D. Comparison of p16INK4a immunocytochemistry with the HPV polymerase chain reaction in predicting high grade cervical squamous intraepithelial lesions. Asian Pac. J. Cancer Prev. 2013, 14, 4989–4992. [Google Scholar] [CrossRef]
- Liao, G.-D.; Sellors, J.W.; Sun, H.-K.; Zhang, X.; Bao, Y.-P.; Jeronimo, J.; Chen, W.; Zhao, F.-H.; Song, Y.; Cao, Z.; et al. p16INK4A immunohistochemical staining and predictive value for progression of cervical intraepithelial neoplasia grade 1: A prospective study in China. Int. J. Cancer 2014, 134, 1715–1724. [Google Scholar] [CrossRef]
- Ma, Y.-Y.; Cheng, X.-D.; Zhou, C.-Y.; Qiu, L.-Q.; Chen, X.-D.; Lü, W.-G.; Xie, X. Value of P16 expression in the triage of liquid-based cervical cytology with atypical squamous cells of undetermined significance and low-grade squamous intraepithelial lesions. Chin. Med. J. 2011, 124, 2443–2447. [Google Scholar]
- Valasoulis, G.; Stasinou, S.-M.; Nasioutziki, M.; Athanasiou, A.; Zografou, M.; Spathis, A.; Loufopoulos, A.; Karakitsos, P.; Paraskevaidis, E.; Kyrgiou, M. Expression of HPV-related biomarkers and grade of cervical intraepithelial lesion at treatment. Acta Obstet. Gynecol. Scand. 2014, 93, 194–200. [Google Scholar] [CrossRef]
- Sarma, N.C. Evidence-based Review, Grade of Recommendation, and Suggested Treatment Recommendations for Melasma. Indian Dermatol. Online J. 2017, 8, 406–442. [Google Scholar] [CrossRef] [PubMed]
- van Baars, R.; Griffin, H.; Wu, Z.; Soneji, Y.; van de Sandt, M.M.; Arora, R.; van der Marel, J.; ter Harmsel, B.; Jach, R.; Okon, K.; et al. Investigating Diagnostic Problems of CIN1 and CIN2 Associated with High-risk HPV by Combining the Novel Molecular Biomarker PanHPVE4 with P16INK4a. Am. J. Surg. Pathol. 2015, 39, 1518–1528. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.S.; Oh, S.; Jung, E.-J.; Park, J.H.; Jeon, H.-W.; Lee, T.S.; Kim, J.H.; Choi, E.; Byeon, S.-J.; Park, I.-A. High-risk human papillomavirus load and biomarkers in cervical intraepithelial neoplasia and cancer. Acta Pathol. Microbiol. Immunol. Scand. 2014, 122, 427–436. [Google Scholar] [CrossRef] [PubMed]
- Koo, Y.-J.; Hahn, H.-S.; Lee, I.-H.; Lim, K.-T.; Lee, K.-H.; Kim, H.-S.; Kim, T.-J.; Chun, Y.-K.; Kim, H.-S.; Hong, S.-R. Dual immunostaining of cervical cytology specimens with atypical squamous cells for p16/Ki-67 does not exclude the existence of a high-grade squamous intraepithelial lesion. Virchows Arch. Int. J. Pathol. 2013, 463, 689–696. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Liu, J.; Gong, L.; Sun, X.; Long, W. Combining HPV DNA load with p16/Ki-67 staining to detect cervical precancerous lesions and predict the progression of CIN1-2 lesions. Virol. J. 2019, 16, 117. [Google Scholar] [CrossRef]
- Jackson, J.A.; Kapur, U.; Erşahin, Ç. Utility of p16, Ki-67, and HPV test in diagnosis of cervical intraepithelial neoplasia and atrophy in women older than 50 years with 3- to 7-year follow-up. Int. J. Surg. Pathol. 2012, 20, 146–153. [Google Scholar] [CrossRef]
- Alhamlan, F.; Obeid, D.; Khayat, H.; Asma, T.; Al-Badawi, I.A.; Almutairi, A.; Almatrrouk, S.; Fageeh, M.; Bakhrbh, M.; Nassar, M.; et al. Prognostic impact of human papillomavirus infection on cervical dysplasia, cancer, and patient survival in Saudi Arabia: A 10-year retrospective analysis. Ann. Saudi Med. 2021, 41, 350–360. [Google Scholar] [CrossRef]
- Rodriguez-Carunchio, L.; Soveral, I.; Steenbergen, R.D.M.; Torné, A.; Martinez, S.; Fusté, P.; Pahisa, J.; Marimon, L.; Ordi, J.; del Pino, M. HPV-negative carcinoma of the uterine cervix: A distinct type of cervical cancer with poor prognosis. BJOG Int. J. Obstet. Gynaecol. 2015, 122, 119–127. [Google Scholar] [CrossRef]
- Lei, J.; Ploner, A.; Lagheden, C.; Eklund, C.; Kleppe, S.N.; Andrae, B.; Elfström, K.M.; Dillner, J.; Sparén, P.; Sundström, K. High-risk human papillomavirus status and prognosis in invasive cervical cancer: A nationwide cohort study. PLoS Med. 2018, 15, e1002666. [Google Scholar] [CrossRef]
- Narisawa-Saito, M.; Kiyono, T. Basic mechanisms of high-risk human papillomavirus-induced carcinogenesis: Roles of E6 and E7 proteins. Cancer Sci. 2007, 98, 1505–1511. [Google Scholar] [CrossRef]
- Haltas, H.; Bayrak, R.; Yenidunya, S.; Yildirim, U. The immunohistochemical detection of P16 and HPV L1 capsid protein on cell block sections from residual PapSpin liquid-based gynecology cytology specimens as a diagnostic and prognostic tool. Eur. Rev. Med. Pharmacol. Sci. 2012, 16, 1588–1595. [Google Scholar] [PubMed]
- Pabuccu, E.G.; Taskin, S.; Ustun, H.; Gungor, M.; Aytac, R.; Yalcin, I.; Ortac, F. Diagnostic performance of p16 staining in atypical squamous cells “cannot exclude high-grade squamous epithelial lesion” in predicting high-grade cervical pathology. J. Obstet. Gynaecol. 2014, 34, 730–734. [Google Scholar] [CrossRef] [PubMed]
- Pacchiarotti, A.; Ferrari, F.; Bellardini, P.; Chini, F.; Collina, G.; Palma, P.D.; Ghiringhello, B.; Maccallini, V.; Musolino, F.; Negri, G.; et al. Prognostic value of p16-INK4A protein in women with negative or CIN1 histology result: A follow-up study. Int. J. Cancer 2014, 134, 897–904. [Google Scholar] [CrossRef] [PubMed]
- Sarma, U.; Biswas, I.; Das, A.; Das, G.C.; Saikia, C.; Sarma, B. p16INK4a Expression in Cervical Lesions Correlates with Histologic Grading—A Tertiary Level Medical Facility Based Retrospective Study. Asian Pac. J. Cancer Prev. 2017, 18, 2643–2647. [Google Scholar] [CrossRef] [PubMed]
- Tsoumpou, I.; Valasoulis, G.; Founta, C.; Kyrgiou, M.; Nasioutziki, M.; Daponte, A.; Koliopoulos, G.; Malamou-Mitsi, V.; Karakitsos, P.; Paraskevaidis, E. High-risk human papillomavirus DNA test and p16(INK4a) in the triage of LSIL: A prospective diagnostic study. Gynecol. Oncol. 2011, 121, 49–53. [Google Scholar] [CrossRef] [PubMed]
- Celewicz, A.; Celewicz, M.; Wężowska, M.; Chudecka-Głaz, A.; Menkiszak, J.; Urasińska, E. Clinical efficacy of p16/Ki-67 dual-stained cervical cytology in secondary prevention of cervical cancer. Pol. J. Pathol. 2018, 69, 42–47. [Google Scholar] [CrossRef]
- Diouf, D.; Diop, G.; Fall, C.; Sarr, S.; Diarra, C.A.T.; Ngom, A.I.; Ka, S.; Lo, S.; Faye, O.; Dem, A. The Association of Molecular Biomarkers in the Diagnosis of Cervical Pre-Cancer and Cancer and Risk Factors in Senegalese. Asian Pac. J. Cancer Prev. 2020, 21, 3221–3227. [Google Scholar] [CrossRef]
- El-Zein, M.; Gotlieb, W.; Gilbert, L.; Hemmings, R.; Behr, M.A.; Franco, E.L.; The STAIN-IT Study Group. Dual staining for p16/Ki-67 to detect high-grade cervical lesions: Results from the Screening Triage Ascertaining Intraepithelial Neoplasia by Immunostain Testing study. Int. J. Cancer 2021, 148, 492–501. [Google Scholar] [CrossRef]
- Frega, A.; Pavone, M.; Sesti, F.; Leone, C.; Bianchi, P.; Cozza, G.; Colombrino, C.; Lukic, A.; Marziani, R.; De Sanctis, L.; et al. Sensitivity and specificity values of high-risk HPV DNA, p16/ki-67 and HPV mRNA in young women with atypical squamous cells of undetermined significance (ASCUS) or low-grade squamous intraepithelial lesion (LSIL). Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 10672–10677. [Google Scholar] [CrossRef]
- Ngugi, C.W.; Schmidt, D.; Wanyoro, K.; Boga, H.; Wanzala, P.; Muigai, A.; Mbithi, J.; Doeberitz, M.V.K.; Reuschenbach, M. p16(INK4a)/Ki-67 dual stain cytology for cervical cancer screening in Thika district, Kenya. Infect. Agent Cancer 2015, 10, 25. [Google Scholar] [CrossRef]
- Stănculescu, R.; Brătilă, E.; Bauşic, V.; Vlădescu, T. The triage of low-grade cytological abnormalities by the immunocytological expression of cyclin-dependent kinase inhibitor p16INK4a versus Human Papillomavirus test: A real possibility to predict cervical intraepithelial neoplasia CIN2 or CIN2+. Rom. J. Morphol. Embryol. 2013, 54, 1061–1065. [Google Scholar] [PubMed]
- Waldstrøm, M.; Christensen, R.K.; Ørnskov, D. Evaluation of p16(INK4a)/Ki-67 dual stain in comparison with an mRNA human papillomavirus test on liquid-based cytology samples with low-grade squamous intraepithelial lesion. Cancer Cytopathol. 2013, 121, 136–145. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.-L.; Chen, W.; Lei, X.-Q.; Qin, Y.; Wu, Z.-N.; Pan, Q.-J.; Zhang, X.; Chang, B.-F.; Zhang, S.-K.; Guo, H.-Q.; et al. Evaluation of p16/Ki-67 dual staining in detection of cervical precancer and cancers: A multicenter study in China. Oncotarget 2016, 7, 21181–21189. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.-L.; Guo, H.-Q.; Lei, X.-Q.; Qin, Y.; Wu, Z.-N.; Kang, L.-N.; Zhang, X.; Qiao, Y.-L.; Chen, W. p16/Ki-67 co-expression associates high risk human papillomavirus persistence and cervical histopathology: A 3-year cohort study in China. Oncotarget 2016, 7, 64810–64819. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.-K.; Jia, M.-M.; Zhao, D.-M.; Wu, Z.-N.; Guo, Z.; Liu, Y.-L.; Guo, P.-P.; Chen, Q.; Cao, X.-Q.; Liu, S.-Z.; et al. Evaluation of p16/Ki-67 dual staining in the detection of cervical precancer and cancer in China. Cancer Epidemiol. 2019, 59, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Ren, C.; Yang, L.; Zhang, X.; Liu, L.; Wang, Z. Performance of p16/Ki67 immunostaining, HPV E6/E7 mRNA testing, and HPV DNA assay to detect high-grade cervical dysplasia in women with ASCUS. BMC Cancer 2019, 19, 271. [Google Scholar] [CrossRef] [PubMed]
- Donà, M.G.; Vocaturo, A.; Giuliani, M.; Ronchetti, L.; Rollo, F.; Pescarmona, E.; Carosi, M.; Vocaturo, G.; Benevolo, M. p16/Ki-67 dual staining in cervico-vaginal cytology: Correlation with histology, Human Papillomavirus detection and genotyping in women undergoing colposcopy. Gynecol. Oncol. 2012, 126, 198–202. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Gong, J.; Xu, H.; Zhang, D.; Xia, N.; Li, H.; Song, K.; Lv, T.; Chen, Y.; Diao, Y.; et al. Good performance of p16/Ki-67 dual-stain cytology for detection and post-treatment surveillance of high-grade CIN/VAIN in a prospective, cross-sectional study. Diagn. Cytopathol. 2020, 48, 635–644. [Google Scholar] [CrossRef]
- Toll, A.D.; Kelly, D.; Maleki, Z. Utility of P16 expression and Ki-67 proliferation index in ASCUS and ASC-H pap tests. Diagn. Cytopathol. 2014, 42, 576–581. [Google Scholar] [CrossRef] [PubMed]
- Gatta, L.B.; Berenzi, A.; Balzarini, P.; Dessy, E.; Angiero, F.; Alessandri, G.; Gambino, A.; Grigolato, P.; Benetti, A. Diagnostic implications of L1, p16, and Ki-67 proteins and HPV DNA in low-grade cervical intraepithelial neoplasia. Int. J. Gynecol. Pathol. 2011, 30, 597–604. [Google Scholar] [CrossRef]
- Chen, S.M.; Lin, W.; Liu, X.; Zhang, Y.Z. Significance of human telomerase RNA gene amplification detection for cervical cancer screening. Asian Pac. J. Cancer Prev. 2012, 13, 2063–2068. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Xu, C.; Xu, M.; Yuan, Y.; Sun, Y.; Zhao, H.; Zhang, X. Genomic amplification of hTERC in paraffin-embedded tissues of cervical intraepithelial neoplasia and invasive cancer. Int. J. Gynecol. Pathol. 2012, 31, 280–285. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Pan, Q.; Pan, J.; Chen, Y.; Cao, L. Study on the correlation between hTREC and HPV load and cervical CINI/II/III lesions and cervical cancer. J. Clin. Lab. Anal. 2020, 34, e23257. [Google Scholar] [CrossRef] [PubMed]
- Ji, W.; Lou, W.; Hong, Z.; Qiu, L.; Di, W. Genomic amplification of HPV, h-TERC and c-MYC in liquid-based cytological specimens for screening of cervical intraepithelial neoplasia and cancer. Oncol. Lett. 2019, 17, 2099–2106. [Google Scholar] [CrossRef]
- Jiang, J.; Wei, L.-H.; Li, Y.-L.; Wu, R.-F.; Xie, X.; Feng, Y.-J.; Zhang, G.; Zhao, C.; Zhao, Y.; Chen, Z. Detection of TERC amplification in cervical epithelial cells for the diagnosis of high-grade cervical lesions and invasive cancer: A multicenter study in China. J. Mol. Diagn. 2010, 12, 808–817. [Google Scholar] [CrossRef]
- Jin, Y.; Li, J.-P.; He, D.; Tang, L.-Y.; Zee, C.-S.; Guo, S.-Z.; Zhou, J.; Chen, J.-N.; Shao, C.-K. Clinical significance of human telomerase RNA gene (hTERC) amplification in cervical squamous cell lesions detected by fluorescence in situ hybridization. Asian. Pac. J. Cancer Prev. 2011, 12, 1167–1171. [Google Scholar]
- Kudela, E.; Visnovsky, J.; Balharek, T.; Farkasova, A.; Zubor, P.; Plank, L.; Danko, J. Different amplification patterns of 3q26 and 5p15 regions in cervical intraepithelial neoplasia and cervical cancer. Ann. Diagn. Pathol. 2018, 35, 16–20. [Google Scholar] [CrossRef]
- Kuglik, P.; Kasikova, K.; Smetana, J.; Vallova, V.; Lastuvkova, A.; Moukova, L.; Cvanova, M.; Brozova, L. Molecular cytogenetic analyses of hTERC (3q26) and MYC (8q24) genes amplifications in correlation with oncogenic human papillomavirus infection in Czech patients with cervical intraepithelial neoplasia and cervical carcinomas. Neoplasma 2015, 62, 130–139. [Google Scholar] [CrossRef][Green Version]
- Li, Y.; Zeng, W.-J.; Ye, F.; Wang, X.-Y.; Lü, W.-G.; Ma, D.; Wei, L.-H.; Xie, X. Application of hTERC in thinprep samples with mild cytologic abnormality and HR-HPV positive. Gynecol. Oncol. 2011, 120, 73–83. [Google Scholar] [CrossRef]
- Li, T.; Tang, L.; Bian, D.; Jia, Y.; Huang, X.; Zhang, X. Detection of hTERC and c-MYC genes in cervical epithelial exfoliated cells for cervical cancer screening. Int. J. Mol. Med. 2014, 33, 1289–1297. [Google Scholar] [CrossRef][Green Version]
- Yin, G.; Li, J.; Zhu, T.; Zhao, X. The detection of hTERC amplification using fluorescence in situ hybridization in the diagnosis and prognosis of cervical intraepithelial neoplasia: A case control study. World J. Surg. Oncol. 2012, 10, 168. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Liang, P.; Zheng, Y.; Yi, P.; Liu, Q.; Han, J.; Huang, Y.; Zhou, Y.; Guo, J.; Li, L. Clinical significance of hTERC gene detection in exfoliated cervical epithelial cells for cervical lesions. Int. J. Gynecol. Cancer 2013, 23, 785–790. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Han, Y.; Tian, T.; Su, P.; Jin, G.; Chen, J.; Cao, Y. MiR-21-5p, miR-34a, and human telomerase RNA component as surrogate markers for cervical cancer progression. Pathol. Res. Pract. 2018, 214, 374–379. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.; Taye, J.; Yu, K.; Park, S.; Kim, J.; Kim, S.; Lee, D.; Wang, H.-Y.; Park, K.H.; Lee, H. HPV E6/E7, hTERT, and Ki67 mRNA RT-qPCR Assay for Detecting High-Grade Cervical Lesion with Microscope Slides. Anal. Cell. Pathol. 2019, 2019, 9365654. [Google Scholar] [CrossRef] [PubMed]
- Zejnullahu, V.A.; Zejnullahu, V.A.; Josifovska, S.; Vukovik, N.; Pakovski, K.; Panov, S. Correlation of hTERT Expression with Cervical Cytological Abnormalities and Human Papillomavirus Infection. Prilozi 2017, 38, 143–151. [Google Scholar] [CrossRef]
- Koeneman, M.M.; Ovestad, I.T.; Janssen, E.A.M.; Ummelen, M.; Kruitwagen, R.F.P.M.; Hopman, A.H.; Kruse, A.J. Gain of Chromosomal Region 3q26 as a Prognostic Biomarker for High-Grade Cervical Intraepithelial Neoplasia: Literature Overview and Pilot Study. Pathol. Oncol. Res. 2019, 25, 549–557. [Google Scholar] [CrossRef]
- Zhou, Y.; Shu, C.; Huang, Y. Fibronectin promotes cervical cancer tumorigenesis through activating FAK signaling pathway. J. Cell. Biochem. 2019, 120, 10988–10997. [Google Scholar] [CrossRef] [PubMed]
- Zheng, R.; Heller, D.S. High-Risk Human Papillomavirus Identification in Precancerous Cervical Intraepithelial Lesions. J. Low. Genit. Tract Dis. 2020, 24, 197–201. [Google Scholar] [CrossRef]
- Leon, K.E.; Tangudu, N.K.; Aird, K.M.; Buj, R. Loss of p16: A Bouncer of the Immunological Surveillance? Life 2021, 11, 309. [Google Scholar] [CrossRef]
- Shah, U.J.; Nasiruddin, M.; Dar, S.; Alam Khan, K.; Akhter, M.R.; Singh, N.; Rabaan, A.A.; Haque, S. Emerging biomarkers and clinical significance of HPV genotyping in prevention and management of cervical cancer. Microb. Pathog. 2020, 143, 104131. [Google Scholar] [CrossRef]
- Cuzick, J.; Bergeron, C.; Doeberitz, M.V.K.; Gravitt, P.; Jeronimo, J.; Lorincz, A.T.; Meijer, C.J.; Sankaranarayanan, R.; Snijders, P.J.; Szarewski, A. New technologies and procedures for cervical cancer screening. Vaccine 2012, 30 (Suppl. 5), F107–F116. [Google Scholar] [CrossRef] [PubMed]
- Savone, D.; Carrone, A.; Riganelli, L.; Merlino, L.; Mancino, P.; Benedetti Panici, P. Management of HPV-related cervical disease: Role of p16INK4a immunochemistry. Review of the literature. Tumori J. 2016, 102, 450–458. [Google Scholar] [CrossRef] [PubMed]
- Bergeron, C.; Ronco, G.; Reuschenbach, M.; Wentzensen, N.; Arbyn, M.; Stoler, M.; von Knebel Doeberitz, M. The clinical impact of using p16(INK4a) immunochemistry in cervical histopathology and cytology: An update of recent developments. Int. J. Cancer 2015, 136, 2741–2751. [Google Scholar] [CrossRef]
- Tornesello, M.L.; Buonaguro, L.; Giorgi-Rossi, P.; Buonaguro, F.M. Viral and cellular biomarkers in the diagnosis of cervical intraepithelial neoplasia and cancer. BioMed Res. Int. 2013, 2013, 519619. [Google Scholar] [CrossRef]
- Bergeron, C.; von Knebel Doeberitz, M. The Role of Cytology in the 21st Century: The Integration of Cells and Molecules. Acta Cytol. 2016, 60, 540–542. [Google Scholar] [CrossRef] [PubMed]
- Peeters, E.; Wentzensen, N.; Bergeron, C.; Arbyn, M. Meta-analysis of the accuracy of p16 or p16/Ki-67 immunocytochemistry versus HPV testing for the detection of CIN2+/CIN3+ in triage of women with minor abnormal cytology. Cancer Cytopathol. 2019, 127, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Darragh, T.M.; Colgan, T.J.; Cox, J.T.; Heller, D.S.; Henry, M.R.; Luff, R.D.; McCalmont, T.; Nayar, R.; Palefsky, J.M.; Stoler, M.H.; et al. Members of LAST Project Work Groups. The Lower Anogenital Squamous Terminology Standardization Project for HPV-Associated Lesions: Background and consensus recommendations from the College of American Pathologists and the American Society for Colposcopy and Cervical Pathology. Arch. Pathol. Lab. Med. 2012, 136, 1266–1297. [Google Scholar] [CrossRef] [PubMed]
- Ravaioli, S.; Tumedei, M.M.; Amadori, A.; Puccetti, M.; Chiadini, E.; Bravaccini, S. Role of Telomerase in Cervical Lesions as Prognostic Marker: A Comparison Between Immunohistochemistry and Fluorescence In Situ Hybridization. J. Low. Genit. Tract Dis. 2017, 21, 42–46. [Google Scholar] [CrossRef]
- Pańczyszyn, A.; Boniewska-Bernacka, E.; Głąb, G. Telomeres and Telomerase During Human Papillomavirus-Induced Carcinogenesis. Mol. Diagn. Ther. 2018, 22, 421–430. [Google Scholar] [CrossRef]
- Heselmeyer-Haddad, K.; Janz, V.; Castle, P.E.; Chaudhri, N.; White, N.; Wilber, K.; Morrison, L.E.; Auer, G.; Burroughs, F.H.; Sherman, M.E.; et al. Detection of genomic amplification of the human telomerase gene (TERC) in cytologic specimens as a genetic test for the diagnosis of cervical dysplasia. Am. J. Pathol. 2003, 163, 1405–1416. [Google Scholar] [CrossRef]
- Heselmeyer-Haddad, K.; Sommerfeld, K.; White, N.M.; Chaudhri, N.; Morrison, L.E.; Palanisamy, N.; Wang, Z.Y.; Auer, G.; Steinberg, W.; Ried, T. Genomic amplification of the human telomerase gene (TERC) in pap smears predicts the development of cervical cancer. Am. J. Pathol. 2005, 166, 1229–1238. [Google Scholar] [CrossRef]
- Han, B.; Wang, H.; Zhang, J.; Tian, J. FNDC3B is associated with ER stress and poor prognosis in cervical cancer. Oncol. Lett. 2020, 19, 406–414. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Voidăzan, S.T.; Dianzani, C.; Husariu, M.A.; Geréd, B.; Turdean, S.G.; Uzun, C.C.; Kovacs, Z.; Rozsnyai, F.F.; Neagu, N. The Role of p16/Ki-67 Immunostaining, hTERC Amplification and Fibronectin in Predicting Cervical Cancer Progression: A Systematic Review. Biology 2022, 11, 956. https://doi.org/10.3390/biology11070956
Voidăzan ST, Dianzani C, Husariu MA, Geréd B, Turdean SG, Uzun CC, Kovacs Z, Rozsnyai FF, Neagu N. The Role of p16/Ki-67 Immunostaining, hTERC Amplification and Fibronectin in Predicting Cervical Cancer Progression: A Systematic Review. Biology. 2022; 11(7):956. https://doi.org/10.3390/biology11070956
Chicago/Turabian StyleVoidăzan, Septimiu Toader, Caterina Dianzani, Mădălina Aurelia Husariu, Bíborka Geréd, Sabin Gligore Turdean, Cosmina Cristina Uzun, Zsolt Kovacs, Florin Francisc Rozsnyai, and Nicoleta Neagu. 2022. "The Role of p16/Ki-67 Immunostaining, hTERC Amplification and Fibronectin in Predicting Cervical Cancer Progression: A Systematic Review" Biology 11, no. 7: 956. https://doi.org/10.3390/biology11070956
APA StyleVoidăzan, S. T., Dianzani, C., Husariu, M. A., Geréd, B., Turdean, S. G., Uzun, C. C., Kovacs, Z., Rozsnyai, F. F., & Neagu, N. (2022). The Role of p16/Ki-67 Immunostaining, hTERC Amplification and Fibronectin in Predicting Cervical Cancer Progression: A Systematic Review. Biology, 11(7), 956. https://doi.org/10.3390/biology11070956







