Visual Monitoring Strategies of Sentinels in a Cooperative Breeder
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. Video Analysis
2.3. Statistical Analysis
3. Results
3.1. Look Duration
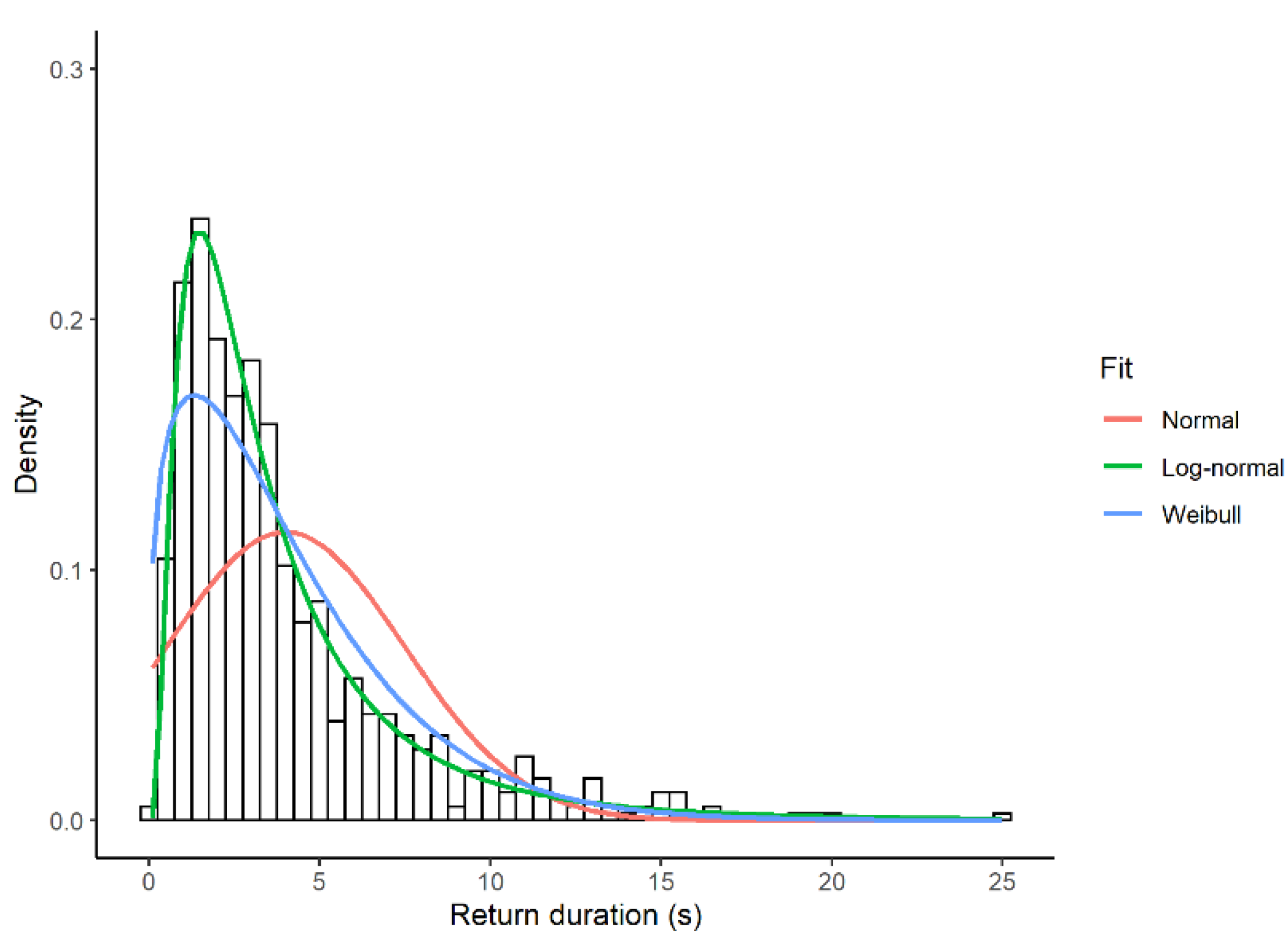
3.2. Return Times
3.3. Head Orientation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Beauchamp, G. Animal Vigilance: Monitoring Predators and Competitors; Academic Press: London, UK, 2015. [Google Scholar]
- Lima, S.L. Vigilance while feeding and its relation to the risk of predation. J. Theor. Biol. 1987, 124, 303–316. [Google Scholar] [CrossRef]
- McNamara, J.M.; Houston, A.I. Evolutionarily stable levels of vigilance as a function of group size. Anim. Behav. 1992, 43, 641–658. [Google Scholar] [CrossRef]
- Sirot, E. Adjustments in compound defensive strategies in response to variation in predation risk. Anim. Behav. 2019, 147, 53–60. [Google Scholar] [CrossRef]
- Jones, K.A.; Krebs, J.R.; Whittingham, M.J. Vigilance in the third dimension: Head movement not scan duration varies in response to different predator models. Anim. Behav. 2007, 74, 1181–1187. [Google Scholar] [CrossRef]
- Fernández-Juricic, E. Sensory basis of vigilance behavior in birds: Synthesis and future prospects. Behav. Proc. 2012, 89, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Juricic, E.; Beauchamp, G.; Treminio, R.; Hoover, M. Making heads turn: Association between head movements during vigilance and perceived predation risk in brown-headed cowbird flocks. Anim. Behav. 2011, 82, 573–577. [Google Scholar] [CrossRef]
- Bekoff, M. Vigilance, flock size, and flock geometry—Information gathering by western evening grosbeaks (Aves, Fringillidae). Ethology 1995, 99, 150–161. [Google Scholar] [CrossRef]
- Lazarus, J. Flock size and behaviour in captive red-billed weaverbirds (Quelea quelea): Implications for social facilitation and the functions of flocking. Behaviour 1979, 71, 127–145. [Google Scholar] [CrossRef]
- Cézilly, F.; Brun, B. Surveillance et picorage chez la tourterelle rieuse, Streptopelia risoria: Effets de la présence d’un congénère et de la dispersion des graines. Behaviour 1989, 110, 146–160. [Google Scholar]
- Favreau, F.-R.; Goldizen, A.W.; Pays, O. Interactions among social monitoring, anti-predator vigilance and group size in eastern grey kangaroos. Proc. R. Soc. Lond. B Biol. Sci. 2010, 277, 2089–2095. [Google Scholar] [CrossRef]
- Hirsch, B.T. Social monitoring and vigilance behavior in brown capuchin monkeys (Cebus apella). Behav. Ecol. Sociobiol. 2002, 52, 458–464. [Google Scholar] [CrossRef]
- Beauchamp, G. Should vigilance always decrease with group size? Behav. Ecol. Sociobiol. 2001, 51, 47–52. [Google Scholar] [CrossRef]
- Coolen, I.; Giraldeau, L.A.; Lavoie, M. Head position as an indicator of producer and scrounger tactics in a ground-feeding bird. Anim. Behav. 2001, 61, 895–903. [Google Scholar] [CrossRef][Green Version]
- Li, M.F.; Arseneau-Robar, T.J.M.; Smeltzer, E.A.; Teichroeb, J.A. Be early or be tolerated: Vervet monkey, Chlorocebus pygerythrus, foraging strategies in a dispersed resource. Anim. Behav. 2021, 176, 1–15. [Google Scholar] [CrossRef]
- Dawkins, M.S. What are birds looking at? Head movements and eye use in chickens. Anim. Behav. 2002, 63, 991–998. [Google Scholar]
- Bednekoff, P.A. Sentinel behavior: A review and prospectus. Adv. Study Behav. 2015, 47, 115–145. [Google Scholar] [CrossRef]
- Bell, M.B.V.; Radford, A.N.; Rose, R.; Wade, H.M.; Ridley, A.R. The value of constant surveillance in a risky environment. Proc. R. Soc. Lond. B Biol. Sci. 2009, 276, 2997–3005. [Google Scholar] [CrossRef]
- Bednekoff, P.A. Mutualism among safe, selfish sentinels: A dynamic game. Am. Nat. 1997, 150, 373–390. [Google Scholar] [CrossRef]
- Woolfenden, G.E.; Fitzpartrick, J.W. The Florida Scrub-Jay: Demography of a Cooperative-Breeding Bird; Princeton University Press: Princeton, NJ, USA, 1984. [Google Scholar]
- McGowan, K.J.; Woolfenden, G.E. A sentinel system in the Florida scrub jay. Anim. Behav. 1989, 37, 1000–1006. [Google Scholar] [CrossRef]
- Hailman, J.P.; McGowan, K.J.; Woolfenden, G.E. Role of helpers in the sentinel behaviour of the Florida scrub jay (Aphelocoma c. coerulescens). Ethology 1994, 97, 119–140. [Google Scholar] [CrossRef]
- Bednekoff, P.A.; Woolfenden, G.E. Florida scrub-jays (Aphelocoma coerulescens) are sentinels more when well-fed (even with no kin nearby). Ethology 2003, 109, 895–903. [Google Scholar] [CrossRef]
- Bednekoff, P.A.; Woolfenden, G.E. Florida Scrub-Jays compensate for the sentinel behavior of flockmates. Ethology 2006, 112, 796–800. [Google Scholar] [CrossRef]
- Woolfenden, G.E.; Fitzpatrick, J.W. Florida Scrub-Jay (Aphelocoma coerulescens). In Birds of the World; Poole, A.F., Gill, F.B., Eds.; Cornell Lab of Ornithology: Ithaca, NY, USA, 2020. [Google Scholar]
- Scannell, J.; Roberts, G.; Lazarus, J. Prey scan at random to evade observant predators. Proc. R. Soc. Lond. B Biol. Sci. 2001, 268, 541–547. [Google Scholar] [CrossRef] [PubMed]
- Bednekoff, P.A.; Lima, S.L. Why are scanning patterns so variable? An overlooked question in the study of anti-predator vigilance. J. Avian Biol. 2002, 33, 143–149. [Google Scholar]
- Fernández-Juricic, E.; O’Rourke, C.; Pitlik, T. Visual coverage and scanning behavior in two corvid species: American crow and Western scrub jay. J. Comp. Physiol. A-Neuroethol. Sens. Neural Behav. Physiol. 2010, 196, 879–888. [Google Scholar] [CrossRef]
- Land, M.F. Motion and vision: Why animals move their eyes. J. Comp. Physiol. A-Neuroethol Sens. Neural Behav. Physiol. 1999, 185, 341–352. [Google Scholar] [CrossRef]
- Beauchamp, G. Half-blind to the risk of predation. Front Ecol. Evol. 2017, 5, 131. [Google Scholar] [CrossRef]
- Yorzinski, J.L. Eye blinking in an avian species is associated with gaze shifts. Sci. Rep. 2016, 6, 32471. [Google Scholar] [CrossRef]
- Dawkins, M.S. How do hens view other hens? The use of lateral and binocular fields in social recognition. Behaviour 1995, 132, 591–606. [Google Scholar]
- Limpert, E.; Stahel, W.A.; Abbt, M. Log-normal distributions across the sciences: Keys and clues. Bioscience 2001, 51, 341–352. [Google Scholar] [CrossRef]
- Lendrem, D.W.; Stretch, D.; Metcalfe, N.B.; Jones, P. Scanning for predators in the purple sandpiper: A time-dependent or time-independent process? Anim. Behav. 1986, 34, 1577–1578. [Google Scholar] [CrossRef]
- Ochs, M.F.; Zamani, M.; Gomes, G.M.R.; de Oliveira Neto, R.C.; Kane, S.A. Sneak peek: Raptors search for prey using stochastic head turns. AUK 2016, 134, 104–115. [Google Scholar] [CrossRef]
- Bürkner, P.-C. brms: An R Package for Bayesian Multilevel Models Using Stan. J. Stat. Softw. 2017, 80, 1–28. [Google Scholar] [CrossRef]
- Vehtari, A.; Gelman, A.; Gabry, J. Practical Bayesian model evaluation using leave-one-out cross-validation and WAIC. Stat. Comput. 2017, 27, 1413–1432. [Google Scholar] [CrossRef]
- Delignette-Muller, M.L.; Dutang, C. fitdistrplus: An R Package for Fitting Distributions. J. Stat. Softw. 2015, 64, 1–34. [Google Scholar] [CrossRef]
- Agostinelli, C.; Lund, U. 2022 R Package ‘Circular’: Circular Statistics, Version 0.4-95. Available online: https://r-forge.r-project.org/projects/circular/ (accessed on 9 November 2022).
- Ruxton, G.D. Testing for departure from uniformity and estimating mean direction for circular data. Biol. Lett. 2017, 13, 20160756. [Google Scholar] [CrossRef] [PubMed]
- Franklin, W.E.; Lima, S.L. Laterality in avian vigilance: Do sparrows have a favourite eye? Anim. Behav. 2001, 62, 879–885. [Google Scholar] [CrossRef]
- Randler, C. Eye preference for vigilance during feeding in coot Fulica atra, and geese Anser anser and Anser cygnoides. Laterality 2005, 10, 535–543. [Google Scholar]
- Fourie, B.; Berezina, E.; Giljov, A.; Karenina, K. Visual lateralization in artiodactyls: A brief summary of research and new evidence on saiga antelope. Laterality 2021, 26, 106–129. [Google Scholar] [CrossRef]
- Beauchamp, G. Foraging success in a wild species of bird varies depending on which eye is used for anti-predator vigilance. Laterality 2013, 18, 194–202. [Google Scholar] [CrossRef]
- Rasa, O.A.E. The costs and effectiveness of vigilance behaviour in the dwarf mongoose: Implications for fitness and optimal group size. Ethol. Ecol. Evol. 1989, 1, 265–282. [Google Scholar] [CrossRef]
- Clutton-Brock, T.H.; O’Riain, M.J.; Brotherton, P.N.M.; Gaynor, D.; Kansky, R.; Griffin, A.S.; Manser, M. Selfish sentinels in cooperative mammals. Science 1999, 284, 1640–1644. [Google Scholar] [CrossRef] [PubMed]
- Ridley, A.R.; Raihani, N.J. Facultative response to a kleptoparasite by the cooperatively breeding pied babbler. Behav. Ecol. 2007, 18, 324–330. [Google Scholar] [CrossRef]
- Pulliam, H.R. On the advantages of flocking. J. Theor. Biol. 1973, 38, 419–422. [Google Scholar] [CrossRef]
- Bertram, B.C.R. Living in groups: Predator and prey. In Behavioural Ecology; Krebs, J.R., Davies, N.B., Eds.; Blackwell: Oxford, UK, 1978; pp. 64–96. [Google Scholar]
- Moore, B.A.; Doppler, M.; Young, J.E.; Fernández-Juricic, E. Interspecific differences in the visual system and scanning behavior of three forest passerines that form heterospecific flocks. J. Comp. Physiol. A-Neuroethol. Sens. Neural. Behav. Physiol. 2013, 199, 263–277. [Google Scholar] [CrossRef]
- Griffin, A.S.; Evans, C.S.; Blumstein, D.T. Learning specificity in acquired predator recognition. Anim. Behav. 2001, 62, 577–589. [Google Scholar] [CrossRef]
- Stankowich, T.; Blumstein, D.T. Fear in animals: A meta-analysis and review of fear assessment. Proc. R. Soc. Lond. B Biol. Sci. 2005, 272, 2627–2634. [Google Scholar]
- Walker, L.A.; York, J.E.; Young, A.J. Sexually selected sentinels? Evidence of a role for intrasexual competition in sentinel behavior. Behav. Ecol. 2016, 27, 1461–1470. [Google Scholar]
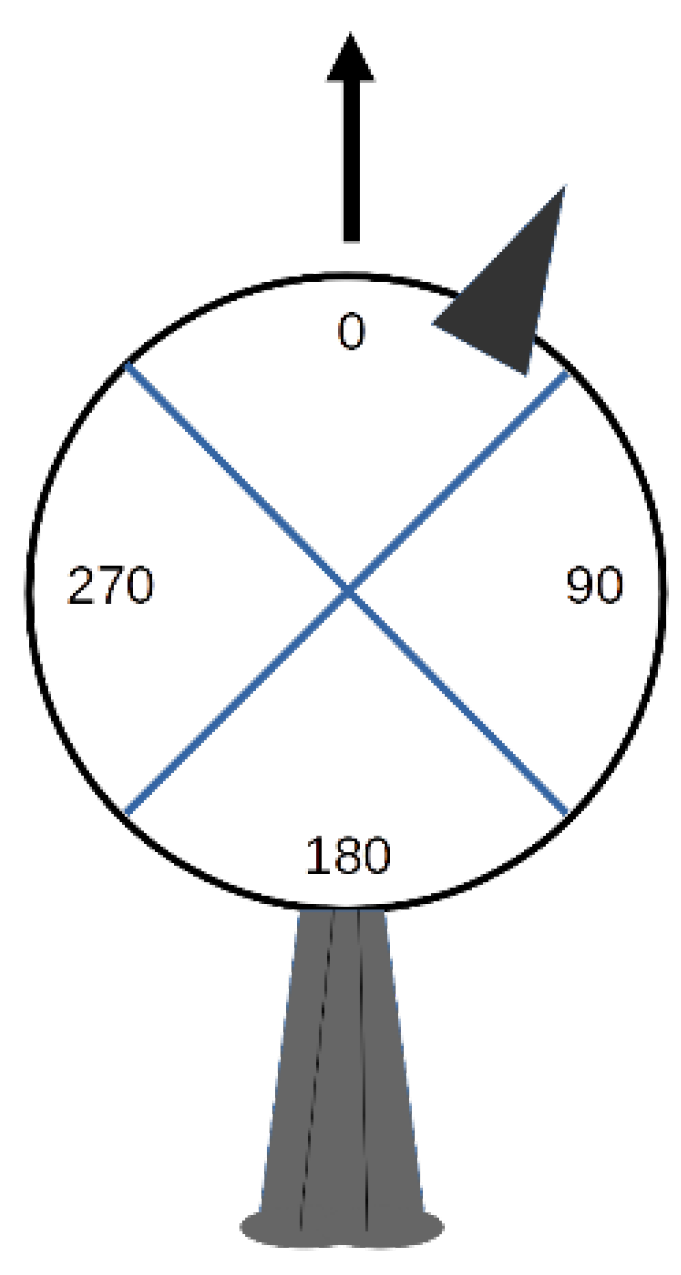
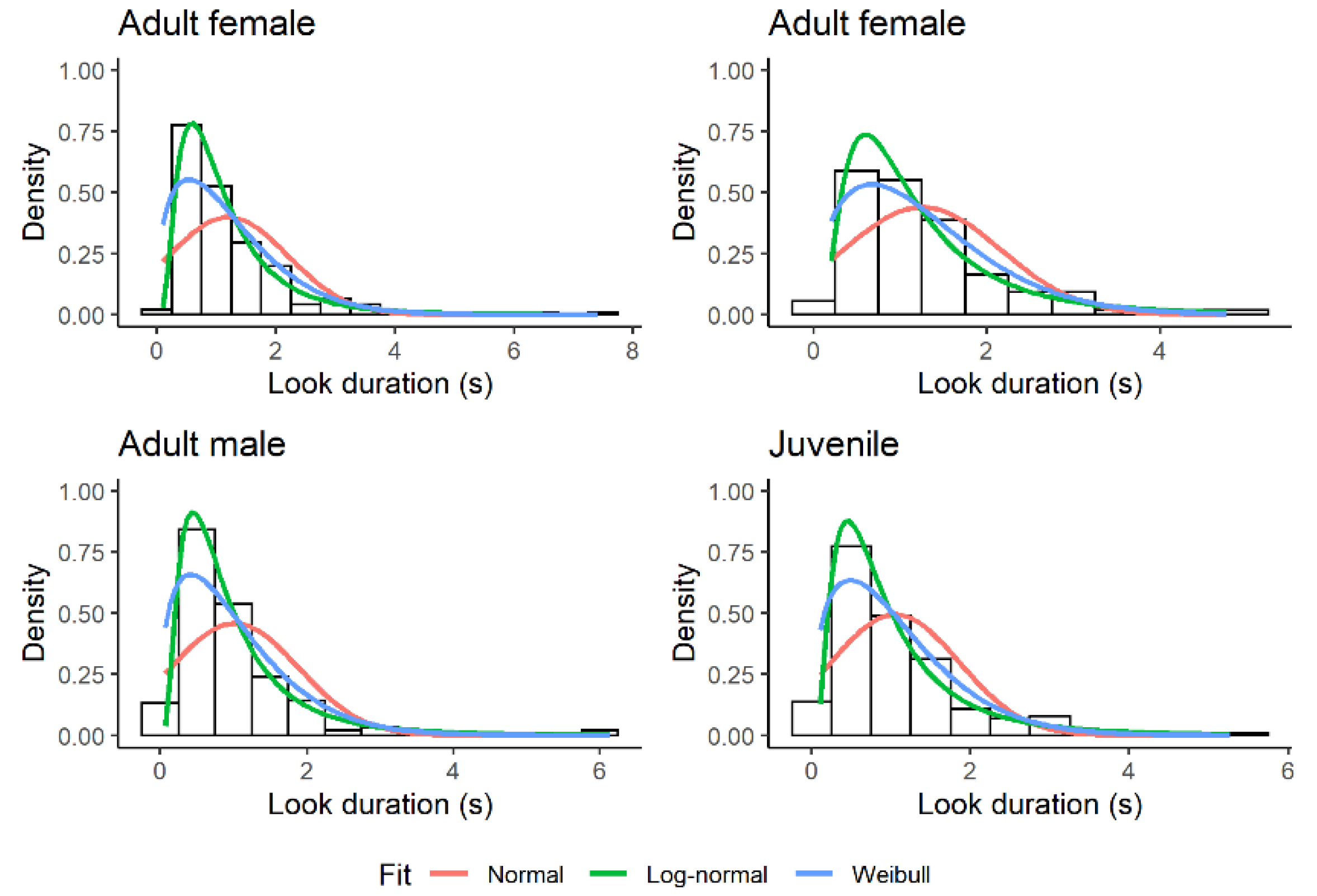
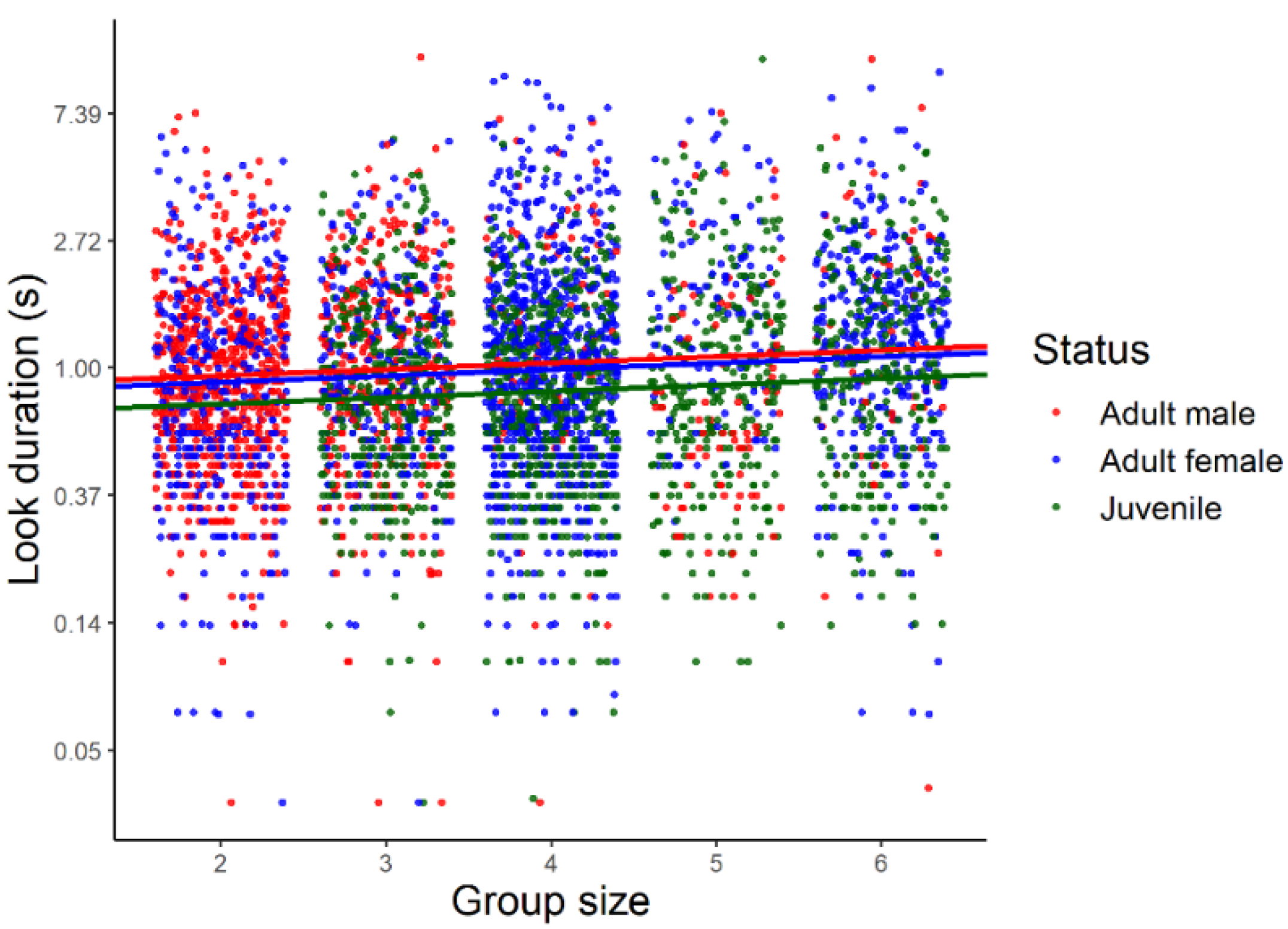
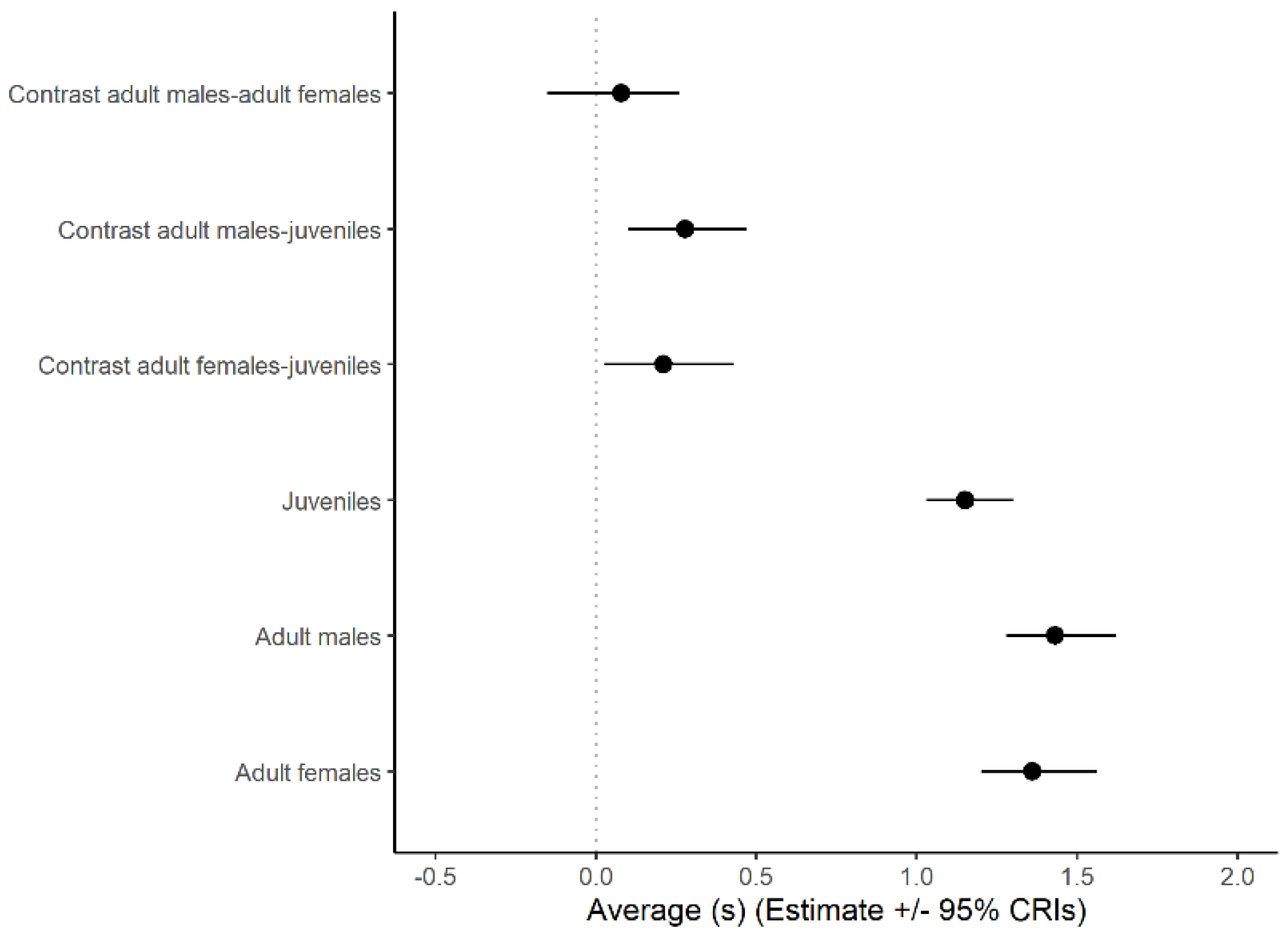
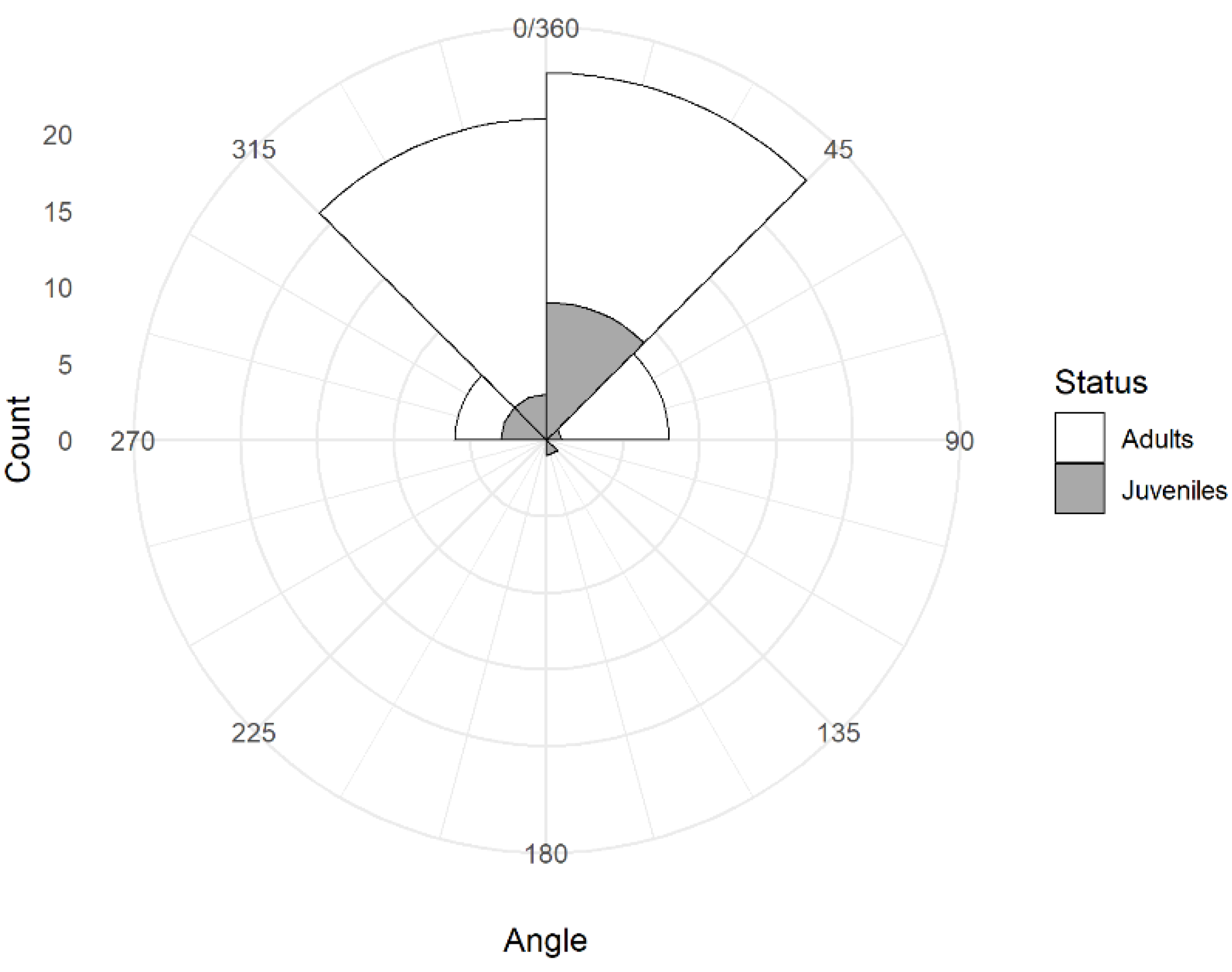
| Status | Bias to the Left | No Bias | Bias to the Right | n |
|---|---|---|---|---|
| Adult females | 6 | 7 | 6 | 19 |
| Adult males | 8 | 7 | 9 | 24 |
| Juveniles | 4 | 8 | 5 | 17 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beauchamp, G.; Bowman, R. Visual Monitoring Strategies of Sentinels in a Cooperative Breeder. Biology 2022, 11, 1769. https://doi.org/10.3390/biology11121769
Beauchamp G, Bowman R. Visual Monitoring Strategies of Sentinels in a Cooperative Breeder. Biology. 2022; 11(12):1769. https://doi.org/10.3390/biology11121769
Chicago/Turabian StyleBeauchamp, Guy, and Reed Bowman. 2022. "Visual Monitoring Strategies of Sentinels in a Cooperative Breeder" Biology 11, no. 12: 1769. https://doi.org/10.3390/biology11121769
APA StyleBeauchamp, G., & Bowman, R. (2022). Visual Monitoring Strategies of Sentinels in a Cooperative Breeder. Biology, 11(12), 1769. https://doi.org/10.3390/biology11121769






