Acute Pharmacological Effects and Oral Fluid Biomarkers of the Synthetic Cannabinoid UR-144 and THC in Recreational Users
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Design and Treatments
2.3. Procedures
2.4. Physiological Effects
2.5. Subjective Effects
2.6. Drugs Disposition in Oral Fluid
2.7. Statistical Analysis
3. Results
3.1. Participants
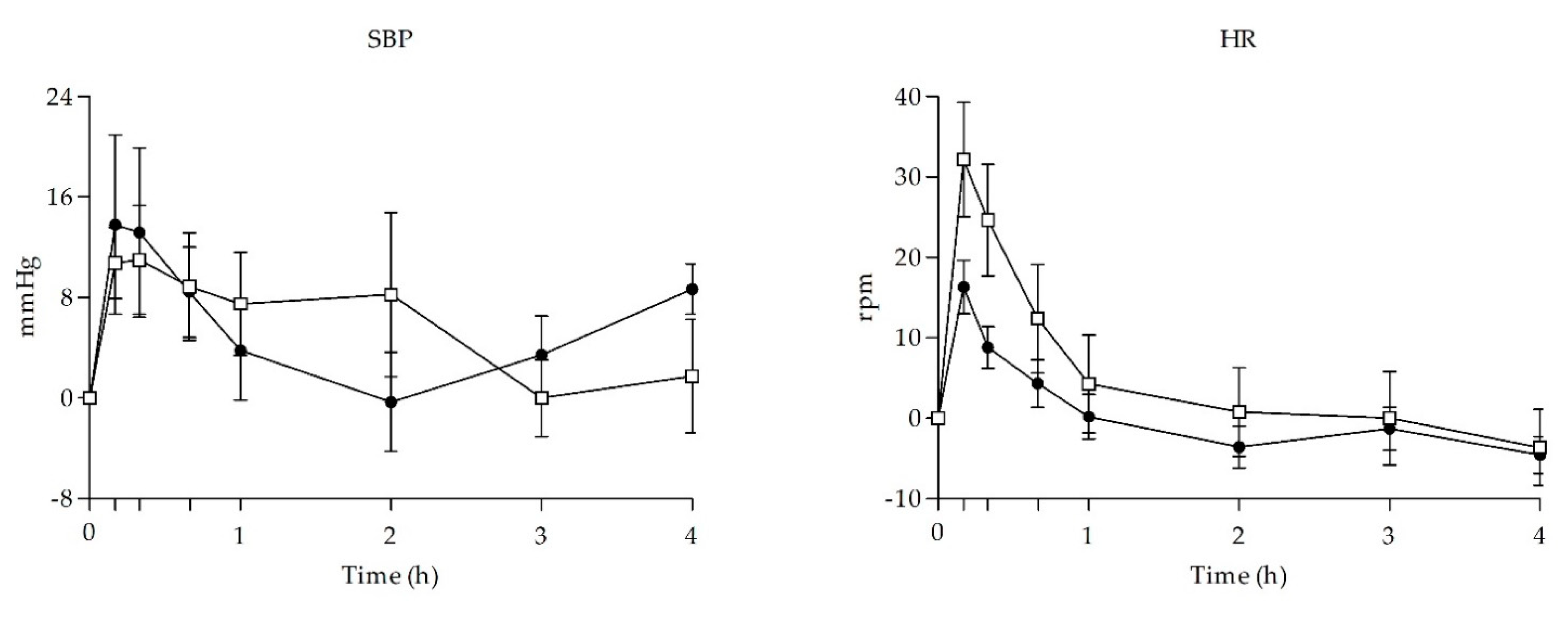
3.2. Physiological Effects
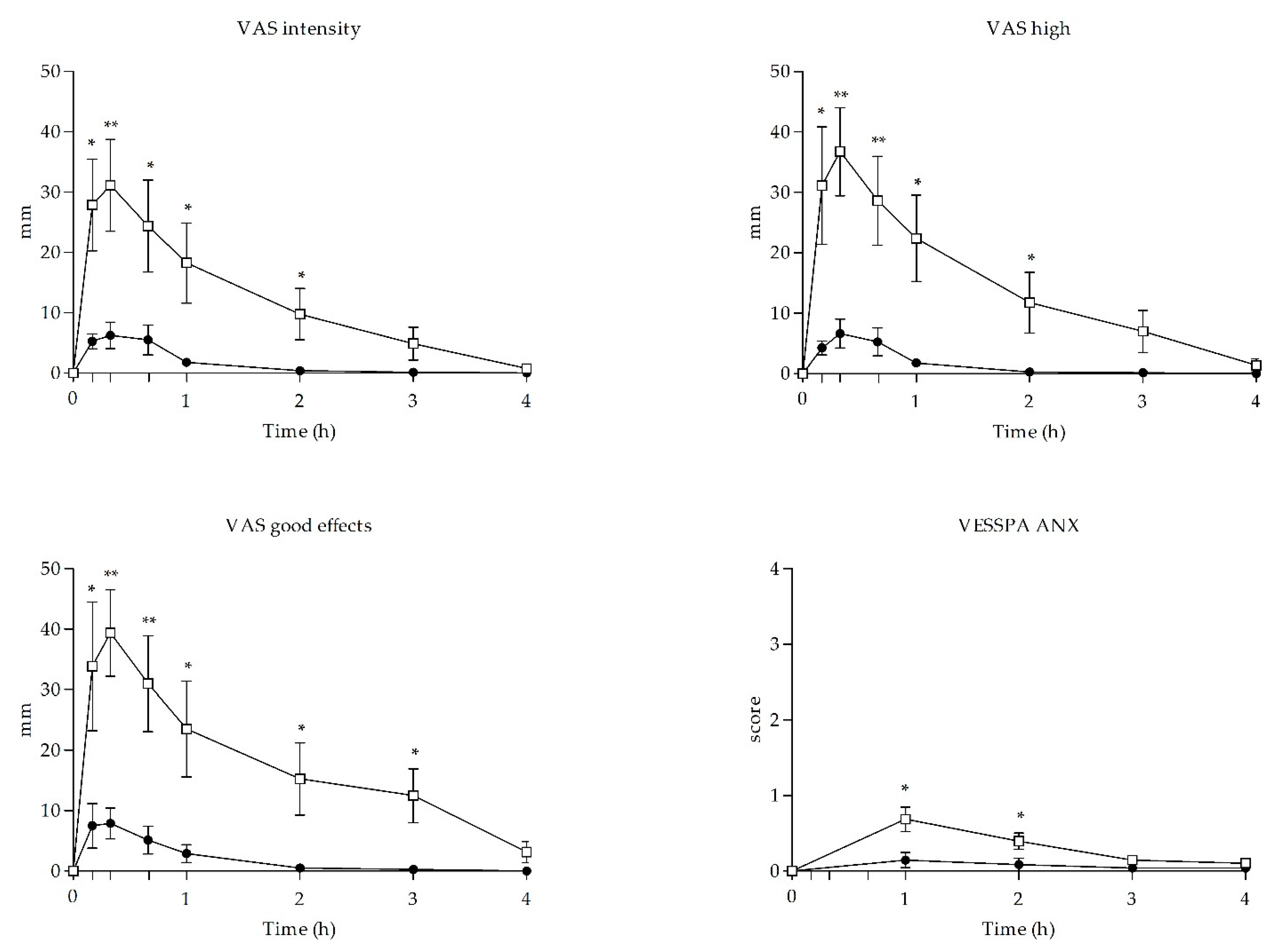
3.3. Subjective Effects
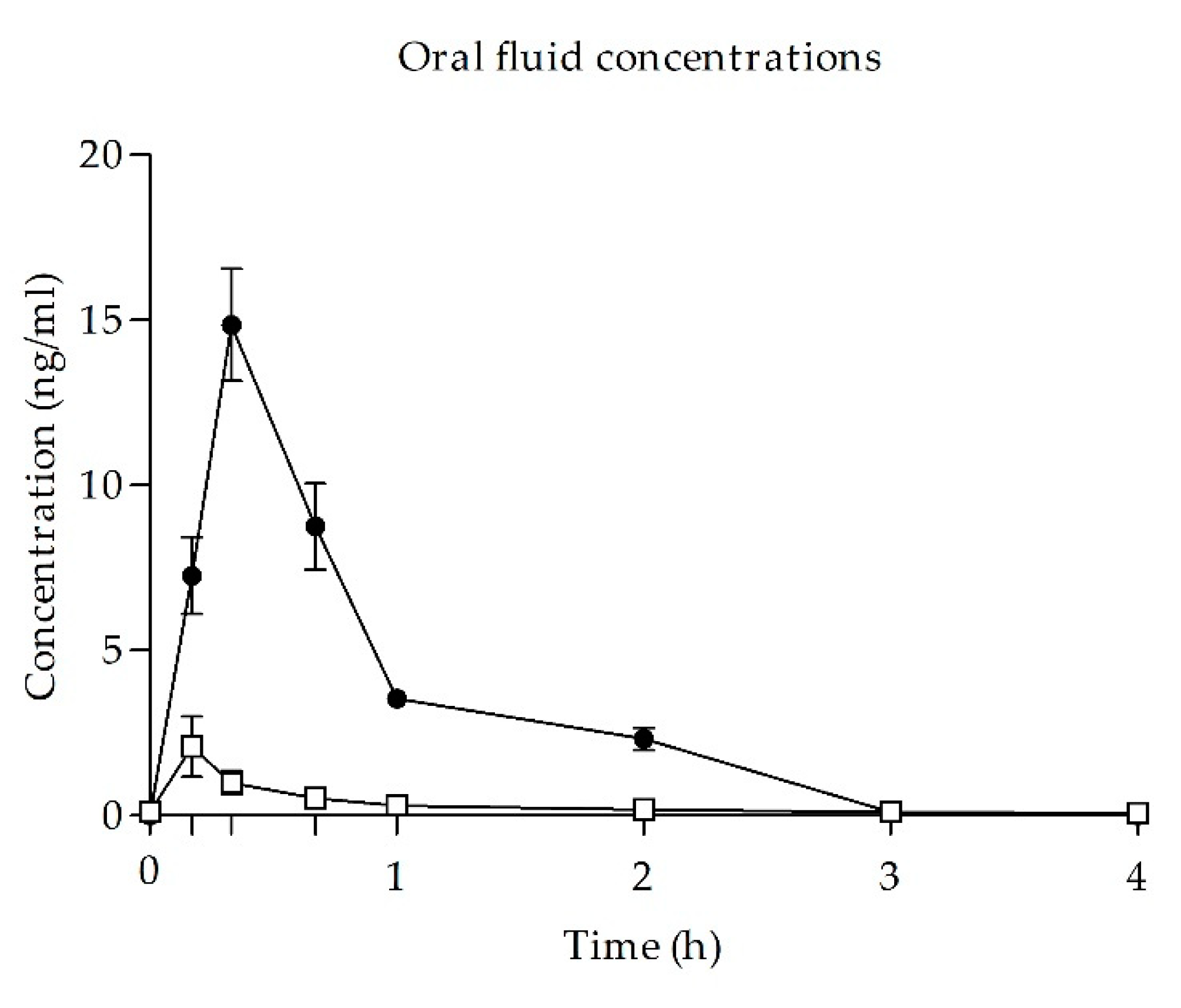
3.4. UR-144 and THC of Concentrations
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- European Monitoring Centre for Drugs and Drug Addiction. European Drug Report 2020: Key Issues; LU: Malmö, Sweden, 2020. [Google Scholar]
- United Nations Office on Drugs and Crime Cannabis: A Short Review. Available online: https://www.unodc.org/documents/drug-prevention-and-treatment/cannabis_review.pdf (accessed on 15 March 2021).
- ElSohly, M.A.; Radwan, M.M.; Gul, W.; Chandra, S.; Galal, A. Phytochemistry of Cannabis sativa L. In Phytocannabinoids: Unraveling the Complex Chemistry and Pharmacology of Cannabis Sativa; Kinghorn, A.D., Falk, H., Gibbons, S., Kobayashi, J., Eds.; Progress in the Chemistry of Organic Natural Products; Springer International Publishing: Cham, Switzerland, 2017; pp. 1–36. ISBN 978-3-319-45541-9. [Google Scholar]
- Wu, J. Cannabis, Cannabinoid Receptors, and Endocannabinoid System: Yesterday, Today, and Tomorrow. Acta Pharmacol. Sin. 2019, 40, 297–299. [Google Scholar] [CrossRef]
- Cohen, K.; Weizman, A.; Weinstein, A. Positive and Negative Effects of Cannabis and Cannabinoids on Health. Clin. Pharmacol. Ther. 2019, 105, 1139–1147. [Google Scholar] [CrossRef] [PubMed]
- Zou, S.; Kumar, U. Cannabinoid Receptors and the Endocannabinoid System: Signaling and Function in the Central Nervous System. Int. J. Mol. Sci. 2018, 19, 833. [Google Scholar] [CrossRef]
- Papaseit, E.; Pérez-Mañá, C.; Pérez-Acevedo, A.P.; Hladun, O.; Torres-Moreno, M.C.; Muga, R.; Torrens, M.; Farré, M. Cannabinoids: From Pot to Lab. Int. J. Med. Sci. 2018, 15, 1286–1295. [Google Scholar] [CrossRef] [PubMed]
- Brents, L.K.; Prather, P.L. The K2/Spice Phenomenon: Emergence, Identification, Legislation and Metabolic Characterization of Synthetic Cannabinoids in Herbal Incense Products. Drug Metab. Rev. 2014, 46, 72–85. [Google Scholar] [CrossRef]
- Synthetic Cannabinoids in Europe (Perspectives on Drugs)|Www.Emcdda.Europa.Eu. Available online: https://www.emcdda.europa.eu/publications/pods/synthetic-cannabinoids_en (accessed on 9 February 2021).
- Synthetic Cannabinoids|Www.Emcdda.Europa.Eu. Available online: https://www.emcdda.europa.eu/keywords/synthetic-cannabinoids (accessed on 9 February 2021).
- Baumann, M.H.; Solis, E.; Watterson, L.R.; Marusich, J.A.; Fantegrossi, W.E.; Wiley, J.L. Baths Salts, Spice, and Related Designer Drugs: The Science Behind the Headlines. J. Neurosci. 2014, 34, 15150–15158. [Google Scholar] [CrossRef]
- World Health Organization UR-144 Critical Review Report. Available online: https://www.who.int/medicines/areas/quality_safety/4_8_Review.pdf (accessed on 15 March 2021).
- Fabregat-Safont, D.; Ibáñez, M.; Baquero, A.; Sancho, J.V.; Hernández, F.; Haro, G. Investigation on the Consumption of Synthetic Cannabinoids among Teenagers by the Analysis of Herbal Blends and Urine Samples. J. Pharm. Biomed. Anal. 2020, 186, 113298. [Google Scholar] [CrossRef] [PubMed]
- Expert Committee on Drug Dependence Thirty-Ninth Meeting Geneva, 6–10 November 2017. World Health Organization. UR-144 Critical Review Report. Available online: https://www.who.int/medicines/access/controlled-substances/CriticalReview_UR144.pdf?ua=1 (accessed on 15 March 2021).
- Nielsen, L.M.; Holm, N.B.; Olsen, L.; Linnet, K. Cytochrome P450-Mediated Metabolism of the Synthetic Cannabinoids UR-144 and XLR-11. Drug Test. Anal. 2016, 8, 792–800. [Google Scholar] [CrossRef] [PubMed]
- Adamowicz, P.; Gieroń, J.; Gil, D.; Lechowicz, W.; Skulska, A.; Tokarczyk, B. The Effects of Synthetic Cannabinoid UR-144 on the Human Body—A Review of 39 Cases. Forensic Sci. Int. 2017, 273, e18–e21. [Google Scholar] [CrossRef] [PubMed]
- Adamowicz, P.; Zuba, D.; Sekuła, K. Analysis of UR-144 and Its Pyrolysis Product in Blood and Their Metabolites in Urine. Forensic Sci. Int. 2013, 233, 320–327. [Google Scholar] [CrossRef]
- Adamowicz, P.; Lechowicz, W. The Influence of Synthetic Cannabinoid UR-144 on Human Psychomotor Performance—A Case Report Demonstrating Road Traffic Risks. Traffic Inj. Prev. 2015, 16, 754–759. [Google Scholar] [CrossRef] [PubMed]
- Tournebize, J.; Gibaja, V.; Kahn, J.-P. Acute Effects of Synthetic Cannabinoids: Update 2015. Subst. Abuse 2017, 38, 344–366. [Google Scholar] [CrossRef] [PubMed]
- Karinen, R.; Tuv, S.S.; Øiestad, E.L.; Vindenes, V. Concentrations of APINACA, 5F-APINACA, UR-144 and Its Degradant Product in Blood Samples from Six Impaired Drivers Compared to Previous Reported Concentrations of Other Synthetic Cannabinoids. Forensic Sci. Int. 2015, 246, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Andreeva-Gateva, P.A.; Nankova, V.H.; Angelova, V.T.; Gatev, T.N. Synthetic Cannabimimetics in Bulgaria 2010–2013. Drug Alcohol Depend. 2015, 157, 200–204. [Google Scholar] [CrossRef] [PubMed]
- Paul, A.B.M.; Simms, L.; Amini, S.; Paul, A.E. Teens and Spice: A Review of Adolescent Fatalities Associated with Synthetic Cannabinoid Use. J. Forensic Sci. 2018, 63, 1321–1324. [Google Scholar] [CrossRef]
- Grifell, M.; Ventura, M.; Carbón, X.; Quintana, P.; Galindo, L.; Palma, Á.; Fornis, I.; Gil, C.; Farre, M.; Torrens, M. Patterns of use and toxicity of new para-halogenated substituted cathinones: 4-CMC (clephedrone), 4-CEC (4-chloroethcatinone) and 4-BMC (brephedrone). Hum. Psychopharmacol. 2017, 32, e2621. [Google Scholar] [CrossRef] [PubMed]
- Papaseit, E.; Farré, M.; Pérez-Mañá, C.; Torrens, M.; Ventura, M.; Pujadas, M.; de la Torre, R.; González, D. Acute Pharmacological Effects of 2C-B in Humans: An Observational Study. Front. Pharmacol. 2018, 9, 206. [Google Scholar] [CrossRef] [PubMed]
- Papaseit, E.; Olesti, E.; Pérez-Mañá, C.; Torrens, M.; Grifell, M.; Ventura, M.; Pozo, O.J.; de Sousa Fernandes Perna, E.B.; Ramaekers, J.G.; de la Torre, R.; et al. Acute Effects of 2C-E in Humans: An Observational Study. Front. Pharmacol. 2020, 11, 233. [Google Scholar] [CrossRef] [PubMed]
- Lamas, X.; Farré, M.; Llorente, M.; Camí, J. Spanish Version of the 49-Item Short Form of the Addiction Research Center Inventory (ARCI). Drug Alcohol Depend. 1994, 35, 203–209. [Google Scholar] [CrossRef]
- González, D.; Torrens, M.; Farré, M. Acute Effects of the Novel Psychoactive Drug 2C-B on Emotions. BioMed Res. Int. 2015, 2015, 643878. [Google Scholar] [CrossRef] [PubMed]
- La Maida, N.; Pellegrini, M.; Papaseit, E.; Pérez-Mañá, C.; Poyatos, L.; Ventura, M.; Galindo, L.; Busardò, F.P.; Pichini, S.; Farré, M.; et al. Determination of the Synthetic Cannabinoids JWH-122, JWH-210, UR-144 in Oral Fluid of Consumers by GC-MS and Quantification of Parent Compounds and Metabolites by UHPLC-MS/MS. Int. J. Mol. Sci. 2020, 21, 9414. [Google Scholar] [CrossRef] [PubMed]
- Pichini, S.; Mannocchi, G.; Gottardi, M.; Pérez-Acevedo, A.P.; Poyatos, L.; Papaseit, E.; Pérez-Mañá, C.; Farré, M.; Pacifici, R.; Busardò, F.P. Fast and Sensitive UHPLC-MS/MS Analysis of Cannabinoids and Their Acid Precursors in Pharmaceutical Preparations of Medical Cannabis and Their Metabolites in Conventional and Non-Conventional Biological Matrices of Treated Individual. Talanta 2020, 209, 120537. [Google Scholar] [CrossRef]
- Brunetti, P.; Lo Faro, A.F.; Pirani, F.; Berretta, P.; Pacifici, R.; Pichini, S.; Busardò, F.P. Pharmacology and legal status of cannabidiol. Ann. Ist. Super. Sanita 2020, 56, 285–291. [Google Scholar] [CrossRef]
- Pagano, S.; Coniglio, M.; Valenti, C.; Federici, M.I.; Lombardo, G.; Cianetti, S.; Marinucci, L. Biological effects of Cannabidiol on normal human healthy cell populations: Systematic review of the literature. Biomed. Pharmacother. 2020, 132, 110728. [Google Scholar] [CrossRef]
- Volkow, N.D.; Baler, R.D.; Compton, W.M.; Weiss, S.R. Adverse health effects of marijuana use. N. Engl. J. Med. 2014, 370, 2219–2227. [Google Scholar] [CrossRef] [PubMed]
- Wiley, J.L.; Marusich, J.A.; Lefever, T.W.; Grabenauer, M.; Moore, K.N.; Thomas, B.F. Cannabinoids in Disguise: Δ9-Tetrahydrocannabinol-like Effects of Tetramethylcyclopropyl Ketone Indoles. Neuropharmacology 2013, 75, 145–154. [Google Scholar] [CrossRef]
- Busardò, F.P.; Pérez-Acevedo, A.P.; Pacifici, R.; Mannocchi, G.; Gottardi, M.; Papaseit, E.; Pérez-Mañá, C.; Martin, S.; Poyatos, L.; Pichini, S.; et al. Disposition of Phytocannabinoids, Their Acidic Precursors and Their Metabolites in Biological Matrices of Healthy Individuals Treated with Vaporized Medical Cannabis. Pharmaceuticals 2021, 14, 59. [Google Scholar] [CrossRef] [PubMed]
- Martínez, L.; La Maida, N.; Papaseit, E.; Pérez-Mañá, C.; Poyatos, L.; Pellegrini, M.; Pichini, S.; Ventura, M.; Galindo, L.; Busardo, F.P.; et al. Acute Pharmacological Effects and Oral Fluid Disposition Biomarkers of the Synthetic Cannabinoids JWH-122 and JWH-210 in Humans. Pharmaceuticals 2021, submitted. [Google Scholar]
- Amaratunga, P.; Thomas, C.; Lemberg, B.L.; Lemberg, D. Quantitative Measurement of XLR11 and UR-144 in Oral Fluid by LC–MS-MS. J. Anal. Toxicol. 2014, 38, 315–321. [Google Scholar] [CrossRef] [PubMed]
| Parameter | Mean ± SD | T StudentComparison Effects UR-114 vs. THC | ANOVAComparison Time-Course Effects UR-144 vs. THC | |||||
|---|---|---|---|---|---|---|---|---|
| UR-144 | THC | t | p-Value | F | p-Value | T-C points | ||
| SBP (mmHg) | Emax | 14.94 ± 22.92 | 17.63 ± 17.74 | −0.262 | 0.797 | |||
| AUC0–4h | 18.36 ± 34.25 | 21.59 ± 39.64 | −0.175 | 0.864 | ||||
| TC | 1.100 | 0.370 | -- | |||||
| DBP (mmHg) | Emax | 11.56 ± 10.15 | 15.31 ± 5.82 | −0.906 | 0.380 | |||
| AUC0–4h | 17.02 ± 22.53 | 28.74 ± 25.77 | −0.969 | 0.380 | ||||
| TC | 1.867 | 0.083 | -- | |||||
| HR (beats/min) | Emax | 10.81 ± 17.37 | 24.13 ± 31.59 | −1.044 | 0.314 | |||
| AUC0–4h | −0.74 ± 26.86 | 17.27 ± 62.54 | −0.748 | 0.467 | ||||
| TC | 3.902 | 0.001 | NS | |||||
| T (°C) | Emax | −0.38 ± 0.44 | 0.10 ± 0.46 | −0.611 | 0.551 | |||
| AUC0–4h | 0.26 ± 0.98 | 0.33 ± 1.18 | −0.117 | 0.908 | ||||
| TC | 1.747 | 0.207 | -- | |||||
| Intensity | Emax | 8.13 ± 6.22 | 39.88 ± 16.76 | −5.023 | <0.001 | |||
| AUC0–4h | 5.92 ± 5.73 | 47.62 ± 42.33 | −2.761 | 0.015 | ||||
| TC | 4.945 | <0.001 | 10, 20, 40, 1, 2 | |||||
| High | Emax | 8.75 ± 5.78 | 43.75 ± 20.99 | −4.548 | <0.001 | |||
| AUC0–4h | 5.63 ± 5.16 | 58.16 ± 48.61 | −3.039 | 0.009 | ||||
| TC | 6.520 | <0.001 | 10, 20, 40, 1, 2 | |||||
| Good effects | Emax | 11.38 ± 10.23 | 46.62 ± 21.71 | −4.154 | 0.001 | |||
| AUC0–4h | 7.56 ± 7.53 | 70.68 ± 56.91 | −3.110 | 0.008 | ||||
| TC | 5.180 | <0.001 | 10, 20, 40, 1, 2, 3 | |||||
| Bad effects | AUC0–4h | 1.25 ± 2.37 | 5.00 ± 6.39 | −1.555 | 0.142 | |||
| TC | 0.84 ± 1.98 | 3.25 ± 5.26 | −1.215 | 0.244 | ||||
| T-C | 0.841 | 0.556 | -- | |||||
| Hunger | Emax | 35.37 ± 26.92 | 30.37 ± 29.87 | 0.352 | 0.730 | |||
| AUC0–4h | 68.46 ± 70.43 | 55.89 ± 66.79 | 0.366 | 0.722 | ||||
| TC | 0.470 | 0.854 | -- | |||||
| Somnolence | Emax | 20.37 ± 23.42 | 11.50 ± 13.20 | 0.934 | 0.366 | |||
| AUC0−4 | 48.81 ± 78.76 | 22.52 ± 32.00 | 0.875 | 0.397 | ||||
| T-C | 0.618 | 0.740 | -- | |||||
| Dizziness | Emax | 3.50 ± 6.09 | 4.00 ± 3.46 | −0.202 | 0.843 | |||
| AUC0–4h | 1.15 ± 1.89 | 3.11 ± 4.67 | −1.099 | 0.290 | ||||
| TC | 1.469 | 0.187 | -- | |||||
| Confusion | Emax | 2.00 ± 4.11 | 3.26 ± 5.01 | −0.546 | 0.594 | |||
| AUC0–4h | 0.58 ± 0.90 | 1.05 ± 1.69 | −0.687 | 0.503 | ||||
| TC | 1.054 | 0.399 | -- | |||||
| Nausea | Emax | 1.37 ± 2.72 | 0.12 ± 0.35 | 1.288 | 0.219 | |||
| AUC0–4h | 0.28 ± 0.45 | 0.21 ± 0.06 | 1.606 | 0.131 | ||||
| TC | 0.517 | 0.993 | -- | |||||
| Vomit | Emax | 0.13 ± 0.35 | 0.00 ± 0.00 | 1.000 | 0.334 | |||
| AUC0–4h | 0.03 ± 0.09 | 0.00 ± 0.00 | 1.000 | 0.334 | ||||
| TC | 1.000 | 0.436 | -- | |||||
| Anxiety | Emax | 4.63 ± 12.68 | 3.50 ± 8.40 | 0.209 | 0.837 | |||
| AUC0–4h | 1.99 ± 5.53 | 1.84 ± 4.46 | 0.060 | 0.953 | ||||
| TC | 0.149 | 0.994 | -- | |||||
| Aggressiveness | Emax | 1.25 ± 3.15 | 0.00 ± 0.00 | 1.122 | 0.281 | |||
| AUC0–4h | 0.00 ± 0.00 | 0.00 ± 0.00 | 1.178 | 0.259 | ||||
| TC | 1.299 | 0.259 | -- | |||||
| Hallucinations-seeing of lights or spots | Emax | 0.36 ± 0.86 | 0.00 ± 0.00 | 0.858 | 0.405 | |||
| AUC0–4h | 0.50 ± 1.41 | 0.66 ± 1.22 | 0.243 | 0.812 | ||||
| TC | 0.758 | 0.623 | -- | |||||
| Hallucinations-hearings of sounds or voices | Emax | 0.00 ± 0.00 | 0.00 ± 0.00 | -- | -- | |||
| AUC0–4h | 0.00 ± 0.00 | 0.00 ± 0.00 | -- | -- | ||||
| TC | -- | -- | -- | |||||
| Hallucinations-seeing animals, things, insects, or people | Emax | 0.13 ± 0.35 | 0.00 ± 0.00 | 1.000 | 0.334 | |||
| AUC0–4h | 0.08 ± 2.37 | 0.00 ± 0.00 | 1.000 | 0.334 | ||||
| TC | 1.000 | 0.436 | -- | |||||
| PCAG | Emax | 2.63 ± 1.60 | 4.63 ± 2.27 | −2.041 | 0.061 | |||
| AUC0–4h | 5.50 ± 4.82 | 8.50 ± 6.88 | −1.011 | 0.329 | ||||
| TC | 1.342 | 0.266 | -- | |||||
| MBG | Emax | 1.00 ± 1.93 | 2.63 ± 2.39 | −1.498 | 0.156 | |||
| AUC0–4h | 2.69 ± 5.99 | 5.94 ± 6.04 | −1.080 | 0.298 | ||||
| TC | 2.331 | 0.067 | -- | |||||
| LSD | Emax | −0.88 ± 1.13 | 0.63 ± 2.97 | −1.335 | 0.203 | |||
| v | −1.31 ± 1.28 | 1.06 ± 4.81 | −1.351 | 0.198 | ||||
| T-C | 3.973 | 0.007 | 10 | |||||
| BG | Emax | 0.88 ± 1.55 | 0.13 ± 2.42 | 0.739 | 0.472 | |||
| AUC0–4h | 0.44 ± 0.98 | 1.38 ± 4.49 | −0.577 | 0.573 | ||||
| TC | 2.507 | 0.052 | -- | |||||
| A | Emax | 1.63 ± 2.39 | 2.25 ± 1.98 | −0.570 | 0.578 | |||
| AUC0–4h | 3.38 ± 5.26 | 5.56 ± 5.29 | −0.829 | 0.421 | ||||
| TC | 0.874 | 0.485 | -- | |||||
| S | Emax | 0.63 ± 0.52 | 0.75 ± 0.49 | −0.487 | 0.633 | |||
| AUC0–4h | 1.43 ± 1.41 | 1.44 ± 0.97 | −0.019 | 0.985 | ||||
| TC | 2.709 | 0.039 | NS | |||||
| ANX | Emax | 0.15 ± 0.29 | 0.69 ± 0.46 | −2.837 | 0.013 | |||
| AUC0–4h | 0.29 ± 0.69 | 1.28 ± 0.82 | −2.596 | 0.021 | ||||
| TC | 6.154 | <0.001 | 1, 2 | |||||
| CP | Emax | 0.02 ± 0.06 | 0.08 ± 0.13 | −1.269 | 0.225 | |||
| AUC0–4h | 0.02 ± 0.60 | 0.08 ± 0.13 | −1.269 | 0.225 | ||||
| TC | 3.444 | 0.014 | NS | |||||
| SOC | Emax | 0.77 ± 0.97 | 0.96 ± 1.13 | −0.361 | 0.724 | |||
| AUC0–4h | 1.49 ± 2.61 | 2.63 ± 3.61 | −0.721 | 0.483 | ||||
| ACT | Emax | 0.31 ± 0.56 | 0.25 ± 0.25 | 0.288 | 0.777 | |||
| AUC0–4h | 0.53 ± 0.89 | 0.54 ± 0.58 | −0.030 | 0.977 | ||||
| TC | 1.066 | 0.382 | NS | |||||
| PS | Emax | 0.11 ± 0.24 | 0.06 ± 0.89 | 0.463 | 0.650 | |||
| AUC0–4h | 0.18 ± 0.44 | 0.64 ± 0.09 | 0.721 | 0.483 | ||||
| TC | 0.409 | 0.801 | NS | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maida, N.L.; Papaseit, E.; Martínez, L.; Pérez-Mañá, C.; Poyatos, L.; Pellegrini, M.; Pichini, S.; Pacifici, R.; Ventura, M.; Galindo, L.; et al. Acute Pharmacological Effects and Oral Fluid Biomarkers of the Synthetic Cannabinoid UR-144 and THC in Recreational Users. Biology 2021, 10, 257. https://doi.org/10.3390/biology10040257
Maida NL, Papaseit E, Martínez L, Pérez-Mañá C, Poyatos L, Pellegrini M, Pichini S, Pacifici R, Ventura M, Galindo L, et al. Acute Pharmacological Effects and Oral Fluid Biomarkers of the Synthetic Cannabinoid UR-144 and THC in Recreational Users. Biology. 2021; 10(4):257. https://doi.org/10.3390/biology10040257
Chicago/Turabian StyleMaida, Nunzia La, Esther Papaseit, Lucia Martínez, Clara Pérez-Mañá, Lourdes Poyatos, Manuela Pellegrini, Simona Pichini, Roberta Pacifici, Mireia Ventura, Liliana Galindo, and et al. 2021. "Acute Pharmacological Effects and Oral Fluid Biomarkers of the Synthetic Cannabinoid UR-144 and THC in Recreational Users" Biology 10, no. 4: 257. https://doi.org/10.3390/biology10040257
APA StyleMaida, N. L., Papaseit, E., Martínez, L., Pérez-Mañá, C., Poyatos, L., Pellegrini, M., Pichini, S., Pacifici, R., Ventura, M., Galindo, L., Busardò, F. P., & Farré, M. (2021). Acute Pharmacological Effects and Oral Fluid Biomarkers of the Synthetic Cannabinoid UR-144 and THC in Recreational Users. Biology, 10(4), 257. https://doi.org/10.3390/biology10040257








