Male Sexual Preference for Female Swimming Activity in the Guppy (Poecilia reticulata)
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Organism
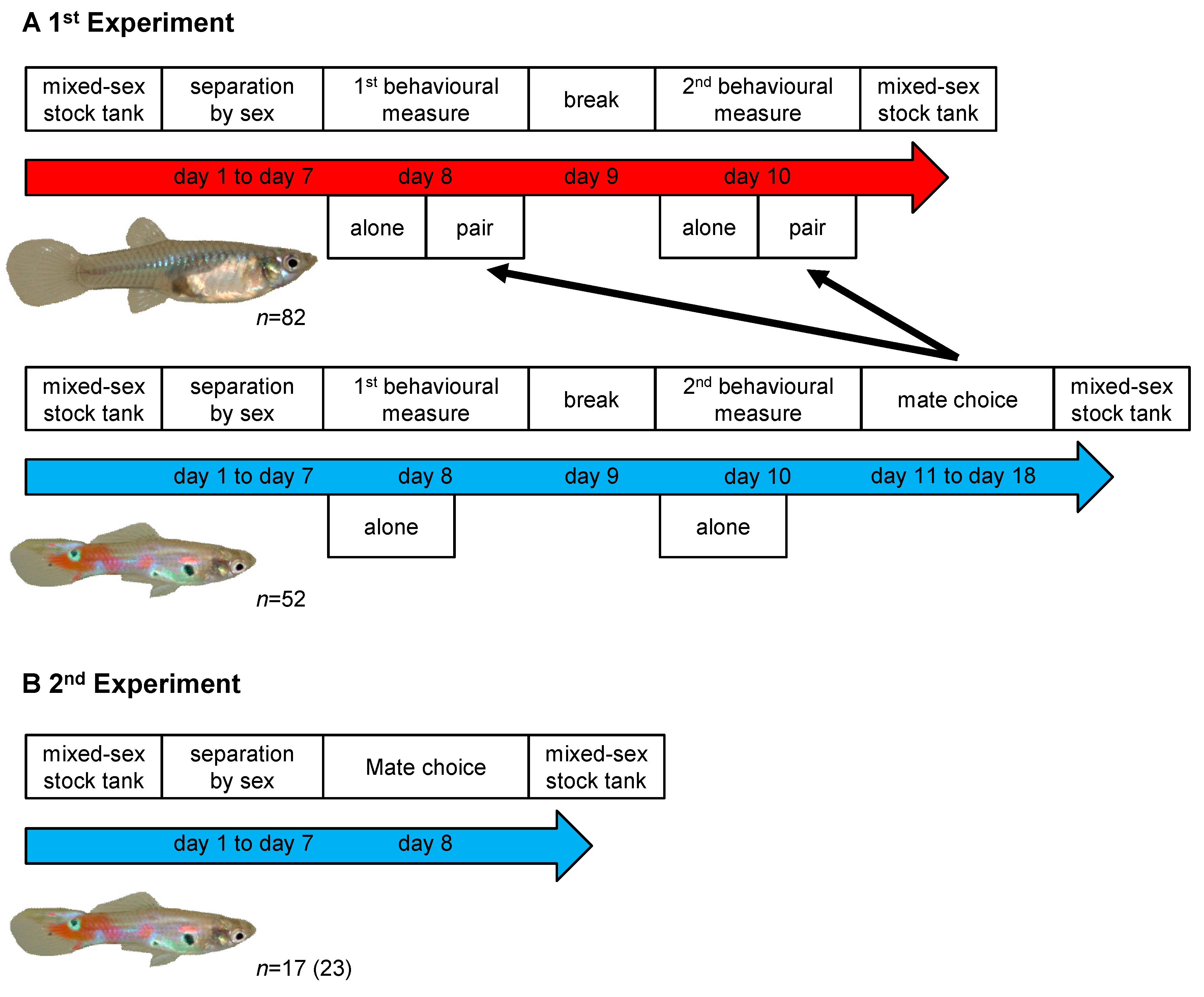
2.2. First Experiment
2.3. Second Experiment
3. Results
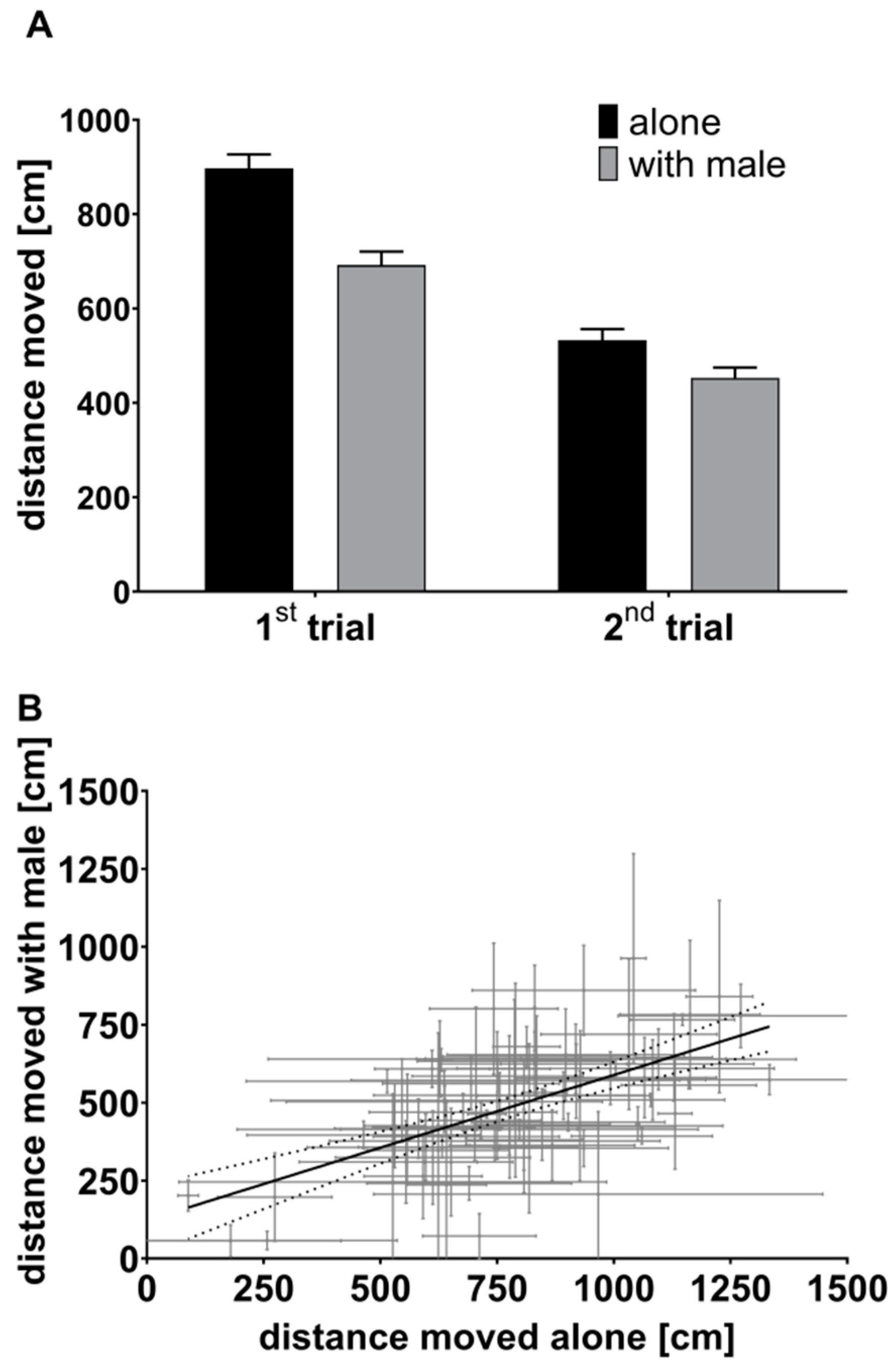
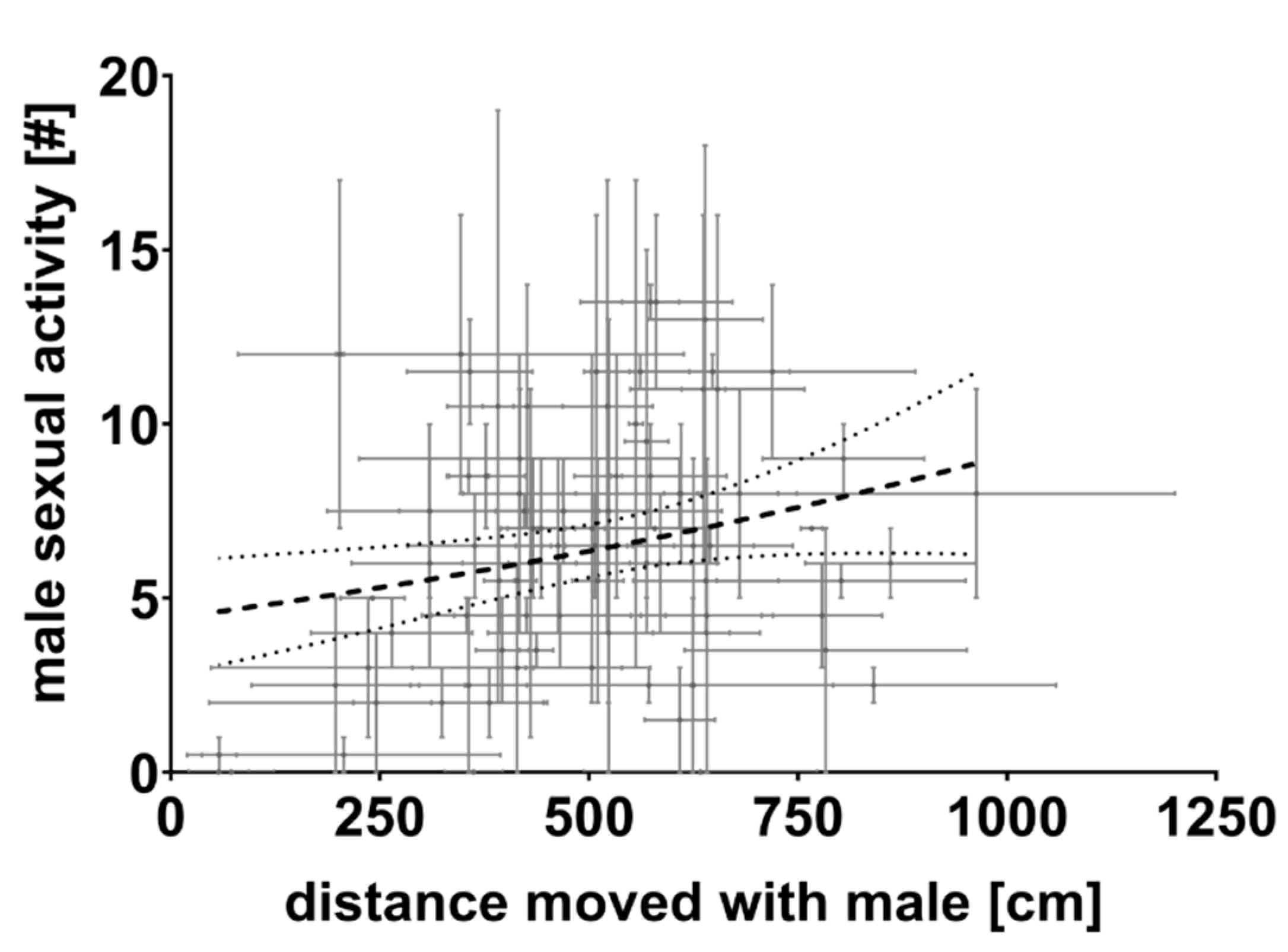
3.1. First Experiment
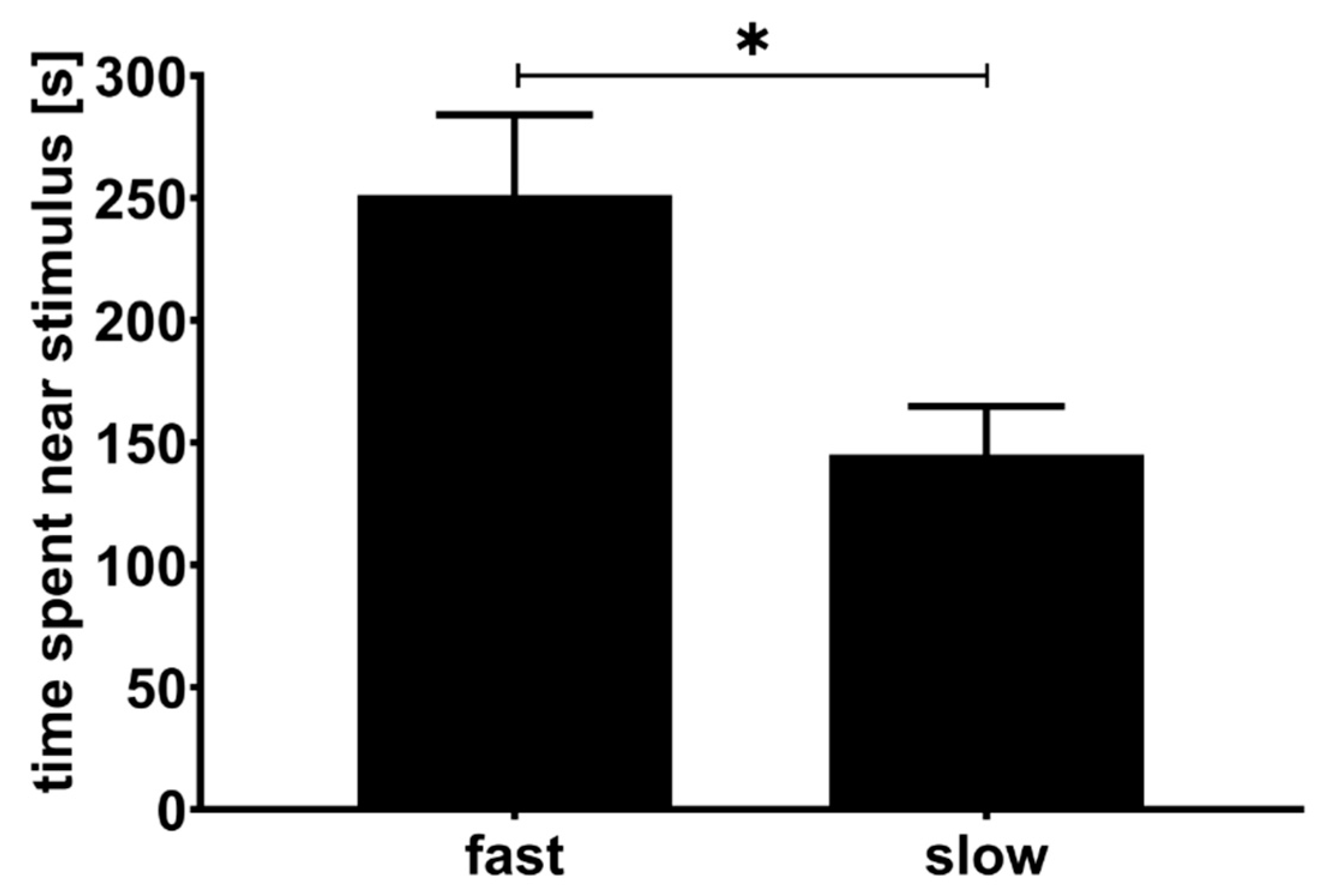
3.2. Second Experiment
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Andersson, M. Sexual Selection; Princeton University Press: Princeton, NJ, USA, 1994. [Google Scholar]
- Edward, D.A.; Chapman, T. The evolution and significance of male mate choice. Trends Ecol. Evol. 2011, 26, 647–654. [Google Scholar] [CrossRef] [PubMed]
- Berglund, A. Many mates make male pipefish choosy. Behaviour 1995, 132, 213–218. [Google Scholar] [CrossRef]
- Scherer, U.; Schuett, W. No male mate choice for female boldness in a bi-parental West African cichlid, the rainbow krib (Pelvicachromis pulcher). PeerJ 2018, 6, e5373. [Google Scholar] [CrossRef] [PubMed]
- Bierbach, D.; Makowicz, A.M.; Schlupp, I.; Geupel, H.; Streit, B.; Plath, M. Casanovas are liars: Behavioral syndromes, sperm competition risk, and the evolution of deceptive male mating behavior in live-bearing fishes [version 3; peer review: 3 approved]. F1000Research 2013, 2, 75. [Google Scholar] [CrossRef] [PubMed]
- Plath, M.; Richter, S.; Tiedemann, R.; Schlupp, I. Male fish deceive competitors about mating preferences. Curr. Biol. 2008, 18, 1138–1141. [Google Scholar] [CrossRef] [PubMed]
- Dosen, L.; Montgomerie, R. Mate preferences by male guppies (Poecilia reticulata) in relation to the risk of sperm competition. Behav. Ecol. Sociobiol. 2004, 55, 266–271. [Google Scholar] [CrossRef]
- Bonduriansky, R. The evolution of male mate choice in insects: A synthesis of ideas and evidence. Biol. Rev. 2001, 76, 305–339. [Google Scholar] [CrossRef]
- Amundsen, T.; Forsgren, E. Male mate choice selects for female coloration in a fish. Proc. Natl. Acad. Sci. USA 2001, 98, 13155–13160. [Google Scholar] [CrossRef]
- Ojanguren, A.F.; Magurran, A.E. Uncoupling the links between male mating tactics and female attractiveness. Proc. R. Soc. Lond. B Biol. Sci. 2004, 271 (Suppl. 6), S427–S429. [Google Scholar] [CrossRef]
- Pampoulie, C.; Lindström, K.; Mary, C.M.S. Have your cake and eat it too: Male sand gobies show more parental care in the presence of female partners. Behav. Ecol. 2004, 15, 199–204. [Google Scholar] [CrossRef]
- Byers, J.; Hebets, E.; Podos, J. Female mate choice based upon male motor performance. Anim. Behav. 2010, 79, 771–778. [Google Scholar] [CrossRef]
- Schuett, W.; Tregenza, T.; Dall, S.R.X. Sexual selection and animal personality. Biol. Rev. 2010, 85, 217–246. [Google Scholar] [CrossRef] [PubMed]
- Snowberg, L.K.; Benkman, C.W. Mate choice based on a key ecological performance trait. J. Evol. Biol. 2009, 22, 762–769. [Google Scholar] [CrossRef] [PubMed]
- Schuett, W.; Godin, J.-G.J.; Dall, S.R.X. Do female zebra finches, Taeniopygia guttata, choose their mates based on their ‘personality’? Ethology 2011, 117, 908–917. [Google Scholar] [CrossRef]
- Teyssier, A.; Bestion, E.; Richard, M.; Cote, J. Partners’ personality types and mate preferences: Predation risk matters. Behav. Ecol. 2014, 25, 723–733. [Google Scholar] [CrossRef]
- Godin, J.G.; Dugatkin, L.A. Female mating preference for bold males in the guppy, Poecilia reticulata. Proc. Natl. Acad. Sci. USA 1996, 93, 10262–10267. [Google Scholar] [CrossRef]
- Smith, B.R.; Blumstein, D.T. Fitness consequences of personality: A meta-analysis. Behav. Ecol. 2008, 19, 448–455. [Google Scholar] [CrossRef]
- Moiron, M.; Laskowski, K.L.; Niemelä, P.T. Individual differences in behaviour explain variation in survival: A meta-analysis. Ecol. Lett. 2020, 23, 399–408. [Google Scholar] [CrossRef]
- Scherer, U.; Godin, J.-G.J.; Schuett, W. Do female rainbow kribs choose males on the basis of their apparent aggression and boldness? A non-correlational mate choice study. Behav. Ecol. Sociobiol. 2020, 74, 1–15. [Google Scholar] [CrossRef]
- Schuett, W.; Dall, S.R.X.; Royle, N.J. Pairs of zebra finches with similar ‘personalities’ make better parents. Anim Behav. 2011, 81, 609–618. [Google Scholar] [CrossRef]
- Ariyomo, T.O.; Watt, P.J. Disassortative mating for boldness decreases reproductive success in the guppy. Behav. Ecol. 2013, 24, 1320–1326. [Google Scholar] [CrossRef]
- Réale, D.; Dingemanse, N.J.; Kazem, A.J.N.; Wright, J. Evolutionary and ecological approaches to the study of personality. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 3937–3946. [Google Scholar] [CrossRef] [PubMed]
- Réale, D.; Reader, S.M.; Sol, D.; McDougall, P.T.; Dingemanse, N.J. Integrating animal temperament within ecology and evolution. Biol. Rev. 2007, 82, 291–318. [Google Scholar] [CrossRef] [PubMed]
- Bell, A.M.; Hankison, S.J.; Laskowski, K.L. The repeatability of behaviour: A meta-analysis. Anim. Behav. 2009, 77, 771–783. [Google Scholar] [CrossRef] [PubMed]
- Careau, V.; Garland, T. Performance, personality, and energetics: Correlation, causation, and mechanism. Physiol. Biochem. Zool. 2012, 85, 543–571. [Google Scholar] [CrossRef]
- Careau, V.; Thomas, D.; Humphries, M.M.; Réale, D. Energy metabolism and animal personality. Oikos 2008, 117, 641–653. [Google Scholar] [CrossRef]
- Polverino, G.; Bierbach, D.; Killen, S.S.; Uusi-Heikkilä, S.; Arlinghaus, R. Body length rather than routine metabolic rate and body condition correlates with activity and risk-taking in juvenile zebrafish Danio rerio. J. Fish Biol. 2016, 89, 2251–2267. [Google Scholar] [CrossRef]
- Stamps, J.A. Growth-mortality tradeoffs and ‘personality traits’ in animals. Ecol. Lett. 2007, 10, 355–363. [Google Scholar] [CrossRef]
- Biro, P.A.; Stamps, J.A. Are animal personality traits linked to life-history productivity? Trends Ecol. Evol. 2008, 23, 361–368. [Google Scholar] [CrossRef]
- Cavigelli, S.A. Animal personality and health. Behaviour 2005, 142, 1223–1244. [Google Scholar] [CrossRef]
- Smith, B.R.; Blumstein, D.T. Behavioral types as predictors of survival in Trinidadian guppies (Poecilia reticulata). Behav. Ecol. 2010, 21, 919–926. [Google Scholar] [CrossRef]
- Plaut, I. Does pregnancy affect swimming performance of female Mosquitofish, Gambusia affinis? Funct. Ecol. 2002, 16, 290–295. [Google Scholar] [CrossRef]
- Ghalambor, C.K.; Reznick, D.N.; Walker, J.A. Constraints on adaptive evolution: The functional trade-off between reproduction and fast-start swimming performance in the Trinidadian guppy (Poecilia reticulata). Am. Nat. 2004, 164, 38–50. [Google Scholar] [CrossRef] [PubMed]
- Krause, J.; Godin, J.-G.J. Predator preferences for attacking particular prey group sizes: Consequences for predator hunting success and prey predation risk. Anim. Behav. 1995, 50, 465–473. [Google Scholar] [CrossRef]
- Ruell, E.W.; Handelsman, C.A.; Hawkins, C.L.; Sofaer, H.R.; Ghalambor, C.K.; Angeloni, L. Fear, food and sexual ornamentation: Plasticity of colour development in Trinidadian guppies. Proc. R. Soc. B Biol. Sci. 2013, 280, 20122019. [Google Scholar] [CrossRef]
- Magurran, A.E. Evolutionary Ecology: The Trinidadian Guppy Oxford Series in Ecology and Evolution; Oxford University Press: Oxford, UK, 2005. [Google Scholar]
- Burns, J.G. The validity of three tests of temperament in guppies Poecilia reticulata). J. Comp. Psychol. 2008, 122, 344–356. [Google Scholar] [CrossRef]
- Harris, S.; Ramnarine, I.W.; Smith, H.G.; Pettersson, L.B. Picking personalities apart: Estimating the influence of predation, sex and body size on boldness in the guppy Poecilia reticulata. Oikos 2010, 119, 1711–1718. [Google Scholar] [CrossRef]
- Lukas, J.; Kalinkat, G.; Miesen, F.W.; Landgraf, T.; Krause, J.; Bierbach, D. Consistent behavioural syndromes across seasons in an invasive freshwater fish. Front. Ecol. Evol. 2020, 8, 583670. [Google Scholar] [CrossRef]
- Head, M.L.; Jacomb, F.; Vega-Trejo, R.; Jennions, M.D. Male mate choice and insemination success under simultaneous versus sequential choice conditions. Anim. Behav. 2015, 103, 99–105. [Google Scholar] [CrossRef]
- Bierbach, D.; Laskowski, K.L.; Wolf, M. Behavioural individuality in clonal fish arises despite near-identical rearing conditions. Nat. Commun. 2017, 8, 15361. [Google Scholar] [CrossRef] [PubMed]
- Reznick, D.; Butler, M.J.; Rodd, F.H.; Ross, P. Life-history evolution in guppies (Poecilia reticulata). Differential mortality as a mechanism for natural selection. Evolution 1996, 50, 1651–1660. [Google Scholar] [PubMed]
- Dingemanse, N.J.; Dochtermann, N.A. Quantifying individual variation in behaviour: Mixed-effect modelling approaches. J. Anim. Ecol. 2013, 82, 39–54. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, S.; Schielzeth, H. Repeatability for Gaussian and non-Gaussian data: A practical guide for biologists. Biol. Rev. 2010, 85, 935–956. [Google Scholar] [CrossRef] [PubMed]
- Herbert-Read, J.E.; Krause, S.; Morrell, L.J.; Schaerf, T.M.; Krause, J.; Ward, A.J.W. The role of individuality in collective group movement. Proc. R. Soc. Lond. B Biol. Sci. 2012, 280, 20122564. [Google Scholar] [CrossRef]
- Bierbach, D.; Sommer-Trembo, C.; Hanisch, J.; Wolf, M.; Plath, M. Personality affects mate choice: Bolder males show stronger audience effects under high competition. Behav. Ecol. 2015, 26, 1314–1325. [Google Scholar] [CrossRef]
- Bierbach, D.; Jung, C.T.; Hornung, S.; Streit, B.; Plath, M. Homosexual behaviour increases male attractiveness to females. Biol. Lett. 2013, 9, 20121038. [Google Scholar] [CrossRef]
- Chouinard-Thuly, L.; Gierszewski, S.; Rosenthal, G.G.; Reader, S.M.; Rieucau, G.; Woo, K.L.; Gerlai, R.; Tedore, C.; Ingley, S.J.; Stowers, J.R.; et al. Technical and conceptual considerations for using animated stimuli in studies of animal behavior. Curr. Zool. 2017, 63, 5–19. [Google Scholar] [CrossRef]
- Gierszewski, S.; Müller, K.; Smielik, I.; Hütwohl, J.-M.; Kuhnert, K.-D.; Witte, K. The virtual lover: Variable and easily guided 3D fish animations as an innovative tool in mate-choice experiments with sailfin mollies-II Validation. Curr. Zool. 2017, 63, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Gierszewski, S.; Baker, D.; Müller, K.; Hütwohl, J.-M.; Kuhnert, K.-D.; Witte, K. Using the fishsim animation toolchain to investigate fish behavior: A case study on mate-choice copying in sailfin mollies. J. Vis. Exp. 2018, e58435. [Google Scholar] [CrossRef]
- Fisher, H.S.; Rosenthal, G.G. Male swordtails court with an audience in mind. Biol. Lett. 2007, 3, 5–7. [Google Scholar] [CrossRef] [PubMed]
- Schlupp, I.; Riesch, R.; Tobler, M.; Plath, M.; Parzefall, J.; Schartl, M. A novel sexually selected trait in poeciliid fishes: Female preference for mustache-like, rostral filaments in male Poecilia sphenops. Behav. Ecol. Sociobiol. 2010, 64, 1849–1855. [Google Scholar] [CrossRef]
- Sommer-Trembo, C.; Plath, M.; Gismann, J.; Helfrich, C.; Bierbach, D. Context-dependent female mate choice maintains variation in male sexual activity. R. Soc. Open Sci. 2017, 4, 170303. [Google Scholar] [CrossRef]
- Walling, C.; Royle, N.J.; Lindström, J.; Metcalfe, N.B.; Lindström, J. Do female association preferences predict the likelihood of reproduction? Behav. Ecol. Sociobiol. 2010, 64, 541–548. [Google Scholar] [CrossRef]
- Brown, C.; Irving, E. Individual personality traits influence group exploration in a feral guppy population. Behav. Ecol. 2014, 25, 95–101. [Google Scholar] [CrossRef]
- Trompf, L.; Brown, C. Personality affects learning and trade-offs between private and social information in guppies, Poecilia reticulata. Anim. Behav. 2014, 88, 99–106. [Google Scholar] [CrossRef]
- Budaev, S.V. “Personality” in the guppy (Poecilia reticulata): A correlational study of exploratory behavior and social tendency. J. Comp. Psychol. 1997, 111, 399–411. [Google Scholar] [CrossRef]
- Jolles, J.W.; Weimar, N.; Landgraf, T.; Romanczuk, P.; Krause, J.; Bierbach, D. Group-level patterns emerge from individual speed as revealed by an extremely social robotic fish. Biol. Lett. 2020, 16, 20200436. [Google Scholar] [CrossRef] [PubMed]
- Bierbach, D.; Landgraf, T.; Romanczuk, P.; Lukas, J.; Nguyen, H.; Wolf, M.; Krause, J. Using a robotic fish to investigate individual differences in social responsiveness in the guppy. R. Soc. Open Sci. 2018, 5, 181026. [Google Scholar] [CrossRef]
- Jolles, J.W.; Boogert, N.J.; Sridhar, V.H.; Couzin, I.D.; Manica, A. Consistent individual differences drive collective behavior and group functioning of schooling fish. Curr. Biol. 2017, 27, 2862–2868.e7. [Google Scholar] [CrossRef] [PubMed]
- Candolin, U. The use of multiple cues in mate choice. Biol. Rev. 2003, 78, 575–595. [Google Scholar] [CrossRef]
- Partan, S.; Marler, P. Issues in the classification of multimodal communication signals. Am. Nat. 2005, 166, 231–245. [Google Scholar] [CrossRef]
- Zahavi, A. Mate selection—A selection for a handicap. J. Theor. Biol. 1975, 53, 205–214. [Google Scholar] [CrossRef]
- Houde, A.E. Sex, Color, and Mate Choice in Guppies; Princeton University Press: Princeton, NJ, USA, 1997. [Google Scholar]
- Liley, N.R. Ethological isolating mechanisms in four sympatric species of Poeciliid fishes. Behav. Suppl. 1966, 13, 1–197. [Google Scholar]
- Schlupp, I.; Ryan, M.J. Male Sailfin mollies (Poecilia latipinna) copy the mate choice of other males. Behav. Ecol. 1997, 8, 104–107. [Google Scholar] [CrossRef]
- Plath, M.; Bierbach, D. Sex and the public: Social eavesdropping, sperm competition risk and male mate choice. Commun. Integr. Biol. 2011, 4, 276–280. [Google Scholar] [CrossRef] [PubMed]
- Magurran, A.E. Sexual Coercion. In Ecology and Evolution of Poeciliid Fishes; Evans, J.P., Pilastro, A., Schlupp, I., Eds.; Chicago University Press: Chicago, IL, USA, 2011; pp. 209–217. [Google Scholar]
- Evans, J.P.; Magurran, A.E. Multiple benefits of multiple mating in guppies. Proc. Natl. Acad. Sci. USA 2000, 97, 10074–10076. [Google Scholar] [CrossRef] [PubMed]
- Killen, S.S.; Croft, D.P.; Salin, K.; Darden, S.K. Male sexually coercive behaviour drives increased swimming efficiency in female guppies. Funct. Ecol. 2015, 30, 576–583. [Google Scholar] [CrossRef] [PubMed]
- Dadda, M. Female social response to male sexual harassment in poeciliid fish: A comparison of six species. Front. Psychol. 2015, 6, 1453. [Google Scholar] [CrossRef] [PubMed]
- Gasparini, C.; Devigili, A.; Pilastro, A. Cross-generational effects of sexual harassment on female fitness in the guppy. Evolution 2011, 66, 532–543. [Google Scholar] [CrossRef]
- Evans, J.P.; Pilastro, A.; Ramnarine, I.W. Sperm transfer through forced matings and its evolutionary implications in natural guppy (Poecilia reticulata) populations. Biol. J. Linn. Soc. 2003, 78, 605–612. [Google Scholar] [CrossRef]
- Laubu, C.; Dechaume-Moncharmont, F.-X.; Motreuil, S.; Schweitzer, C. Mismatched partners that achieve postpairing behavioral similarity improve their reproductive success. Sci. Adv. 2016, 2, e1501013. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, E.L.E.; Souza, C.R.N.; Ward, A.J.W.; Seebacher, F. Exercise changes behaviour. Funct. Ecol. 2014, 28, 652–659. [Google Scholar] [CrossRef]
- Brown, C. Familiarity with the test environment improves escape responses in the crimson spotted rainbowfish, Melanotaenia duboulayi. Anim. Cogn. 2001, 4, 109–113. [Google Scholar] [CrossRef]
- Kniel, N.; Godin, J.J. Does individual personality predict male mating preference for female body size in the Trinidadian guppy? Ethology 2020, 126, 1019–1030. [Google Scholar] [CrossRef]
- Jennions, M.D.; Petrie, M. Variation in mate choice and mating preferences: A review of causes and consequences. Biol. Rev. 1997, 72, 283–327. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bierbach, D.; Wenchel, R.; Gehrig, S.; Wersing, S.; O’Connor, O.L.; Krause, J. Male Sexual Preference for Female Swimming Activity in the Guppy (Poecilia reticulata). Biology 2021, 10, 147. https://doi.org/10.3390/biology10020147
Bierbach D, Wenchel R, Gehrig S, Wersing S, O’Connor OL, Krause J. Male Sexual Preference for Female Swimming Activity in the Guppy (Poecilia reticulata). Biology. 2021; 10(2):147. https://doi.org/10.3390/biology10020147
Chicago/Turabian StyleBierbach, David, Ronja Wenchel, Stefan Gehrig, Serafina Wersing, Olivia L. O’Connor, and Jens Krause. 2021. "Male Sexual Preference for Female Swimming Activity in the Guppy (Poecilia reticulata)" Biology 10, no. 2: 147. https://doi.org/10.3390/biology10020147
APA StyleBierbach, D., Wenchel, R., Gehrig, S., Wersing, S., O’Connor, O. L., & Krause, J. (2021). Male Sexual Preference for Female Swimming Activity in the Guppy (Poecilia reticulata). Biology, 10(2), 147. https://doi.org/10.3390/biology10020147





