Alkaloid Escholidine and Its Interaction with DNA Structures
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Melting Experiments
2.2. Fluorescence Titrations
2.3. NMR Spectroscopy
2.4. Molecular Modeling Studies
3. Results and Discussion
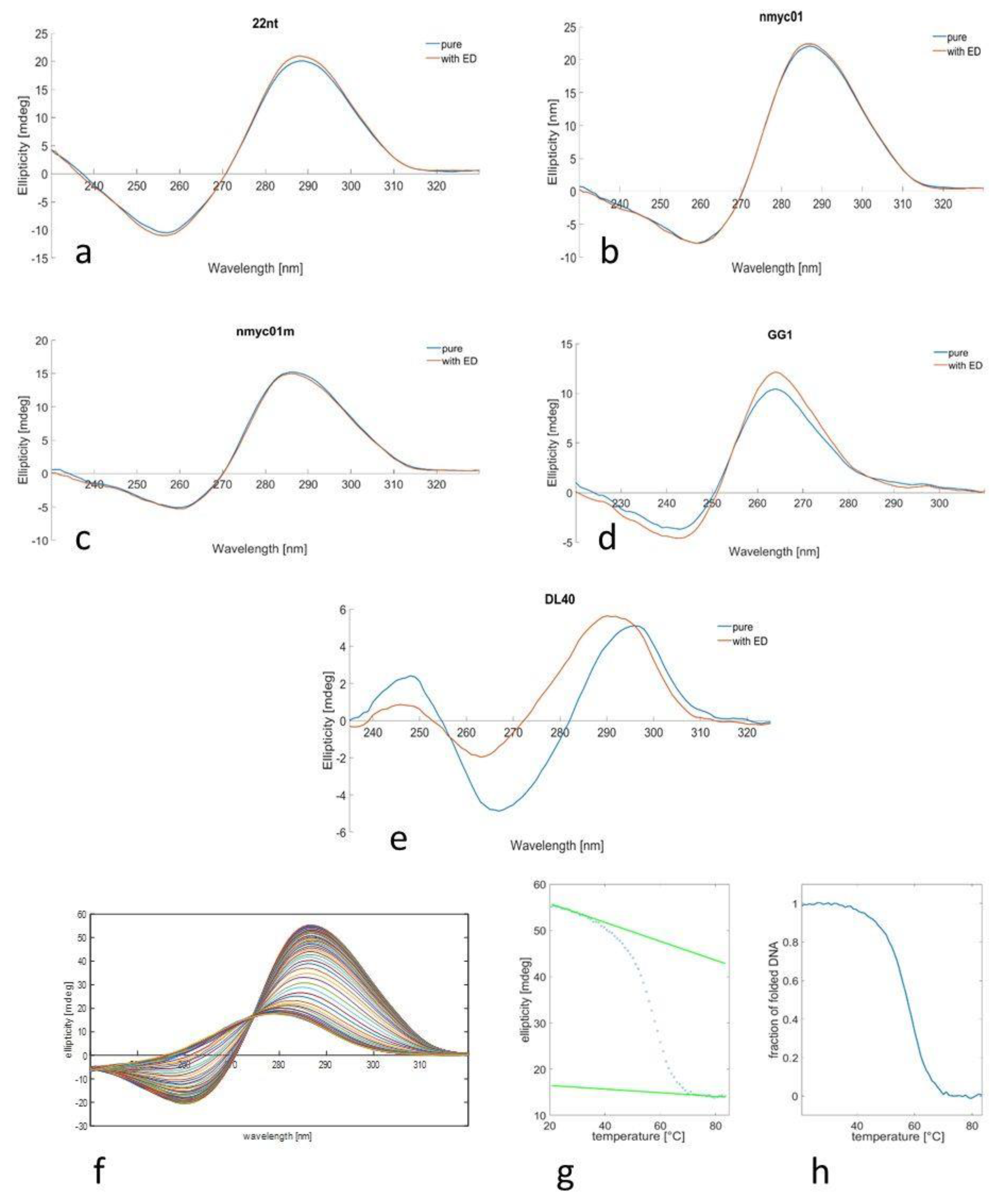
3.1. CD Spectra and Meltings
3.2. Fluorescence Titrations
3.3. NMR Analysis
3.4. Molecular Modeling Studies
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rajecky, M.; Slaninova, I.; Mokrisova, P.; Urbanova, J.; Palkovsky, M.; Taborska, E.; Taborsky, P. Alkaloid chelirubine and DNA: Blue and red luminescence. Talanta 2013, 105, 317–319. [Google Scholar] [CrossRef]
- Rybakova, S.; Rajecky, M.; Urbanova, J.; Pencikova, K.; Taborska, E.; Gargallo, R.; Taborsky, P. Interaction of oligonucleotides with benzo c phenanthridine alkaloid sanguilutine. Chem. Pap. 2013, 67, 568–572. [Google Scholar] [CrossRef]
- Sandor, R.; Slanina, J.; Midlik, A.; Sebrlova, K.; Novotna, L.; Carnecka, M.; Slaninova, I.; Taborsky, P.; Taborska, E.; Pes, O. Sanguinarine is reduced by NADH through a covalent adduct. Phytochemistry 2018, 145, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Rajecky, M.; Sebrlova, K.; Mravec, F.; Taborsky, P. Influence of Solvent Polarity and DNA-Binding on Spectral Properties of Quaternary Benzo c phenanthridine Alkaloids. PLoS ONE 2015, 10, e0129925. [Google Scholar] [CrossRef]
- Leitao da-Cunha, E.V.; Fechine, I.M.; Guedes, D.N.; Barbosa-Filho, J.M.; Sobral da Silva, M. Protoberberine Alkaloids. In The Alkaloids: Chemistry and Biology; Cordell, G.A., Ed.; Academic Press: Cambridge, MA, USA, 2005; Volume 62, pp. 1–75. [Google Scholar]
- Grycová, L.; Dostál, J.; Marek, R. Quarternary Protoberberine Alkaloids; Elsevier: Amsterdam, The Netherlands, 2007; Volume 68, pp. 150–175. [Google Scholar]
- Dostál, J.; Slavík, J. Novější Poznatky o Sanguinarinu a Příbuzných Alkaloidech; Chemické Listy: Praha, Czech Republic, 2000; pp. 15–20. [Google Scholar]
- Majidzadeh, H.; Araj-Khodaei, M.; Ghaffari, M.; Torbati, M.; Ezzati Nazhad Dolatabadi, J.; Hamblin, M.R. Nano-based delivery systems for berberine: A modern anti-cancer herbal medicine. Colloids Surf. B Biointerfaces 2020, 194, 111188. [Google Scholar] [CrossRef]
- Ponnusamy, L.; Kothandan, G.; Manoharan, R. Berberine and Emodin abrogates breast cancer growth and facilitates apoptosis through inactivation of SIK3-induced mTOR and Akt signaling pathway. Biochim. Biophys. Acta (BBA)—Mol. Basis Dis. 2020, 1866, 165897. [Google Scholar] [CrossRef]
- Ma, W.; Zhang, Y.; Yu, M.; Wang, B.; Xu, S.; Zhang, J.; Li, X.; Ye, X. In-vitro and in-vivo anti-breast cancer activity of synergistic effect of berberine and exercise through promoting the apoptosis and immunomodulatory effects. Int. Immunopharmacol. 2020, 87, 106787. [Google Scholar] [CrossRef]
- Gu, S.; Song, X.; Xie, R.; Ouyang, C.; Xie, L.; Li, Q.; Su, T.; Xu, M.; Xu, T.; Huang, D.; et al. Berberine inhibits cancer cells growth by suppressing fatty acid synthesis and biogenesis of extracellular vesicles. Life Sci. 2020, 257, 118122. [Google Scholar] [CrossRef]
- Chudík, S.; Marek, R.; Seckárová, P.; Necas, M.; Dostál, J.; Slavík, J. Revision of the structure of escholidine. J. Nat. Prod. 2006, 69, 954–956. [Google Scholar] [CrossRef] [PubMed]
- Slavík, J.; Dolejš, L.; Sedmera, P. Alkaloids of the Papaveraceae, XLIV. Quaternary alkaloids from roots of three Escholtzia species and from the aerial part of Hunnemannia fumariaefolia SWEET: Constitution of escholidine. Collect. Czechoslov. Chem. Commun. 1970, 35, 2597–2612. [Google Scholar] [CrossRef]
- Schäfer, H.L.; Schäfer, H.; Schneider, W.; Elstner, E.F. Sedative action of extract combinations of Eschscholtzia californica and Corydalis cava. Arzneim.-Forsch. 1995, 45, 124–126. [Google Scholar]
- Rolland, A.; Fleurentin, J.; Lanhers, M.C.; Younos, C.; Misslin, R.; Mortier, F.; Pelt, J.M. Behavioural effects of the American traditional plant Eschscholzia californica: Sedative and anxiolytic properties. Planta Med. 1991, 57, 212–216. [Google Scholar] [CrossRef] [PubMed]
- Jain, L.; Tripathi, M.; Pandey, V.B. Alkaloids of Eschscholtzia californica. Planta Med. 1996, 62, 188. [Google Scholar] [CrossRef]
- Bochman, M.L.; Paeschke, K.; Zakian, V.A. DNA secondary structures: Stability and function of G-quadruplex structures. Nat. Rev. Genet. 2012, 13, 770–780. [Google Scholar] [CrossRef] [Green Version]
- Takahashi, S.; Sugimoto, N. Quantitative Analysis of Stall of Replicating DNA Polymerase by G-Quadruplex Formation. In G-Quadruplex Nucleic Acids: Methods and Protocols; Yang, D., Lin, C., Eds.; Springer: New York, NY, USA, 2019; pp. 257–274. [Google Scholar]
- Abou Assi, H.; Garavís, M.; González, C.; Damha, M.J. i-Motif DNA: Structural features and significance to cell biology. Nucleic Acids Res. 2018, 46, 8038–8056. [Google Scholar] [CrossRef] [Green Version]
- Spiegel, J.; Adhikari, S.; Balasubramanian, S. The Structure and Function of DNA G-Quadruplexes. Trends Chem. 2020, 2, 123–136. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blackburn, G.M. Nucleic Acids in Chemistry and Biology, 3rd ed.; Royal Society of Chemistry: Cambridge, UK, 2006. [Google Scholar]
- Khandelwal, P.; Panchal, S.C.; Radha, P.K.; Hosur, R.V. Solution structure and dynamics of GCN4 cognate DNA: NMR investigations. Nucleic Acids Res. 2001, 29, 499–505. [Google Scholar] [CrossRef] [Green Version]
- Phan, A.T.; Mergny, J.L. Human telomeric DNA: G-quadruplex, i-motif and watson-crick double helix. Nucleic Acids Res. 2002, 30, 4618–4625. [Google Scholar] [CrossRef]
- Neidle, S. 2—The Building-Blocks of DNA and RNA. In Principles of Nucleic Acid Structure; Academic Press: New York, NY, USA, 2008; pp. 20–37. [Google Scholar]
- Burge, S.; Parkinson, G.N.; Hazel, P.; Todd, A.K.; Neidle, S. Quadruplex DNA: Sequence, topology and structure. Nucleic Acids Res. 2006, 34, 5402–5415. [Google Scholar] [CrossRef] [Green Version]
- Vojtylová, T.; Dospivová, D.; Třísková, O.; Pilařová, I.; Lubal, P.; Farková, M.; Trnková, L.; Táborský, P. Spectroscopic study of protonation of oligonucleotides containing adenine and cytosine. Chem. Pap. 2009, 63, 731–737. [Google Scholar] [CrossRef]
- Dzatko, S.; Krafcikova, M.; Hänsel-Hertsch, R.; Fessl, T.; Fiala, R.; Loja, T.; Krafcik, D.; Mergny, J.-L.; Foldynova-Trantirkova, S.; Trantirek, L. Evaluation of the Stability of DNA i-Motifs in the Nuclei of Living Mammalian Cells. Angew. Chem. Int. Ed. 2018, 57, 2165–2169. [Google Scholar] [CrossRef] [Green Version]
- Alba, J.J.; Sadurní, A.; Gargallo, R. Nucleic Acid i-Motif Structures in Analytical Chemistry. Crit. Rev. Anal. Chem. 2016, 46, 443–454. [Google Scholar] [CrossRef] [Green Version]
- Jin, K.S.; Shin, S.R.; Ahn, B.; Rho, Y.; Kim, S.J.; Ree, M. pH-Dependent Structures of an i-Motif DNA in Solution. J. Phys. Chem. B 2009, 113, 1852–1856. [Google Scholar] [CrossRef]
- Wright, E.P.; Huppert, J.L.; Waller, Z.A.E. Identification of multiple genomic DNA sequences which form i-motif structures at neutral pH. Nucleic Acids Res. 2017, 45, 2951–2959. [Google Scholar] [CrossRef] [PubMed]
- Gurung, S.P.; Schwarz, C.; Hall, J.P.; Cardin, C.J.; Brazier, J.A. The importance of loop length on the stability of i-motif structures. Chem. Commun. 2015, 51, 5630–5632. [Google Scholar] [CrossRef] [Green Version]
- Brooks, T.A.; Kendrick, S.; Hurley, L. Making sense of G-quadruplex and i-motif functions in oncogene promoters. FEBS J. 2010, 277, 3459–3469. [Google Scholar] [CrossRef] [Green Version]
- Leroy, J.-L.; Guéron, M.; Mergny, J.-L.; Hélène, C. Intramolecular folding of a fragment of the cytosine-rich strand of telomeric DNA into an i-motif. Nucleic Acids Res. 1994, 22, 1600–1606. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khan, N.; Aviñó, A.; Tauler, R.; González, C.; Eritja, R.; Gargallo, R. Solution equilibria of the i-motif-forming region upstream of the B-cell lymphoma-2 P1 promoter. Biochimie 2007, 89, 1562–1572. [Google Scholar] [CrossRef] [Green Version]
- Simonsson, T.; Pribylova, M.; Vorlickova, M. A Nuclease Hypersensitive Element in the Human c-myc Promoter Adopts Several Distinct i-Tetraplex Structures. Biochem. Biophys. Res. Commun. 2000, 278, 158–166. [Google Scholar] [CrossRef] [PubMed]
- Brazier, J.A.; Shah, A.; Brown, G.D. I-Motif formation in gene promoters: Unusually stable formation in sequences complementary to known G-quadruplexes. Chem. Commun. 2012, 48, 10739–10741. [Google Scholar] [CrossRef]
- Zeraati, M.; Langley, D.B.; Schofield, P.; Moye, A.L.; Rouet, R.; Hughes, W.E.; Bryan, T.M.; Dinger, M.E.; Christ, D. I-motif DNA structures are formed in the nuclei of human cells. Nat. Chem. 2018, 10, 631–637. [Google Scholar] [CrossRef]
- Feng, L.; Dong, Z.; Tao, D.; Zhang, Y.; Liu, Z. The acidic tumor microenvironment: A target for smart cancer nano-theranostics. Nat. Sci. Rev. 2018, 5, 269–286. [Google Scholar] [CrossRef] [Green Version]
- Jarosova, P.; Sandor, R.; Slaninkova, A.; Vido, M.; Pes, O.; Taborsky, P. Quaternary protoberberine alkaloids and their interactions with DNA. Chem. Pap. 2019, 73, 2965–2973. [Google Scholar] [CrossRef]
- Jarosova, P.; Paroulek, P.; Rajecky, M.; Rajecka, V.; Taborska, E.; Eritja, R.; Aviñó, A.; Mazzini, S.; Gargallo, R.; Taborsky, P. Naturally occurring quaternary benzo[c]phenanthridine alkaloids selectively stabilize G-quadruplexes. Pchys. Chem. Chem. Phys. 2018, 20, 21772–21782. [Google Scholar] [CrossRef] [Green Version]
- Adamcik, J.; Valle, F.; Witz, G.; Rechendorff, K.; Dietler, G. The promotion of secondary structures in single-stranded DNA by drugs that bind to duplex DNA: An atomic force microscopy study. Nanotechnology 2008, 19, 384016. [Google Scholar] [CrossRef] [PubMed]
- Wright, E.P.; Day, H.A.; Ibrahim, A.M.; Kumar, J.; Boswell, L.J.E.; Huguin, C.; Stevenson, C.E.M.; Pors, K.; Waller, Z.A.E. Mitoxantrone and Analogues Bind and Stabilize i-Motif Forming DNA Sequences. Sci. Rep. 2016, 6, 39456. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mazzini, S.; Bellucci, M.C.; Mondelli, R. Mode of binding of the cytotoxic alkaloid berberine with the double helix oligonucleotide d(AAGAATTCTT)2. Bioorganic Med. Chem. 2003, 11, 505–514. [Google Scholar] [CrossRef]
- Gargallo, R.; Aviñó, A.; Eritja, R.; Jarosova, P.; Mazzini, S.; Scaglioni, L.; Taborsky, P. Study of alkaloid berberine and its interaction with the human telomeric i-motif DNA structure. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2021, 248, 119185. [Google Scholar] [CrossRef]
- Fernando, H.; Reszka, A.P.; Huppert, J.; Ladame, S.; Rankin, S.; Venkitaraman, A.R.; Neidle, S.; Balasubramanian, S. A Conserved Quadruplex Motif Located in a Transcription Activation Site of the Human c-kit Oncogene. Biochemistry 2006, 45, 7854–7860. [Google Scholar] [CrossRef] [Green Version]
- Kuryavyi, V.; Majumdar, A.; Shallop, A.; Chernichenko, N.; Skripkin, E.; Jones, R.; Patel, D.J. A double chain reversal loop and two diagonal loops define the architecture of unimolecular DNA quadruplex containing a pair of stacked G(syn)·G(syn)·G(anti)·G(anti) tetrads flanked by a G·(T-T) triad and a T·T·T triple1. J. Mol. Biol. 2001, 310, 181–194. [Google Scholar] [CrossRef]
- Benabou, S.; Ferreira, R.; Aviñó, A.; González, C.; Lyonnais, S.; Solà, M.; Eritja, R.; Jaumot, J.; Gargallo, R. Solution equilibria of cytosine- and guanine-rich sequences near the promoter region of the n-myc gene that contain stable hairpins within lateral loops. Biochim. Biophys. Acta (BBA)—Gen. Subj. 2014, 1840, 41–52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bell, E.; Chen, L.; Liu, T.; Marshall, G.M.; Lunec, J.; Tweddle, D.A. MYCN oncoprotein targets and their therapeutic potential. Cancer Lett. 2010, 293, 144–157. [Google Scholar] [CrossRef]
- Mergny, J.-L.; Riou, J.-F.; Mailliet, P.; Teulade-Fichou, M.-P.; Gilson, E. Natural and pharmacological regulation of telomerase. Nucleic Acids Res. 2002, 30, 839–865. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Datta, B.; Armitage, B.A. Hybridization of PNA to Structured DNA Targets: Quadruplex Invasion and the Overhang Effect. J. Am. Chem. Soc. 2001, 123, 9612–9619. [Google Scholar] [CrossRef]
- Puglisi, J.D.; Tinoco, I. Absorbency melting curves of RNA. Methods Enzymol. 1989, 180, 304–325. [Google Scholar]
- Breslauer, K.J. Extracting thermodynamic data from equilibrium melting curves for oligonucleotide order-disorder transitions. Energetics Biol. Macromol. 1995, 259, 221–242. [Google Scholar]
- Dyson, R.M.; Kaderli, S.; Lawrance, G.A.; Maeder, M. Second order global analysis: The evaluation of series of spectrophotometric titrations for improved determination of equilibrium constants. Anal. Chim. Acta 1997, 353, 381–393. [Google Scholar] [CrossRef]
- Kuryavyi, V.; Phan, A.T.; Patel, D.J. Solution structures of all parallel-stranded monomeric and dimeric G-quadruplex scaffolds of the human c-kit2 promoter. Nucleic Acids Res. 2010, 38, 6757–6773. [Google Scholar] [CrossRef] [Green Version]
- Moraca, F.; Amato, J.; Ortuso, F.; Artese, A.; Pagano, B.; Novellino, E.; Alcaro, S.; Parrinello, M.; Limongelli, V. Ligand binding to telomeric G-quadruplex DNA investigated by funnel-metadynamics simulations. Proc. Natl. Acad. Sci. USA 2017, 114, E2136–E2145. [Google Scholar] [CrossRef] [Green Version]
- Morris, G.M.; Goodsell, D.S.; Halliday, R.S.; Huey, R.; Hart, W.E.; Belew, R.K.; Olson, A.J. Automated docking using a Lamarckian genetic algorithm and an empirical binding free energy function. J. Comput. Chem. 1998, 19, 1639–1662. [Google Scholar] [CrossRef] [Green Version]
- Morris, G.M.; Huey, R.; Lindstrom, W.; Sanner, M.F.; Belew, R.K.; Goodsell, D.S.; Olson, A.J. AutoDock4 and AutoDockTools4: Automated Docking with Selective Receptor Flexibility. J. Comput. Chem. 2009, 30, 2785–2791. [Google Scholar] [CrossRef] [Green Version]
- Gasteiger, J.; Marsili, M. Iterative Partial Equalization of Orbital Electronegativity—A Rapid Access to Atomic Charges. Tetrahedron 1980, 36, 3219–3228. [Google Scholar] [CrossRef]
- Sanner, M.F. Python: A programming language for software integration and development. J. Mol. Graph. Model. 1999, 17, 57–61. [Google Scholar]
- Goddard, T.D.; Huang, C.C.; Meng, E.C.; Pettersen, E.F.; Couch, G.S.; Morris, J.H.; Ferrin, T.E. UCSF ChimeraX: Meeting modern challenges in visualization and analysis. Protein Sci. 2018, 27, 14–25. [Google Scholar] [CrossRef] [PubMed]
- Ambrus, A.; Chen, D.; Dai, J.; Bialis, T.; Jones, R.A.; Yang, D. Human telomeric sequence forms a hybrid-type intramolecular G-quadruplex structure with mixed parallel/antiparallel strands in potassium solution. Nucleic Acids Res. 2006, 34, 2723–2735. [Google Scholar] [CrossRef] [Green Version]
- Benabou, S.; Aviñó, A.; Lyonnais, S.; González, C.; Eritja, R.; De Juan, A.; Gargallo, R. i-motif structures in long cytosine-rich sequences found upstream of the promoter region of the SMARCA4 gene. Biochimie 2017, 140, 20–33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, S.; Chen, J.; Cheng, T.J.; Gindulyte, A.; He, J.; He, S.Q.; Li, Q.L.; Shoemaker, B.A.; Thiessen, P.A.; Yu, B.; et al. PubChem in 2021: New data content and improved web interfaces. Nucleic Acids Res. 2021, 49, D1388–D1395. [Google Scholar] [CrossRef] [PubMed]
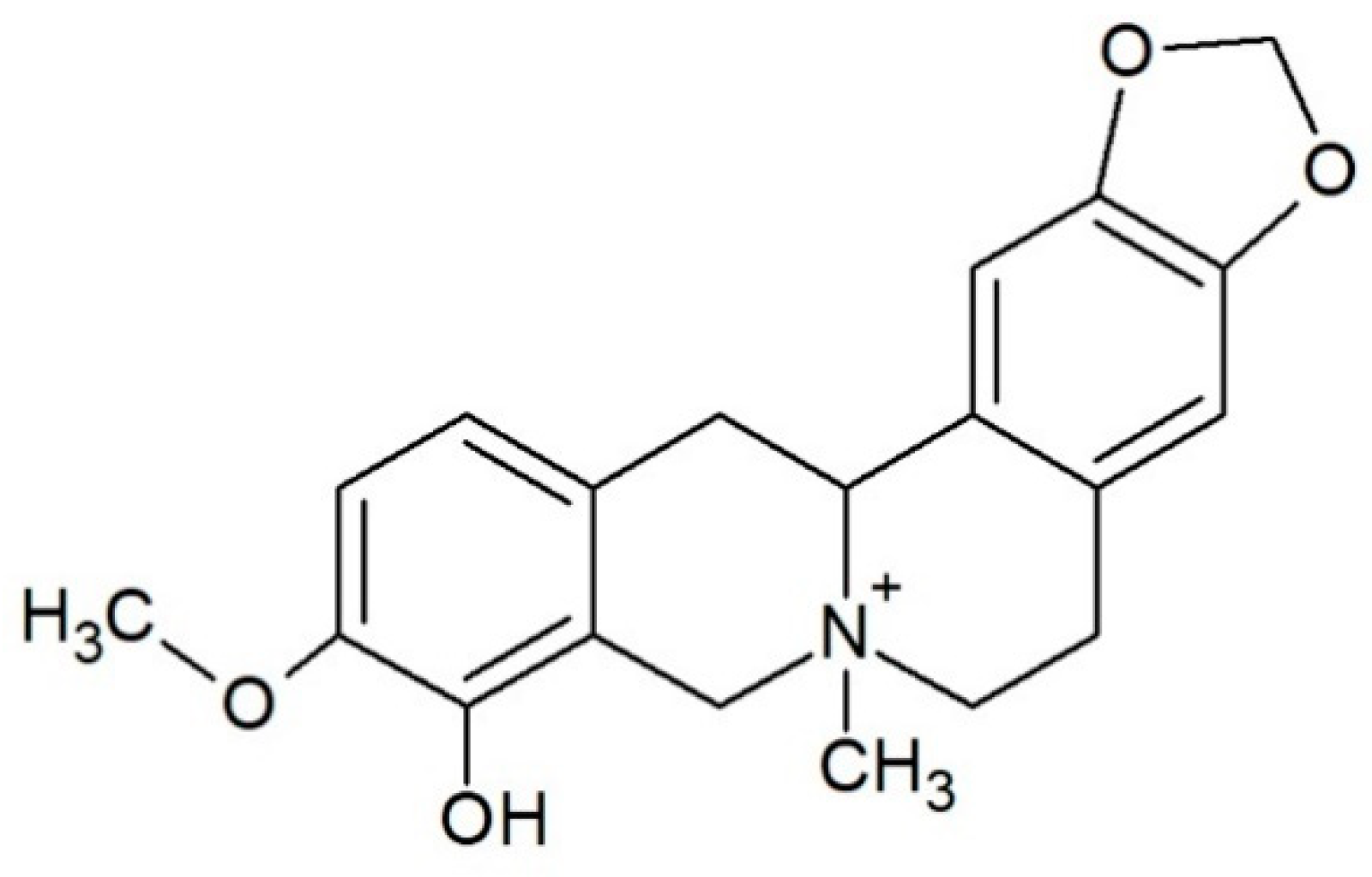
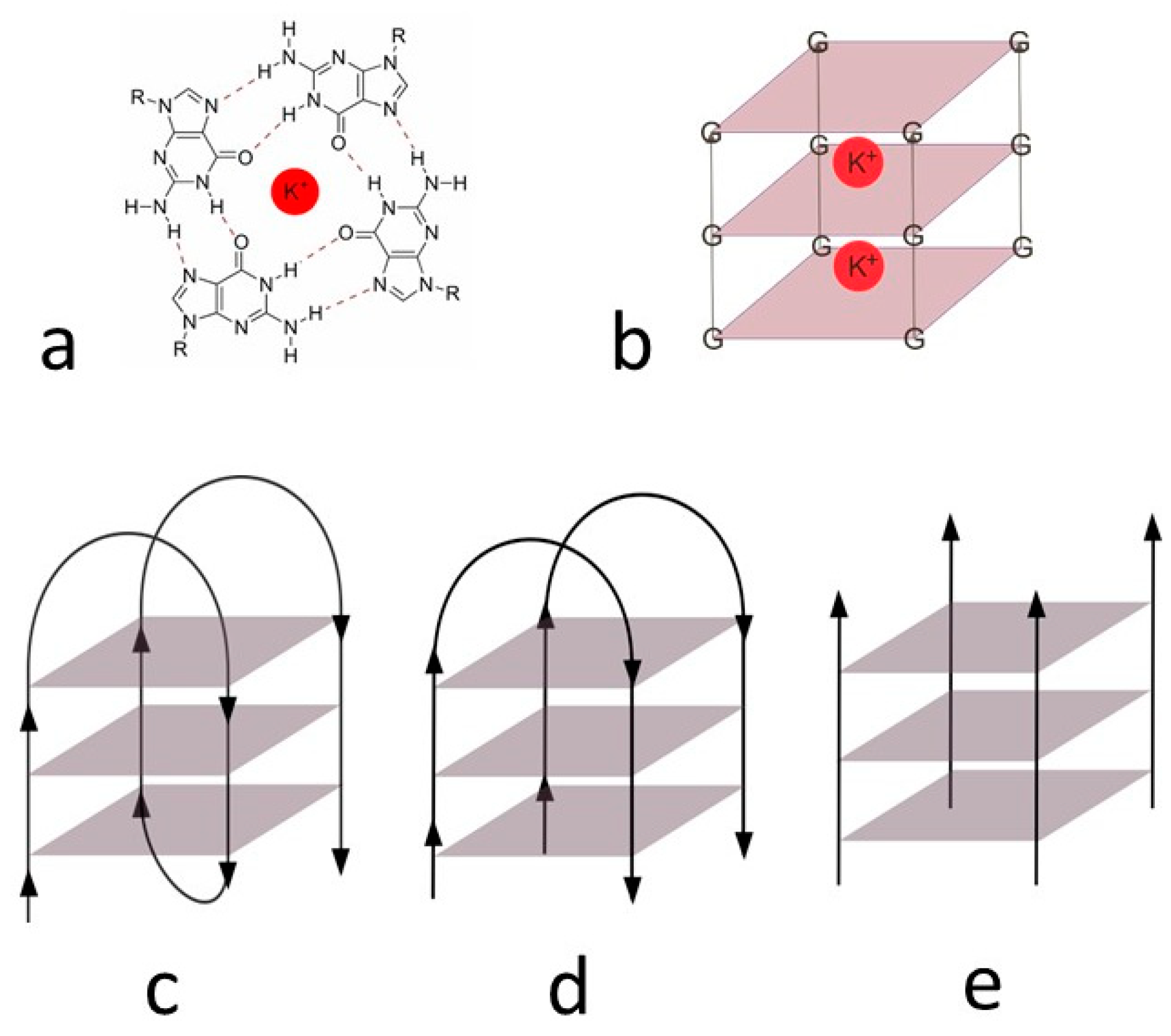
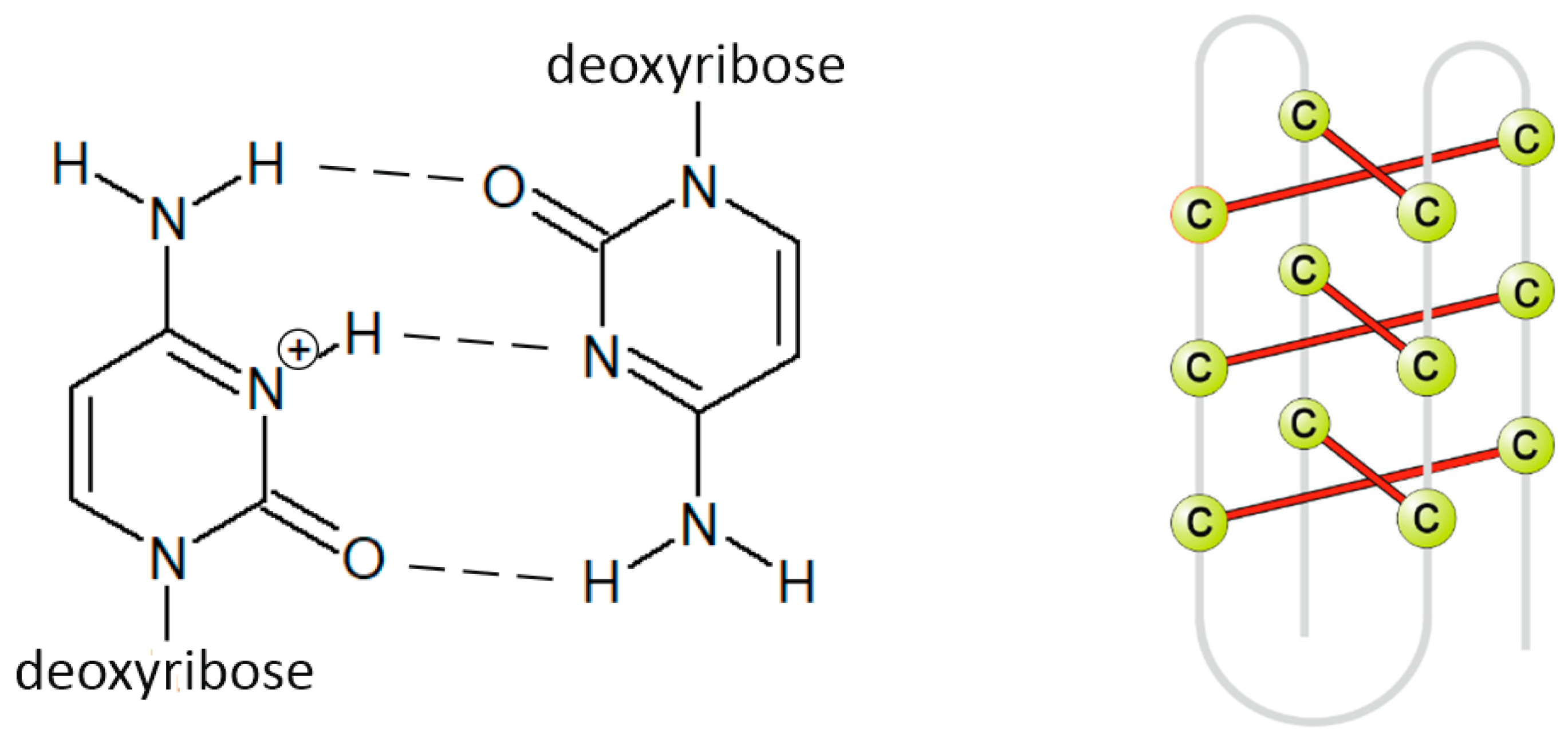
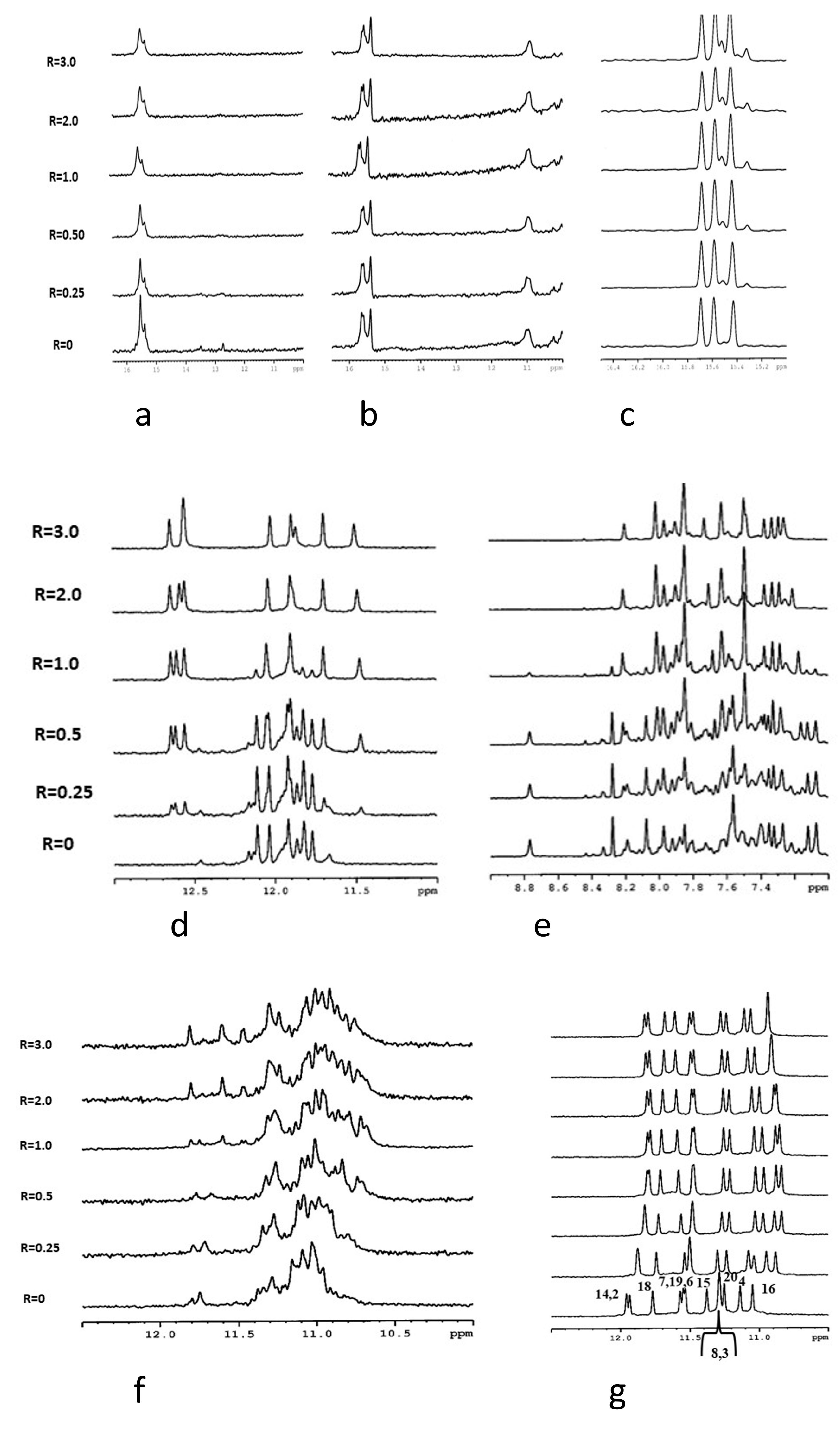
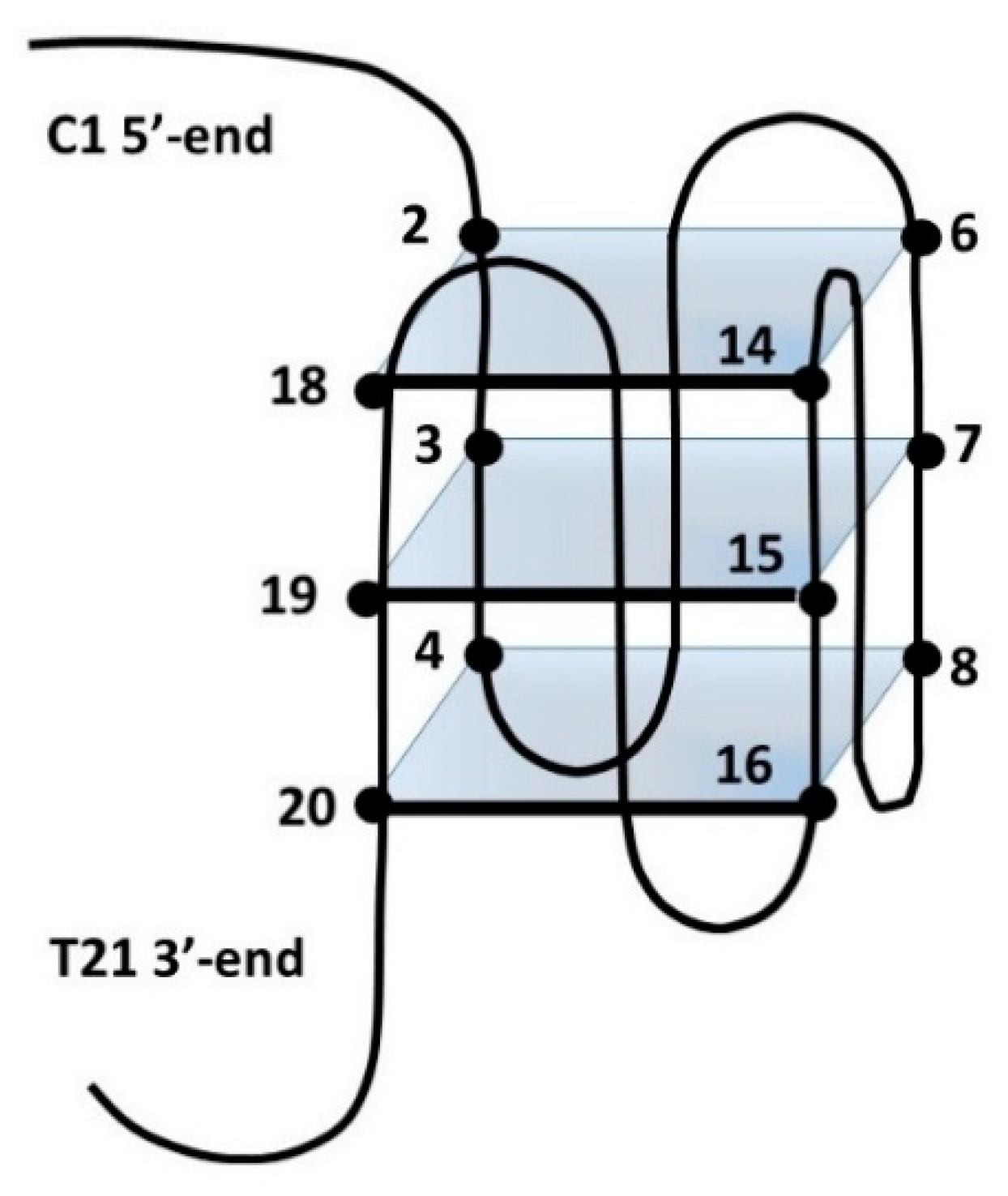
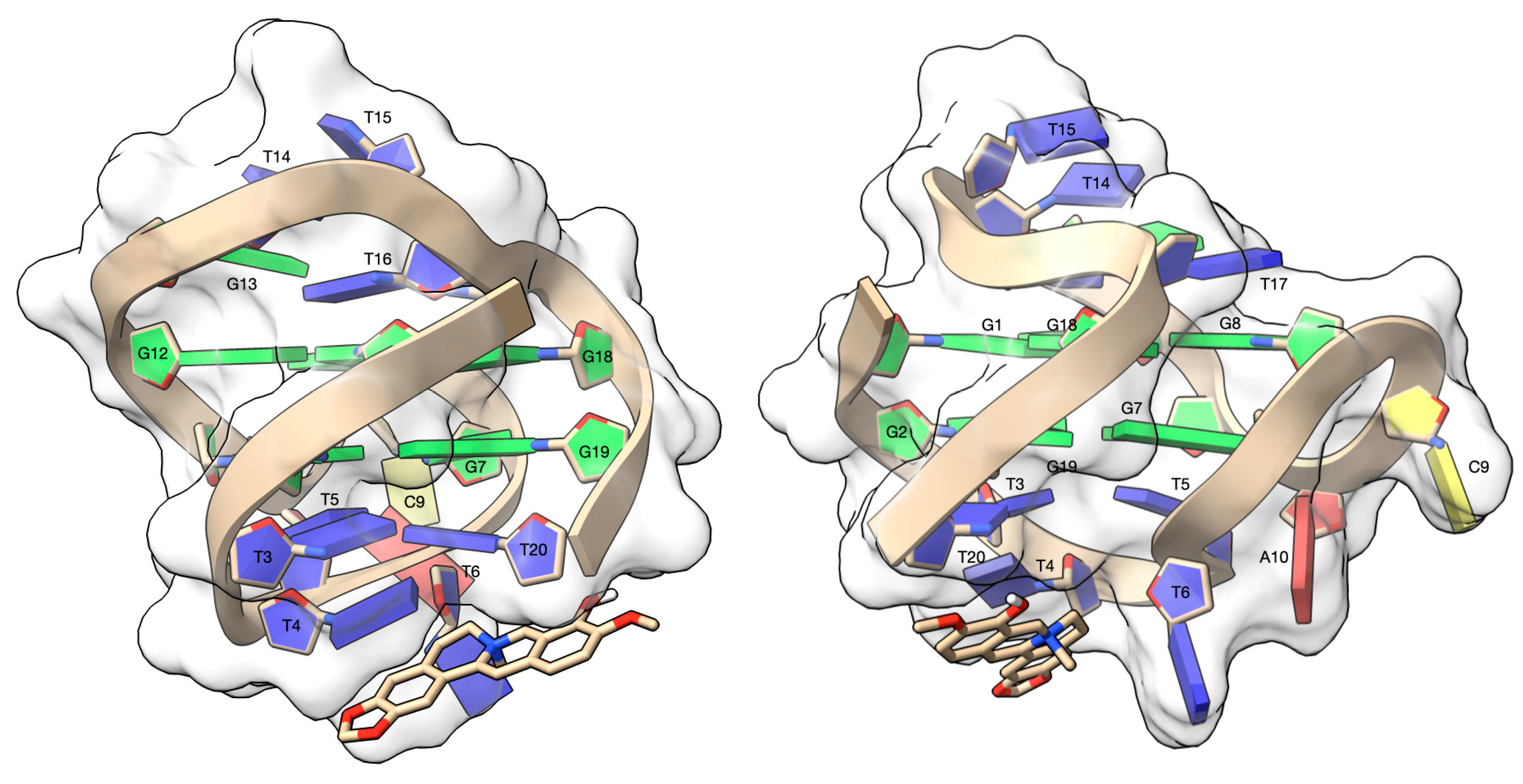
| Code | Sequence (5′–3′) | Type |
|---|---|---|
| GG1 | GGGCGGGCGCGAGGGAGGGT | Parallel G-quadruplex [45] (pH 7) |
| DL40 | GGTTTTGGCAGGGTTTTGGT | Antiparallel G-quadruplex [46] (pH 7) |
| nmyc01 | ACCCCCTGCATCTGCATGCCCCCTCCCACCCCCT | i-motif + duplex hairpin [47] (pH 5) |
| nmyc01m | ACCCCCTGCATCTTTTTGCCCCCTCCCACCCCCT | I-motif [47] (pH 5) |
| 22nt | CCCTAACCCTAACCCTAACCCT | I-motif [23] (pH 5) |
| Oligonucleotide | Tm [°C] | ΔTm [°C] | |
|---|---|---|---|
| Pure | with ED | ||
| GG1 | 48.1 ± 0.4 | 49.8 ± 1.4 | 1.7 |
| DL40 | 28.8 ± 0.9 | 34.0 ± 0.8 | 5.2 |
| nmyc01 | 58.1 ± 0.4 | 57.8 ± 1.6 | −0.3 |
| nmyc01m | 55.8 ± 0.9 | 55.2 ± 1.1 | −0.6 |
| 22nt | 51.5 ± 0.1 | 52.2 ± 0.5 | 0.7 |
| Oligonucleotide | Pure | with ED | ||||
|---|---|---|---|---|---|---|
| ΔG (37) | ΔH [kcal/mol] | ΔS [cal/K·mol] | ΔG (37) | ΔH [kcal/mol] | ΔS [cal/K·mol] | |
| GG1 | −1.4 | −40.8 | −127.1 | −1.7 | −42.5 | −131.5 |
| DL40 | 1.7 | −62.3 | −206.5 | 0.6 | −56.2 | −183.0 |
| nmyc01 | −5.4 | −85.0 | −256.7 | −4.4 | −70.1 | −211.8 |
| nmyc01m | −4.4 | −77.9 | −236.8 | −3.8 | −67.3 | −205.0 |
| 22nt | −3.2 | −71.4 | −219.9 | −3.7 | −78.9 | −242.4 |
| Oligonucleotide | Stoichiometry | Log Ka |
|---|---|---|
| GG1 | 1:5 | 22.39 ± 0.03 |
| DL40 | 1:4 | 18.46 ± 0.05 |
| nmyc01 | 1:4 | 18.21 ± 0.17 |
| nmyc01m | 1:5 | 22.61 ± 0.08 |
| 22nt | 1:4 | 17.64 ± 0.12 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jarošová, P.; Hannig, P.; Kolková, K.; Mazzini, S.; Táborská, E.; Gargallo, R.; Borgonovo, G.; Artali, R.; Táborský, P. Alkaloid Escholidine and Its Interaction with DNA Structures. Biology 2021, 10, 1225. https://doi.org/10.3390/biology10121225
Jarošová P, Hannig P, Kolková K, Mazzini S, Táborská E, Gargallo R, Borgonovo G, Artali R, Táborský P. Alkaloid Escholidine and Its Interaction with DNA Structures. Biology. 2021; 10(12):1225. https://doi.org/10.3390/biology10121225
Chicago/Turabian StyleJarošová, Petra, Pavel Hannig, Kateřina Kolková, Stefania Mazzini, Eva Táborská, Raimundo Gargallo, Gigliola Borgonovo, Roberto Artali, and Petr Táborský. 2021. "Alkaloid Escholidine and Its Interaction with DNA Structures" Biology 10, no. 12: 1225. https://doi.org/10.3390/biology10121225
APA StyleJarošová, P., Hannig, P., Kolková, K., Mazzini, S., Táborská, E., Gargallo, R., Borgonovo, G., Artali, R., & Táborský, P. (2021). Alkaloid Escholidine and Its Interaction with DNA Structures. Biology, 10(12), 1225. https://doi.org/10.3390/biology10121225








