1. Introduction
Sensory information processing is not fully blocked during sleep since animals need to identify relevant stimuli for their survival. However, when compared to wakefulness, focused attention to the environment is lost while we sleep. Such reduced sensitivity in the detection of external signals leads to a decrease in sensory awareness. Still, sleep is a vital recurrent state that reduces brain energy demands by slowing down the metabolic rate [
1]. Behaviorally, this is clearly observed in the cessation of actions since sleep holds back reproduction, exploration, protection and nurture of the offspring. Electrophysiologically, the deep phases of sleep (NREM sleep) are distinguished from wakefulness based on the electrical activity emerging from neuronal populations. For instance, extracellular cortical recordings obtained during wakefulness show low-amplitude–high-frequency voltage fluctuations whereas during NREM sleep, and also during deep anesthesia [
2,
3,
4], these fluctuations exhibit high-amplitude–low-frequency components and are bimodal [
3,
4,
5,
6,
7,
8]. The bimodality appears due to the alternation between periods of persistent firing activity (Up states) and silent periods (Down states), which provides the idiosyncratic dynamical features of NREM sleep, also known as slow wave activity (SWA). This leads to a reduction of the overall firing rate of both excitatory and inhibitory neuronal cell types in the time course of NREM sleep [
4,
5,
6,
7,
8].
Despite these differences, neurons in the primary auditory cortex show comparable responses during sleep and wakefulness when pure tones are delivered to rats [
7] and primates [
9]. These experimental results challenge the hypothesis that during sleep cortical responses are weakened through thalamic gating, i.e., the mechanism by which thalamic spindles could prevent a reliable transmission of external signals to sensory cortices during sleep [
10]. Interestingly, in higher-order cortical areas during sleep—in the perirhinal cortex in rats [
8] and the human cingulate and prefrontal cortex [
11] and during anesthesia—in the association cortex in humans [
12], auditory responses are attenuated. These studies imply that information gating must occur within brain structures higher up the stream of sensory processing and seem to be more consistent with a breakdown of functional cortical connectivity rather than of thalamocortical connectivity. Indeed, fMRI data collected in humans indicate a reduced global effective connectivity during NREM sleep [
13]. Even in the presence of cycling alternating patterns (CAP), a sign of sleep instability, EEG-based functional connectivity shows a small-world network type of structure [
14]. Moreover, cortical responses to transcranial magnetic stimulation (TMS) have been shown to be local during NREM sleep and do not propagate to connected brain regions [
15,
16].
One possible neural correlate of decreased sensory responsiveness in the non-primary cortex during NREM sleep could be the downscaling of the conductance of excitatory synapses, a hypothesis known as synaptic homeostasis hypothesis (SHY) [
17,
18,
19,
20,
21]. Experimental evidence [
22,
23,
24,
25] shows that potentiation of synapses during wakefulness caused by synaptic plasticity is compensated during NREM sleep with a general reduction of synaptic strength. We implement this synaptic conductance downscaling in our computational study by tuning a single parameter that is biophysically equivalent to an excitatory synaptic conductance. Moreover, decreasing its value shifts the model from wakefulness to NREM sleep. To our knowledge, this is the first mathematical model, among all the different proposals [
26,
27,
28,
29,
30], that reconciles these two important findings of sleep research: synaptic downscaling and the simultaneous emergence of slow rhythms as the brain enters the deep phases of sleep from wakefulness.
We aim at understanding, within the neural mass model formalism used in this study and others [
27,
31], whether SHY not only explains the aforementioned change in brain dynamics, but also in the propagation span of evoked activities occurring throughout the sleep–waking cycle (SWC). To do so, we have simulated the local field potential (LFP) signals of two cortical columns operating in either NREM sleep or wakefulness. One cortical column—the perturbed one—responds to an externally applied excitatory input, reproducing the activation of a primary auditory area by a sound or a superficial region of the cerebral cortex stimulated by TMS. A second cortical column—the unperturbed one—is bidirectionally coupled to the perturbed column and does not directly receive the external input, as occurs with non-primary auditory areas and neighboring regions. We quantify the propagation of this external input as a function of the strength of the inter-column connectivity in each brain state. We show that the upscaling of the conductance of excitatory synapses shifts the LFP dynamics from NREM sleep to wakefulness in both columns, as they are intrinsically identical. However, a general build-up of this conductance does not lead to the propagation of the external input from the perturbed cortical column to the unperturbed cortical column. Interestingly, we have found that the unperturbed cortical column responds only significantly when the increase in the excitatory conductance selectively takes place in the inter-column synapses with respect to the intra-column connectivity. This finding points to a novel and different interpretation of SHY, namely that the tuning of synaptic conductances could be distance-dependent.
Our results are presented as follows. First, we introduce the model for a single cortical column in order to validate the parameter space where the system exhibits NREM and wakefulness dynamics. Second, we extend the network to two cortical columns coupled bidirectionally with excitatory synapses to generalize the results to a more complex connectivity diagram. Note that now the change in synaptic conductance that allows the system to shift dynamics, also affects the inter-column connectivity and thus directly influences signal transmission between columns. Finally, we show how efficient input propagation observed during wakefulness, and reduced during NREM sleep, can be obtained.
4. Discussion
There is compelling experimental evidence that auditory stimuli elicit firing responses in the primary auditory cortex that are similar in NREM sleep and wakefulness [
7,
9]. This poses a challenge to the understanding of auditory perception across the wake–sleep cycle because sounds seem to bypass the thalamus in both brain states. However, the observation that neuronal responses to either auditory stimuli [
7,
8,
9,
11,
12] and non-sensory stimuli [
15,
16] are elicited in areas located at distances from the stimulation site that are larger in wakefulness than in NREM sleep opens the door to a new hypothesis. In particular, researchers have suggested that it is the propagation of sounds throughout the cortex that is compromised during sleep, given that a neuronal response is detected in primary brain areas at the initial stages of auditory processing. This hypothesis is supported by the fact that effective connectivity during sleep is reduced with respect to wakefulness, a feature that has also been explored in large-scale models [
13,
39].
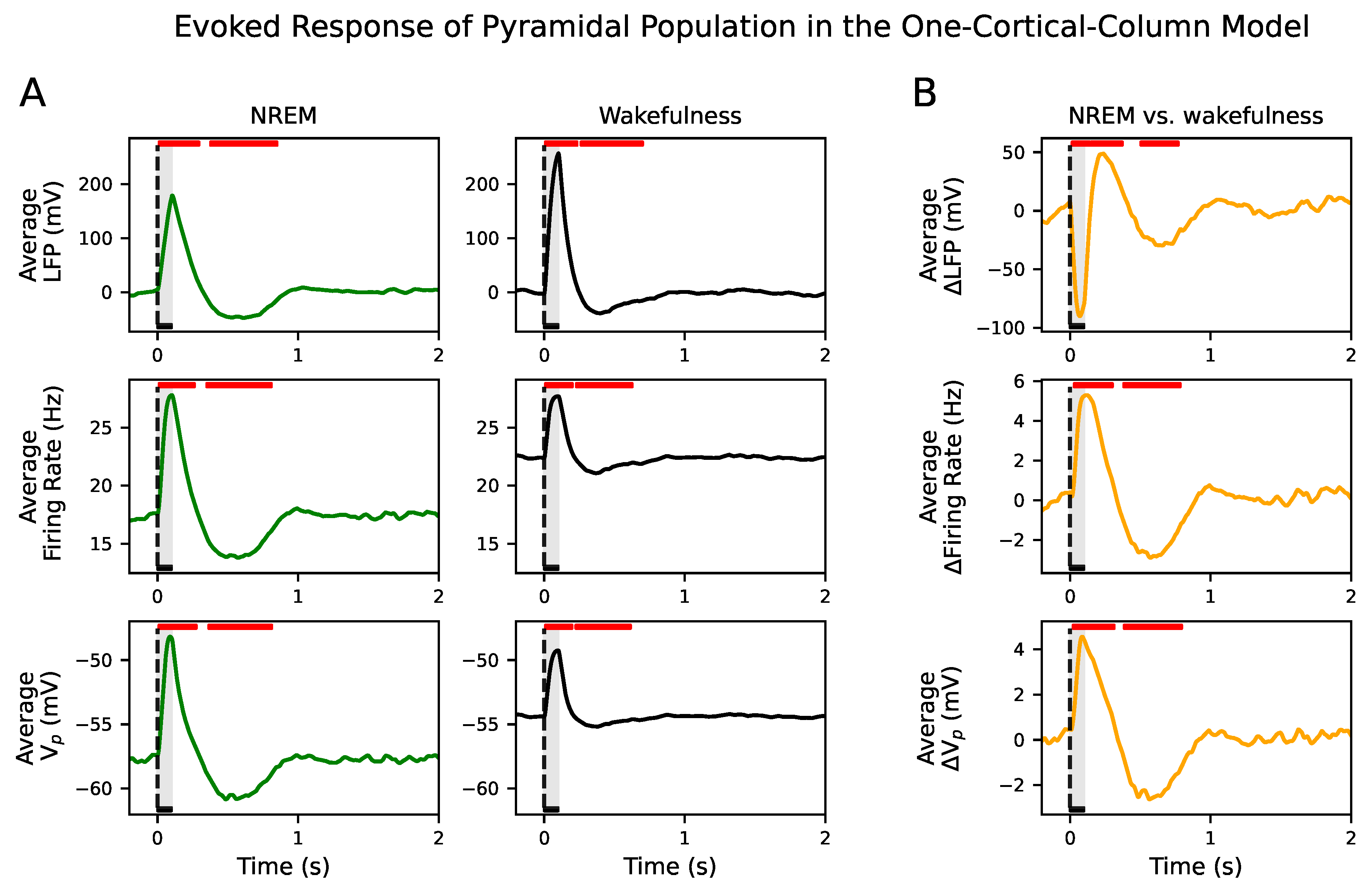
Here, we have used a computational approach to understand how input propagation is disrupted, beyond primary sensory encoding, by tuning the conductance of synaptic connections. By means of a neuronal mass model operating in two distinct brain dynamics, NREM sleep and wakefulness, we investigate the response of a neural system composed of two cortical columns to an external perturbation. Our simulated signals—LFP, firing rate and membrane potential—have a bimodal distribution in the parametric space belonging to NREM dynamics, which is indicative of the alternation between Up and Down states and the presence of slow oscillations. The higher power at low frequencies in NREM versus the higher power at higher frequencies in wakefulness of the LFP also validates the existence of these two different dynamical regimes.
Few models exhibiting both regimes have been published up to date [
26,
27,
28,
29,
54]. Despite the fact that any computational model with multiple parameters exhibits a rich variety of bifurcations, we show that the sleep–waking transition can be obtained with the minimum set of possible changes. Naturally, the simplest solution is to vary a single parameter, in contrast with the proposal of [
27,
31]. Here, an increase in the AMPAergic conductance
suffices to move the system from NREM-like to waking-like dynamics. This is so because in the model, the membrane potential relaxes back to its steady state value producing damped oscillations in response to a perturbation for the parameter space of the NREM state (see
Figure S10) due to the presence of a stable focus [
55]. This behavior generates the slow oscillation in the presence of noise. Moreover, this change is consistent with the waxing and waning of synaptic strengths across the wake–sleep transition that SHY proposes [
17,
18,
19,
20,
21,
22,
23,
24,
25].
In this study, the connectivity between the two cortical columns is excitatory and symmetric, but only one of the pyramidal populations receives a transient external input. We investigate how this perturbation is transmitted to the other column in such a way that, in agreement with the aforementioned experiments, its response is enhanced in the awake state when compared to NREM. Previous computational work [
30] also aimed at reproducing the distinct propagation patterns that arise across the SWC, obtaining similar results to the ones described here. In particular, a wider propagation of the external perturbation occurs during wakefulness than during NREM sleep. In our case, this is quantified by the response of the unperturbed population, whereas [
30] uses the perturbational complexity index (PCI) [
56]. However, the set of parameters’ changes used by [
30] focus on an adaptation current that weakens during wakefulness, which in our opinion is less experimentally justified. Moreover, the authors showed that during wakefulness the ratio between poststimulus and prestimulus firing rate can be greater in an area that is not directly stimulated than in the stimulation site, which requires further experimental verification.
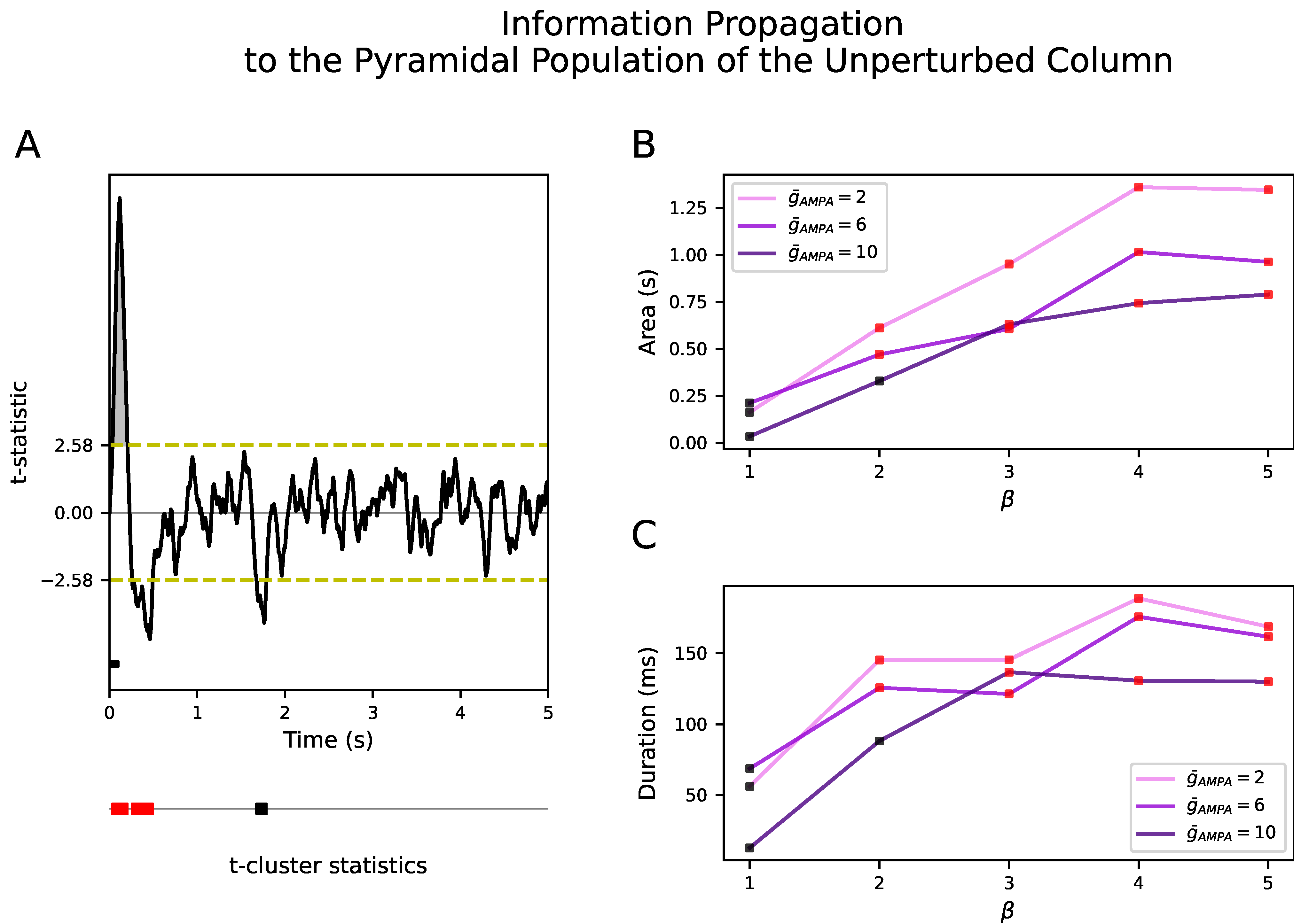
We show that when the conductance of the excitatory synapses,
, within a cortical column and between neighboring cortical columns are equal, there is no propagation of the input to the unperturbed cortical column. Propagation only occurs when the synaptic conductance between cortical columns is selectively boosted with respect to the synaptic conductance within cortical columns, a ratio that we have quantified with the parameter
. It is important to note that this difference between the inter- and intra-AMPAergic conductance does not directly result in a higher AMPAergic external than local synaptic afferent current. In fact, in the model, local synaptic currents surpass external ones both during NREM sleep and wakefulness. However, during wakefulness, this synaptic imbalance increases. Therefore, our results suggest that the synaptic homeostasis hypothesis should be heterogeneously applied, that is, that synaptic potentiation has to be spatially organized and distant synaptic conductances have to be scaled with respect to the local ones. Although this result needs experimental validation, it has been reported that, in the cortex, perforated synapses enlarge their axon–spine interface after waking relative to sleep, in contrast to the lack of change found in non-perforated synapses [
25]. This morphological change specific to perforated synapses also speaks for their major role in synaptic plasticity and the selective implementation of synaptic homeostasis that we propose.
Despite the fact that our model cannot account for whole brain interactions and is restricted to the dialogue between two networks, our results are derived from physiological assumptions and reproduce verified experimental observations. A more complex connectivity diagram comprising other cortical and subcortical structures could, nonetheless, provide spatial information about stimulus propagation. Subcortical structures, such as the brainstem and basal forebrain, are the substrate for the changes in the concentration of neuromodulators that occur throughout the sleep–weak cycle [
57]. These neuromodulators target vesicular release at the presynaptic site or transmitter receptors at the postsynaptic site and alter synaptic strength [
58,
59]. We have phenomenologically implemented their role by tuning synaptic conductances but the precise effect of these substances (serotonin, acetylcholine, histamine, etc.) on target brain areas is out of the scope of this study, partly because the exact microcircuitry of the ascending arousal system is still unknown.
It is important to note that we have excluded REM sleep from our work, which is a short but relevant stage of sleep that follows NREM epochs. The LFP and EEG dynamics recorded during REM sleep show similar features to wakefulness. Since firing responses in the primary auditory cortex are preserved across the SWC, including REM sleep [
7,
8], computationally, we could use the same set of parameters to model REM than wakefulness at a single cortical column level. However, tracking these responses up to the perirhinal cortex, shows that their amplitude is attenuated from wakefulness to NREM sleep, and has intermediate values in REM sleep [
8]. In our model, this could be replicated by using a
value for REM sleep smaller than for wakefulness (see
Figure 3B,C). However, experimentally, SHY has only proven to be true when comparing wakefulness and NREM sleep [
25]. Therefore, we need to be cautious before extending our conclusions to REM sleep.
Despite the limitations of our model, our work opens the door to explore which rules need to be implemented to reproduce experimental data and guide computational principles. This is particularly important while we still lack a clear understanding of how inputs scattered along the wide dendritic tree of a pyramidal neuron are added to combine synaptic sources at short and long distances with respect to the soma.