In Situ Produced Bacterial Cellulose Nanofiber-Based Hybrids for Nanocomposites
Abstract
:1. Introduction
2. Experimental
2.1. Design of Samples and Tests
2.2. Microorganism and Culture Media
2.3. In Situ Fabrication of BC
2.4. In Situ Fabrication of BC-MFC Hybrid Fibrous Structure
2.5. In Situ Fabrication of BC-Sisal Hybrid Fibrous Structure
2.6. Fabrication of BC-MFC-SPI Nanocomposites and BC-Sisal-SPI Microbond Specimens
2.7. Characterization
2.7.1. Tensile Test
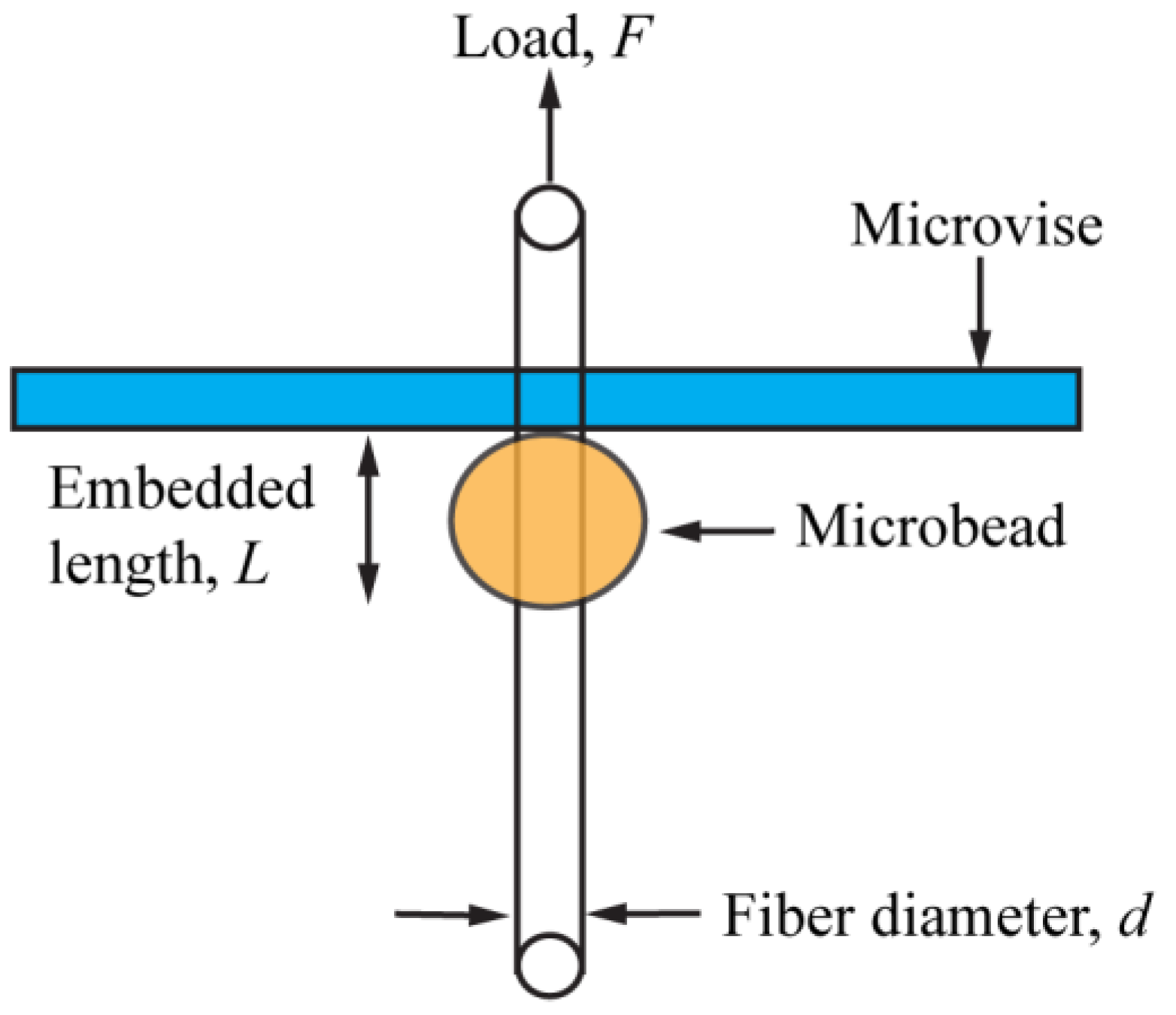
2.7.2. Interfacial Shear Strength (IFSS) Test
3. Results and Discussion
3.1. Mechanisms of In Situ Formation of BC-MFC and BC-Sisal Hybrid Fibrous Structures
3.2. Tensile Properties of BC-MFC Hybrid and BC-MFC-SPI Nanocomposite
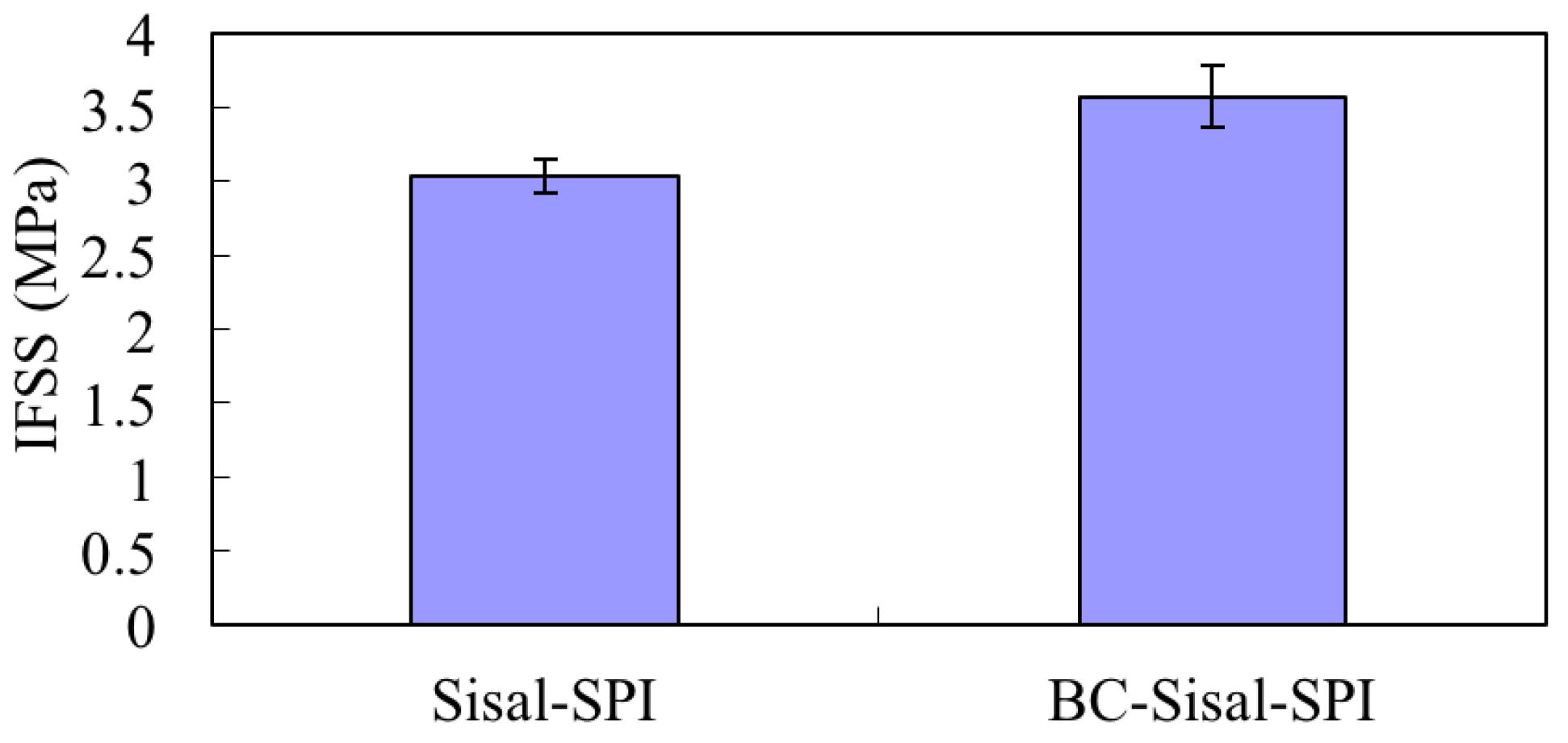
3.3. Comparison of IFSS for Sisal/SPI and BC-Sisal/SPI Microbond Specimens
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Qiu, K.; Netravali, A.N. A review of fabrication and applications of bacterial cellulose based nanocomposites. Polym. Rev. 2014, 54, 598–626. [Google Scholar] [CrossRef]
- Liebner, F.; Aigner, N.; Schimper, C.; Potthast, A.; Rosenau, T. Bacterial cellulose aerogels: From lightweight dietary food to functional materials. In Functional Materials from Renewable Sources; American Chemical Society: Washington, DC, USA, 2012; Volume 1107, pp. 57–74. [Google Scholar]
- Qiu, K.; Netravali, A.N. Polyvinyl alcohol based biodegradable polymer nanocomposites. In Biodegradable Polymers; Chu, C.-C., Ed.; Nova Science Publishers: Hauppauge, NY, USA, 2015; Volume 1, pp. 325–379. [Google Scholar]
- Qiu, K.; Netravali, A.N. ˝Green˝ composites based on bacterial cellulose produced using novel low-cost carbon source and soy protein resin. In Recent Advances in Adhesion Science and Technology in Honor of Dr. Kash Mittal; CRC Press: Boca Raton, FL, USA, 2014; pp. 193–208. [Google Scholar]
- Qiu, K.; Netravali, A.N. Bacterial cellulose-based membrane-like biodegradable composites using cross-linked and noncross-linked polyvinyl alcohol. J. Mater. Sci. 2012, 47, 6066–6075. [Google Scholar] [CrossRef]
- Bäckdahl, H.; Helenius, G.; Bodin, A.; Nannmark, U.; Johansson, B.R.; Risberg, B.; Gatenholm, P. Mechanical properties of bacterial cellulose and interactions with smooth muscle cells. Biomaterials 2006, 27, 2141–2149. [Google Scholar] [CrossRef] [PubMed]
- Klemm, D.; Schumann, D.; Udhardt, U.; Marsch, S. Bacterial synthesized cellulose—Artificial blood vessels for microsurgery. Prog. Polym. Sci. 2001, 26, 1561–1603. [Google Scholar] [CrossRef]
- Klemm, D.; Heublein, B.; Fink, H.-P.; Bohn, A. Cellulose: Fascinating biopolymer and sustainable raw material. Angew. Chem. Int. Ed. 2005, 44, 3358–3393. [Google Scholar] [CrossRef] [PubMed]
- Wan, W.K.; Hutter, J.L.; Milton, L.; Guhados, G. Bacterial cellulose and its nanocomposites for biomedical applications. In Cellulose Nanocomposites; American Chemical Society: Washingdon, DC, USA, 2006; Volume 938, pp. 221–241. [Google Scholar]
- Qiu, K.; Netravali, A.N. Fabrication and characterization of biodegradable composites based on microfibrillated cellulose and polyvinyl alcohol. Compos. Sci. Technol. 2012, 72, 1588–1594. [Google Scholar] [CrossRef]
- Wan, Y.Z.; Hong, L.; Jia, S.R.; Huang, Y.; Zhu, Y.; Wang, Y.L.; Jiang, H.J. Synthesis and characterization of hydroxyapatite—Bacterial cellulose nanocomposites. Compos. Sci. Technol. 2006, 66, 1825–1832. [Google Scholar] [CrossRef]
- Qiu, K. Biobased and Biodegrable Polymer Nanocomposites; Cornell University: Ithaca, NY, USA, 2012. [Google Scholar]
- Netravali, A.N.; Qiu, K. Bacterial Cellulose Based ‘Green’ Composites. US Patent No. 9,499,686 B2, 22 November 2016. [Google Scholar]
- Fontana, J.D.; De Souza, A.M.; Fontana, C.K.; Torriani, I.L.; Moreschi, J.C.; Gallotti, B.J.; De Souza, S.J.; Narcisco, G.P.; Bichara, J.A.; Farah, L.F.X. Acetobacter cellulose pellicle as a temporary skin substitute. Appl. Biochem. Biotechnol. 1990, 24, 253–264. [Google Scholar] [CrossRef] [PubMed]
- Svensson, A.; Nicklasson, E.; Harrah, T.; Panilaitis, B.; Kaplan, D.L.; Brittberg, M.; Gatenholm, P. Bacterial cellulose as a potential scaffold for tissue engineering of cartilage. Biomaterials 2005, 26, 419–431. [Google Scholar] [CrossRef] [PubMed]
- Fink, H.P.; Weigel, P.; Purz, H.J.; Ganster, J. Structure formation of regenerated cellulose materials from nmmo-solutions. Prog. Polym. Sci. 2001, 26, 1473–1524. [Google Scholar] [CrossRef]
- Shibazaki, H.; Kuga, S.; Onabe, F.; Usuda, M. Bacterial cellulose membrane as separation medium. J. Appl. Polym. Sci. 1993, 50, 965–969. [Google Scholar] [CrossRef]
- Hong, F.; Qiu, K. An alternative carbon source from konjac powder for enhancing production of bacterial cellulose in static cultures by a model strain acetobacter aceti subsp. Xylinus atcc 23770. Carbohydr. Polym. 2008, 72, 545–549. [Google Scholar] [CrossRef]
- Pommet, M.; Juntaro, J.; Heng, J.Y.Y.; Mantalaris, A.; Lee, A.F.; Wilson, K.; Kalinka, G.; Shaffer, M.S.P.; Bismarck, A. Surface modification of natural fibers using bacteria: Depositing bacterial cellulose onto natural fibers to create hierarchical fiber reinforced nanocomposites. Biomacromolecules 2008, 9, 1643–1651. [Google Scholar] [CrossRef] [PubMed]
- Grande, C.J.; Torres, F.G.; Gomez, C.M.; Troncoso, O.P.; Canet-Ferrer, J.; Martínez-Pastor, J. Development of self-assembled bacterial cellulose—Starch nanocomposites. Mater. Sci. Eng. C 2009, 29, 1098–1104. [Google Scholar] [CrossRef]
- Juntaro, J.; Pommet, M.; Kalinka, G.; Mantalaris, A.; Shaffer, M.S.P.; Bismarck, A. Creating hierarchical structures in renewable composites by attaching bacterial cellulose onto sisal fibers. Adv. Mater. 2008, 20, 3122–3126. [Google Scholar] [CrossRef]
- Lee, K.-Y.; Bharadia, P.; Blaker, J.J.; Bismarck, A. Short sisal fibre reinforced bacterial cellulose polylactide nanocomposites using hairy sisal fibres as reinforcement. Compos. Part A 2012, 43, 2065–2074. [Google Scholar] [CrossRef]
- Nakagaito, A.N.; Yano, H. The effect of fiber content on the mechanical and thermal expansion properties of biocomposites based on microfibrillated cellulose. Cellulose 2008, 15, 555–559. [Google Scholar] [CrossRef]
- Nakagaito, A.N.; Fujimura, A.; Sakai, T.; Hama, Y.; Yano, H. Production of microfibrillated cellulose (mfc)-reinforced polylactic acid (pla) nanocomposites from sheets obtained by a papermaking-like process. Compos. Sci. Technol. 2009, 69, 1293–1297. [Google Scholar] [CrossRef]
- Huang, X.; Netravali, A. Biodegradable green composites made using bamboo micro/nano-fibrils and chemically modified soy protein resin. Compos. Sci. Technol. 2009, 69, 1009–1015. [Google Scholar] [CrossRef]
- Qiu, K.; Netravali, A.N. A composting study of membrane-like polyvinyl alcohol based resins and nanocomposites. J. Polym. Environ. 2013, 21, 658–674. [Google Scholar] [CrossRef]
- Stenstad, P.; Andresen, M.; Tanem, B.S.; Stenius, P. Chemical surface modifications of microfibrillated cellulose. Cellulose 2008, 15, 35–45. [Google Scholar] [CrossRef]
- Nakagaito, A.N.; Yano, H. The effect of morphological changes from pulp fiber towards nano-scale fibrillated cellulose on the mechanical properties of high-strength plant fiber based composites. Appl. Phys. A 2004, 78, 547–552. [Google Scholar] [CrossRef]
- Nakagaito, A.N.; Yano, H. Novel high-strength biocomposites based on microfibrillated cellulose having nano-order-unit web-like network structure. Appl. Phys. A 2005, 80, 155–159. [Google Scholar] [CrossRef]
- Nakagaito, A.N.; Iwamoto, S.; Yano, H. Bacterial cellulose: The ultimate nano-scalar cellulose morphology for the production of high-strength composites. Appl. Phys. A 2005, 80, 93–97. [Google Scholar] [CrossRef]
- Kim, J.T.; Netravali, A.N. Effect of protein content in soy protein resins on their interfacial shear strength with ramie fibers. J. Adhes. Sci. Technol. 2010, 24, 203–215. [Google Scholar] [CrossRef]
- Kim, J.R.; Netravali, A.N. Self-healing properties of protein resin with soy protein isolate-loaded poly(d,l-lactide-co-glycolide) microcapsules. Adv. Funct. Mater. 2016, 26, 4786–4796. [Google Scholar] [CrossRef]
- Netravali, A.N.; Chabba, S. Composites get greener. Mater. Today 2003, 6, 22–29. [Google Scholar] [CrossRef]
- Lodha, P.; Netravali, A.N. Characterization of stearic acid modified soy protein isolate resin and ramie fiber reinforced ‘green’ composites. Compos. Sci. Technol. 2005, 65, 1211–1225. [Google Scholar] [CrossRef]
- Nam, S.; Netravali, A.N. Characterization of ramie fiber/soy protein concentrate (spc) resin interface. J. Adhes. Sci. Technol. 2004, 18, 1063–1076. [Google Scholar] [CrossRef]
- Nam, S.; Netravali, A.N. Green composites. II. Environment-friendly, biodegradable composites using ramie fibers and soy protein concentrate (spc) resin. Fibers Polym. 2006, 7, 380–388. [Google Scholar] [CrossRef]
- Warner, S.B. Fiber Science; Prentice Hall: Upper Saddle River, NJ, USA, 1995. [Google Scholar]
- Lodha, P. Fundamental Approaches to Improving Performance of Soy Protein Isolate Based ‘Green’ Plastics and Composites; Cornell University: Ithaca, NY, USA, 2004. [Google Scholar]


| Young′s Modulus (MPa) | Theoretical Young′s Modulus (MPa) | Tensile Strength (MPa) | Theoretical Tensile Strength (MPa) | Fracture Strain (%) | |
|---|---|---|---|---|---|
| BC | 2490 (9.6) * | 79.1 (13.6) | 5.6 (17.8) | ||
| BC-MFC (MFC 50%) | 2826 (6.9) | 84.1 (14.7) | 6.0 (22.3) | ||
| SPI resin | 218 (8.3) | 9.7 (9.5) | 103.5 (24.1) | ||
| BC-SPI (SPI 50%) | 1284 (15.1) | 1265 | 41.8 (2.9) | 41.7 | 5.5 (20.6) |
| BC-MFC-SPI (SPI 50%) | 1408 (19.4) | 1420 | 44.0 (12.9) | 44.0 | 5.8 (11.3) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qiu, K.; Netravali, A. In Situ Produced Bacterial Cellulose Nanofiber-Based Hybrids for Nanocomposites. Fibers 2017, 5, 31. https://doi.org/10.3390/fib5030031
Qiu K, Netravali A. In Situ Produced Bacterial Cellulose Nanofiber-Based Hybrids for Nanocomposites. Fibers. 2017; 5(3):31. https://doi.org/10.3390/fib5030031
Chicago/Turabian StyleQiu, Kaiyan, and Anil Netravali. 2017. "In Situ Produced Bacterial Cellulose Nanofiber-Based Hybrids for Nanocomposites" Fibers 5, no. 3: 31. https://doi.org/10.3390/fib5030031
APA StyleQiu, K., & Netravali, A. (2017). In Situ Produced Bacterial Cellulose Nanofiber-Based Hybrids for Nanocomposites. Fibers, 5(3), 31. https://doi.org/10.3390/fib5030031







