Comparison of Engineered Peptide-Glycosaminoglycan Microfibrous Hybrid Scaffolds for Potential Applications in Cartilage Tissue Regeneration
Abstract
:1. Introduction
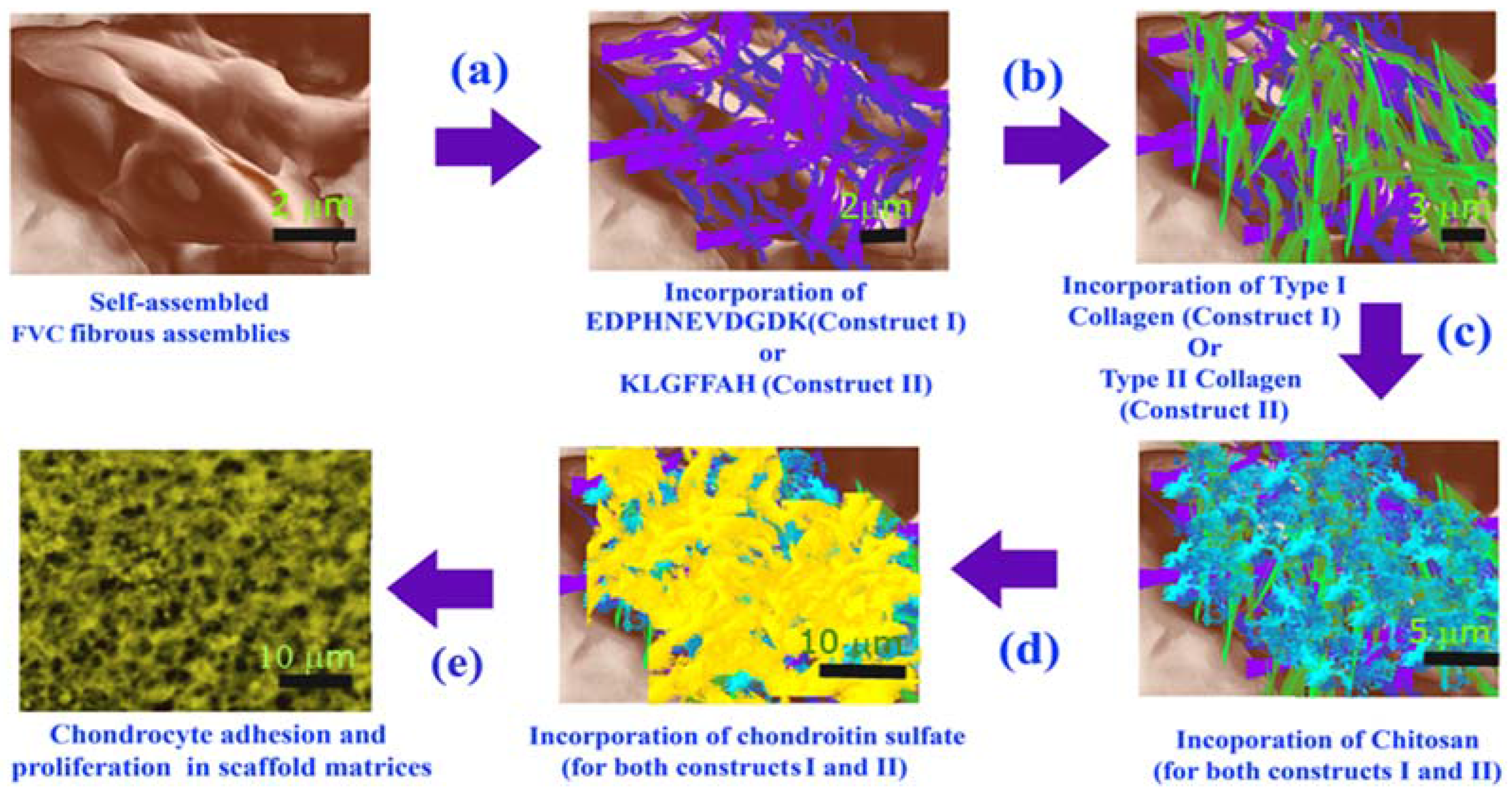
2. Experimental Section
2.1. Materials
2.2. Methods
2.2.1. Preparation of Fmoc-Valyl-Cetylamide (FVC) Fibrous Assemblies
2.2.2. Layer-by-Layer Assembly of Construct I (FVC-DSp-Col-I-Ch-CS)
2.2.3. Layer-by-Layer Assembly of Construct II (FVC-CP-Col-II-Ch-CS)
2.3. Characterization
2.3.1. Scanning Electron Microscopy (SEM)
2.3.2. Fourier Transform Infrared Spectroscopy (FTIR)
2.3.3. Differential Scanning Calorimetry
2.3.4. Mechanical Testing of Constructs
2.4. Cell Studies
2.4.1. Cell Imaging
2.4.2. Cell Proliferation
2.4.3. Chondrogenesis Assay
2.4.4. Soluble Collagen Assay
2.5. Statistical Analysis
3. Results
3.1. Formation of Scaffold Constructs


3.2. FTIR Spectroscopy
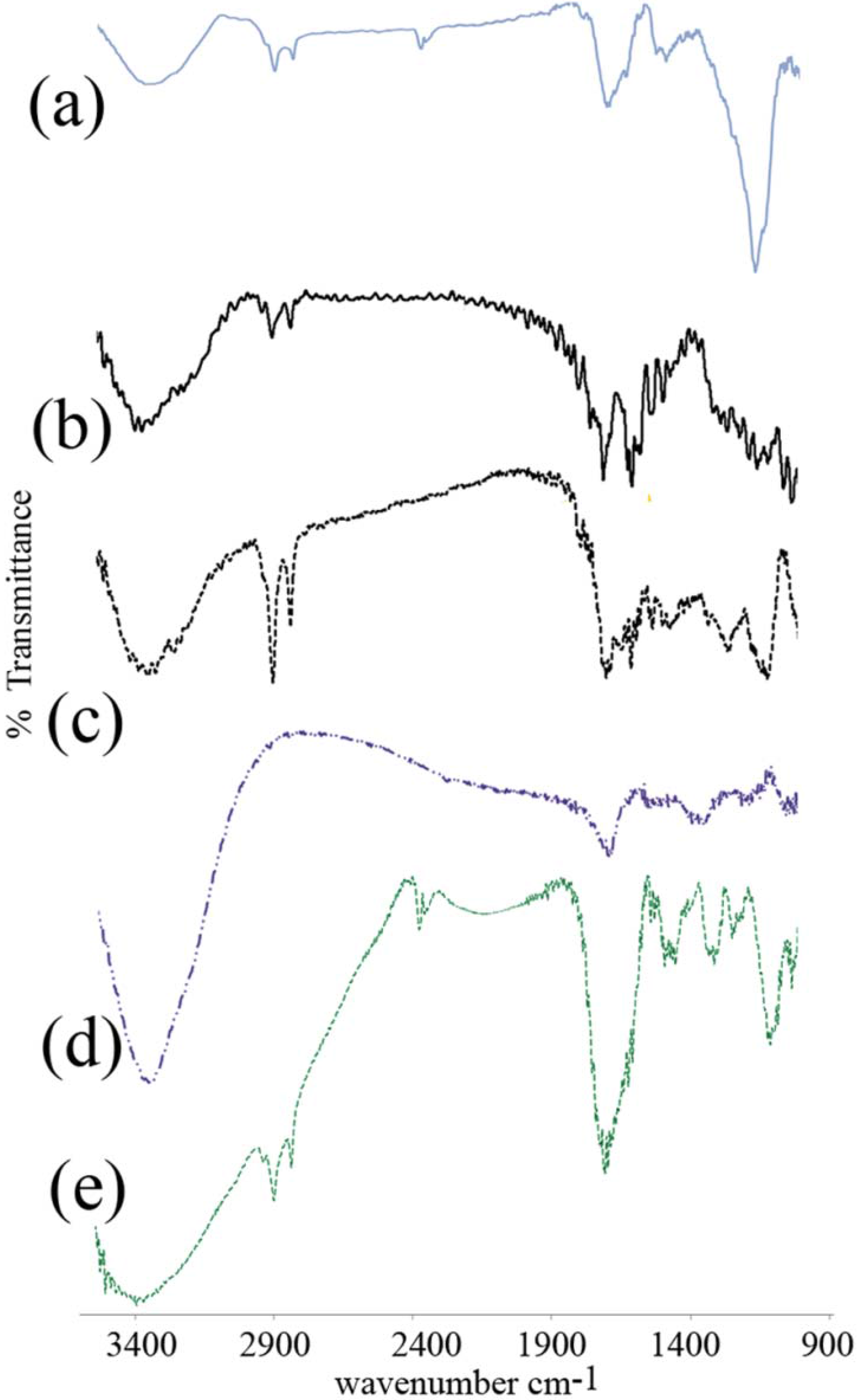
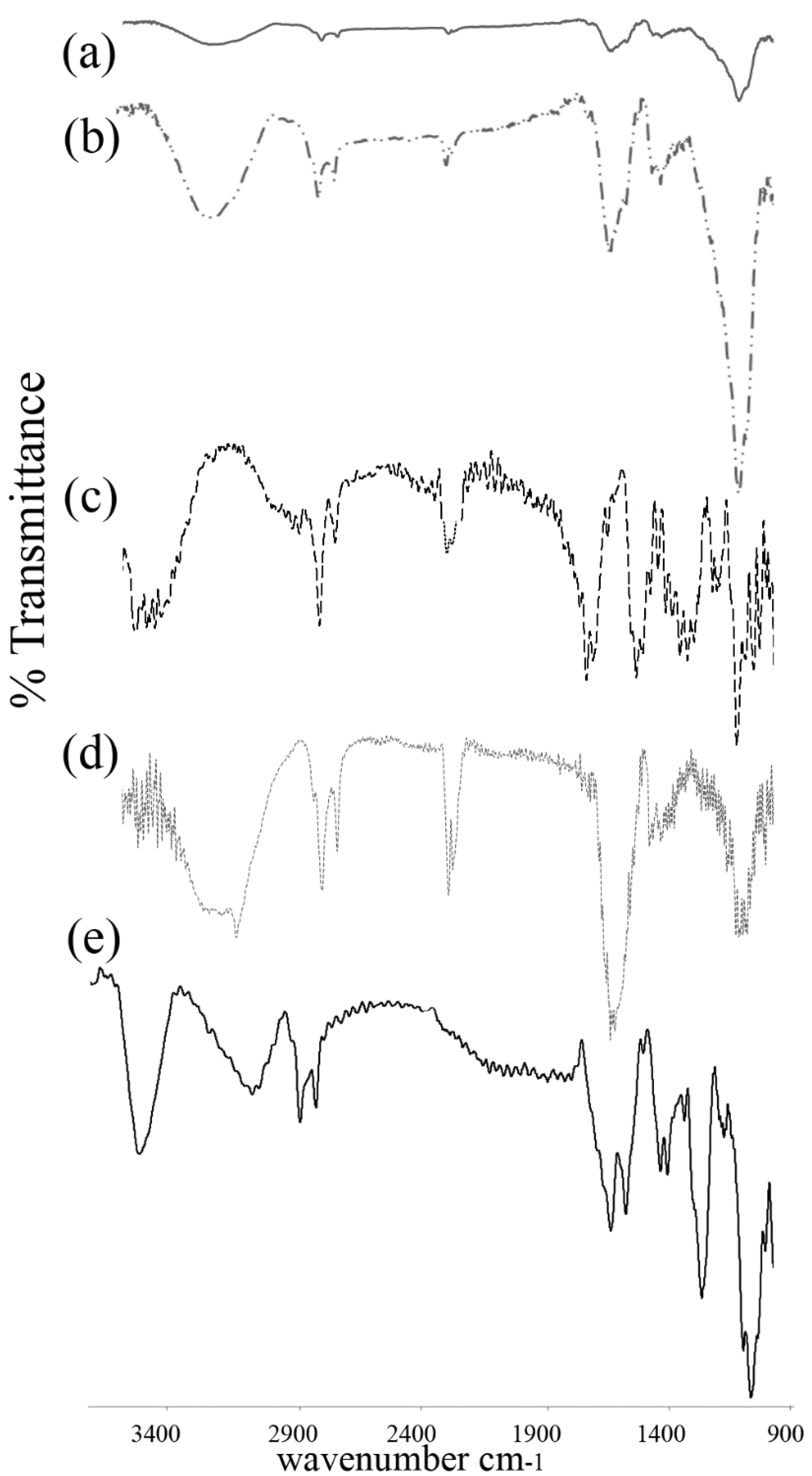
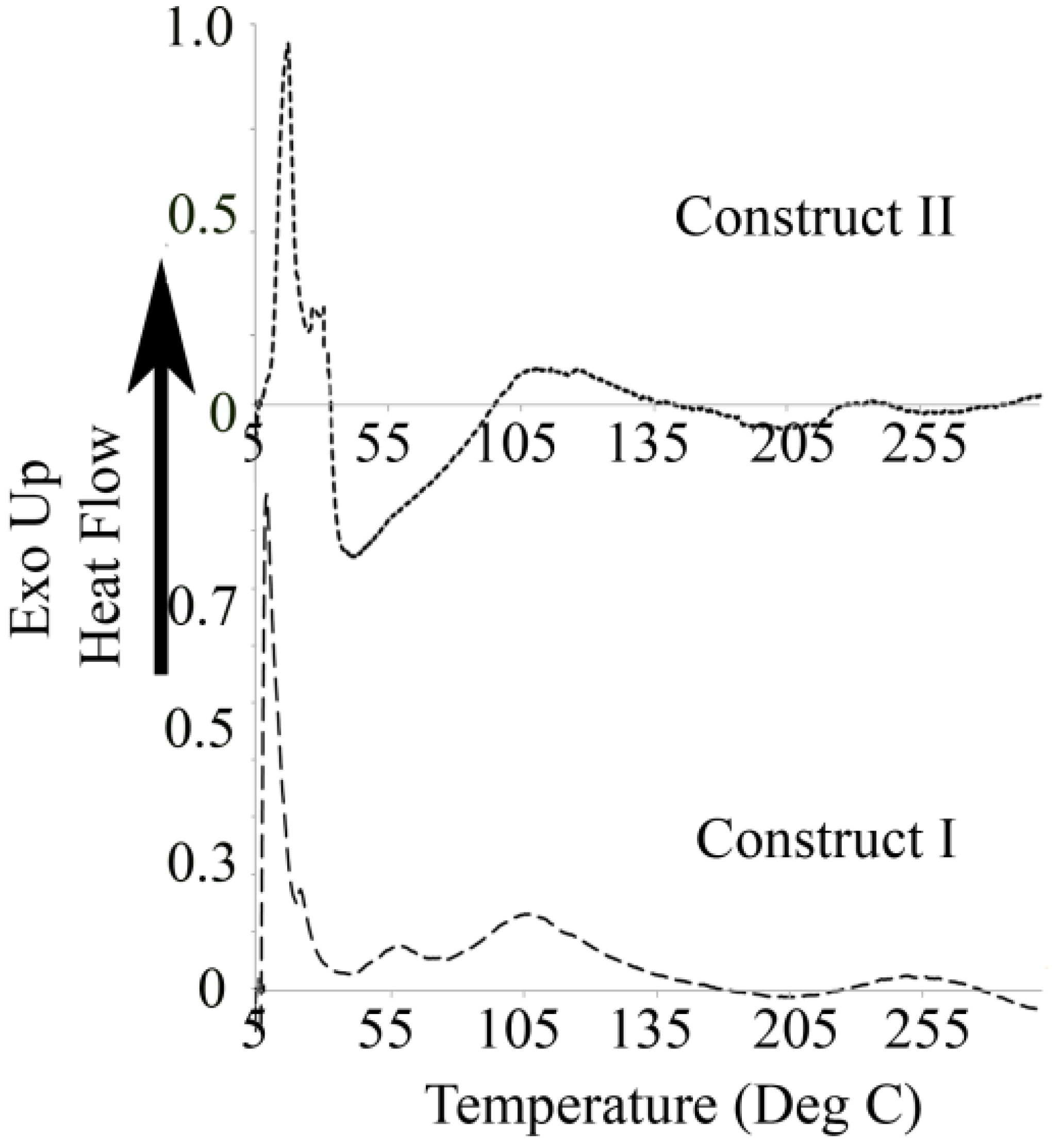
3.3. Differential Scanning Calorimetry
3.4. AFM imaging and Nanomechanical Properties
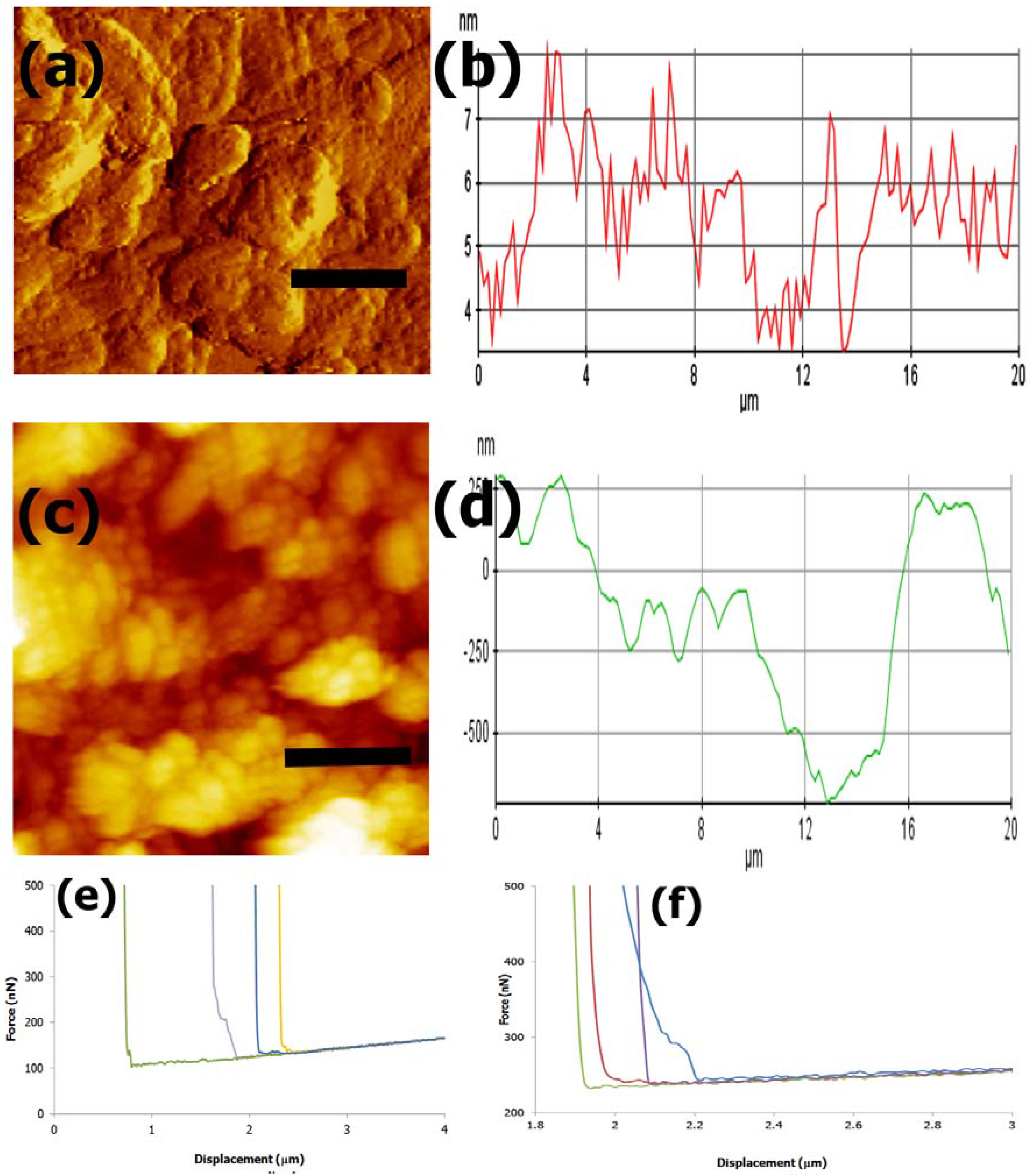
3.5. Cell Studies
3.5.1. Cell Morphology and Attachment to Constructs

3.5.2. Cell Proliferation Studies
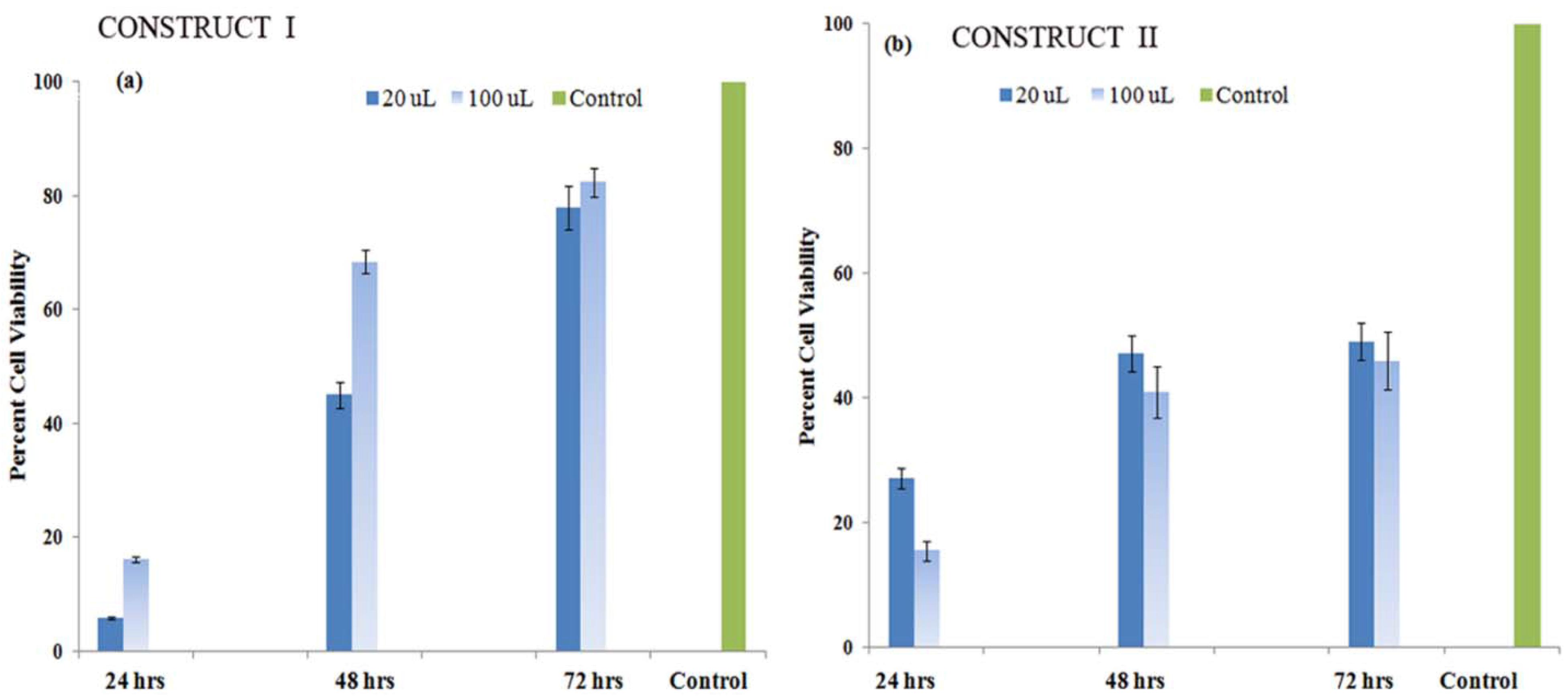
3.5.3. Formation of Proteoglycans
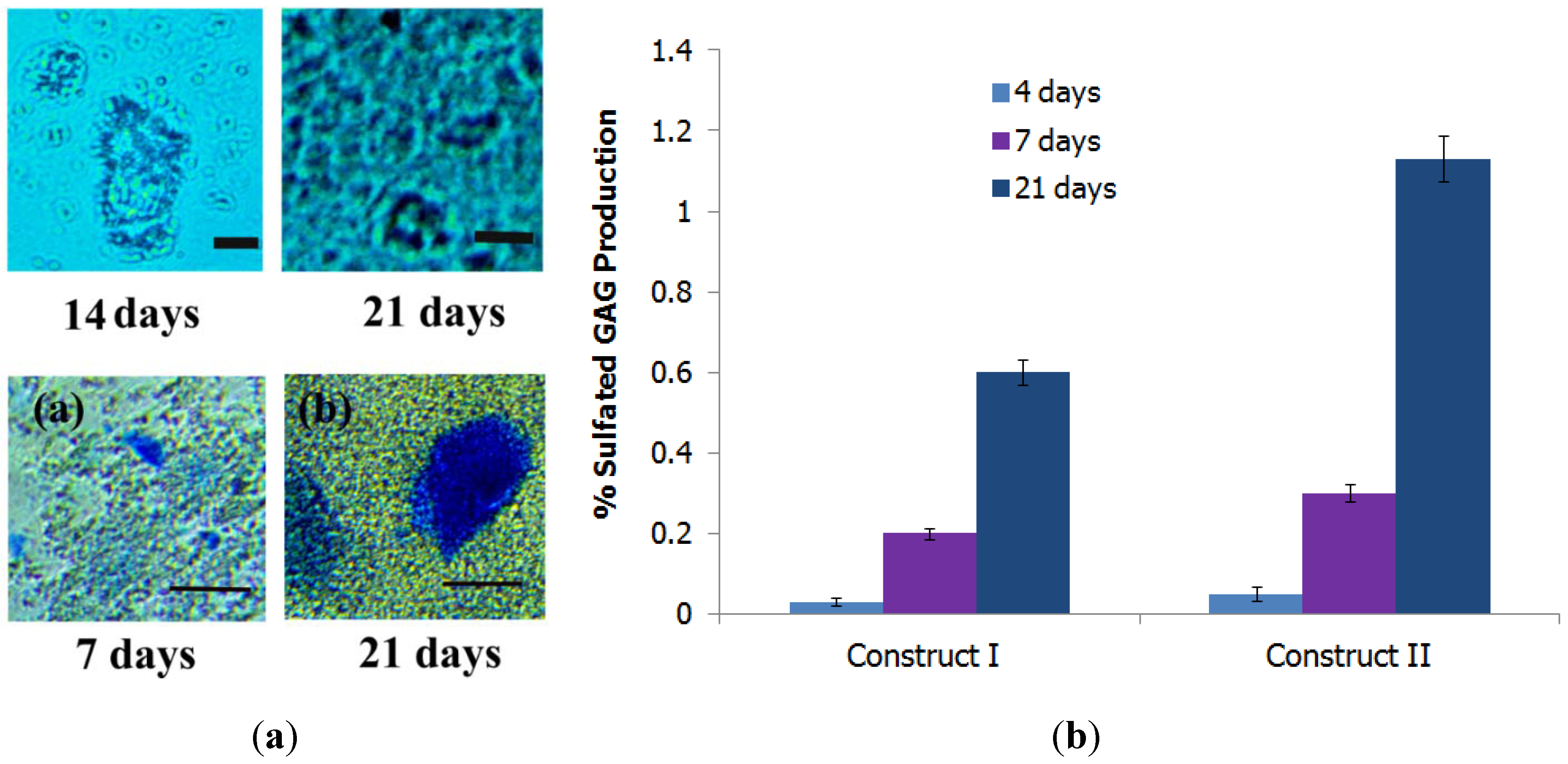
3.5.4. Induction of Soluble Collagen
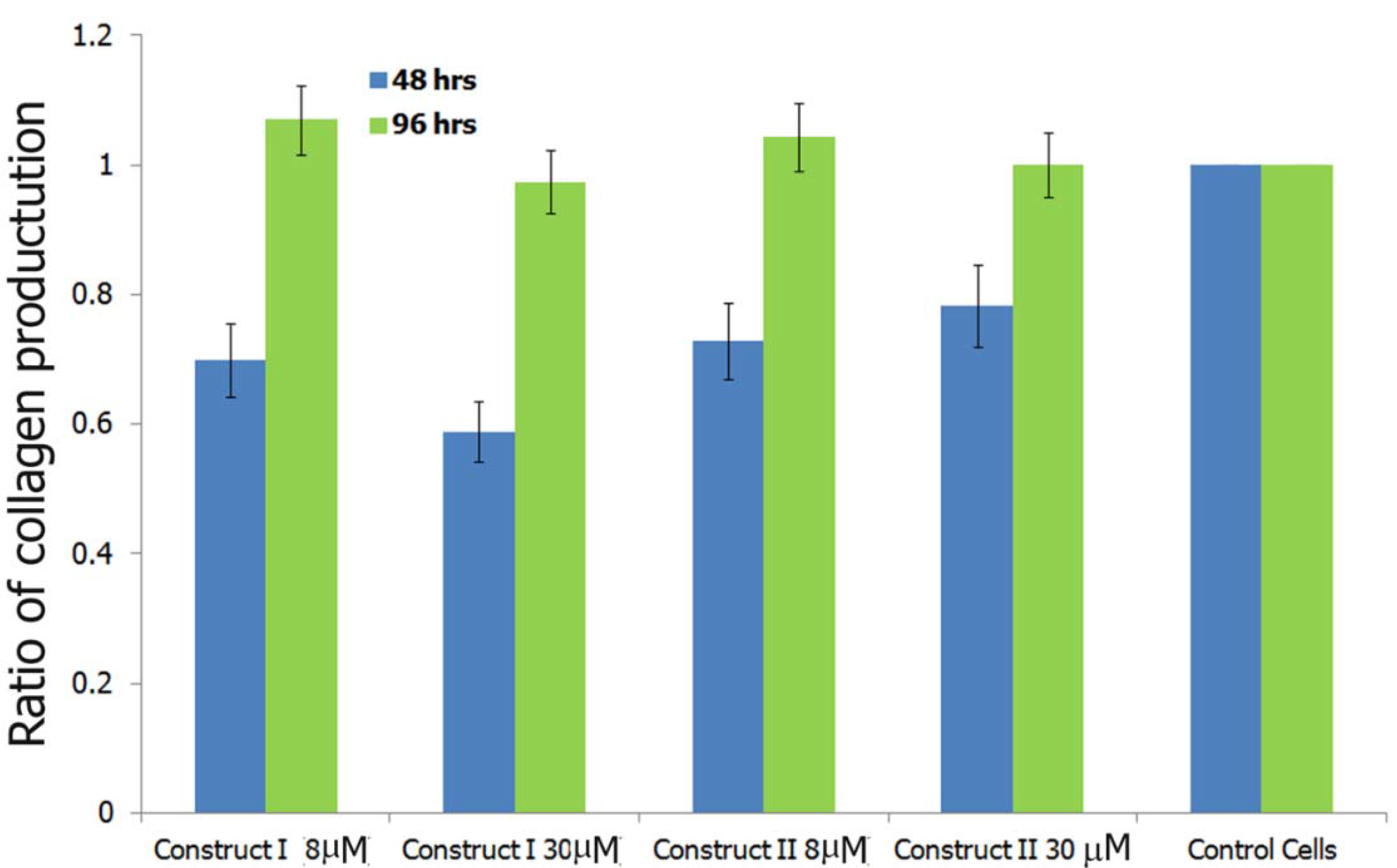
4. Discussion
4.1. Development of Scaffolds
4.2. FTIR Spectroscopy
4.3. Differential Scanning Calorimetry
4.4. AFM Imaging and Nanoindentation Analysis
4.5. Cell Morphology and Adhesion
4.6. Proteoglycan Synthesis
4.7. Soluble Collagen Synthesis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Khademhosseini, A.; Vacanti, J.P.; Langer, R. Progress in tissue engineering. Sci. Am. 2009, 300, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Healy, K.E.; Guldberg, R.E. Bone tissue engineering. J. Musculoskelet. Neuronal Interact. 2007, 7, 328–330. [Google Scholar] [PubMed]
- Moreira-Teixeira, L.S.; Georgi, N.; Leijten, J.; Wu, L.; Karperien, M. Cartilage tissue engineering. Endocr. Dev. 2011, 21, 102–115. [Google Scholar] [PubMed]
- Shi, J.; Votruba, A.R.; Farokhzad, O.C.; Langer, R. Nanotechnology in drug delivery and tissue engineering: From discovery to applications. Nano. Lett. 2010, 10, 3223–3230. [Google Scholar] [CrossRef] [PubMed]
- Lipkin, R. Tissue engineering: Replacing damaged organs with new tissues. Sci. News 1995, 148, 24–26. [Google Scholar] [CrossRef]
- Huey, D.J.; Hu, J.C.; Athanasiou, K.A. Unlike bone, cartilage regeneration remains elusive. Science 2012, 338, 917–921. [Google Scholar] [CrossRef] [PubMed]
- Pullig, O.; Weseloh, G.; Klatt, A.R.; Wagener, R.; Swobda, B. Matrilin-3 in human articular cartilage: Increased expression in osteoarthritis. Osteoarthr. Cartil. 2002, 10, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Buckwalter, J.A.; Mankin, H.J. Articular cartilage: Degeneration and osteoarthritis, repair, regeneration and transplantation. Instr. Course Lect. 1998, 47, 487–504. [Google Scholar] [PubMed]
- Hjelle, K.; Solheim, E.; Strand, T.; Muri, R.; Brittberg, M. Articular cartilage defects in 1,000 knee arthroscopies. Arthrosc. J. Arthrosc. Relat. Surg. 2002, 18, 730–734. [Google Scholar] [CrossRef]
- Buckwalter, J.A.; Mankin, H.J. Articular cartilage: Tissue design and chondrocyte-matrix interactions. Instr. Course Lect. 1998, 47, 477–486. [Google Scholar] [PubMed]
- Newman, A.P. Articular cartilage repair. Am. J. Sports Med. 1998, 26, 309–324. [Google Scholar] [PubMed]
- Hunzinker, E.B. Articular cartilage repair: Basic science and clinical progress. A review on the current status and prospectives. Osteoarthr. Cartil. 2001, 10, 432–463. [Google Scholar] [CrossRef] [PubMed]
- Darling, E.M.; Athanasiou, K.A. Biomedical strategies for articular cartilage regeneration. Ann. Biomed. Eng. 2003, 31, 1114–1124. [Google Scholar] [CrossRef] [PubMed]
- Suh, J.K.; Matthew, H.W.T. Application of chitosan-based polysaccharide biomaterials in cartilage tissue engineering: A review. Biomaterials 2000, 21, 2589–2598. [Google Scholar] [PubMed]
- Hutchmacher, D.W. Scaffolds in tissue engineering bone and cartilage. Biomaterials 2000, 21, 2529–2543. [Google Scholar] [CrossRef]
- Choi, B.; Kim, S.; Lin, B.; Wu, B.M.; Lee, M. Cartilaginous extracellular matrix-modified chitosan hydrogels for cartilage tissue engineering. ACS Appl. Mater. Interfaces 2014, 6, 20110–20121. [Google Scholar] [CrossRef] [PubMed]
- Van Vlierbergh, S.; Dubruel, P.; Schacht, E. Biopolymer-based hydrogels as scaffolds for tissue engineering applications: A review. Biomacromolecules 2011, 12, 1387–1408. [Google Scholar] [CrossRef] [PubMed]
- Yamaoka, H.; Asato, H.; Ogasawara, T.; Nishizawa, S.; Takahashi, T.; Nakatsuka, T.; Koshima, I.; Nakamura, K.; Kawaguchi, H.; Chung, U.; et al. Cartilage tissue engineering using human auricular chondrocytes embedded in different hydrogel materials. J. Biomed. Mater. Res. A 2006, 78, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Chaipinyo, K.; Oakes, B.W.; Van Damme, M.P. The use of derbrided human articular cartilage for autologous chondrocyte implantation: Maintenance of chondrocyte differentiation and proliferation in type I collagen gels. J. Orthop. Res. 2004, 22, 446–455. [Google Scholar] [CrossRef] [PubMed]
- Kisiday, J.; Jin, M.; Kurz, B.; Hung, H.; Semino, C.; Zhang, S.; Grodzinsky, A.J. Self-assembling peptide hydrogel fosters chondrocyte extracellular matrix production and cell division: Implications for cartilage tissue repair. Proc. Natl. Acad. Sci. USA 2002, 99, 9996–10001. [Google Scholar] [CrossRef] [PubMed]
- Mart, R.J.; Osborne, R.D.; Stevens, M.M.; Ulijn, R.V. Peptide-based stimuli-responsive biomaterials. Soft Mater 2006, 2, 822–835. [Google Scholar] [CrossRef]
- Bryant, S.J.; Anseth, K.S. Controlling the spatial distribution of ECM components in degradable PEG hydrogels for tissue engineering cartilage. J. Biomed. Mater. Res. A 2003, 64, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Drury, J.L.; Mooney, D.J. Hydrogels for tissue engineering: Scaffold design variables and applications. Biomaterials 2003, 24, 4337–4351. [Google Scholar] [CrossRef]
- Noth, U.; Rackwitz, L.; Heymer, A.; Weber, M.; Baumann, B.; Steinhert, A.; Schutze, N.; Jakob, F.; Eulert, J. Chondrogenic differentiation of human mesenchymal stem cells in collagen type I hydrogels. J. Biomed. Mater. Res. A 2007, 83, 626–635. [Google Scholar] [CrossRef] [PubMed]
- Balakrishnan, B.; Banerjee, R. Biopolymer-based hydrogels for cartilage tissue engineering. Chem. Rev. 2011, 111, 4453–4474. [Google Scholar] [CrossRef] [PubMed]
- Nehrer, S.; Domayer, S.; Dorotka, R.; Schatz, K.; Bindreiter, U.; Kotz, R. Three-year clinical outcome after chondrocyte transplantation using a hyaluronan matrix for cartilage repair. Eur. J. Radiol. 2006, 57, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Mattioli-Belmonte, M.; Gigante, A.; Muzzarelli, R.A.; Politano, R.; De Benedittis, A.; Specchia, N.; Buffa, A.; Biagani, G.; Greco, F. N,N-dicarboxymethyl chitosan as delivery agent for bone morphogenetic protein in the repair of articular cartilage. Med. Biol. Eng. Comput. 1999, 37, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Fan, H.; Hu, Y.; Zhang, C.; Li, X.; Lv, R.; Qin, L.; Zhu, R. Cartilage regeneration using mesenchymal stem cells and a PLGA-gelatin/chondroitin/hyaluronate hybrid scaffold. Biomaterials 2006, 27, 4573–4580. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.R.; Grad, S.; Gorna, K.; Gogolewski, S.; Goessl, A.; Alini, M. Fibrin-polyurethane composites for articular cartilage tissue engineering: A preliminary analysis. Tissue Eng. 2005, 11, 1562–1573. [Google Scholar] [CrossRef] [PubMed]
- Paramonov, S.E.; Jun, H.W.; Hargerink, J.D. Self-assembly of peptide-amphiphile nanofibers: The roles of hydrogen bonding and amphiphilic packing. J. Am. Chem. Soc. 2006, 128, 7291–7298. [Google Scholar] [CrossRef] [PubMed]
- Jun, H.W.; Paramonov, S.E.; Hartgerink, J.D. Biomimetic self-assembled nanofibers. Soft Matter 2006, 2, 177–181. [Google Scholar] [CrossRef]
- Slaughter, B.V.; Kurshid, S.S.; Fisher, O.Z.; Khademhosseini, A.; Peppas, N.A. Hydrogels in regenerative medicine. Adv. Mater 2009, 21, 3307–3329. [Google Scholar] [CrossRef] [PubMed]
- Shin, H.; Jo, S.; Mikos, A.G. Biomimetic materials for tissue engineering. Biomaterials 2003, 24, 4353–4364. [Google Scholar] [CrossRef]
- Hersel, U.; Dahmen, C.; Kessler, H. RGD modified polymers: Biomaterials for stimulated cell adhesion and beyond. Biomaterials 2003, 24, 4385–4415. [Google Scholar] [CrossRef]
- Ryan, D.M.; Nilsson, B.L. Self-assembled amino acids and dipeptides as non covalent hydrogels for tissue engineering. Polym. Chem. 2012, 3, 18–33. [Google Scholar] [CrossRef]
- Jayawarna, V.; Richardson, S.M.; Hirst, A.R.; Hodson, N.W.; Saiani, A.; Gough, J.E.; Ulijn, R.V. Introducing chemical functionality in Fmoc-peptide gels for cell culture. Acta Biomater. 2009, 5, 934–943. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Smith, A.M.; Das, A.K.; Hodson, N.W.; Collins, R.F.; Ulijn, R.V.; Gough, J.E. Self-assembled peptide-based hydrogels as scaffolds for anchorage-dependent cells. Biomaterials 2009, 30, 2523–2530. [Google Scholar] [CrossRef] [PubMed]
- Romanelli, S.M.; Hartnett, J.W.; Banerjee, I.A. Effects of amide side chains on nanoassembly formation and gelation of Fmoc-valine conjugates. Powder Technol. 2015, 271, 76–87. [Google Scholar] [CrossRef]
- Romanelli, S.M.; Fath, K.R.; Phekoo, A.P.; Knoll, G.A.; Banerjee, I.A. Layer-by-layer assembly of peptide based bioorganic-inorganic hybrid scaffolds and their interactions with osteoblastic MC3T3-E1 cells. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 51, 316–328. [Google Scholar] [CrossRef] [PubMed]
- Qin, C.; Brunn, J.C.; Cadena, E.; Ridall, A.; Tsujigiwa, H.; Nagatsuka, H.; Nagai, N.; Butler, W.T. The expression of dentin sialophosphoprotein gene in bone. J. Dent. Res. 2002, 81, 392–394. [Google Scholar] [CrossRef] [PubMed]
- Ferré, F.C.; Larjava, H.; Loison-Robert, L.S.; Berbar, T.; Owen, G.R.; Berdal, A.; Cherafi, H.; Gogly, B.; Hakkinen, L.; Fournier, P.J.B. Formation of cartilage and synovial tissue by human gingival stem cells. Stem. Cells Dev. 2014, 23, 2895–2906. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Gibson, M.P.; Sun, H.; Qin, C. Dentin sialophosphoprotein (DSPP) plays an essential role in postnatal development and maintenance of mouse mandibular condylar cartilage. J. Histochem. Cytochem. 2013, 61, 749–758. [Google Scholar]
- Choi, Y.S.; Lee, J.Y.; Suh, J.S.; Lee, G.; Chung, C.P.; Park, Y.J. The mineralization inducing peptide derived from dentin sialophosphoprotein for bone regeneration. J. Biomed. Mater. Res. 2013, 101A, 590–598. [Google Scholar] [CrossRef] [PubMed]
- Yagihashi, K.; Miyazawa, K.; Togari, K.; Goto, S. Demineralized dentin matrix acts as a scaffold for repair of articular cartilage defects. Calcif. Tissue Int. 2009, 84, 210–220. [Google Scholar] [CrossRef] [PubMed]
- Fisher, L.W.; Tochia, D.A.; Fohr, B.; Young, M.F.; Fedarko, N.S. Flexible structures of SIBLING proteins, bone sialophosphortein and osteopontin. Biochem. Biophys. Res. Commun. 2001, 280, 460–465. [Google Scholar] [CrossRef] [PubMed]
- Prasad, M.; Zhu, Q.; Sun, Y.; Wang, X.; Kulkarni, A.; Boskey, A.; Feng, J.Q.; Qin, C. Expression of dentin sialophosphoprotein in non-mineralized tissues. J. Histochem. Cytochem. 2011, 59, 1009–1021. [Google Scholar] [CrossRef] [PubMed]
- Ramadass, S.K.; Perumal, S.; Gopinath, A.; Nisal, A.; Subramanjan, S.; Madhan, B. Sol-gel assisted fabrication of collagen hydrolysate composite scaffold: A novel therapeutic alternative to the traditional collagen scaffold. ACS Appl. Mater. Interfaces 2014, 6, 15015–15025. [Google Scholar] [CrossRef] [PubMed]
- Eyre, D.R.; Muir, H. Quantitative analysis of types I and II collagen in human intervertebral discs at various ages. Biochim. Biophys. Acta 1977, 492, 29–42. [Google Scholar] [CrossRef]
- Hong, Y.; Song, H.; Gong, Y.; Mao, Z.; Gao, C.; Shen, J. Covalently crosslinked chitosan hydrogel: Properties of in vitro degradation and chondrocyte encapsulation. Acta Biomater. 2007, 3, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Prydz, K.; Dalen, K.T. Synthesis and sorting of proteoglycans. J. Cell Sci. 2000, 113, 193–205. [Google Scholar] [PubMed]
- Di Martino, A.; Sittinger, M.; Risbud, M.V. Chitosan: A versatile biopolymer for orthapaedic tissue-engineering. Biomaterials 2005, 26, 5983–5990. [Google Scholar] [CrossRef] [PubMed]
- Jerosch, J. Effects of glucosamine and chondroitin sulfate on cartilage metabolism in OA: Outlook on other nutrient partners especially omega-3-fatty acids. Int. J. Rheumatol. 2011, 2011, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Wang, J.; Su, S.; Wang, E.; Qiu, J. Tough and fully cyclic recoverable biomimetic hydrogels. J. Mater. Chem. B 2015, 3, 5284–5290. [Google Scholar] [CrossRef]
- Lipiello, L. Glucosamine and chondroitin sulfate: Biological response modifiers of chondrocytes under simualted conditions of joint stress. Osteoarthr. Cartil. 2003, 11, 335–342. [Google Scholar] [CrossRef]
- Uebelhart, D.; Thonar, E.; Zhang, J.; Williams, J.M. Protective effect of exogenous chondroitin 4,6-sulfate in the acute degradation of articular cartilage in the rabbit. Osteoarthr. Cartil. 1998, 6, 6–13. [Google Scholar] [CrossRef]
- Sechriest, V.F.; Miao, Y.J.; Niyibizi, C.; Westerhausen-Larson, A.; Matthew, H.W.; Evans, C.H.; Fuh, F.H.; Suh, J.K. GAG-augmented polysaccharide hydrogel: A novel biocompatible and biodegradable material to support chondrogenesis. J. Biomed. Mater. Res. 2000, 49, 534–541. [Google Scholar] [CrossRef]
- Camper, L.; Hellman, U.; Lundgren-Akerlund, E. Isolation, cloning and sequence analysis of the integrin subunit α10, a β1-assocaited collagen binding integrin expressed on chondrocytes. J. Biol. Chem. 1998, 273, 20383–20389. [Google Scholar] [CrossRef] [PubMed]
- Eyre, D.R.; Weis, M.A.; Wu, J.J. Articular cartilage collagen: An irreplaceable framework? Eur. Cells Mater. 2006, 12, 57–63. [Google Scholar] [CrossRef]
- Wachsmuth, L.; Soder, S.; Fan, Z.; Finger, F; Aigner, T. Immunolocalization of matrix proteins in different cartilage subtypes. Histo. Histopathol. 2006, 21, 477–485. [Google Scholar]
- Billinghurst, R.C.; Dahlberg, L.; Ionescu, M.; Reiner, A.; Bourne, R.; Rorabeck, C.; Mitchell, P.; Hambor, J.; Diekmann, O.; Tschesche, H.; et al. Enhanced cleavage of type II collagen by collagenases in osteoarthritic articular cartilage. J. Clin. Investig. 1997, 99, 1534–1545. [Google Scholar] [CrossRef] [PubMed]
- Nehrer, S.; Breinan, H.A.; Ramappa, A.; Shortkroff, S.; Young, G.; Minas, T.; Sledge, C.B.; Yannas, I.V.; Spector, M. Canine chondrocytes seeded in type I and type II collagen implants investigated in vitro. J. Biomed. Mater. Res. 1997, 38, 95–104. [Google Scholar] [CrossRef]
- Bosnakovski, D.; Mizuno, M.; Kim, G.; Takagi, S.; Okumura, M.; Fujinaga, T. Chondrogenic differentiation of bovine bone marrow mesenchymal stem cells (MSCs) in different hydrogels: Influence of collagen type II extracellular matrix on MSC chondrogenesis. Biotechnol. Bioeng. 2006, 93, 1152–1163. [Google Scholar] [CrossRef] [PubMed]
- Hertz, H. Uber die beruhring fester elastischer korper. Reine Angew. Math. 1882, 92, 156–171. [Google Scholar]
- Sneddon, I.N. The relation between load and penetration in the axisymmetric boussinesq problem for a punch of arbitrary profile. Int. J. Eng. Sci. 1965, 3, 47–57. [Google Scholar] [CrossRef]
- Matsui, Y.; Sugiyama, K.; Kamei, M.; Takahashi, T.; Suzuki, T.; Katagata, Y. Ito Extraction of passion fruit (Passiflora edulis) seed containing high amounts of picetannol inhibits melanogenesis and promotes collagen synthesis. J. Agric. Food Chem. 2010, 58, 11112–11118. [Google Scholar] [CrossRef] [PubMed]
- Vesentini, S.; Redaelli, A.; Gautieri, A. Nanomechanics of collagen microfibrils. Muscle Ligaments Tendons J. 2013, 3, 23–34. [Google Scholar] [CrossRef] [PubMed]
- Zandomeneghi, G.; Krebs, M.R.H.; McCammon, M.G.; Fändrich, M. FTIR reveals structural differences between native β-sheet proteins and amyloid fibers. Protein Sci. 2004, 13, 3314–3321. [Google Scholar] [CrossRef] [PubMed]
- Susi, H.; Byler, D.M. Fourier transform infrared study of proteins with parallel β-chains. Arch. Biochem. Biophys. 1987, 258, 465–469. [Google Scholar] [CrossRef]
- Khodzhaeva, V.L. Study of liquid crystalline state of p-n-alkyloxybenzoic acids according to their IR absorption spectra. Bull. Acad. Sci. USSR 1969, 18, 2257–2262. [Google Scholar] [CrossRef]
- Konshina, E.A.; Vangonen, A.I. Features of the vibrational spectra of diamond-like and polymer-like a C:H films. Semiconductors 2005, 39, 585–590. [Google Scholar] [CrossRef]
- Parkih, S.J.; Kubicki, J.D.; Jonsson, C.M.; Jonsson, C.L.; Hazen, R.M.; Sverjensky, D.A.; Sparks, D.L. Evaluating glutamate and aspartate binding mechanisms to rutile (α-TiO2) via ATR-FTIR spectroscopy and quantum chemical calculations. Langmuir 2011, 27, 1778–1787. [Google Scholar] [CrossRef] [PubMed]
- Belbachir, K.; Noreen, R.; Gouspillou, G.; Petibois, C. Collagen types analysis and differentiation by FTIR spectroscopy. Anal. Bioanal. Chem. 2009, 395, 829–837. [Google Scholar] [CrossRef] [PubMed]
- Vitagliano, L.; Berisio, R.; Mazzarella, L.; Zagari, A. Structural basis of collagen stabilization induced by proline hydroxylation. Biopolymers 2001, 58, 459–464. [Google Scholar] [CrossRef]
- Sedaghat, S. Synthesis and characterization of new biocompatible copolymer: Chitosan-graft polyaniline. Int. Nano Lett. 2014, 4. [Google Scholar] [CrossRef]
- Mucha, M.; Piekielna, J.; Wieczorek, A. Characterization and morphology of biodegradable chitosan/synthetic polymer blends. Macromol. Symp. 1999, 144, 391–412. [Google Scholar] [CrossRef]
- Kolhe, P.; Kannan, R.M. Improvement in ductility of chitosan through blending and copolymerization with PEG: FTIR investigation of molecular interactions. Biomacromolecules 2003, 4, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.L.; Ni, H.C.; Wang, L.F.; Chen, J.S. Biodegradable amphiphilic copolymers based on poly(ε-caprolactone)-graft chondroitin sulfate as drug carriers. Biomacromolecules 2009, 9, 2477–2457. [Google Scholar]
- Hanifi, A.; McCarthy, H.; Roberts, S.; Pleshko, N. Fourier transform infrared imaging and infrared fiber optic probe spectroscopy identify collagen type in connective tissues. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Gargiulo, B.J.; Cragg, P.; Richardson, J.B.; Ashton, B.A.; Johnson, W.E.B. Phenotypic modulation of human articular chondrocytes by bistratene A. Eur. Cells. Mater. 2002, 3, 9–18. [Google Scholar]
- Joyce, M.E.; Roberts, A.B.; Sporn, M.B.; Bolander, M.E. Transforming Growth Factor-beta and the Initiation of Chondrogenesis and Osteogenesis in the Rat Femur. J. Cell Biol. 1990, 110, 2195–2207. [Google Scholar] [CrossRef] [PubMed]
- Kadler, K.E.; Baldock, C.; Bella, J.; Boot-Handford, R.P. Collagens at a glance. J. Cell Sci. 2007, 120, 1955–1958. [Google Scholar] [CrossRef] [PubMed]
- Orbach, R.; Adler-Abramovich, L.; Zigerson, S.; Mironi-Harpaz, I.; Seliktar, D.; Gazit, E. Self-assembled Fmoc-peptides as a platform for the formation of nanostructures and hydrogels. Biomacromolecules 2009, 10, 2646–2651. [Google Scholar] [CrossRef] [PubMed]
- Javid, N.; Roy, S.; Zelzer, M.; Yang, Z.; Sefcik, J.; Ulijn, R.V. Cooperative self-assembly of peptide gelators and proteins. Biomacromolecules 2013, 14, 4368–4376. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, M.; Sheshadri, B.S.; Vekatappa, M.P. Interactions of proteins with detergents: Binding of cationic detergents with lysozyme. J. Biosci. 1986, 10, 359–371. [Google Scholar] [CrossRef]
- Nakayaka, K.; Ushiwata, A.; Nakamura, Y. Interaction between proteins and detergents which contain a hydrocarbon chain longer than 16 carbon atoms. III. Competitive inhibition of trypsin by cetyldimethylbenzylammonium chloride. Biochim. Biophys. Acta 1976, 439, 116–124. [Google Scholar] [CrossRef]
- Xie, Y.; Wang, X.; Huang, R.; Qi, W.; Wang, Y.; Su, R.; He, Z. Electrostatic and aromatic interaction-directed supramolecular self-assembly of a designed Fmoc-tripeptide into helican ribbons. Langmuir 2015, 31, 2885–2894. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.; Ulijn, R.; Saiani, A. Self-assembly and gelation properties of glycine/leucine Fmoc-dipeptides. Eur. Phys. J. E 2013, 36, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Riley, G.P.; Harrall, R.L.; Constant, C.R.; Chard, M.D.; Cawston, T.E.; Hazleman, B.L. Glycosaminoglycans of human rotator cuff tendons: Changes with age and in chronic rotator cuff tendonitis. Ann. Rheum. Dis. 1994, 53, 367–376. [Google Scholar] [CrossRef] [PubMed]
- Zondlo, N.J. Aromatic-proline interactions: Electronically tunable CH/π interactions. Acc. Chem. Res. 2013, 46, 1039–1049. [Google Scholar] [CrossRef] [PubMed]
- Kar, K.; Wang, Y.H.; Brodsky, B. Sequence dependence of kinetics and morphology of collagen model peptide self-assembly into higher order structures. Protein Sci. 2008, 17, 1086–1095. [Google Scholar] [CrossRef] [PubMed]
- Gong, J.; Zhou, Z.; Hu, X.; Wong, M.K.; Du, Z. Self-assembled chitosan nanotemplates for biomineralization of controlled calcite nanoarchitectures. Appl. Mater. Inter. 2009, 1, 26–29. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, V.; Yuan, Y.; Rigney, D.A.; Puckett, A.D.; Ong, J.L.; Yang, Y.; Elder, S.H.; Bumgardner, J.D. Characterization of chitosan films and effects on fibroblast cell attachment and proliferation. J. Mater. Sci. Med. 2006, 17, 1373–1381. [Google Scholar] [CrossRef] [PubMed]
- Kuijpers, A.J.; Engbers, G.H.M.; Meyvis, T.K.L.; de Smedt, S.S.C.; Demeester, J.; Krijgsveld, J.; Zaat, S.A.J.; Dankert, J.; Feijen, J. Combined gelatin-chondroitin sulfate hydrogels for controlled release of cationic antibacterial proteins. Macromolecules 2000, 33, 3705–3713. [Google Scholar] [CrossRef]
- Oldber, A.; Ruoslahti, E. Interactions between chondroitin sulfate proteoglycan, fibronectin and collagen. J. Biol. Chem. 1982, 257, 4859–4863. [Google Scholar]
- Ma, M.; Kuang, Y.; Gao, Y.; Zhang, Y.; Gao, P.; Xu, B. Aromatic-aromatic interactions induce the self-assembly of pentapeptidic derivatives in water to form nanofibers and supramolecular hydrogels. J. Am. Chem. Soc. 2010, 132, 2719–2728. [Google Scholar] [CrossRef] [PubMed]
- Burley, S.K.; Petsko, G.A. Aromatic-aromatic interaction: A mechanism of protein structure stabilization. Science 1985, 229, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Hulmes, D.J.S. Building collagen molecules, fibrils and suprafibrillar structures. J. Struct. Biol. 2002, 137, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Fertala, A.; Sieron, A.L.; Hojima, Y.; Ganguly, A.; Prockop, D.J. Self-assembly into fibrils of collagen II by enzymic cleavage of recombinant procollagen II. J. Biol. Chem. 1994, 269, 11584–11589. [Google Scholar] [PubMed]
- Piez, K.A. Primary Structure. In Biochemistry of Collagen; Ramachandran, G.N., Reddi, A.H., Eds.; Plenum Press: New York, NY, USA, 1976; pp. 1–44. [Google Scholar]
- Jackson, M.; Choo, L.P.; Watson, P.H.; Halliday, W.C.; Mantsch, H.H. Beware of connective tissue proteins: Assignment and implications of collagen absorptions in infrared spectra of human tissues. Biochim. Biophys. Acta 1995, 1270, 1–6. [Google Scholar] [CrossRef]
- McClain, P.E.; Wiley, E.R. Differential scanning calorimeter studies of the thermal transitions of collagen. J. Biol. Chem. 1972, 247, 692–697. [Google Scholar] [PubMed]
- Nakmura, K.; Hatakeyama, T.; Hatakeyama, H. Studies on bound water of cellulose by differential scanning calorimetry. Text. Res. J. 1981, 51, 607–613. [Google Scholar] [CrossRef]
- Kittur, F.S.; Prashanth, K.V.H.; Sankar, K.U.; Tharanathan, R.N. Characterization of chitin, chitosan and their carboxymethyl derivatives by differential scanning calorimetry. Carbohydr. Polym. 2002, 49, 185–193. [Google Scholar] [CrossRef]
- Kacurakova, M.; Belton, P.S.; Wilson, R.H.; Hirsch, J.; Ebringerova, A. Hydration properties of xylan-type structures: An FTIR study of xylooligosaccharides. J. Sci. Food Agric. 1998, 77, 38–44. [Google Scholar] [CrossRef]
- Shanmugasundaram, N.; Ravichandran, P.; Reddy, P.N.; Ramamurthy, N.; Pal, S.; Rao, K.P. Collagen-chitosan polymeric scaffolds for in vitro culture of human epidermoid carcinoma cells. Biomaterials 2001, 22, 1943–1951. [Google Scholar] [CrossRef]
- Yang, H.Y.; Zhang, Q.Q. Preparation and characterization of collagen-GAG bioactive matrices for tissue engineering. J. Mater. Sci. Technol. 2001, 17, 495–500. [Google Scholar]
- Engler, A.J.; Sen, S.; Sweeney, H.L.; Discher, D.E. Matrix elasticity directs stem cell lineage specification. Cell 2006, 126, 677–689. [Google Scholar] [CrossRef] [PubMed]
- Xin, X.; Hussain, M.; Mao, J.J. Continuing differentiation of human mesenchymal stem cells and induced chondrogenic and osteogenic lineages in electrospun PLGA nanofiber scaffold. Biomaterials 2007, 28, 316–325. [Google Scholar] [CrossRef] [PubMed]
- Djokic, J.D.; Kojovic, A.; Stojanovic, D.; Marinkovic, A.; Vukovic, G.; Aleksic, R.; Uskokovic, P.S. Processing and Nanomechanical properties of chitosan/poly (ethylene oxide) blend films. J. Serb. Chem. Soc. 2012, 77, 1723–1733. [Google Scholar] [CrossRef]
- Palchesko, R.N.; Zhang, L.; Sun, Y.; Feinberg, A.W. Development of Polydimethylsiloxane substrates with tunable elastic modulus to study cell mechanobiology in muscle and nerve. PLoS ONE 2012, 7. [Google Scholar] [CrossRef] [PubMed]
- Candiello, J.; Balasubramani, M.; Schreiber, E.M.; Cole, G.J.; Mayer, U.; Halfter, W.; Lin, H. Biomechanical properties of native basement membranes. FEBS J. 2007, 274, 2897–2908. [Google Scholar] [CrossRef] [PubMed]
- Constantinides, G.; Kalcioglu, Z.I.; McFarland, M.; Smith, J.F.; Van Vliet, K.J. Probing mechanical properties of fully hydrated gels and biological tissues. J. Biomech. 2008, 41, 3285–3289. [Google Scholar] [CrossRef] [PubMed]
- Ko, C.S.; Wu, C.H.; Huang, H.H.; Chu, I.M. Genipin cross-linking of type II collagen-chondroitin sulfate-hyaluronan scaffold for articular cartilage therapy. J. Med. Biol. Eng. 2007, 27, 7–14. [Google Scholar]
- Nesti, L.J.; Li, W.J.; Shanti, R.M.; Jiang, Y.J.; Jackson, W.; Freedman, B.A.; Kuklo, T.R.; Giuliani, J.R.; Tuan, R.S. Intervertebral disc tissue engineering using a novel hyaluronic acid-nanofibrous scaffold (HANFS) amalgam. Tissue Eng. A 2008, 14, 1527–1537. [Google Scholar] [CrossRef] [PubMed]
- Cherry, R.S.; Papoutsakis, E.T. Modeling of contact-inhibited animal cell growth on flat surfaces and spheres. Biotechnol. Bioeng. 1989, 33, 300–305. [Google Scholar] [CrossRef] [PubMed]
- Goldring, M.B.; Tsuchimochi, K.; Ijuru, K. The Control of Chondrogenesis. J. Cell. Biochem. 2006, 97, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Ahrens, P.B.; Solurush, M.; Reiter, R.S. Stage-related capacity for limb chondrogenesis in cell culture. Dev. Biol. 1977, 60, 69–82. [Google Scholar] [CrossRef]
- Hassell, J.R.; Horigan, E.A. Chondrogenesis: A model developmental system for measuring teratogenic potential of compounds. Teratog. Carcinog. Mutagen. 1982, 2, 325–331. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Romanelli, S.M.; Knoll, G.A.; Santora, A.M.; Brown, A.M.; Banerjee, I.A. Comparison of Engineered Peptide-Glycosaminoglycan Microfibrous Hybrid Scaffolds for Potential Applications in Cartilage Tissue Regeneration. Fibers 2015, 3, 265-295. https://doi.org/10.3390/fib3030265
Romanelli SM, Knoll GA, Santora AM, Brown AM, Banerjee IA. Comparison of Engineered Peptide-Glycosaminoglycan Microfibrous Hybrid Scaffolds for Potential Applications in Cartilage Tissue Regeneration. Fibers. 2015; 3(3):265-295. https://doi.org/10.3390/fib3030265
Chicago/Turabian StyleRomanelli, Steven M., Grant A. Knoll, Anthony M. Santora, Alexandra M. Brown, and Ipsita A. Banerjee. 2015. "Comparison of Engineered Peptide-Glycosaminoglycan Microfibrous Hybrid Scaffolds for Potential Applications in Cartilage Tissue Regeneration" Fibers 3, no. 3: 265-295. https://doi.org/10.3390/fib3030265





