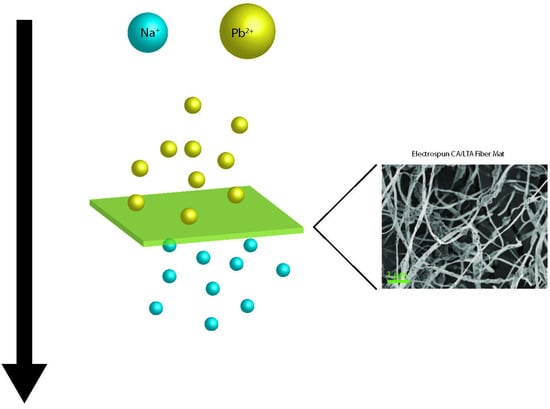
Electrospun Zeolite/Cellulose Acetate Fibers for Ion Exchange of Pb2+
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Experimental
3. Results
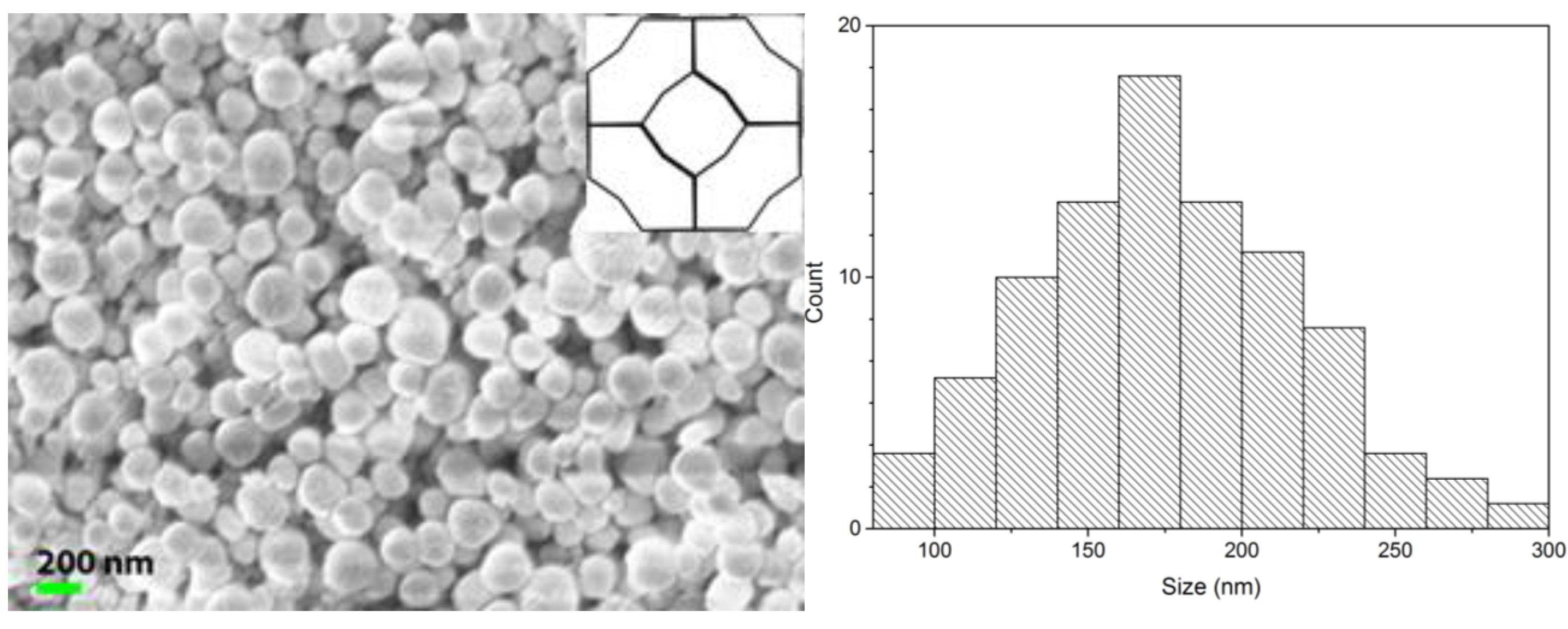
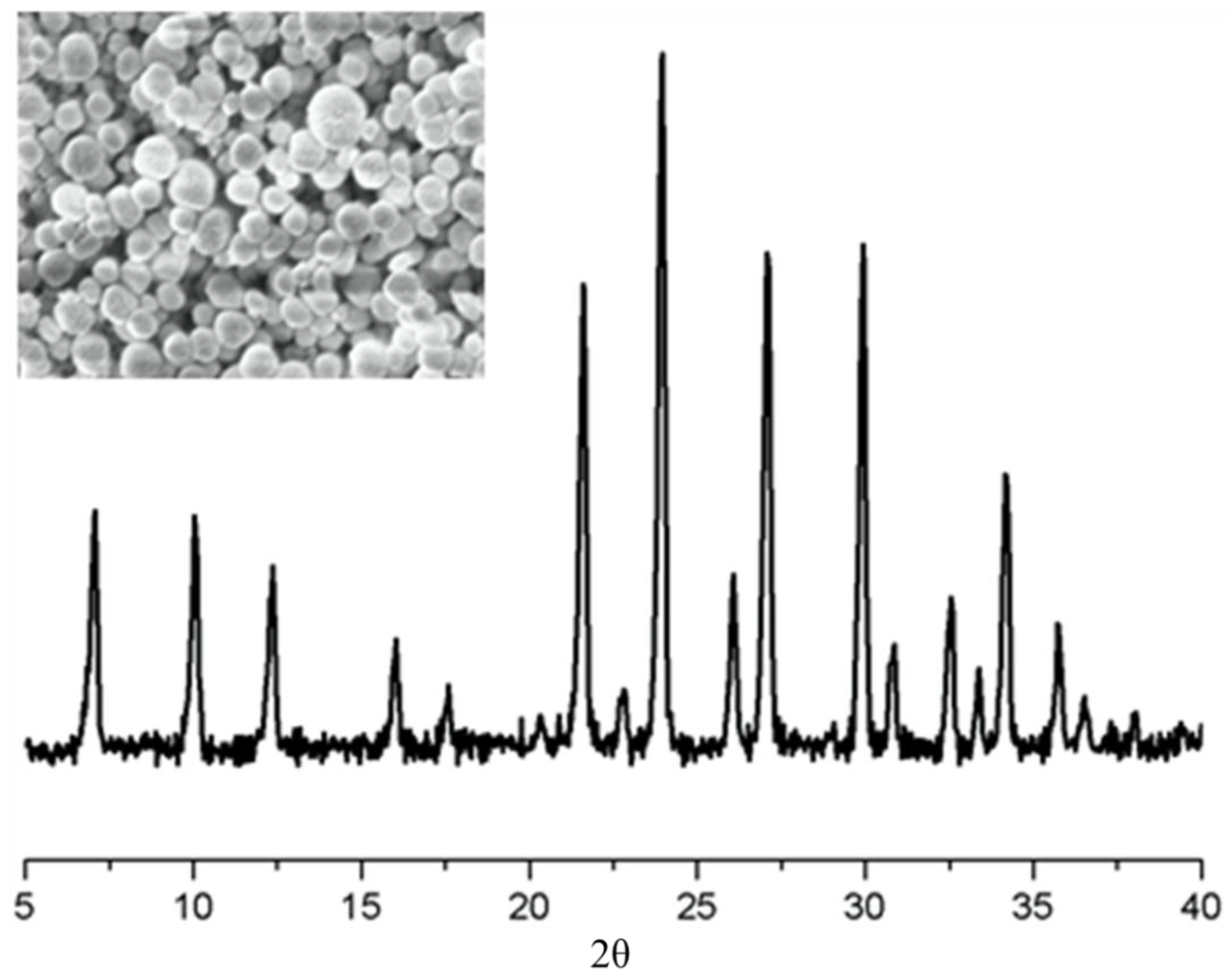
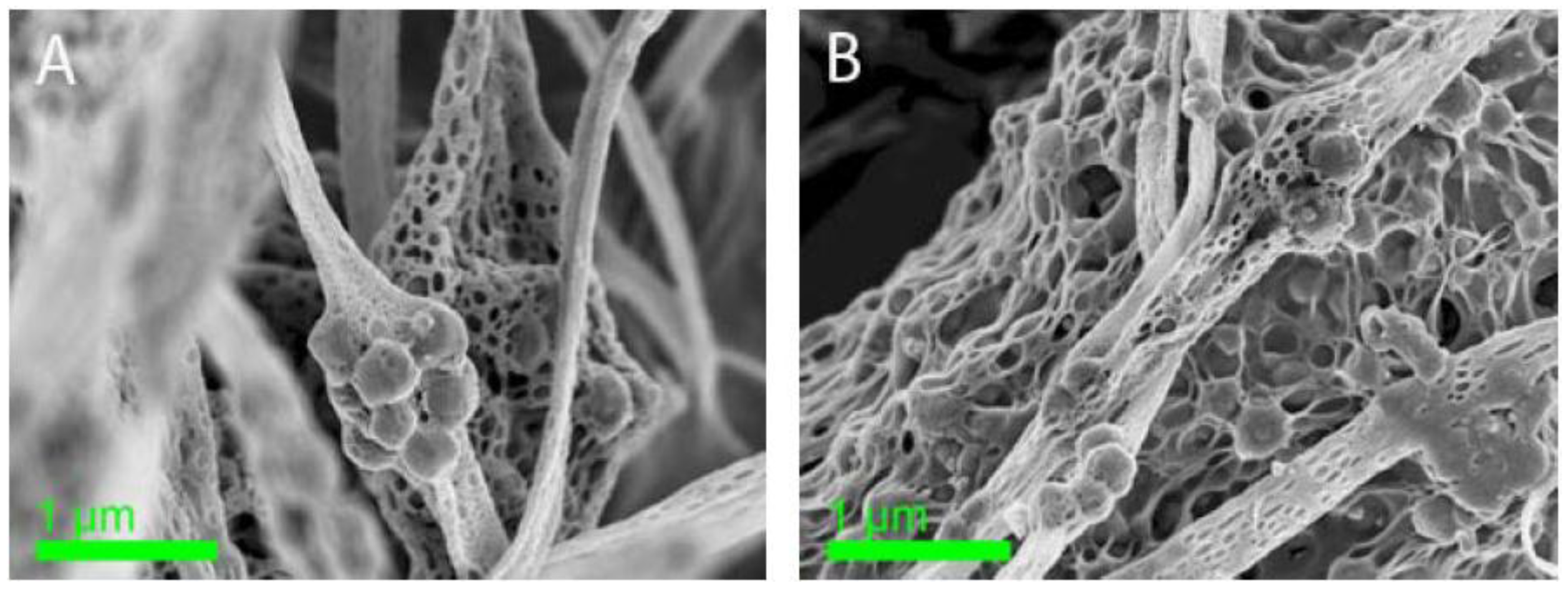
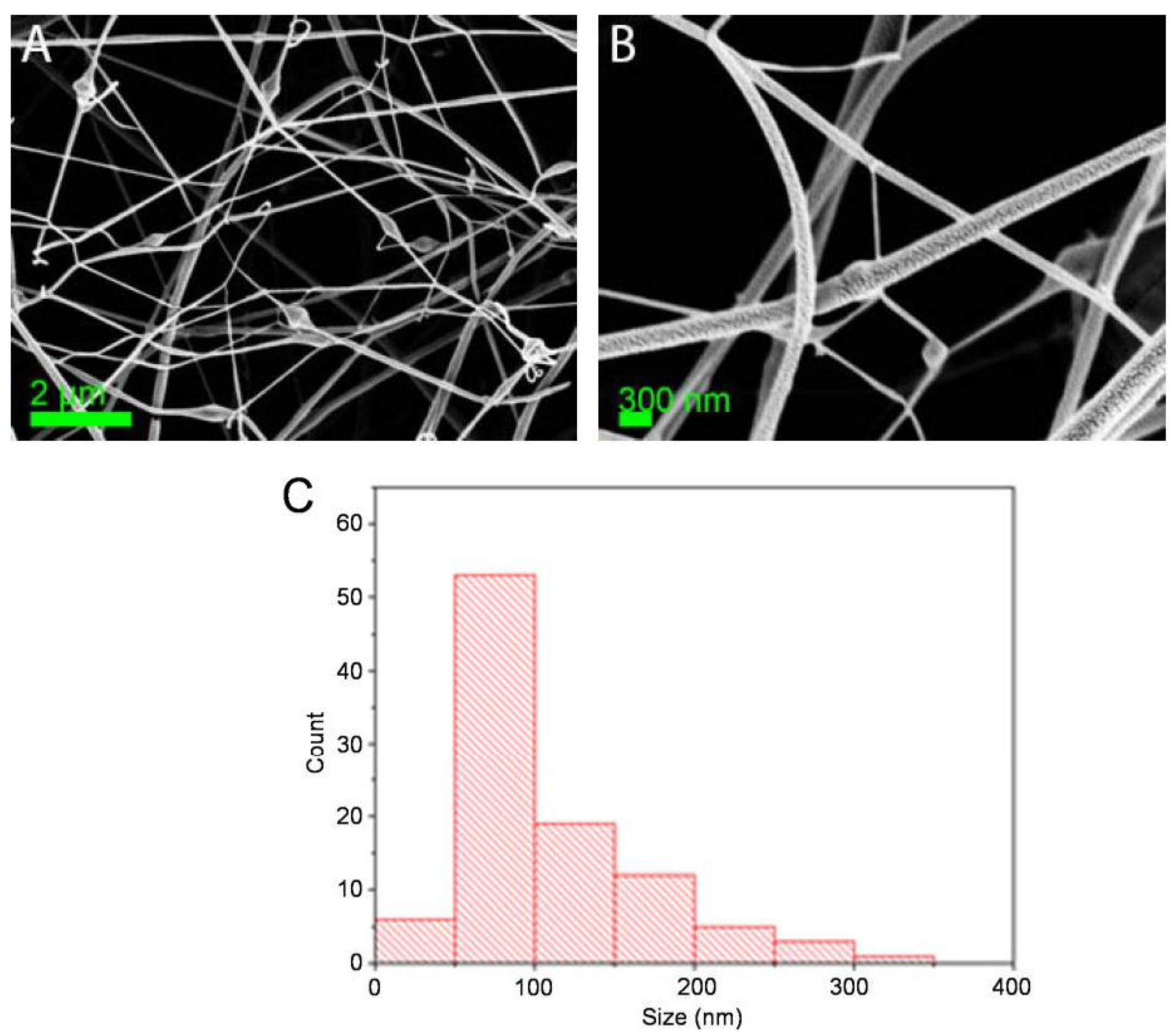
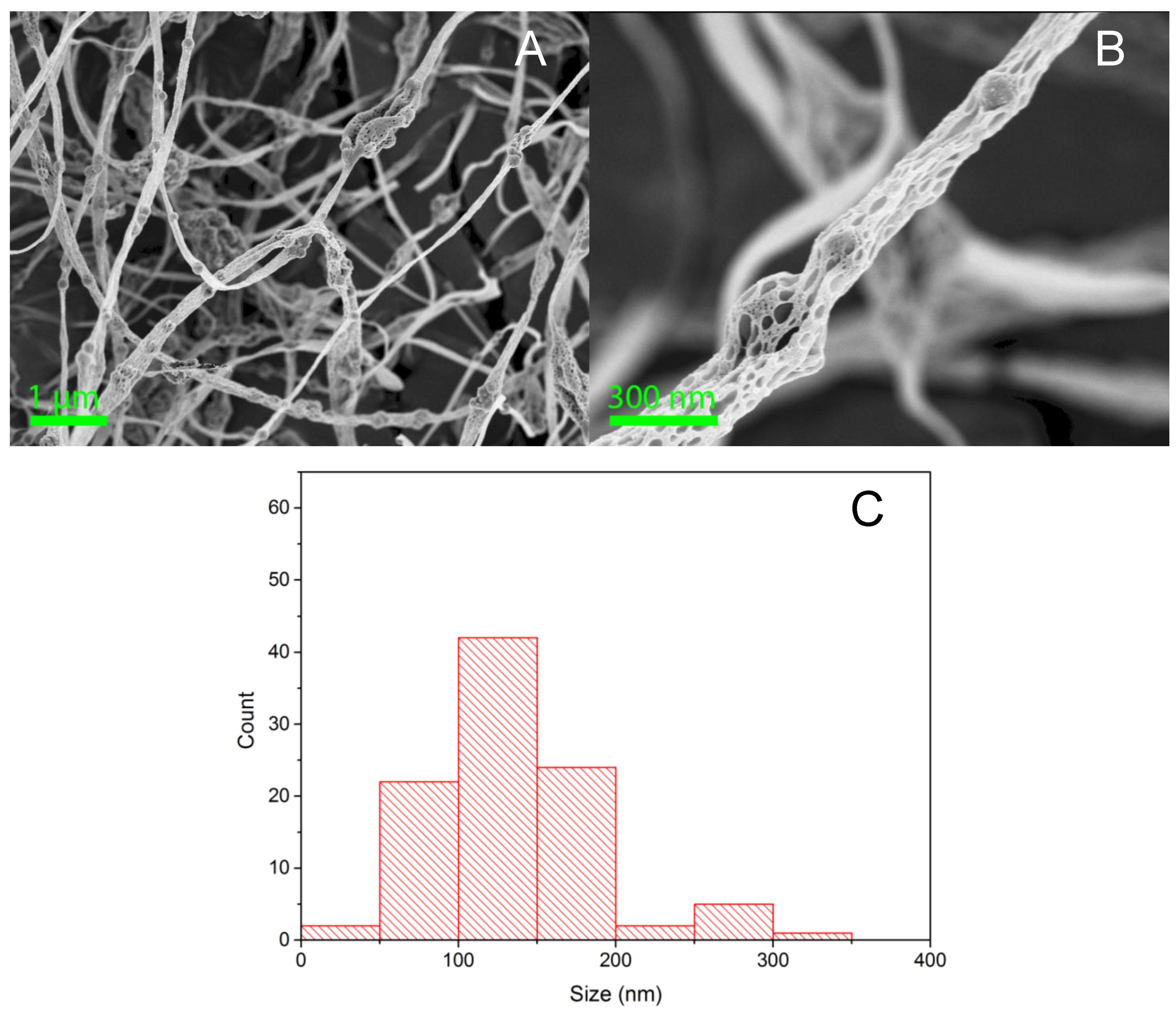
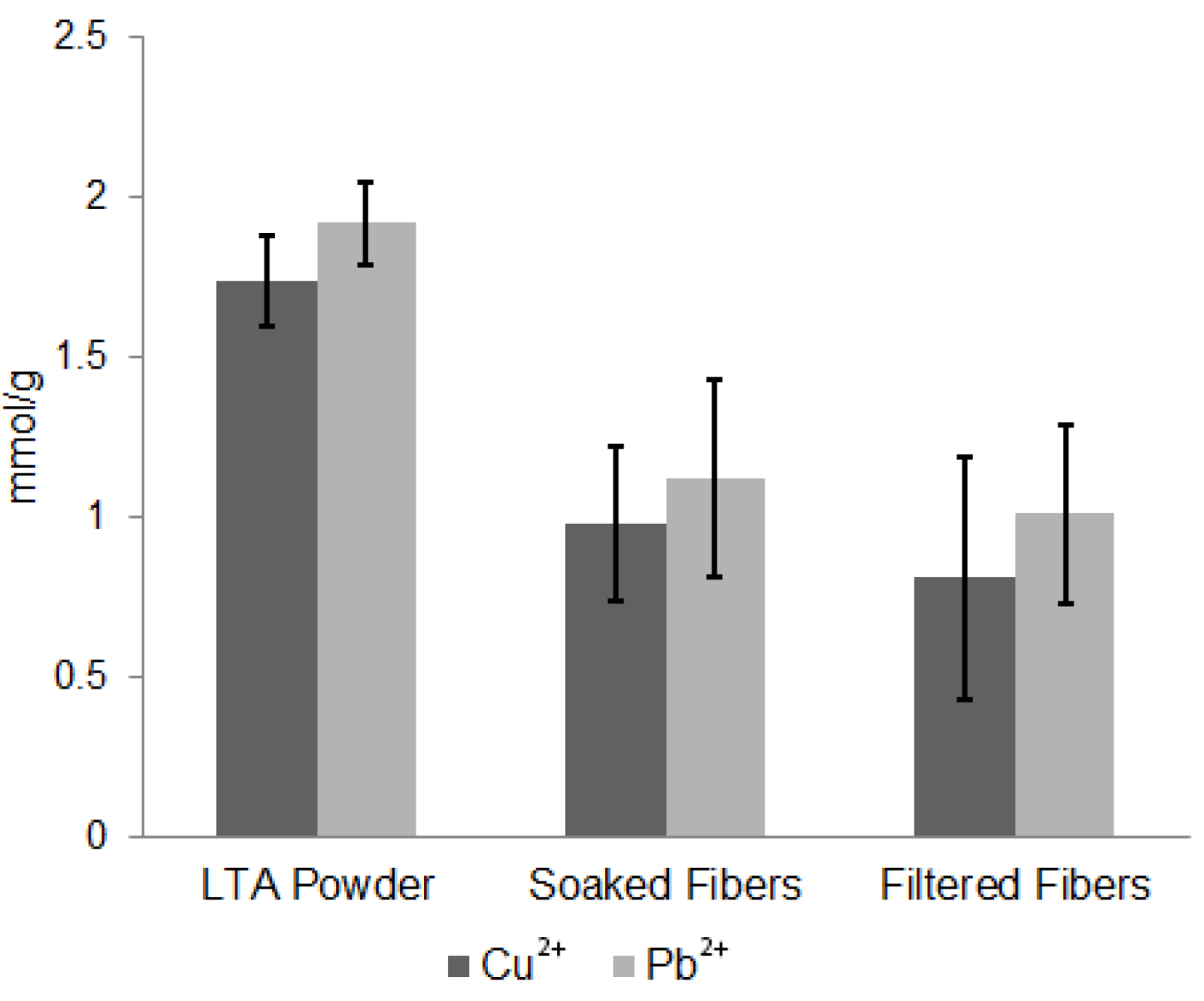
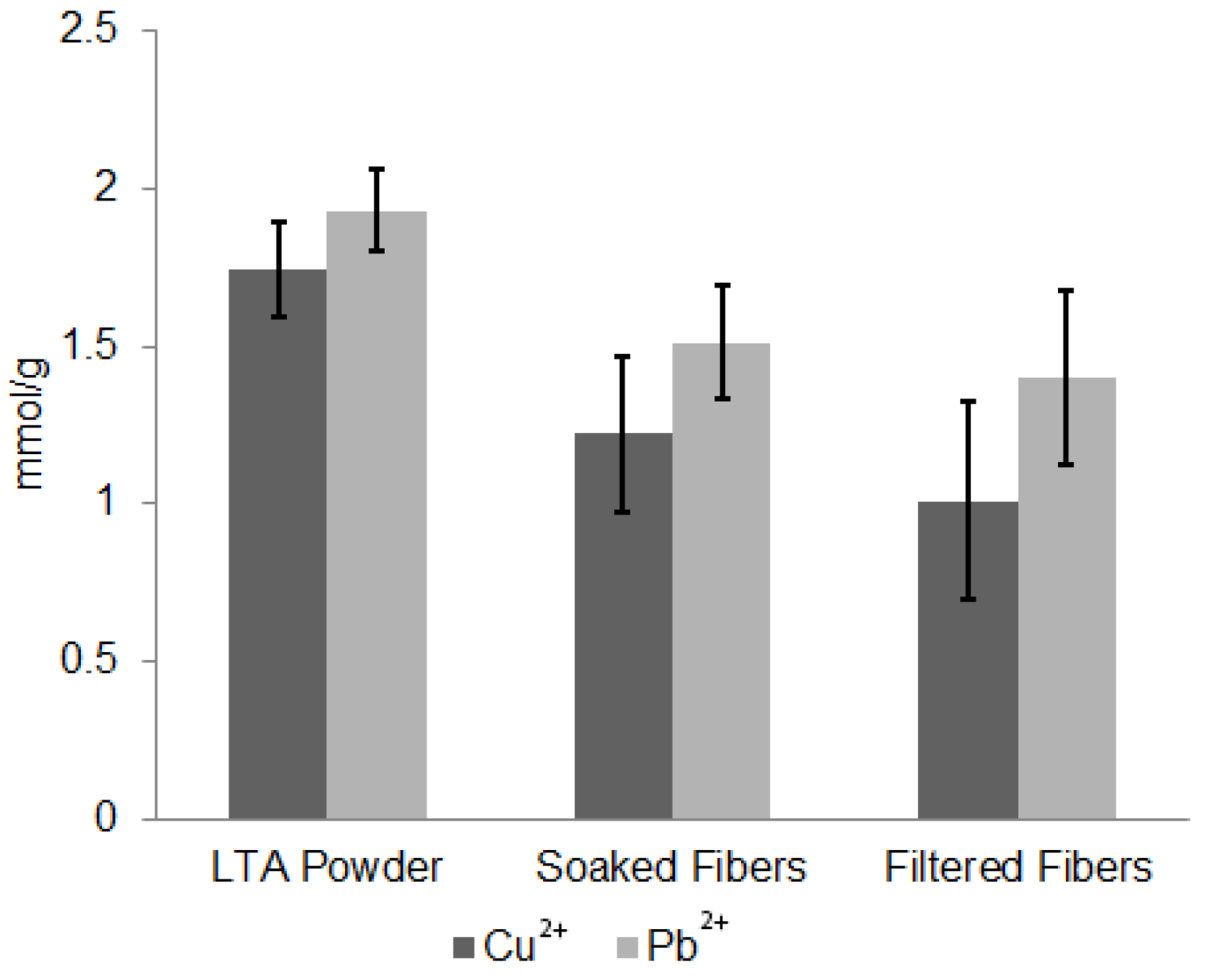
| Sample | Cu2+ (mmol/g) | Pb2+ (mmol/g) |
|---|---|---|
| LTA Powder | 1.74 ± 0.14 | 1.92 |
| Solid CA/LTA Fibers Soaked | 0.98 ± 0.24 | 1.12 ± 0.31 |
| Solid CA/LTA Fibers Filtered | 0.81 ± 0.38 | 1.01 ± 0.28 |
| Porous CA/LTA Fibers Soaked | 1.22 ± 0.25 | 1.51 ± 0.18 |
| Porous CA/LTA Fibers Filtered | 1.01 ± 0.31 | 1.4 ± 0.28 |
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Needleman, H. Lead poisoning. Annu. Rev. Med. 2004, 55, 209–222. [Google Scholar] [CrossRef]
- Goyer, R.A. Toxic and essential metal interactions. Annu. Rev. Nutr. 1997, 17, 37–50. [Google Scholar] [CrossRef]
- Sharma, A.K.; Lee, B.-K. Lead sorption onto acrylamide modified titanium nanocomposite from aqueous media. J. Environ. Manag. 2013, 128, 787–797. [Google Scholar] [CrossRef]
- Wiers, B.H.; Grosse, R.J.; Cilley, W.A. Divalent and trivalent ion exchange with zeolite A. Environ. Sci. Technol. 1982, 16, 617–624. [Google Scholar] [CrossRef]
- Fu, F.; Wang, Q. Removal of heavy metal ions from wastewaters: A review. J. Environ. Manag. 2011, 92, 407–418. [Google Scholar] [CrossRef]
- An, H.; Shin, C.; Chase, G.G. Ion exchanger using electrospun polystyrene nanofibers. J. Membrane Sci. 2006, 283, 84–87. [Google Scholar] [CrossRef]
- Li, H.; Wang, J.; Xu, J.; Meng, X.; Xu, B.; Yang, J.; Li, S.; Lu, J.; Zhang, Y.; He, X. Synthesis of zeolite NaA membranes with high performance and high reproducibility on coarse macroporous supports. J. Membrane Sci. 2013, 444, 513–522. [Google Scholar] [CrossRef]
- Alexandratos, S.D. Ion-Exchange resins: A retrospective from industrial and engineering chemistry research. Ind. Eng. Chem. Res. 2009, 48, 388–398. [Google Scholar] [CrossRef]
- Mauritz, K.A.; Moore, R.B. State of understanding of Nafion. Chem. Rev. 2004, 104, 4535–4585. [Google Scholar] [CrossRef]
- Abels, C.; Carstensen, F.; Wessling, M. Membrane processes in biorefinery applications. J. Membrane Sci. 2013, 444, 285–317. [Google Scholar] [CrossRef]
- Liu, B.S.; Tang, D.C.; Au, C.T. Fabrication of analcime zeolite fibers by hydrothermal synthesis. Microporous Mesoporous Mater. 2005, 86, 106–111. [Google Scholar] [CrossRef]
- Di, J.; Chen, H.; Wang, X.; Zhao, Y.; Jiang, L.; Yu, J.; Xu, R. Fabrication of zeolite hollow fibers by coaxial electrospinning. Chem. Mater. 2008, 20, 3543–3545. [Google Scholar] [CrossRef]
- Ke, C.; Yang, W.L.; Ni, Z.; Wang, Y.J.; Tang, Y.; Gu, Y.; Gao, Z. Electrophoretic assembly of nanozeolites: Zeolite coated fibers and hollow zeolite fibers. Chem. Commun. 2001, 2001, 783–784. [Google Scholar] [CrossRef]
- Reneker, D.H.; Fong, H.; Koombhongse, S.; Yarin, A.L. Bending instability of electrically charged liquid jets of polymer solutions in electrospinning. J. Appl. Phys. 2000, 87, 4531–4547. [Google Scholar] [CrossRef]
- Li, D.; Xia, Y. Electrospinning of nanofibers: Reinventing the wheel? Adv. Mater. 2004, 16, 1151–1170. [Google Scholar] [CrossRef]
- Liu, H.A.; Balkus, K.J., Jr. Novel delivery system for the bioregulatory agent nitric oxide. Chem. Mater. 2009, 21, 5032–5041. [Google Scholar] [CrossRef]
- Cucchi, I.; Spano, F.; Giovanella, U.; Catellani, M.; Varesano, A.; Calzaferri, G.; Botta, C. Fluorescent electrospun nanofibers embedding dye-loaded zeolite crystals. Small 2007, 3, 305–309. [Google Scholar] [CrossRef]
- Tian, Y.; Wu, M.; Liu, R.; Li, Y.; Wang, D.; Tan, J.; Wu, R.; Huang, Y. Electrospun membrane of cellulose acetate for heavy metal ion adsorption in water treatment. Carbohydr. Polym. 2011, 83, 743–748. [Google Scholar] [CrossRef]
- Xiang, T.; Zhang, Z.; Liu, H.; Yin, Z.; Li, L.; Liu, X.M. Characterization of cellulose-based electrospun nanofiber membrane and its adsorptive behaviours using Cu(II), Cd(II), Pb(II) as models. Sci. China Chem. 2013, 56, 567–575. [Google Scholar] [CrossRef]
- Zhou, W.; He, J.; Cui, S.; Gao, W. Preparation of electrospun silk fibroin/Cellulose Acetate blend nanofibers and their applications to heavy metal ions adsorption. Fibers Polym. 2011, 12, 431–437. [Google Scholar] [CrossRef]
- Mintova, S.; Olson, N.H.; Valtchev, V.; Bein, T. Mechanism of zeolite a nanocrystal growth from colloids at room temperature. Science 1999, 283, 958–960. [Google Scholar] [CrossRef]
- Vasimalai, N.; John, S.A. Spectrofluorimetric determination of picogram level Pb(II) using a dimercaptothiadiazole fluorophore. Spectrochim. Acta Part. A 2011, 82, 153–158. [Google Scholar] [CrossRef]
- Megelski, S.; Stephens, J.S.; Chase, D.B.; Rabolt, J.F. Micro- and nanostructured surface morphology on electrospun polymer fibers. Macromolecules 2002, 35, 8456–8466. [Google Scholar] [CrossRef]
- Cerjan-Stefanovič, S.; Kaštelan-Macan, M. The effect of temperature on the ion-exchange of Cd(II) and Pb(II) from waters. Fresenius Zeitschrift Anal. Chem. 1988, 332, 817–818. [Google Scholar] [CrossRef]
- Reid, C.E.; Breton, E.J. Water and ion flow across cellulosic membranes. J. Appl. Polym. Sci. 1959, 1, 133–143. [Google Scholar] [CrossRef]
- Wu, Y.; Li, N.B.; Luo, H.Q. Electrochemical determination of Pb(II) at a gold electrode modified with a self-assembled monolayer of 2,5-dimercapto-1,3,4-thiadiazole. Microchim. Acta 2008, 160, 185–190. [Google Scholar] [CrossRef]
- Da̧browski, A.; Hubicki, Z.; Podkościelny, P.; Robens, E. Selective removal of the heavy metal ions from waters and industrial wastewaters by ion-exchange method. Chemosphere 2004, 56, 91–106. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tran, D.N.; Marti, A.M.; Balkus, K.J., Jr. Electrospun Zeolite/Cellulose Acetate Fibers for Ion Exchange of Pb2+. Fibers 2014, 2, 308-317. https://doi.org/10.3390/fib2040308
Tran DN, Marti AM, Balkus KJ Jr. Electrospun Zeolite/Cellulose Acetate Fibers for Ion Exchange of Pb2+. Fibers. 2014; 2(4):308-317. https://doi.org/10.3390/fib2040308
Chicago/Turabian StyleTran, Daniel N., Anne M. Marti, and Kenneth J. Balkus, Jr. 2014. "Electrospun Zeolite/Cellulose Acetate Fibers for Ion Exchange of Pb2+" Fibers 2, no. 4: 308-317. https://doi.org/10.3390/fib2040308
APA StyleTran, D. N., Marti, A. M., & Balkus, K. J., Jr. (2014). Electrospun Zeolite/Cellulose Acetate Fibers for Ion Exchange of Pb2+. Fibers, 2(4), 308-317. https://doi.org/10.3390/fib2040308