Distribution of Al Element of Ti–6Al–4V Joints by Fiber Laser Welding
Abstract
:1. Introduction
2. Experimental
3. Results and Discussions
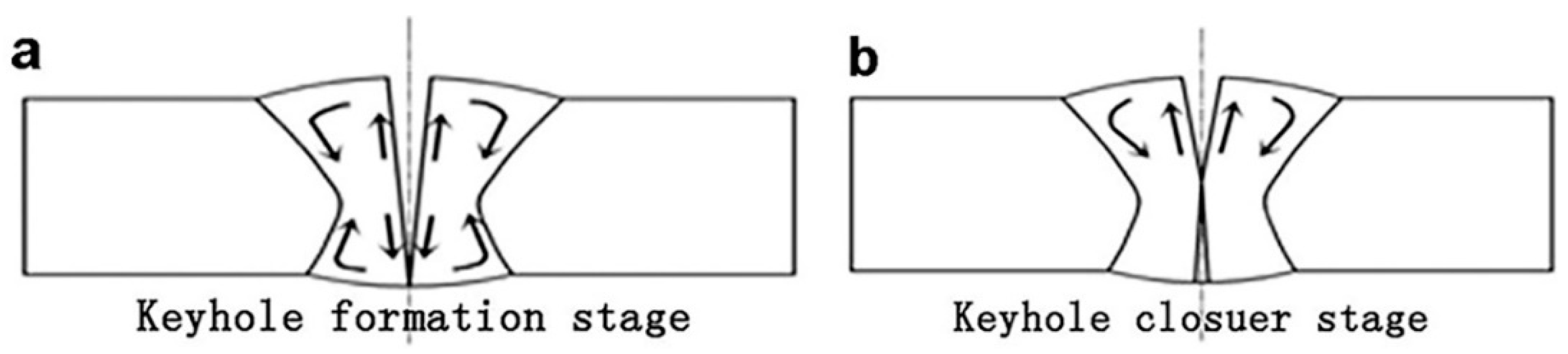
3.1. Morphology Analysis
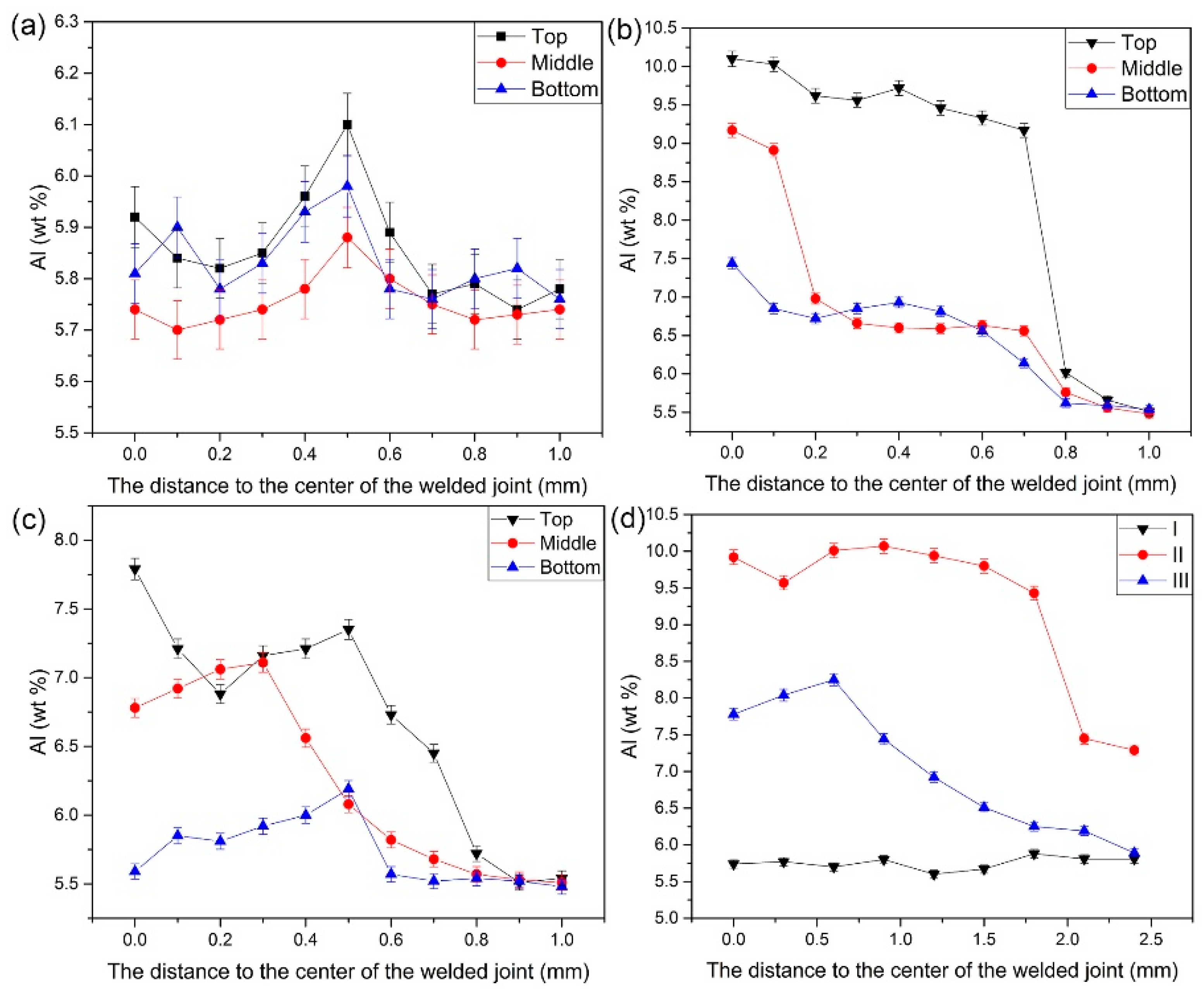
3.2. Al Solute Element Distribution
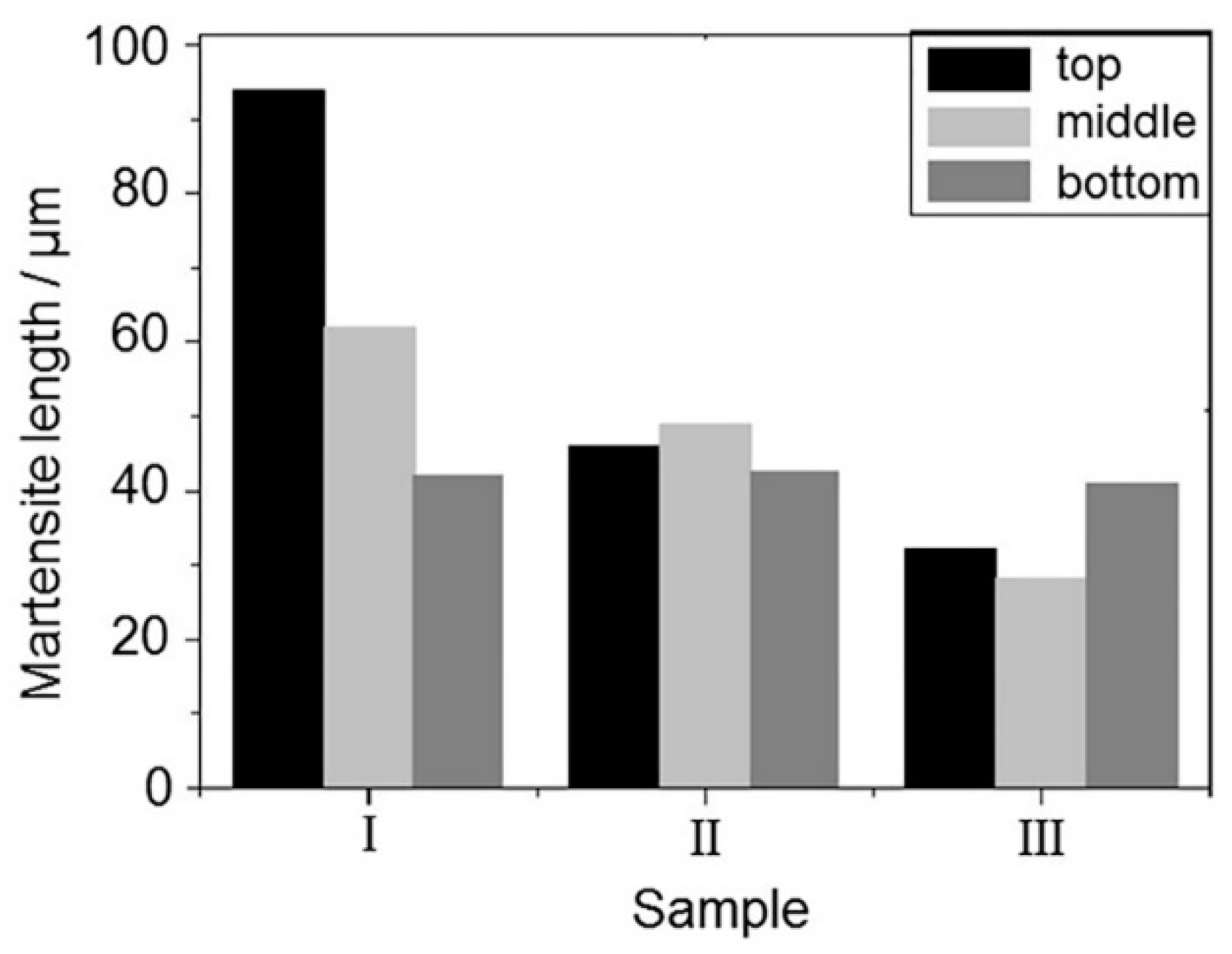
3.3. Microstructure
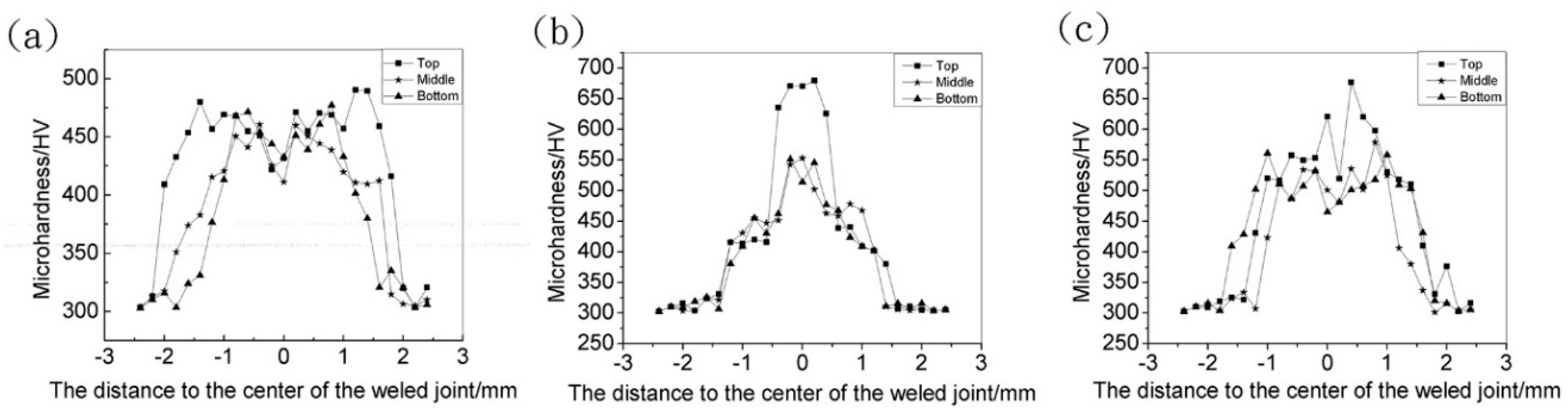
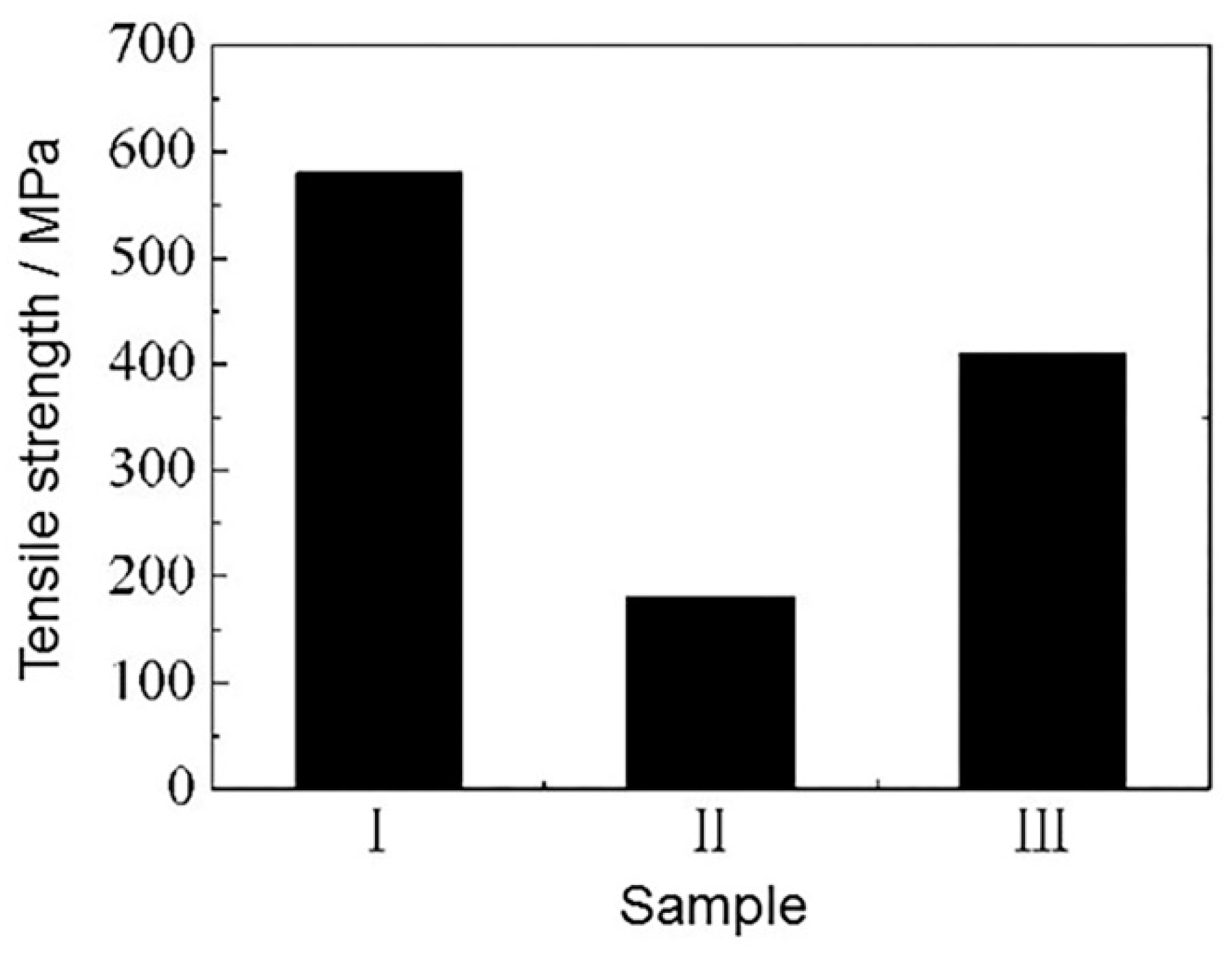
3.4. Mechanical Properties
4. Conclusions
- The distribution of solute elements in the laser welding seam is mainly determined by the flow of the molten pool and the self-diffusion. And the flow of molten pool has greater influence on the distribution of solute elements than the diffusion of the solute elements themselves. The addition of Si can significantly improve the fluidity of the molten pool, which makes the movement of the weld pool more intense, so that the distribution of the Al solute element in the weld is more uniform.
- In the study, the microstructure of the acicular martensite was explored, and the variation of the size was taken as a measurement of the nonuniform of the weld. The result showed that the grain size of weld with Al added was smaller than that without elemental added and with the smallest amount Al and Si added. The experimental results showed that the length of martensite in the top changed obviously and in the middle and bottom regions changed slightly.
- The addition of alloy powder not only changed the microhardness profiles of the weld, but also changed the distribution of the microhardness in different regions. Moreover, higher concentration of Al element may have resulted in higher microhardness, while the welded joint became more brittle and the tensile strength was lower. The addition of Si powder could avoid the decrease in joint tensile strength to some extent, but the strength was still lower than that of the welded joint without powder addition. Therefore, the tensile strength of sample III weld joint was the biggest in the case of powder addition, and its comprehensive mechanical properties were better.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Akman, E.; Demir, A.; Canel, T.; Sınmazçelik, T. Laser welding of Ti6Al4V titanium alloys. J. Mater. Process. Technol. 2009, 209, 3705–3713. [Google Scholar] [CrossRef]
- Bolzoni, L.; Ruiz-Navas, E.; Gordo, E. Feasibility study of the production of biomedical Ti–6Al–4V alloy by powder metallurgy. Mater. Sci. Eng. C 2015, 49, 400–407. [Google Scholar] [CrossRef] [PubMed]
- Kashaev, N.; Ventzke, V.; Fomichev, V.; Fomin, F.; Riekehr, S. Effect of Nd:YAG laser beam welding on weld morphology and mechanical properties of Ti–6Al–4V butt joints and T-joints. Opt. Lasers Eng. 2016, 86, 172–180. [Google Scholar] [CrossRef]
- Zhou, L.; Zhang, M.; Jin, X.; Zhang, H.; Mao, C. Study on the burning loss of magnesium in fiber laser welding of an Al-Mg alloy by optical emission spectroscopy. Int. J. Adv. Manuf. Technol. 2017, 88, 1373–1381. [Google Scholar] [CrossRef]
- Simonds, B.; Sowards, J.; Williams, P. Laser-induced fluorescence applied to laser welding of austenitic stainless steel for dilute alloying element detection. J. Phys. D Appl. Phys. 2017, 50. [Google Scholar] [CrossRef]
- Abe, Y.; Mizutani, M.; Kawahito, Y.; Katayama, S. Deep penetration welding with high power laser under vacuum. J. Laser Appl. 2010, 648–653. [Google Scholar] [CrossRef]
- Pastor, M.; Zhao, H.; Martukanitz, R.; Debroy, T. Porosity, underfill and magnesium loss during continuous wave Nd:YAG laser welding of thin plates of aluminum alloys 5182 and 5754. Weld. J. 1999, 78. [Google Scholar] [CrossRef]
- Fuerschbach, P.W.; Eisler, G.R. Effect of laser spot weld energy and duration on melting and absorption. Sci. Technol. Weld. Join. 2002, 7, 241–246. [Google Scholar] [CrossRef]
- Leo, P.; Renna, G.; Casalino, G.; Olabi, A.G. Effect of power distribution on the weld quality during hybrid laser welding of an Al–Mg alloy. Opt. Laser Technol. 2015, 73, 118–126. [Google Scholar] [CrossRef]
- Zhao, L.; Sugino, T.; Arakane, G.; Tsukamoto, S. Influence of welding parameters on distribution of wire feeding elements in CO2 laser GMA hybrid welding. Sci. Technol. Weld. Join. 2009, 14, 457–467. [Google Scholar] [CrossRef]
- Ju, H.; Lin, C.X.; Tian, Y.; Liu, Z.J.; Jiang, H.L.; Sun, D.P. Mechanical properties study of Fe-Mn-Si shape memory alloys welding seam formed by laser welding with filler powder. Materials 2018, 11, 1454. [Google Scholar] [CrossRef] [PubMed]
- Abderrazak, K.; Bannour, S.; Mhiri, H.; Lepalec, G.; Autric, M. Numerical and experimental study of molten pool formation during continuous laser welding of AZ91 magnesium alloy. Comput. Mater. Sci. 2009, 44, 858–866. [Google Scholar] [CrossRef]
- Suthakar, T.K.R.B.; Sankaranarayanasamy, K.; Naidu, B.G. Studies on weld shape formation and Marangoni convection in Nd:YAG laser welding. Int. J. Mechatron. Manuf. Syst. 2012, 5, 226–246. [Google Scholar] [CrossRef]
- Gao, Z.M.; Jiang, P.; Mi, G.Y.; Cao, L.C.; Liu, W. Investigation on the weld bead profile transformation with the keyhole and molten pool dynamic behavior simulation in high power laser welding. Int. J. Heat Mass Transf. 2018, 116, 1304–1313. [Google Scholar] [CrossRef]
- Peng, G.C.; Li, L.Q.; Xia, H.B.; Gong, J.F. Study on keyhole and melt flow behaviors of laser welding of aluminum under reduced ambient pressures. J. Phys. Conf. Ser. 2018, 1063, 012071. [Google Scholar] [CrossRef] [Green Version]
- Fotovvati, B.; Wayne, S.; Lewis, G.; Asadi, E. A review on melt-pool characteristics in laser welding of metals. Adv. Mater. Sci. Eng. 2018, 2018, 1–18. [Google Scholar] [CrossRef]
- Zou, J.; Wu, S.; He, Y.; Yang, W.; Xu, J.; Xiao, R. Distinct morphology of keyhole wall during high power fibre laser deep penetration welding. Sci. Technol. Weld. Join. 2015, 20, 655–658. [Google Scholar] [CrossRef]
- Zhang, G.; Wu, C.S.; Liu, Z. Experimental observation of both keyhole and its surrounding thermal field in plasma arc welding. Int. J. Heat Mass Transf. 2014, 70, 439–448. [Google Scholar] [CrossRef]
- Hanumanthu, R.; Stebe, K.J. Transient and persistent Marangoni–Bénard convection in the presence of surfactants. Colloids Surf. A Physicochem. Eng. Asp. 2011, 391, 51–61. [Google Scholar] [CrossRef]
- Wang, S.; Wu, X. Investigation on the microstructure and mechanical properties of Ti–6Al–4V alloy joints with electron beam welding. Mater. Des. 2012, 36, 663–670. [Google Scholar] [CrossRef]
- Zhang, Z.; Dong, S.; Wang, Y.; Xu, B.; Fang, J.; He, P. Microstructure characteristics of thick aluminum alloy plate joints welded by fiber laser. Mater. Des. 2015, 84, 173–177. [Google Scholar] [CrossRef]
- Guo, P.; Zhao, Y.; Zeng, W.; Hong, Q. The effect of microstructure on the mechanical properties of TC4-DT titanium alloys. Mater. Sci. Eng. A 2013, 563, 106–111. [Google Scholar] [CrossRef]
- Liang, S.X.; Yin, L.X.; Jiang, R.J.; Zhang, X.Y.; Ma, M.Z.; Liu, R.P. Strengthening mechanism of two-phase titanium alloys with basketweave microstructure. J. Alloy Compd. 2014, 603, 42–47. [Google Scholar] [CrossRef]
- Cai, Z.; Wang, R.; Zhang, C.; Peng, C.; Wang, L. Microstructure and properties of Al/Sip composites for thermal management applications. J. Mater. Sci. Mater. Electron. 2015, 26, 4234–4240. [Google Scholar] [CrossRef]




| Al | V | Fe | C | N | H | O | Ti |
|---|---|---|---|---|---|---|---|
| 5.50–6.80 | 3.50–4.50 | ≤0.30 | ≤0.10 | ≤0.05 | ≤0.015 | ≤0.20 | Bal. |
| Sample I | Sample II | Sample III |
|---|---|---|
| Without powder | 100% Al + 0% Si | 90% Al + 10% Si |
| Power (kW) | Welding Speed (m/min) | Defocusing Amount (mm) | Gas Flow (m3/h) |
|---|---|---|---|
| 2.5 | 2.5 | 0 | 2 |
| Sample | Top | Middle | Bottom |
|---|---|---|---|
| II | 0.61 | 2.66 | 1.05 |
| III | 0.93 | 0.65 | 0.61 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, J.; Hu, R.; Pang, S.; Huang, A. Distribution of Al Element of Ti–6Al–4V Joints by Fiber Laser Welding. Coatings 2019, 9, 566. https://doi.org/10.3390/coatings9090566
Zhang J, Hu R, Pang S, Huang A. Distribution of Al Element of Ti–6Al–4V Joints by Fiber Laser Welding. Coatings. 2019; 9(9):566. https://doi.org/10.3390/coatings9090566
Chicago/Turabian StyleZhang, Jiajia, Renzhi Hu, Shengyong Pang, and Anguo Huang. 2019. "Distribution of Al Element of Ti–6Al–4V Joints by Fiber Laser Welding" Coatings 9, no. 9: 566. https://doi.org/10.3390/coatings9090566





