Hydrophobic Modification of Magnesium Hydroxide Coating Deposited Cathodically on Magnesium Alloy and its Corrosion Protection
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Effects of Modifying Parameters
3.1.1. Concentration of stearic acid
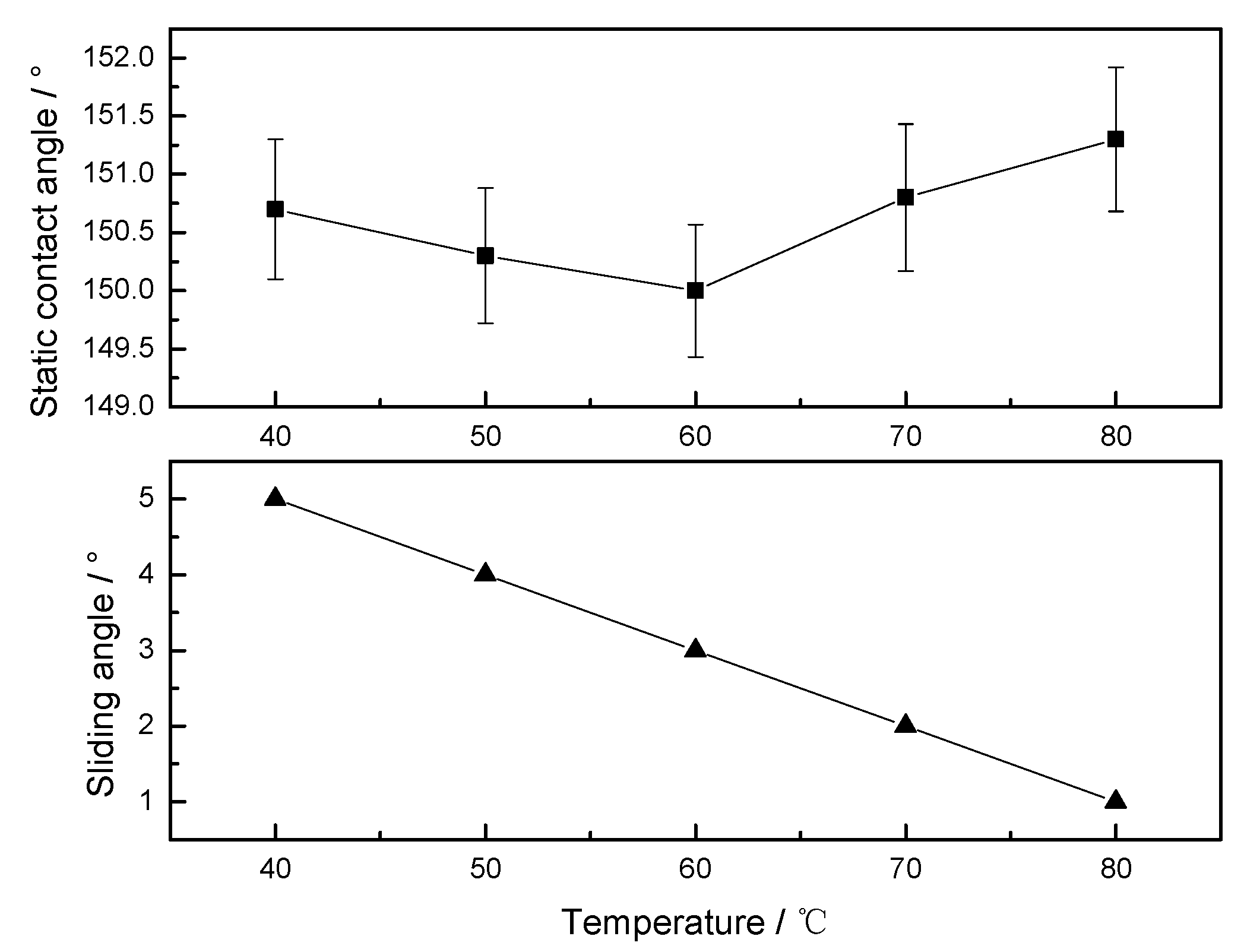
3.1.2. Solution Temperature
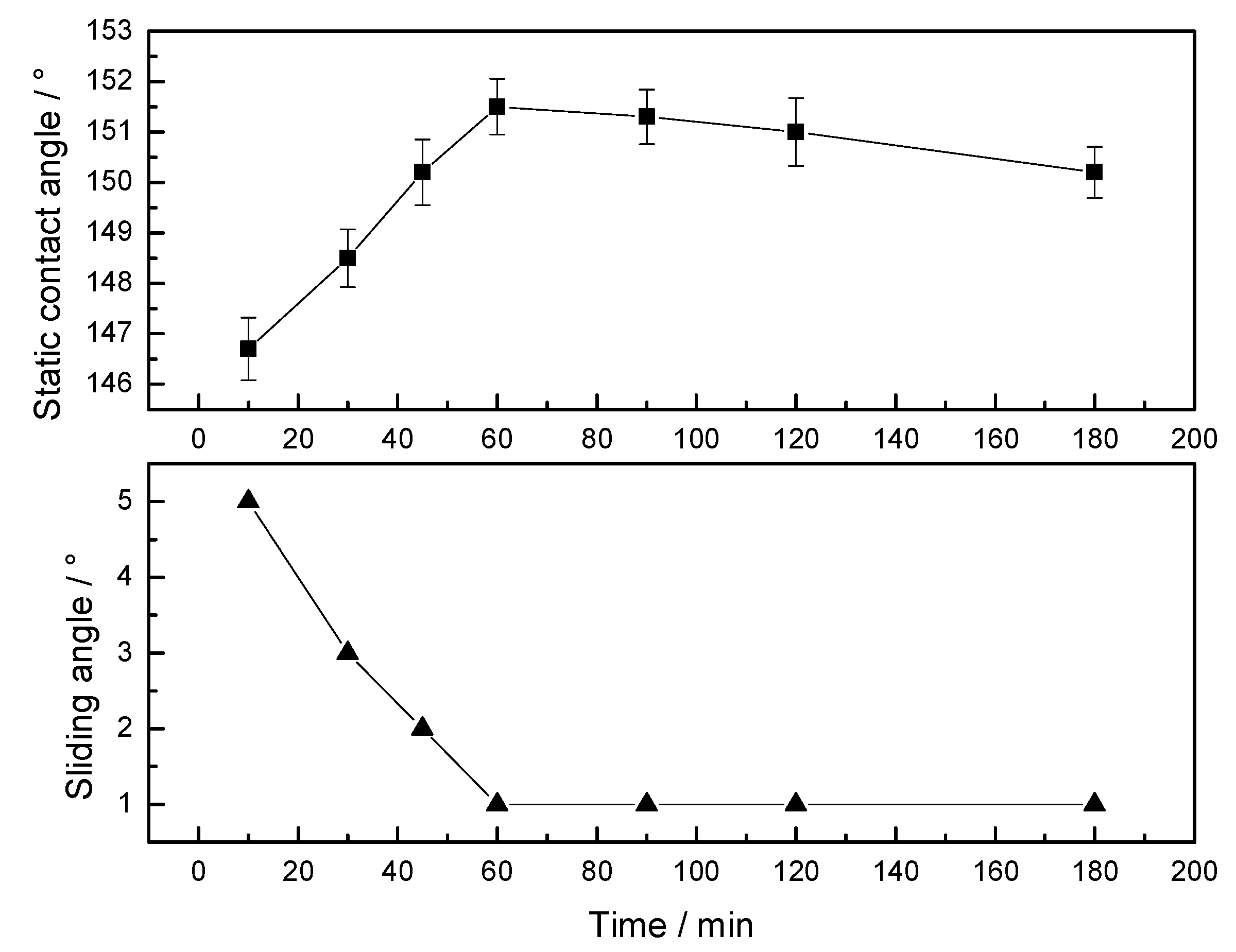
3.1.3. Treatment Time
3.2. Corrosion Protection
3.2.1. Droplet Test
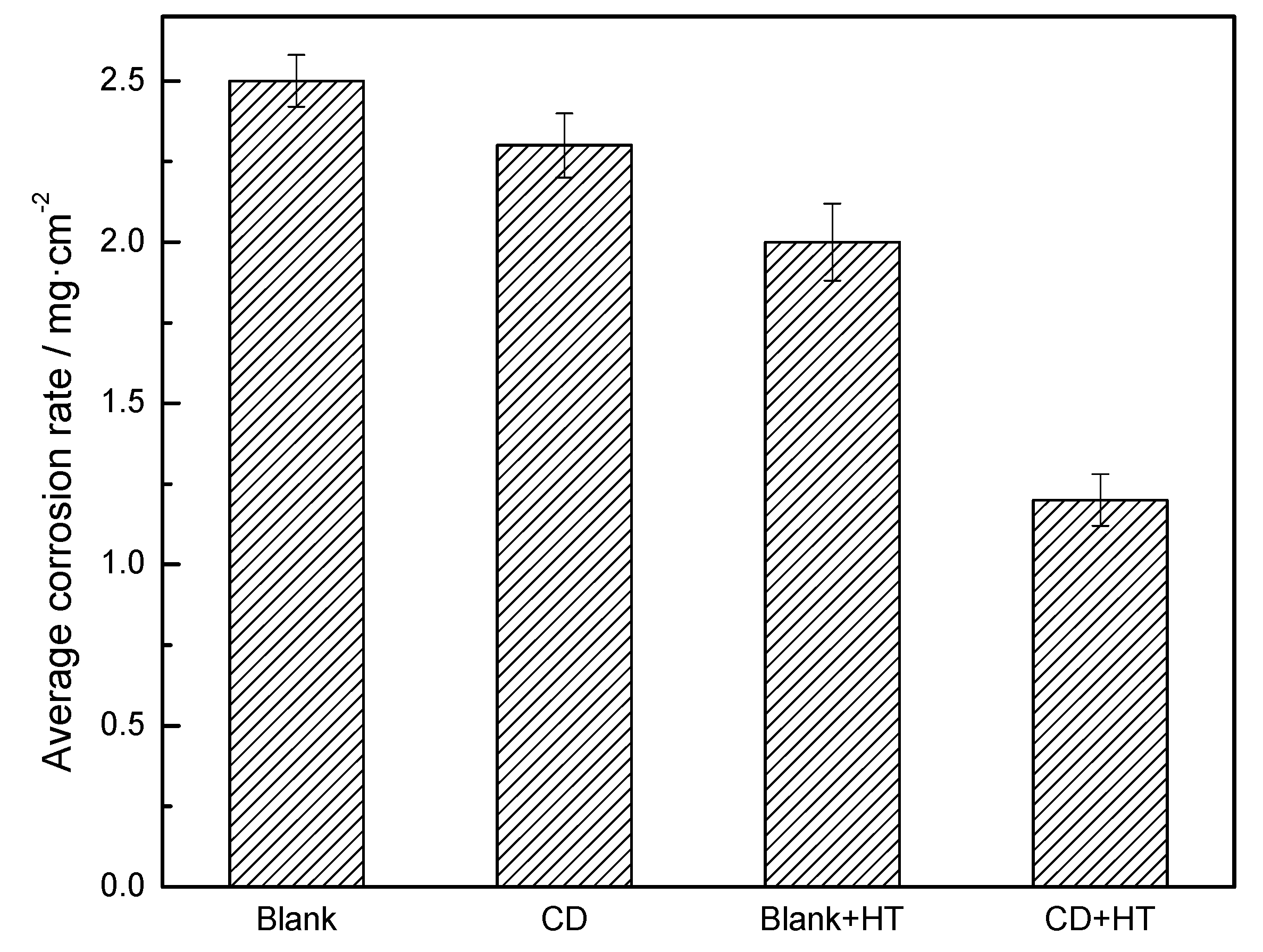
3.2.2. Total Immersion Corrosion Test
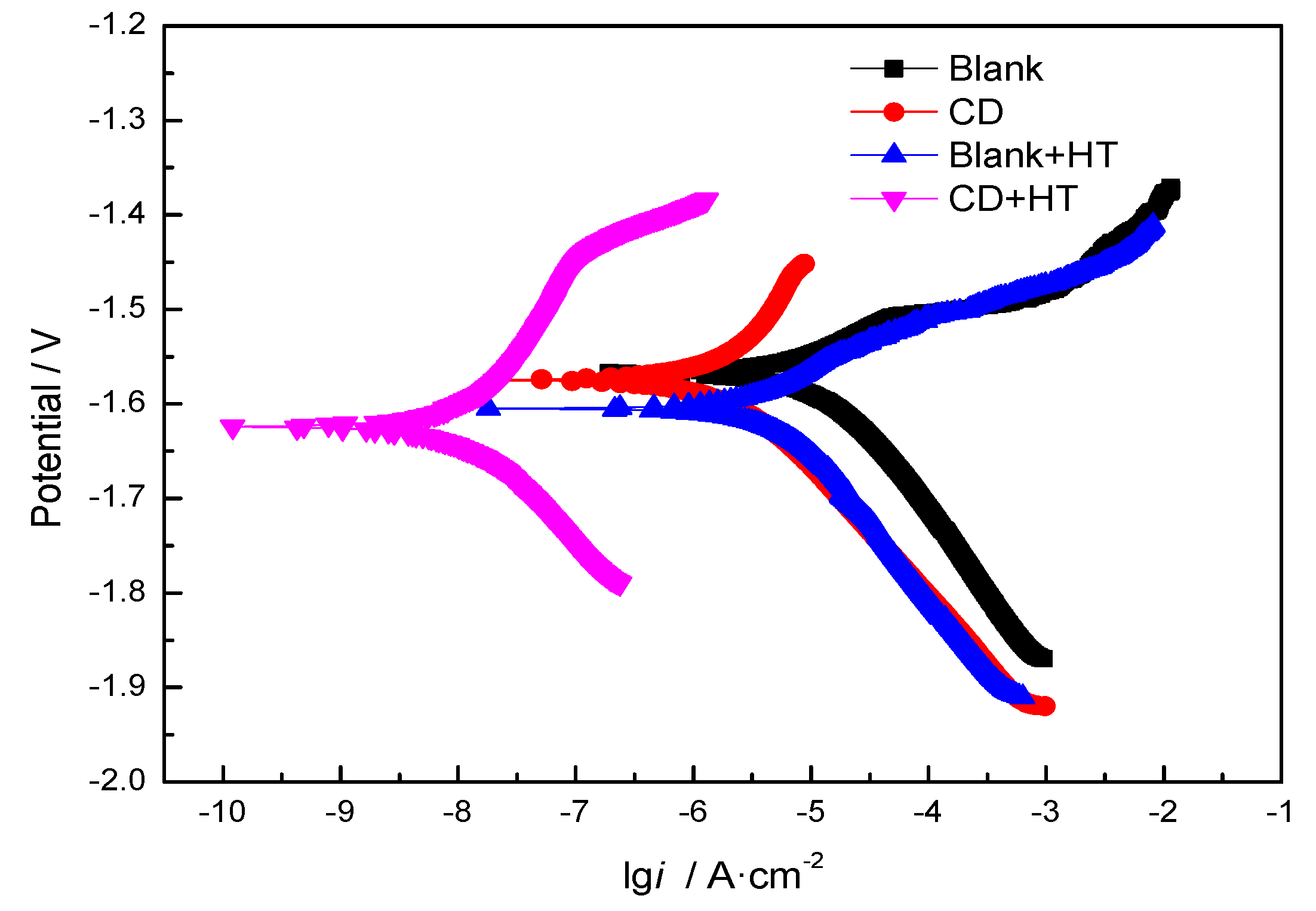
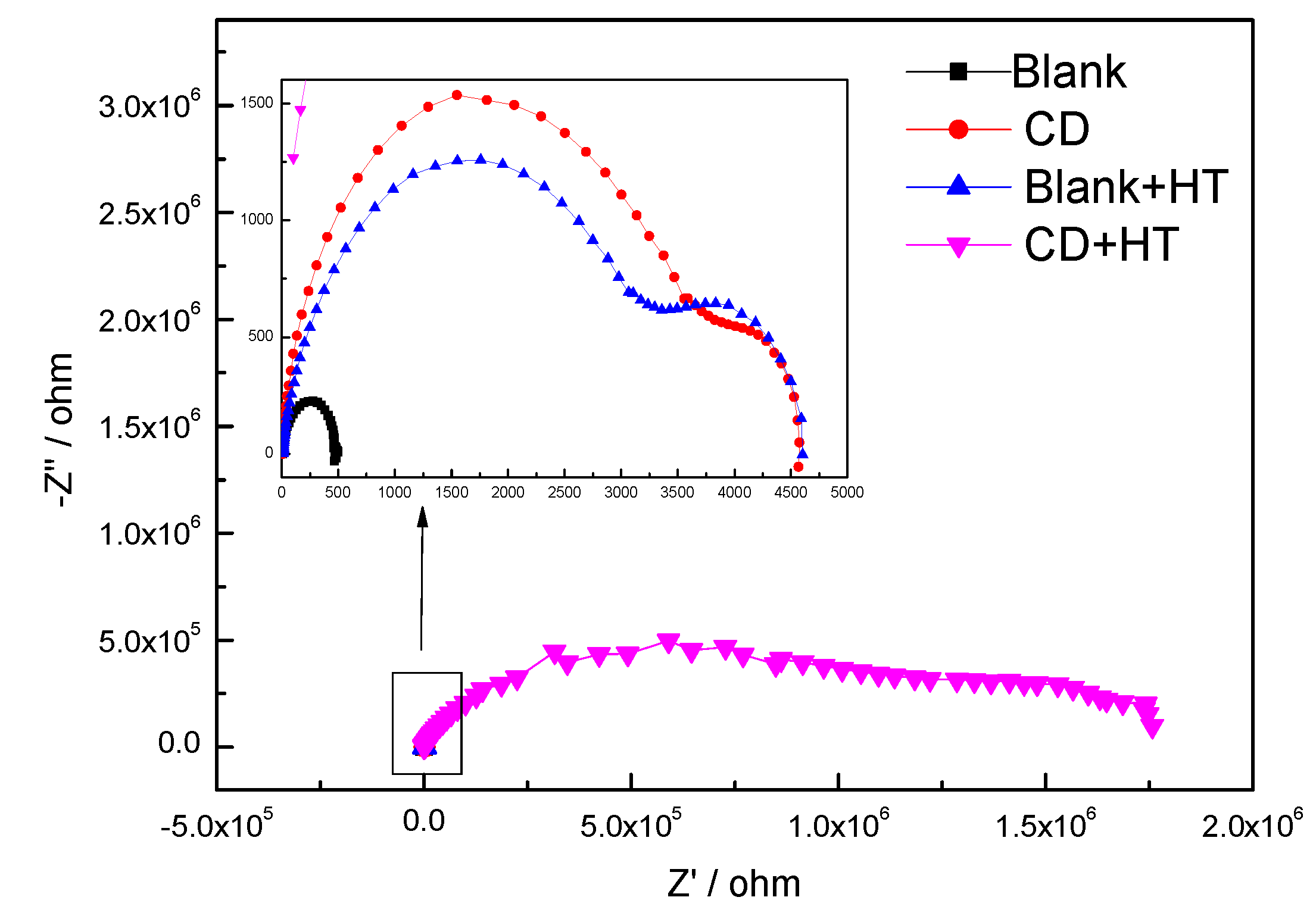
3.2.3. Electrochemical Measurements
3.3. Surface Wettability
3.4. Coating Adhesion
3.5. Micromorphology
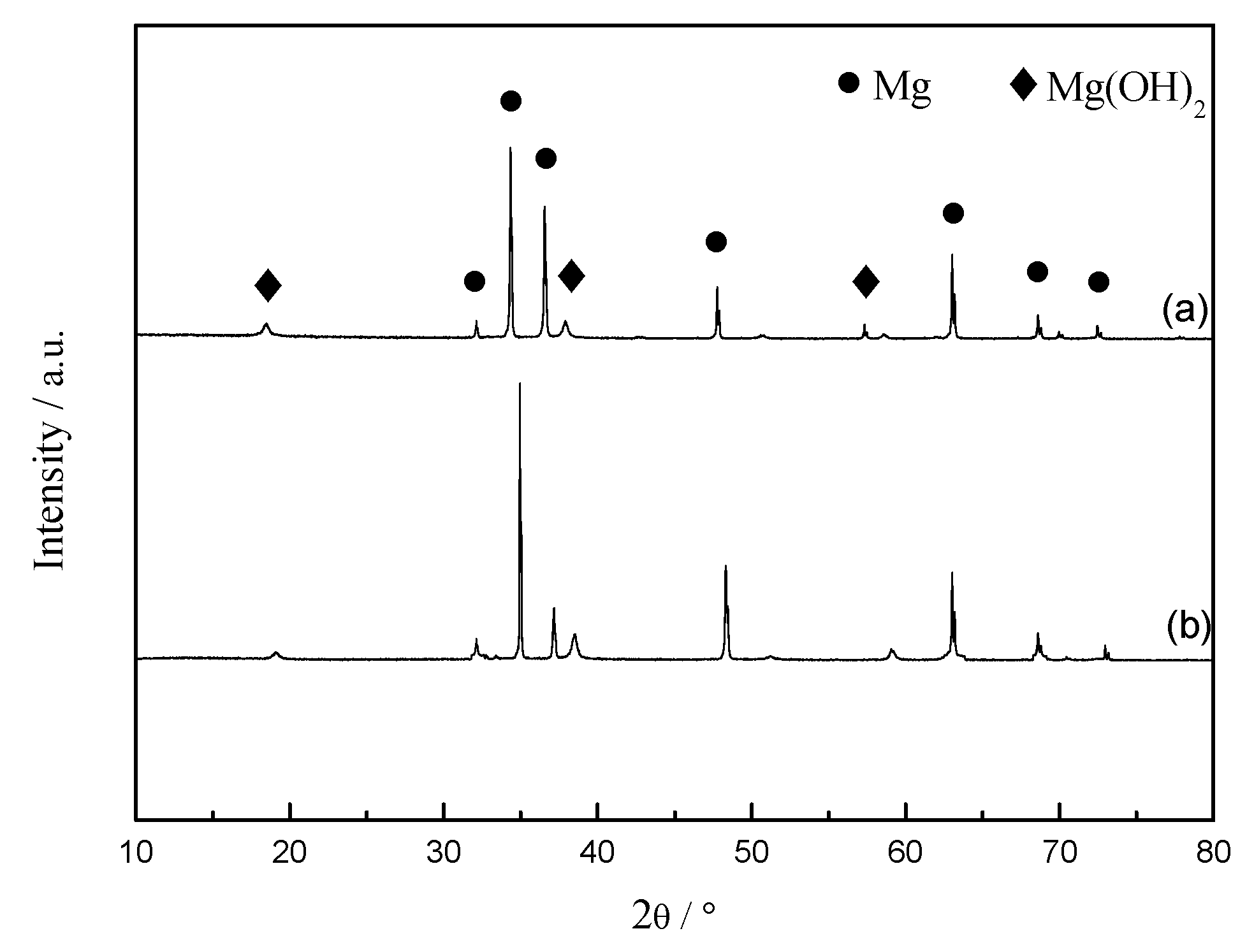
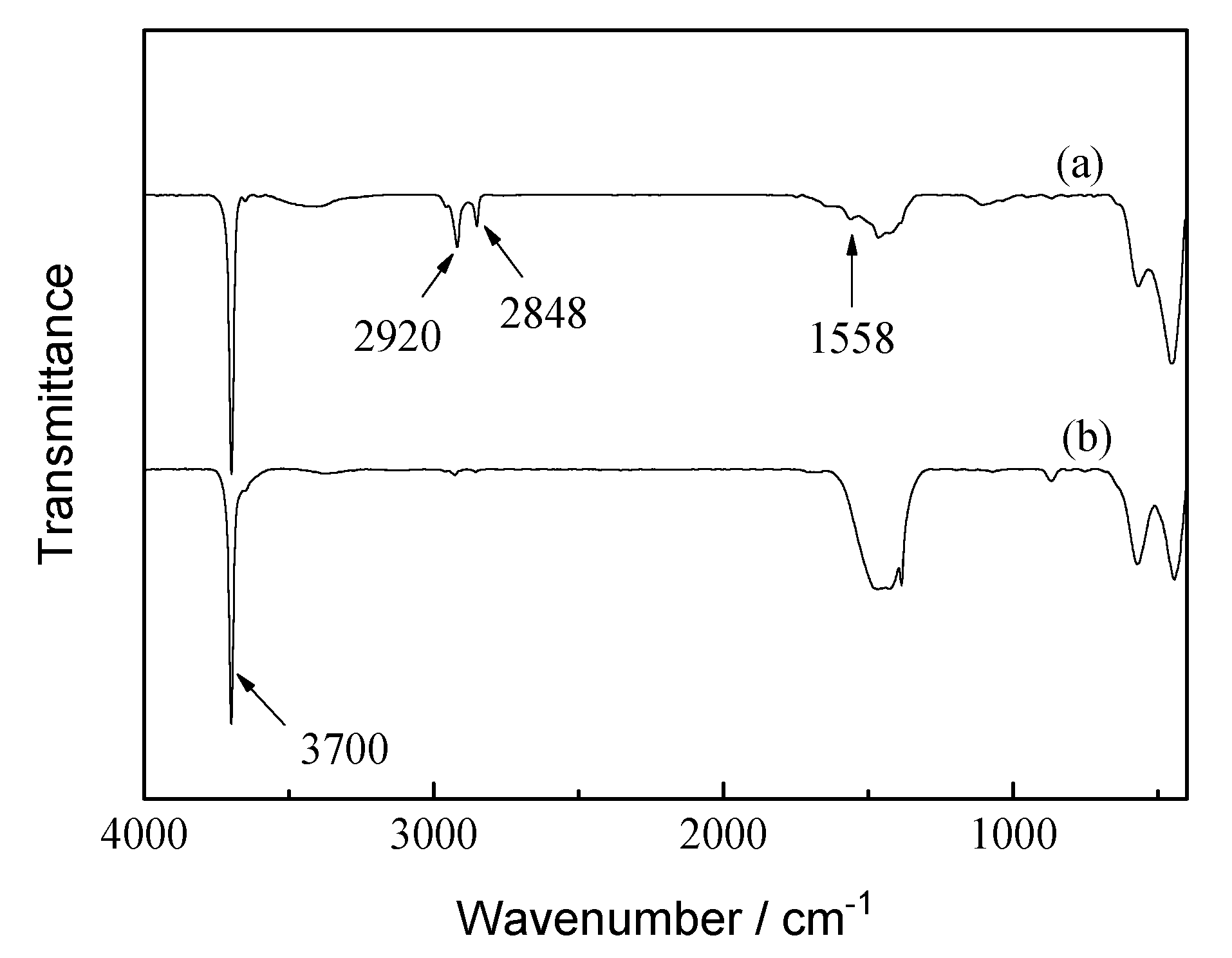
3.6. Chemical Composition
4. Conclusions
- Superhydrophobic surface with admirable 151.5° water static contact angle and 1° rolling angle could be obtained on Mg alloy substrate by CD + HT composite treatment in which Mg(OH)2 film was prepared by cathodic deposition firstly and then a simple immersion was done in a low-concentration ethanol solution of stearic acid for a short period. The optimal surface modifying parameters were 5 g/L of stearic acid at 80 °C for 1 h of immersion.
- Only CD/HT composite modification could realize surface superhydrophobization of Mg alloy, whereas single cathodic deposition or single hydrophobic treatment could not.
- SEM observation showed that Mg(OH)2 coating modified by stearic acid consisted of blade-like structures with micro/nano scale sizes. This kind of micro/nano structures were interlaced to form a large number of voids, which effectively inhibited surface contact with water and fluids and significantly improved the corrosion resistance of Mg alloy.
- The FT-IR spectrum test results showed that new –CH2– peaks appeared in the infrared spectrum of the CD + HT sample, indicating that stearic acid had reacted with the Mg(OH)2 film in a self-assembled or chemically bonded manner.
Author Contributions
Funding
Conflicts of Interest
References
- Wang, Z.W.; Li, Q.; She, Z.X.; Chen, F.N.; Li, L.Q.; Zhang, X.X.; Zhang, P. Facile and fast fabrication of superhydrophobic surface on magnesium alloy. Appl. Surf. Sci. 2013, 271, 182–192. [Google Scholar] [CrossRef]
- Song, G.L. Corrosion and Protection of Mg Alloy, 1st ed.; Chemical Industry Press: Beijing, China, 2006. (In Chinese) [Google Scholar]
- Xi, T.F.; Zheng, Y.F. Current situation and development and tendency of interventional medical device. Mater. China 2010, 29, 17–26. (In Chinese) [Google Scholar]
- Tan, Y. An overview of techniques for characterizing in homogeneities in organic surface films and under film localized corrosion. Prog. Org. Coat. 2013, 76, 791–803. [Google Scholar] [CrossRef]
- Kumar, P.S.; Sundaramurthy, J.; Zhang, X.; Mangalaraj, D.; Thavasi, V.; Ramakrishna, S. Superhydrophobic and antireflecting behavior of densely packed and size controlled ZnO nanorods. J. Alloy. Compd. 2013, 553, 375–382. [Google Scholar] [CrossRef]
- Qian, H.C.; Li, H.Y.; Zhang, D.W. Research progress of superhydrophobic surface technologies in the field of corrosion protection. Surf. Technol. 2015, 44, 15–24. [Google Scholar]
- Celia, E.; Darmanin, T.; Givenchy, E.T.; Amigoni, S.; Guittard, F. Recent advances in designing superhydrophobic surfaces. J. Colloid Interface Sci. 2013, 402, 1–18. [Google Scholar] [CrossRef]
- Darmanin, T.; Guittard, F. Recent advances in the potential applications of bioinspired superhydrophobic materials. J. Mater. Chem. A 2014, 2, 16319–16359. [Google Scholar] [CrossRef]
- Song, J.L.; Lu, Y.; Huang, S.; Liu, X.; Wu, L.B.; Xu, W.J. A simple immersion approach for fabricating superhydrophobic Mg alloy surfaces. Appl. Surf. Sci. 2013, 266, 445–450. [Google Scholar] [CrossRef]
- Zhang, Q.; Cao, K.; Chen, F.M. Fabrication of superhydrophobic surface on metallic substrate by pulse Cu electroplating and silver plating. China Surf. Eng. 2015, 28, 1–8. (In Chinese) [Google Scholar]
- Zhang, W.; Lu, P.; Qian, L. Fabrication of superhydrophobic paper surface via wax mixture coating. Chem. Eng. J. 2014, 250, 431–436. [Google Scholar] [CrossRef]
- Zhou, M.; Pang, X.; Wei, L.; Gao, K. Insitu grown superhydrophobic Zn-Al layered double hydroxides films on magnesium alloy to improve corrosion properties. Appl. Surf. Sci. 2015, 337, 172–177. [Google Scholar] [CrossRef]
- Shirtcliffe, N.J.; McHale, G.; Newton, M.I.; Perry, C.C. Intrinsically superhydrophobic organosilica sol-gel foams. Langmuir 2003, 19, 5626–5631. [Google Scholar] [CrossRef]
- Yang, J.; Zhang, Z.; Men, X.; Xu, X.; Zhu, X. Reversible superhydrobicity to superhydrophilicity switching of a carbon nanotube film via alternation of UV irradiation and dark storage. Langmuir 2010, 26, 10198–10202. [Google Scholar] [CrossRef]
- Xi, W.; Qiao, Z.; Zhu, C.; Jia, A.; Li, M. The preparation of lotus-like super-hydrophobic copper surfaces by electroplating. Appl. Surf. Sci. 2009, 255, 4836–4839. [Google Scholar] [CrossRef]
- Chu, F.; Wu, X. Fabrication and condensation characteristics of metallic superhydrophobic surfaces with hierarchical micro-nano structures. Appl. Surf. Sci. 2016, 371, 322–328. [Google Scholar] [CrossRef]
- Zhang, H.M.; Wang, T. Preparation and hydrophobic properties of the micro-nano structure of butterfly wing surface. China Surf. Eng. 2014, 27, 131–136. [Google Scholar]
- Chen, Z.; Hao, L.; Chen, C. A fast electrodeposition method for fabrication of lanthanum superhydrophobic surface with hierarchical micro-nanostructures. Colloids Surf. A. 2012, 401, 1–7. [Google Scholar] [CrossRef]
- Huang, Y.; Sarkar, D.K.; Grant Chen, X. Superhydrophobic aluminum alloy surfaces prepared by chemical etching process and their corrosion resistance properties. Appl. Surf. Sci. 2015, 356, 1012–1024. [Google Scholar] [CrossRef]
- Jia, S.G. Study on cathodic deposition of Mg(OH)2 for surface modification of Mg alloy. Master’s Thesis, South China University of Technology, Guangzhou, China, 2015. [Google Scholar]
- Zhong, Y.; Hu, J.; Zhang, Y.; Tang, S. The one-step electroposition of superhydrophobic surface on AZ31 magnesium alloy and its time-dependence corrosion resistance in NaCl solution. Appl. Surf. Sci. 2018, 427, 1193–1201. [Google Scholar] [CrossRef]
- Zhang, Y.; Tang, S.; Hu, J.; Lin, T. Formation mechanism and corrosion resistance of the hydrophobic coating on anodized magnesium. Corros. Sci. 2016, 111, 334–343. [Google Scholar] [CrossRef]
- Gao, R.; Liu, Q.; Wang, J.; Zhang, X.; Yang, W.; Liu, J.; Liu, L. Fabrication of fibrous szaibelyite with hierarchical structure superhydrophobic coating on AZ31 magnesium alloy for corrosion protection. Chem. Eng. J. 2014, 241, 352–359. [Google Scholar] [CrossRef]
- Liu, Y.; Yin, X.; Zhang, J. An electro-deposition process for fabrication of biomimetic super-hydrophobic surface and its corrosion resistance on Mg alloy. Electrochem. Acta 2014, 125, 395–403. [Google Scholar] [CrossRef]
- Zhu, Y.L.; Fan, W.B.; Feng, L.B. Anti-adhesion and corrosion resistance of superhydrophobic Mg alloy surface. J. Mater. Eng. 2016, 44, 66–70. [Google Scholar]
- Han, M.; Go, S.; Ahn, Y. Fabrication of superhydrophobic surface on magnesium substrate by chemical etching. Bull. Korean Chem. Soc. 2012, 33, 1363–1366. [Google Scholar] [CrossRef]
- Ishizaki, T.; Sakamoto, M. Facile formation of biomimetic color-tuned superhydrophobic magnesium alloy with corrosion resistance. Langmuir 2011, 27, 2375–2381. [Google Scholar] [CrossRef]
- ASTM D3359–09 Standard Test Methods for Measuring Adhesion by Tape Test; ASTM International: West Conshohocken, PA, USA, 2009.




| Percent Area Removed | 0% | Less Than 5% | 5%–15% | 15%–35% | 35%–65% | Greater Than 65% |
|---|---|---|---|---|---|---|
| Classification | 5B | 4B | 3B | 2B | 1B | 0B |
| Sample | Blank | CD | Blank + HT | CD + HT |
|---|---|---|---|---|
| Discoloration time, s | 40 | 550 | 134 | 1037 |
| Sample | Ecorr (V) | icorr (μA/cm2) |
|---|---|---|
| Blank | −1.5675 | 5.3660 |
| CD | −1.5748 | 2.5588 |
| Blank + HT | −1.6049 | 2.4051 |
| CD + HT | −1.6240 | 6.2955 × 10−3 |
| Sample | Blank | CD | Blank + HT | CD + HT |
|---|---|---|---|---|
| |Z|0.1 Hz | 4.66 × 102 | 4.14 × 103 | 3.18 × 103 | 1.45 × 106 |
| Sample | Ratio of Film Spalling | Adhesion Level | Image of Film Spalling |
|---|---|---|---|
| CD | 0% | 5B |  |
| CD + HT | 0% | 5B |  |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.; Cao, H.; Huang, H.; Wang, Z. Hydrophobic Modification of Magnesium Hydroxide Coating Deposited Cathodically on Magnesium Alloy and its Corrosion Protection. Coatings 2019, 9, 477. https://doi.org/10.3390/coatings9080477
Zhang Y, Cao H, Huang H, Wang Z. Hydrophobic Modification of Magnesium Hydroxide Coating Deposited Cathodically on Magnesium Alloy and its Corrosion Protection. Coatings. 2019; 9(8):477. https://doi.org/10.3390/coatings9080477
Chicago/Turabian StyleZhang, Yongjun, Hui Cao, Hanxiong Huang, and Zhiping Wang. 2019. "Hydrophobic Modification of Magnesium Hydroxide Coating Deposited Cathodically on Magnesium Alloy and its Corrosion Protection" Coatings 9, no. 8: 477. https://doi.org/10.3390/coatings9080477
APA StyleZhang, Y., Cao, H., Huang, H., & Wang, Z. (2019). Hydrophobic Modification of Magnesium Hydroxide Coating Deposited Cathodically on Magnesium Alloy and its Corrosion Protection. Coatings, 9(8), 477. https://doi.org/10.3390/coatings9080477




