Characterization of Sol-Gel Derived Calcium Hydroxyapatite Coatings Fabricated on Patterned Rough Stainless Steel Surface
Abstract
1. Introduction
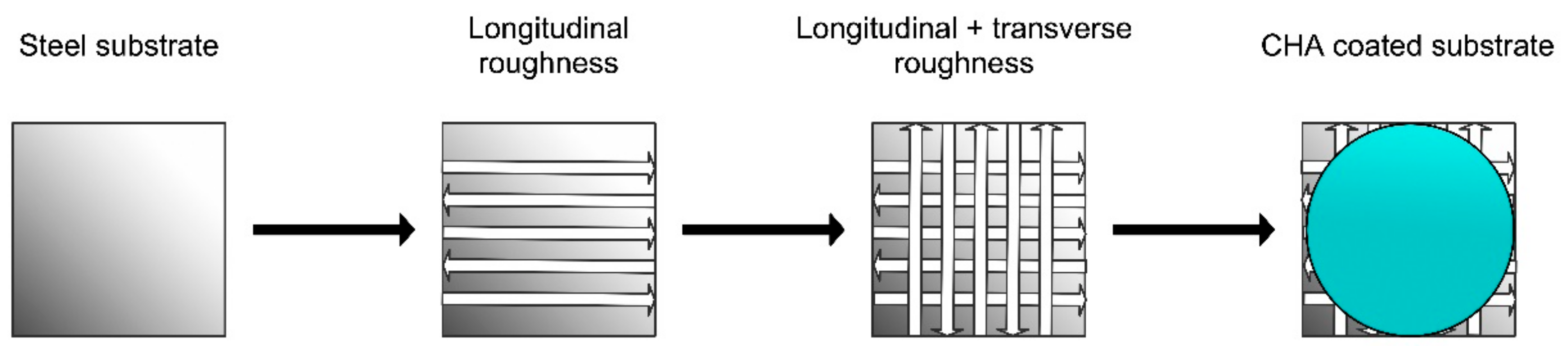
2. Experimental
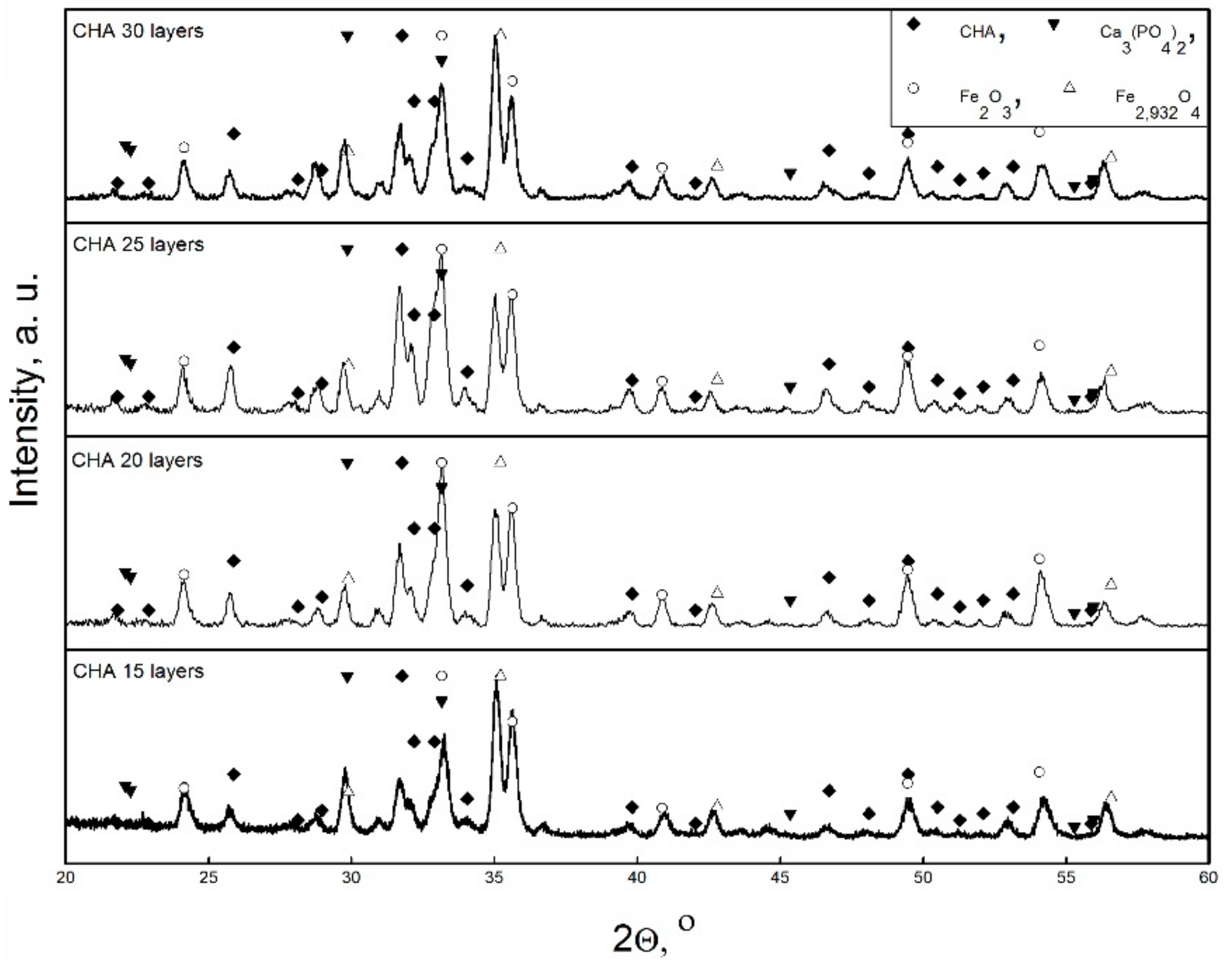
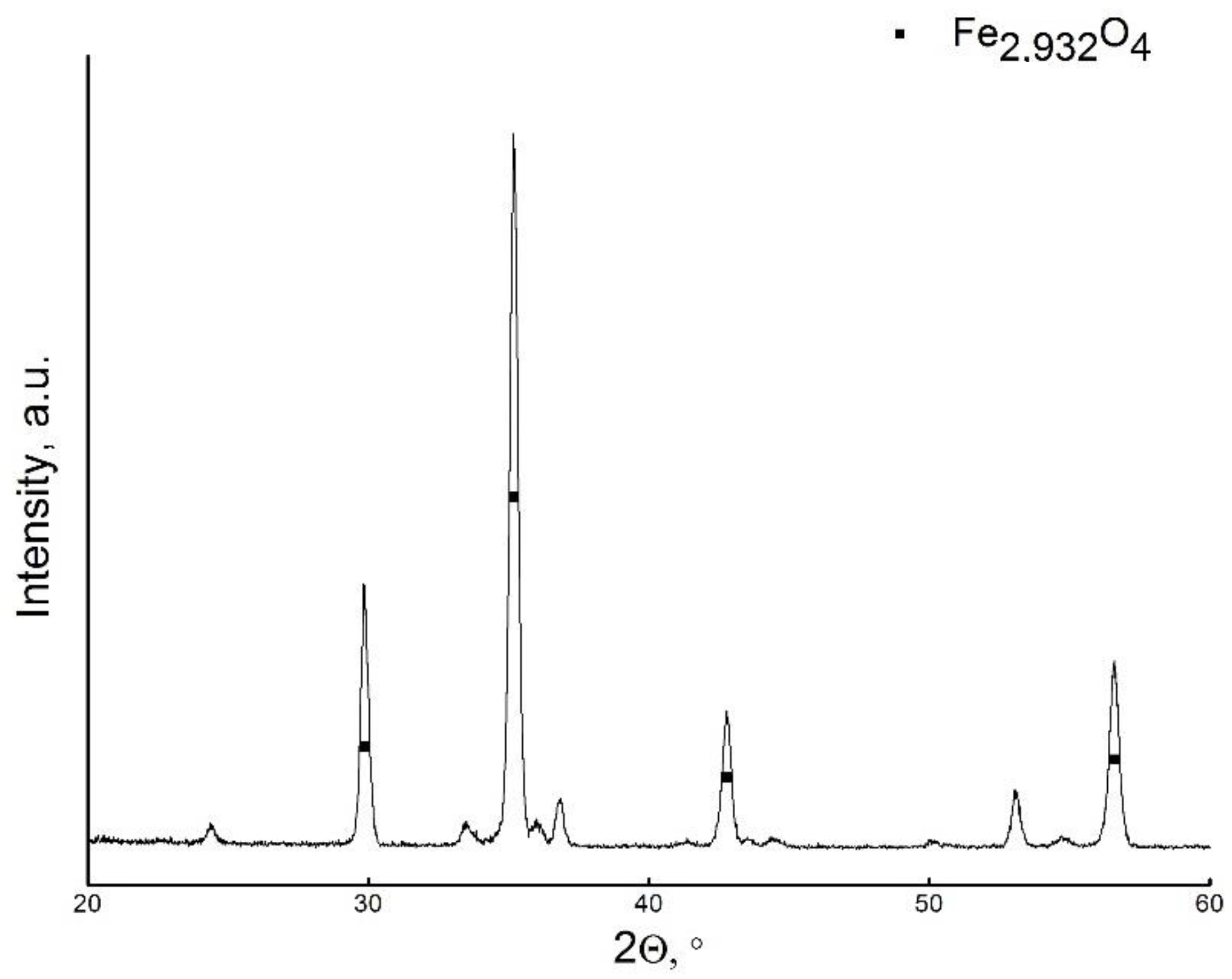
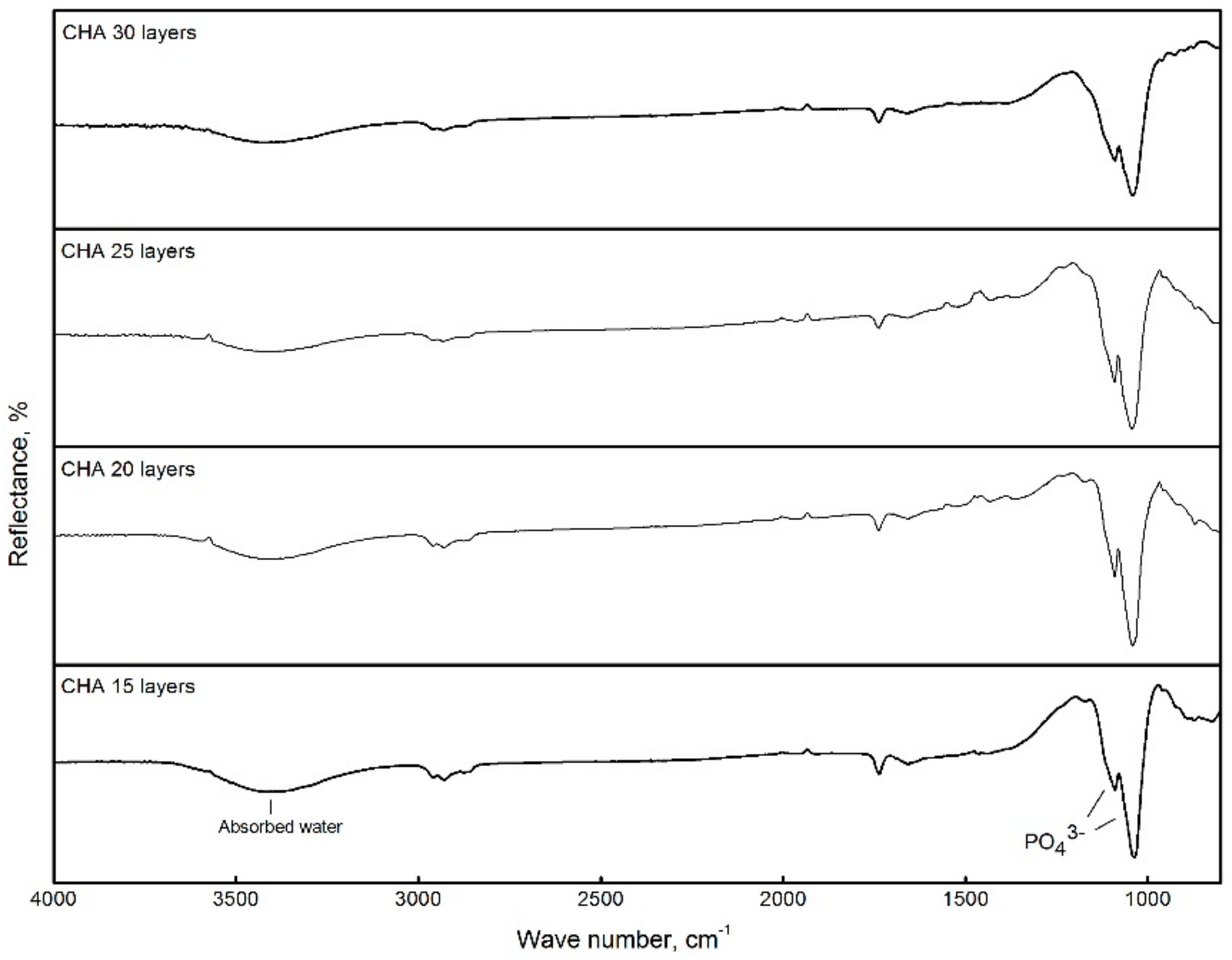
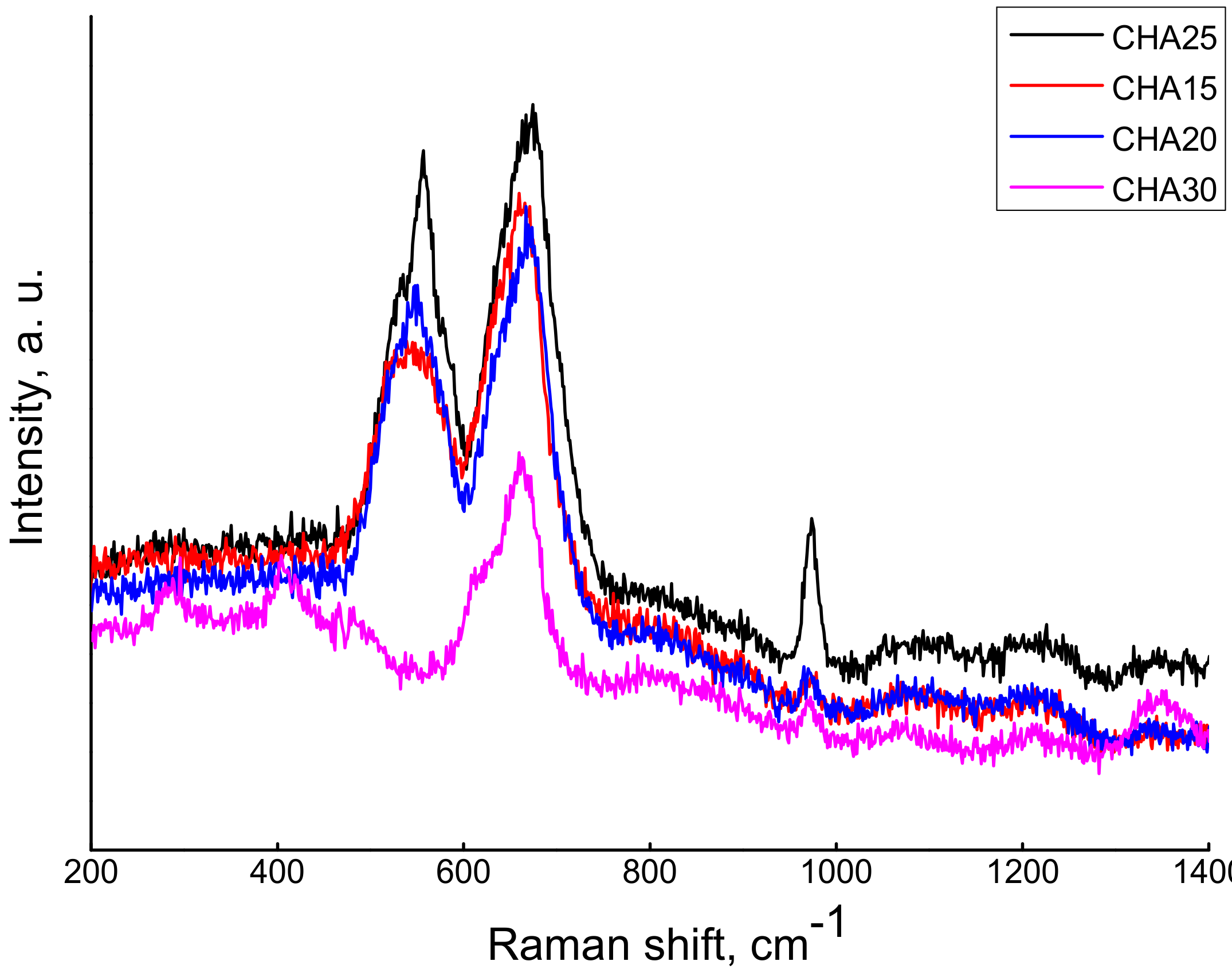
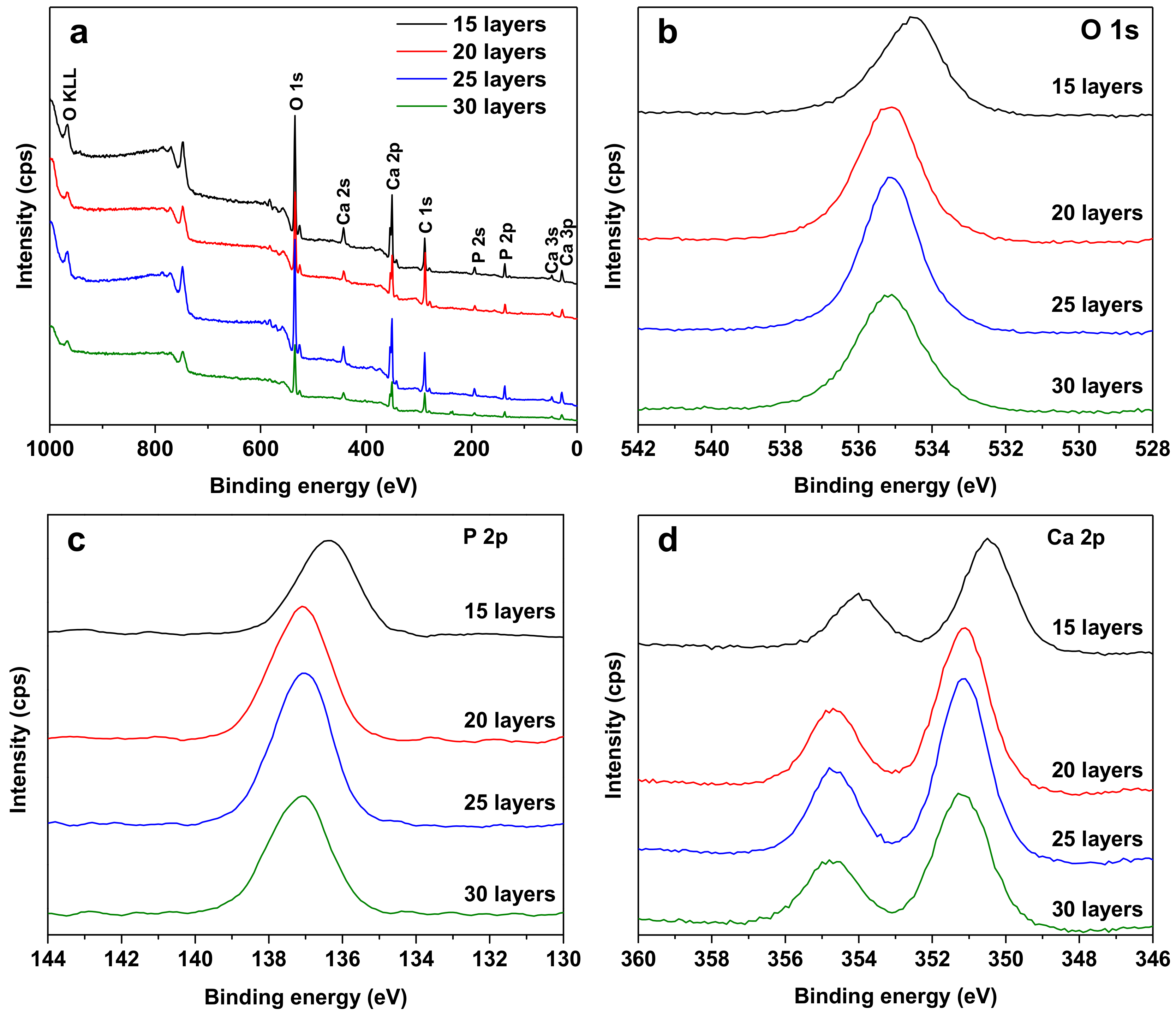
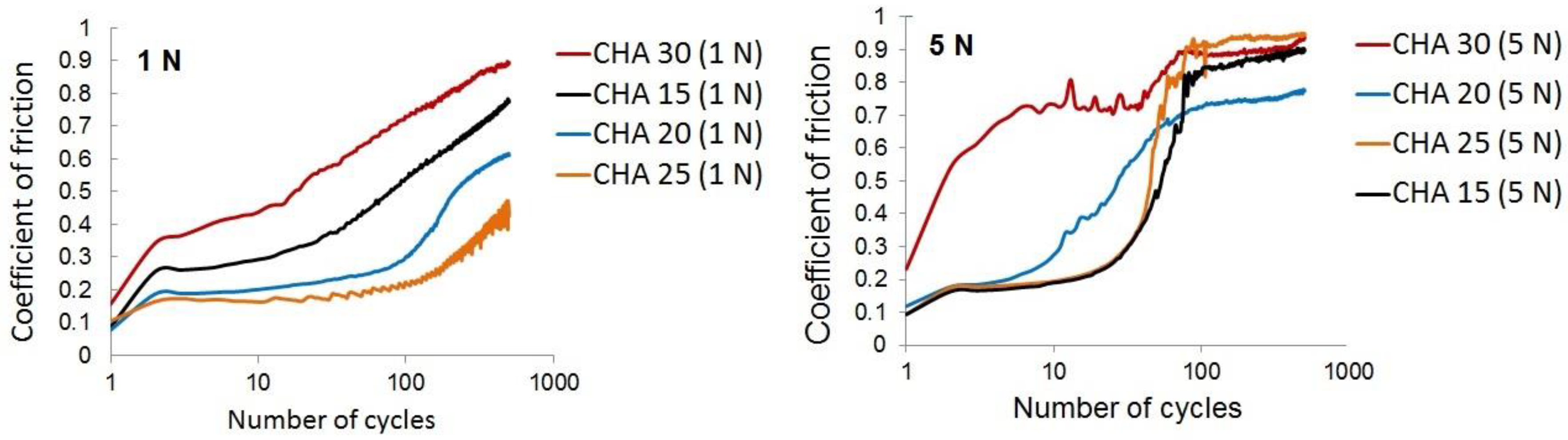
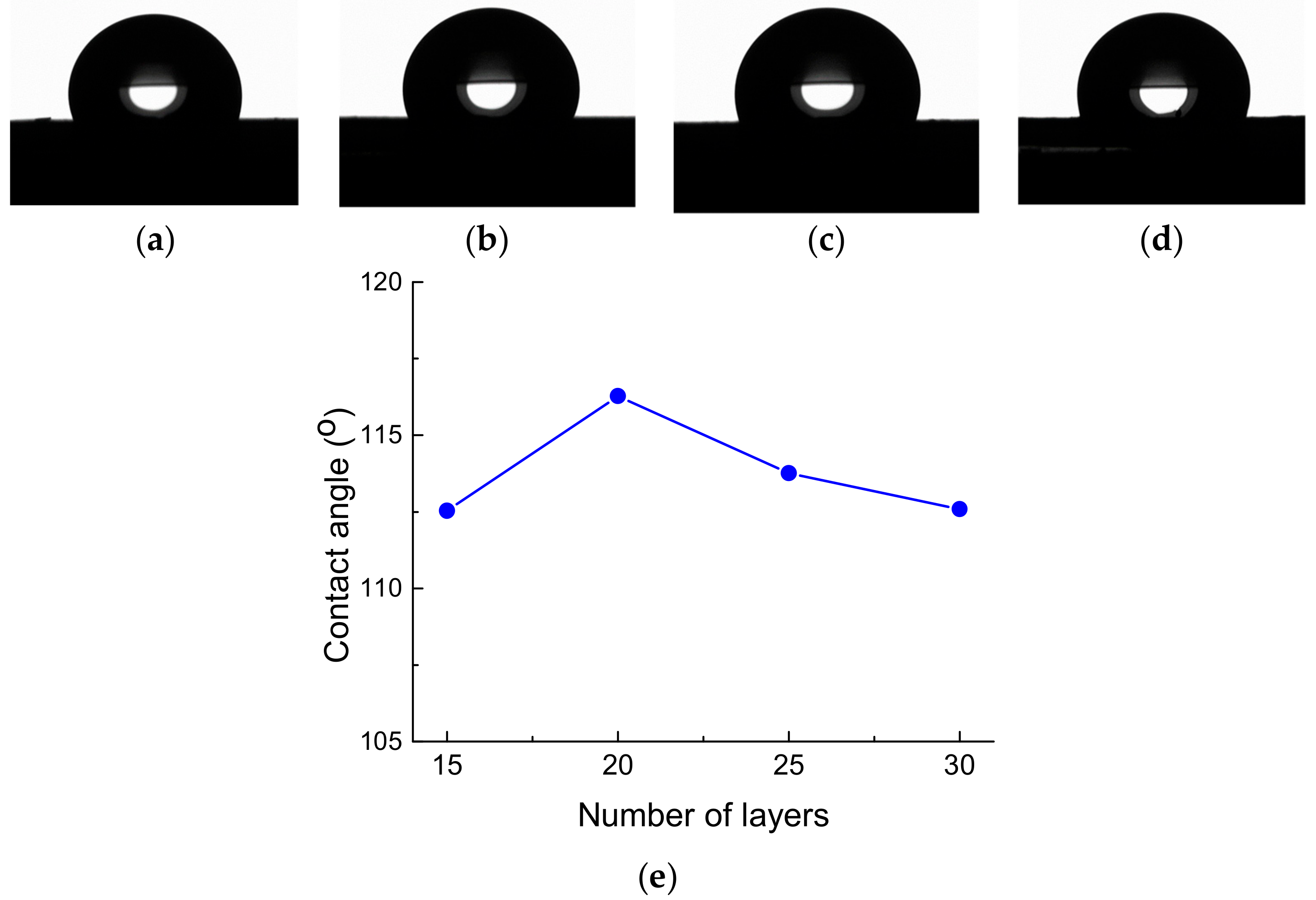
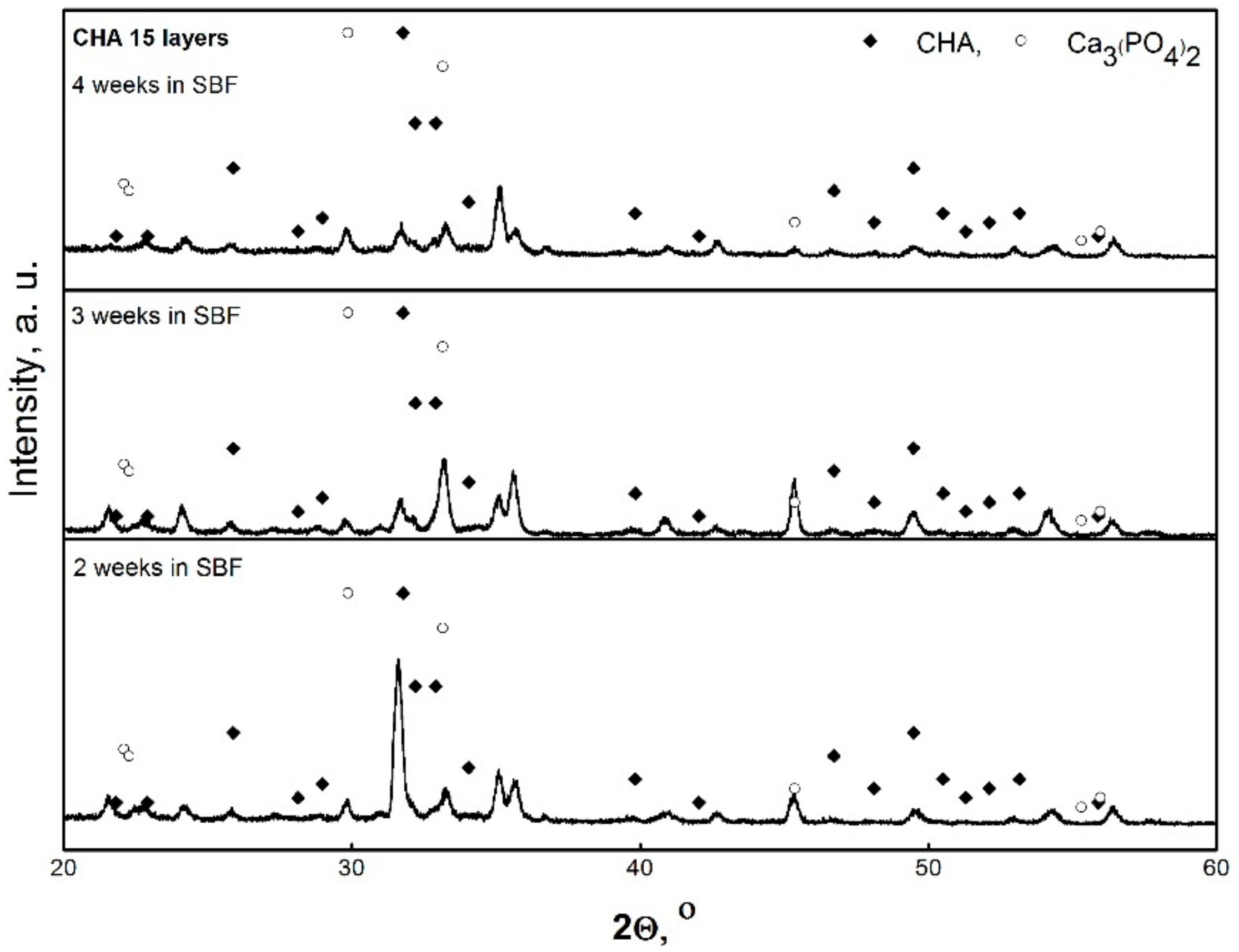
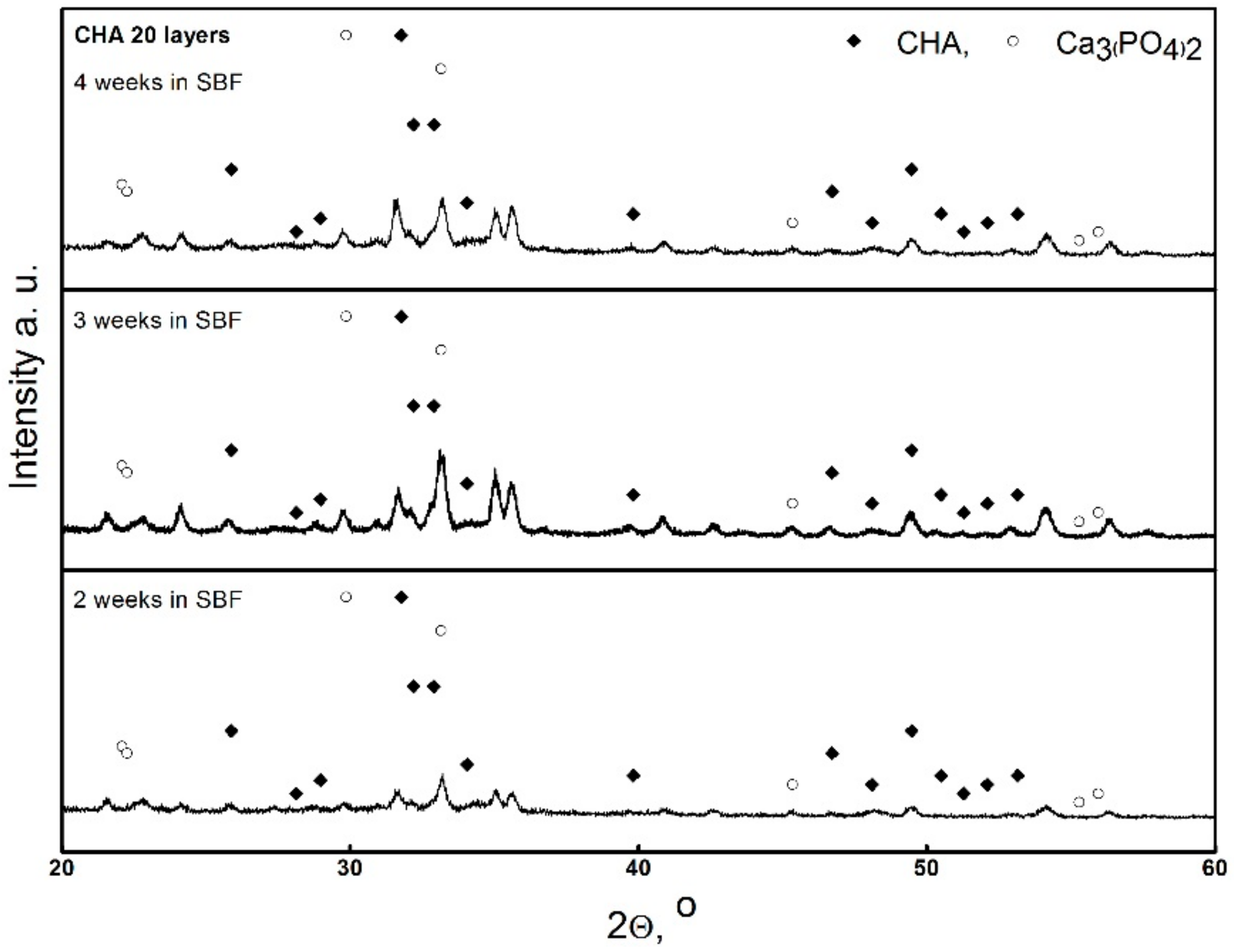
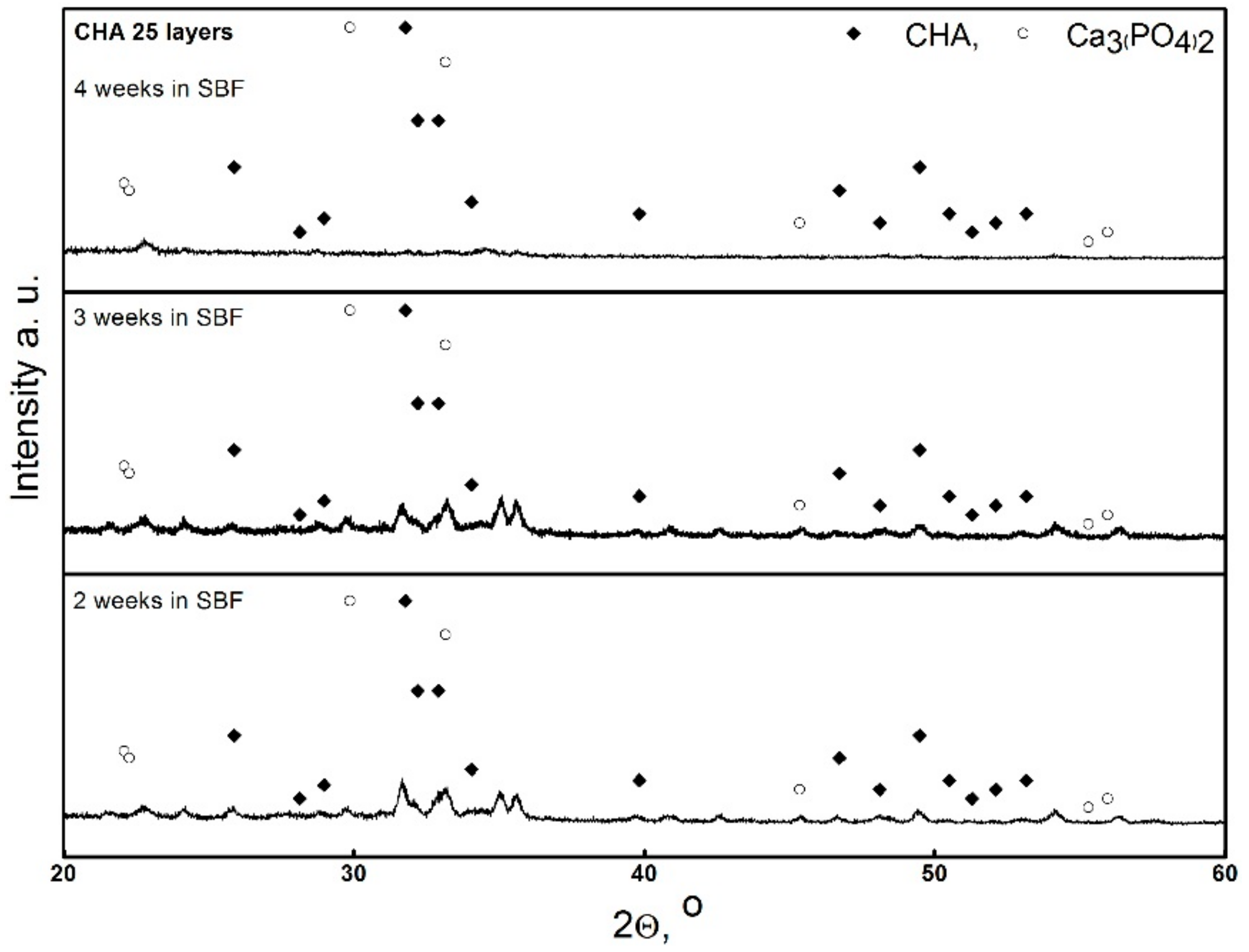
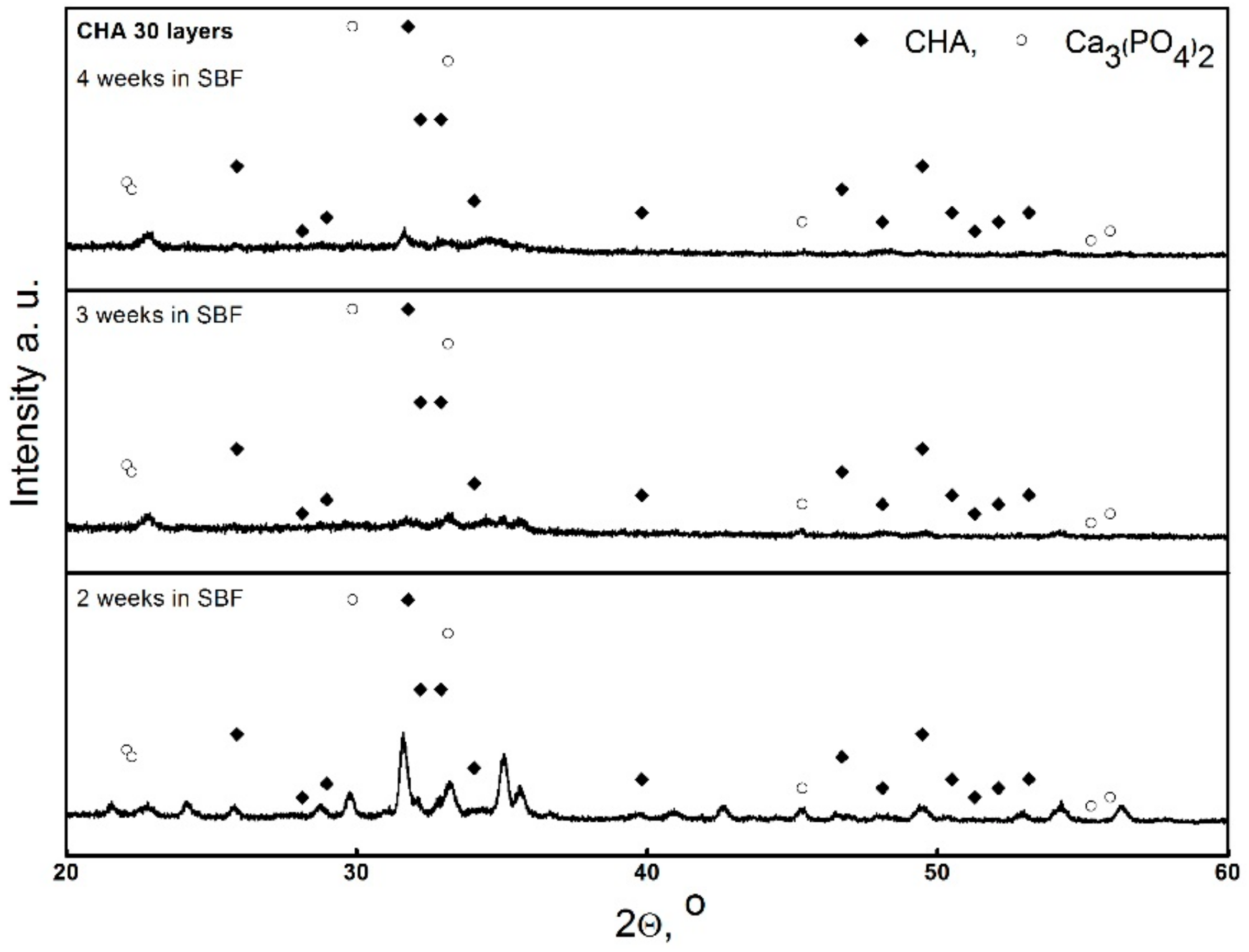
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Furkó, M.; Balázsi, K.; Balázsi, C. Comparative study on preparation and characterization of bioactive coatings for biomedical applications—A review on recent patents and literature. Rev. Adv. Mater. Sci. 2017, 48, 25–51. [Google Scholar]
- Daud, N.M.; Al-Ashwal, R.H.; Kadir, M.R.A.; Saidin, S. Polydopamine-assisted chlorhexidine immobilization on medical grade stainless steel 316L: Apatite formation and in vitro osteoblastic evaluation. Ann. Anat. 2018, 220, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Kokubo, T.; Takadama, H. How useful is SBF in predicting in vivo bone bioactivity? Biomaterials 2006, 27, 2907–2915. [Google Scholar] [CrossRef]
- Bekmurzayeva, A.; Duncansond, W.J.; Azevedo, H.S.; Kanayeva, D. Surface modification of stainless steel for biomedical applications: Revisiting a century-old material. Mater. Sci. Eng. C 2018, 93, 1073–1089. [Google Scholar] [CrossRef] [PubMed]
- Dayan, A.; Lamed, R.; Benayahu, D.; Fleminger, G. RGD-modified dihydrolipoamide dehydrogenase as a molecular bridge for enhancing the adhesion of bone forming cells to titanium dioxide implant surfaces. J. Biomed. Mater. Res. Part A 2019, 107, 545–551. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.M.; Yang, Q.; Troczynski, T. Sol-gel hydroxyapatite coatings on stainless steel substrates. Biomaterials 2002, 23, 691–698. [Google Scholar] [CrossRef]
- Shen, J.; Jin, B.; Qi, Y.C.; Jiang, Q.Y.; Gao, X.F. Carboxylated chitosan/silver-hydroxyapatite hybrid microspheres with improved antibacterial activity and cytocompatibility. Mater. Sci. Eng. C 2017, 78, 589–597. [Google Scholar] [CrossRef] [PubMed]
- Iconaru, S.L.; Chifiriuc, M.C.; Groza, A. Structural and antimicrobial evaluation of silver doped hydroxyapatite-polydimethylsiloxane thin layers. J. Nanomater. 2017, 2017, 7492515. [Google Scholar] [CrossRef]
- Predoi, D.; Popa, C.L.; Chapon, P.; Groza, A.; Iconaru, S. Evaluation of the antimicrobial activity of different antibiotics enhanced with silver-doped hydroxyapatite thin films. Materials 2016, 9, 778. [Google Scholar] [CrossRef] [PubMed]
- Takechi, M.; Ishikawa, K.; Miyamoto, Y.; Nagayama, M.; Suzuki, K. Tissue responses to anti-washout apatite cement using chitosan when implanted in the rat tibia. J. Mater. Sci. Mater. Med. 2001, 12, 597–602. [Google Scholar] [CrossRef] [PubMed]
- Shah, F.A.; Thomsen, P.; Palmquist, A. Osseointegration and current interpretations of the bone-implant interface. Acta Biomater. 2019, 84, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Sadtler, K.; Wolf, M.T.; Ganguly, S.; Moad, C.A.; Chung, L.; Majumdar, S.; Housseau, F.; Pardoll, D.M.; Elisseeff, J.H. Divergent immune responses to synthetic and biological scaffolds. Biomaterials 2019, 192, 405–415. [Google Scholar] [CrossRef] [PubMed]
- Shariff, K.A.; Tsuru, K.; Ishikawa, K. Fabrication of dicalcium phosphate dihydrate-coated beta-TCP granules and evaluation of their osteoconductivity using experimental rats. Mater. Sci. Eng. C 2017, 75, 1411–1419. [Google Scholar] [CrossRef] [PubMed]
- Fujioka-Kobayashi, M.; Tsuru, K.; Nagai, H.; Fujisawa, K.; Kudoh, T.; Ohe, G.; Ishikawa, K.; Miyamoto, Y. Fabrication and evaluation of carbonate apatite-coated calcium carbonate bone substitutes for bone tissue engineering. J. Tissue Eng. Regener. Med. 2018, 12, 2077–2087. [Google Scholar] [CrossRef] [PubMed]
- Kargozar, S.; Hamzehlou, S.; Baino, F. Can bioactive glasses be useful to accelerate the healing of epithelial tissues? Mater. Sci. Eng. C 2019, 97, 1009–1020. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Han, J.; Sun, Y.; Huang, Y.; Zhou, M. MC3T3-E1 cell response to stainless steel 316L with different surface treatments. Mater. Sci. Eng. C 2015, 56, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Akindoyo, J.O.; Beg, M.D.; Ghazali, S.; Alam, A.K.M.M.; Heim, H.P.; Feldmann, M. Synergized poly(lactic acid)-hydroxyapatite composites: Biocompatibility study. J. Appl. Polym. Sci. 2019, 136, 47400. [Google Scholar] [CrossRef]
- Dai, G.; Wan, W.; Chen, J.; Wu, J.; Shuai, X.; Wang, Y. Enhanced osteogenic differentiation of MC3T3-E1 on rhBMP-2 immobilized titanium surface through polymer-mediated electrostatic interaction. Appl. Surf. Sci. 2019, 471, 986–998. [Google Scholar] [CrossRef]
- Balla, V.K.; Das, M.; Bose, S.; Ram, G.J.; Manna, I. Laser surface modification of 316 L stainless steel with bioactive hydroxyapatite. Mater. Sci. Eng. C 2013, 33, 4594–4598. [Google Scholar] [CrossRef] [PubMed]
- Prosolov, K.A.; Belyavskaya, O.A.; Muehle, U.; Sharkeev, Y.P. Thin bioactive Zn substituted hydroxyapatite coating deposited on ultrafine-grained titanium substrate: Structure analysis. Front. Mater. 2018, 5, 3. [Google Scholar] [CrossRef]
- Xin, F.; Jian, C.; Zou, J.P.; Qian, W.; Zhou, Z.C.; Ruan, J.M. Bone-like apatite formation on HA/316L stainless steel composite surface in simulated body fluid. Transac. Nonferr. Metals Soc. China 2009, 19, 347–352. [Google Scholar] [CrossRef]
- Sarkar, C.; Kumari, P.; Anuvrat, K.; Sahu, S.K.; Chakraborty, J.; Garai, S. Synthesis and characterization of mechanically strong carboxymethyl cellulose-gelatin-hydroxyapatite nanocomposite for load-bearing orthopedic application. J. Mater. Sci. 2018, 53, 230–246. [Google Scholar] [CrossRef]
- Testori, T.; Iezzi, G.; Manzon, L.; Fratto, G.; Piattelli, A.; Weinstein, R.L. High temperature-treated bovine porous hydroxyapatite in sinus augmentation procedures: A case report. Int. J. Periodont. Restor. Dent. 2012, 32, 295–301. [Google Scholar]
- Abueva, C.D.; Padalhin, A.R.; Min, Y.K.; Lee, B.T. Preformed chitosan cryogel-biphasic calcium phosphate: A potential injectable biocomposite for pathologic fracture. J. Biomater. Appl. 2015, 30, 182–192. [Google Scholar] [CrossRef] [PubMed]
- Durham III, J.W.; Allen, M.J.; Rabiei, A. Preparation, characterization and in vitro response of bioactive coatings on polyether ether ketone. J. Biomed. Mater. Res. Part B Appl. Biomater. 2017, 105, 560–567. [Google Scholar] [CrossRef] [PubMed]
- Malakauskaite-Petruleviciene, M.; Stankeviciute, Z.; Beganskiene, A.; Kareiva, A. Sol-gel synthesis of calcium hydroxyapatite thin films on quartz substrate using dip-coating and spin-coating techniques. J. Sol Gel Sci. Technol. 2014, 71, 437–446. [Google Scholar] [CrossRef]
- Malakauskaite-Petruleviciene, M.; Stankeviciute, Z.; Niaura, G.; Prichodko, A.; Kareiva, A. Synthesis and characterization of sol-gel derived calcium hydroxyapatite thin films spin-coated on silicon substrate. Ceram. Int. 2015, 41, 7421–7428. [Google Scholar] [CrossRef]
- Usinskas, P.; Stankeviciute, Z.; Beganskiene, A.; Kareiva, A. Sol-gel derived porous and hydrophilic calcium hydroxyapatite coating on modified titanium substrate. Surf. Coat. Technol. 2016, 307, 935–940. [Google Scholar] [CrossRef]
- Jonauske, V.; Prichodko, A.; Skaudzius, R.; Kareiva, A. Sol-gel derived calcium hydroxyapatite thin films on 316L stainless steel substrate: Comparison of spin-coating and dip-coating techniques. Chemija 2016, 27, 192–201. [Google Scholar]
- Rao, P.S.; Murmu, B.; Agarwal, S. Effects of surface roughness and non-newtonian micropolar fluid squeeze film between truncated conical bearings. J. Nanofluids 2019, 8, 1338–1344. [Google Scholar] [CrossRef]
- Matijošius, T.; Ručinskienė, A.; Selskis, A.; Stalnionis, G.; Leinartas, K.; Asadauskas, S. Friction reduction by nanothin titanium layers on anodized alumina. Surf. Coat. Tech. 2016, 307, 610–621. [Google Scholar] [CrossRef]
- Garskaite, E.; Gross, K.A.; Yang, S.W.; Yang, T.C.K.; Yang, J.C.; Kareiva, A. Effect of processing conditions on the crystallinity and structure of carbonated calcium hydroxyapatite (CHAp). CrystEngComm 2014, 16, 3950–3959. [Google Scholar] [CrossRef]
- Mujahid, M.; Sarfraz, S.; Amin, S. On the formation of hydroxyapatite nano crystals prepared using cationic surfactant. Mater. Res. 2015, 18, 468–472. [Google Scholar] [CrossRef]
- Natasha, A.N.; Sopyan, I.; Zuraid, A. Fourier transform infrared study on sol-gel derived manganese-doped hydroxyapatite. Adv. Mater. Res. 2008, 47, 1185–1188. [Google Scholar] [CrossRef]
- Malakauskaite-Petruleviciene, M.; Stankeviciute, Z.; Niaura, G.; Garskaite, E.; Beganskiene, A.; Kareiva, A. Characterization of sol-gel processing of calcium phosphate thin films on silicon substrate by FTIR spectroscopy. Vibr. Spectrosc. 2016, 85, 16–21. [Google Scholar] [CrossRef]
- Usinskas, P.; Stankeviciute, Z.; Niaura, G.; Maminskas, J.; Juodzbalys, G.; Kareiva, A. Sol-gel processing of calcium hydroxyapatite thin films on silicon nitride (Si3N4) substrate. J. Sol Gel Sci. Technol. 2017, 83, 268–274. [Google Scholar] [CrossRef]
- Karampas, I.A.; Kontoyannis, C.G. Characterization of calcium phosphates mixtures. Vibr. Spectrosc. 2013, 64, 126–133. [Google Scholar] [CrossRef]
- Sofronia, A.M.; Baies, R.; Anghel, E.M.; Marinescu, C.A.; Tanasescu, S. Thermal and structural characterization of synthetic and natural nanocrystalline hydroxyapatite. Mater. Sci. Eng. C 2014, 43, 153–163. [Google Scholar] [CrossRef] [PubMed]
- Chernozem, R.V.; Surmeneva, M.A.; Krause, B.; Baumbach, T.; Ignatov, V.P.; Tyurin, A.I.; Loza, K.; Epple, M.; Surmenev, R.A. Hybrid biocomposites based on titania nanotubes and a hydroxyapatite coating deposited by RF-magnetron sputtering: Surface topography, structure, and mechanical properties. Appl. Surf. Sci. 2017, 426, 229–237. [Google Scholar] [CrossRef]
- Ramesh, B.; Dillip, G.R.; Rambabu, B.; Joo, S.W.; Raju, B.D.P. Structural studies of a green-emitting terbium doped calcium zinc phosphate phosphor. J. Molec. Struct. 2018, 1155, 568–572. [Google Scholar] [CrossRef]
- Huang, J.; Fan, X.; Xiong, D.; Li, J.; Zhu, H.; Huang, M. Characterization and one-step synthesis of hydroxyapatite-Ti(C,N)-TiO2 composite coating by cathodic plasma electrolytic saturation and accompanying electrochemical deposition on titanium alloy. Surf. Coat. Technol. 2017, 324, 463–470. [Google Scholar] [CrossRef]
- Brazda, L.; Rohanova, D.; Helebrant, A. Kinetics of dissolution of calcium phosphate (Ca–P) bioceramics. Process. Appl. Ceram. 2008, 2, 57–62. [Google Scholar] [CrossRef]
- Wu, M.; Wang, T.; Wang, Q.; Huang, W. Preparation of bio-inspired polydopamine coating on hydrated tricalcium silicate substrate to accelerate hydroxyapatite mineralization. Mater. Lett. 2019, 236, 120–123. [Google Scholar] [CrossRef]
- Xie, L.; Yang, Y.; Fu, Z.; Li, Y.; Shi, J.; Ma, D.; Liu, S.L.; Luo, D. Fe/Zn-modified tricalcium phosphate (TCP) biomaterials: Preparation and biological properties. RSC Adv. 2019, 9, 781–789. [Google Scholar] [CrossRef]
- Chen, Z.; Zhai, J.; Wang, D.; Chen, C. Bioactivity of hydroxyapatite/wollastonite composite films deposited by pulsed laser. Ceram. Int. 2018, 44, 10204–10209. [Google Scholar] [CrossRef]
- Shahrezaee, M.; Raz, M.; Shishehbor, S.; Moztarzadeh, F.; Baghbani, F.; Sadeghi, A.; Bajelani, K.; Tondnevis, F. Synthesis of magnesium doped amorphous calcium phosphate as a bioceramic for biomedical application: In vitro study. Silicon 2018, 10, 1171–1179. [Google Scholar] [CrossRef]
- Heimann, R.B. Plasma-sprayed hydroxylapatite coatings as biocompatible intermediaries between inorganic implant surfaces and living tissue. J. Therm. Spray Technol. 2018, 27, 1212–1237. [Google Scholar] [CrossRef]
- Roy, M.; Bandyopadhyay, A.; Bose, S. Induction plasma sprayed nano hydroxyapatite coatings on titanium for orthopaedic and dental implants. Surf. Coat. Technol. 2011, 205, 2785–2792. [Google Scholar] [CrossRef]
- Xiao, G.Y.; Lü, Y.P.; Zhu, R.F.; Xu, W.H.; Jiao, Y. Fabrication of hydroxyapatite microspheres with poor crystallinity using a novel flame-drying method. Transac. Nonfer. Metals Soc. China 2012, 22, S169–S174. [Google Scholar] [CrossRef]
- Zhu, H.; Guo, D.; Sun, L.; Li, H.; Hanaor, D.A.; Schmidt, F.; Xu, K. Nanostructural insights into the dissolution behavior of Sr-doped hydroxyapatite. J. Eur. Ceram. Soc. 2018, 38, 5554–5562. [Google Scholar] [CrossRef]


| Ion | Ion Concentration (mM) | |
|---|---|---|
| Blood Plasma | SBF | |
| Na+ | 142.0 | 142.0 |
| K+ | 5.0 | 5.0 |
| Mg2+ | 1.5 | 1.5 |
| Ca2+ | 2.5 | 2.5 |
| Cl− | 103.0 | 147.8 |
| HCO3− | 27.0 | 4.2 |
| HPO42− | 1.0 | 1.0 |
| SO42− | 0.5 | 0.5 |
| pH | 7.2–7.4 | 7.40 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jonauske, V.; Stanionyte, S.; Chen, S.-W.; Zarkov, A.; Juskenas, R.; Selskis, A.; Matijosius, T.; Yang, T.C.K.; Ishikawa, K.; Ramanauskas, R.; et al. Characterization of Sol-Gel Derived Calcium Hydroxyapatite Coatings Fabricated on Patterned Rough Stainless Steel Surface. Coatings 2019, 9, 334. https://doi.org/10.3390/coatings9050334
Jonauske V, Stanionyte S, Chen S-W, Zarkov A, Juskenas R, Selskis A, Matijosius T, Yang TCK, Ishikawa K, Ramanauskas R, et al. Characterization of Sol-Gel Derived Calcium Hydroxyapatite Coatings Fabricated on Patterned Rough Stainless Steel Surface. Coatings. 2019; 9(5):334. https://doi.org/10.3390/coatings9050334
Chicago/Turabian StyleJonauske, Vilma, Sandra Stanionyte, Shih-Wen Chen, Aleksej Zarkov, Remigijus Juskenas, Algirdas Selskis, Tadas Matijosius, Thomas C. K. Yang, Kunio Ishikawa, Rimantas Ramanauskas, and et al. 2019. "Characterization of Sol-Gel Derived Calcium Hydroxyapatite Coatings Fabricated on Patterned Rough Stainless Steel Surface" Coatings 9, no. 5: 334. https://doi.org/10.3390/coatings9050334
APA StyleJonauske, V., Stanionyte, S., Chen, S.-W., Zarkov, A., Juskenas, R., Selskis, A., Matijosius, T., Yang, T. C. K., Ishikawa, K., Ramanauskas, R., & Kareiva, A. (2019). Characterization of Sol-Gel Derived Calcium Hydroxyapatite Coatings Fabricated on Patterned Rough Stainless Steel Surface. Coatings, 9(5), 334. https://doi.org/10.3390/coatings9050334









