Electrodeposition of Co-B/SiC Composite Coatings: Characterization and Evaluation of Wear Volume and Hardness
Abstract
1. Introduction
2. Materials and Methods
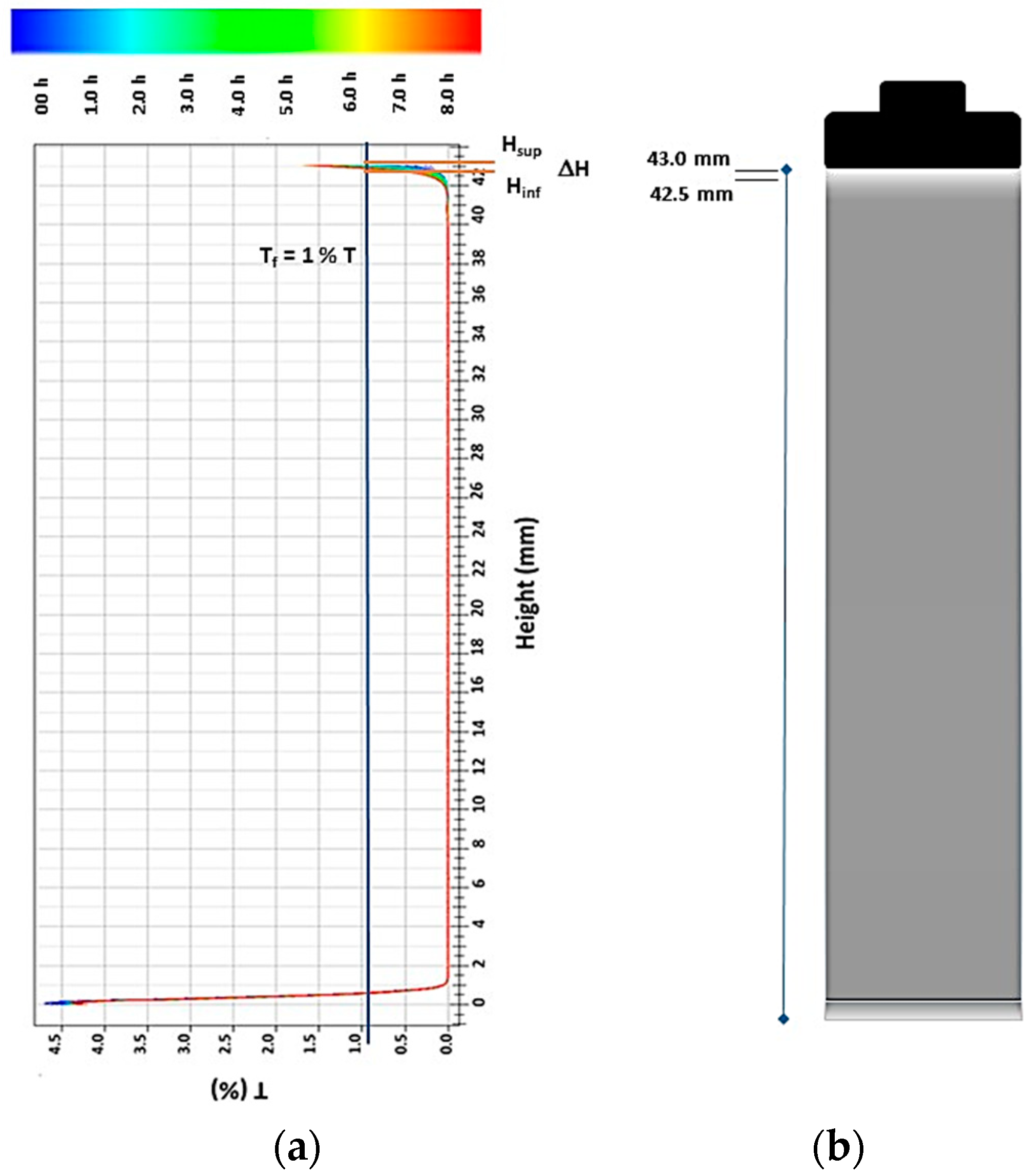
2.1. Study of the Stability of SiC Particles in Colloidal Suspension
2.2. Electrodeposition and Characterization of the Co-B/SiC Composite Coatings
3. Results and Discussion
3.1. Stability Analyses of the System SiC Particles/CTAB in Colloidal Suspensions
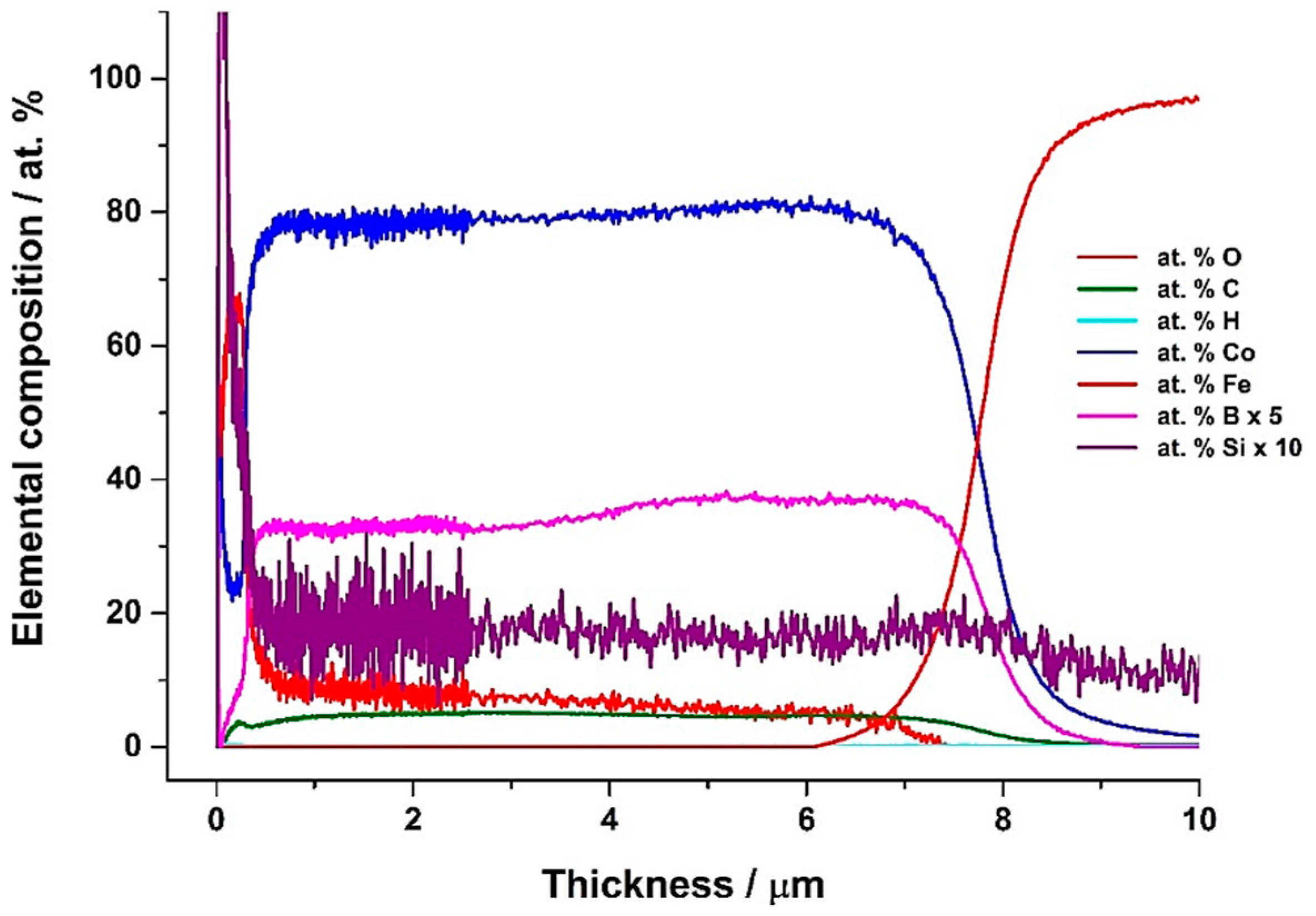
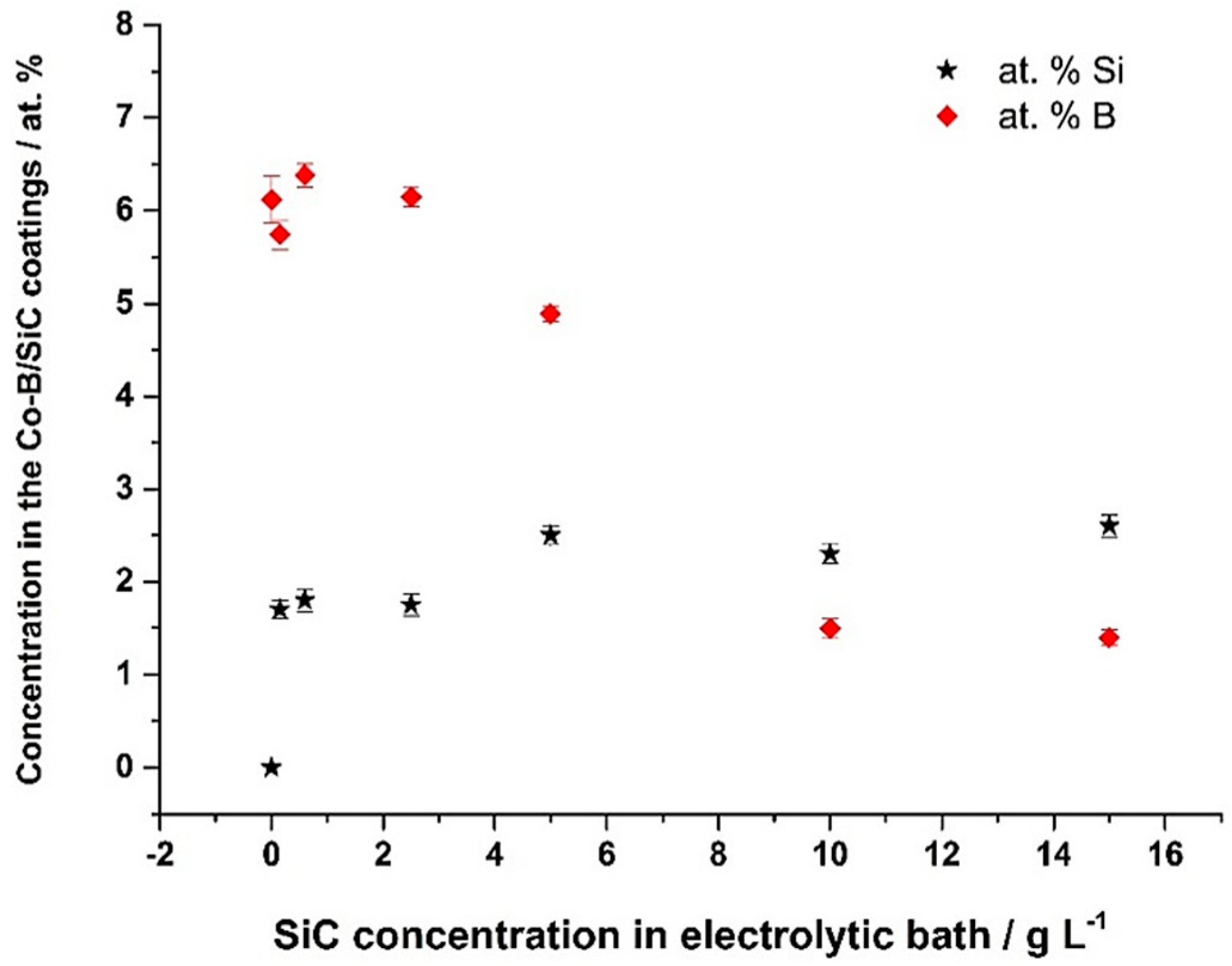
3.2. Co-B/SiC Composite Coating Composition
3.3. Co-B/SiC Composite Coating Morphology
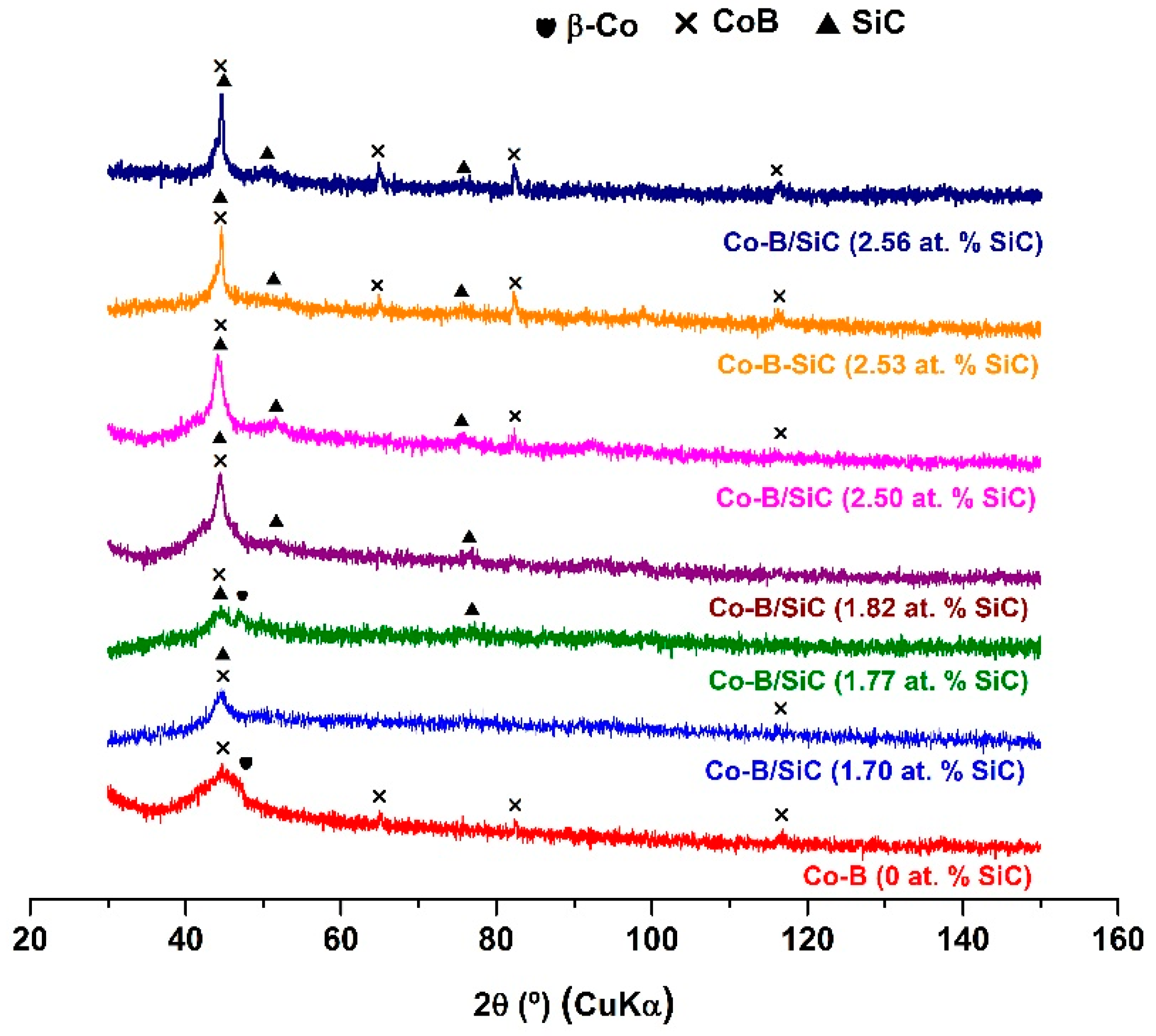
3.4. XRD Analysis
3.5. Microhardness of the Co-B/SiC Composite Coatings
3.6. Tribological Behavior
3.7. Study of Heat Treatment (HT) onto Hardness, Wear Volume, and Friction Coefficients (μ)
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Eskin, S.; Berkh, O.; Rogalsky, G.; Zahavi, J. Co-W alloys for replacement of conventional hard chromium. Plat. Surf. Finish. 1998, 85, 79–83. [Google Scholar]
- Hovestad, A.; Janssen, L.J.J. Electrochemical codeposition of inert particles in a metallic matrix. J. Appl. Electrochem. 1995, 25, 519–527. [Google Scholar] [CrossRef]
- Wang, W.; Hou, F.-Y.; Wang, H.; Guo, H.-T. Fabrication and characterization of Ni-ZrO2 composite nano-coatings by pulse electrodeposition. Scr. Mater. 2005, 53, 613–618. [Google Scholar] [CrossRef]
- Shahri, Z.; Allahkaram, S.R. Effect of plating parameters on microstructure and tribological properties of Co-B(hexagonal) nano composite coatings. Trans. Nonferrous Met. Soc. China 2013, 23, 2929–2938. [Google Scholar] [CrossRef]
- Venkataraman, B.; Sundararajan, G. The sliding wear behavior of Al-SiC particulate composites-I. Macrobehavior. Acta Mater. 1996, 44, 451–460. [Google Scholar] [CrossRef]
- Ortiz-Merino, J.L.; Todd, R.I. Relationship between wear rate, surface pullout and microstructure during abrasive wear of alumina and alumina/SiC nanocomposites. Acta Mater. 2005, 53, 3345–3357. [Google Scholar] [CrossRef]
- Sanchez Egea, A.J.; Martynenko, V.; Abate, G.; Deferrari, N.; Martínez Krahmer, D.; López de Lacalle, L.N. Friction capabilities of graphite-based lubricants at room and over 1400 K temperatures. Int. J. Adv. Manuf. Technol. 2019, 1–11. [Google Scholar] [CrossRef]
- Lee, H.-K.; Lee, H.-Y.; Jeon, J.-M. Electrolytic deposition behaviors of Ni-SiC composite coatings containing submicron-sized SiC particles. Met. Mater. Int. 2008, 14, 599–605. [Google Scholar] [CrossRef]
- Chou, M.-C.; Ger, M.-D.; Ke, S.-T.; Huang, Y.-R.; Wu, S.-T. The Ni–P–SiC composite produced by electro-codeposition. Mater. Chem. Phys. 2005, 92, 146–151. [Google Scholar] [CrossRef]
- Ogihara, H.; Wang, H.; Saji, T. Electrodeposition of Ni-B/SiC composite films with high hardness and wear resistance. Appl. Surf. Sci. 2014, 296, 108–113. [Google Scholar] [CrossRef]
- Balaraju, J.N.; Seshadri, S.K. Synthesis and corrosion behavior of electroless Ni-P-Si3N4 composite coatings. J. Materials. Sci. Lett. 1998, 17, 1297–1299. [Google Scholar] [CrossRef]
- Aslayan, I.R.; Bonino, J.-P.; Celis, J.-P. Effect of submicron SiC particles on the wear of electrolytic NiP coatings Part 1. Uni-directional sliding. Surf. Coat. Technol. 2006, 200, 2909–2916. [Google Scholar] [CrossRef]
- Garcia, I.; Fransaer, J.; Celis, J.-P. Electrodeposition and sliding wear resistance of nickel composite coatings containing micron and submicron SiC particles. Surf. Coat. Technol. 2001, 148, 171–178. [Google Scholar] [CrossRef]
- Bozzini, B.; Martini, C.; Cavalloti, P.L.; Lanzoni, E. Relationships among crystallographic structure, mechanical properties and tribological behavior of electroless Ni-P(9%)/B4C. Wear 1999, 225, 806–813. [Google Scholar] [CrossRef]
- Prado, R.A.; Facchini, D.; Mahalanobis, N.; Gonzalez, F.; Palumbo, G. Electrodeposition of nanocrystalline cobalt alloy coatings as a hard chrome alternative. In Proceedings of the DoD Corrosion Conference, Gaylord National, Washington, DC, USA, 10–14 August 2009. [Google Scholar]
- Friedman, H.; Eidelman, O.; Feldman, Y.; Moshkovich, A.; Perfiliev, V.; Rapoport, I.; Cohen, H.; Yoffe, A.; Tenne, R. Fabrication of self-lubricating cobalt coatings on metal surface. Nanotechnology 2007, 18, 115703–115710. [Google Scholar] [CrossRef]
- Duruibe, J.O.; Ogwuegbu, M.O.C.; Egwurugwu, J.N. Heavy metal pollution and human biotoxic effects. Int. J. Phys. Sci. 2007, 2, 112–118. [Google Scholar]
- Martínez-Hernández, A.; Meas, Y.; Pérez-Bueno, J.; Ortíz-Frade, L.; Flores-Segura, J.; Méndez-Albores, A.; Trejo, G. Electrodeposition of Co-B hard coatings: Characterization and tribological properties. Int. J. Electrochem. Sci. 2017, 12, 1863–1873. [Google Scholar] [CrossRef]
- Méndez-Albores, A.; González-Arellano, S.G.; Reyes-Vidal, Y.; Torres, J.; Talu, S.; Cercado, B.; Trejo, G. Electrodeposited chrome/silver nanoparticle (Cr/AgNPs) composite coatings: Characterization and antibacterial activity. J. Alloy. Compd. 2017, 710, 302–311. [Google Scholar] [CrossRef]
- ASTM G99-05 Standard Test Method for Wear Testing with a Pin-on-Disk Apparatus; ASTM International: West Conshohocken, PA, USA, 2005.
- Cerruti, B.; de Souza, C.; Castellan, A.; Ruggiero, R.; Frollini, E. Carboxymethyl lignin as stabilizing agent in aqueous ceramic suspensions. Ind. Crops. Prod. 2012, 36, 108–115. [Google Scholar] [CrossRef]
- Malfati, C.F.; Ferreira, J.Z.; Oliveira, C.T.; Rieder, E.S.; Bonino, J.P. Electrochemical behavior of Ni-P/SiC composite coatings: Effect of heat treatment and SiC particle incorporation. Mater. Corros. 2012, 63, 36–43. [Google Scholar] [CrossRef]
- Hou, K.-H.; Hwu, W.-H.; Ke, S.-T.; Ger, M.-D. Ni-P/SiC composite produced by pulse and direct current plating. Mater. Chem. Phys. 2006, 100, 54–59. [Google Scholar] [CrossRef]
- González-Arellano, S.G.; Avilés-Arellano, L.M.R.; Manríquez, J.; Torres, J.; Ortega, R.; Meas, Y.; Trejo, G.; Méndez-Albores, A. Study of the stability of silver particles suspended using Cetylpyridinium Bromide (CPB) as surfactant. Chem. Sci. Rev. Lett. 2015, 4, 809–816. [Google Scholar]
- Bozzini, B.; Cavallotti, P.L.; Parisi, G. Corrosion and erosion of electrodeposited Ni-P/B4C composites. Brit. Corros. J. 2001, 36, 49–55. [Google Scholar] [CrossRef]
- Bozzini, B.; Lecis, N.; Cavallotti, P.L. Structure and Magnetic properties of nanocrystalline Ni-P prepared by autocatalytic chemical deposition. J. Phys. IV France 1998, 8, 371–374. [Google Scholar] [CrossRef]
- Lim, T.; Kim, J.J. Effect of B and W contents on hardness of electroless Co alloys thin films. Korean Chem. Eng. Res. 2018, 56, 895–900. [Google Scholar] [CrossRef]
- Wang, L.; Gao, Y.; Xu, T.; Xue, Q. Corrosion resistance and lubricated sliding wear behavior of novel Ni-P graded alloy as alternative to hard Cr deposits. Appl. Surf. Sci. 2006, 252, 7361–7372. [Google Scholar] [CrossRef]
- Lee, K.H.; Chang, D.S.; Know, C. Properties of electrodeposited nanocrystalline Ni-B alloys films. Electrochim. Acta 2005, 50, 4538–4543. [Google Scholar] [CrossRef]
- Aslanyan, I.R.; Bonino, J.-P.; Celis, J.-P. Effect of reinforcing submicron SiC particles on the wear of electrolytic NiP coatings, Part 2: Bi-directional sliding. Surf. Coat. Technol. 2006, 201, 581–589. [Google Scholar] [CrossRef]
- Krishnaveni, K.; Sankara Narayanan, T.S.N.; Seshadri, S.K. Electrodeposited Ni-B coatings: formation and evaluation of hardness and wear resistance. Mater. Chem. Phys. 2006, 99, 300–308. [Google Scholar] [CrossRef]
- Monteiro, O.R.; Murugesan, S.; Khabashesku, V. Electrodeposited Ni-B and Ni-B metal matrix diamond nanocomposite coatings. Surf. Coat. Technol. 2015, 272, 291–297. [Google Scholar] [CrossRef]
- Benea, L.; Bonora, P.L.; Berello, A.; Martelli, S. Wear corrosion properties of nanostructured SiC nickel composite coatings obtained by electroplating. Wear 2002, 249, 995–1003. [Google Scholar] [CrossRef]



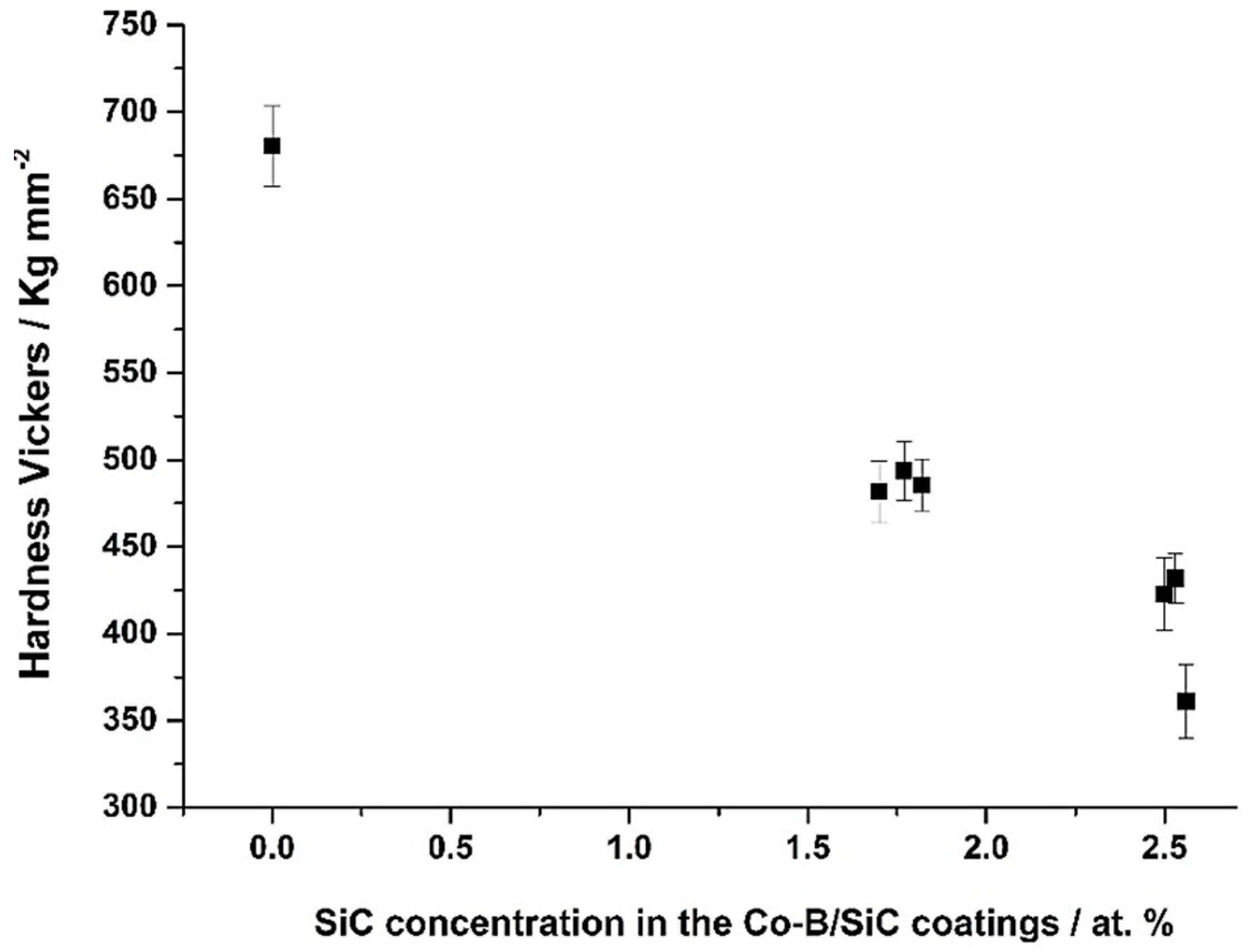

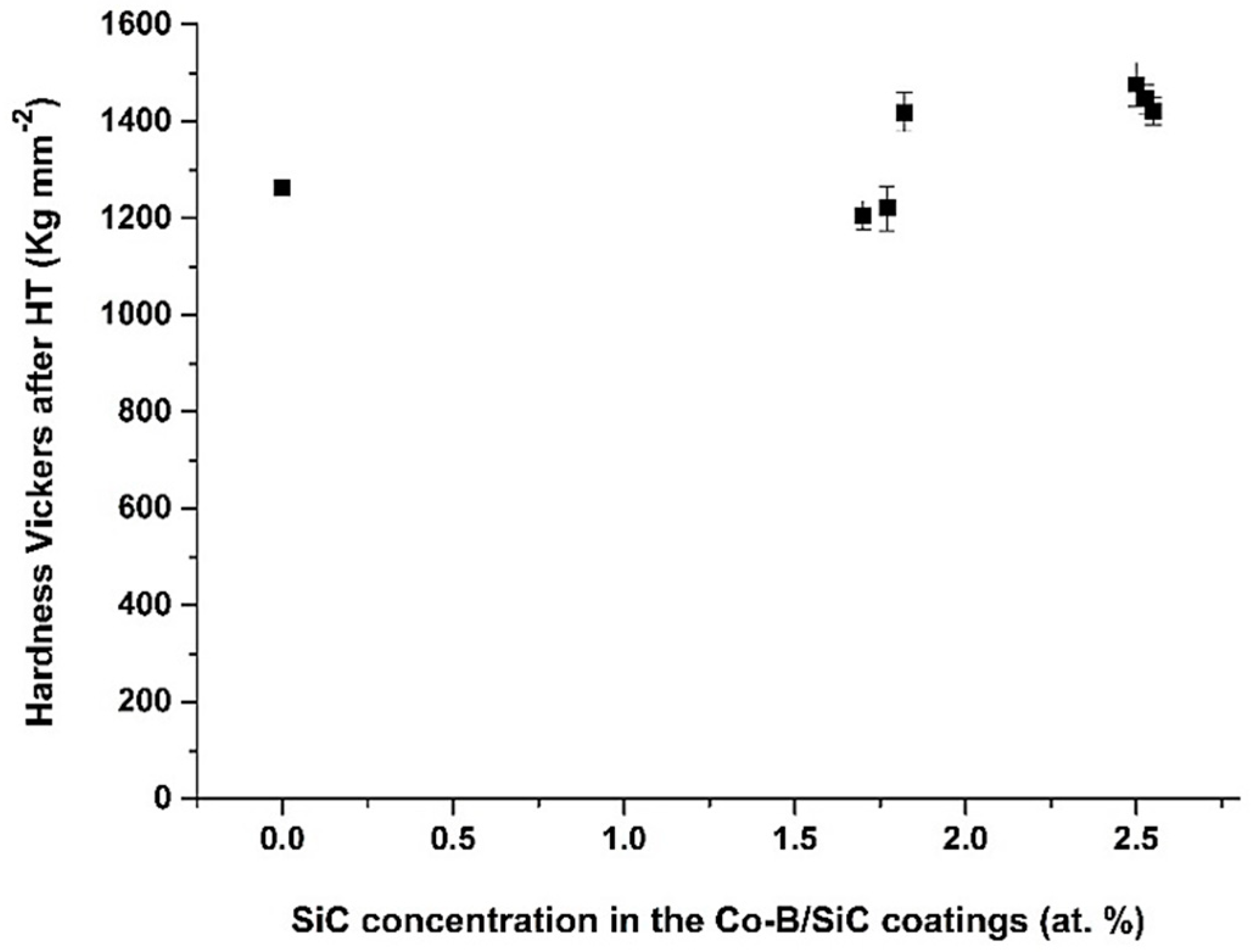
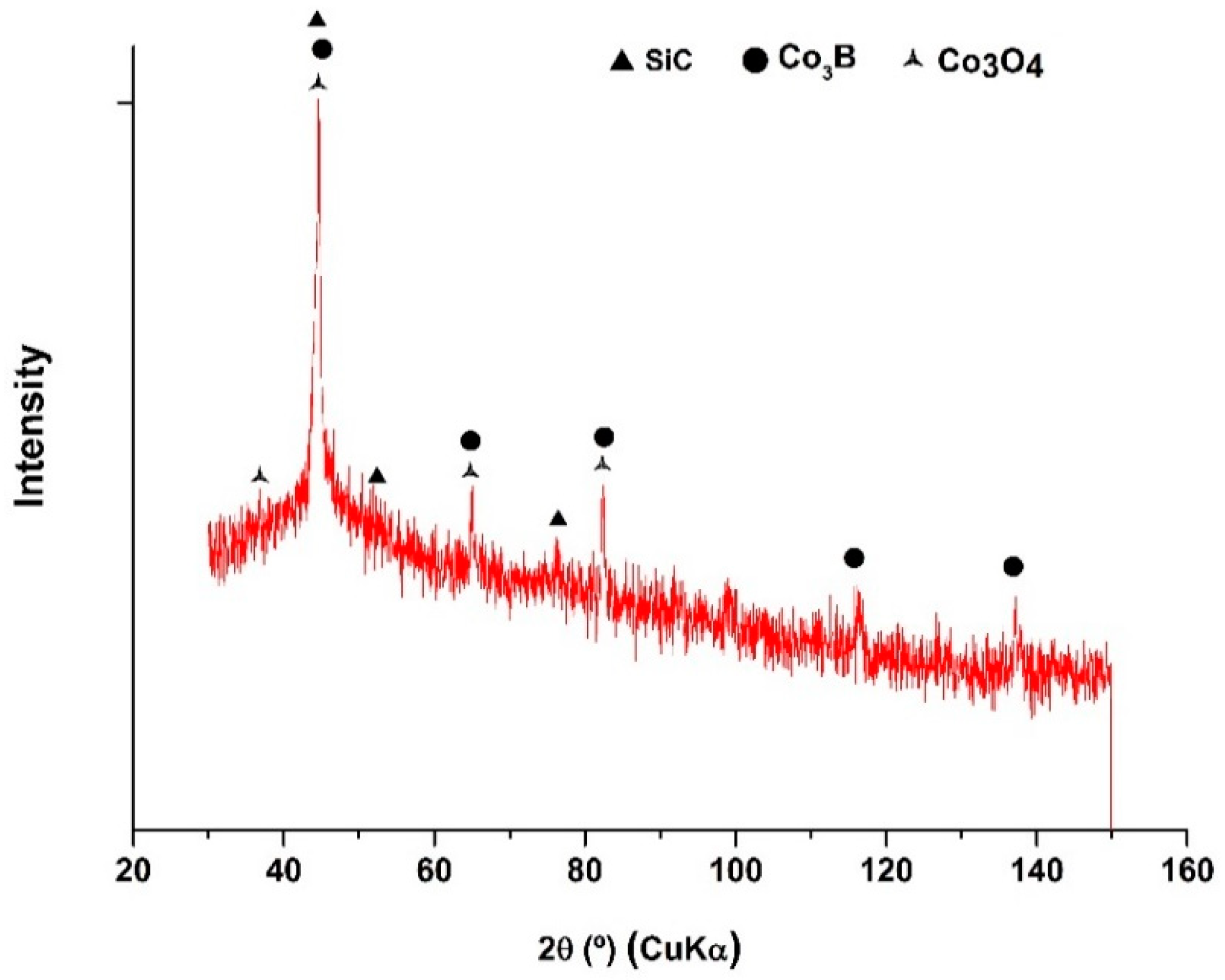
| SiC Concentration in the Colloidal Suspensions (g L−1) | SiC in the Coating (at.%) | Wear Volumen Loss (×10−7 mm3 N−1 m−1) | Friction Coefficient (μ) |
|---|---|---|---|
| 0 | 0 | 11.80 ± 0.13 | 0.444 ± 0.03 |
| 0.15 | 1.70 | 3.49 ± 0.19 | 0.417 ± 0.008 |
| 0.60 | 1.77 | 2.83 ± 0.21 | 0.353 ± 0.012 |
| 2.50 | 1.82 | 0.46 ± 0.12 | 0.304 ± 0.008 |
| 5.00 | 2.50 | 0.23 ± 0.08 | 0.312 ± 0.010 |
| 10.00 | 2.54 | 0.23 ± 0.10 | 0.313 ± 0.010 |
| 15.00 | 2.56 | 0.27 ± 0.11 | 0.312 ± 0.082 |
| SiC Concentration in the Colloidal Suspensions (g L−1) | SiC in the Coating (at.%) | Wear Volumen Loss after HT (×10−7 mm3 N−1 m−1) | Friction Coefficient after HT (μ) |
|---|---|---|---|
| 0 | 0 | 3.82 ± 0.09 | 0.260 ± 0.03 |
| 0.15 | 1.70 | 2.72 ± 0.11 | 0.255 ± 0.01 |
| 0.60 | 1.77 | 1.32 ± 0.08 | 0.245 ± 0.03 |
| 2.50 | 1.82 | 0.93 ± 0.10 | 0.238 ± 0.03 |
| 5.00 | 2.50 | 0.44 ± 0.08 | 0.213 ± 0.08 |
| 10.00 | 2.54 | 0.35 ± 0.10 | 0.215 ± 0.02 |
| 15.00 | 2.56 | 0.19 ± 0.07 | 0.208 ± 0.01 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Villa-Mondragón, A.; Martínez-Hernández, A.; Manríquez, F.; Meas, Y.; Pérez-Bueno, J.J.; Rodríguez-Valadez, F.J.; Ballesteros, J.C.; Morales-Hernández, J.; Méndez-Albores, A.; Trejo, G. Electrodeposition of Co-B/SiC Composite Coatings: Characterization and Evaluation of Wear Volume and Hardness. Coatings 2019, 9, 279. https://doi.org/10.3390/coatings9040279
Villa-Mondragón A, Martínez-Hernández A, Manríquez F, Meas Y, Pérez-Bueno JJ, Rodríguez-Valadez FJ, Ballesteros JC, Morales-Hernández J, Méndez-Albores A, Trejo G. Electrodeposition of Co-B/SiC Composite Coatings: Characterization and Evaluation of Wear Volume and Hardness. Coatings. 2019; 9(4):279. https://doi.org/10.3390/coatings9040279
Chicago/Turabian StyleVilla-Mondragón, A., A. Martínez-Hernández, F. Manríquez, Y. Meas, J. J. Pérez-Bueno, Francisco J. Rodríguez-Valadez, J.C. Ballesteros, J. Morales-Hernández, Alia Méndez-Albores, and G. Trejo. 2019. "Electrodeposition of Co-B/SiC Composite Coatings: Characterization and Evaluation of Wear Volume and Hardness" Coatings 9, no. 4: 279. https://doi.org/10.3390/coatings9040279
APA StyleVilla-Mondragón, A., Martínez-Hernández, A., Manríquez, F., Meas, Y., Pérez-Bueno, J. J., Rodríguez-Valadez, F. J., Ballesteros, J. C., Morales-Hernández, J., Méndez-Albores, A., & Trejo, G. (2019). Electrodeposition of Co-B/SiC Composite Coatings: Characterization and Evaluation of Wear Volume and Hardness. Coatings, 9(4), 279. https://doi.org/10.3390/coatings9040279







