Microstructures and Mechanical Properties of Cu-Coated SiC Particles Reinforced AZ61 Alloy Composites
Abstract
1. Introduction
2. Experimental Procedure
2.1. Raw Materials
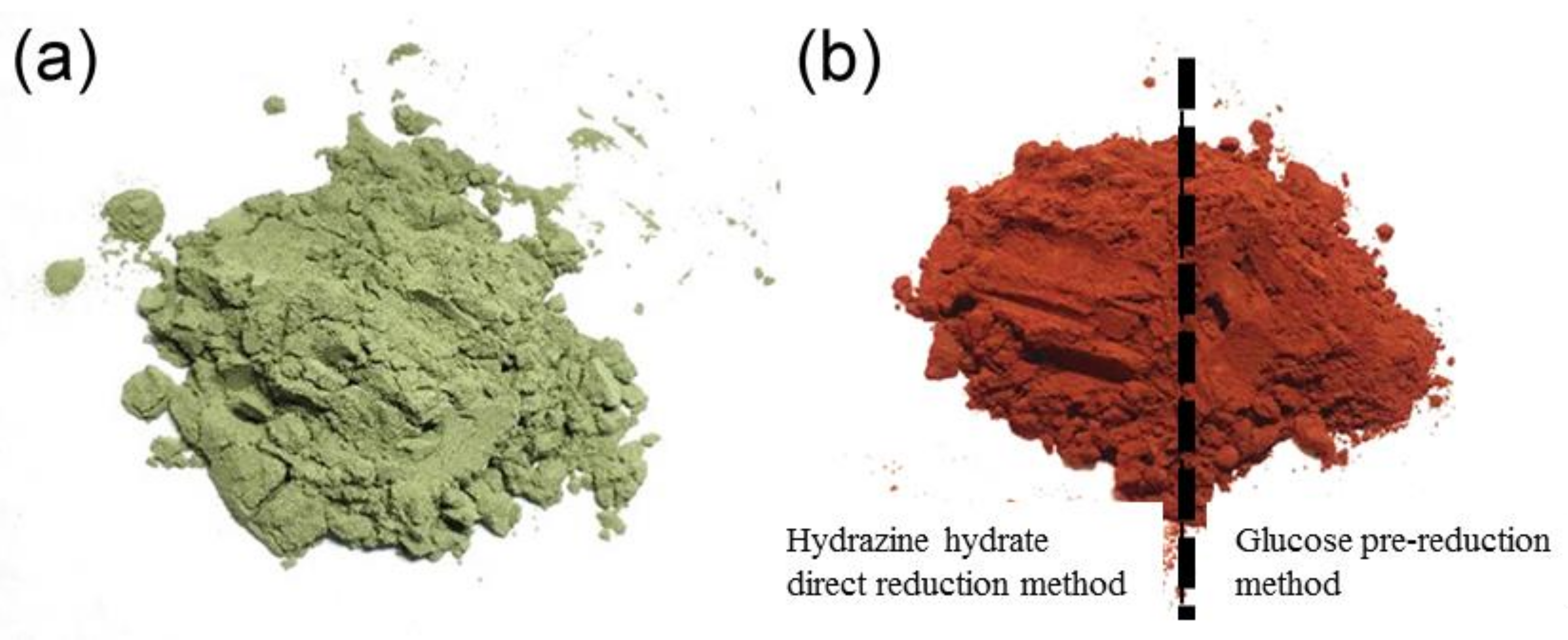
2.2. Surface Modification of SiCP
- (1)
- Hydrazine hydrate direct reduction method.
- (2)
- Glucose pre-reduction method.
2.3. Preparation of SiCP/AZ61 Composites
2.4. Materials Characterization
3. Results and Discussion
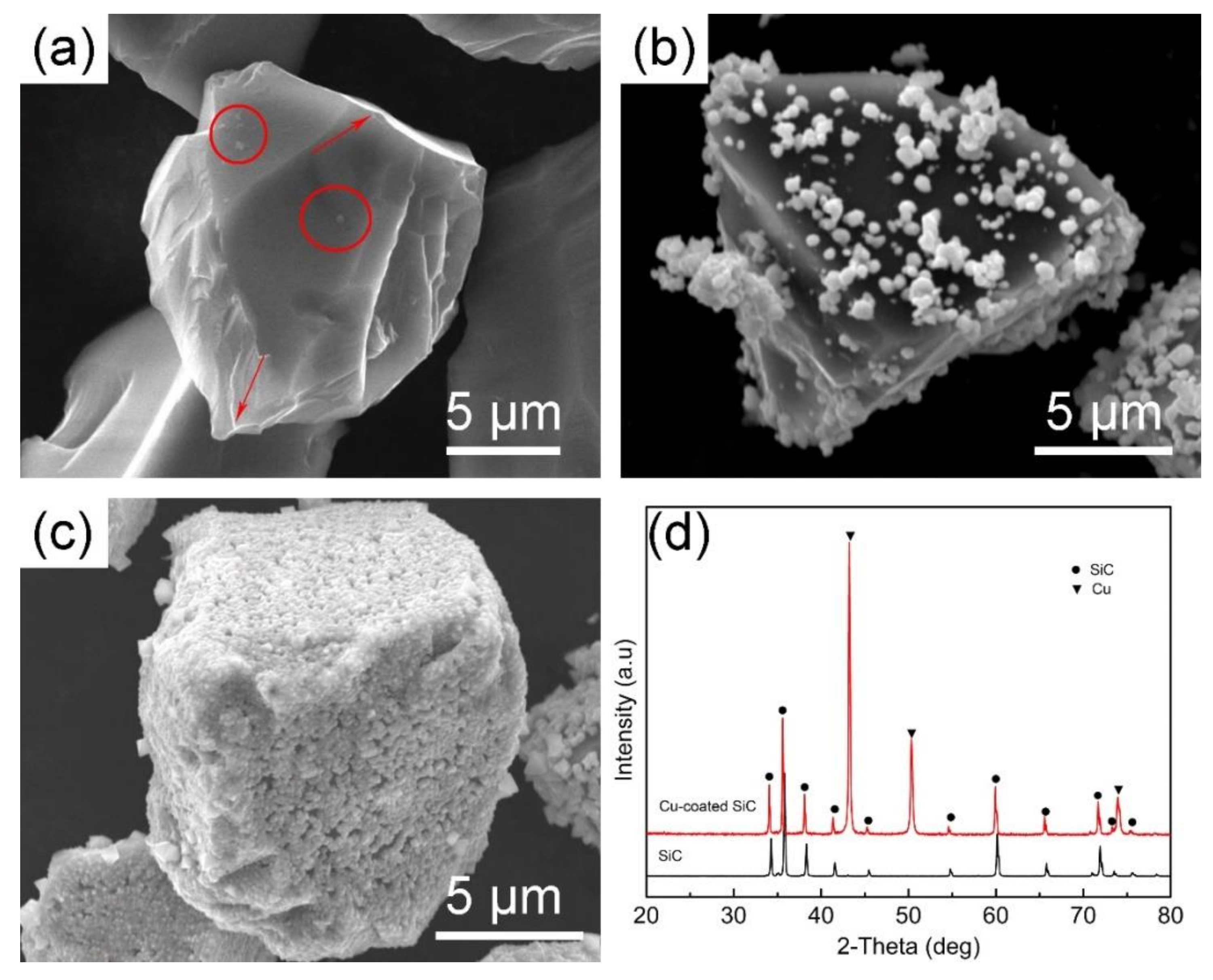
3.1. Investigation on Electroless Methods of Cu Plating
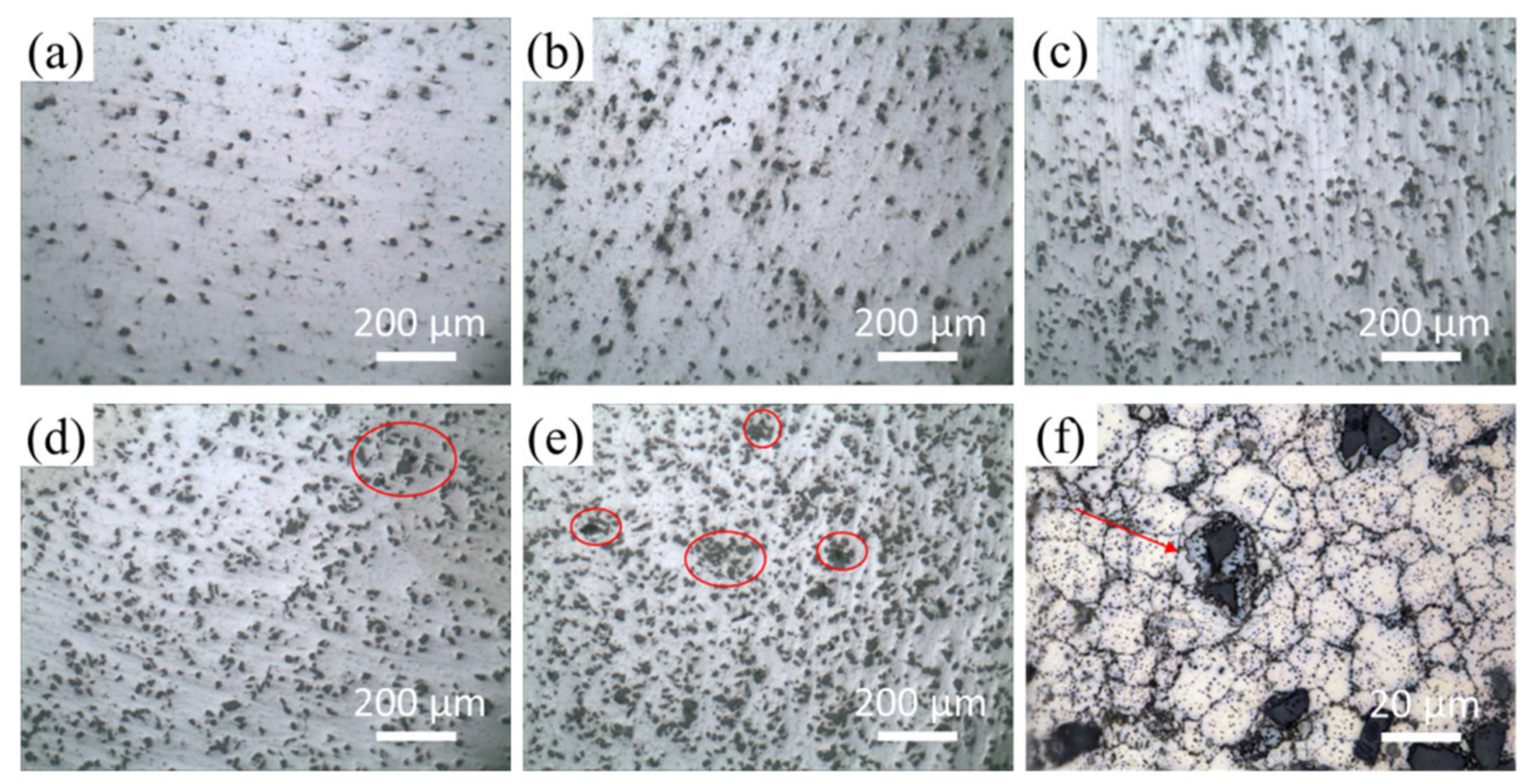
3.2. Microstructures of SiCP/AZ61 Composites
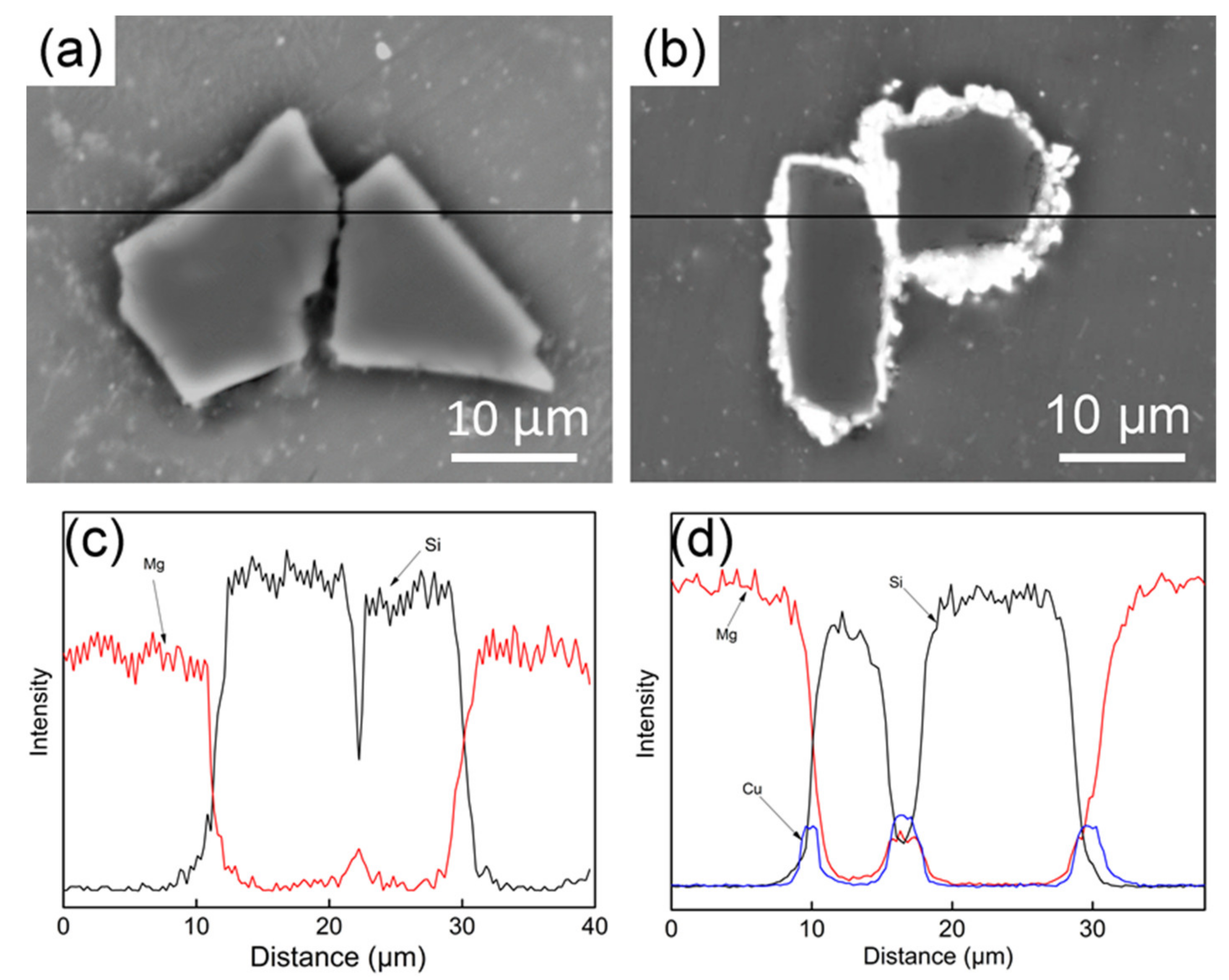
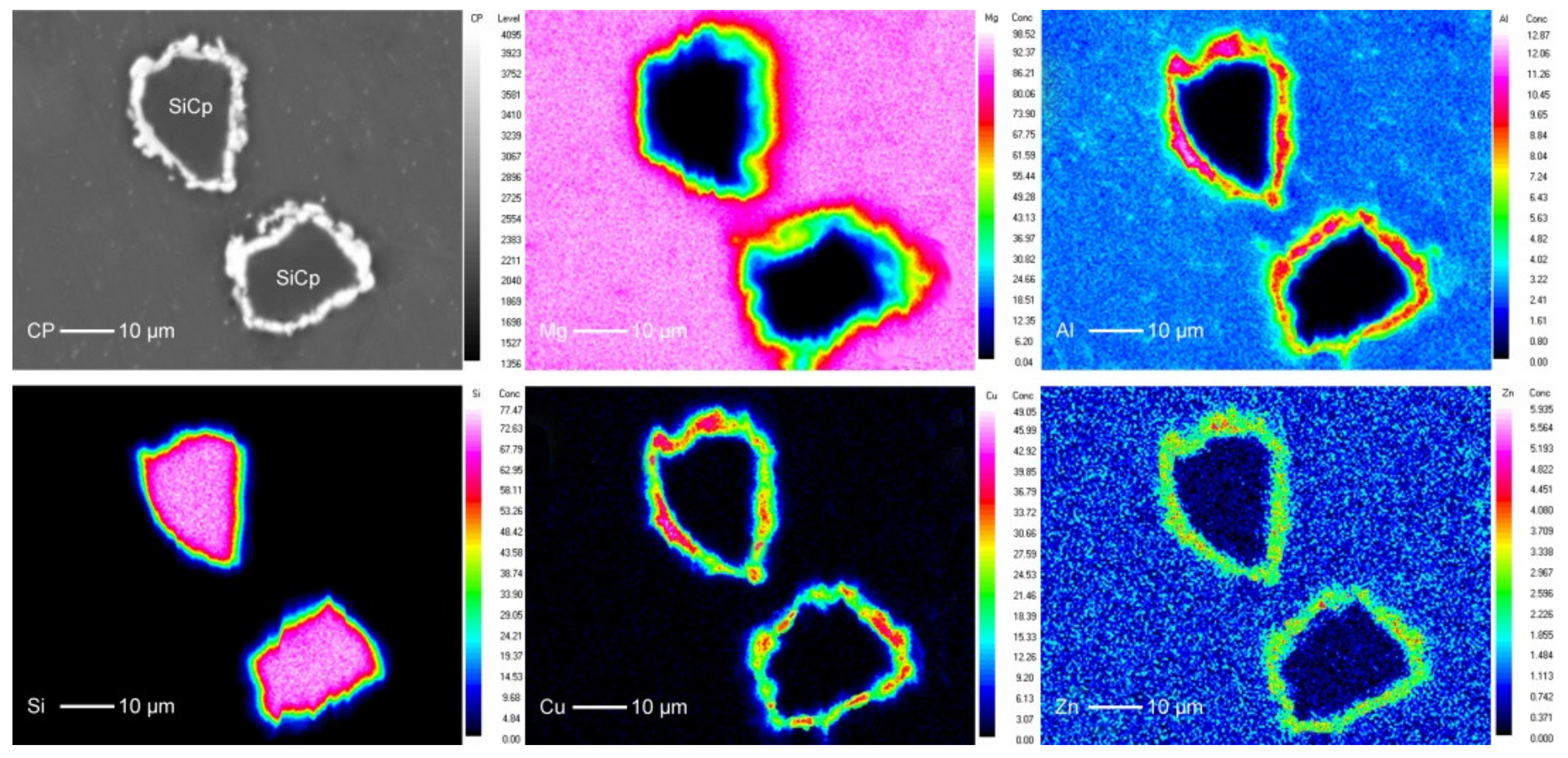
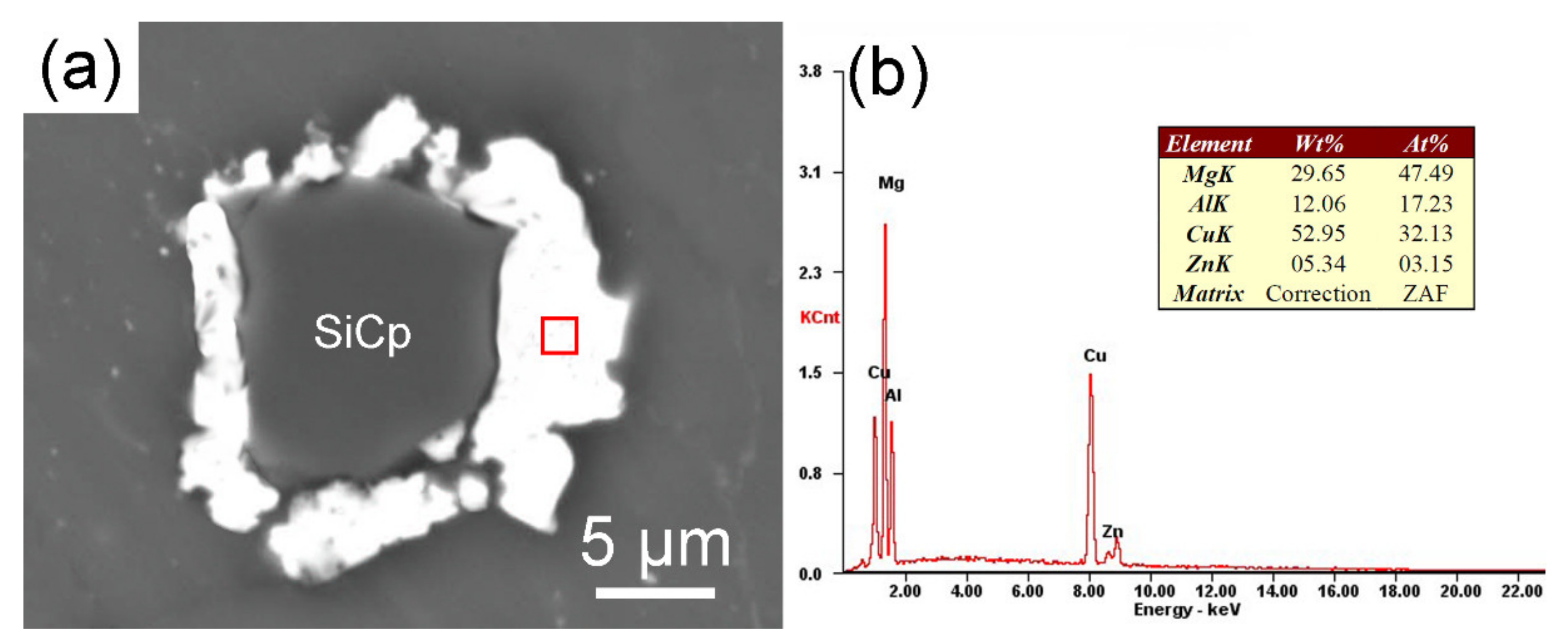
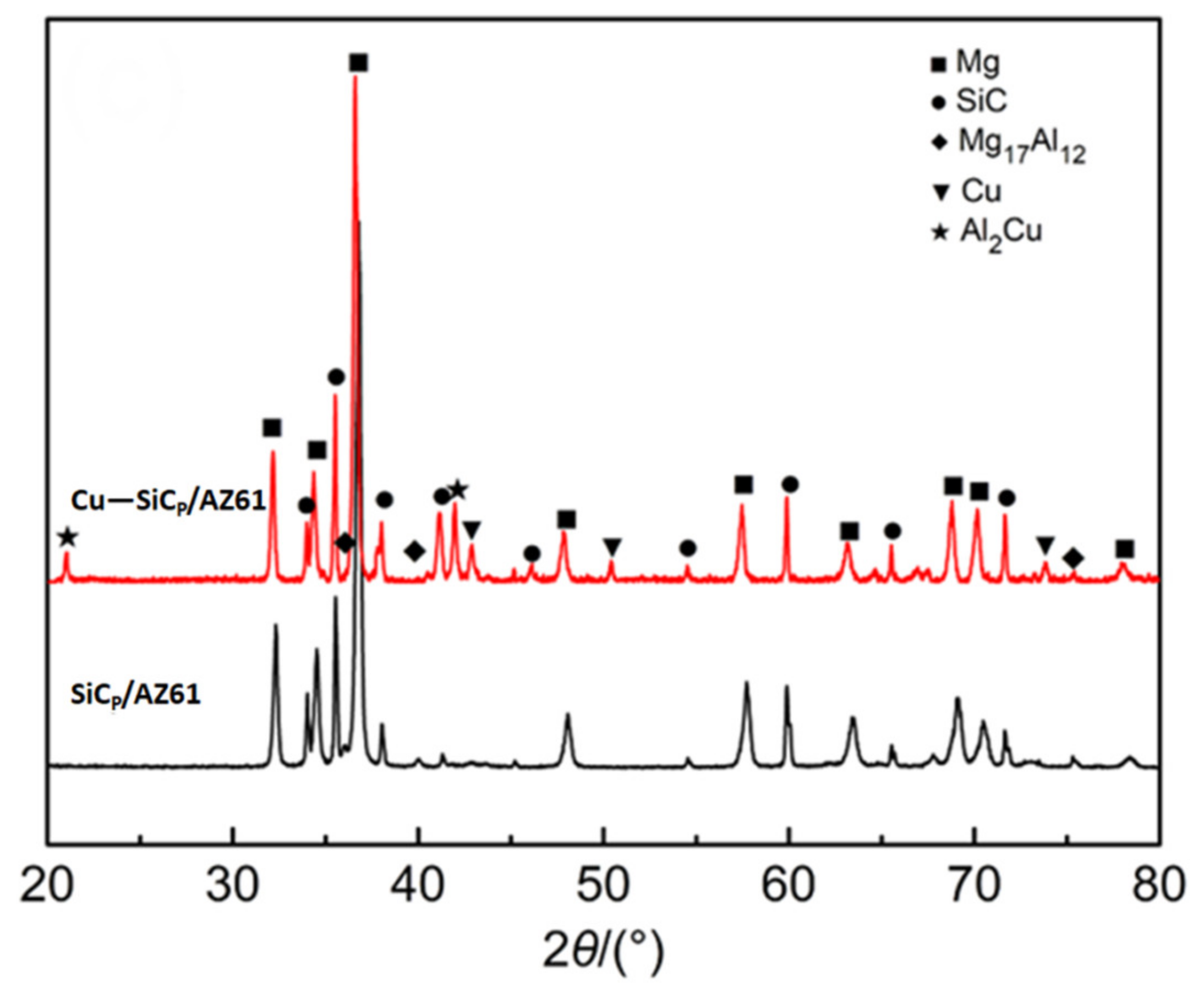
3.3. Interface of SiCP/AZ61 Composites
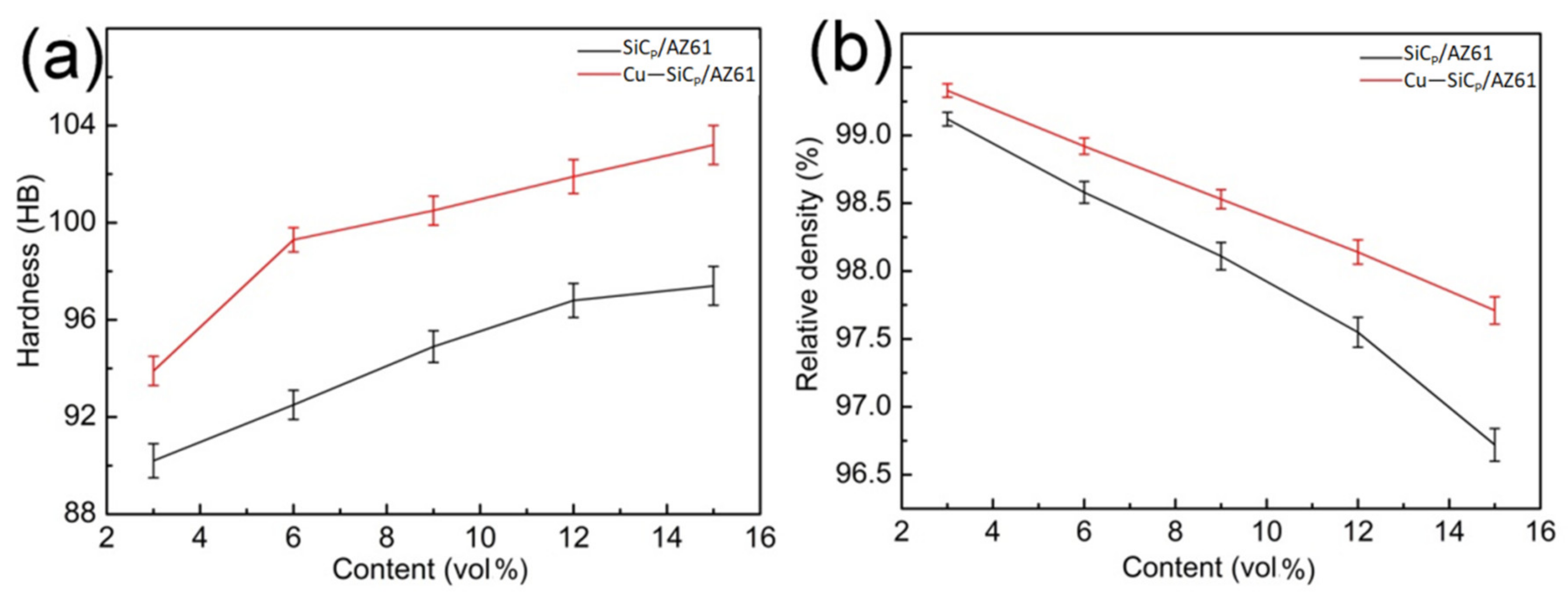
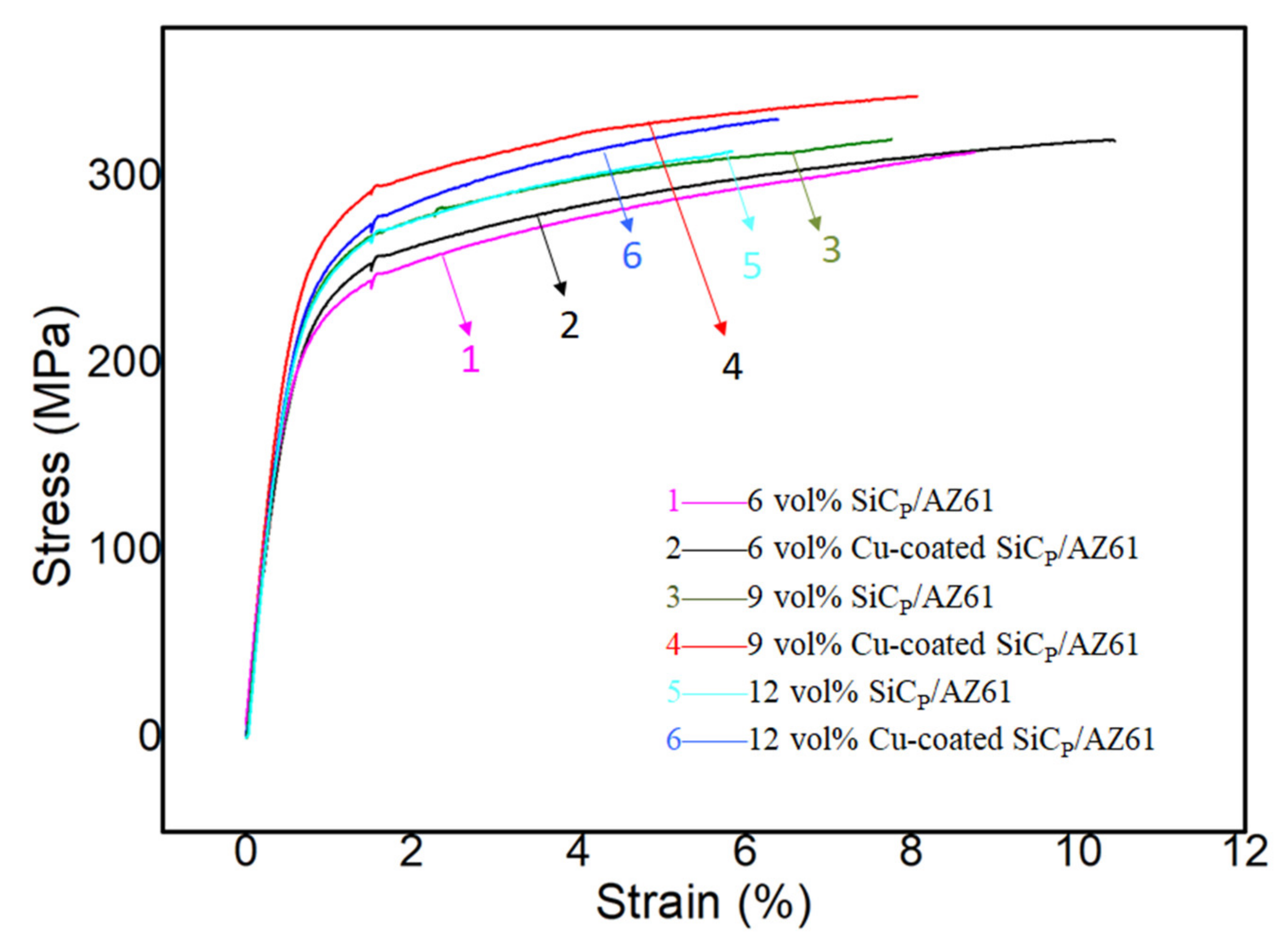
3.4. Mechanical Properties of SiCP/AZ61 Composites
4. Conclusions
- (1)
- The Cu layer prepared by the pre-reduction method of glucose using hydrazine hydrate as reducing agent is more uniform and complete than the one prepared by direct reduction of hydrazine hydrate. Therefore, the pre-reduction method is more suitable for copper plating on the surface of SiCP.
- (2)
- The Cu layer fabricated by electroless plating is beneficial for the wettability and interfacial bonding strength between the SiCP and AZ61 matrix. The microstructure of Cu–SiCP/AZ61 composites shows a significant decrease in micro-porosity and other defects.
- (3)
- XRD phase analysis shows that θ phase (Al2Cu) has formed in Cu–SiCP/AZ61. Combined with the EPMA element mapping and EDS spectrum quantitative analysis, it is further inferred the formation of η phase (MgZn2).
- (4)
- Cu plating on the surface of SiCP has positive effect on the hardness, relative density, tensile strength, and elongation of the composite. With the increase of SiCP volume content, defects such as voidsare more likely to occur in the composite. While the Cu layer on the surface of SiCP can reduce the porosity effectively and enhanced the interfacial bonding strength between SiCP and AZ61 matrix. Besides, the Cu coating on the surface of SiCP plays as a transition layer to make sure the load transfersfrom the matrix to the SiC particles effectively, thereby increasing the ultimate tensile strength, and elongation of the composite. The best mechanical properties are obtained when the appropriate volume fraction of SiCP is 9% and the hardness, ultimate tensile strength, and elongation are 100.5 HB, 335.67 MPa, and 7.91%, respectively.
Author Contributions
Funding
Conflicts of Interest
References
- Cui, Z.; Zhang, Y.; Cheng, Y.; Gong, D.; Wang, W. Microstructure, mechanical, corrosion properties and cytotoxicity of betacalcium polyphosphate reinforced ZK61 magnesium alloy composite by spark plasma sintering. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 99, 1035–1047. [Google Scholar] [CrossRef] [PubMed]
- Lim, P.N.; Lam, R.N.; Zheng, Y.F.; Thian, E.S. Magnesium-calcium/hydroxyapatite (Mg-Ca/HA) composites with enhanced bone differentiation properties for orthopedic applications. Mater. Lett. 2016, 172, 193–197. [Google Scholar] [CrossRef]
- Czerwinski, F. Controlling the ignition and flammability of magnesium for aerospace applications. Corros. Sci. 2014, 86, 1–16. [Google Scholar] [CrossRef]
- Mallick, A.; Tun, K.S.; Gupta, M. Deformation behaviour of Mg/Y2O3 nanocomposite at elevated temperatures. Mater. Sci. Eng. A 2012, 551, 222–230. [Google Scholar] [CrossRef]
- Bommala, V.K.; Krishna, M.G.; Rao, C.T. Magnesium matrix composites for biomedical applications: A review. J. Magnes. Alloys 2019, 7, 72–79. [Google Scholar] [CrossRef]
- Ye, H.Z.; Liu, X.Y. Review of recent studies in magnesium matrix composites. J. Mater. Sci. 2004, 39, 6153–6171. [Google Scholar] [CrossRef]
- Chen, Q.; Chen, G.; Han, F.; Xia, X.; Wu, Y. Microstructures, mechanical properties, and wear resistances of thixoextruded SiCP/WE43 magnesium matrix composites. Miner. Met. Meter. Soc. ASM Int. 2017, 48, 3497–3513. [Google Scholar] [CrossRef]
- Zhao, W.; Huang, S.-J.; Wu, Y.-J.; Kang, C.-W. Particle Size and Particle Percentage Effect of AZ61/SiCp Magnesium Matrix Micro- and Nano-Composites on Their Mechanical Properties Due to Extrusion and Subsequent Annealing. Metals 2017, 7, 293. [Google Scholar] [CrossRef]
- Zulkoffli, Z.; Syarif, J.; Sajuri, Z. Fabrication of AZ61/SiC composites by powder metallurgy process. Int. J. Mech. Mater. Eng. 2009, 4, 156–159. [Google Scholar]
- Zhou, T.W.; Liu, X.R.; Zhang, R.L.; Su, Z.Z. Preparation and Properties of Magnesium-Aluminum Matrix Composites Reinforced with Silicon Carbon Particles. Adv. Mater. Res. 2012, 583, 3–8. [Google Scholar] [CrossRef]
- Yong, H.; He, B.L.; Hong, Y. Rheological behavior of semi-solid Mg 2Si/AM60 magnesium matrix composites at steady state. Trans. Nonferr. Met. Soc. Chin. 2010, 20, s883–s887. [Google Scholar]
- Wang, H.; Zhang, R.; Xu, H.; Lu, H.; Guan, S. Preparation of Al-based Metal Matrix Composites Reinforced by Cu Coated SiC Particles. Key Eng. Mater. 2005, 280, 1493–1496. [Google Scholar]
- Gupta, M.; Lai, M.O.; Saravanaranganathan, D. Synthesis, microstructure and properties characterization of disintegrated melt deposited Mg/SiC composites. J. Mater. Sci. 2000, 35, 2155–2165. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, Q.; Liao, W.; Guo, W.; Ye, B.; Li, W.; Jiang, H.; Ding, W. Effects of cyclic extrusion and compression on the microstructure and mechanical properties of AZ91D magnesium composites reinforced by SiC nanoparticles. Mater. Charact. 2017, 126, 17–27. [Google Scholar] [CrossRef]
- Esmaily, M.; Mortazavi, N.; Svensson, J.E.; Halvarsson, M.; Wessén, M.; Johansson, L.G.; Jarfors, A.E. A new semi-solid casting technique for fabricating SiC-reinforced Mg alloys matrix composites. Compos. Part B Eng. 2016, 94, 176–189. [Google Scholar] [CrossRef]
- Trojanová, Z.; Gärtnerová, V.; Jäger, A.; Námešný, A.; Chalupová, M.; Palček, P.; Lukáč, P. Mechanical and fracture properties of an AZ91 Magnesium alloy reinforced by Si and SiC particles. Compos. Sci. Technol. 2009, 69, 2256–2264. [Google Scholar]
- Song, H.Y.; Zha, X.W. Influence of nickel coating on the interfacial bonding characteristics of carbon nanotube–aluminum composites. Comput. Mater. Sci. 2010, 49, 899–903. [Google Scholar] [CrossRef]
- Guo, S.Q.; Wang, R.C.; Peng, C.Q.; Cai, Z.Y.; Dong, C.G. Microstructures and mechanical properties of Ni-coated SiC particles reinforced AZ61 alloy composites. Trans. Nonferr. Met. Soc. Chin. 2019, 29, 1854–1863. [Google Scholar] [CrossRef]
- Chen, S.Y.; Li, X.R.; An, D.; Liang, J.; Liu, C.S. Preparation and Wear Performance of Novel Graphite/Copper Alloy-Matrix Self-Lubricating Composite Materials. Adv. Mater. Res. 2014, 941, 284–287. [Google Scholar] [CrossRef]
- Hong, T.W.; Kim, S.K.; Ha, H.S.; Kim, M.G.; Lee, D.B.; Kim, Y.J. Microstructural evolution and semisolid forming of SiC particulate reinforced AZ91HP magnesium composites. Mater. Sci. Technol. 2000, 16, 887–892. [Google Scholar] [CrossRef]
- Chowdhury, P.P.; Shaik, A.H.; Chakraborty, J. Preparation of stable sub 10 nm copper nanopowders redispersible in polar and non-polar solvents. Colloids Surf. A Physicochem. Eng. Asp. 2015, 466, 189–196. [Google Scholar] [CrossRef]
- Saikova, S.V.; Vorob’ev, S.A.; Nikolaeva, R.B.; Mikhlin, Y.L. Conditions for the formation of copper nanoparticles by reduction of copper (II) ions with hydrazine hydrate solutions. Russ. J Gen. Chem. 2010, 80, 1122–1127. [Google Scholar] [CrossRef]
- Peng, Y.H.; Lee, C.H.; Popuri, S.R.; Kumar, K.N.S. Preparation of High-Purity Ultrafine Copper Powder in Mass-Production by Chemical Reduction Method: Taguchi Robust Design Optimization. Powder Metall. Met. Ceram. 2016, 55, 386–396. [Google Scholar] [CrossRef]
- Hassan, S.F.; Gupta, M. Development of high strength magnesium based composites using elemental nickel particulates as reinforcement. J. Mater. Sci. 2002, 37, 2467–2474. [Google Scholar] [CrossRef]
- Chen, L.Y.; Konishi, H.; Fehrenbacher, A.; Ma, C.; Xu, J.Q.; Choi, H.; Xu, H.F.; Pfefferkorn, F.E.; Li, X.C. Novel nanoprocessing route for bulk graphene nanoplatelets reinforced metal matrix nanocomposites. Scr. Mater. 2012, 67, 29–32. [Google Scholar] [CrossRef]
- Yang, N.; Boselli, J.; Sinclair, I. Simulation and quantitative assessment of homogeneous and inhomogeneous particle distributions in particulate metal matrix composites. J. Microsc. 2001, 201, 189–200. [Google Scholar] [CrossRef]
- Iacob, G.; Ghica, V.G.; Buzatu, M.; Buzatu, T.; Petrescu, M.I. Studies on wear rate and micro-hardness of the Al/Al2O3/Gr hybrid composites produced via powder metallurgy. Compos. Part B Eng. 2015, 69, 603–611. [Google Scholar] [CrossRef]
- Kim, T.W. Determination of densification behavior of Al–SiC metal matrix composites during consolidation processes. Mater. Sci. Eng. A 2008, 483, 648–651. [Google Scholar] [CrossRef]
- Chaubey, A.K.; Mohapatra, S.; Jayasankar, K.; Pradhan, S.K.; Satpati, B.; Sahay, S.S.; Mishra, B.K.; Mukherjee, P.S. Effect of cerium addition on microstructure and mechanical properties of Al-Zn-Mg-Cu alloy. Trans. Indian Inst. Met. 2009, 62, 539–543. [Google Scholar] [CrossRef]
- Li, H.; Cao, F.; Guo, S.; Ning, Z.; Liu, Z.; Jia, Y.; Scudino, S.; Gemming, T.; Sun, J. Microstructures and properties evolution of spray-deposited Al-Zn-Mg-Cu-Zr alloys with scandium addition. J. Alloys. Compd. 2017, 691, 482–488. [Google Scholar] [CrossRef]
- Ohno, M.; Mirkovic, D.; Schmidfetzer, R. Liquidus and solidus temperatures of Mg-rich Mg–Al–Mn–Zn alloys. Acta Mater. 2006, 54, 3883–3891. [Google Scholar] [CrossRef]
- Xu, Z.; Ma, L.; Yan, J.; Chen, W.; Yang, S. Solidification microstructure of SiC particulate reinforced Zn–Al composites under ultrasonic exposure. Mater. Chem. Phys. 2014, 148, 824–832. [Google Scholar] [CrossRef]
- Chen, B.; Li, S.; Imai, H.; Jia, L.; Umeda, J.; Takahashi, M.; Kondoh, K. Load transfer strengthening in carbon nanotubes reinforced metal matrix composites via in-situ tensile tests. Compos. Sci. Technol. 2015, 113, 1–8. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dong, C.; Wang, R.; Guo, S. Microstructures and Mechanical Properties of Cu-Coated SiC Particles Reinforced AZ61 Alloy Composites. Coatings 2019, 9, 820. https://doi.org/10.3390/coatings9120820
Dong C, Wang R, Guo S. Microstructures and Mechanical Properties of Cu-Coated SiC Particles Reinforced AZ61 Alloy Composites. Coatings. 2019; 9(12):820. https://doi.org/10.3390/coatings9120820
Chicago/Turabian StyleDong, Cuige, Richu Wang, and Suqing Guo. 2019. "Microstructures and Mechanical Properties of Cu-Coated SiC Particles Reinforced AZ61 Alloy Composites" Coatings 9, no. 12: 820. https://doi.org/10.3390/coatings9120820
APA StyleDong, C., Wang, R., & Guo, S. (2019). Microstructures and Mechanical Properties of Cu-Coated SiC Particles Reinforced AZ61 Alloy Composites. Coatings, 9(12), 820. https://doi.org/10.3390/coatings9120820



