Anti-Graffiti Behavior of Oleo/Hydrophobic Nano-Filled Coatings Applied on Natural Stone Materials
Abstract
1. Introduction
2. Materials and Methods
2.1. Protective Products, Stone Materials, and Staining Agents
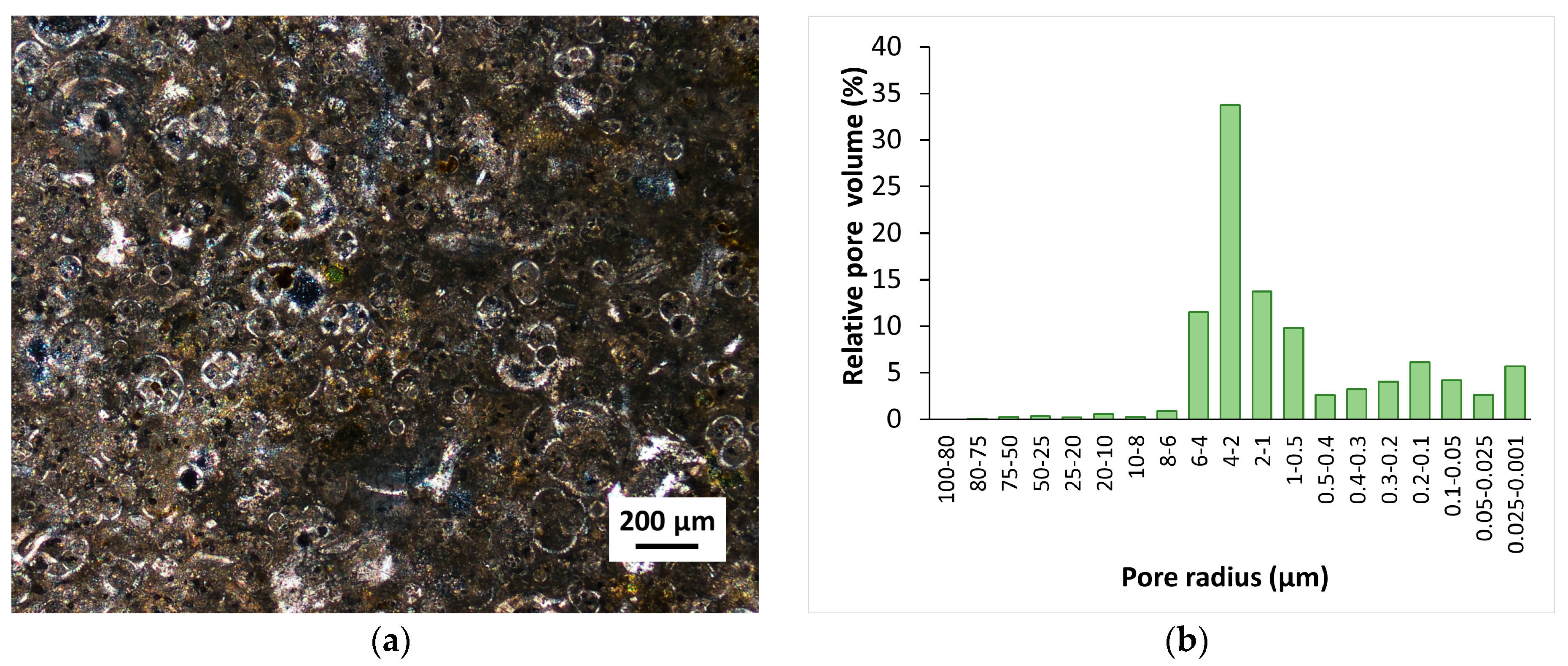
2.2. Stone Specimens
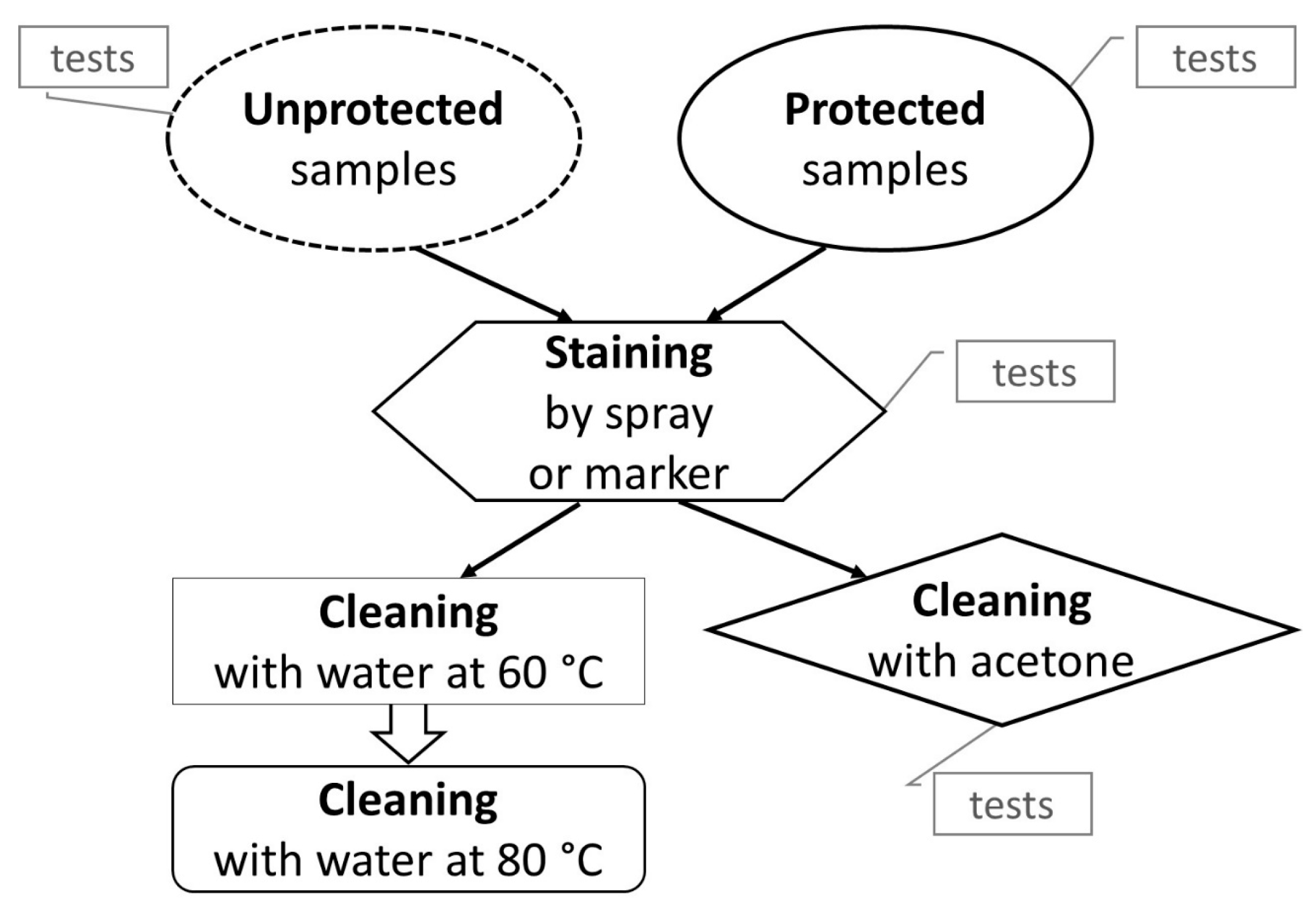
2.3. Staining Methods and Removal Procedures
2.3.1. Spray Paint
2.3.2. Felt-Tip Paint Marker
2.4. Analytical Investigations
3. Results and Discussion
3.1. Assessment of Basic Prerequisite for Antigraffiti Protection
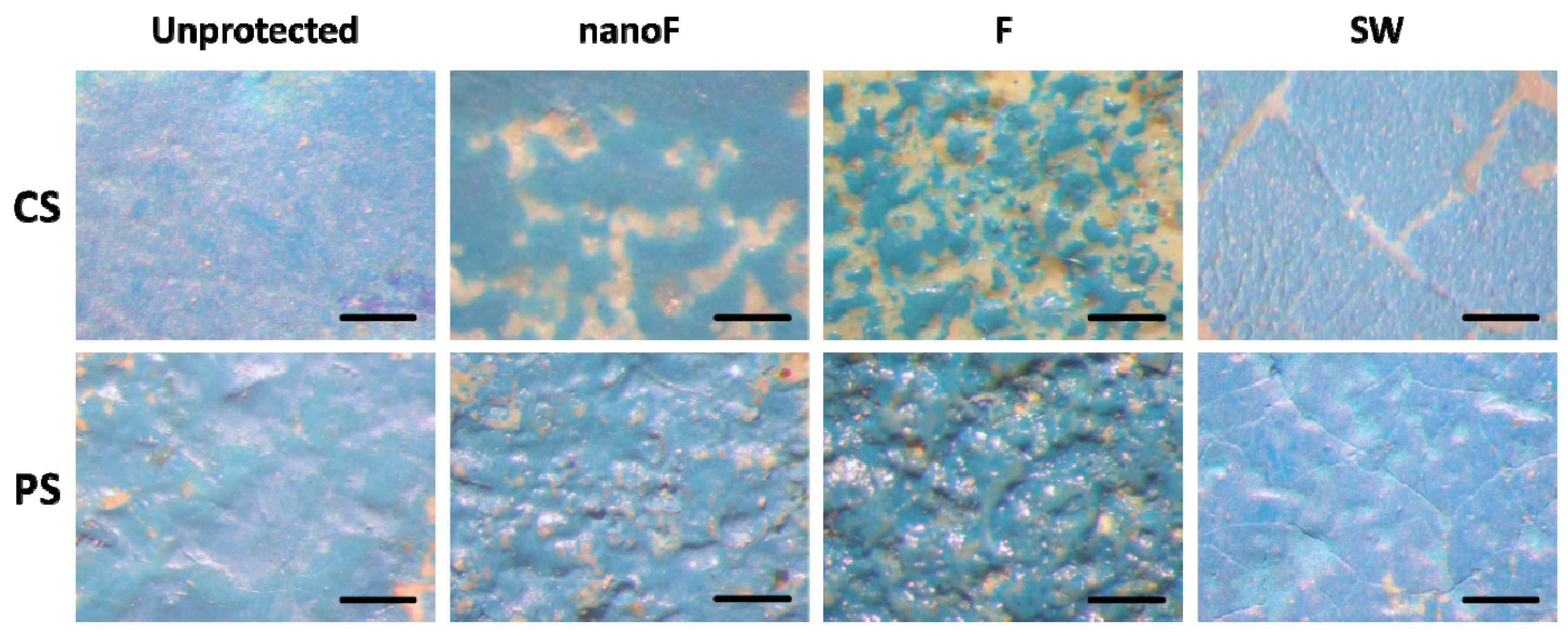
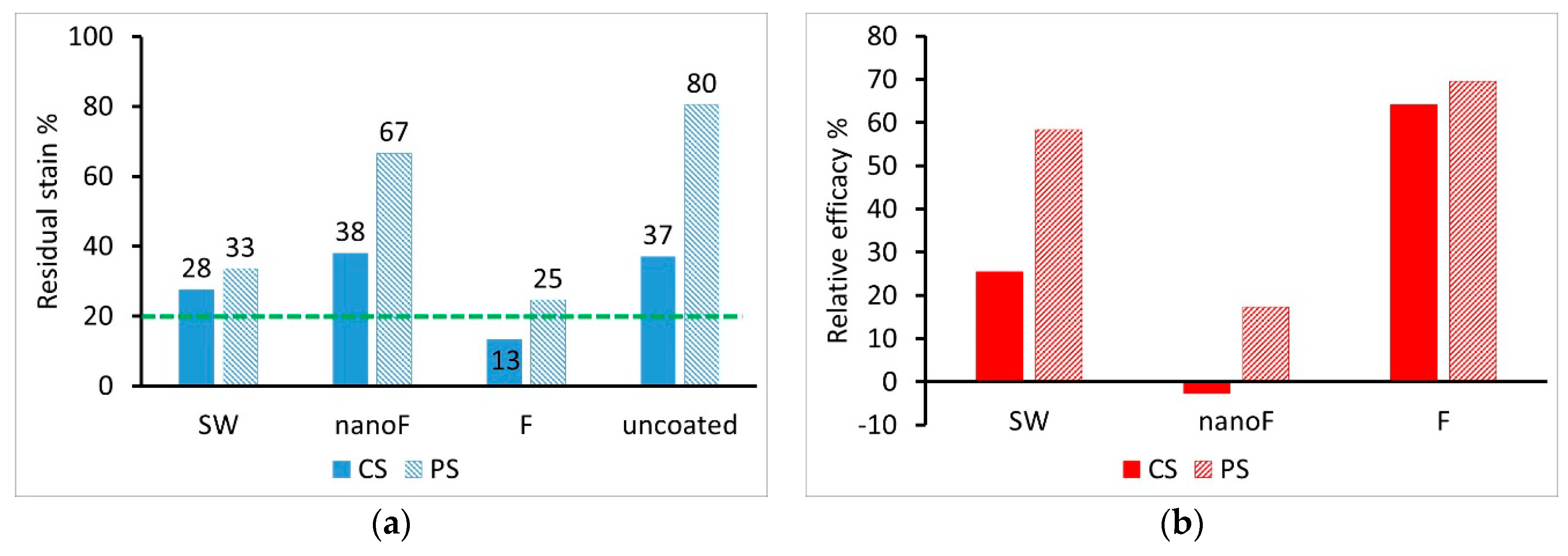
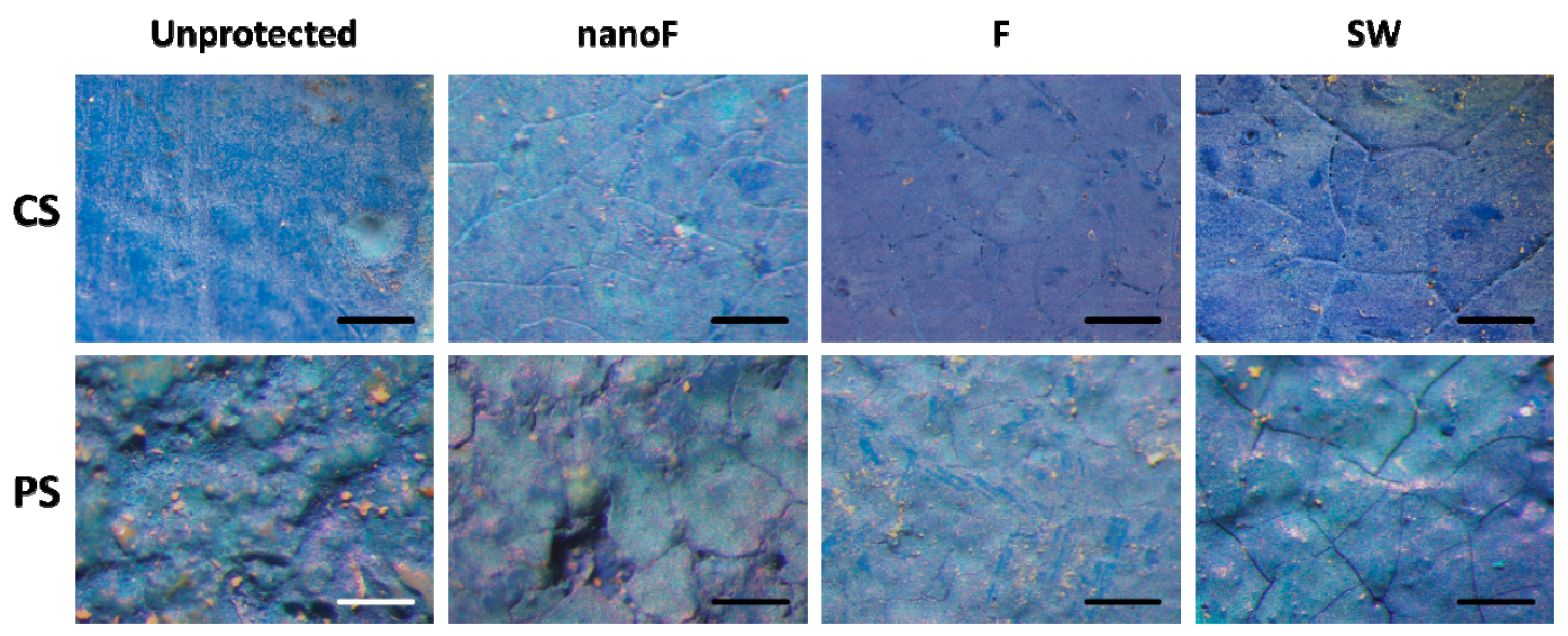
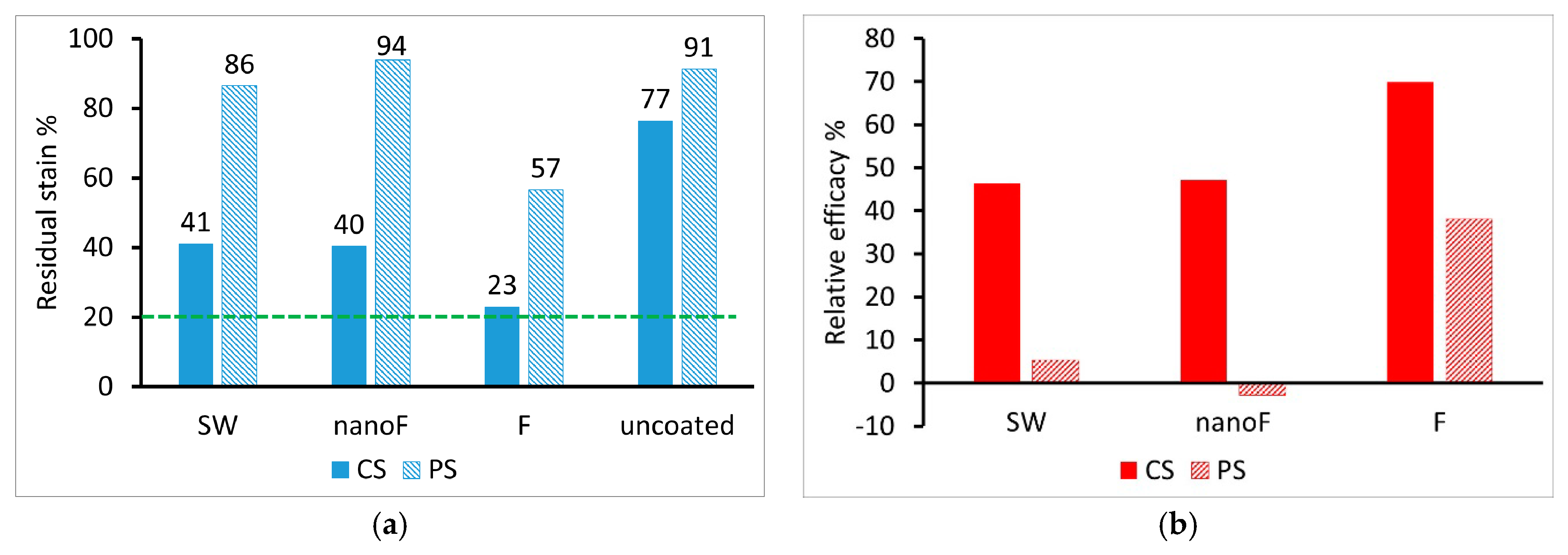
3.2. Staining by Spray Paint and Removal
3.3. Staining by Felt-Tip Marker and Removal
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- GRAFFOLUTION, Awareness and Prevention Solutions against Graffiti Vandalism in Public Areas and Transport. SSP (Policy Oriented Research) of the Seventh European Programme of the European Commission. FP7-SEC-2013-1. 2016. Available online: http://project.graffolution.eu/po/ (accessed on 26 September 2019).
- Macchia, A.; Ruffolo, S.A.; Rivaroli, L.; Malagodi, M.; Licchelli, M.; Rovella, N.; Randazzo, L.; La Russa, M.F. Comparative study of protective coatings for the conservation of Urban Art. J. Cult. Herit. 2019, in press. [Google Scholar] [CrossRef]
- Bosi, A.; Ciccola, A.; Serafini, I.; Guiso, M.; Ripanti, F.; Postorino, P.; Curini, R.; Bianco, A. Street art graffiti: Discovering their composition and alteration by FTIR and micro-Raman spectroscopy. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2020, 225, 117474. [Google Scholar] [CrossRef] [PubMed]
- Sanmartín, P.; Cappitelli, F. Evaluation of accelerated ageing tests for metallic and non-metallic graffiti paints applied to stone. Coatings 2017, 7, 180. [Google Scholar] [CrossRef]
- Dionísio, A.; Ribeiro, T. When graffiti is not art: The damage of alkyd sprays on calcareous stones employed in Cultural Heritage. In Cultural Heritage: Protection, Developments and International Perspectives (Focus on Civilizations and Cultures); Nova Science Publishers: Hauppauge, NY, USA, 2013; pp. 279–291. ISBN 978-1-62808-822-9. [Google Scholar]
- Gomes, V.; Dionísio, A.; Pozo-Antonio, J.S. Conservation strategies against graffiti vandalism on Cultural Heritage stones: Protective coatings and cleaning methods. Prog. Org. Coat. 2017, 113, 90–109. [Google Scholar] [CrossRef]
- Sanmartín, P.; Cappitelli, F.; Mitchell, R. Current methods of graffiti removal: A review. Constr. Build. Mater. 2014, 71, 363–374. [Google Scholar] [CrossRef]
- Rivas, T.; Pozo, S.; Fiorucci, M.P.; López, A.J.; Ramil, A. Nd: YVO4 laser removal of graffiti from granite. Influence of paint and rock properties on cleaning efficacy. Appl. Surf. Sci. 2012, 263, 563–572. [Google Scholar] [CrossRef]
- Samolik, S.; Walczak, M.; Plotek, M.; Sarzynski, A.; Pluska, I.; Marczak, J. Investigation into the removal of graffiti on mineral supports: Comparison of nanosecond Nd: YAG laser cleaning with traditional mechanical and chemical methods. Stud. Conserv. 2015, 60, S58–S64. [Google Scholar] [CrossRef]
- Rossi, S.; Fedel, M.; Petrolli, S.; Deflorian, F. Behaviour of different removers on permanent anti-graffiti organic coatings. J. Build. Eng. 2016, 5, 104–113. [Google Scholar] [CrossRef]
- Pozo-Antonio, J.S.; Rivas, T.; Fiorucci, M.P.; López, A.J.; Ramil, A. Effectiveness and harmfulness evaluation of graffiti cleaning by mechanical, chemical and laser procedures on granite. Microchem. J. 2016, 125, 1–9. [Google Scholar] [CrossRef]
- Sanmartín, P.; Bosch-Roig, P. Biocleaning to remove graffiti: A real possibility? Advances towards a complete protocol of action. Coatings 2019, 9, 104. [Google Scholar] [CrossRef]
- Germinario, G.; van der Werf, I.D.; Palazzo, G.; Regidor Ros, J.L.; Montes-Estelles, R.M.; Sabbatini, L. Bioremoval of marker pen inks by exploiting lipase hydrolysis. Prog. Org. Coat. 2017, 110, 162–171. [Google Scholar] [CrossRef]
- García, O.; Malaga, K. Definition of the procedure to determine the suitability and durability of an anti-graffiti product for application on cultural heritage porous materials. J. Cult. Herit. 2012, 13, 77–82. [Google Scholar] [CrossRef]
- Licchelli, M.; Marzolla, S.J.; Poggi, A.; Zanchi, C. Crosslinked fluorinated polyurethanes for the protection of stone surfaces from graffiti. J. Cult. Herit. 2011, 12, 34–43. [Google Scholar] [CrossRef]
- Liu, H.; Gao, L.; Shang, Q.; Xiao, G. Preparation and characterization of polyurethane clearcoats and investigation into their antigraffiti property. J. Coat. Technol. Res. 2013, 10, 775–784. [Google Scholar] [CrossRef]
- Rabea, A.M.; Mohseni, M.; Mirabedini, S.M.; Tabatabaei, M.H. Surface analysis and anti-graffiti behavior of a weathered polyurethane-based coating embedded with hydrophobic nano silica. Appl. Surf. Sci. 2012, 258, 4391–4396. [Google Scholar] [CrossRef]
- Manvi, G.N.; Singh, A.R.; Jagtap, R.N.; Kothari, D.C. Isocyanurate based fluorinated polyurethane dispersion for anti-graffiti coatings. Prog. Org. Coat. 2012, 75, 139–146. [Google Scholar] [CrossRef]
- Carmona-Quiroga, P.M.; Rubio, J.; Sánchez, M.J.; Martínez-Ramírez, S.; Blanco-Varela, M.T. Surface dispersive energy determined with IGC-ID in anti-graffiti-coated building materials. Prog. Org. Coat. 2011, 71, 207–212. [Google Scholar] [CrossRef]
- García, O.; Rz-Maribona, I.; Gardei, A.; Riedl, M.; Vanhellemont, Y.; Santarelli, M.L.; Suput, J.S. Comparative study of the variation of the hydric properties and aspect of natural stone and brick after the application of 4 types of anti-graffiti. Mater. Constr. 2010, 60, 69–82. [Google Scholar] [CrossRef]
- Carmona-Quiroga, P.M.; Jacobs, R.M.J.; Martínez-Ramírez, S.; Viles, H.A. Durability of anti-graffiti coatings on stone: Natural vs accelerated weathering. PLoS ONE 2017, 12, e0172347. [Google Scholar] [CrossRef]
- Kharitonov, A.P.; Simbirtseva, G.V.; Nazarov, V.G.; Stolyarov, V.P.; Dubois, M.; Peyroux, J. Enhanced anti-graffiti or adhesion properties of polymers using versatile combination of fluorination and polymer grafting. Prog. Org. Coat. 2015, 88, 127–136. [Google Scholar] [CrossRef]
- Esposito Corcione, C.; De Simone, N.; Santarelli, M.L.; Frigione, M. Protective properties and durability characteristics of experimental and commercial organic coatings for the preservation of porous stone. Prog. Org. Coat. 2017, 103, 193–203. [Google Scholar] [CrossRef]
- Heinisch, M.; Miricescu, D. Innovative industrial technologies for preventive anti-graffiti coating. In MATEC Web of Conferenes; EDP Sciences: Les Ulis, France, 2017; Volume 121, p. 03009. [Google Scholar]
- Licchelli, M.; Malagodi, M.; Weththimuni, M.; Zanchi, C. Anti-Graffiti nanocomposite materials for surface protection of a very porous stone. Appl. Phys. A 2014, 116, 1525–1539. [Google Scholar] [CrossRef]
- Moura, A.; Flores-Colen, I.; De Brito, J. Study of the effect of three anti-graffiti products on the physical properties of different substrates. Constr. Build. Mater. 2016, 107, 157–164. [Google Scholar] [CrossRef]
- Fedel, M.; Rossi, S.; Deflorian, F. Polymethyl (hydro)/polydimethylsilazane-derived coatings applied on AA1050: Effect of the dilution in butyl acetate on the structural and electrochemical properties. J. Coat. Technol. Res. 2019, 16, 1013–1019. [Google Scholar] [CrossRef]
- Melquiades, F.L.; Appoloni, C.R.; Andrello, A.C.; Spagnuolo, E. Non-Destructive analytical techniques for the evaluation of cleaning and protection processes on white marble surfaces. J. Cult. Herit. 2019, 37, 54–62. [Google Scholar] [CrossRef]
- Frigione, M.; Lettieri, M. Novel attribute of organic–inorganic hybrid coatings for protection and preservation of materials (stone and wood) belonging to cultural heritage. Coatings 2018, 8, 319. [Google Scholar] [CrossRef]
- Kronlund, D.; Lindén, M.; Smått, J.-H. A sprayable protective coating for marble with water-repellent and anti-graffiti properties. Prog. Org. Coat. 2016, 101, 359–366. [Google Scholar] [CrossRef]
- Bayer, S.I. On the durability and wear resistance of transparent superhydrophobic coatings. Coatings 2017, 7, 12. [Google Scholar] [CrossRef]
- Bormashenko, E. Physics of solid–liquid interfaces: From the Young equation to the superhydrophobicity. Low Temp. Phys. 2016, 42, 622–635. [Google Scholar] [CrossRef]
- Simpson, J.T.; Hunter, S.R.; Aytug, T. Superhydrophobic materials and coatings: A review. Rep. Prog. Phys. 2015, 78, 086501. [Google Scholar] [CrossRef]
- Milionis, A.; Loth, E.; Bayer, I.S. Recent advances in the mechanical durability of superhydrophobic materials. Adv. Colloid Interface Sci. 2016, 229, 57–79. [Google Scholar] [CrossRef] [PubMed]
- Cohen, N.; Dotan, A.; Dodiuk, H.; Kenig, S. Superhydrophobic coatings and their durability. Mater. Manuf. Process. 2016, 31, 1143–1155. [Google Scholar] [CrossRef]
- Manoudis, P.N.; Tsakalof, A.; Karapanagiotis, I.; Zuburtikudis, I.; Panayiotou, C. Fabrication of super-hydrophobic surfaces for enhanced stone protection. Surf. Coat. Technol. 2009, 203, 1322–1328. [Google Scholar] [CrossRef]
- Milionis, A.; Bayer, I.S.; Loth, E. Recent advances in oil-repellent surfaces. Int. Mater. Rev. 2016, 61, 101–126. [Google Scholar] [CrossRef]
- Hosseini, M.; Karapanagiotis, I. (Eds.) Advanced Materials for the Conservation of Stone; Springer: Cham, Switzerland, 2018; ISBN 978-3-319-72260-3. [Google Scholar]
- Godeau, G.; Guittard, F.; Darmanin, T. Surfaces bearing fluorinated nucleoperfluorolipids for potential anti-graffiti surface properties. Coatings 2017, 7, 220. [Google Scholar] [CrossRef]
- Haas, K.-H.; Amberg-Schwab, S.; Rose, K. Functionalized coating materials based on inorganic-organic polymers. Thin Solid Film. 1999, 351, 198–203. [Google Scholar] [CrossRef]
- Badila, M.; Kohlmayr, M.; Zikulnig-Rusch, E.M.; Dolezel-Horwath, E.; Kandelbauer, A. Improving the cleanability of melamine-formaldehyde-based decorative laminates. J. Appl. Polym. Sci. 2014, 131, 40964. [Google Scholar] [CrossRef]
- Xu, F.; Li, X.; Li, Y.; Sun, J. Oil-repellent antifogging films with water-enabled functional and structural healing ability. ACS Appl. Mater. Interfaces 2017, 9, 27955–27963. [Google Scholar] [CrossRef]
- Carmona-Quiroga, P.M.; Martínez-Ramírez, S.; Sánchez-Cortés, S.; Oujja, M.; Castillejo, M.; Blanco-Varela, M.T. Effectiveness of antigraffiti treatments in connection with penetration depth determined by different techniques. J. Cult. Herit. 2010, 11, 297–303. [Google Scholar] [CrossRef]
- Khan, F.; Khan, A.; Tuhin, M.O.; Rabnawaz, M.; Li, Z.; Naveed, M. A novel dual-layer approach towards omniphobic polyurethane coatings. RSC Adv. 2019, 9, 26703–26711. [Google Scholar] [CrossRef]
- Malaga, K.; Mueller, U. Relevance of hydrophobic and oleophobic properties of antigraffiti systems on their cleaning efficiency on concrete and stone surfaces. J. Mater. Civ. Eng. 2013, 25, 755–762. [Google Scholar] [CrossRef]
- Scheerder, J.; Visscher, N.; Nabuurs, T.; Overbeek, A. Novel, water-based fluorinated polymers with excellent antigraffiti properties. J. Coat. Technol. Res. 2005, 2, 617–625. [Google Scholar] [CrossRef]
- Lettieri, M.; Masieri, M.; Morelli, A.; Pipoli, M.; Frigione, M. Oleo/hydrophobic coatings containing nano-particles for the protection of stone materials having different porosity. Coatings 2018, 8, 429. [Google Scholar] [CrossRef]
- Bugani, S.; Camaiti, M.; Morselli, L.; Van de Casteele, E.; Janssens, K. Investigation on porosity changes of Lecce stone due to conservation treatments by means of X-ray nano- and improved micro-computed tomography: Preliminary results. X-Ray Spectrom. 2007, 36, 316–320. [Google Scholar] [CrossRef]
- Föllmi, K.B.; Hofmann, H.; Chiaradia, M.; de Kaenel, E.; Frijia, G.; Parente, M. Miocene phosphate-rich sediments in Salento (southern Italy). Sediment. Geol. 2015, 327, 55–71. [Google Scholar] [CrossRef]
- Tiano, P.; Accolla, P.; Tomaselli, L. Phototrophic biodeteriogens on lithoid surfaces: An ecological study. Microb. Ecol. 1995, 29, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Dunham, R.J. Classification of carbonate rocks according to depositional textures. AAPG Mem. 1962, 1, 108–121. [Google Scholar]
- NORMAL Rec. 4/80 Distribuzione del Volume dei Pori in Funzione del Loro Diametro; CNR/ICR: Rome, Italy, 1980.
- UNI 10921. Beni Culturali Materiali Lapidei Naturali ed Artificiali—Prodotti Idrorepellenti—Applicazione su Provini e Determinazione in Laboratorio Delle Loro Caratteristiche; Ente Italiano di normazione: Milan, Italy, 2001.
- ASTM D 6578. Standard Practice for Determination of Graffiti Resistance; ASTM International: West Conshohocken, PA, USA, 2000.
- Baglioni, M.; Poggi, G.; Jaidar Benavides, Y.; Martínez Camacho, F.; Giorgi, R.; Baglioni, P. Nanostructured fluids for the removal of graffiti—A survey on 17 commercial spray-can paints. J. Cult. Herit. 2018, 34, 218–226. [Google Scholar] [CrossRef]
- EN 15802. Conservation of Cultural Property—Test Methods—Determination of Static Contact Angle; CEN (European Committee for Standardization): Brussels, Belgium, 2010. [Google Scholar]
- Aslanidou, D.; Karapanagiotis, I.; Panayiotou, C. Tuning the wetting properties of siloxane-nanoparticle coatings to induce superhydrophobicity and superoleophobicity for stone protection. Mater. Des. 2016, 108, 736–744. [Google Scholar] [CrossRef]
- Facio, D.S.; Carrascosa, L.A.M.; Mosquera, M.J. Producing lasting amphiphobic building surfaces with self-cleaning properties. Nanotechnology 2017, 28, 265601. [Google Scholar] [CrossRef]
- Aslanidou, D.; Karapanagiotis, I.; Lampakis, D. Waterborne superhydrophobic and superoleophobic coatings for the protection of marble and sandstone. Materials 2018, 11, 585. [Google Scholar] [CrossRef] [PubMed]
- EN 15886. Conservation of Cultural Property–Test Methods—Colour Measurement of Surfaces; CEN (European Committee for Standardization): Brussels, Belgium, 2010.
- Sahasrabudhe, S.N.; Rodriguez-Martinez, V.; O’Meara, M.; Farkas, B.E. Density, viscosity, and surface tension of five vegetable oils at elevated temperatures: Measurement and modeling. Int. J. Food Prop. 2017, 20, 1965–1981. [Google Scholar] [CrossRef]
- Rabea, A.M.; Mirabedini, S.M.; Mohseni, M. Investigating the surface properties of polyurethane based anti-graffiti coatings against UV exposure. J. Appl. Polym. Sci. 2012, 124, 3082–3091. [Google Scholar] [CrossRef]
- Shang, B.; Chen, M.; Wu, L. One-step synthesis of statically amphiphilic/dynamically amphiphobic fluoride-free transparent coatings. ACS Appl. Mater. Interfaces 2018, 10, 41824–41830. [Google Scholar] [CrossRef] [PubMed]
- Zhong, X.; Hu, H.; Yang, L.; Sheng, J.; Fu, H. Robust hyperbranched polyester-based anti-smudge coatings for self-cleaning, anti-graffiti, and chemical shielding. ACS Appl. Mater. Interfaces 2019, 11, 14305–14312. [Google Scholar] [CrossRef] [PubMed]
- Carvalhão, M.; Dionísio, A. Evaluation of mechanical soft-abrasive blasting and chemical cleaning methods on alkyd-paint graffiti made on calcareous stones. J. Cult. Herit. 2015, 16, 579–590. [Google Scholar] [CrossRef]
- Gherardi, F.; Colombo, A.; D’Arienzo, M.; Di Credico, B.; Goidanich, S.; Morazzoni, F.; Simonutti, R.; Toniolo, L. Efficient self-cleaning treatments for built heritage based on highly photo-active and well-dispersible TiO2 nanocrystals. Microchem. J. 2016, 126, 54–62. [Google Scholar] [CrossRef]
- Gomes, V.; Dionísio, A.; Pozo-Antonio, J.S. The influence of the SO2 ageing on the graffiti cleaning effectiveness with chemical procedures on a granite substrate. Sci. Total Environ. 2018, 625, 233–245. [Google Scholar] [CrossRef]
- Masieri, M.; Lettieri, M. Influence of the distribution of a spray paint on the efficacy of anti-graffiti coatings on a highly porous natural stone material. Coatings 2017, 7, 18. [Google Scholar] [CrossRef]
- Tarnowski, A.; Zhang, X.; Mcnamara, C.; Martin, S.; Mitchell, R. Biodeterioration and performance of anti-graffiti coatings on sandstone and marble. J. Can. Assoc. Conserv. J CAC 2007, 32, 3–16. [Google Scholar]
- Pozo-Antonio, J.S.; Rivas, T.; Jacobs, R.M.J.; Viles, H.A.; Carmona-Quiroga, P.M. Effectiveness of commercial anti-graffiti treatments in two granites of different texture and mineralogy. Prog. Org. Coat. 2018, 116, 70–82. [Google Scholar] [CrossRef]
- Poli, T.; Toniolo, L. The challenge of protecting monuments from atmospheric attack. In Fracture and Failure of Natural Building Stones: Applications in the Restoration of Ancient Monuments; Kourkoulis, S.K., Ed.; Springer Netherlands: Dordrecht, The Netherlands, 2006; pp. 553–563. ISBN 978-1-4020-5077-0. [Google Scholar]
- Selwitz, C. Epoxy Resins in Stone Conservation; Getty Publications: Marina del Rey, CA, USA, 1992; Volume 7, ISBN 0-89236-238-3. [Google Scholar]
- Sena da Fonseca, B.; Piçarra, S.; Ferreira Pinto, A.P.; Ferreira, M.J.; Montemor, M.F. TEOS-based consolidants for carbonate stones: The role of N1-(3-trimethoxysilylpropyl)diethylenetriamine. New J. Chem. 2017, 41, 2458–2467. [Google Scholar] [CrossRef]
- Borsoi, G.; Lubelli, B.; van Hees, R.; Veiga, R.; Silva, A.S.; Colla, L.; Fedele, L.; Tomasin, P. Effect of solvent on nanolime transport within limestone: How to improve in-depth deposition. Colloids Surf. Physicochem. Eng. Asp. 2016, 497, 171–181. [Google Scholar] [CrossRef]
- Domaslowski, W. The mechanism of polymer migration in porous stones. Wien. Ber. Über Nat. Kunst 1988, 4, 402–425. [Google Scholar]
- Hansen, E.F.; Lowinger, R.; Sadoff, E. Consolidation of porous paint in a vapor-saturated atmosphere a technique for minimizing changes in the appearance of powdering, matte paint. J. Am. Inst. Conserv. 1993, 32, 1–14. [Google Scholar]
- Sena da Fonseca, B.; Piçarra, S.; Pinto, A.P.; Montemor, M.d.F. Polyethylene glycol oligomers as siloxane modificators in consolidation of carbonate stones. Pure Appl. Chem. 2016, 88, 1117. [Google Scholar] [CrossRef]
- Giorgiutti-Dauphiné, F.; Pauchard, L. Painting cracks: A way to investigate the pictorial matter. J. Appl. Phys. 2016, 120, 065107. [Google Scholar] [CrossRef]
- Krzemień, L.; Łukomski, M.; Bratasz, Ł.; Kozłowski, R.; Mecklenburg, M.F. Mechanism of craquelure pattern formation on panel paintings. Stud. Conserv. 2016, 61, 324–330. [Google Scholar] [CrossRef]
- Atkinson, A.; Guppy, R.M. Mechanical stability of sol-gel films. J. Mater. Sci. 1991, 26, 3869–3873. [Google Scholar] [CrossRef]
- Moura, A.R.; Flores-Colen, I.; de Brito, J. Anti-Graffiti products for porous surfaces. An overview. In Proceedings of the Hydrophobe VII 7th International Conference on Water Repellent Treatment and Protective Surface Technology for Building Materials, LNEC (Laboratório Nacional de Engenharia Civil), Lisbon, Portugal, 11–12 September 2014; pp. 225–233. [Google Scholar]
- Moretti, P.; Germinario, G.; Doherty, B.; van der Werf, I.D.; Sabbatini, L.; Mirabile, A.; Sgamellotti, A.; Miliani, C. Disclosing the composition of historical commercial felt-tip pens used in art by integrated vibrational spectroscopy and pyrolysis-gas chromatography/mass spectrometry. J. Cult. Herit. 2019, 35, 242–253. [Google Scholar] [CrossRef]
- Lettieri, M.; Masieri, M. Surface characterization and effectiveness evaluation of anti-graffiti coatings on highly porous stone materials. Appl. Surf. Sci. 2014, 288, 466–477. [Google Scholar] [CrossRef]






| Stone Substrate | Product | Applied Amount (g/m2) | ΔE*ab (CIELAB unit) | ΔP (%) |
|---|---|---|---|---|
| CS | nanoF | 58 | 1.72 | +15 |
| F | 60 | 2.50 | −5 | |
| SW | 109 | 2.56 | −38 | |
| PS | nanoF | 155 | 1.38 | +14 |
| F | 160 | 3.97 | −6 | |
| SW | 313 | 3.51 | −51 |
| Samples | CS | PS | ||
|---|---|---|---|---|
| WCA | OCA | WCA | OCA | |
| Unprotected | 40 ± 8 | 13 ± 1 | Not determinable | Not determinable |
| nanoF | 139 ± 5 | 114 ± 1 | 142 ± 5 | 122 ± 7 |
| F | 106 ± 4 | 93 ± 4 | 119 ± 3 | 114 ± 4 |
| SW | 114 ± 4 | 56 ± 1 | 122 ± 4 | 56 ± 2 |
| Samples | CS | PS | ||
|---|---|---|---|---|
| After Staining | After Removal | After Staining | After Removal | |
| Unprotected | 54.53 ± 0.33 | 20.19 ± 2.27 | 55.39 ± 1.14 | 44.58 ± 4.36 |
| nanoF | 40.72 ± 3.56 | 21.72 ± 1.85 | 55.73 ± 3.96 | 37.11 ± 3.35 |
| F | 45.23 ± 5.18 | 5.99 ± 1.58 | 56.13 ± 2.61 | 13.78 ± 1.53 |
| SW | 55.28 ± 0.44 | 15.27 ± 1.95 | 59.51 ± 1.99 | 19.93 ± 2.96 |
| Stone Support | Samples | Before Treatments | After Protection | After Staining | After Removal |
|---|---|---|---|---|---|
| CS | Unprotected | 55 ± 10 | – | 93 ± 1 | 76 ± 3 |
| nanoF | 38 ± 7 | 140 ± 5 | 98 ± 3 | 115 ± 4 | |
| F | 44 ± 7 | 105 ± 5 | 92 ± 1 | 116 ± 4 | |
| SW | 39 ± 7 | 112 ± 3 | 101 ± 4 | 111 ± 4 | |
| PS | Unprotected | n.d. | – | 98 ± 5 | 43 ± 14 |
| nanoF | n.d. | 144 ± 4 | 106 ± 7 | 126 ± 4 | |
| F | n.d. | 120 ± 3 | 95 ± 3 | 128 ± 4 | |
| SW | n.d. | 121 ± 4 | 101 ± 4 | 120 ± 4 |
| Samples | CS | PS | ||
|---|---|---|---|---|
| After Staining | After Removal | After Staining | After Removal | |
| Unprotected | 66.19 ± 0.37 | 50.65 ± 1.98 | 71.22 ± 0.70 | 65.03 ± 1.93 |
| nanoF | 58.52 ± 0.46 | 23.67 ± 9.57 | 70.61 ± 0.56 | 66.33 ± 0.52 |
| F | 57.95 ± 1.83 | 13.35 ± 4.40 | 72.88 ± 0.32 | 41.23 ± 12.22 |
| SW | 48.24 ± 1.87 | 19.81 ± 6.49 | 72.38 ± 0.57 | 62.60 ± 2.99 |
| Stone Support | Samples | Before Treatments | After Protection | After Staining | After Removal |
|---|---|---|---|---|---|
| CS | Unprotected | 55 ± 10 | – | 89 ± 2 | 92 ± 3 |
| nanoF | 38 ± 7 | 140 ± 5 | 80 ± 3 | 101 ± 9 | |
| F | 44 ± 7 | 105 ± 5 | 79 ± 5 | 106 ± 2 | |
| SW | 39 ± 7 | 112 ± 3 | 80 ± 3 | 110 ± 2 | |
| PS | Unprotected | n.d. | – | 98 ± 5 | 106 ± 7 |
| nanoF | n.d. | 144 ± 4 | 83 ±13 | 122 ± 8 | |
| F | n.d. | 120 ± 3 | 90 ± 12 | 117 ± 4 | |
| SW | n.d. | 121 ± 4 | 70 ± 12 | 106 ± 6 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lettieri, M.; Masieri, M.; Pipoli, M.; Morelli, A.; Frigione, M. Anti-Graffiti Behavior of Oleo/Hydrophobic Nano-Filled Coatings Applied on Natural Stone Materials. Coatings 2019, 9, 740. https://doi.org/10.3390/coatings9110740
Lettieri M, Masieri M, Pipoli M, Morelli A, Frigione M. Anti-Graffiti Behavior of Oleo/Hydrophobic Nano-Filled Coatings Applied on Natural Stone Materials. Coatings. 2019; 9(11):740. https://doi.org/10.3390/coatings9110740
Chicago/Turabian StyleLettieri, Mariateresa, Maurizio Masieri, Mariachiara Pipoli, Alessandra Morelli, and Mariaenrica Frigione. 2019. "Anti-Graffiti Behavior of Oleo/Hydrophobic Nano-Filled Coatings Applied on Natural Stone Materials" Coatings 9, no. 11: 740. https://doi.org/10.3390/coatings9110740
APA StyleLettieri, M., Masieri, M., Pipoli, M., Morelli, A., & Frigione, M. (2019). Anti-Graffiti Behavior of Oleo/Hydrophobic Nano-Filled Coatings Applied on Natural Stone Materials. Coatings, 9(11), 740. https://doi.org/10.3390/coatings9110740







