Low-Temperature Vapor-Solid Growth of ZnO Nanowhiskers for Electron Field Emission
Abstract
1. Introduction
2. Materials and Methods
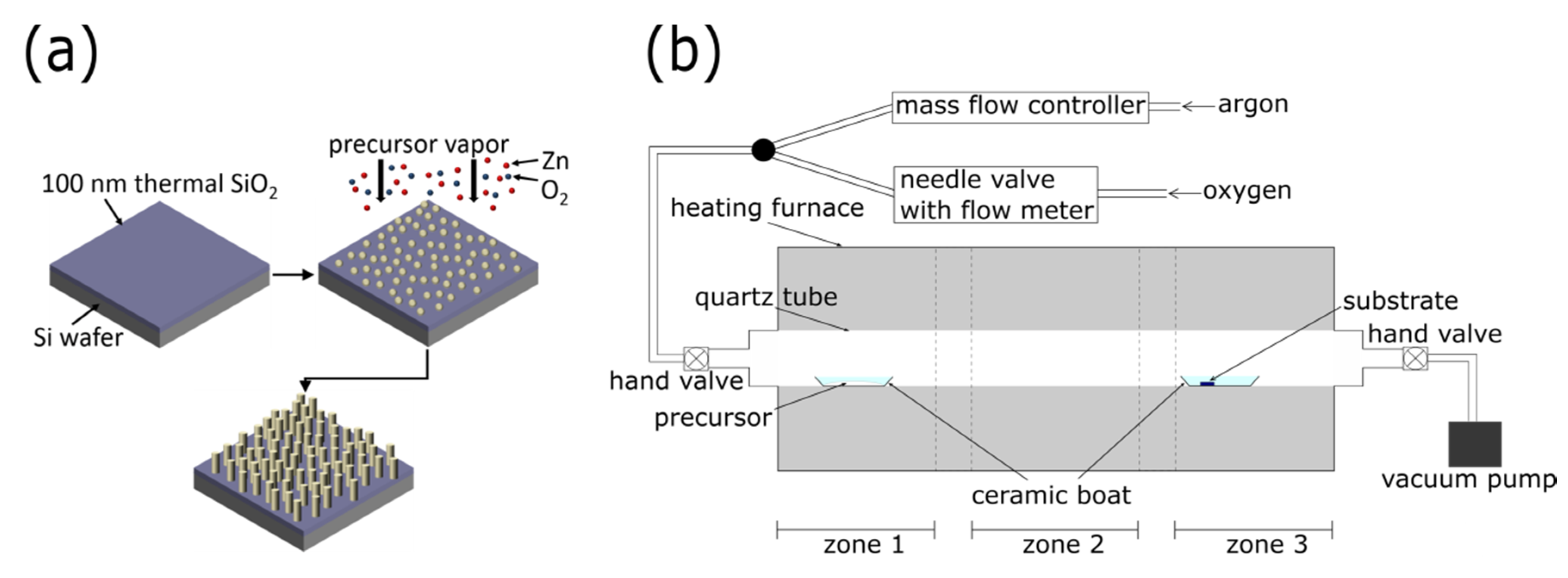
2.1. Nanostructured ZnO Growth
2.2. Structural Characterization
2.3. Electron Field Emission Measurements
3. Results and Discussion: Process Optimization
3.1. Substrate Position
3.2. Growth Temperature
3.3. Oxygen and Argon Flow
3.4. Precursor Amount and Growth Time
3.5. Substrate Type
3.6. Summary of Optimized Growth Parameters
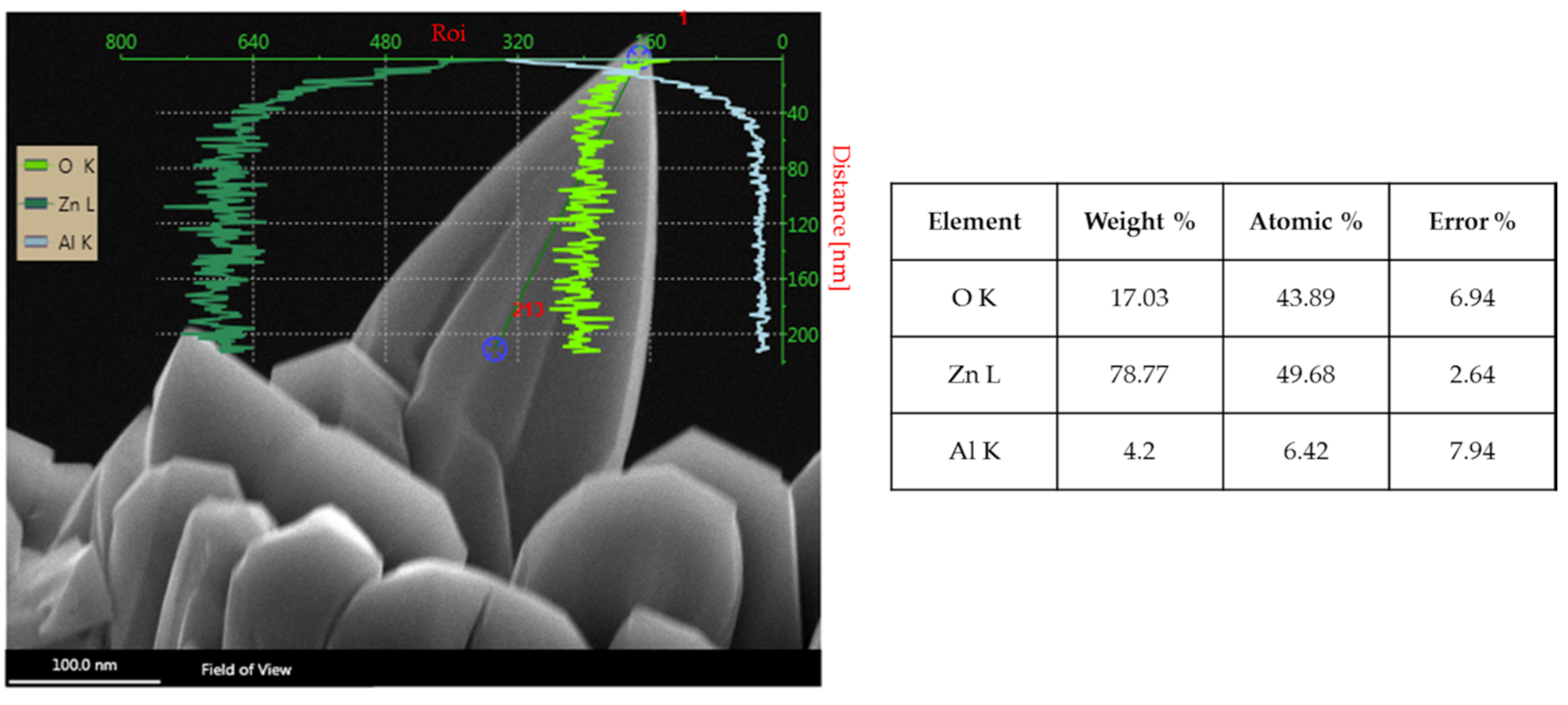
3.7. Structural and Compositional Analysis of Optimized ZnO Nanowhiskers
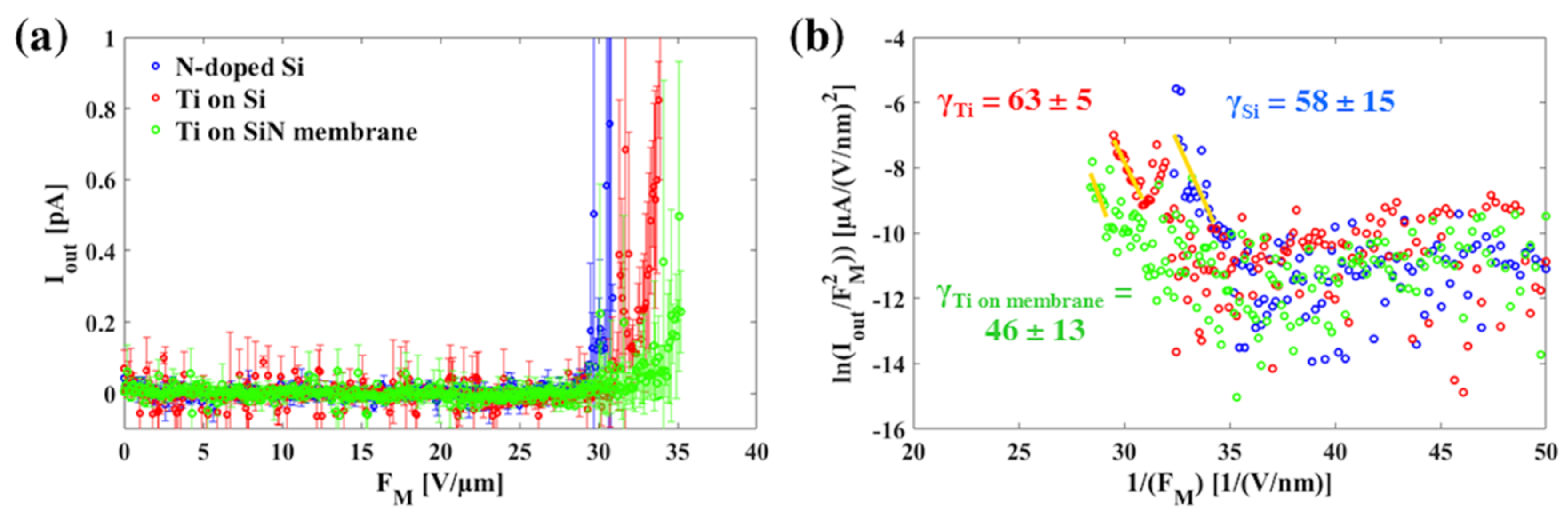
4. Field Emission Measurements
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Djurišić, A.B.; Leung, Y.H. Optical Properties of ZnO Nanostructures. Small 2006, 2, 944–961. [Google Scholar] [CrossRef] [PubMed]
- Park, W.I. Controlled synthesis and properties of ZnO nanostructures grown by metalorganic chemical vapor deposition: A review. Met. Mater. Int. 2008, 14, 659–665. [Google Scholar] [CrossRef]
- Kołodziejczak-Radzimska, A.; Jesionowski, T. Zinc Oxide—From Synthesis to Application: A Review. Materials 2014, 7, 2833–2881. [Google Scholar] [CrossRef] [PubMed]
- Ding, M.; Guo, Z.; Zhou, L.; Fang, X.; Zhang, L.; Zeng, L.; Xie, L.; Zhao, H. One-Dimensional Zinc Oxide Nanomaterials for Application in High-Performance Advanced Optoelectronic Devices. Crystals 2018, 8, 223. [Google Scholar] [CrossRef]
- Hsu, C.-L.; Chang, S.-J.; Hung, H.-C.; Lin, Y.-R.; Huang, C.-J.; Tseng, Y.-K.; Chen, I.-C. Well-Aligned, Vertically Al-Doped ZnO Nanowires Synthesized on ZnO:Ga/Glass Templates. J. Electrochem. Soc. 2005, 152, G378–G381. [Google Scholar] [CrossRef]
- Tynell, T.; Karppinen, M. Atomic layer deposition of ZnO: A review. Semicond. Sci. Technol. 2014, 29, 043001. [Google Scholar] [CrossRef]
- Özgür, Ü.; Alivov, Y.I.; Liu, C.; Teke, A.; Reshchikov, M.A.; Doğan, S.; Avrutin, V.; Cho, S.-J.; Morkoç, H. A comprehensive review of ZnO materials and devices. J. Appl. Phys. 2005, 98, 041301. [Google Scholar] [CrossRef]
- Gomez, J.L.; Tigli, O. Zinc oxide nanostructures: From growth to application. J. Mater. Sci. 2013, 48, 612–624. [Google Scholar] [CrossRef]
- Nasr, B.; Dasgupta, S.; Wang, D.; Mechau, N.; Kruk, R.; Hahn, H. Electrical resistivity of nanocrystalline Al-doped zinc oxide films as a function of Al content and the degree of its segregation at the grain boundaries. J. Appl. Phys. 2010, 108, 103721. [Google Scholar] [CrossRef]
- Park, W.I.; Yi, G.-C.; Kim, M.; Pennycook, S.J. ZnO Nanoneedles Grown Vertically on Si Substrates by Non-Catalytic Vapor-Phase Epitaxy. Adv. Mater. 2002, 14, 1841–1843. [Google Scholar] [CrossRef]
- Djurišić, A.B.; Ng, A.M.C.; Chen, X.Y. ZnO nanostructures for optoelectronics: Material properties and device applications. Prog. Quantum Electron. 2010, 34, 191–259. [Google Scholar] [CrossRef]
- Carretero-Genevrier, A.; Mestres, N.; National, S. Encyclopedia of Nanotechnology; Bhushan, B., Ed.; Springer: Dordrecht, The Netherlands, 2016; ISBN 978-94-017-9779-5. [Google Scholar]
- Sui, M.; Gong, P.; Gu, X. Review on one-dimensional ZnO nanostructures for electron field emitters. Front. Optoelectron. 2013, 6, 386–412. [Google Scholar] [CrossRef]
- Rivero, P.; Garcia, J.; Quintana, I.; Rodriguez, R. Design of Nanostructured Functional Coatings by Using Wet-Chemistry Methods. Coatings 2018, 8, 76. [Google Scholar] [CrossRef]
- Tao, Y.; Pescarmona, P. Nanostructured Oxides Synthesised via scCO2-Assisted Sol-Gel Methods and Their Application in Catalysis. Catalysts 2018, 8, 212. [Google Scholar] [CrossRef]
- Greene, L.E.; Law, M.; Goldberger, J.; Kim, F.; Johnson, J.C.; Zhang, Y.; Saykally, R.J.; Yang, P. Low-Temperature Wafer-Scale Production of ZnO Nanowire Arrays. Angew. Chem. Int. Ed. 2003, 42, 3031–3034. [Google Scholar] [CrossRef] [PubMed]
- Ko, S.H.; Lee, D.; Hotz, N.; Yeo, J.; Hong, S.; Nam, K.H.; Grigoropoulos, C.P. Digital Selective Growth of ZnO Nanowire Arrays from Inkjet-Printed Nanoparticle Seeds on a Flexible Substrate. Langmuir 2012, 28, 4787–4792. [Google Scholar] [CrossRef]
- Ko, S.H.; Lee, D.; Kang, H.W.; Nam, K.H.; Yeo, J.Y.; Hong, S.J.; Grigoropoulos, C.P.; Sung, H.J. Nanoforest of Hydrothermally Grown Hierarchical ZnO Nanowires for a High Efficiency Dye-Sensitized Solar Cell. Nano Lett. 2011, 11, 666–671. [Google Scholar] [CrossRef]
- Wan, H.; Ruda, H.E. A study of the growth mechanism of CVD-grown ZnO nanowires. J. Mater. Sci. Mater. Electron. 2010, 21, 1014–1019. [Google Scholar] [CrossRef]
- Hamzah, N.A.B.; Pung, S.Y.; Sreekantan, S.; Aziz, S.N.Q.A.B.A. Synthesis and Growth Mechanism of Catalyst-Free ZnO Nanowires Using Chemical Vapour Deposition. Adv. Mater. Res. 2012, 620, 320–324. [Google Scholar] [CrossRef]
- Satoh, M.; Tanaka, N.; Ueda, Y.; Ohshio, S.; Saitoh, H. Epitaxial Growth of Zinc Oxide Whiskers by Chemical-Vapor Deposition under Atmospheric Pressure. Jpn. J. Appl. Phys. 1999, 38, L586–L589. [Google Scholar] [CrossRef]
- Baxter, J.B.; Aydil, E.S. Metallorganic Chemical Vapor Deposition of ZnO Nanowires from Zinc Acetylacetonate and Oxygen. J. Electrochem. Soc. 2009, 156, H52–H58. [Google Scholar] [CrossRef]
- Liu, S.-C.; Wu, J.-J. Low-temperature and catalyst-free synthesis of well-aligned ZnO nanorods on Si (100). J. Mater. Chem. 2002, 12, 3125–3129. [Google Scholar] [CrossRef]
- Wagner, R.S.; Ellis, W.C. Vapor-Liquid-Solid Mechanism of Single Crystal Growth. Appl. Phys. Lett. 1964, 4, 89–90. [Google Scholar] [CrossRef]
- Wu, Y.; Yang, P. Direct Observation of Vapor−Liquid−Solid Nanowire Growth. J. Am. Chem. Soc. 2001, 123, 3165–3166. [Google Scholar] [CrossRef]
- Huang, M.H.; Wu, Y.; Feick, H.; Tran, N.; Weber, E.; Yang, P. Catalytic Growth of Zinc Oxide Nanowires by Vapor Transport. Adv. Mater. 2001, 13, 113–116. [Google Scholar] [CrossRef]
- Wu, J.-J.; Liu, S.-C. Low-Temperature Growth of Well-Aligned ZnO Nanorods by Chemical Vapor Deposition. Adv. Mater. 2002, 14, 215–218. [Google Scholar] [CrossRef]
- Wu, J.-J.; Liu, S.-C. Catalyst-Free Growth and Characterization of ZnO Nanorods. J. Phys. Chem. B 2002, 106, 9546–9551. [Google Scholar] [CrossRef]
- Garry, S.; McCarthy, É.; Mosnier, J.-P.; McGlynn, E. Influence of ZnO nanowire array morphology on field emission characteristics. Nanotechnology 2014, 25, 135604. [Google Scholar] [CrossRef]
- Fang, Y.; Wong, K.M.; Lei, Y. Synthesis and field emission properties of different ZnO nanostructure arrays. Nanoscale Res. Lett. 2012, 7, 197. [Google Scholar] [CrossRef]
- Kuo, S.-Y.; Lin, H.-I. Field emission characteristics of zinc oxide nanowires synthesized by vapor-solid process. Nanoscale Res. Lett. 2014, 9, 70. [Google Scholar] [CrossRef]
- Li, S.Y.; Lin, P.; Lee, C.Y.; Tseng, T.Y. Field emission and photofluorescent characteristics of zinc oxide nanowires synthesized by a metal catalyzed vapor-liquid-solid process. J. Appl. Phys. 2004, 95, 3711–3716. [Google Scholar] [CrossRef]
- Wang, X.D.; Zhou, J.; Lao, C.S.; Song, J.H.; Xu, N.S.; Wang, Z.L. In Situ Field Emission of Density-Controlled ZnO Nanowire Arrays. Adv. Mater. 2007, 19, 1627–1631. [Google Scholar] [CrossRef]
- Zhu, Y.W.; Zhang, H.Z.; Sun, X.C.; Feng, S.Q.; Xu, J.; Zhao, Q.; Xiang, B.; Wang, R.M.; Yu, D.P. Efficient field emission from ZnO nanoneedle arrays. Appl. Phys. Lett. 2003, 83, 144–146. [Google Scholar] [CrossRef]
- Zhang, Z.; Yuan, H.; Zhou, J.; Liu, D.; Luo, S.; Miao, Y.; Gao, Y.; Wang, J.; Liu, L.; Song, L.; et al. Growth Mechanism, Photoluminescence, and Field-Emission Properties of ZnO Nanoneedle Arrays. J. Phys. Chem. B 2006, 110, 8566–8569. [Google Scholar] [CrossRef]
- Shen, G.Z.; Bando, Y.; Liu, B.D.; Golberg, D.; Lee, C.-J. Characterization and Field-Emission Properties of Vertically Aligned ZnO Nanonails and Nanopencils Fabricated by a Modified Thermal-Evaporation Process. Adv. Funct. Mater. 2006, 16, 410–416. [Google Scholar] [CrossRef]
- Tseng, Y.-K.; Huang, C.-J.; Cheng, H.-M.; Lin, I.-N.; Liu, K.-S.; Chen, I.-C. Characterization and Field-Emission Properties of Needle-like Zinc Oxide Nanowires Grown Vertically on Conductive Zinc Oxide Films. Adv. Funct. Mater. 2003, 13, 811–814. [Google Scholar] [CrossRef]
- Li, C.; Fang, G.; Li, J.; Ai, L.; Dong, B.; Zhao, X. Effect of Seed Layer on Structural Properties of ZnO Nanorod Arrays Grown by Vapor-Phase Transport. J. Phys. Chem. C 2008, 112, 990–995. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Menzel, A.; Goldberg, R.; Burshtein, G.; Lumelsky, V.; Subannajui, K.; Zacharias, M.; Lifshitz, Y. Role of Carrier Gas Flow and Species Diffusion in Nanowire Growth from Thermal CVD. J. Phys. Chem. C 2012, 116, 5524–5530. [Google Scholar] [CrossRef]
- Hassan, N.K.; Hashim, M.R.; Mahdi, M.A.; Allam, N.K. A Catalyst-Free Growth of ZnO Nanowires on Si (100) Substrates: Effect of Substrate Position on Morphological, Structural and Optical Properties. ECS J. Solid State Sci. Technol. 2012, 1, P86–P89. [Google Scholar] [CrossRef]
- Babu, E.S.; Saravanakumar, B.; Ravi, G.; Yuvakkumar, R.; Ganesh, V.; Guduru, R.K.; Kim, S. Zinc oxide nanotips growth by controlling vapor deposition on substrates. J. Mater. Sci. Mater. Electron. 2018, 29, 6149–6156. [Google Scholar] [CrossRef]
- Lupan, O.; Emelchenko, G.A.; Ursaki, V.V.; Chai, G.; Redkin, A.N.; Gruzintsev, A.N.; Tiginyanu, I.M.; Chow, L.; Ono, L.K.; Roldan Cuenya, B.; et al. Synthesis and characterization of ZnO nanowires for nanosensor applications. Mater. Res. Bull. 2010, 45, 1026–1032. [Google Scholar] [CrossRef]
- Huber, F.; Puchinger, A.; Ahmad, W.; Madel, M.; Bauer, S.; Thonke, K. Controlled growth of ZnO layers and nanowires using methane as reducing precursor. J. Mater. Res. 2017, 32, 4087–4094. [Google Scholar] [CrossRef]
- Mohanta, S.K.; Kim, D.C.; Cho, H.K.; Chua, S.J.; Tripathy, S. Structural and optical properties of ZnO nanorods grown by metal organic chemical vapor deposition. J. Cryst. Growth 2008, 310, 3208–3213. [Google Scholar] [CrossRef]
- Menzel, A.; Subannajui, K.; Bakhda, R.; Wang, Y.; Thomann, R.; Zacharias, M. Tuning the Growth Mechanism of ZnO Nanowires by Controlled Carrier and Reaction Gas Modulation in Thermal CVD. J. Phys. Chem. Lett. 2012, 3, 2815–2821. [Google Scholar] [CrossRef]
- Cha, S.N.; Song, B.G.; Jang, J.E.; Jung, J.E.; Han, I.T.; Ha, J.H.; Hong, J.P.; Kang, D.J.; Kim, J.M. Controlled growth of vertically aligned ZnO nanowires with different crystal orientation of the ZnO seed layer. Nanotechnology 2008, 19, 235601. [Google Scholar] [CrossRef]
- Liang, S.-D. Quantum Tunneling and Field Electron Emission Theories; World Scientific: Singapure, 2014; ISBN 978-981-4440-21-9. [Google Scholar]
- Minami, T.; Miyata, T.; Yamamoto, T. Work function of transparent conducting multicomponent oxide thin films prepared by magnetron sputtering. Surf. Coatings Technol. 1998, 108–109, 583–587. [Google Scholar] [CrossRef]
- Forbes, R.G.; Edgcombe, C.; Valdrè, U. Some comments on models for field enhancement. Ultramicroscopy 2003, 95, 57–65. [Google Scholar] [CrossRef]
- Biswas, D. A universal formula for the field enhancement factor. Phys. Plasmas 2018, 25, 043113. [Google Scholar] [CrossRef]
- Jo, S.H.; Lao, J.Y.; Ren, Z.F.; Farrer, R.A.; Baldacchini, T.; Fourkas, J.T. Field-emission studies on thin films of zinc oxide nanowires. Appl. Phys. Lett. 2003, 83, 4821–4823. [Google Scholar] [CrossRef]
- Zhao, Q.; Zhang, H.Z.; Zhu, Y.W.; Feng, S.Q.; Sun, X.C.; Xu, J.; Yu, D.P. Morphological effects on the field emission of ZnO nanorod arrays. Appl. Phys. Lett. 2005, 86, 203115. [Google Scholar] [CrossRef]
- Ahmad, A.; Tripathi, V.K.; Shang, X.; Wang, M.; Qu, S. Model calculation of the scanned field enhancement factor of CNTs. Nanotechnology 2006, 17, 3798–3801. [Google Scholar] [CrossRef]
- Lee, C.J.; Lee, T.J.; Lyu, S.C.; Zhang, Y.; Ruh, H.; Lee, H.J. Field emission from well-aligned zinc oxide nanowires grown at low temperature. Appl. Phys. Lett. 2002, 81, 3648–3650. [Google Scholar] [CrossRef]
- Park, J.; Qin, H.; Scalf, M.; Hilger, R.T.; Westphall, M.S.; Smith, L.M.; Blick, R.H. A mechanical nanomembrane detector for time-of-flight mass spectrometry. Nano Lett. 2011, 11, 3681–3684. [Google Scholar] [CrossRef]
- Huang, J.-M.; Tsai, S.-Y.; Ku, C.-S.; Lin, C.-M.; Chen, S.-Y.; Lee, H.-Y. Enhanced electrical properties and field emission characteristics of AZO/ZnO-nanowire core–shell structures. Phys. Chem. Chem. Phys. 2016, 18, 15251–15259. [Google Scholar] [CrossRef]
- Yang, J.H.; Lee, S.Y.; Song, W.S.; Shin, Y.S.; Park, C.; Kim, H.-J.; Cho, W.; An, K.-S. Field emission properties of ZnO nanorods coated with NiO film. J. Vac. Sci. Technol. B 2008, 26, 1021. [Google Scholar] [CrossRef]
- Henkel, C.; Zierold, R.; Kommini, A.; Haugg, S.; Thomason, C.; Aksamija, Z.; Blick, R.H. Resonant Tunneling Induced Enhancement of Electron Field Emission by Ultra-Thin Coatings. Sci. Rep. 2019, 9, 6840. [Google Scholar] [CrossRef]








© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hedrich, C.; Haugg, S.; Pacarizi, L.; P. Furlan, K.; Blick, R.H.; Zierold, R. Low-Temperature Vapor-Solid Growth of ZnO Nanowhiskers for Electron Field Emission. Coatings 2019, 9, 698. https://doi.org/10.3390/coatings9110698
Hedrich C, Haugg S, Pacarizi L, P. Furlan K, Blick RH, Zierold R. Low-Temperature Vapor-Solid Growth of ZnO Nanowhiskers for Electron Field Emission. Coatings. 2019; 9(11):698. https://doi.org/10.3390/coatings9110698
Chicago/Turabian StyleHedrich, Carina, Stefanie Haugg, Leutrim Pacarizi, Kaline P. Furlan, Robert H. Blick, and Robert Zierold. 2019. "Low-Temperature Vapor-Solid Growth of ZnO Nanowhiskers for Electron Field Emission" Coatings 9, no. 11: 698. https://doi.org/10.3390/coatings9110698
APA StyleHedrich, C., Haugg, S., Pacarizi, L., P. Furlan, K., Blick, R. H., & Zierold, R. (2019). Low-Temperature Vapor-Solid Growth of ZnO Nanowhiskers for Electron Field Emission. Coatings, 9(11), 698. https://doi.org/10.3390/coatings9110698





