A Rapid Synthesis of Mesoporous Mn2O3 Nanoparticles for Supercapacitor Applications
Abstract
1. Introduction
2. Materials and Methods
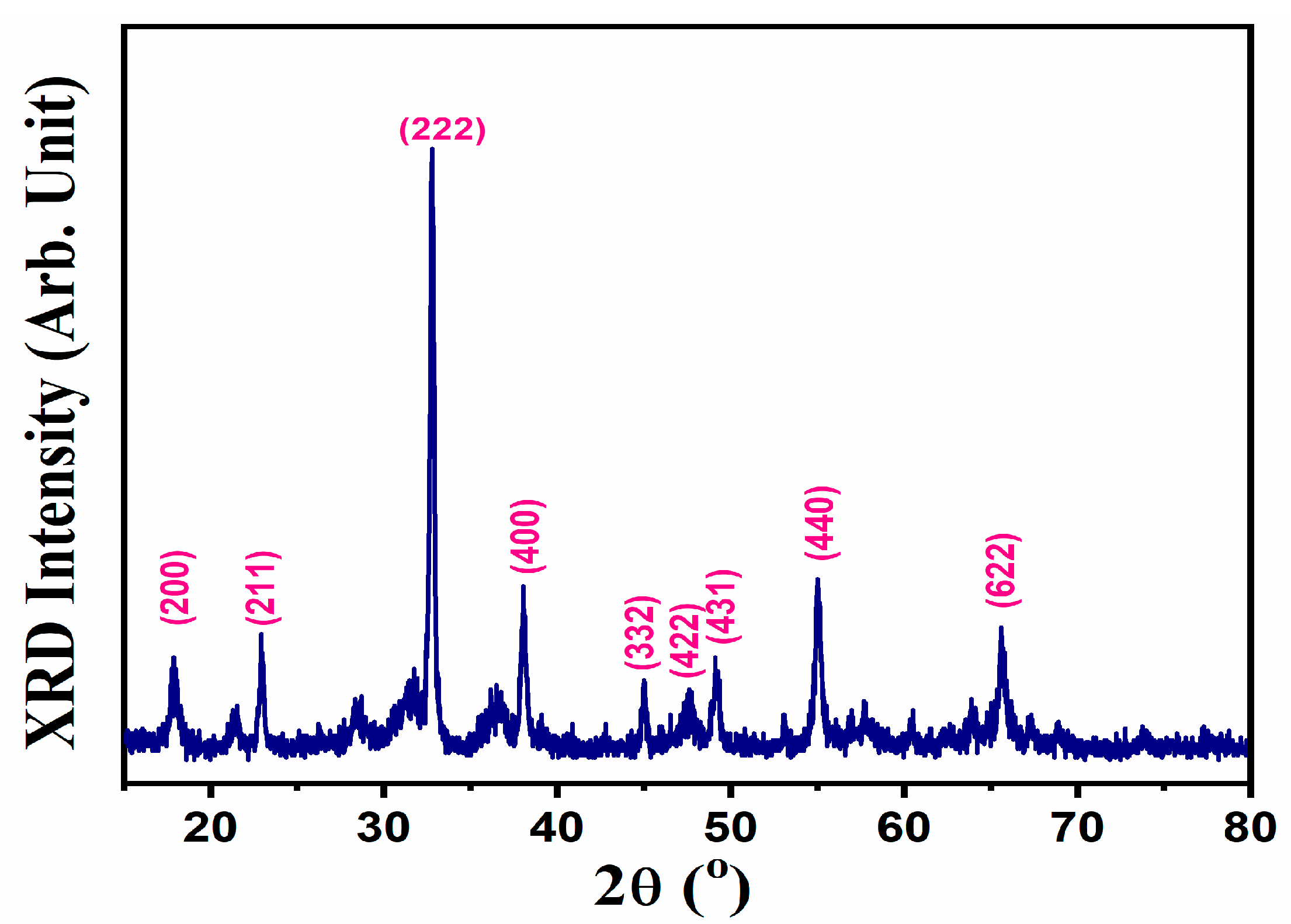
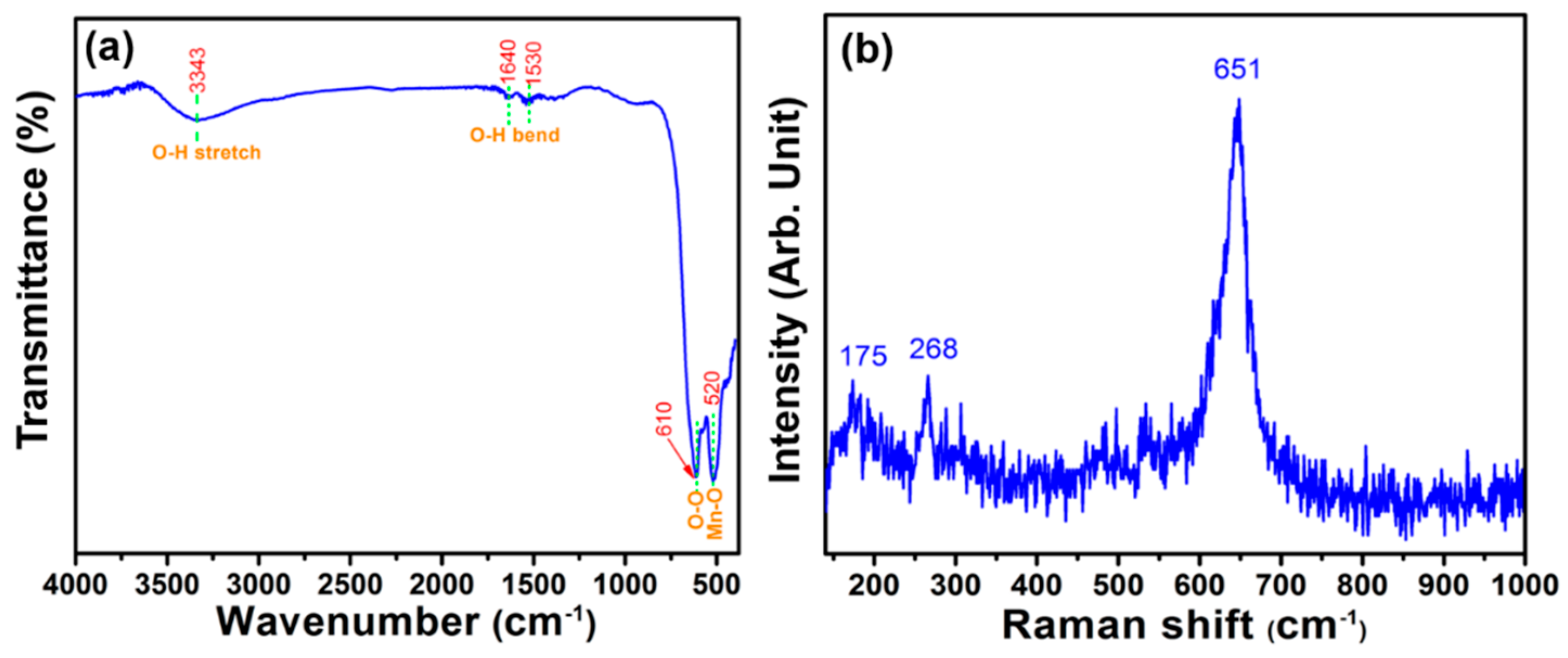
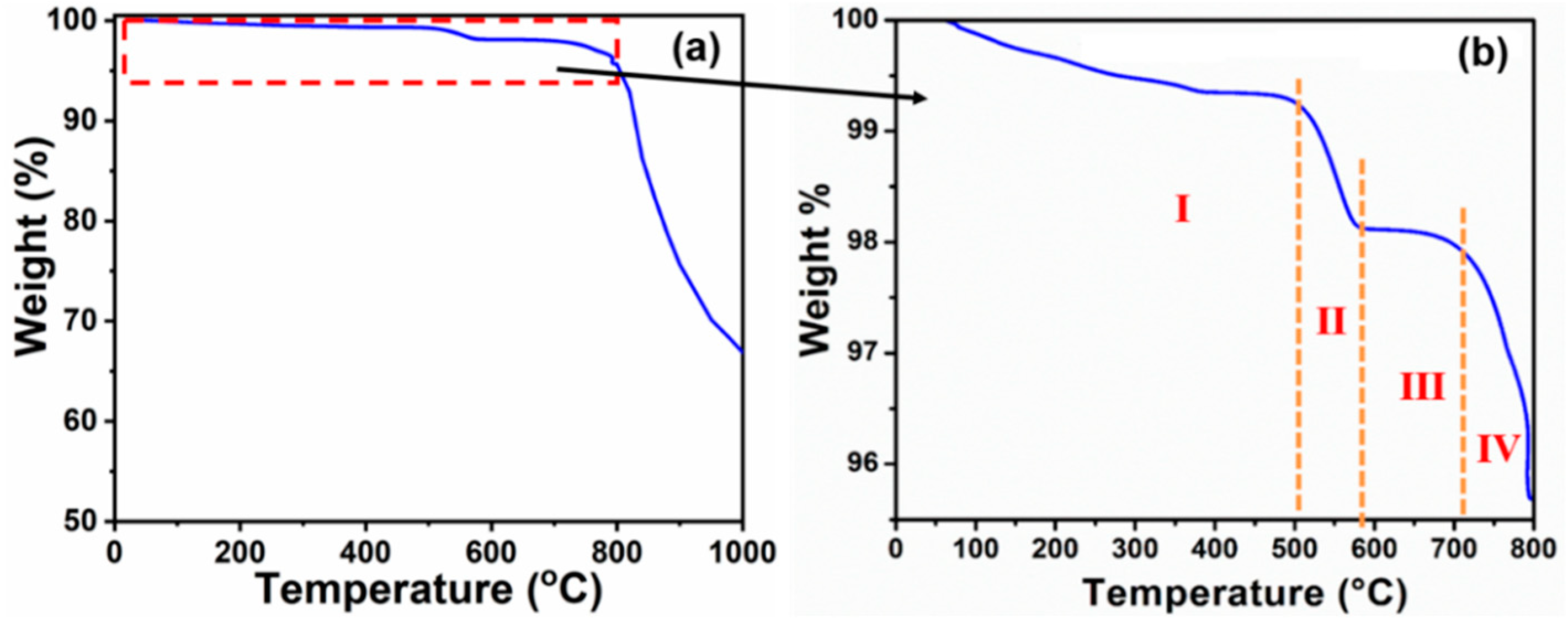
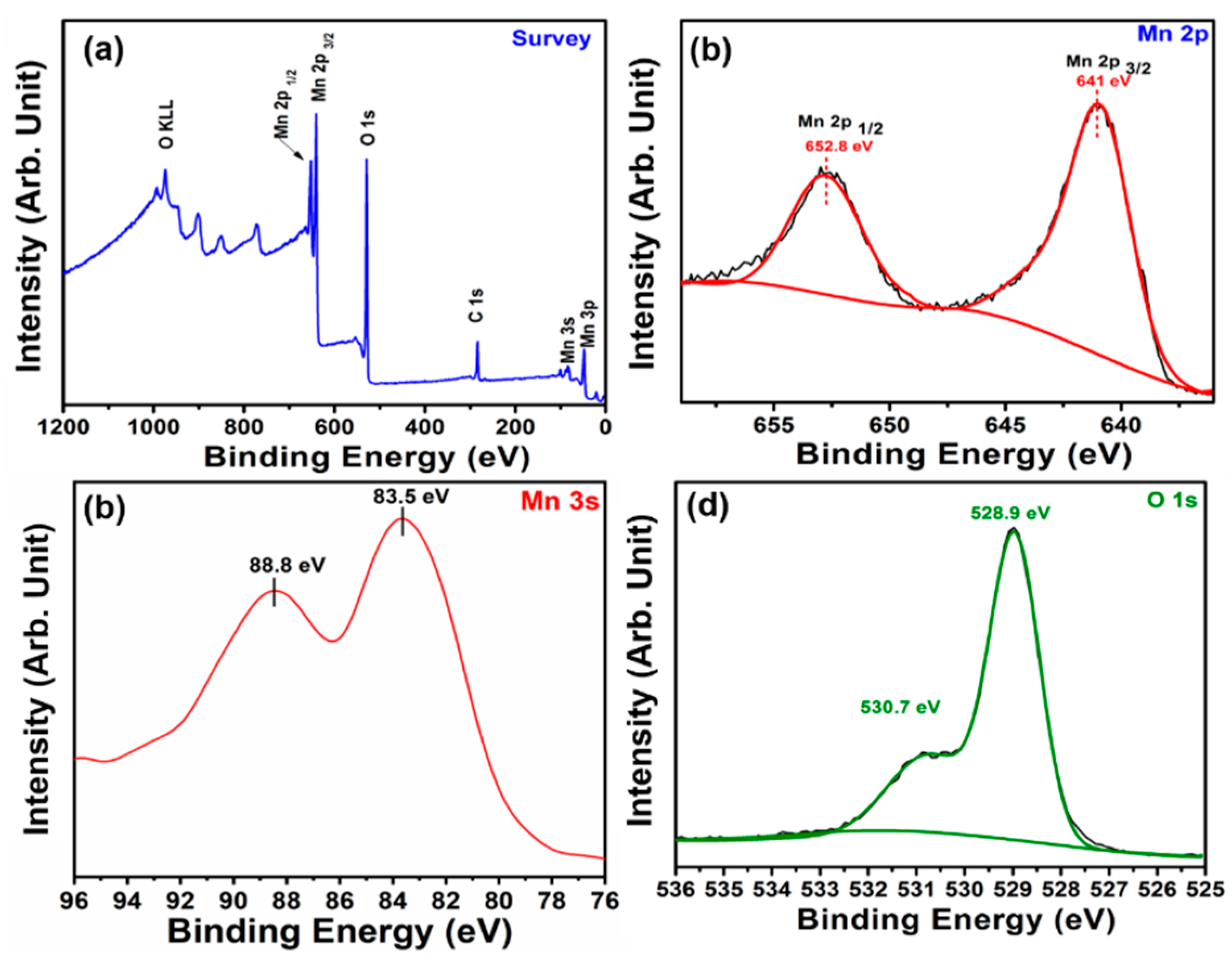
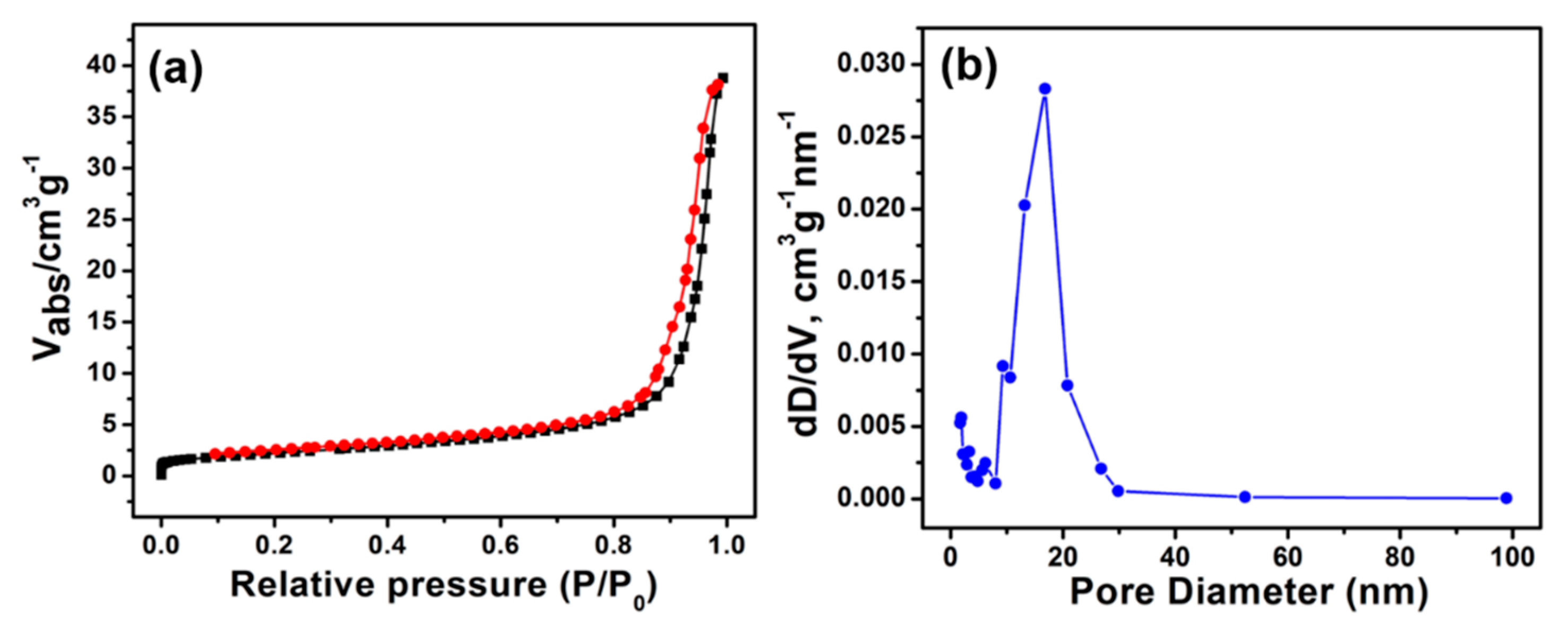
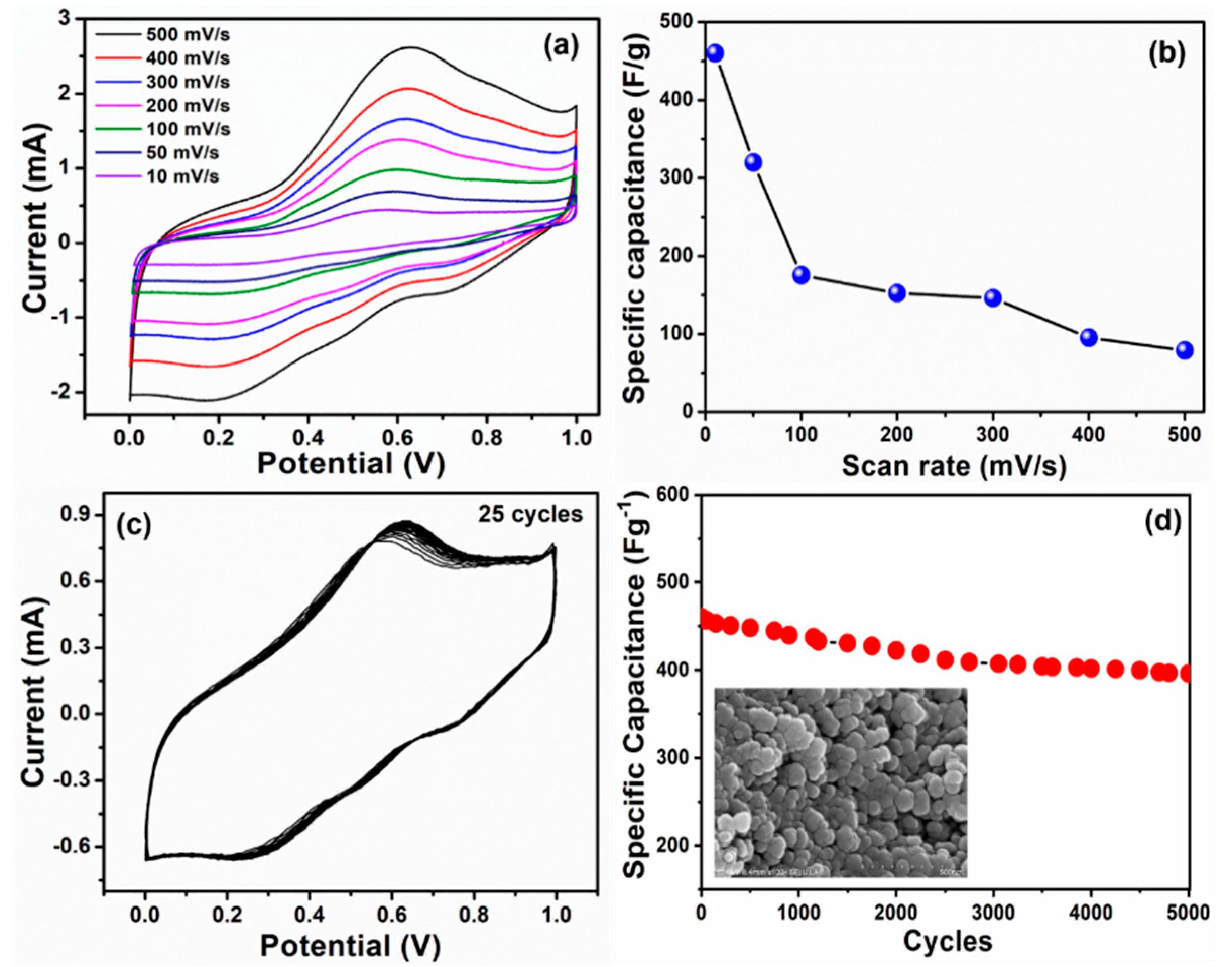
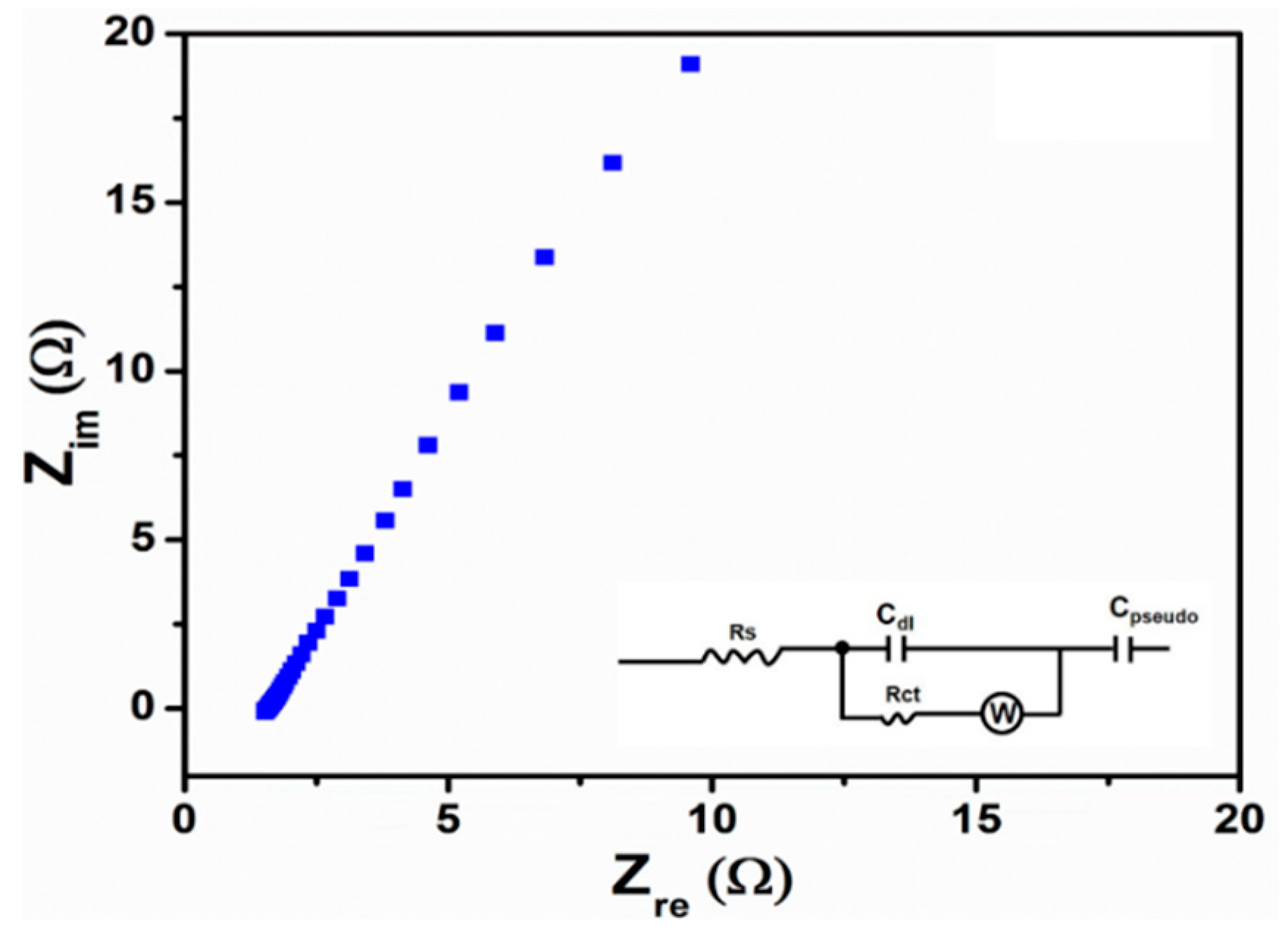
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Miller, J.R.; Simon, P. Electrochemical capacitors for energy management. Science 2008, 321, 651–652. [Google Scholar] [CrossRef] [PubMed]
- Simon, P.; Gogotsi, Y. Materials for electrochemical capacitors. Nat. Mater. 2008, 7, 845–854. [Google Scholar] [CrossRef]
- Wei, D.; Scherer, M.R.; Bower, C.; Andrew, P.; Ryhanen, T.; Steiner, U. A nanostructured electrochromic supercapacitor. Nano Lett. 2012, 12, 1857–1862. [Google Scholar] [CrossRef] [PubMed]
- Yuan, C.Z.; Wu, H.B.; Xie, Y.; Lou, X.W. Mixed transition-metal oxides: Design, synthesis, and energy-related applications. Angew. Chem. Int. Ed. 2014, 53, 1488–1504. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Li, C.; Ma, J. Polyaniline–MnO2 coaxial nanofiber with hierarchical structure for high-performance supercapacitors. J. Mater. Chem. 2012, 22, 2751. [Google Scholar] [CrossRef]
- Devaraj, S.; Munichandraiah, N. High capacitance of electrodeposited MnO2 by the effect of a surface-active agent. Electrochem. Solid State Lett. 2005, 8, 373–377. [Google Scholar] [CrossRef]
- Zhao, D.; Tian, J.S.; Ji, Q.Q.; Zhang, J.T.; Zhao, X.S.; Guo, P.Z. Mn2O3 nanomaterials: Facile synthesis and electrochemical properties. Chin. J. Inorg. Chem. 2010, 26, 832–838. [Google Scholar]
- Jang, G.; Ameen, S.; Akhtar, M.S.; Kim, E.-B.; Shin, H.S. Electrochemical Investigations of Hydrothermally Synthesized Porous Cobalt Oxide (Co3O4) Nanorods: Supercapacitor Application. ChemistrySelect 2017, 2, 8941–8949. [Google Scholar] [CrossRef]
- Li, Z.Y.; Bui, P.T.M.; Kwak, D.H.; Akhtar, M.S.; Yang, O.B. Enhanced electrochemical activity of low temperature solution process synthesized Co3O4 nanoparticles for pseudo-supercapacitors applications. Ceram. Int. 2016, 42, 1879–1885. [Google Scholar] [CrossRef]
- Li, Z.Y.; Akhtar, M.S.; Yang, O.B. Supercapacitors with ultrahigh energy density based on mesoporous carbon nanofibers: Enhanced double-layer electrochemical properties. J. Alloy. Compd. 2015, 653, 212–218. [Google Scholar] [CrossRef]
- Seo, M.K.; Saouab, A.; Park, S.J. Effect of annealing temperature on electrochemical characteristics of ruthenium oxide/multi-walled carbon nanotube composites. Mater. Sci. Eng. B 2010, 167, 65–69. [Google Scholar] [CrossRef]
- Shambharkar, B.H.; Umare, S.S. Production and characterization of polyaniline/Co3O4 nanocomposite as a cathode of Zn–polyaniline battery. Mater. Sci. Eng. B 2010, 175, 120–128. [Google Scholar] [CrossRef]
- Zheng, Z.; Huang, L.; Zhou, Y.; Hu, X.W.; Ni, X.M. Large-scale synthesis of mesoporous CoO-doped NiO hexagonal nanoplatelets with improved electrochemical performance. Solid State Sci. 2009, 11, 1439–1443. [Google Scholar] [CrossRef]
- Prasad, K.R.; Miura, N. Electrochemical synthesis and characterization of nanostructured tin oxide for electrochemical redox supercapacitors. Electrochem. Commun. 2004, 6, 849–852. [Google Scholar] [CrossRef]
- Jiang, H.; Zhao, T.; Ma, J.; Yan, C.; Li, C. Ultrafine manganese dioxide nanowire network for high-performance supercapacitors. Chem. Commun. 2011, 47, 1264. [Google Scholar] [CrossRef] [PubMed]
- Rakhi, R.B.; Chen, W.; Cha, D.; Alshareef, H.N. Substrate dependent self-organization of mesoporous cobalt oxide nanowires with remarkable pseudocapacitance. Nano Lett. 2012, 12, 2559. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.; Lattanzi, M.W.; Chen, Y.; Kou, X.; Li, W.; Fan, X.; Unruh, K.M.; Chen, J.G.; Xiao, J.Q. Supercapacitor electrodes with high-energy and power densities prepared from monolithic NiO/Ni nanocomposites. Angew. Chem. 2011, 50, 6847–6850. [Google Scholar] [CrossRef]
- Benson, J.; Boukhalfa, S.; Magasinski, A.; Kvit, A.; Yushin, G. Chemical vapor deposition of aluminum nanowires on metal substrates for electrical energy storage applications. ACS Nano 2012, 6, 118–125. [Google Scholar] [CrossRef]
- Xie, K.; Li, J.; Lai, Y.; Lu, W.; Zhang, Z.; Liu, Y.; Zhou, L.; Huang, H. Highly ordered iron oxide nanotube arrays as electrodes for electrochemical energy storage. Electrochem. Commun. 2011, 13, 657–660. [Google Scholar] [CrossRef]
- Ghosh, S.; Polaki, S.R.; Sahoob, G.; Jin, E.-M.; Kamruddin, M.; Cho, J.S.; Jeong, S.M. Designing metal oxide-vertical graphene nanosheets structures for 2.6 V aqueous asymmetric electrochemical capacitor. J. Ind. Eng. Chem. 2019, 72, 107–116. [Google Scholar] [CrossRef]
- Nagamuthu, S.; Ryu, K.-S. MOF-derived microstructural interconnected network porous Mn2O3/C as negative electrode material for asymmetric supercapacitor device. CrystEngComm 2019, 21, 1442–1451. [Google Scholar] [CrossRef]
- Sui, N.; Duan, Y.; Jiao, X.; Chen, D. Large-scale preparation and catalytic properties of one-dimensional α/β-MnO2 nanostructures. J. Phys. Chem. C 2009, 113, 8560–8565. [Google Scholar] [CrossRef]
- Kolathodi, M.S.; Rao, S.N.H.; Natarajana, T.S.; Singh, G. Beaded manganese oxide (Mn2O3) nanofibers: Preparation and application for capacitive energy storage. J. Mater. Chem. A 2016, 4, 7883–7891. [Google Scholar] [CrossRef]
- Nakayama, M.; Tanaka, A.; Sato, Y.; Tonosaki, T.; Ogura, K. Electrodeposition of manganese and molybdenum mixed oxide thin films and their charge storage properties. Langmuir 2005, 21, 5907. [Google Scholar] [CrossRef]
- Wu, Z.-S.; Ren, W.; Wang, D.-W.; Li, F.; Liu, B. High-energy MnO2 nanowire/graphene and graphene asymmetric electrochemical capacitors. ACS Nano 2010, 4, 5835–5842. [Google Scholar] [CrossRef]
- Fischer, A.E.; Pettigrew, K.A.; Rolison, D.R.; Stroud, R.M.; Long, J.W. Incorporation of homogeneous, nanoscale MnO2 within ultraporous carbon structures via self-limiting electroless deposition: Implications for electrochemical capacitors. Nano Lett. 2007, 7, 281–286. [Google Scholar] [CrossRef]
- Ghodbane, O.; Pascal, J.-L.; Favier, F. Microstructural effects on charge-storage properties in MnO2-based electrochemical supercapacitors. ACS Appl. Mater. Interfaces 2009, 1, 1130–1139. [Google Scholar] [CrossRef]
- Zhi, M.; Xiang, C.; Li, J.; Li, M.; Wu, N. Nanostructured carbon–metal oxide composite electrodes for supercapacitors: A review. Nanoscale 2013, 5, 72–88. [Google Scholar] [CrossRef]
- Jiang, J.; Li, Y.; Liu, J.; Huang, X.; Yuan, C.; Lou, X.W. Recent advances in metal oxide-based electrode architecture design for electrochemical energy storage. Adv. Mater. 2012, 24, 5166–5180. [Google Scholar] [CrossRef]
- Han, Z.J.; Seo, D.H.; Yick, S.; Chen, J.H.; Ostrikov, K. MnOx/carbon nanotube/reduced graphene oxide nanohybrids as high-performance supercapacitor electrodes. NPG Asia Mater. 2014, 6, e140. [Google Scholar] [CrossRef]
- Demir, M.; Ashourirad, B.; Mugumya, J.H.; Saraswata, S.K.; El-Kaderi, H.M.; Gupta, R.B. Nitrogen and oxygen dual-doped porous carbons prepared from pea protein as electrode materials for high performance supercapacitors. Int. J. Hydrog. Energy 2018, 43, 18549–18558. [Google Scholar] [CrossRef]
- Cheng, F.; Shen, J.; Ji, W.; Tao, Z.; Chen, J. Selective synthesis of manganese oxide nanostructures for electrocatalytic oxygen reduction. ACS Appl. Mater. Interfaces 2009, 1, 460–466. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, R. Introduction to X-Ray Powder Diffractometry; John Wiley & Sons, Inc.: New York, NY, USA, 1996; p. 90. [Google Scholar]
- Gillot, B.; El Guendouzi, M.; Laarj, M. Particle size effects on the oxidation–reduction behavior of Mn3O4 hausmannite. Mater. Chem. Phys. 2001, 70, 54–60. [Google Scholar] [CrossRef]
- Chen, Z.W.; Lai, J.K.L.; Shek, C.H. Influence of grain size on the vibrational properties in Mn2O3 nanocrystals. J. Non Cryst. Solids 2006, 352, 3285–3289. [Google Scholar] [CrossRef]
- Han, Y.-F.; Ramesh, K.; Chen, L.; Widjaja, E.; Chilukoti, S.; Chen, F. Controlled synthesis, characterization, and catalytic properties of Mn2O3 and Mn3O4 nanoparticles supported on mesoporous silica SBA-15. J. Phys. Chem. B 2007, 111, 2830–2833. [Google Scholar]
- Zhang, Y.; Yan, Y.; Wang, X.; Li, G.; Deng, D.; Jiang, L.; Shu, C.; Wang, C. Facile synthesis of porous Mn2O3 nanoplates and their electrochemical behavior as anode materials for lithium ion batteries. Chem. A Eur. J. 2014, 20, 6126–6130. [Google Scholar] [CrossRef]
- Jahan, M.; Tominaka, S.; Henzie, J. Phase pure α-Mn2O3 prisms and their bifunctional electrocatalytic activity in oxygen evolution and reduction reactions. Dalton Trans. 2016, 45, 18494–18501. [Google Scholar] [CrossRef]
- Ilton, E.S.; Post, J.E.; Heaney, P.J.; Ling, F.T.; Kerisit, S.N. XPS determination of Mn oxidation states in Mn (hydr)oxides. Appl. Surf. Sci. 2016, 366, 475–485. [Google Scholar] [CrossRef]
- Toupin, M.; Brousse, T.; Bélanger, D. Influence of microstucture on the charge storage properties of chemically synthesized manganese dioxide. Chem. Mater. 2002, 14, 3946–3952. [Google Scholar] [CrossRef]
- Toupin, M.; Brousse, T.; Bélanger, D. Charge storage mechanism of MnO2 electrode used in aqueous electrochemical capacitor. Chem. Mater. 2004, 16, 3184–3190. [Google Scholar] [CrossRef]
- Rajkumar, M.; Hsu, C.-T.; Wu, T.-H.; Chen, M.-G.; Hu, C.-C. Advanced materials for aqueous supercapacitors in the asymmetric design. Prog. Nat. Sci. Mater. Int. 2015, 25, 527. [Google Scholar] [CrossRef]
- Gopalakrishnan, I.K.; Bagkar, N.; Ganguly, R.; Kulshreshtha, S.K. Synthesis of superparamagnetic Mn3O4 nanocrystallites by ultrasonic irradiation. J. Cryst. Growth 2005, 280, 436. [Google Scholar] [CrossRef]
- Li, Z.-Y.; Akhtar, M.S.; Bui, P.T.M.; Yang, O.-B. Predominance of two dimensional (2D) Mn2O3 nanowalls thin film for high performance electrochemical supercapacitors. Chem. Eng. J. 2017, 330, 1240–1247. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Son, Y.-H.; Bui, P.T.M.; Lee, H.-R.; Akhtar, M.S.; Shah, D.K.; Yang, O.-B. A Rapid Synthesis of Mesoporous Mn2O3 Nanoparticles for Supercapacitor Applications. Coatings 2019, 9, 631. https://doi.org/10.3390/coatings9100631
Son Y-H, Bui PTM, Lee H-R, Akhtar MS, Shah DK, Yang O-B. A Rapid Synthesis of Mesoporous Mn2O3 Nanoparticles for Supercapacitor Applications. Coatings. 2019; 9(10):631. https://doi.org/10.3390/coatings9100631
Chicago/Turabian StyleSon, You-Hyun, Phuong T. M. Bui, Ha-Ryeon Lee, Mohammad Shaheer Akhtar, Deb Kumar Shah, and O-Bong Yang. 2019. "A Rapid Synthesis of Mesoporous Mn2O3 Nanoparticles for Supercapacitor Applications" Coatings 9, no. 10: 631. https://doi.org/10.3390/coatings9100631
APA StyleSon, Y.-H., Bui, P. T. M., Lee, H.-R., Akhtar, M. S., Shah, D. K., & Yang, O.-B. (2019). A Rapid Synthesis of Mesoporous Mn2O3 Nanoparticles for Supercapacitor Applications. Coatings, 9(10), 631. https://doi.org/10.3390/coatings9100631






