Realization of Graphene on the Surface of Electroless Ni–P Coating for Short-Term Corrosion Prevention
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
3. Results and Discussion
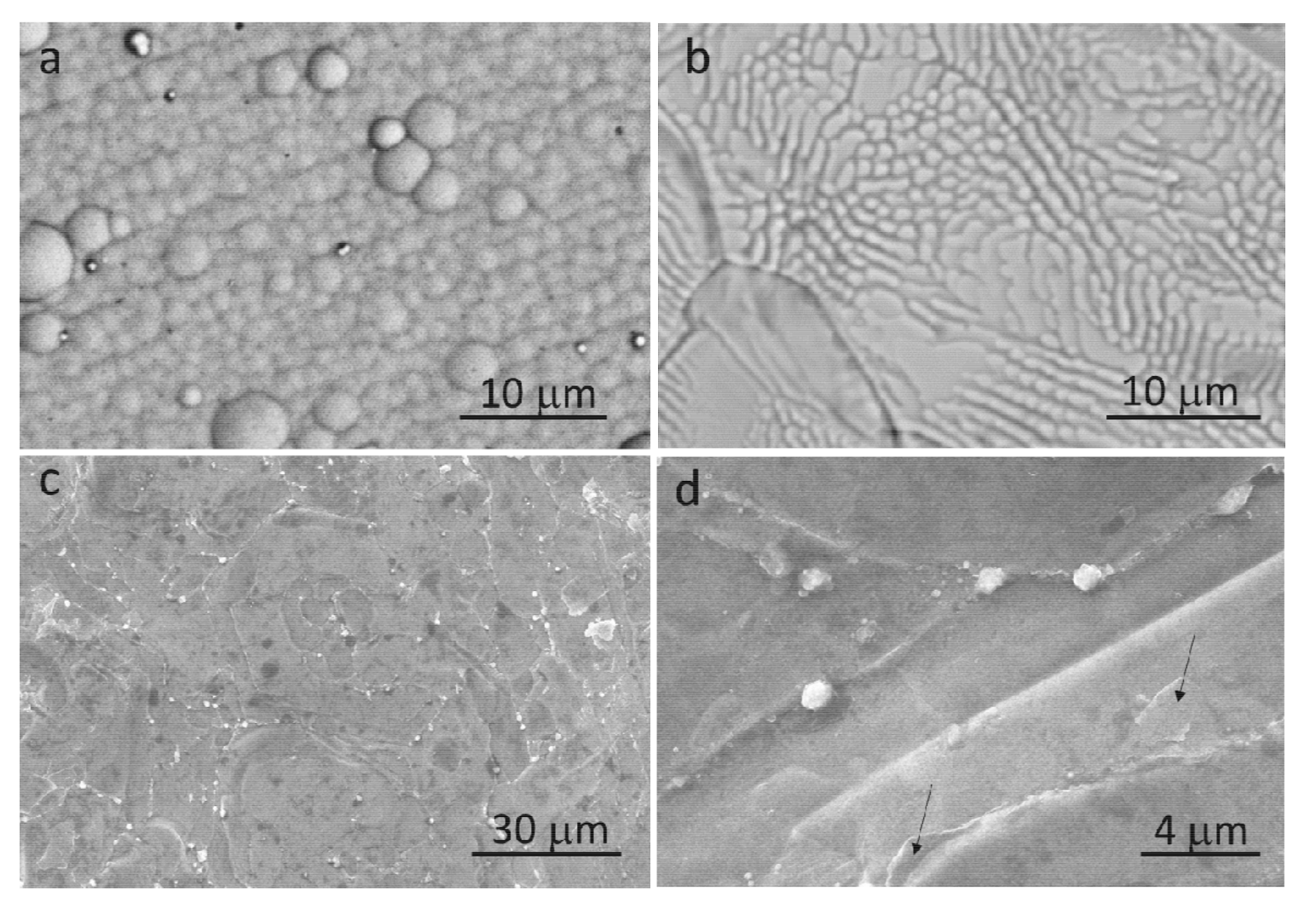
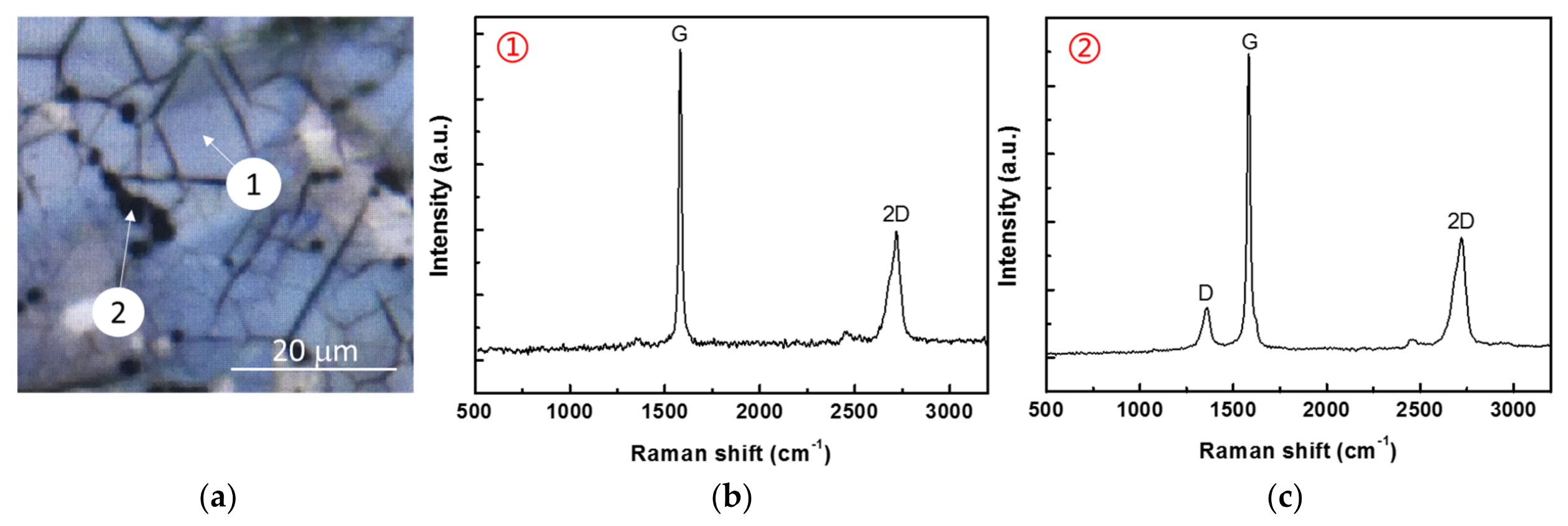
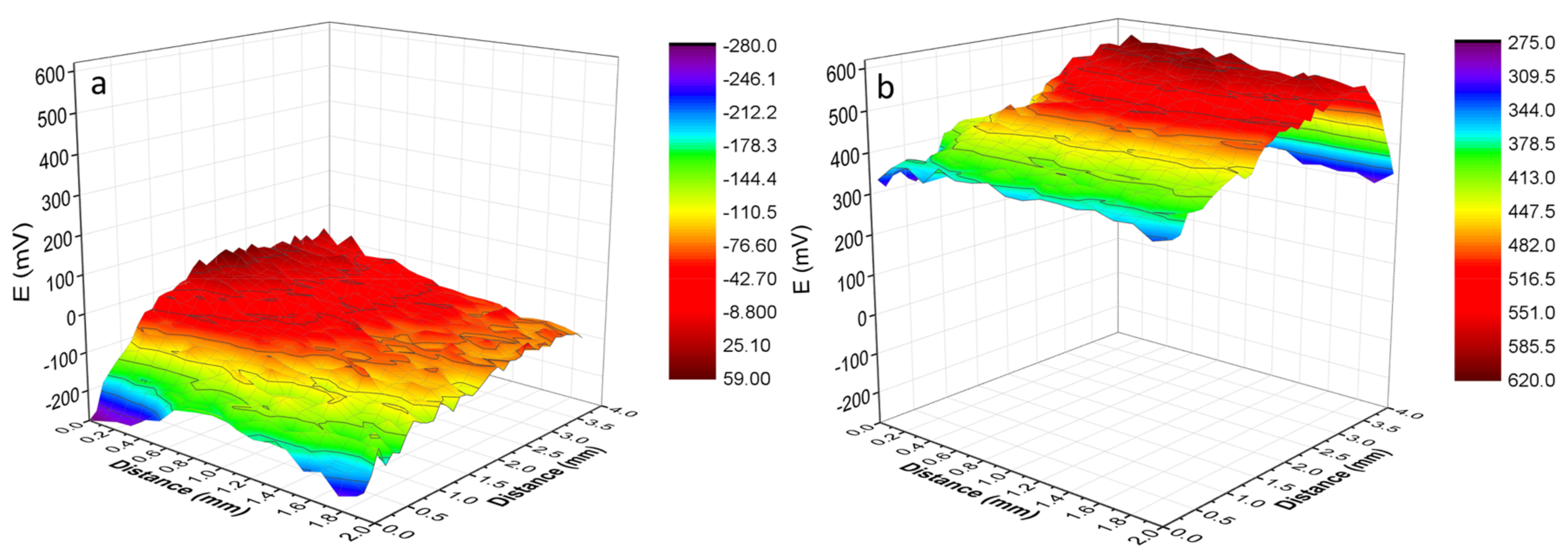
3.1. Morphology
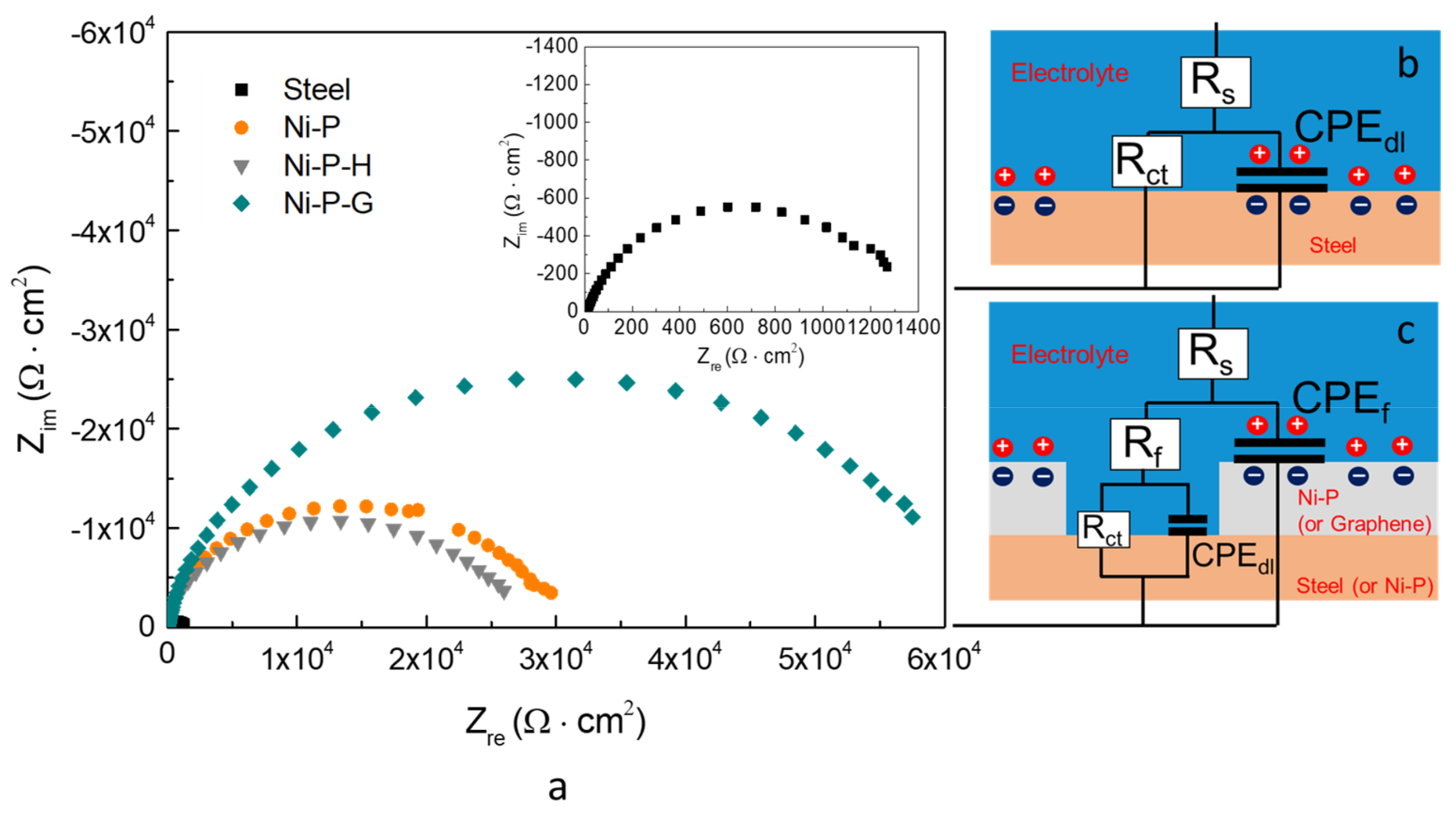
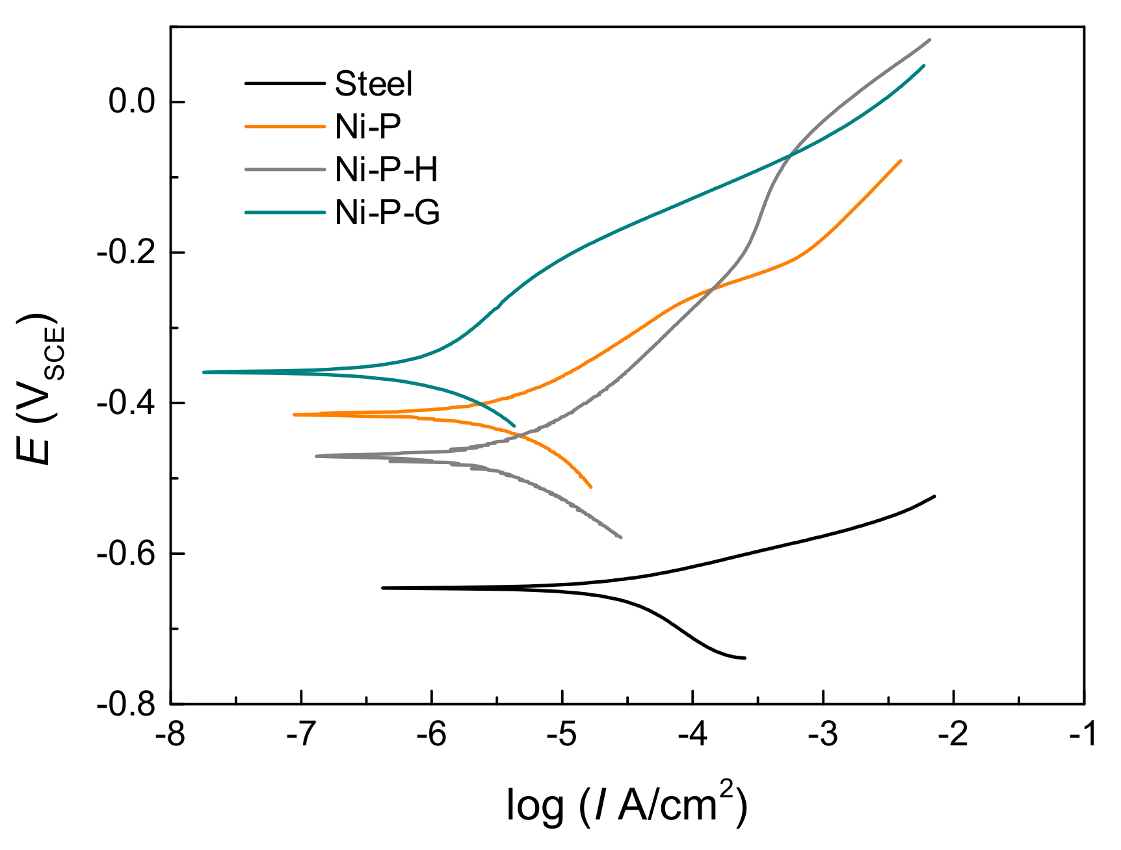
3.2. Electrochemical Tests
4. Conclusions
- (1)
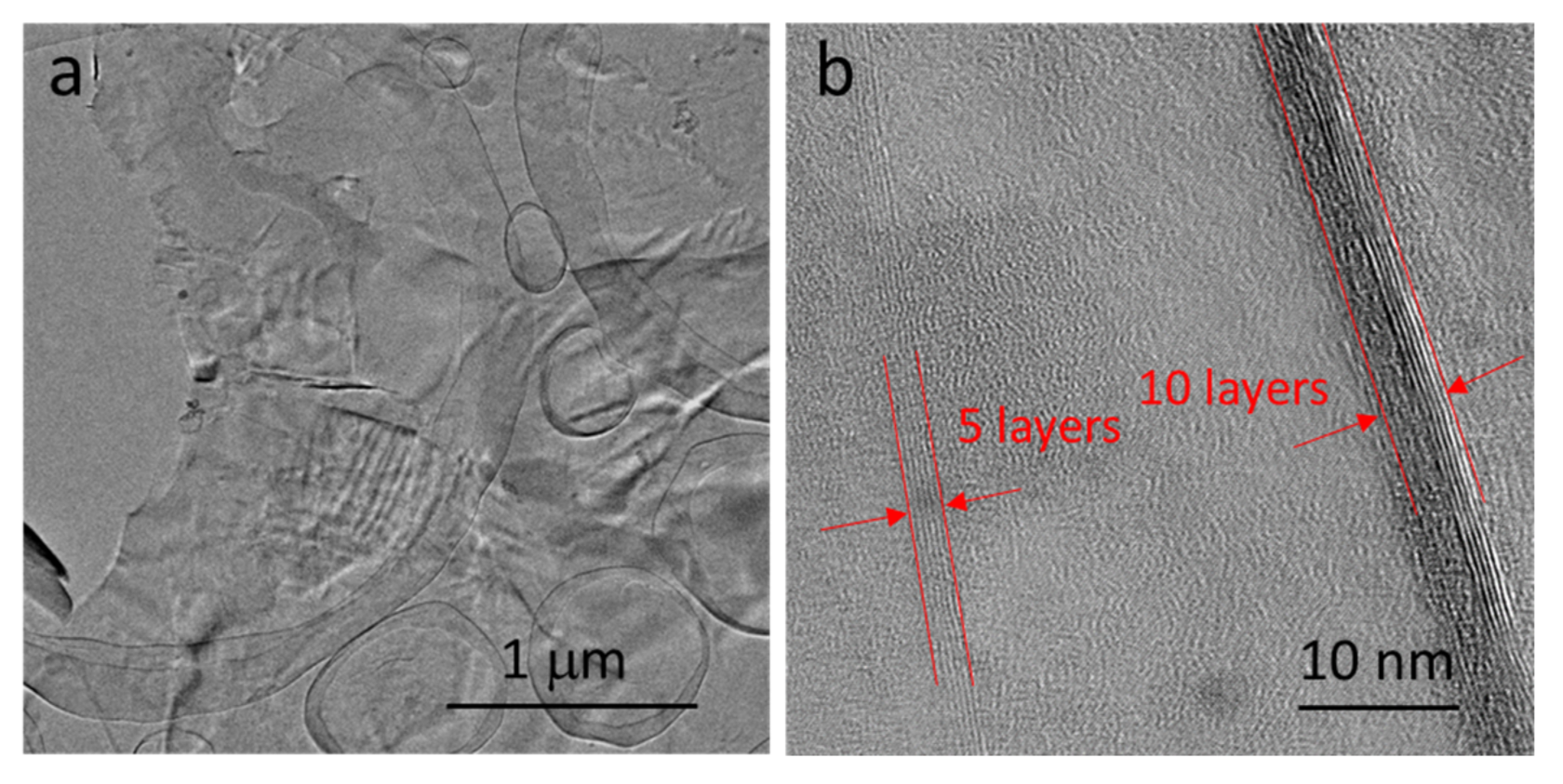
- Graphene achieved on the surface of Ni–P–G was multilayered with the number of layers around 5–10, which was determined by the IG/I2D ratio from Raman spectra and the observation of HRTEM. The ID/IG ratio revealed that the graphene at the grain region of Ni–P–H had large grain size, while that at the grain boundary contained slight defects and had smaller grain size because there were more nucleation sites there.
- (2)
- The corrosion resistance of Ni–P–G is more than two times higher than that of Ni–P and Ni–P–H, displaying a higher Rp around 6.09 × 104 Ω·cm2, a lower CPEdl, and a lower corrosion rate of 0.011 mm/year, which is ascribed to the impermeable and inert ionic barrier of graphene on the surface. Graphene also reduced the corrosion tendency of Ni–P–G by shifting its surface potential.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wang, Q.Y.; Wang, X.Z.; Luo, H.; Luo, J.L. A study on corrosion behaviors of Ni–Cr–Mo laser coating, 316 stainless steel and X70 steel in simulated solutions with H2S and CO2. Surf. Coat. Technol. 2016, 291, 250–257. [Google Scholar] [CrossRef]
- Sudagar, J.; Lian, J.; Sha, W. Electroless nickel, alloy, composite and nano coatings—A critical review. J. Alloys Compd. 2013, 571, 183–204. [Google Scholar] [CrossRef]
- Choi, W.; Lahiri, I.; Seelaboyina, R.; Kang, Y.S. Synthesis of graphene and its applications: A review. Crit. Rev. Solid State 2010, 35, 52–71. [Google Scholar] [CrossRef]
- Ming, H.; Wang, J.; Zhang, Z.; Wang, S.; Han, E.H.; Ke, W. Multilayer graphene: A potential anti-oxidation barrier in simulated primary water. J. Mater. Sci. Technol. 2014, 30, 1084–1087. [Google Scholar] [CrossRef]
- Hu, J.; Ji, Y.; Shi, Y.; Hui, F.; Duan, H.; Lanza, M.A. A review on the use of graphene as a protective coating against corrosion. Ann. J. Mater. Sci. Eng. 2014, 1, 16. [Google Scholar]
- Raman, R.S.; Banerjee, P.C.; Lobo, D.E.; Gullapalli, H.; Sumandasa, M.; Kumar, A.; Majumder, M. Protecting copper from electrochemical degradation by graphene coating. Carbon 2012, 50, 4040–4045. [Google Scholar] [CrossRef]
- Prasai, D.; Tuberquia, J.C.; Harl, R.R.; Jennings, G.K.; Bolotin, K.I. Graphene: Corrosion-inhibiting coating. ACS Nano 2012, 6, 1102–1108. [Google Scholar] [CrossRef] [PubMed]
- Stoot, A.C.; Camilli, L.; Spiegelhauer, S.A.; Yu, F.; Bøggild, P. Multilayer graphene for long-term corrosion protection of stainless steel bipolar plates for polymer electrolyte membrane fuel cell. J. Power Sources 2015, 293, 846–851. [Google Scholar] [CrossRef]
- Schriver, W.R.M.; Gannett, W.J.; Zaniewski, A.M.; Crommie, M.F.; Zettl, A. Graphene as a long-term metal oxidation barrier: Worse than nothing. ACS Nano 2013, 7, 5763–5768. [Google Scholar] [CrossRef] [PubMed]
- Yu, F.; Stoot, A.C.; Bøggild, P.; Camilli, L. Failure of multi-layer graphene coatings in acidic media. RSC Adv. 2016, 6, 21497–21502. [Google Scholar] [CrossRef]
- Li, X.; Cai, W.; An, J.; Kim, S.; Nah, J.; Yang, D.; Piner, R.; Velamakanni, A.; Jung, I.; Tutuc, E. Large-area synthesis of high-quality and uniform graphene films on copper foils. Science 2009, 324, 1312–1314. [Google Scholar] [CrossRef] [PubMed]
- Chem, A. Evolution of graphene growth on Ni and Cu by carbon isotope labeling. Nano Lett. 2009, 9, 4268–4272. [Google Scholar]
- John, R.; Ashokreddy, A.; Vijayan, C.; Pradeep, T. Single- and few-layer graphene growth on stainless steel substrates by direct thermal chemical vapor deposition. Nanotechnology 2011, 22, 165701. [Google Scholar] [CrossRef] [PubMed]
- Zhan, N.; Wang, G.; Liu, J. Cobalt-assisted large-area epitaxial graphene growth in thermal cracker enhanced gas source molecular beam epitaxy. Appl. Phys. A 2011, 105, 341–345. [Google Scholar] [CrossRef]
- Que, Y.; Xiao, W.; Fei, X.; Chen, H.; Huang, L.; Du, S.X.; Gao, H.J. Epitaxial growth of large-area bilayer graphene on Ru(0001). Appl. Phys. Lett. 2014, 104, 093110. [Google Scholar] [CrossRef]
- Liu, M.; Gao, Y.; Zhang, Y.; Zhang, Y.; Ma, D.; Ji, Q.; Gao, T.; Chen, Y.; Liu, Z. Single and polycrystalline graphene on Rh(111) following different growth mechanisms. Small 2013, 9, 1360–1366. [Google Scholar] [CrossRef] [PubMed]
- Romani, E.C.; Larrude, D.G.; Nachez, L.; Vilani, C.; de Campos, J.B.; Peripolli, S.B.; Freire, F.L. Graphene grown by chemical vapour deposition on steel substrates: Friction behaviour. Tribol. Lett. 2017, 65, 96. [Google Scholar] [CrossRef]
- Liu, Z.D.; Yin, Z.Y.; Du, Z.H.; Yang, Y.; Zhu, M.M.; Xie, L.H.; Huang, W. Low temperature growth of graphene on Cu–Ni alloy nanofibers for stable, flexible electrodes. Nanoscale 2014, 6, 5110–5115. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Fu, Y.; Edwards, M.; Jeppson, K.; Ye, L.; Liu, J. Chemical vapor deposition grown graphene on Cu-Pt alloys. Mater. Lett. 2017, 193, 255–258. [Google Scholar] [CrossRef]
- Dai, B.; Fu, L.; Zou, Z.; Wang, M.; Xu, H.; Wang, S.; Liu, Z. Rational design of a binary metal alloy for chemical vapour deposition growth of uniform single-layer graphene. Nat. Commun. 2011, 2, 522. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.Y.; Xi, Y.C.; Xu, J.; Liu, S.; Lin, Y.H.; Zhao, Y.H.; Bai, S.L. Study on properties of double-layered Ni–P–Cr composite coating prepared by the combination of electroless plating and pack cementation. J. Alloys Compd. 2017, 729, 787–795. [Google Scholar] [CrossRef]
- Sadeghzadeh-Attar, A.; AyubiKia, G.; Ehteshamzadeh, M. Improvement in tribological behavior of novel sol-enhanced electroless Ni–P-SiO2 nanocomposite coatings. Surf. Coat. Technol. 2016, 307, 837–848. [Google Scholar] [CrossRef]
- Chen, C.K.; Feng, H.M.; Lin, H.C.; Hon, M.H. The effect of heat treatment on the microstructure of electroless Ni–P coatings containing SiC particles. Thin Solid Films 2002, 416, 31–37. [Google Scholar] [CrossRef]
- Prikhod’ko, N.G.; Lesbaev, B.T.; Auelkhankyzy, M.; Mansurov, Z.A. Synthesis of graphene films in a flame. Rus. J. Phys. Chem. B 2014, 8, 61–64. [Google Scholar] [CrossRef]
- Li, Z.; Xu, Y.; Cao, B.; Qi, L.; He, S.; Wang, C.; Zhang, J.; Wang, J.; Xu, K. Raman spectra investigation of the defects of chemical vapor deposited multilayer graphene and modified by oxygen plasma treatment. Superlattice Microstruct. 2016, 99, 125–130. [Google Scholar] [CrossRef]
- Terasawa, T.; Saiki, K. Growth of graphene on Cu by plasma enhanced chemical vapor deposition. Carbon 2012, 50, 869–874. [Google Scholar] [CrossRef]
- Luo, H.; Dong, C.F.; Li, X.G.; Xiao, K. The electrochemical behaviour of 2205 duplex stainless steel in alkaline solutions with different pH in the presence of chloride. Electrochim. Acta 2012, 64, 211–220. [Google Scholar] [CrossRef]
- Ashassi-Sorkhabi, H.; Rafizadeh, S.H. Effect of coating time and heat treatment on structures and corrosion characteristics of electroless Ni–P alloy deposits. Surf. Coat. Technol. 2004, 176, 318–326. [Google Scholar] [CrossRef]
- Trabelsi, W.; Triki, E.; Dhouibi, L.; Ferreira, M.G.S.; Zheludkevich, M.L.; Montemor, M.F. The use of pre-treatments based on doped silane solutions for improved corrosion resistance of galvanised steel substrates. Surf. Coat. Technol. 2006, 200, 4240–4250. [Google Scholar] [CrossRef]
- Banerjee, P.C.; Raman, R.S. Electrochemical impedance spectroscopic investigation of the role of alkaline pre-treatment in corrosion resistance of a silane coating on magnesium alloy, ZE41. Electrochim. Acta 2011, 56, 3790–3798. [Google Scholar] [CrossRef]
- Qian, M.; Li, D.; Liu, S.B.; Gong, S.L. Corrosion performance of laser-remelted Al–Si coating on magnesium alloy AZ91D. Corros. Sci. 2010, 52, 3554–3560. [Google Scholar] [CrossRef]
- Wang, H.L.; Fan, H.B.; Zheng, J.S. Corrosion inhibition of mild steel in hydrochloric acid solution by a mercapto-triazole compound. Mater. Chem. Phys. 2003, 77, 655–661. [Google Scholar] [CrossRef]
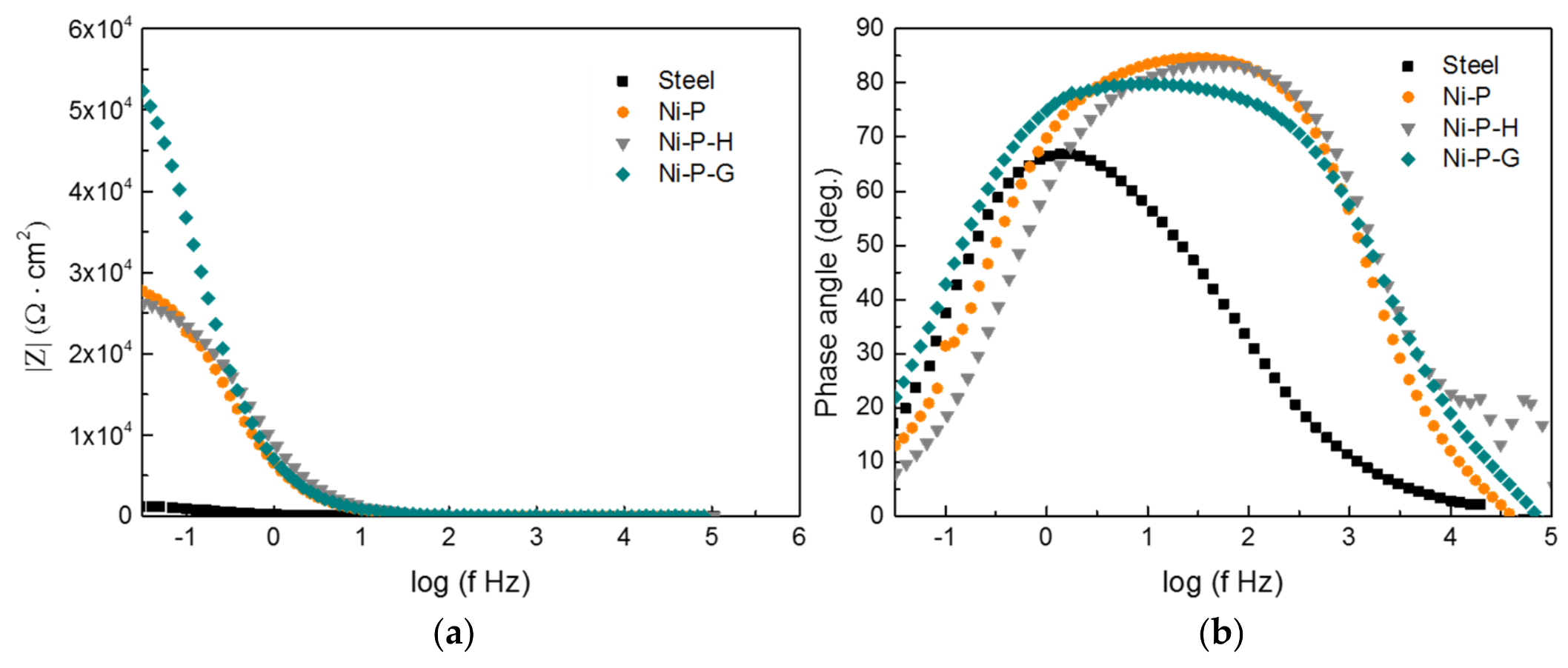
| Sample | Rs (Ω·cm2) | CPEf | Rf (Ω·cm2) | CPEdl | Rct (Ω·cm2) | Rp (Ω·cm2) | ||
|---|---|---|---|---|---|---|---|---|
| Y0 (Ω−1·cm−2·sn) | n | Y0 (Ω−1·cm−2·sn) | n | |||||
| Q235 | 5.9 | - | - | - | 1.14 × 10−3 | 0.79 | 1.51 × 103 | 1.51 × 103 |
| Ni–P | 5.4 | 2.4 × 10−5 | 0.96 | 1.66 × 104 | 4.97 × 10−5 | 0.48 | 1.58 × 104 | 3.24 × 104 |
| Ni–P–H | 6.5 | 1.73 × 10−5 | 0.93 | 2.27 × 104 | 2.55 × 10−4 | 0.83 | 4.32 × 103 | 2.70 × 104 |
| Ni–P–G | 5.9 | 1.01 × 10−5 | 0.96 | 12.33 | 1.78 × 10−5 | 0.84 | 6.09 × 104 | 6.09 × 104 |
| Samples | Q235 Steel | Ni–P | Ni–P–H | Ni–P–G |
|---|---|---|---|---|
| Average atomic mass (g/mol) | 56.0 | 89.7 | 89.7 | 89.7 |
| Icorr (A/cm2) | 5.30 × 10−5 | 3.46 × 10−6 | 3.38 × 10−6 | 9.77 × 10−7 |
| Corrosion rate (mm/year) | 0.381 | 0.039 | 0.040 | 0.011 |
| η (%) | 97.1 | 72.5 | - | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Q.-Y.; Yu, J.; Xu, J.; Fang, H.-M.; Liu, S.; Tang, Y.-R.; Xi, Y.-C.; Bai, S.-L. Realization of Graphene on the Surface of Electroless Ni–P Coating for Short-Term Corrosion Prevention. Coatings 2018, 8, 130. https://doi.org/10.3390/coatings8040130
Wang Q-Y, Yu J, Xu J, Fang H-M, Liu S, Tang Y-R, Xi Y-C, Bai S-L. Realization of Graphene on the Surface of Electroless Ni–P Coating for Short-Term Corrosion Prevention. Coatings. 2018; 8(4):130. https://doi.org/10.3390/coatings8040130
Chicago/Turabian StyleWang, Qin-Ying, Jing Yu, Jiao Xu, Hao-Ming Fang, Shuang Liu, Yi-Rong Tang, Yu-Chen Xi, and Shu-Lin Bai. 2018. "Realization of Graphene on the Surface of Electroless Ni–P Coating for Short-Term Corrosion Prevention" Coatings 8, no. 4: 130. https://doi.org/10.3390/coatings8040130
APA StyleWang, Q.-Y., Yu, J., Xu, J., Fang, H.-M., Liu, S., Tang, Y.-R., Xi, Y.-C., & Bai, S.-L. (2018). Realization of Graphene on the Surface of Electroless Ni–P Coating for Short-Term Corrosion Prevention. Coatings, 8(4), 130. https://doi.org/10.3390/coatings8040130





