Synthesis of Eugenol-Based Silicon-Containing Benzoxazines and Their Applications as Bio-Based Organic Coatings
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Measurements
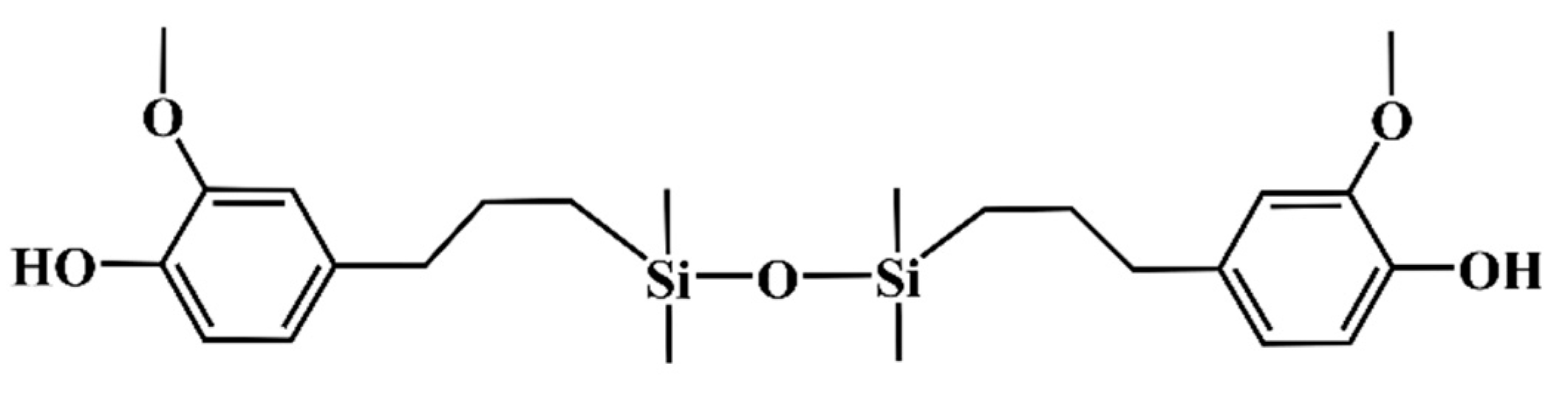
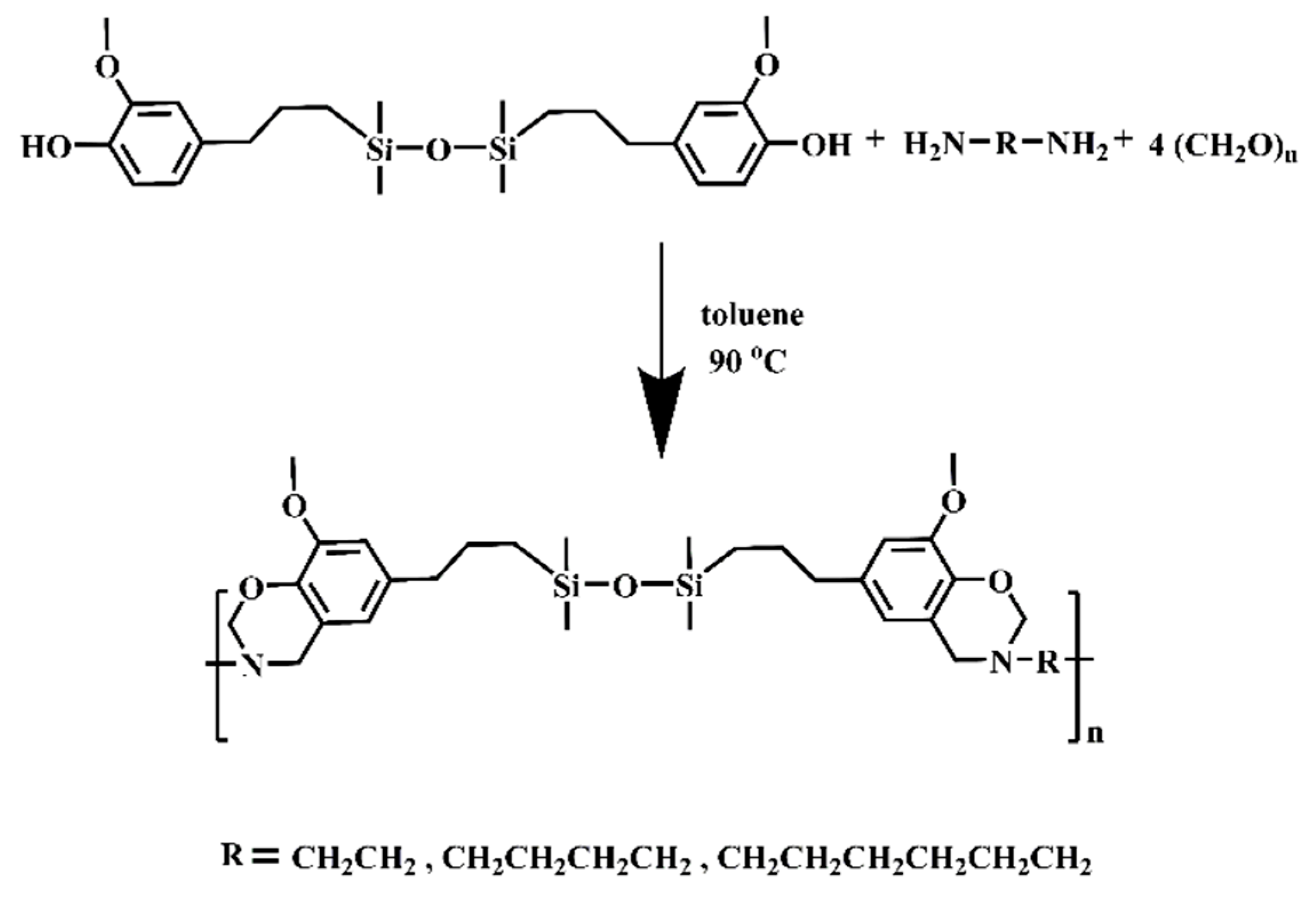
2.3. Synthesis of Different Benzoxazine Oligomers
2.4. Curing of Poly (SIE-e), Poly (SIE-b), Poly (SIE-d), and Coating Preparation
3. Results and Discussion
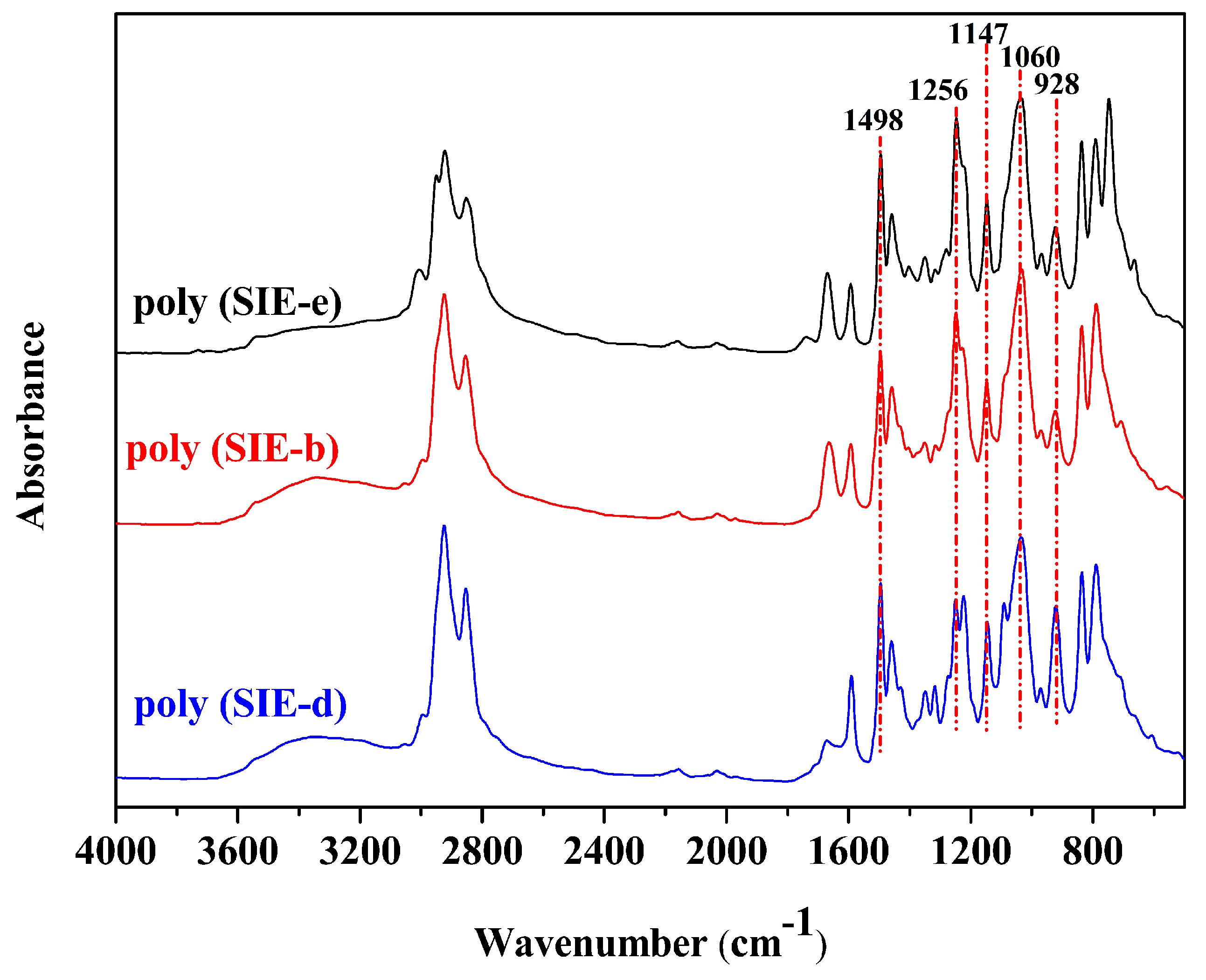
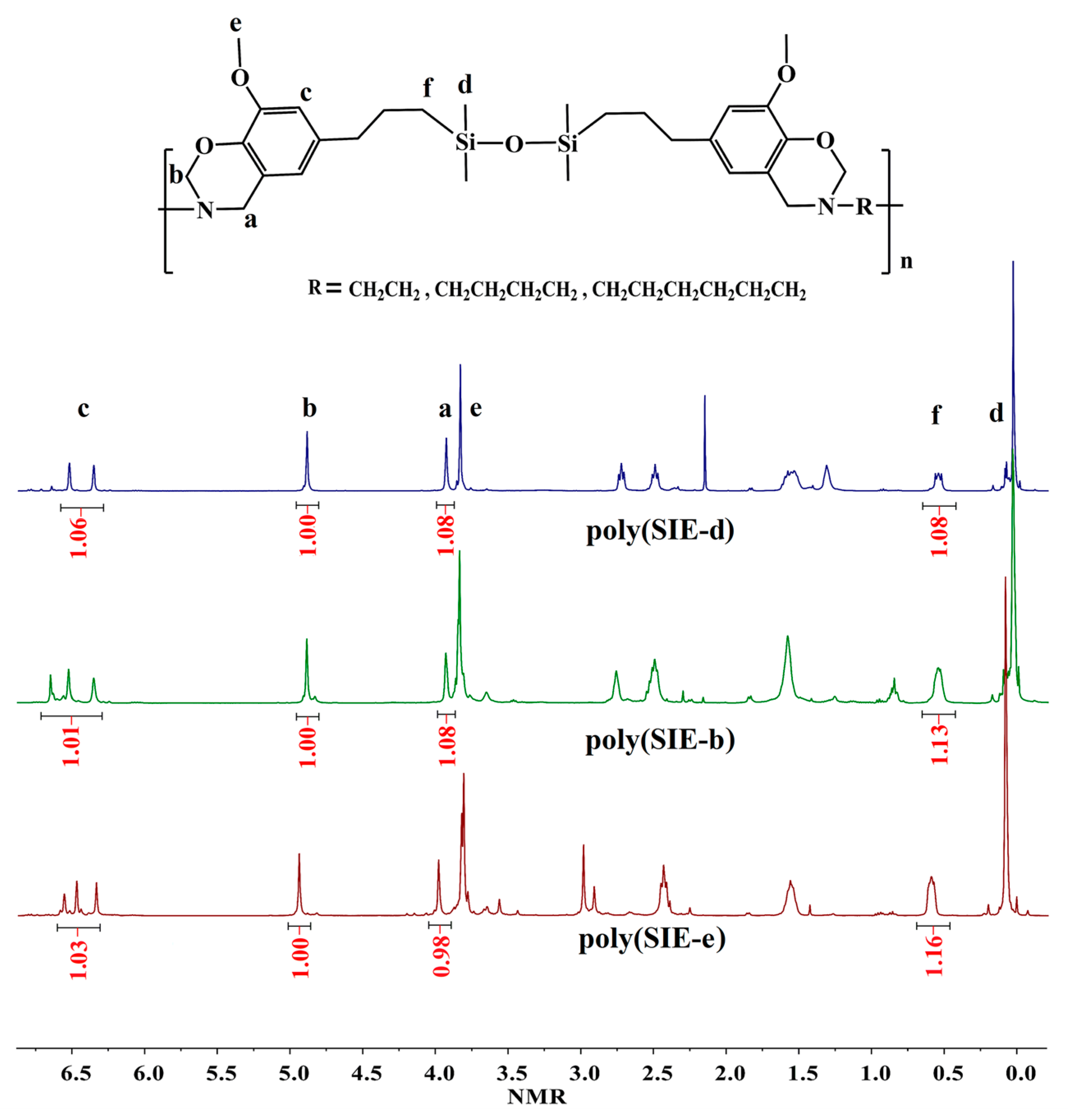
3.1. Characterization of Different Benzoxazine Oligomers
3.2. Curing Behavior of Poly (SIE-e), Poly (SIE-b), and Poly (SIE-d)
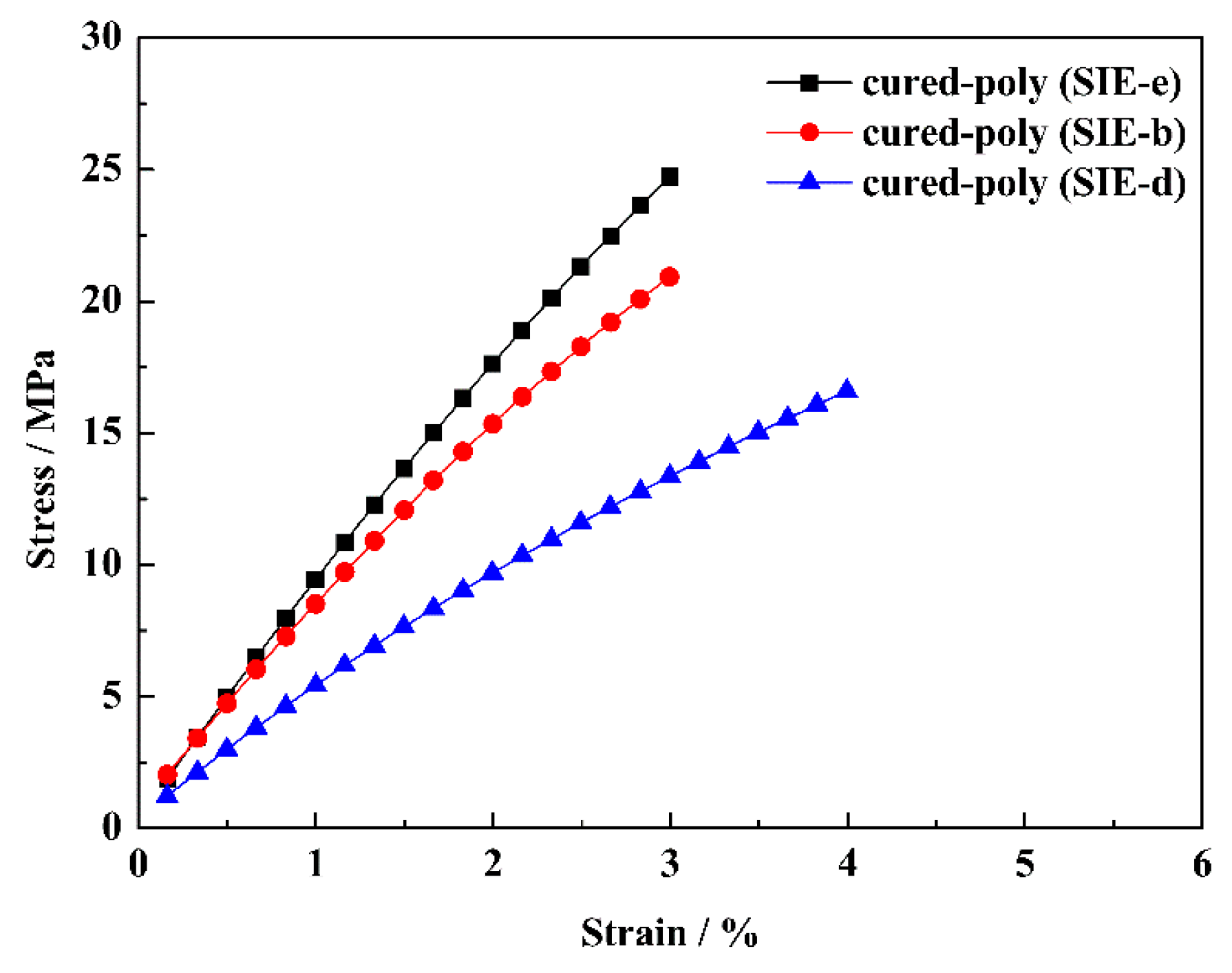
3.3. Mechanical Properties of the Cured Oligomers
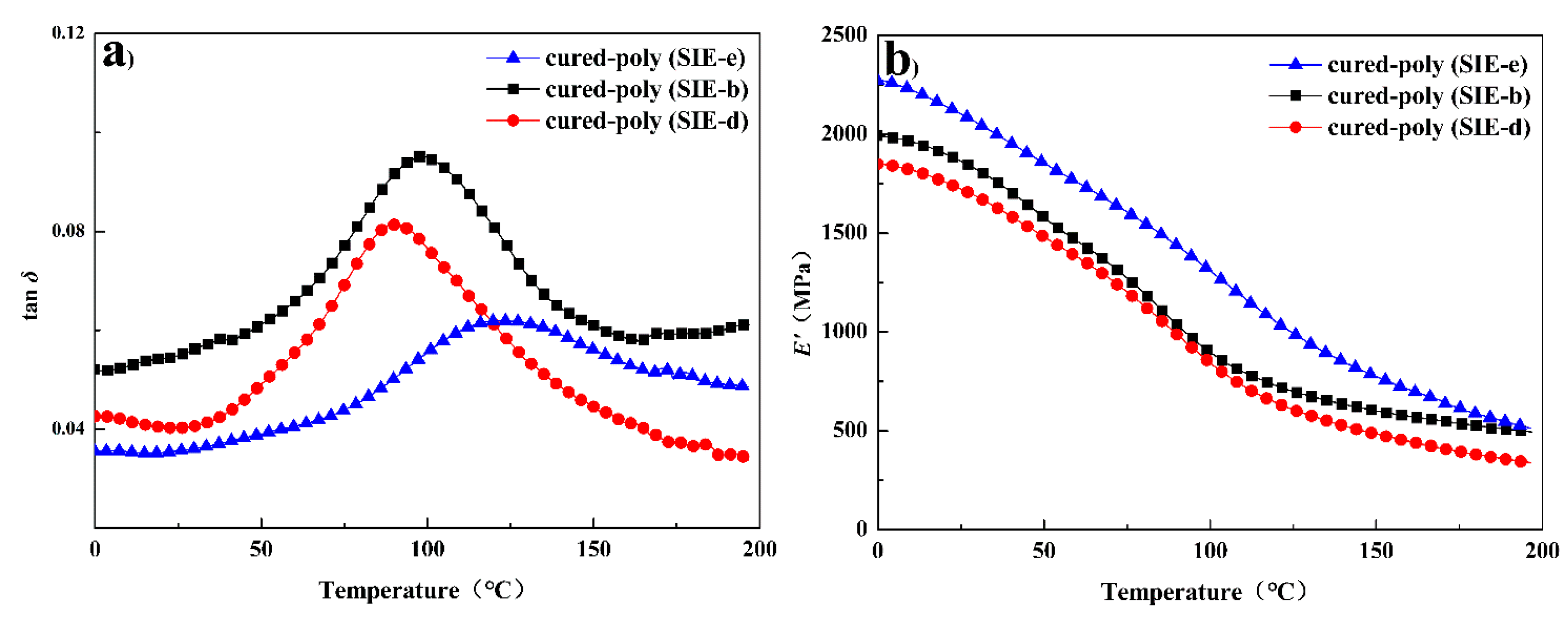
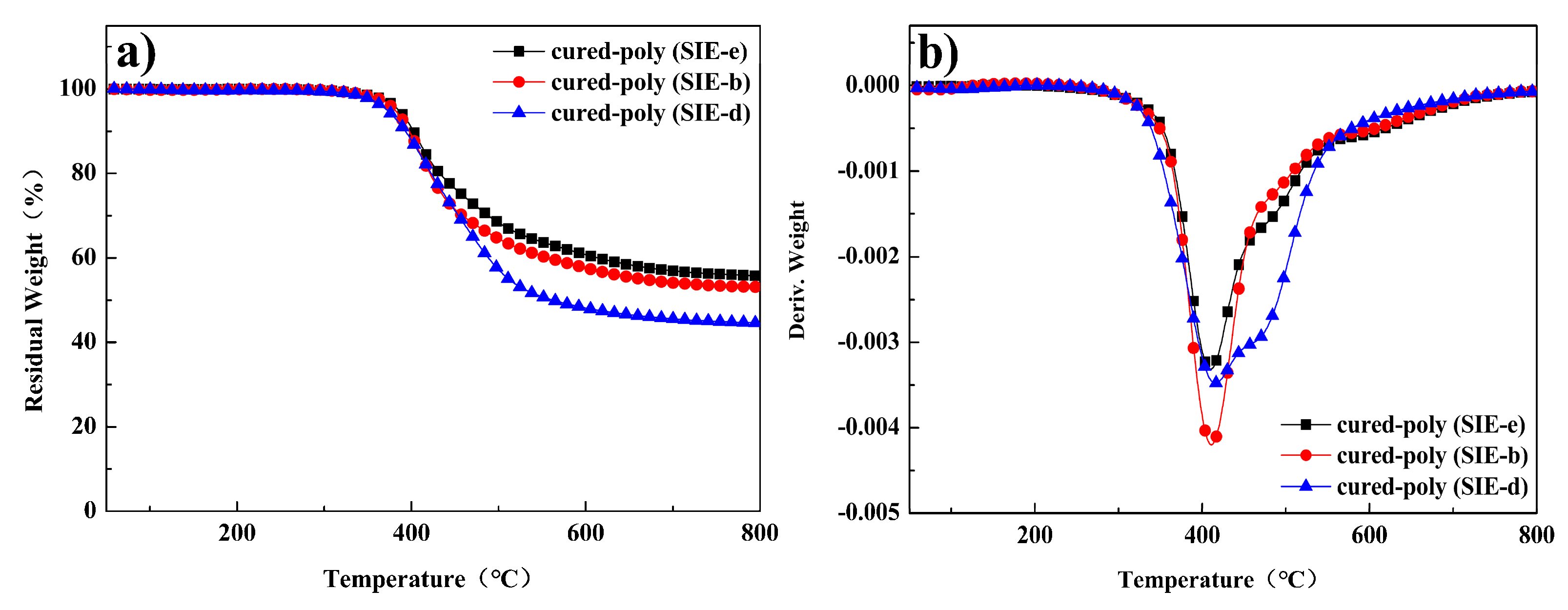
3.4. Dynamic Mechanical Properties and Thermal Stability of Cured Benzoxazine Oligomers
3.5. Coating Properties of the Cured Oligomers
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ishida, H.; Allen, D.J. Physical and mechanical characterization of near-zero shrinkage polybenzoxazines. J. Polym. Sci. Part B Polym. Phys. 1996, 34, 1019–1030. [Google Scholar] [CrossRef]
- Ishida, H.; Sanders, D.P. Regioselectivity and network structure of difunctional alkyl-substituted aromatic amine-based polybenzoxazines. Macromolecules 2000, 33, 8149–8157. [Google Scholar] [CrossRef]
- Nair, C.R. Advances in addition-cure phenolic resins. Prog. Polym. Sci. 2004, 29, 401–498. [Google Scholar] [CrossRef]
- Low, H.Y.; Ishida, H. Improved thermal stability of polybenzoxazines by transition metals. Polym. Degrad. Stabil. 2006, 91, 805–815. [Google Scholar] [CrossRef]
- Chou, C.I.; Liu, Y.L. High performance thermosets from a curable Diels–Alder polymer possessing benzoxazine groups in the main chain. J. Polym. Sci. Part A Polym. Chem. 2008, 46, 6509–6517. [Google Scholar] [CrossRef]
- Aydogan, C.; Kiskan, B.; Hacioglu, S.O.; Toppare, L.; Yagci, Y. Electrochemical manipulation of adhesion strength of polybenzoxazines on metal surfaces: From strong adhesion to dismantling. RSC Adv. 2014, 4, 27545–27551. [Google Scholar] [CrossRef]
- Zhu, Y.Q.; Romain, C.; Williams, C.K. Sustainable polymers from renewable resources. Nature 2016, 540, 354–362. [Google Scholar] [CrossRef] [PubMed]
- Worthington, M.J.H.; Kucera, R.L.; Chalker, J.M. Green chemistry and polymers made from sulfur. Green Chem. 2017, 19, 2748–2761. [Google Scholar] [CrossRef]
- Rahman, M.A.; Lokupitiya, H.N.; Ganewatta, M.S.; Yuan, L.; Stefik, M.; Tang, C.B. Designing block copolymer architectures toward tough bioplastics from natural rosin. Macromolecules 2017, 50, 2069–2077. [Google Scholar] [CrossRef]
- Dumas, L.; Bonnaud, L.; Olivier, M.; Poorteman, M.; Dubois, P. Arbutin-based benzoxazine: En route to an intrinsic water soluble biobased resin. Green Chem. 2016, 18, 4954–4960. [Google Scholar] [CrossRef]
- Minigher, A.; Benedetti, E.; De Giacomo, O.; Campaner, P.; Aroulmoji, V. Synthesis and characterization of novel cardanol based benzoxazines. Nat. Prod. Comm. 2009, 4, 521–528. [Google Scholar]
- Rao, B.S.; Palanisamy, A. Monofunctional benzoxazine from cardanol for bio-composite applications. React. Funct. Polym. 2011, 71, 148–154. [Google Scholar] [CrossRef]
- Zúñiga, C.; Larrechi, M.S.; Lligadas, G.; Ronda, J.C.; Galià, M.; Cádiz, V. Polybenzoxazines from renewable diphenolic acid. J. Polym. Sci. Part A Polym. Chem. 2011, 49, 1219–1227. [Google Scholar] [CrossRef]
- Zúñiga, C.; Lligadas, G.; Ronda, J.C.; Galià, M.; Cádiz, V. Renewable polybenzoxazines based in diphenolic acid. Polymer 2012, 53, 1617–1623. [Google Scholar] [CrossRef]
- Zúñiga, C.; Lligadas, G.; Ronda, J.C.; Galià, M.; Cádiz, V. Self-foaming diphenolic acid benzoxazine. Polymer 2012, 53, 3089–3095. [Google Scholar] [CrossRef]
- Wang, C.F.; Sun, J.Q.; Liu, X.D.; Sudo, A.; Endo, T. Synthesis and copolymerization of fully bio-based benzoxazines from guaiacol, furfurylamine and stearylamine. Green Chem. 2012, 14, 2799–2806. [Google Scholar] [CrossRef]
- Van, A.; Chiou, K.; Ishida, H. Use of renewable resource vanillin for the preparation of benzoxazine resin and reactive monomeric surfactant containing oxazine ring. Polymer 2014, 55, 1443–1451. [Google Scholar] [CrossRef]
- Sini, N.K.; Bijwe, J.; Varma, I.K. Renewable benzoxazine monomer from Vanillin: Synthesis, characterization, and studies on curing behavior. J. Polym. Sci. Part A Polym. Chem. 2014, 52, 7–11. [Google Scholar] [CrossRef]
- Liu, Y.L.; Chou, C.-I. High performance benzoxazine monomers and polymers containing furan groups. J. Polym. Sci. Part A Polym. Chem. 2005, 43, 5267–5282. [Google Scholar] [CrossRef]
- Dumas, L.; Bonnaud, L.; Olivier, M.; Poorteman, M.; Dubois, P. Eugenol-based benzoxazine: From straight synthesis to taming of the network properties. J. Mater. Chem. A 2015, 3, 6012–6018. [Google Scholar] [CrossRef]
- Periyasamy, T.; Asrafali, S.P.; Muthusamy, S. New benzoxazines containing polyhedral oligomeric silsesquioxane from eugenol, guaiacol and vanillin. New J. Chem. 2015, 39, 1691–1702. [Google Scholar] [CrossRef]
- Ručigaj, A.; Gradišar, Š.; Krajnc, M. Kinetic investigation of a complex curing of the guaiacol bio-based benzoxazine system. e-Polymers 2016, 16, 199–206. [Google Scholar] [CrossRef]
- Lu, Y.B.; Li, M.M.; Zhang, Y.J.; Hu, D.; Ke, L.L.; Xu, W.J. Synthesis and curing kinetics of benzoxazine containing fluorene and furan groups. Thermochim. Acta 2011, 515, 32–37. [Google Scholar] [CrossRef]
- Wang, H.; Wang, J.; He, X.Y.; Feng, T.T.; Ramdani, N.; Luan, M.J.; Liu, W.B.; Xu, X.D. Synthesis of novel furan-containing tetrafunctional fluorene-based benzoxazine monomer and its high performance thermoset. RSC Adv. 2014, 4, 64798–64801. [Google Scholar] [CrossRef]
- Dumas, L.; Bonnaud, L.; Olivier, M.; Poorteman, M.; Dubois, P. High performance bio-based benzoxazine networks from resorcinol and hydroquinone. Eur. Polym. J. 2016, 75, 486–494. [Google Scholar] [CrossRef]
- Kiskan, B.; Yagci, Y.; Ishida, H. Synthesis, characterization, and properties of new thermally curable polyetheresters containing benzoxazine moieties in the main chain. J. Polym. Sci. Part A Polym. Chem. 2008, 46, 414–420. [Google Scholar] [CrossRef]
- Philp, J. OECD policies for bioplastics in the context of a bioeconomy, 2013. Ind. Biotechnol. 2014, 10, 19–21. [Google Scholar] [CrossRef]
- Telysheva, G.; Dobele, G.; Meier, D.; Dizhbite, T.; Rossinska, G.; Jurkjane, V. Characterization of the transformations of lignocellulosic structures upon degradation in planted soil. J. Anal. Appl. Pyrol. 2007, 79, 52–60. [Google Scholar] [CrossRef]
- Kuroda, K.-I.; Inoue, Y.; Sakai, K. Analysis of lignin by pyrolysis-gas chromatography. I. Effect of inorganic substances on guaiacol-derivative yield from softwoods and their lignins. J. Anal. Appl. Pyrol. 1990, 18, 59–69. [Google Scholar] [CrossRef]
- Sun, L.; Singh, S.; Joo, M.; Vega-Sanchez, M.; Ronald, P.; Simmons, B.A.; Adams, P.; Auer, M. Non-invasive imaging of cellulose microfibril orientation within plant cell walls by polarized Raman microspectroscopy. Biotechnol. Bioeng. 2016, 113, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-X.; Ishida, H. Synthesis and properties of new thermoplastic polymers from substituted 3,4-dihydro-2H-1,3-benzoxazines. Macromolecules 2000, 33, 2839–2847. [Google Scholar] [CrossRef]
- Dumas, L.; Bonnaud, L.; Olivier, M.; Poorteman, M.; Dubois, P. Bio-based high performance thermosets: Stabilization and reinforcement of eugenol-based benzoxazine networks with BMI and CNT. Eur. Polym. J. 2015, 67, 494–502. [Google Scholar] [CrossRef]
- Wang, Y.X.; Ishida, H. Development of low-viscosity benzoxazine resins and their polymers. J. Appl. Polym. Sci. 2002, 86, 2953–2966. [Google Scholar] [CrossRef]
- Jang, J.; Yang, H. Toughness improvement of carbon-fibre/polybenzoxazine composites by rubber modification. Compos. Sci. Technol. 2000, 60, 457–463. [Google Scholar] [CrossRef]
- Cui, Y.J.; Chen, Y.; Wang, X.L.; Tian, G.H.; Tang, X.Z. Synthesis and characterization of polyurethane/polybenzoxazine-based interpenetrating polymer networks (IPNs). Polym. Int. 2003, 52, 1246–1248. [Google Scholar] [CrossRef]
- Yeganeh, H.; Razavi-Nouri, M.; Ghaffari, M. Investigation of thermal, mechanical, and electrical properties of novel polyurethanes/high molecular weight polybenzoxazine blends. Polym. Advan. Technol. 2008, 19, 1024–1032. [Google Scholar] [CrossRef]
- Ardhyananta, H.; Kawauchi, T.; Takeichi, T.; Ismail, H. Preparation and properties of polybenzoxazine-poly(dimethylsiloxane-co-diphenylsiloxane) hybrids as high performance polymers. High Perform. Polym. 2010, 22, 609–632. [Google Scholar] [CrossRef]
- Ardhyananta, H.; Wahid, M.H.; Sasaki, M.; Agag, T.; Kawauchi, T.; Ismail, H.; Takeichi, T. Performance enhancement of polybenzoxazine by hybridization with polysiloxane. Polymer 2008, 49, 4585–4591. [Google Scholar] [CrossRef]
- Thirukumaran, P.; Parveen, A.S.; Sarojadevi, M. Synthesis of eugenol-based polybenzoxazine–POSS nanocomposites for low dielectric applications. Polym. Compos. 2015, 36, 1973–1982. [Google Scholar] [CrossRef]
- Alhwaige, A.A.; Agag, T.; Ishida, H. Biobased chitosan/polybenzoxazine cross-linked films: Preparation in aqueous media and synergistic improvements in thermal and mechanical properties. Biomacromolecules 2013, 14, 1806–1815. [Google Scholar] [CrossRef] [PubMed]
- Agag, T.; Geiger, S.; Alhassan, S.M.; Qutubuddin, S.; Ishida, H. Low-viscosity polyether-based main-chain benzoxazine polymers: Precursors for flexible thermosetting polymers. Macromolecules 2010, 43, 7122–7127. [Google Scholar] [CrossRef]
- Lin, C.H.; Chang, S.L.; Shen, T.Y.; Shih, Y.S.; Lin, H.T.; Wang, C.F. Flexible polybenzoxazine thermosets with high glass transition temperatures and low surface free energies. Polym. Chem. 2012, 3, 935–945. [Google Scholar] [CrossRef]
- Liu, J.; Agag, T.; Ishida, H. Handbook of Benzoxazine Resins; Elsevier: Oxford, UK, 2011. [Google Scholar]
- Chernykh, A.; Liu, J.P.; Ishida, H. Synthesis and properties of a new crosslinkable polymer containing benzoxazine moiety in the main chain. Polymer 2006, 47, 7664–7669. [Google Scholar] [CrossRef]
- Takeichi, T.; Kano, T.; Agag, T. Synthesis and thermal cure of high molecular weight polybenzoxazine precursors and the properties of the thermosets. Polymer 2005, 46, 12172–12180. [Google Scholar] [CrossRef]
- ASTM D3363-00 Standard Test Method for Film Hardness by Pencil Test; ASTM International: West Conshohocken, PA, USA, 2000.
- ASTM D4145-10 Standard Test Method for Coating Flexibility of Prepainted Sheet; ASTM International: West Conshohocken, PA, USA, 2010.
- ASTM D3359-02 Standard Test Methods for Measuring Adhesion by Tape Test; ASTM International: West Conshohocken, PA, USA, 2002.
- ASTM D5402-06 Standard Practice for Assessing the Solvent Resistance of Organic Coatings Using Solvent Rubs; ASTM International: West Conshohocken, PA, USA, 2006.
- Liu, J.; Agag, T.; Ishida, H. Main-chain benzoxazine oligomers: A new approach for resin transfer moldable neat benzoxazines for high performance applications. Polymer 2010, 51, 5688–5694. [Google Scholar] [CrossRef]
- Li, C.; Wan, J.T.; Kalali, E.N.; Fan, H.; Wang, D.-Y. Synthesis and characterization of functional eugenol derivative based layered double hydroxide and its use as a nanoflame-retardant in epoxy resin. J. Mater. Chem. A 2015, 3, 3471–3479. [Google Scholar] [CrossRef]
- Pan, X.; Webster, D.C. New biobased high functionality polyols and their use in polyurethane coatings. ChemSusChem 2012, 5, 419–429. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.Q.; Xin, W.B.; Zhang, J.W. Rosin-based acid anhydrides as alternatives to petrochemical curing agents. Green Chem. 2009, 11, 1018–1025. [Google Scholar] [CrossRef]
- Shen, X.B.; Dai, J.Y.; Liu, Y.; Liu, X.Q.; Zhu, J. Synthesis of high performance polybenzoxazine networks from bio-based furfurylamine: Furan vs benzene ring. Polymer 2017, 122, 258–269. [Google Scholar] [CrossRef]
- Rao, B.S.; Palanisamy, A. Synthesis of bio based low temperature curable liquid epoxy, benzoxazine monomer system from cardanol: Thermal and viscoelastic properties. Eur. Polym. J. 2013, 49, 2365–2376. [Google Scholar] [CrossRef]
- Thirukumaran, P.; Shakila Parveen, A.; Kumudha, K.; Sarojadevi, M. Synthesis and characterization of new polybenzoxazines from renewable resources for bio-composite applications. Polym. Compos. 2016, 37, 1821–1829. [Google Scholar] [CrossRef]
- Zhang, L.; Yang, Y.; Chen, Y.X.; Lu, H.W. Cardanol-capped main-chain benzoxazine oligomers for resin transfer molding. Eur. Polym. J. 2017, 93, 284–293. [Google Scholar] [CrossRef]
- Tüzün, A.; Lligadas, G.; Ronda, J.C.; Galià, M.; Cádiz, V. Integrating plant oils into thermally curable main-chain benzoxazine polymers via ADMET polymerization. Eur. Polym. J. 2015, 67, 503–512. [Google Scholar] [CrossRef]
- Van Krevelen, D.W.; Te Nijenhuis, K. Typology of polymer. In Properties of Polymers: Their Correlation with Chemical Structure; Their Numerical Estimation and Prediction from Additive Group Contributions, 4th ed.; Elsevier: Amsterdam, The Netherlands, 2009. [Google Scholar]
- Aydogan, B.; Sureka, D.; Kiskan, B.; Yagci, Y. Polysiloxane-containing benzoxazine moieties in the main chain. J. Polym. Sci. Part A: Polym. Chem 2010, 48, 5156–5162. [Google Scholar] [CrossRef]
- Liu, R.; Zhang, X.P.; Zhu, J.J.; Liu, X.Y.; Wang, Z.; Yan, J.L. UV-curable coatings from multiarmed cardanol-based acrylate oligomers. ACS Sustain. Chem. Eng. 2015, 3, 1313–1320. [Google Scholar] [CrossRef]
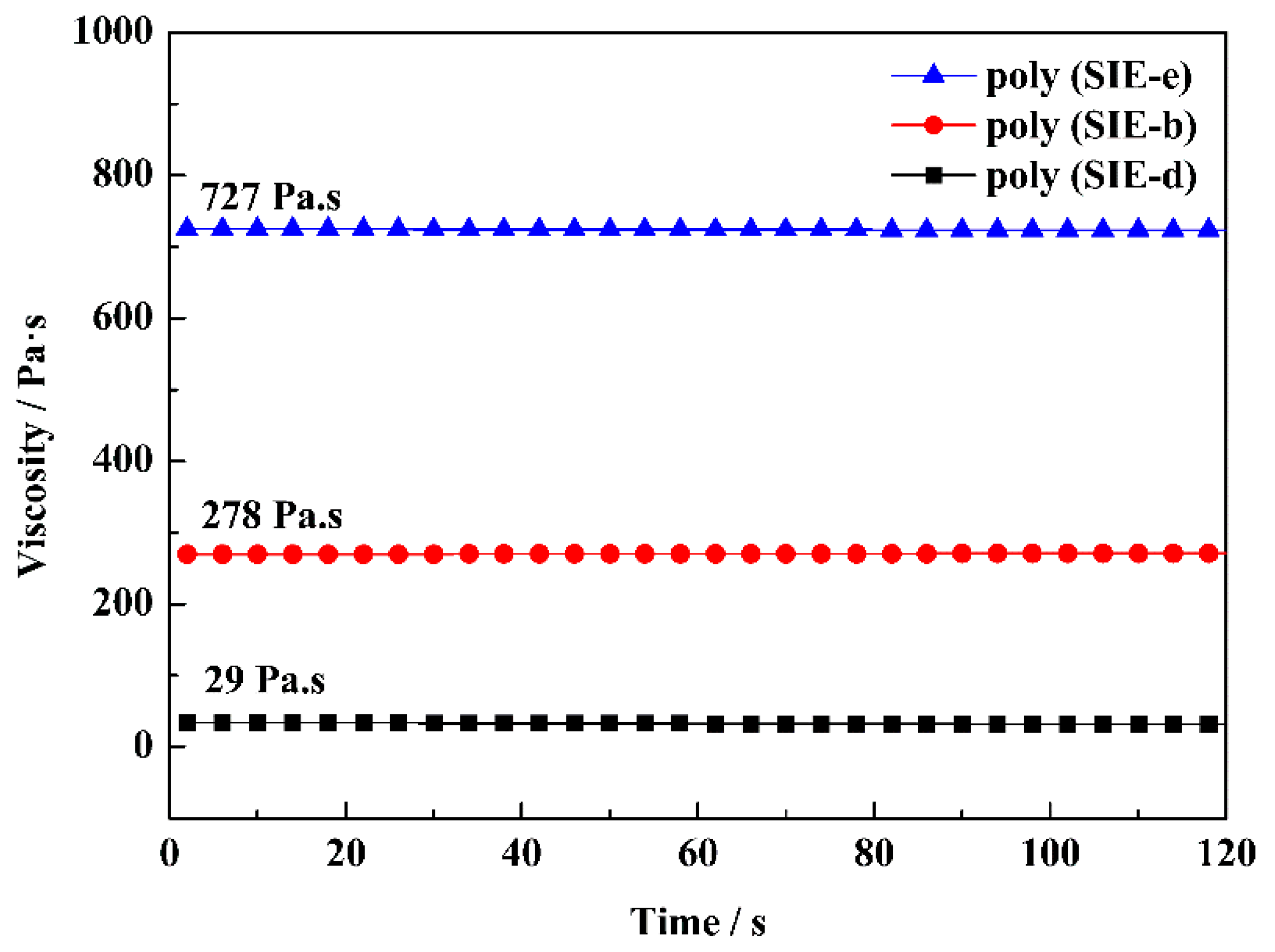
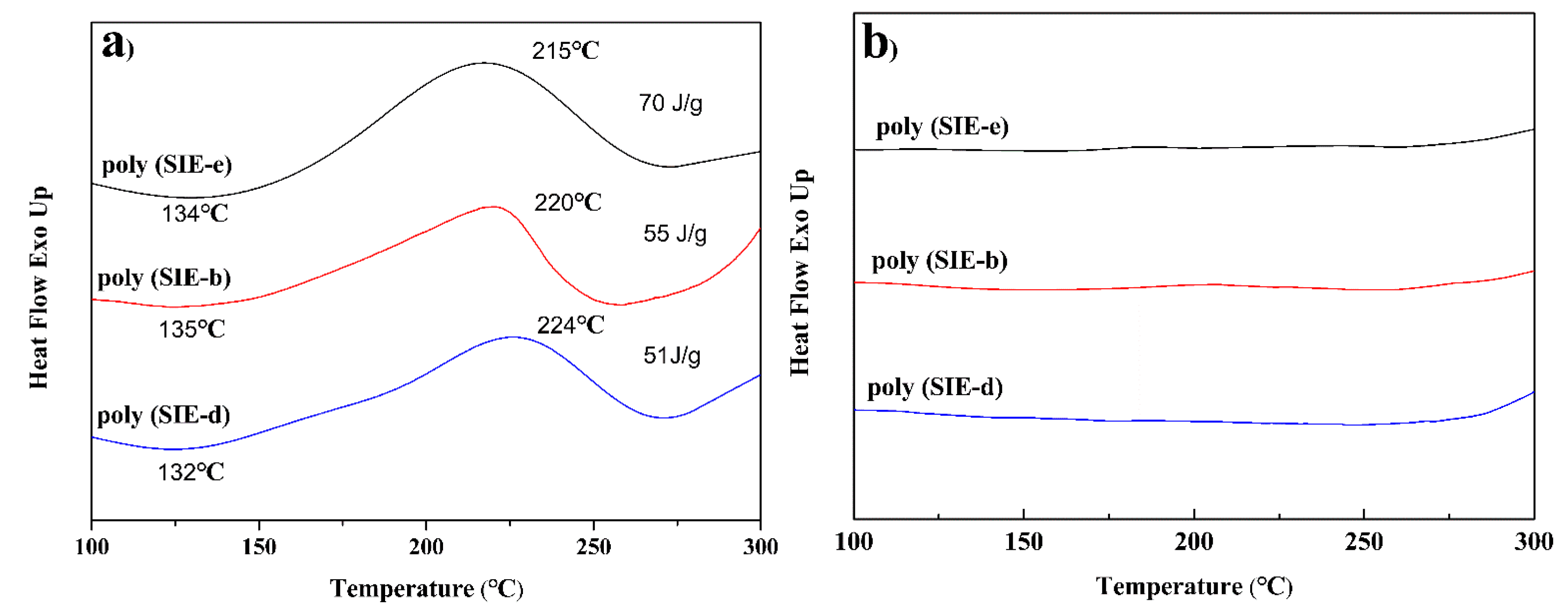
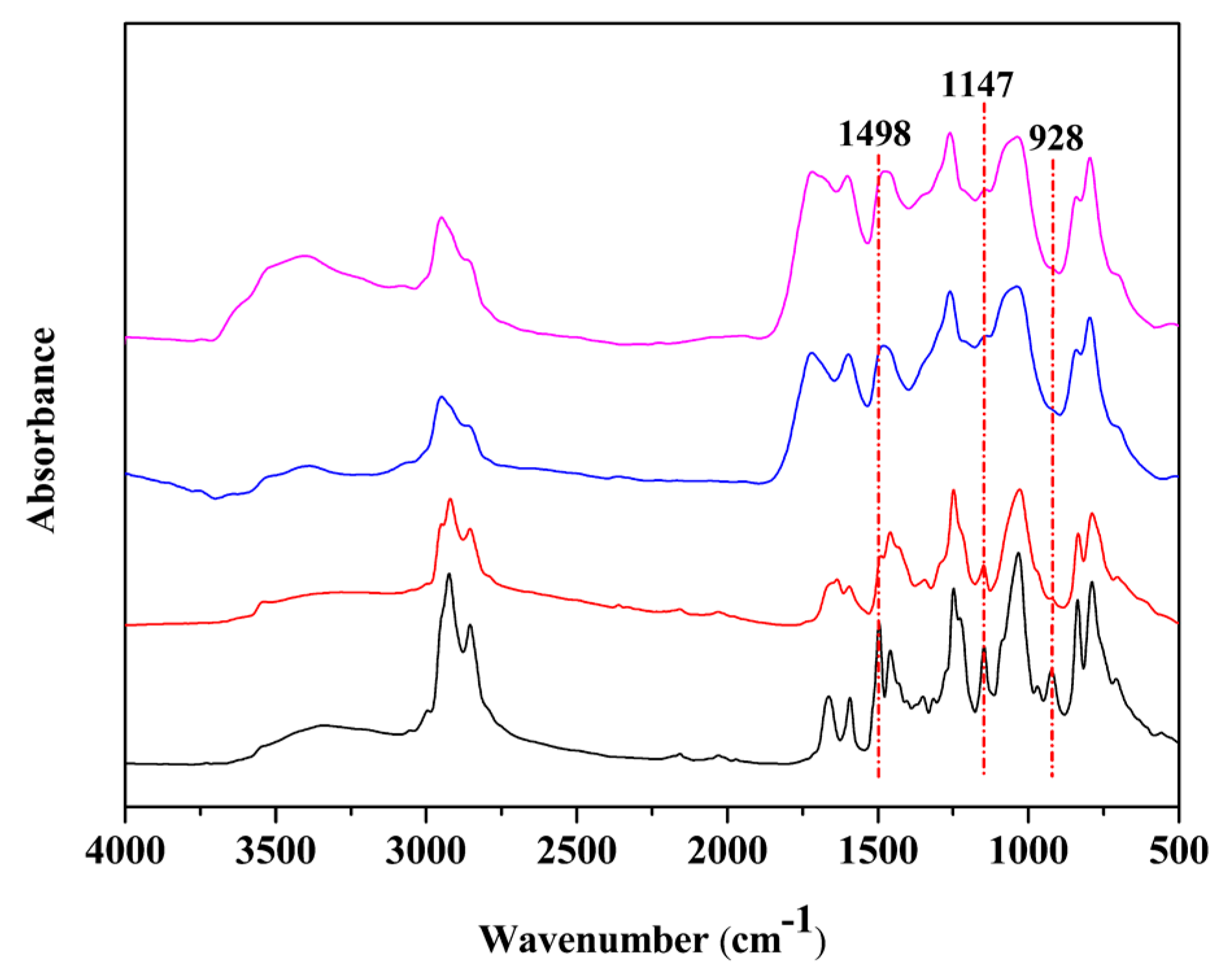
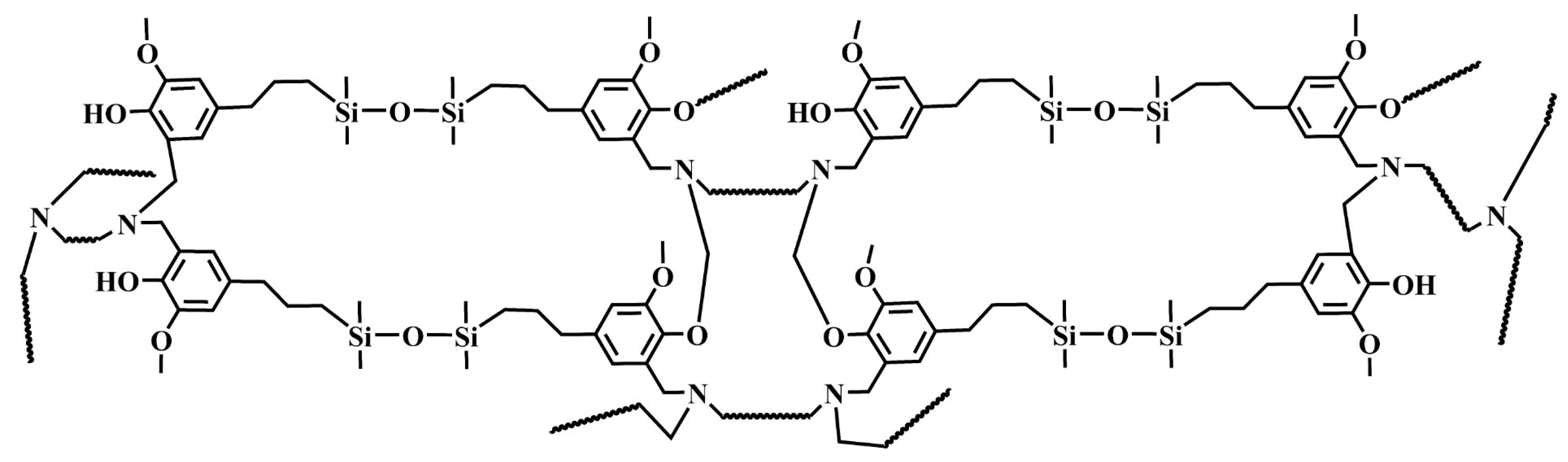


| Sample | Diamine | Mn | Mw/Mn | Viscosity at 30 °C (Pa·s) | Bio-Based Content (%) | Yield |
|---|---|---|---|---|---|---|
| poly (SIE-e) | 1,2-ethylenediamine | 960 | 1.9 | 730 | 60 | 56% |
| poly (SIE-b) | 1,4-butanediamine | 1000 | 1.5 | 280 | 57 | 59% |
| poly (SIE-d) | 1,6-hexanediamine | 1260 | 1.7 | 30 | 55 | 71% |
| Cured-Samples | Tg a (°C) | E’ b (MPa) | νe c/103 (mol·m−3) | σ d (MPa) | ε e (%) | Td10% (°C) | Tdmax f (°C) | R800 (%) |
|---|---|---|---|---|---|---|---|---|
| cured-poly (SIE-e) | 123 | 2102 | 47.0 | 24.7 ± 1.5 | 2.9 ± 0.2 | 402 | 409 | 56 |
| cured-poly (SIE-b) | 96 | 1863 | 43.9 | 20.9 ± 0.9 | 3.0 ± 0.1 | 397 | 411 | 53 |
| cured-poly (SIE-d) | 89 | 1723 | 30.6 | 16.6 ± 1.1 | 3.9 ± 0.2 | 392 | 415 | 45 |
| Samples | Thickness (um) | Pencil Hardness | Flexibility | Adhesion | Water Sbsorption | MEK Resistance | Ethanol Resistance |
|---|---|---|---|---|---|---|---|
| cured-poly (SIE-e) | 50 | 3H | 1T | 5B | 1.2% | 400 | 400 |
| cured-poly (SIE-b) | 50 | 3H | 1T | 5B | 1.1% | 400 | 400 |
| cured-poly (SIE-d) | 50 | 3H | 1T | 5B | 0.7% | 400 | 400 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dai, J.; Yang, S.; Teng, N.; Liu, Y.; Liu, X.; Zhu, J.; Zhao, J. Synthesis of Eugenol-Based Silicon-Containing Benzoxazines and Their Applications as Bio-Based Organic Coatings. Coatings 2018, 8, 88. https://doi.org/10.3390/coatings8030088
Dai J, Yang S, Teng N, Liu Y, Liu X, Zhu J, Zhao J. Synthesis of Eugenol-Based Silicon-Containing Benzoxazines and Their Applications as Bio-Based Organic Coatings. Coatings. 2018; 8(3):88. https://doi.org/10.3390/coatings8030088
Chicago/Turabian StyleDai, Jinyue, Shimin Yang, Na Teng, Yuan Liu, Xiaoqing Liu, Jin Zhu, and Jun Zhao. 2018. "Synthesis of Eugenol-Based Silicon-Containing Benzoxazines and Their Applications as Bio-Based Organic Coatings" Coatings 8, no. 3: 88. https://doi.org/10.3390/coatings8030088
APA StyleDai, J., Yang, S., Teng, N., Liu, Y., Liu, X., Zhu, J., & Zhao, J. (2018). Synthesis of Eugenol-Based Silicon-Containing Benzoxazines and Their Applications as Bio-Based Organic Coatings. Coatings, 8(3), 88. https://doi.org/10.3390/coatings8030088




