Simple Coatings to Render Polystyrene Protein Resistant
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Sample Preparation
2.3. Surface Modification with Polydopamine
2.4. Secondary Polymer Layer Deposition on Polydopamine
2.5. QCM-D Experiments
2.6. X-ray Photoelectron Spectroscopy (XPS)
2.7. Atomic Force Microscopy (AFM)
2.8. Contact Angle Goniometry
2.9. Statistics
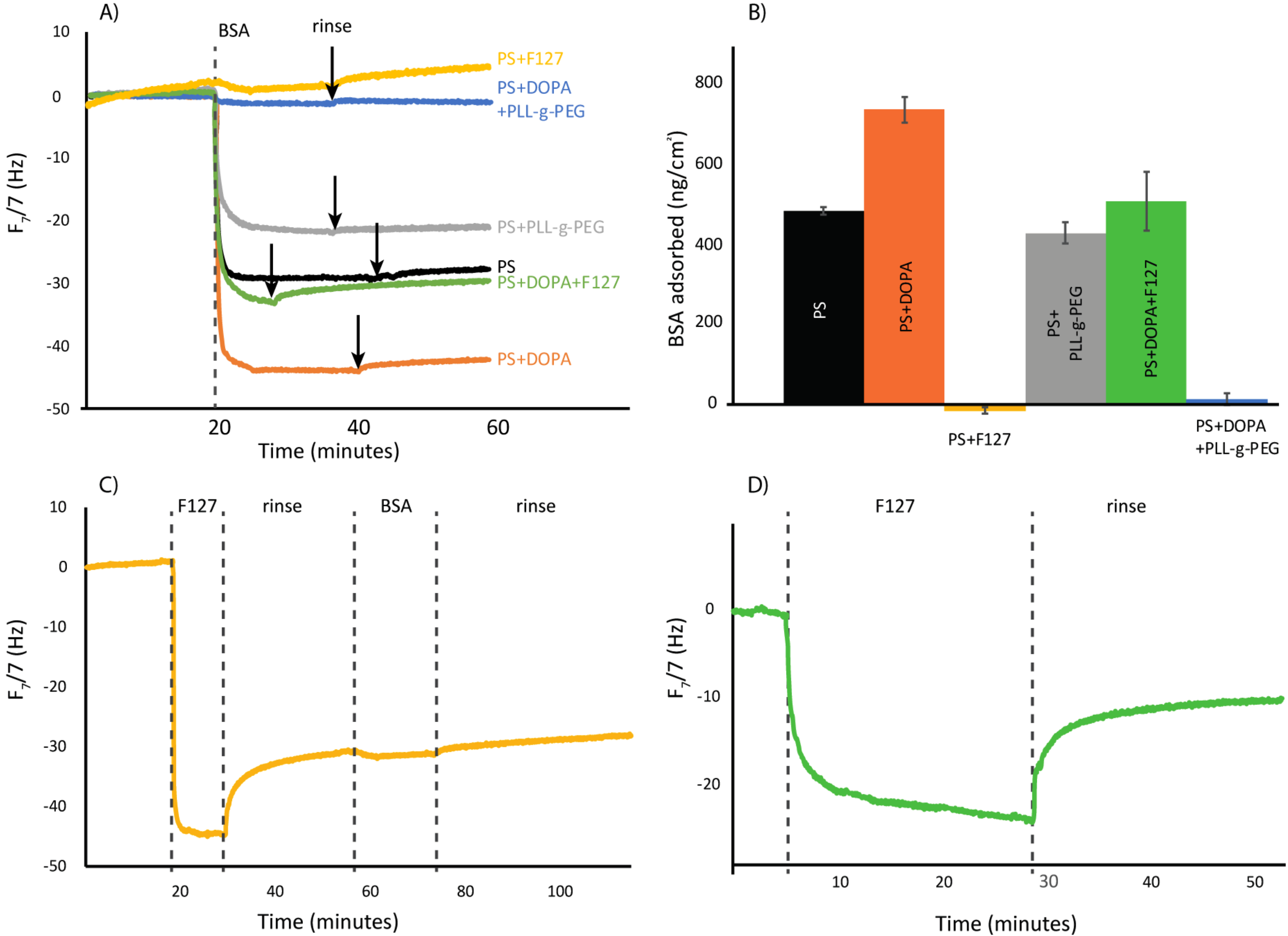
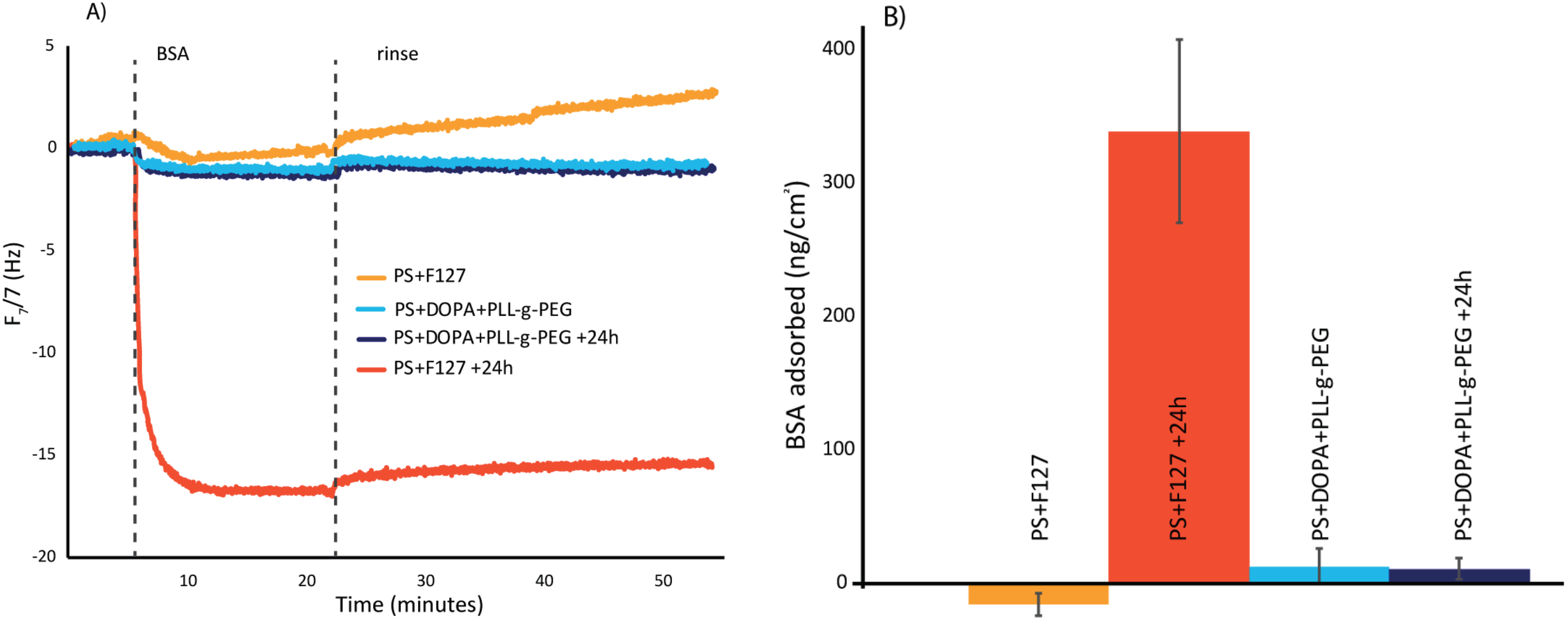
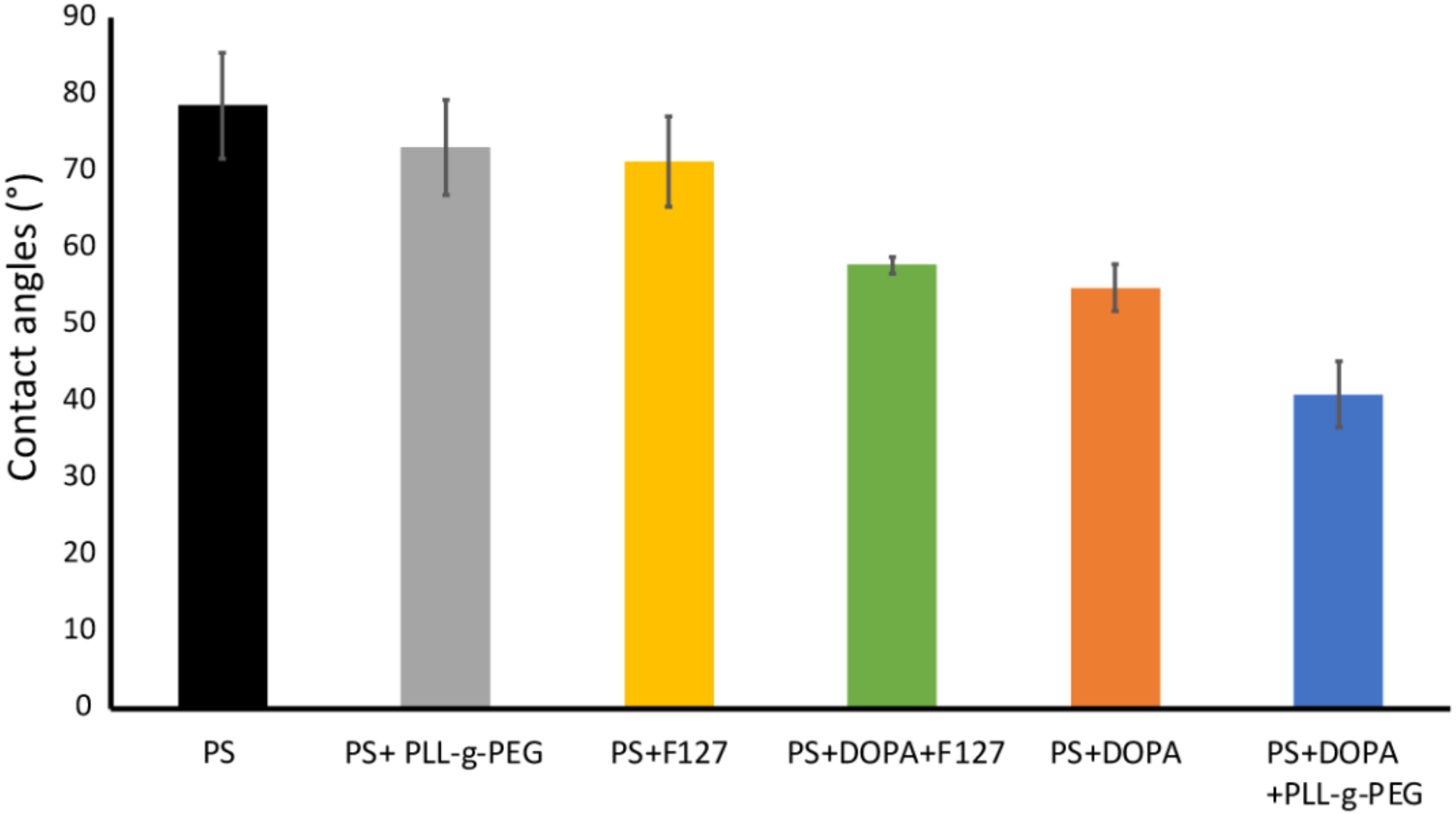
3. Results and Discussion
3.1. QCM-D Analysis
3.2. Sample Characterisation
4. Conclusions
- F127 significantly reduces BSA adsorption in short-term studies. However, after soaking only 24 h in buffer, the resistance is significantly diminished.
- The polydopamine intermediary film produced, cannot provide the necessary attraction to bind F127 to the surface. Instead, bonds between the Pluronic and the surface are weakened, causing more protein adsorption than on unmodified polystyrene.
- By adsorbing the same “assisting” polydopamine film and subsequently grafting PLL-g-PEG, we can successfully prevent 97% of protein adsorption on a polystyrene substrate.
- This modified surface is equally as protein-rejecting after 24 h in buffer, and thus a promising simple coating for long term protein rejection of polystyrene.
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hatakeyama, E.S.; Ju, H.; Gabriel, C.J.; Lohr, J.L.; Bara, J.E.; Noble, R.D.; Freeman, B.D.; Gin, D.L. New protein-resistant coatings for water filtration membranes based on quaternary ammonium and phosphonium polymers. J. Membr. Sci. 2009, 330, 104–116. [Google Scholar] [CrossRef]
- Lowe, S.; O’Brien-Simpson, N.M.; Connal, L.A. Antibiofouling polymer interfaces: Poly(ethylene glycol) and other promising candidates. Polym. Chem. 2015, 6, 198–212. [Google Scholar] [CrossRef]
- Poncin-Epaillard, F.; Vrlinic, T.; Debarnot, D.; Mozetic, M.; Coudreuse, A.; Legeay, G.; El Moualij, B.; Zorzi, W. Surface treatment of polymeric materials controlling the adhesion of biomolecules. J. Funct. Biomater. 2012, 3, 528–543. [Google Scholar] [CrossRef] [PubMed]
- Malmsten, M. Ellipsometry studies of protein layers adsorbed at hydrophobic surfaces. J. Colloid Interface Sci. 1994, 166, 333–342. [Google Scholar] [CrossRef]
- Roach, P.; Farrar, D.; Perry, C.C. Interpretation of protein adsorption: Surface-induced conformational changes. J. Am. Chem. Soc. 2005, 127, 8168–8173. [Google Scholar] [CrossRef] [PubMed]
- Wertz, C.F.; Santore, M.M. Effect of surface hydrophobicity on adsorption and relaxation kinetics of albumin and fibrinogen: Single-species and competitive behavior. Langmuir 2001, 17, 3006–3016. [Google Scholar] [CrossRef]
- Chou, Y.N.; Wen, T.C.; Chang, Y. Zwitterionic surface grafting of epoxylated sulfobetaine copolymers for the development of stealth biomaterial interfaces. Acta Biomater. 2016, 40, 78–91. [Google Scholar] [CrossRef] [PubMed]
- Sundaram, H.S.; Han, X.; Nowinski, A.K.; Ella-Menye, J.R.; Wimbish, C.; Marek, P.; Senecal, K.; Jiang, S. One-step dip coating of zwitterionic sulfobetaine polymers on hydrophobic and hydrophilic surfaces. ACS Appl. Mater. Interfaces 2014, 6, 6664–6671. [Google Scholar] [CrossRef] [PubMed]
- Raynor, J.E.; Capadona, J.R.; Collard, D.M.; Petrie, T.A.; García, A.J. Polymer brushes and self-assembled monolayers: Versatile platforms to control cell adhesion to biomaterials Review. Biointerphases 2009, 4, FA3–FA16. [Google Scholar] [CrossRef] [PubMed]
- Brittain, W.J.; Minko, S. A structural definition of polymer brushes. J. Polym. Sci. Part A Polym. Chem. 2007, 45, 3505–3512. [Google Scholar] [CrossRef]
- Bridgett, M.J.; Davies, M.C.; Denyer, S.P. Control of staphylococcal adhesion to polystyrene surfaces by polymer surface modification with surfactants. Biomaterials 1992, 13, 411–416. [Google Scholar] [CrossRef]
- Green, R.J.; Tasker, S.; Davies, J.; Davies, M.C.; Roberts, C.J.; Tendler, S.J.B. Adsorption of PEO-PPO-PEO triblock copolymers at the solid/liquid interface: A surface plasmon resonance study. Langmuir 1997, 13, 6510–6515. [Google Scholar] [CrossRef]
- Song, X.; Zhao, S.; Fang, S.; Ma, Y.; Duan, M. Mesoscopic simulations of adsorption and association of PEO-PPO-PEO triblock copolymers on a hydrophobic surface: From mushroom hemisphere to rectangle brush. Langmuir 2016, 32, 11375–11385. [Google Scholar] [CrossRef] [PubMed]
- Marsh, L.H.; Coke, M.; Dettmar, P.W.; Ewen, R.J.; Havler, M.; Nevell, T.G.; Smart, J.D.; Smith, J.R.; Timmins, B.; Tsibouklis, J.; et al. Adsorbed poly(ethyleneoxide)-poly(propyleneoxide) copolymers on synthetic surfaces: Spectroscopy and microscopy of polymer structures and effects on adhesion of skin-borne bacteria. J. Biomed. Mater. Res. 2002, 61, 641–652. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Song, W.; Huang, H.; Chen, H. Protein adsorption and cell adhesion on polyurethane/Pluronic® surface with lotus leaf-like topography. Colloids Surf. B Biointerfaces 2010, 77, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.; Wang, L.; Yang, X. A simple swelling and anchoring method for preparing dense and stable poly(ethylene oxide) layers on polystyrene surfaces. Appl. Surf. Sci. 2008, 254, 4606–4610. [Google Scholar] [CrossRef]
- Huang, N.P.; Michel, R.; Voros, J.; Textor, M.; Hofer, R.; Rossi, A.; Elbert, D.L.; Hubbell, J.A.; Spencer, N.D. Poly(l-lysine)-g-poly(ethylene glycol) layers on metal oxide surfaces: Surface-analytical characterization and resistance to serum and fibrinogen adsorption. Langmuir 2001, 17, 489–498. [Google Scholar] [CrossRef]
- Pasche, S.; De Paul, S.M.; Vörös, J.; Spencer, N.D.; Textor, M. Poly(l-lysine)-graft-poly(ethylene glycol) assembled monolayers on niobium oxide surfaces: A quantitative study of the influence of polymer interfacial architecture on resistance to protein adsorption by ToF-SIMS and in situ OWLS. Langmuir 2003, 19, 9216–9225. [Google Scholar] [CrossRef]
- Malmström, J.; Agheli, H.; Kingshott, P.; Sutherland, D.S. Viscoelastic modeling of highly hydrated laminin layers at homogeneous and nanostructured surfaces: Quantification of protein layer properties using QCM-D and SPR. Langmuir 2007, 23, 9760–9768. [Google Scholar] [CrossRef] [PubMed]
- Nonckreman, C.J.; Fleith, S.; Rouxhet, P.G.; Dupont-Gillain, C.C. Competitive adsorption of fibrinogen and albumin and blood platelet adhesion on surfaces modified with nanoparticles and/or PEO. Colloids Surf. B Biointerfaces 2010, 77, 139–149. [Google Scholar] [CrossRef] [PubMed]
- Reimhult, K.; Petersson, K.; Krozer, A. QCM-D analysis of the performance of blocking agents on gold and polystyrene surfaces. Langmuir 2008, 24, 8695–8700. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Dellatore, S.M.; Miller, W.M.; Messersmith, P.B. Mussel-inspired surface chemistry for multifunctional coatings. Science 2007, 318, 426–430. [Google Scholar] [CrossRef] [PubMed]
- Dalsin, J.L.; Hu, B.H.; Lee, B.P.; Messersmith, P.B. Mussel adhesive protein mimetic polymers for the preparation of nonfouling surfaces. J. Am. Chem. Soc. 2003, 125, 4253–4258. [Google Scholar] [CrossRef] [PubMed]
- Barclay, T.G.; Hegab, H.M.; Clarke, S.R.; Ginic‐Markovic, M. Versatile surface modification using polydopamine and related polycatecholamines: Chemistry, structure, and applications. Adv. Mater. Interfaces 2017, 4, 1601192. [Google Scholar] [CrossRef]
- Liu, M.; Zeng, G.; Wang, K.; Wan, Q.; Tao, L.; Zhang, X.; Wei, Y. Recent developments in polydopamine: An emerging soft matter for surface modification and biomedical applications. Nanoscale 2016, 8, 16819–16840. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Huang, Q.; Deng, F.; Huang, H.; Wan, Q.; Liu, M.; Wei, Y. Mussel-inspired fabrication of functional materials and their environmental applications: Progress and prospects. Appl. Mater. Today 2017, 7, 222–238. [Google Scholar] [CrossRef]
- Chien, C.Y.; Tsai, W.B. Poly(dopamine)-assisted immobilization of Arg-Gly-Asp peptides, hydroxyapatite, and bone morphogenic protein-2 on titanium to improve the osteogenesis of bone marrow stem cells. ACS Appl. Mater. Interfaces 2013, 5, 6975–6983. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Zheng, Y.; Zhao, X.; Ge, Y.; Chen, T.; Liu, Y.; Zhou, Y. Osteoinductive effects of free and immobilized bone forming peptide-1 on human adipose-derived stem cells. PLoS ONE 2016, 11, e0150294. [Google Scholar] [CrossRef] [PubMed]
- Luo, R.; Wang, X.; Deng, J.; Zhang, H.; Maitz, M.F.; Yang, L.; Wang, J.; Huang, N.; Wang, Y. Dopamine-assisted deposition of poly (ethylene imine) for efficient heparinization. Colloids Surf. B Biointerfaces 2016, 144, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Pan, C.; Chen, L.; Liu, S.; Zhang, Y.; Zhang, C.; Zhu, H.; Wang, Y. Dopamine-assisted immobilization of partially hydrolyzed poly(2-methyl-2-oxazoline) for antifouling and biocompatible coating. J. Mater. Sci. 2016, 51, 2427–2442. [Google Scholar] [CrossRef]
- Tsai, W.B.; Chien, C.Y.; Thissen, H.; Lai, J.Y. Dopamine-assisted immobilization of poly(ethylene imine) based polymers for control of cell-surface interactions. Acta Biomater. 2011, 7, 2518–2525. [Google Scholar] [CrossRef] [PubMed]
- Yuan, S.; Li, Z.; Zhao, J.; Luan, S.; Ma, J.; Song, L.; Shi, H.; Jin, J.; Yin, J. Enhanced biocompatibility of biostable poly(styrene-b-isobutylene-b-styrene) elastomer via poly(dopamine)-assisted chitosan/hyaluronic acid immobilization. RSC Adv. 2014, 4, 31481–31488. [Google Scholar] [CrossRef]
- Gao, C.; Li, G.; Xue, H.; Yang, W.; Zhang, F.; Jiang, S. Functionalizable and ultra-low fouling zwitterionic surfaces via adhesive mussel mimetic linkages. Biomaterials 2010, 31, 1486–1492. [Google Scholar] [CrossRef] [PubMed]
- Kuang, J.; Messersmith, P.B. Universal surface-initiated polymerization of antifouling zwitterionic brushes using a mussel-mimetic peptide initiator. Langmuir 2012, 28, 7258–7266. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Zhang, L.; Wang, Y.; Long, Y.; Gao, H.; Zhang, X.; Zhao, N.; Cai, Y.; Xu, J. Mussel-inspired chemistry for robust and surface-modifiable multilayer films. Langmuir 2011, 27, 13684–13691. [Google Scholar] [CrossRef] [PubMed]
- Kord Forooshani, P.; Lee, B.P. Recent approaches in designing bioadhesive materials inspired by mussel adhesive protein. J. Polym. Sci. Part A Polym. Chem. 2017, 55, 9–33. [Google Scholar] [CrossRef] [PubMed]
- Sundaram, H.S.; Han, X.; Nowinski, A.K.; Brault, N.D.; Li, Y.; Ella‐Menye, J.R.; Amoaka, K.A.; Cook, K.E.; Marek, P.; Senecal, K.; et al. Achieving one-step surface coating of highly hydrophilic poly(carboxybetaine methacrylate) polymers on hydrophobic and hydrophilic surfaces. Adv. Mater. Interfaces 2014, 1, 1400071. [Google Scholar] [CrossRef] [PubMed]
- Höök, F.; Kasemo, B.; Nylander, T.; Fant, C.; Sott, K.; Elwing, H. Variations in coupled water, viscoelastic properties, and film thickness of a Mefp-1 protein film during adsorption and cross-linking: A quartz crystal microbalance with dissipation monitoring, ellipsometry, and surface plasmon resonance study. Anal. Chem. 2001, 73, 5796–5804. [Google Scholar] [CrossRef] [PubMed]
- Höök, F.; Vörös, J.; Rodahl, M.; Kurrat, R.; Böni, P.; Ramsden, J.J.; Textor, M.; Spencer, N.D.; Tengvall, P.; Gold, J.; et al. A comparative study of protein adsorption on titanium oxide surfaces using in situ ellipsometry, optical waveguide lightmode spectroscopy, and quartz crystal microbalance/dissipation. Colloids Surf. B Biointerfaces 2002, 24, 155–170. [Google Scholar] [CrossRef]
- Höök, F.; Rodahl, M.; Brzezinski, P.; Kasemo, B. Energy dissipation kinetics for protein and antibody-antigen adsorption under shear oscillation on a quartz crystal microbalance. Langmuir 1998, 14, 729–734. [Google Scholar] [CrossRef]
- Sauerbrey, G. Verwendung Von Schwingquarzen Zur Wagung Dunner Schichten Und Zur Mikrowagung. Zeitschrift Fur Physik 1959, 155, 206–222. (In German) [Google Scholar] [CrossRef]
- Jin, J.; Huang, F.; Hu, Y.; Jiang, W.; Ji, X.; Liang, H.; Yin, J. Immobilizing PEO-PPO-PEO triblock copolymers on hydrophobic surfaces and its effect on protein and platelet: A combined study using QCM-D and DPI. Colloids Surf. B Biointerfaces 2014, 123, 892–899. [Google Scholar] [CrossRef] [PubMed]
- Baty, A.M.; Leavitt, P.K.; Siedlecki, C.A.; Tyler, B.J.; Suci, P.A.; Marchant, R.E.; Geesey, G.G. Adsorption of adhesive proteins from the marine mussel, Mytilus edulis, on polymer films in the hydrated state using angle dependent X-ray photoelectron spectroscopy and atomic force microscopy. Langmuir 1997, 13, 5702–5710. [Google Scholar] [CrossRef]
- Baty, A.M.; Suci, P.A.; Tyler, B.J.; Geesey, G.G. Investigation of mussel adhesive protein adsorption on polystyrene and poly(octadecyl methacrylate) using angle dependent XPS, ATR-FTIR, and AFM. J. Colloid Interface Sci. 1996, 177, 307–315. [Google Scholar] [CrossRef]
- Lee, H.; Scherer, N.F.; Messersmith, P.B. Single-molecule mechanics of mussel adhesion. Proc. Natl. Acad. Sci. USA 2006, 103, 12999–13003. [Google Scholar] [CrossRef] [PubMed]
- Moulay, S. Dopa/catechol-tethered polymers: Dioadhesives and biomimetic adhesive materials. Polym. Rev. 2014, 54, 436–513. [Google Scholar] [CrossRef]
- Jiang, J.; Zhu, L.; Zhu, L.; Zhu, B.; Xu, Y. Surface characteristics of a self-polymerized dopamine coating deposited on hydrophobic polymer films. Langmuir 2011, 27, 14180–14187. [Google Scholar] [CrossRef] [PubMed]
- Liebscher, J.; Mrówczyński, R.; Scheidt, H.A.; Filip, C.; Hadade, N.D.; Turcu, R.; Bende, A.; Beck, S. Structure of polydopamine: A never-ending story? Langmuir 2013, 29, 10539–10548. [Google Scholar] [CrossRef] [PubMed]
- Mian, S.A.; Yang, L.M.; Saha, L.C.; Ahmed, E.; Ajmal, M.; Ganz, E. A fundamental understanding of catechol and water adsorption on a hydrophilic silica surface: Exploring the underwater adhesion mechanism of mussels on an atomic scale. Langmuir 2014, 30, 6906–6914. [Google Scholar] [CrossRef] [PubMed]
- Wattendorf, U.; Kreft, O.; Textor, M.; Sukhorukov, G.B.; Merkle, H.P. Stable stealth function for hollow polyelectrolyte microcapsules through a poly(ethylene glycol) grafted polyelectrolyte adlayer. Biomacromolecules 2008, 9, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Zangmeister, R.A.; Morris, T.A.; Tarlov, M.J. Characterization of polydopamine thin films deposited at short times by autoxidation of dopamine. Langmuir 2013, 29, 8619–8628. [Google Scholar] [CrossRef] [PubMed]
- Heuberger, R.; Sukhorukov, G.; Vörös, J.; Textor, M.; Möhwald, H. Biofunctional polyelectrolyte multilayers and microcapsules: Control of non-specific and bio-specific protein adsorption. Adv. Funct. Mater. 2005, 15, 357–366. [Google Scholar] [CrossRef]
| Surface | XPS Atomic Percent (%) | |||||
|---|---|---|---|---|---|---|
| C(1s) | O(1s) | N(1s) | Si(2p) | Na(1s) | S(2p) | |
| PS | 94.5 ± 0.5 | 4.3 ± 0.3 | 0.8 ± 0.1 | 0.3 ± 0.1 | – | 0.1 ± 0.1 |
| PS + DOPA | 79.1 ± 0.5 | 15.3 ± 0.4 | 5.6 ± 0.1 | – | – | – |
| PS + DOPA + PLL-g-PEG | 73.8 ± 0.4 | 19.3 ± 0.3 | 6.4 ± 0.2 | 0.3 * | 0.2 ± 0.0 | – |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hecker, M.; Ting, M.S.H.; Malmström, J. Simple Coatings to Render Polystyrene Protein Resistant. Coatings 2018, 8, 55. https://doi.org/10.3390/coatings8020055
Hecker M, Ting MSH, Malmström J. Simple Coatings to Render Polystyrene Protein Resistant. Coatings. 2018; 8(2):55. https://doi.org/10.3390/coatings8020055
Chicago/Turabian StyleHecker, Marcelle, Matthew Sheng Hao Ting, and Jenny Malmström. 2018. "Simple Coatings to Render Polystyrene Protein Resistant" Coatings 8, no. 2: 55. https://doi.org/10.3390/coatings8020055
APA StyleHecker, M., Ting, M. S. H., & Malmström, J. (2018). Simple Coatings to Render Polystyrene Protein Resistant. Coatings, 8(2), 55. https://doi.org/10.3390/coatings8020055





