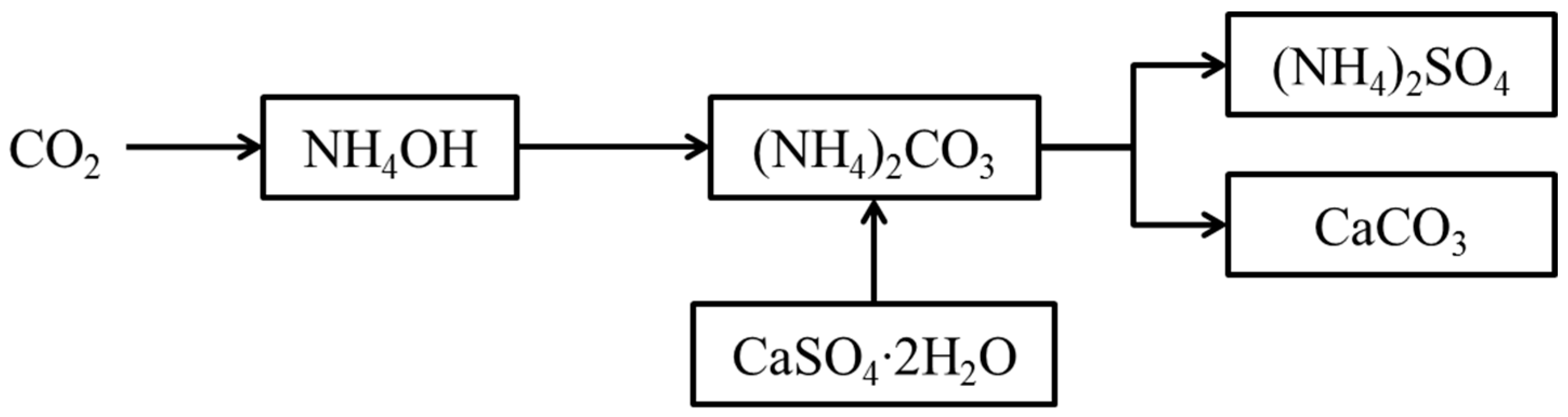
1. Introduction
In many fields such as polymers, paints, thermoplastic resins, and composite materials, a variety of fillers are used for cost-saving and product quality improvement. Among these fillers, the most commonly used is CaCO
3, which is generally a white crystalline solid abundant in nature [
1]. For a long time, CaCO
3 was used primarily as an abrasive or a soil conditioner. Since the early 20th century, it has been used industrially as paint, white pigment, and dye color. In the 1950s, its use as a filler mixed with polymer products increased dramatically in parallel with advances in plastic engineering [
2]. More recently, it has been used primarily as a filler or an additive in many industries such as cement, rubber, plastic, building material, and paper [
3,
4,
5]. CaCO
3, as a filler, is used to improve the physical properties of polymer products, improve processability, increase the surface hardness, add functionality, and save costs associated with the volume increase by mixing it with the polymer matrix [
6]. CaCO
3 is used predominantly in these applications because it is inexpensive and easily applicable, and has adequate specific gravity and a high degree of whiteness as a filler, while having superior physical properties, such as noncombustibility, compared to those of other inorganic materials [
7,
8,
9].
Increasing the effectiveness of CaCO
3 for use in more industries requires added functionality that can satisfy the physicochemical properties to fit that purpose [
10]. Until now, methods to add or improve the functionalities of CaCO
3 were typically based on controlling its particle size or morphology. The CaCO
3 filler used in actual industrial applications in Korea mostly consists of heavy CaCO
3 obtained from limestone ores with the particle size controlled by pulverizing. In recent times, morphology control using a chemical precipitation reaction and the production of precipitated CaCO
3 in ultrafine powder form have also been actively pursued. Atomization of the powder increases the hydrophilic characteristics and diffusion energy on the surface, which can lead to a low dispersion and agglomeration when mixed in an organic polymer matrix [
11]. As a result, the usability of CaCO
3 as a filler is limited because of its irregular surface and low bonding force. The goal of surface modification of CaCO
3 is to address these issues by adding functionalities or modifying the physical properties through changes in the surface properties of the powder to increase its miscibility with a polymer matrix and reduce the surface tension to enhance the physical properties, mechanical performance, thermal resistance, or dispersibility of the powder. Depending on the intended use, the goal of surface modification of CaCO
3 includes improved handling, such as enhanced adhesion, agglomeration, fluidity, and dispersibility [
12,
13,
14]. In addition, combined effects can also be achieved through the addition of optical and surface activation functionality and increased mechanical strength. Such modification effects are often dependent on the types and number of modifiers used. Industrial modifiers that have been primarily used include fatty acids and resin acid, and a wide range of coupling agents, surfactants, amine salts, and esters and polyethylene synthetic compounds are currently in use [
15,
16].
Recent studies have reported on the use of bio-mineralization. These studies have focused on controlling the physicochemical properties of CaCO
3. According to Tester et al., the particle size of amorphous CaCO
3 (ACC) can be controlled by using liposomes with phospholipid vesicles [
17]. They reported that the use of liposomes enabled good control of ACC nucleation and growth, and could determine the ACC particle size due to its reaction kinetic effect. Research on mesocrystal formation was reported by Yu et al. [
18]. In their study, high-magnesium calcite was used to form mesocrystals in aqueous solution. In general, particle size was closely related to reaction time and thereby with crystal growth or formation. Furthermore, phosphatidylcholine showed similar performance with liposomes when Ca
2+ ion interaction was considered. According to Yu et al., control of particle size was effected by the difference in energy as the driving force. Tester et al. and Yu et al. used amorphous materials to form CaCO
3, which was then coated with liposomes. In these processes, amorphous CaCO
3 changed to crystalline phase such as calcite and aragonite. When the driving force decreased, the size of crystals formed was also found to decrease [
18].
This study aims to modify the surface of CaCO3 powder. As mentioned, the physicochemical properties of CaCO3 need to be modified to render it suitable for practical application in various industries. Various methodologies and properties were considered in experiments to improve its properties such as dispersibility, hydrophobicity, surface activation, and physical properties including strain, elongation, and tensile strength. This study used a chemical method to dissolve various modifiers such as fatty acids, surfactants, silica, and organosilanes in organic solvents for admixing with CaCO3, in addition to the mechanochemical method of mixing CaCO3 and the modifier on a rotating mill to modify the surface. The aim of these methods was to improve the surface coating effects by increasing the adsorption efficiency under conditions that would eliminate the high level of energy consumption required in conventional dry and wet processes. In the chemical method, considering the solubility and ease of treatment of the modifiers, the adsorption characteristics were derived according to the concentration, reaction speed, and temperature of the modifier in the organic solvent, while the degree of the coating and changes in surface characteristics, surface energy of the powder before and after modification, and physical and chemical characteristics were investigated. Moreover, the increase in the hydrophobic and dispersion effects on the modified powder according to various factors such as the amount of modifier, reaction speed, and reaction temperature were discussed, along with the spectroscopic physicochemical characteristics and functional improvement effects on the products that used the modified powder.
3. Results and Discussion
As mentioned, the CaCO
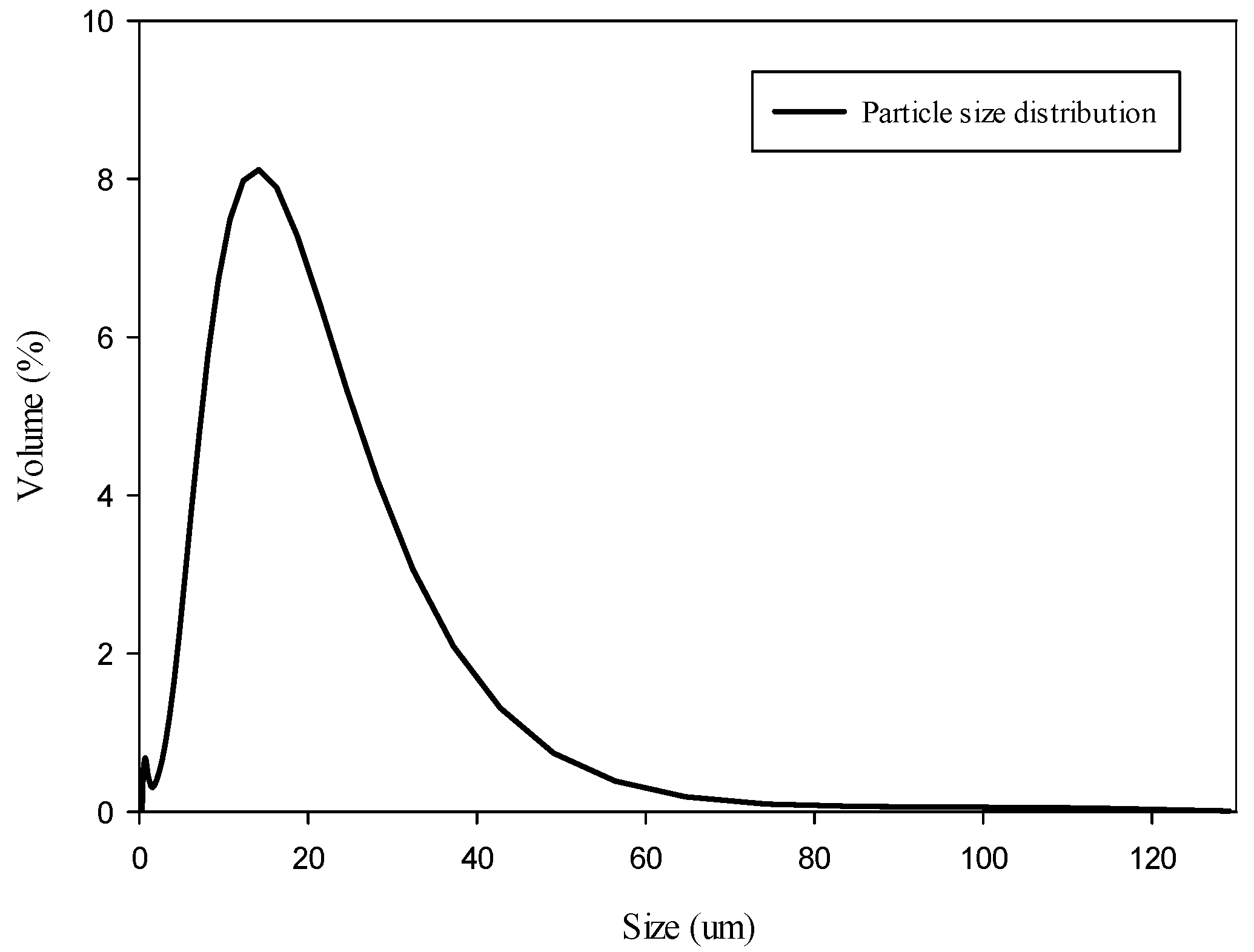
3 used in the experiment was obtained from a pilot plant by KIGAM (Korea Institute of Geoscience and Mineral Resources, Daejeon, Korea). With respect to the particle size distribution, 90% of the unprocessed CaCO
3 obtained from the plant process showed a particle size distribution of <30 μm, while 50% showed a particle size <12.58 μm (
Figure 2). In addition, 90% of the CaCO
3 atomized through the pulverization process showed a particle size distribution of <7.65 μm, while 50% showed a particle size of <7.2 μm. Generally, the particle size of the precipitate CaCO
3 (PCC) was smaller than that of the ground CaCO
3 (GCC). PCC, for its chemical formation, generally uses soluble precursors, and thus has a small particle size; however, GCC was obtained from limestone, in limestone mining, for example. Therefore, the PCC particle size was mostly uniform in contrast to the GCC particle size. As mentioned in the experimental section, we used PCC for the pilot plant. As a result, its distribution was not different. Additionally, the CaCO
3 we used had no relationship with the flotation effect because its distribution was uniform. In other words, we assumed that the main effect of flotation was the degree of surface coverage. Therefore, the CaCO
3 used did not have a classification because we considered the practicability. Moreover, the specific surface area was analyzed to check the effect of using fatty acids. This estimation was calculated using the Mastersize 2000 model (Malvern Co. Ltd., Malvern, AR, USA). Additionally, its definition is the total surface area divided by the total volume. The specific surface areas of the untreated and pulverization-treated CaCO
3 were 1.02 m
2/g and 1.62 m
2/g, respectively [
24]. Most of the crystalline structures of the CaCO
3 samples determined by X-ray diffraction were in the calcite form, while trace amounts of the vaterite form were observed. The purity of CaCO
3 was determined by thermogravimetric analysis, by converting the amount decreased by decomposition at high temperatures to the amount of carbon dioxide present in CaCO
3 and by determining the unreacted sulfur content; this showed similar results: 94% and 96%, respectively [
24]. Although the CaCO
3 that we used had been pretreated by heat, it did not change into CaO powder. In general, CaCO
3 decomposes at high temperatures over 850 °C. However, our study did not use high temperatures. Our previous study also mentioned that this is calcite [
24]. The solvent was used only to dissolve the modifier. In this process, only fatty acids were used and, thus, reformation could not be obtained.
3.1. Pretreatment Activation Effect of CaCO3 Powder
When a powder is treated at high temperatures, the adsorption area becomes activated, resulting in an increase in the specific surface area [
25]. To investigate this effect, the changes in the specific surface area were determined according to the temperature of the CaCO
3 powder samples that underwent a pulverization treatment compared with the untreated samples [
24]. Through the specific surface area analysis, we could guess the effect of modification caused by the fatty acid.
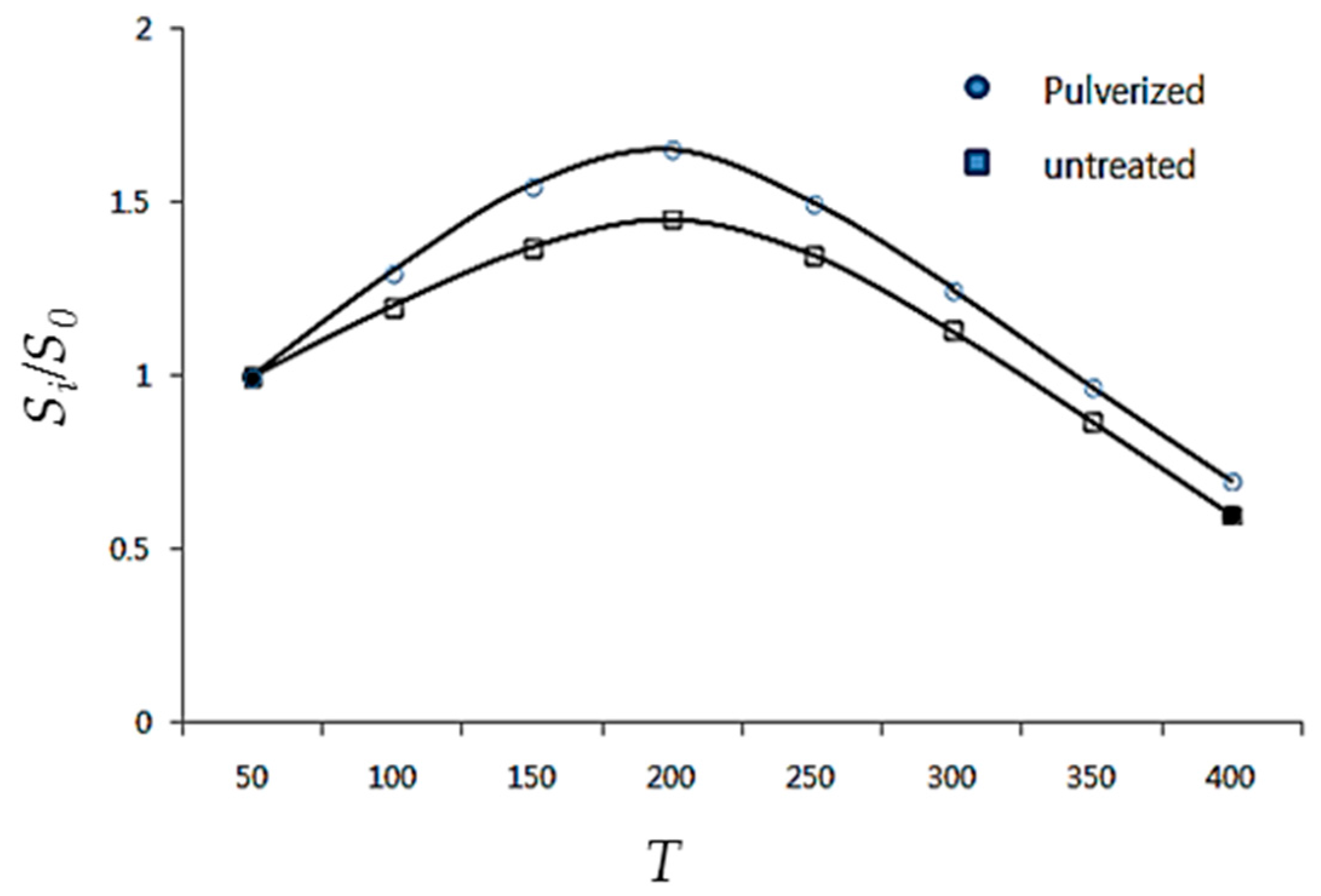
Figure 3 shows the relative ratio of the specific surface area of the pulverized (increased in temperature) CaCO
3 sample to that of the untreated sample.
Figure 3 shows a prominent increase in the specific surface area of the CaCO
3 sample heat-treated up to 180 °C followed by a drastic decrease in the specific surface area with further increases in the heat treatment temperature. The increase in the specific surface area of the heat-treated CaCO
3 up to 180 °C was attributed to the adsorption area being activated by the removal of foreign substances (primarily water) from the absorption area within the pores of the fine particles or powder surface, whereas the decrease in the specific surface area observed in the samples heat treated at temperatures above 180 °C is believed to be the result of fine-sized sintering according to the surface expansion of unstable ions present on the powder surface [
26]. This shows that, even over the temperature range well below the normal decomposition temperature of 900 °C for CaCO
3, atoms on the activated surface area can rearrange themselves to change the adsorption properties of the surface. This can be observed even more distinctly in nanosized powders, where the nanosized CaCO
3 generally has a large surface area to volume ratio and a larger percentage of surface-activated ions with unsaturated ionic bonds. Therefore, the functional properties such as adsorption, adhesion, and agglomeration are enhanced, whereas the handling and storage become increasingly difficult [
27,
28]. The active ratio was “0” in this study because the nontreated CaCO
3 was precipitated in water. This study aims to modify CaCO
3 to ascertain the possibility of its reusability in various industries. For this reason, we used fatty acids to modify CaCO
3. General CaCO
3 is hydrophilic and, thus, nontreated CaCO
3 does not have resistance to water. Therefore, nontreated CaCO
3 was precipitated in water, the result of which was different from that obtained with treated CaCO
3. Fatty acids were used to change the CaCO
3 properties. The fatty acids were dissolved in IPA (IsoPropyl Alcohol) and chloroform because they do not dissolve in water. IPA and chloroform have different end groups; thus, the solubility of each fatty acid was different. Each fatty acid was therefore selected by taking into account its chemical properties.
3.2. Modification Effects of Calcite Using Fatty Acids as Modifier
Among the fatty acid modifiers, stearic acid is the one most often used for nonpolarization of the CaCO
3 powder surface because it contains a nonpolar long-chain
R group, which can facilitate nonpolar modification effects at the lowest decomposition melting temperature [
29]. All results were compared to the duplicated results, and the difference was within 10%. The results are supposed to relate to the interaction of stearic acid with minerals. However, this reaction mechanism was not clearly investigated.
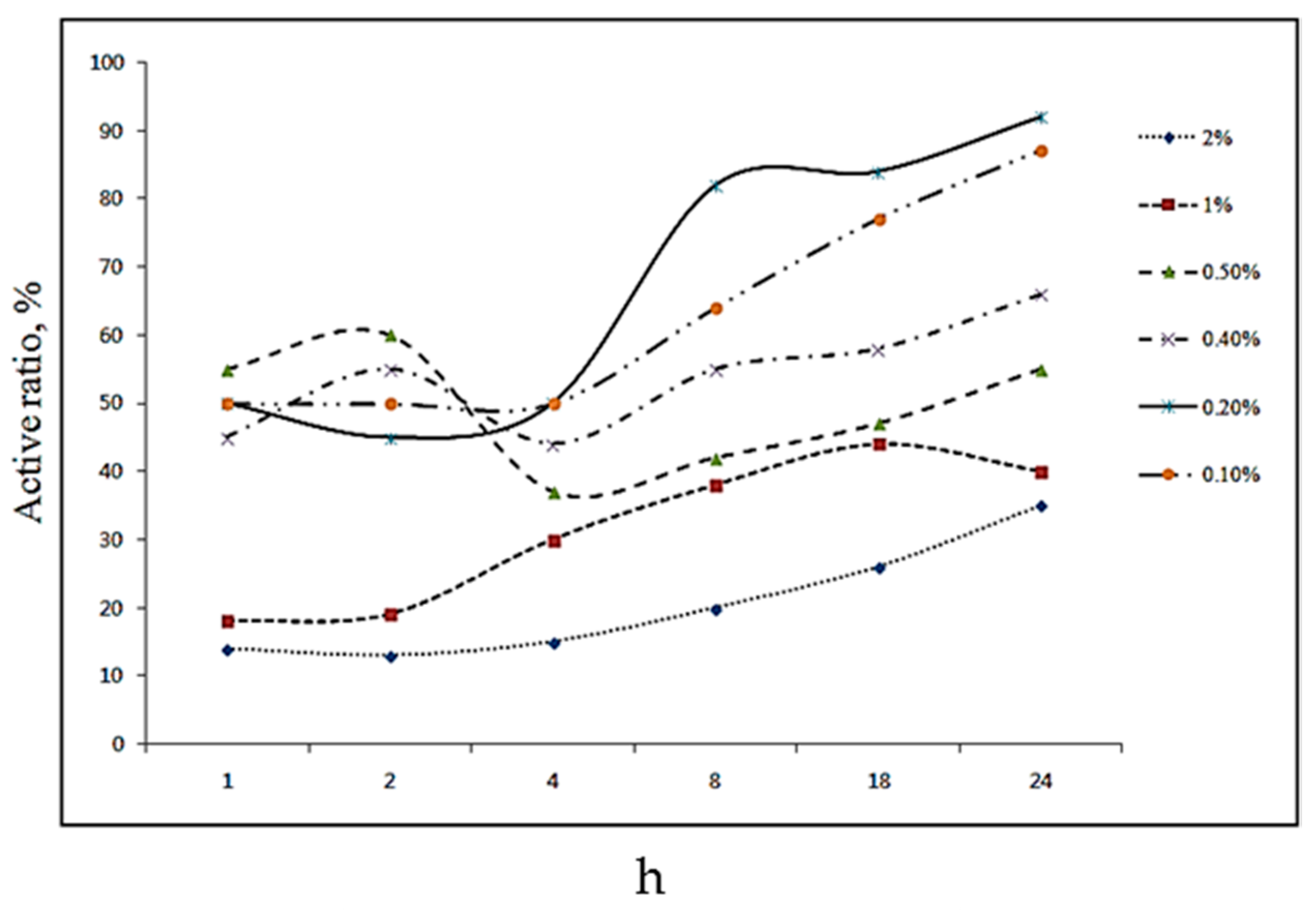
Figure 4 and
Figure 5 (chloroform solvent) show changes in the active ratio according to the reaction time at each modifier solution concentration, which was modified by dissolving stearic acid in 2-propanol and chloroform solvent. 2-Propanol and chloroform solvent are well-known organic solvents. They were used to dissolve the fatty acids. In particular, the solubility of mineral particles in organic solvents was not considered because of its insignificance. Surface coverage and optimum treatment time were closely related to the dissolved stearic acid concentration and H-bonding status of the type of solvents used rather than the solubility.
The reaction times in the 2-propanol solvent were 4, 8, 18, and 24 h, whereas they were 1, 2, 4, and 8 h in chloroform, which shows that the adsorption equilibrium is achieved within a shorter time in chloroform than in 2-propanol. A closer examination of the results obtained using 2-propanol as the solvent, as shown in
Figure 4, revealed an increase in the adsorption time with increasing active ratio. However, when chloroform was used as the solvent, as shown in
Figure 5, the best active ratio was observed at a treatment of 1 h, with the active ratio showing a distinct decrease with increasing treatment time. Therefore, the two solvents showed active ratio results based on the treatment time that were completely contradictory to each other. In general, 2-propanol shows an upward pattern and chloroform shows a decreasing trend with respect to the treatment time. Moreover, although 2-propanol showed the highest active ratio when it was treated at relatively dilute concentrations for a long time, chloroform showed the highest active ratio when it was treated at high concentrations for a short time. This was attributed to hydrogen bonding between the solvent and modifier because 2-propanol possesses an intramolecular hydroxyl group (–OH), which allows the formation of a single hydrogen bond between the molecules, whereas stearic acid has structural characteristics that allow it to form double hydrogen bonds [
7]. When the concentration of stearic acid in the 2-propanol solvent is very low, the hydrogen bonds between the stearic acid molecules are weak; this allows stearic acid with a relatively large dipole moment to be dominant in adsorption competition on the adsorption area of the CaCO
3 surface, thereby showing high adsorption efficiency. It is also suspected that when the concentration of stearic acid increases, the formation of intermolecular double hydrogen bonds is promoted. This causes the terminal activation area to become nonpolar and have weak affinity to CaCO
3, resulting in 2-propanol being polar-dominant to preoccupy the adsorption area of CaCO
3. Consequently, a low concentration treated for a longer duration resulted in superior modification effects than the treatment at high concentrations. In contrast, in the nonpolar chloroform solvent, fatty acids have a much greater affinity to CaCO
3 than to chloroform. Therefore, adsorption competition with the solvent is negligible, and a strong and rapid adsorption equilibrium can be expected in high concentrations of the modifier solution. Moreover, a decrease in the active ratio, which was observed when the treatment time was maintained for a long time, is believed to have originated from the stronger affinity between the nonpolar chloroform and nonpolar stearate than the binding force between the absorption group of stearate and the adsorption area on the CaCO
3 powder surface. This caused the adsorption equilibrium between CaCO
3–stearate–chloroform in the adsorption area of CaCO
3 to shift toward the direction in which the fatty acids adsorbed on CaCO
3 dissociate, leading to a lower active ratio.
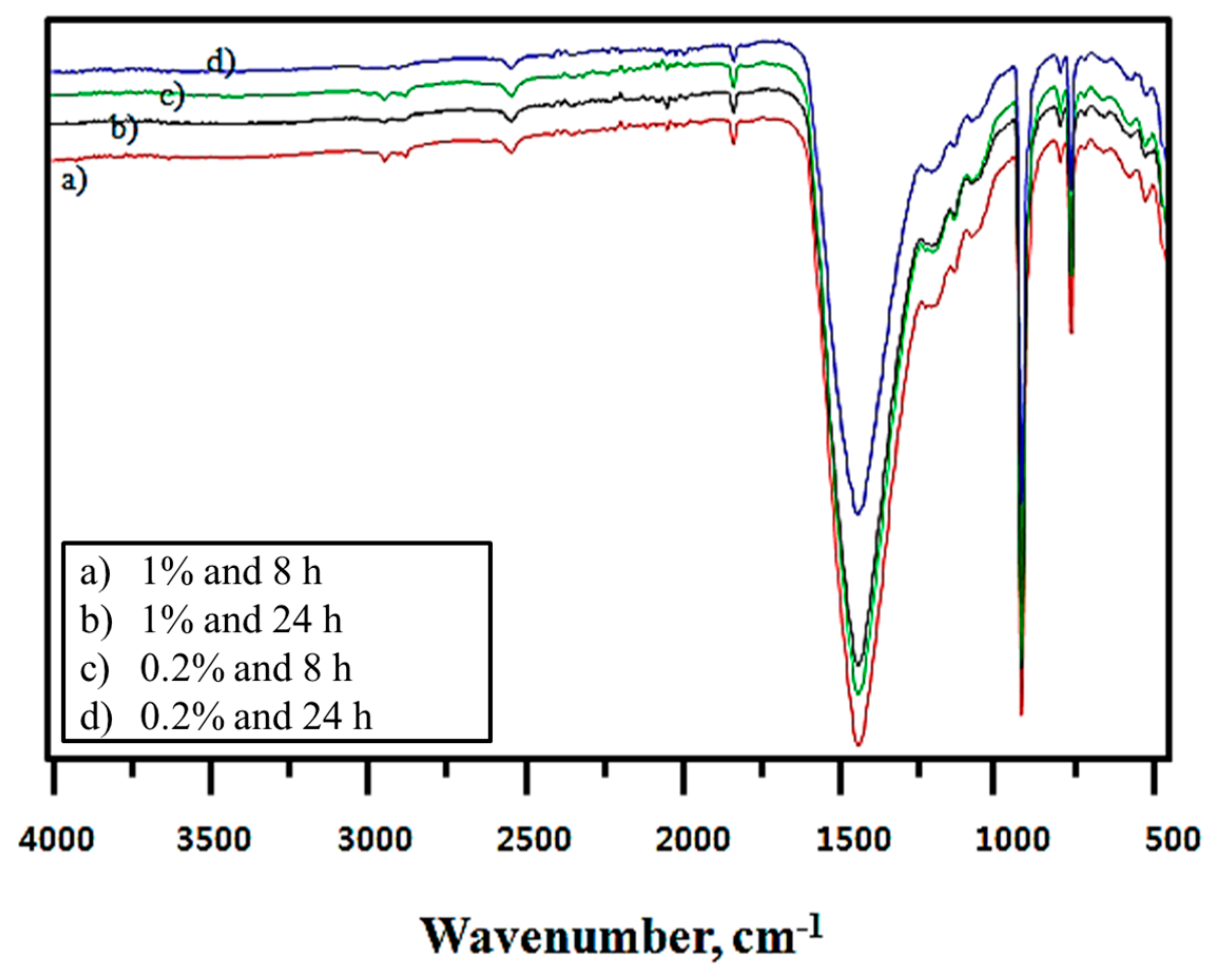
Figure 6 presents the Fourier transform infrared (FT-IR) spectra of modified CaCO
3 in 2-propanol solvent obtained based on changes in the treatment time and modifier concentration. In
Figure 6, the modifier concentration and treatment time for modified CaCO
3 spectra were assigned to the –CH
2 and –CH
3 stretching vibrations, which are indicators of the degree of modification. The intensity of these two peaks was highest where marked a) and lowest where marked d).
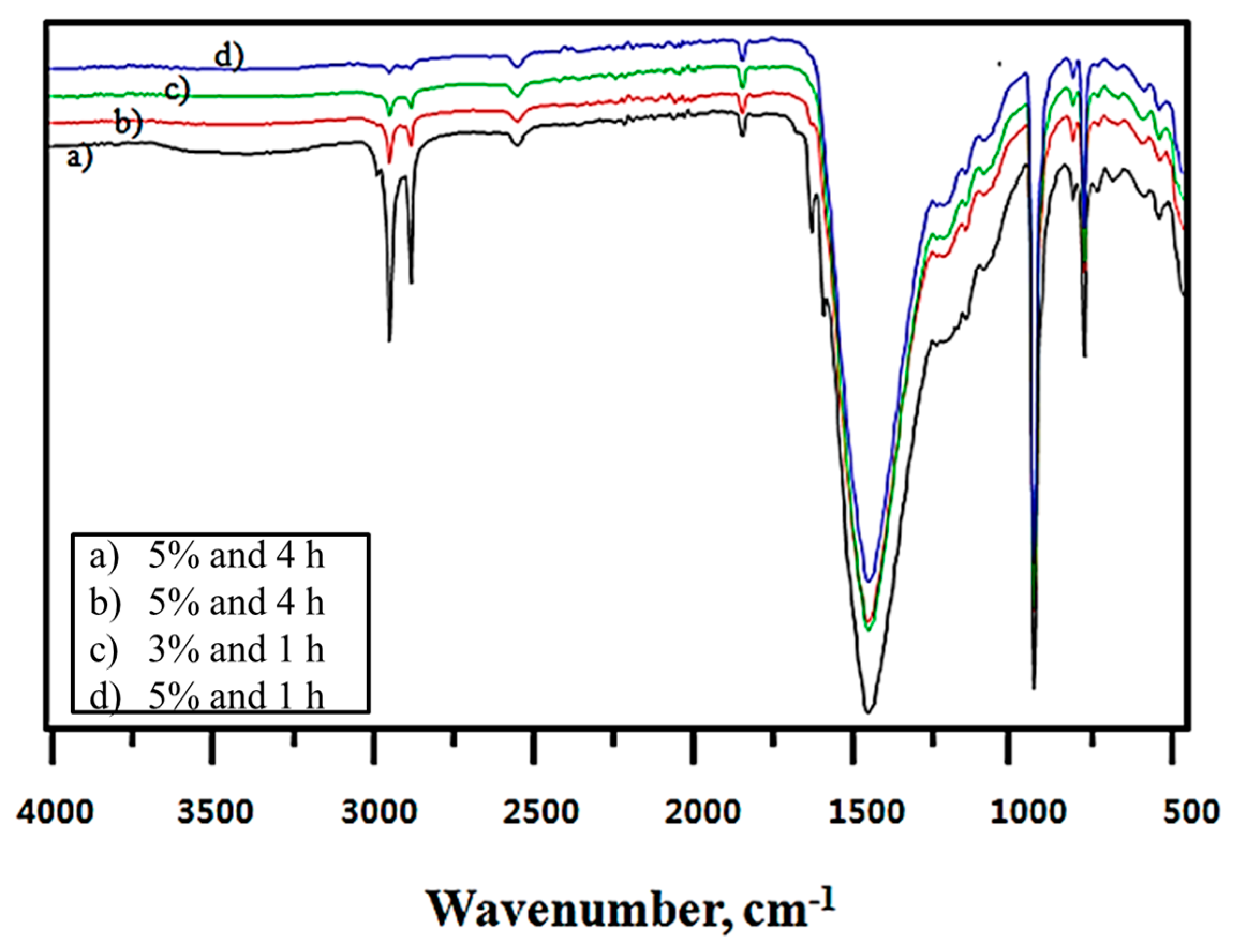
Figure 7 presents the modified CaCO
3 FT-IR spectra obtained when chloroform was used as the solvent; the modifier solution concentrations at the reaction times were larger and more distinct than those obtained for 2-propanol, as shown in
Figure 6. Because the FTIR spectrum obtained at high concentrations and short reaction times (a) was dominant, the results were consistent with the active ratio shown earlier. To summarize the aforementioned active ratio and FT-IR results, the –CH
2 and –CH
3 absorption peaks observed in the chloroform solvent, which are dependent on the amount adsorbed, were much larger than those of 2-propanol. This suggests a larger amount of stearate adsorbed on the CaCO
3 powder surface, whereas both solvents showed an excellent active ratio of ≥90% under their respective optimal conditions. The effective active ratio that took into account the unit adsorption amount was actually higher in 2-propanol.
Table 1 lists the modification effects of the CaCO
3 surface modified using various fatty acids on the active and dispersing ratios. These results show the modification effects obtained under the optimal conditions for the concentration of fatty acid modifiers and treatment, which were obtained from the optimal solvent considering the solubility and dispersibility. From
Table 1, we can see that the selected modifiers showed a better performance than the others. In particular, 2-propanol was better than chloroform in terms of saturated concentration because the saturated concentration of chloroform in each modifier was greater than that of 2-propanol by more than 35 times. Therefore, we presumed that 2-propanol has sufficient potential for application in industry from an economic viewpoint. According to Tran et al., fatty salt can improve surface modification when applied to PCC [
3]. They used sodium series fatty acids to modify nano-PCC using a carbonation process. As a result, fatty acids improved the physicochemical properties and changed hydrophilic CaCO
3 into hydrophobic CaCO
3. Further details are provided in the next section.
3.3. Evaluation of the Contact Angle Characteristics
The contact angle refers to the angle formed when liquid achieves thermodynamic equilibrium on a solid surface. This manifests in the wettability of a solid surface, with a larger contact angle corresponding to lower wettability, highly hydrophobic properties, and low surface energy value; a smaller contact angle corresponds to high hydrophilicity and relatively large surface energy [
30]. The equation that expresses the relationship between the contact angle and interfacial energy is the Young’s equation obtained under uniform, flat, and ideal surface conditions. This equation is used to obtain uniform values with high reproducibility in repeated measures [
31]:
where γ
LV denotes the liquid–gas interfacial tension; θ denotes the contact angle; γ
SV denotes the solid–gas interfacial tension; and γ
SL denotes the solid–liquid interfacial tension.
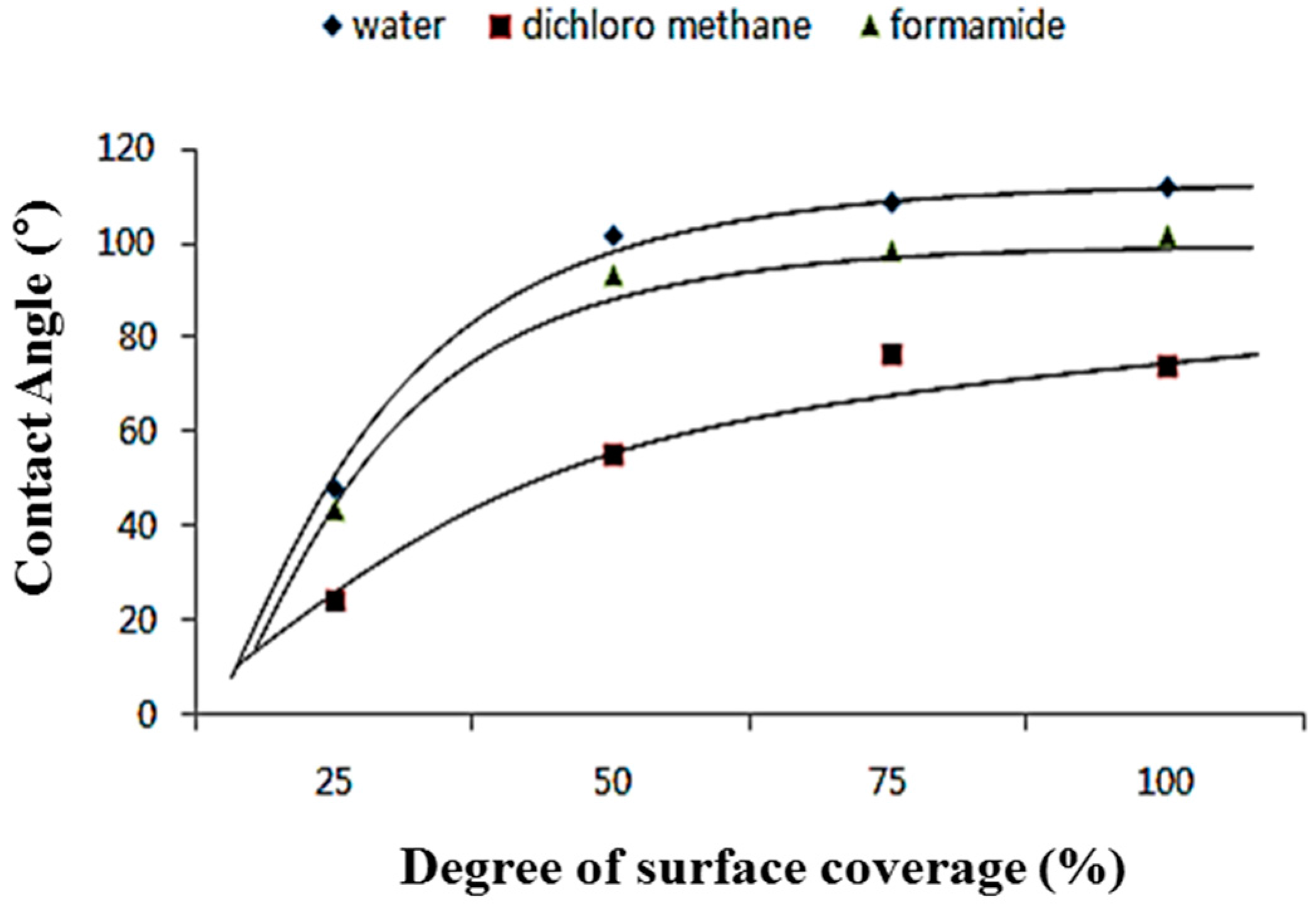
Figure 8 presents contact angles according to the surface coating rate of stearic acid. Unmodified CaCO
3 showed complete wettability in all solvents because the surface energy of the powder, which comprised the spreading factor and polar factor, was very high [
15]. The contact angle increased with increasing surface coating rate of stearic acid. According to published reports, when a fatty acid forms a complete single-molecule adsorption layer on CaCO
3 powder, the maximum contact angles in water and formamide are approximately 103° and 95°, respectively. This shows that the values almost reached the maximum value at a surface coating rate of ≥75% [
32]. Although the fatty acid used had a high concentration, it did not exhibit any significant difference. According to Tran et al., the hydrophobic alkyl chain remains in the first layer of the CaCO
3 coating of stearic acid [
3]. Through this reaction, stearic acid was stored as a multilayer. Thus, a hydrophilic chain remained at the outer layer but most of it was removed by the dry process. Consequently, the modified CaCO
3 exhibited hydrophilic characteristics. In this study, we also performed a similar process to modify CaCO
3. Consequently, the results were in agreement with the results of other researchers. Therefore, even at a surface coating rate of approximately 75%, a value similar to the surface energy in complete single-molecule adsorption was achieved and the results showed that fatty acids adsorbed on the powder surface were oriented vertically on the surface.
4. Conclusions
Surface characteristics of CaCO3 obtained from a mineral carbonation plant using desulfurized gypsum, an industrial byproduct, were modified to increase its usability as a filler for a range of industrial products such as paints, plastics, rubber, and paper. The chemical method involved modification using various types of fatty acids as modifiers, which were dissolved in organic solvents and admixed with CaCO3 to determine the optimal conditions according to various factors, such as the reaction speed, temperature, modifier concentration, and the specific solubility of the solvent. The effects on the hydrophobicity and dispersibility as well as changes in the physicochemical surface characteristics of the modified powder were investigated. All treatment processes were conducted at room temperature to eliminate the high thermal energy consumption required in conventional processes and the surface modification of CaCO3 was induced using a low-cost and high-efficiency method. The obtained modified CaCO3 powder showed a change in surface characteristics from a hydrophilic polar form with high surface energy to a hydrophobic nonpolar form with low surface energy, and exhibited high affinity and dispersibility when added as a filler in a polymer matrix. Therefore, we believe that this study will assist various industries in the field of CaCO3 usage.
In this study, we assumed that fatty acids have sufficient potential to modify CaCO3. However, this study used only two types of fatty acids, which means that other modification chemicals would need to be developed. Besides this, the practical application of modified CaCO3 is to be investigated. Since modified CaCO3 was coated with hydrophobic materials, we intend to use it in paper products and diapers to check its permeability and adhesion characteristics.