Graphene Coating on Copper by Electrophoretic Deposition for Corrosion Prevention
Abstract
1. Introduction
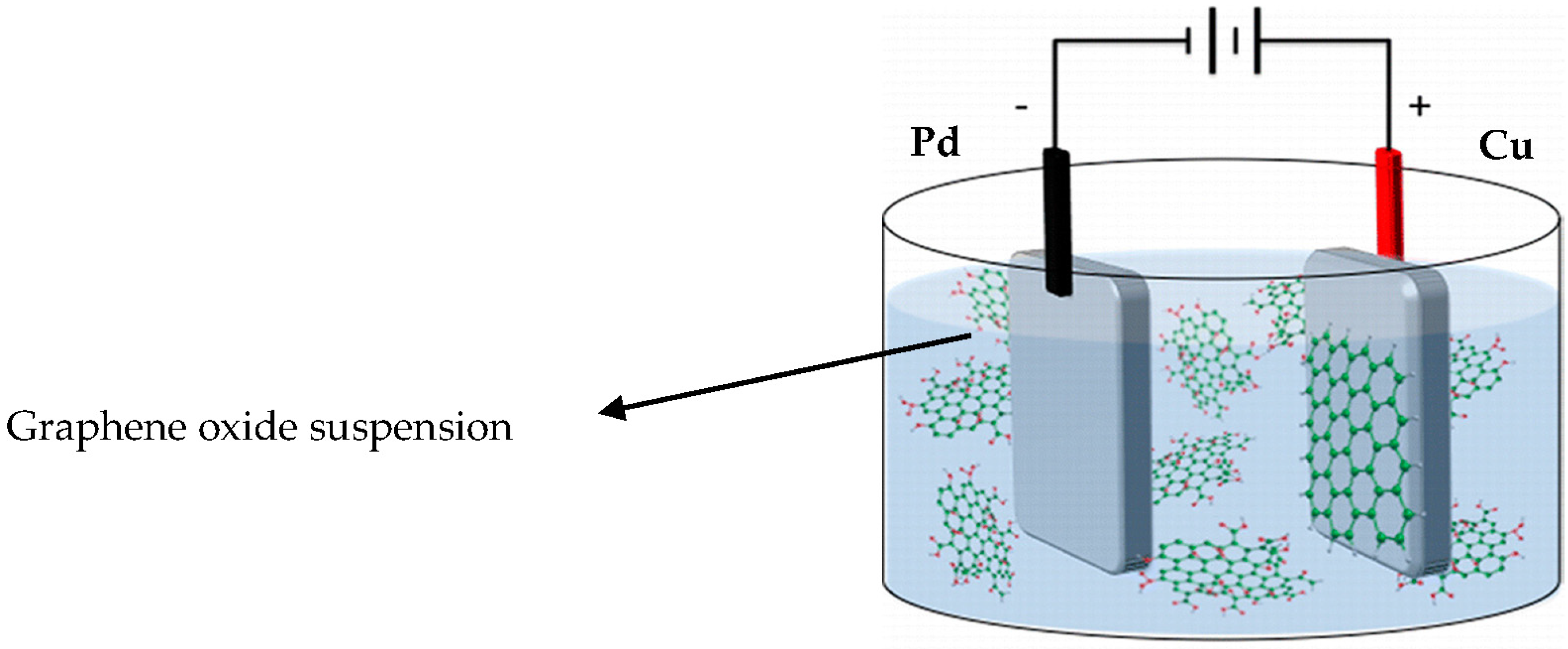
2. Materials and Methods
3. Results and Discussion
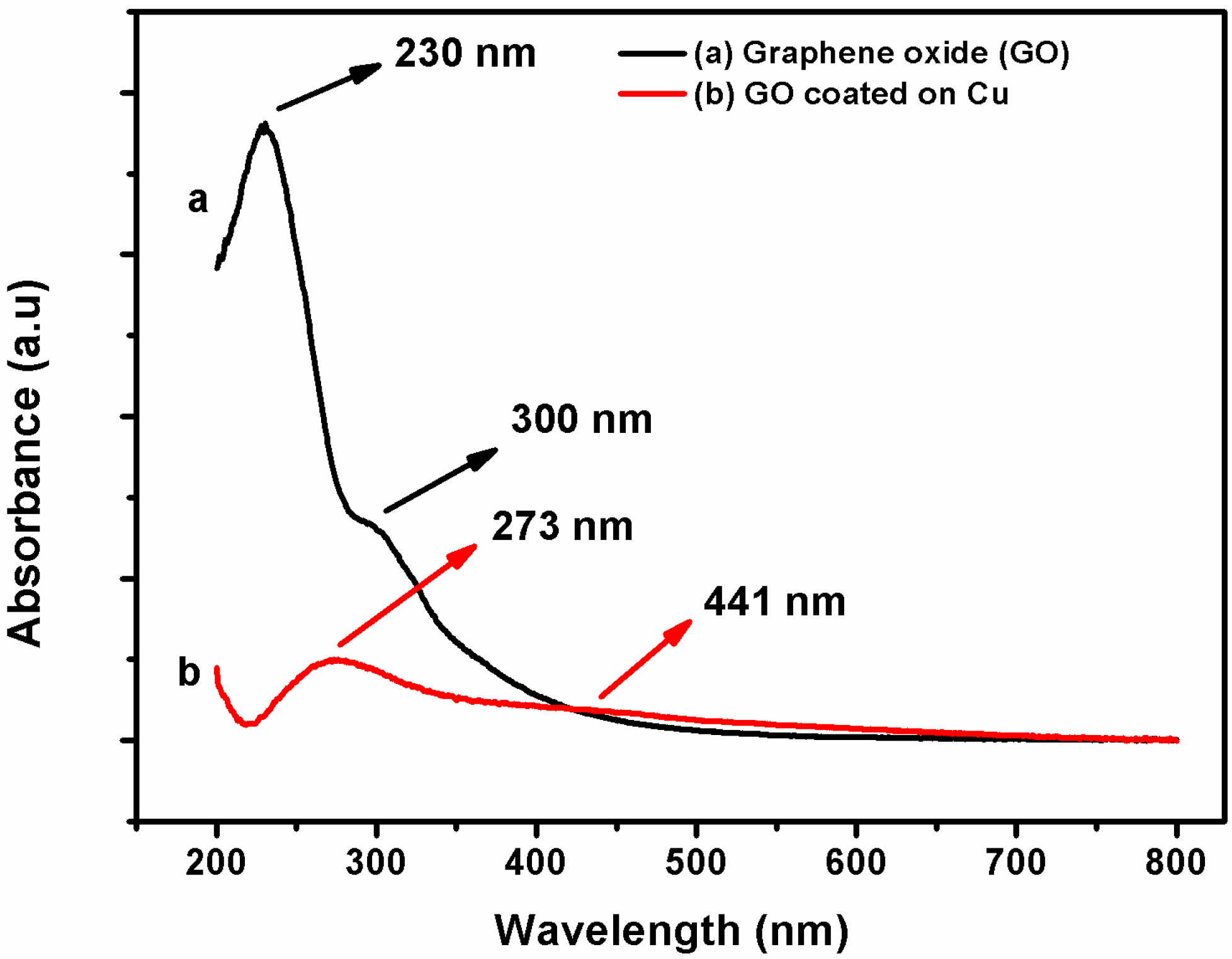
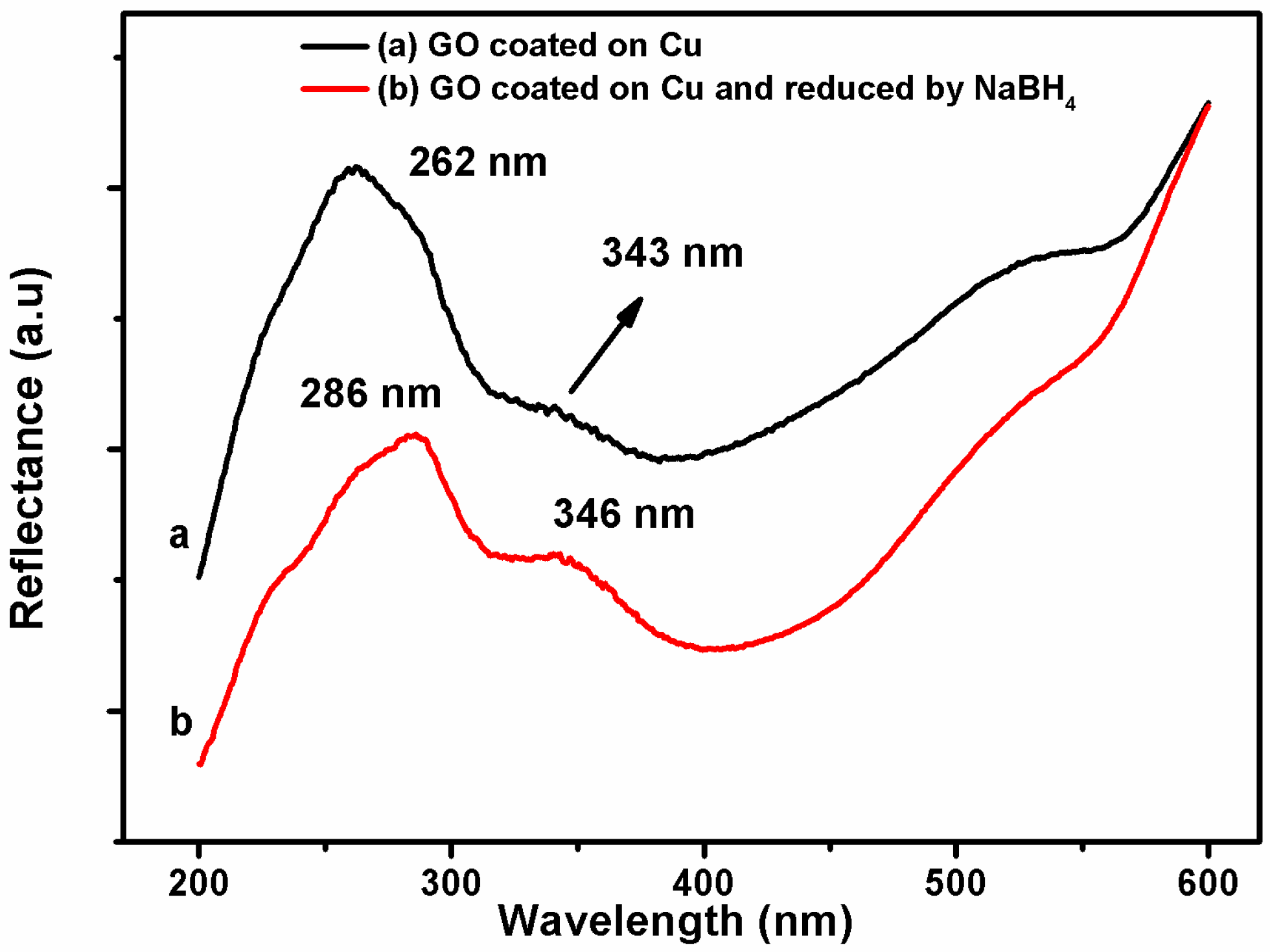
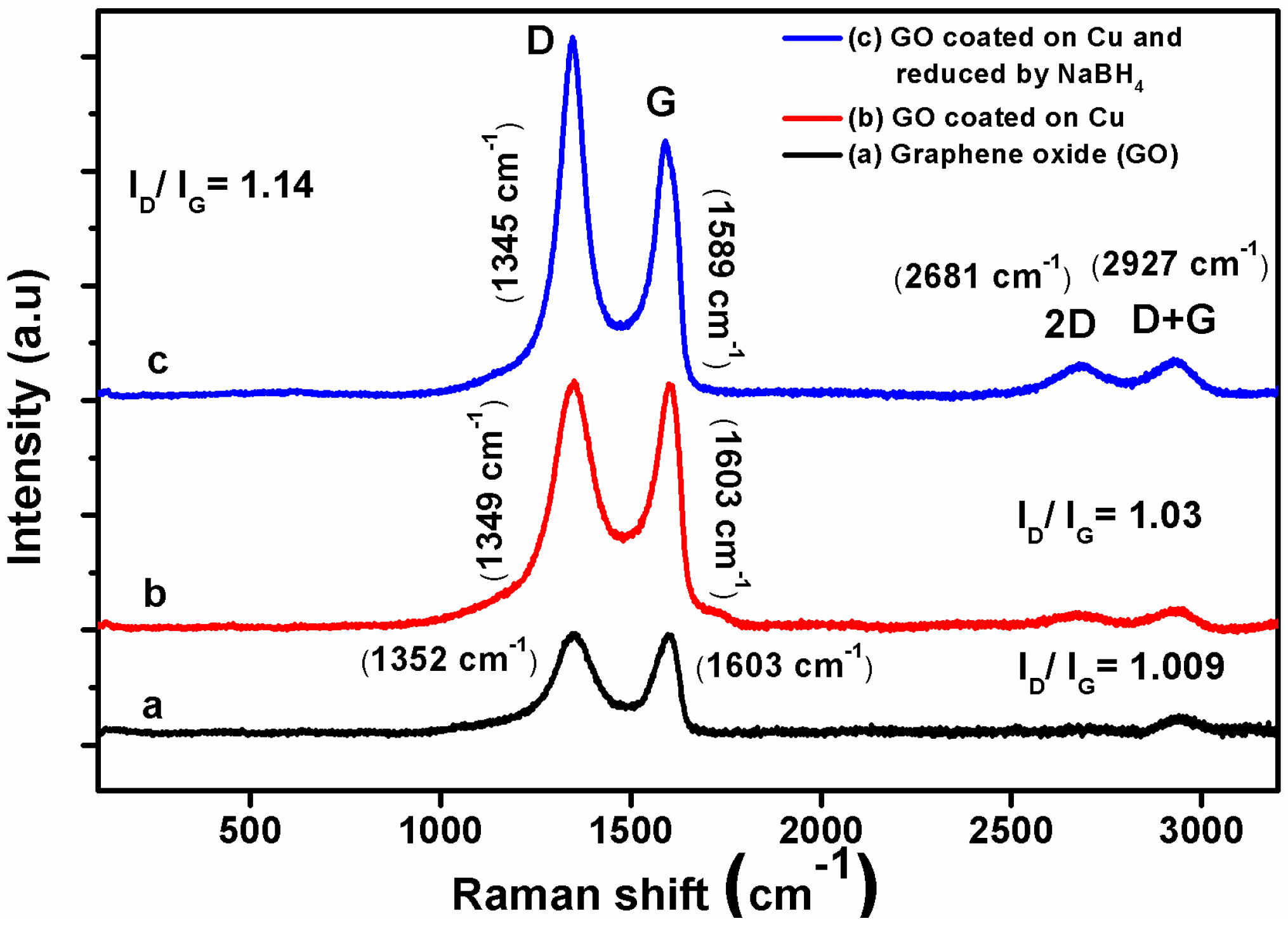
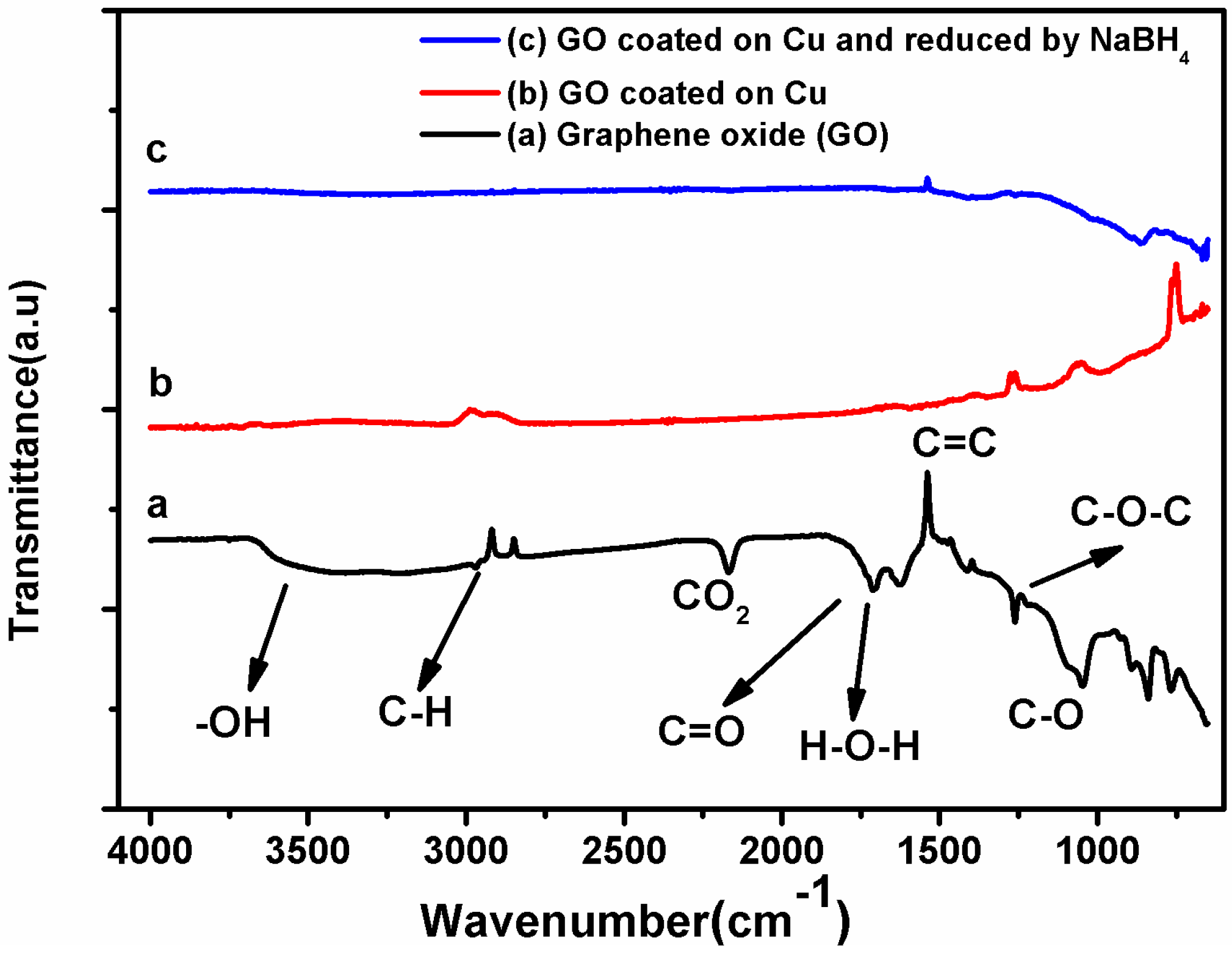
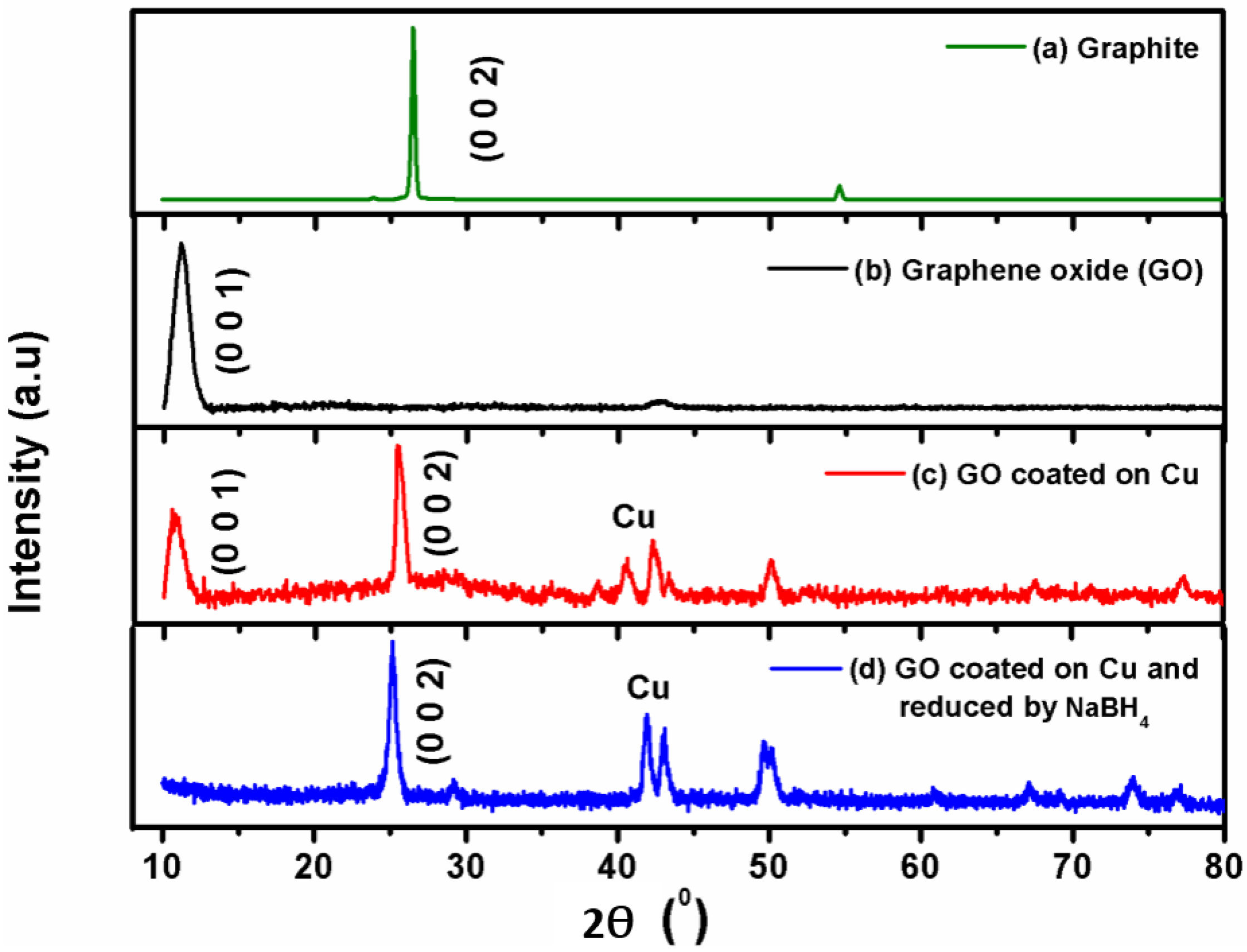
3.1. Characterization of Coating
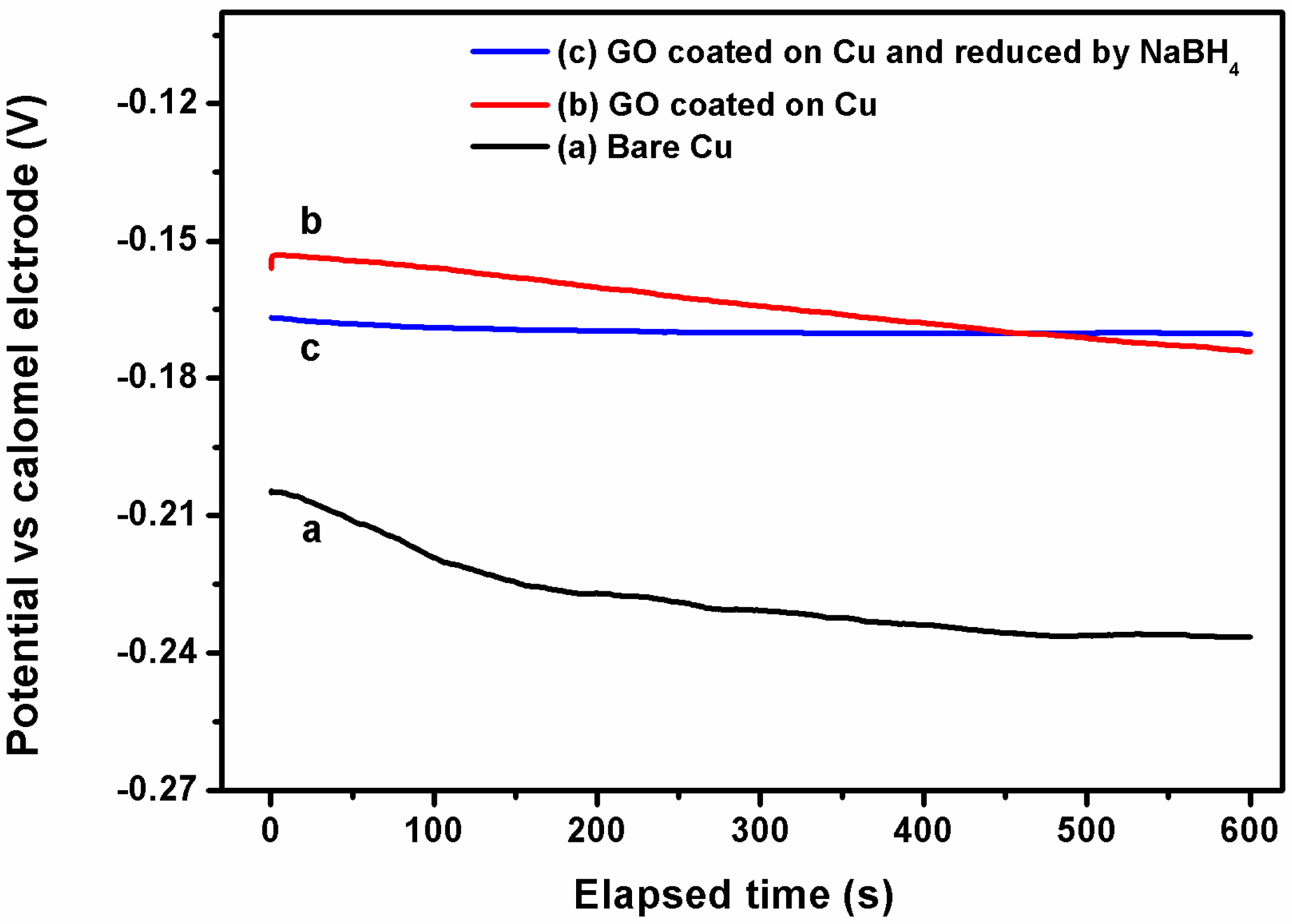
3.2. Corrosion Resistance
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rahmanto, W.H.; Gunawan, R.N. Corrosion rate of copper and iron in seawater based on resistance measurement. J. Coast. Dev. 2002, 5, 67–74. [Google Scholar]
- Singh, B.P.; Nayak, S.; Nanda, K.K.; Jena, B.K.; Bhattacharjee, S.; Besra, L. The production of a corrosion resistant graphene reinforced composite coating on copper by electrophoretic deposition. Carbon 2013, 61, 47–56. [Google Scholar] [CrossRef]
- Basu, S.; Singh, P.K.; Huang, J.-J.; Wang, Y.-H. Liquid-Phase Deposition of Al2O3 Thin Films on GaN. J. Electrochem. Soc. 2007, 154, H1041–H1046. [Google Scholar] [CrossRef][Green Version]
- Silickas, P.; Valiulis, A.V. Liquid-phase deposition and the properties of thin zirconium dioxide films on different substrates. Mater. Sci. 2008, 44, 211–215. [Google Scholar] [CrossRef]
- Talo, A.; Passiniemi, P.; Forsén, O.; Yläsaari, S. Polyaniline/epoxy coatings with good anti-corrosion properties. Synth. Met. 1997, 85, 1333–1334. [Google Scholar] [CrossRef]
- Li, X.; Yu, X.; Han, Y. Polymer thin films for antireflection coatings. J. Mater. Chem. C 2013, 1, 2266–2285. [Google Scholar] [CrossRef]
- Geim, A.K.; Novoselov, K.S. The rise of graphene. Nat. Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.; Joung, D.; Zhai, L.; Das, S.; Khondaker, S.I.; Seal, S. Graphene based materials: Past, present and future. Prog. Mater. Sci. 2011, 56, 1178–1271. [Google Scholar] [CrossRef]
- Sanjinés, R.; Vâju, C.; Smajda, R.; Mionić, M.; Magrez, A. Electrical properties and applications of carbon based nanocomposite materials: An overview. Surf. Coat. Technol. 2011, 206, 727–733. [Google Scholar] [CrossRef]
- Jeon, I.-Y.; Choi, H.-J.; Bae, S.-Y.; Change, D.W.; Baek, J.-B. Wedging graphite into graphene and graphene-like platelets by dendritic. J. Mater. Chem. 2011, 21, 7820–7826. [Google Scholar] [CrossRef]
- Singh, B.P.; Jena, B.K.; Bhattacharjee, S.; Besra, L. Development of oxidation and corrosion resistance hydrophobic graphene oxide-polymer composite coating on copper. Surf. Coat. Technol. 2013, 232, 475–481. [Google Scholar] [CrossRef]
- Tong, Y.; Bohmb, S.; Song, M. Graphene Based Materials and Their Composites as Coatings. Aust. J. Nanomed. Nanotechnol. 2013, 1, 1003. [Google Scholar]
- Yivlialin, R.; Bussetti, G.; Brambilla, L.; Castiglioni, C.; Tommasini, M.; Duò, L.; Passoni, M.; Ghidelli, M.; Casari, C.S.; Li Bassi, A. Microscopic Analysis of the Different Perchlorate Anions Intercalation Stages of Graphite. J. Phys. Chem. C 2017, 121, 14246–14253. [Google Scholar] [CrossRef]
- Dikin, D.A.; Stankovich, S.; Zimney, E.J.; Piner, R.D.; Dommett, G.H.B.; Evmenenko, G.; Nguyen, S.T.; Ruoff, R.S. Preparation and characterization of graphene oxide paper. Nature 2007, 448, 457–460. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhi, L.; Müllen, K. Transparent, conductive graphene electrodes for dye-sensitized solar cells. Nano Lett. 2008, 8, 323–327. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhang, G.; Bai, X.; Sun, X.; Wang, X.; Wang, E.; Dai, H. Highly conducting graphene sheets and Langmuir–Blodgett films. Nat. Nanotechnol. 2008, 3, 538–542. [Google Scholar] [CrossRef] [PubMed]
- Gilje, S.; Han, S.; Wang, M.; Wang, K.L.; Kaner, R.B. A chemical route to graphene for device applications. Nano Lett. 2007, 7, 3394–3398. [Google Scholar] [CrossRef] [PubMed]
- Tung, V.C.; Allen, M.J.; Yang, Y.; Kaner, R.B. High-throughput solution processing of large-scale graphene. Nat. Nanotechnol. 2009, 4, 25–29. [Google Scholar] [CrossRef] [PubMed]
- Prasai, D.; Tuberquia, J.C.; Harl, R.R.; Jennings, G.K.; Rogers, B.R.; Bolotin, K.I. Graphene: Corrosion-inhibiting coating. ACS Nano 2012, 6, 1102–1108. [Google Scholar] [CrossRef] [PubMed]
- Narula, U.; Tan, C. Determining the Parameters of Importance of a Graphene Synthesis Process Using Design-of-Experiments Method. Appl. Sci. 2016, 6, 204. [Google Scholar] [CrossRef]
- Singh Raman, R.K.; Chakraborty Banerjee, P.; Lobo, D.E.; Gullapalli, H.; Sumandasa, M.; Kumar, A.; Choudhary, L.; Tkacz, R.; Ajayan, P.M.; Majumder, M. Protecting copper from electrochemical degradation by graphene coating. Carbon 2012, 50, 4040–4045. [Google Scholar] [CrossRef]
- Kirkland, N.T.; Schiller, T.; Medhekar, N.; Birbilis, N. Exploring graphene as a corrosion protection barrier. Corros. Sci. 2012, 56, 1–4. [Google Scholar] [CrossRef]
- Wang, G.; Zhang, M.; Zhu, Y.; Ding, G.; Jiang, D.; Guo, Q.; Liu, S.; Xie, X.; Chu, P.K.; Di, Z.; et al. Direct growth of graphene film on germanium substrate. Sci. Rep. 2013, 3, 2465. [Google Scholar] [CrossRef] [PubMed]
- Geckle, R.J.; Mroczkowski, R.S. Corrosion of Precious Metal Plated Copper Alloys Due to Mixed Flowing Gas Exposure. IEEE Trans. Compon. Hybrids Manuf. Technol. 1991, 14, 162–169. [Google Scholar] [CrossRef]
- Chen, S.; Brown, L.; Levendorf, M.; Cai, W.; Ju, S.-Y.; Edgeworth, J.; Li, X.; Magnuson, C.W.; Velamakanni, A.; Piner, R.D.; et al. Oxidation resistance of graphene-coated Cu and Cu Ni alloy. ACS Nano 2011, 5, 1321–1327. [Google Scholar] [CrossRef] [PubMed]
- Yang, L. Coatings for Corrosion Protection of Metals and Alloys. Master’s Thesis, McMaster University, Hamilton, ON, Canada, 2012. [Google Scholar]
- Zhou, X. Graphene Oxidation Barrier Coating. Bachelor’s Thesis, University of Colorado, Boulder, CO, USA, 2011. [Google Scholar]
- Besra, L.; Liu, M. A review on fundamentals and applications of electrophoretic deposition (EPD). Prog. Mater. Sci. 2007, 52, 1–61. [Google Scholar] [CrossRef]
- Besra, L.; Uchikoshi, T.; Suzuki, T.S.; Sakka, Y. Bubble-free aqueous electrophoretic deposition (EPD) by pulse-potential application. J. Am. Ceram. Soc. 2008, 91, 3154–3159. [Google Scholar] [CrossRef]
- Ferrari, B.; Moreno, R. Electrophoretic deposition of aqueous alumina slips. J. Eur. Ceram. Soc. 1997, 17, 549–556. [Google Scholar] [CrossRef]
- An, S.J.; Zhu, Y.; Lee, S.H.; Stoller, M.D.; Emilsson, T.; Park, S.; Velamakanni, A.; An, J.; Ruoff, R.S. Thin film fabrication and simultaneous anodic reduction of deposited graphene oxide platelets by electrophoretic deposition. J. Phys. Chem. Lett. 2010, 1, 1259–1263. [Google Scholar] [CrossRef]
- Park, J.H.; Park, J.M. Electrophoretic deposition of graphene oxide on mild carbon steel for anti-corrosion application. Surface Coat. Technol. 2014, 254, 167–174. [Google Scholar] [CrossRef]
- He, W.; Zhu, L.; Chen, H.; Wang, Y.; Li, W.; Liu, H.; Wang, Y. Electrophoretic deposition of graphene oxide as a corrosion inhibitor for sintered NdFeB. Appl. Surface Sci. 2013, 279, 416–423. [Google Scholar] [CrossRef]
- Singh, D. Electrophoretic Deposition of Graphene on Copper and its Corrosion Behaviour. Master’s Thesis, NIT Rourkela, Odisha, India, 2017. [Google Scholar]
- Raza, M.A.; Rehman, Z.U.; Ghauri, F.A.; Ahmad, A.; Ahmad, R.; Raffi, M. Corrosion study of electrophoretically deposited graphene oxide coatings on copper metal. Thin Solid Films 2016, 620, 150–159. [Google Scholar] [CrossRef]
- Raza, M.A.; Ali, A.; Ghauri, F.A.; Aslam, A.; Yaqoob, K.; Wasay, W.; Raffi, M. Electrochemical behavior of graphene coatings deposited on copper metal by electrophoretic deposition and chemical vapor deposition. Surface Coat. Technol. 2017. In press. [Google Scholar] [CrossRef]
- Al-Sammarraie, A.M.A.; Raheema, M.H. Electrodeposited Reduced Graphene Oxide Films on Stainless Steel, Copper, and Aluminum for Corrosion Protection Enhancement. Int. J. Corros. 2017, 2017, 6939354. [Google Scholar] [CrossRef]
- Diba, M.; Fam, D.W.H.; Boccaccini, A.; Shaffer, M.S.P. Electrophoretic deposition of graphene-related materials: A review of the fundamentals. Prog. Mater. Sci. 2016, 82, 83–117. [Google Scholar] [CrossRef]
- Shahriary, L.; Athawale, A. Graphene Oxide Synthesized by using Modified Hummers Approach. Int. J. Renew. Energy Environ. Eng. 2014, 2, 58–63. [Google Scholar]
- Kang, J.H.; Kim, T.; Choi, J.; Park, J.; Kim, Y.S.; Chang, M.S.; Jung, H.; Park, K.T.; Yang, S.J.; Park, C.R. Hidden Second Oxidation Step of Hummers Method. Chem. Mater. 2016, 28, 756–764. [Google Scholar] [CrossRef]
- Yang, Z.; Zheng, Q.; Qiu, H.; LI, J.; Yang, J. A simple method for the reduction of graphene oxide by sodium borohydride with CaCl2 as a catalyst. New Carbon Mater. 2015, 30, 41–47. [Google Scholar] [CrossRef]
- De, D.; Chakraborty, M.; Majumdar, S.; Giri, S. Bandgap engineering through nanocrystalline magnetic alloy grafting on reduced graphene oxide. Phys. Chem. Chem. Phys. 2014, 16, 19661–19667. [Google Scholar] [CrossRef] [PubMed]
- Lih, E.T.Y.; Zaid, R.B.M.; Ling, T.L.; Chong, K.F. Facile Corrosion Protection Coating from Graphene. Int. J. Chem. Eng. Appl. 2012, 3, 453. [Google Scholar] [CrossRef]
- Sobon, G.; Sotor, J.; Jagiello, J.; Kozinski, R.; Zdrojek, M.; Holdynski, M.; Paletko, P.; Boguslawski, J.; Lipinska, L.; Abramski, K.M. Graphene oxide vs. reduced graphene oxide as saturable absorbers for Er-doped passively mode-locked fiber laser. Opt. Express 2012, 20, 19463–19473. [Google Scholar] [CrossRef] [PubMed]
- Loryuenyong, V.; Totepvimarn, K.; Eimburanapravat, P.; Boonchompoo, W.; Buasri, A. Preparation and Characterization of Reduced Graphene Oxide Sheets via Water-Based Exfoliation and Reduction Methods. Adv. Mater. Sci. Eng. 2013, 2013, 923403. [Google Scholar] [CrossRef]
- Fu, C.; Zhao, G.; Zhang, H.; Li, S. Evaluation and Characterization of Reduced Graphene Oxide Nanosheets as Anode Materials for Lithium-Ion Batteries. Int. J. Electrochem. Sci 2013, 8, 6269–6280. [Google Scholar] [CrossRef]
- Thema, F.T.; Moloto, M.J.; Dikio, E.D.; Nyangiwe, N.N.; Kotsedi, L.; Maaza, M.; Khenfouch, M. Synthesis & characterization of graphene thin films by chemical reduction of exfoliated and intercalated graphite oxide. J. Chem. 2013, 2013, 150536. [Google Scholar] [CrossRef]
- Antony, R.P.; Preethi, L.K.; Gupta, B.; Mathews, T.; Dash, S.; Tyagi, A.K. Efficient electrocatalytic performance of thermally exfoliated reduced graphene oxide-Pt hybrid. Mater. Res. Bull. 2015, 70, 60–67. [Google Scholar] [CrossRef]
- Mišković-Stanković, V.; Jevremović, I.; Jung, I.; Rhee, K. Electrochemical study of corrosion behavior of graphene coatings on copper and aluminum in a chloride solution. Carbon 2014, 75, 335–344. [Google Scholar] [CrossRef]
- ASTM G102-89(2015)e1 Standard Practice for Calculation of Corrosion Rates and Related Information from Electrochemical Measurements; ASTM (American Society for Testing and Materials) International: West Conshohocken, PA, USA, 2015.
- Qi, Y.; Xia, T.; Li, Y.; Duan, L.; Chen, W. Colloidal Stability of Reduced Graphene Oxide Materials Prepared Using Different Reducing Agents. Environ. Sci. Nano 2016, 3, 1062–1071. [Google Scholar] [CrossRef]
- Krishnamoorthy, K.; Ramadoss, A.; Kim, S.-J. Graphene Oxide Nanosheets for Corrosion-Inhibiting Coating. Sci. Adv. Mater. 2013, 5, 406–410. [Google Scholar] [CrossRef]
- Sai Pavan, A.S.; Ramanan, S.R. A study on corrosion resistant graphene films on low alloy steel. Appl. Nanosci. 2016, 6, 1175–1181. [Google Scholar] [CrossRef]


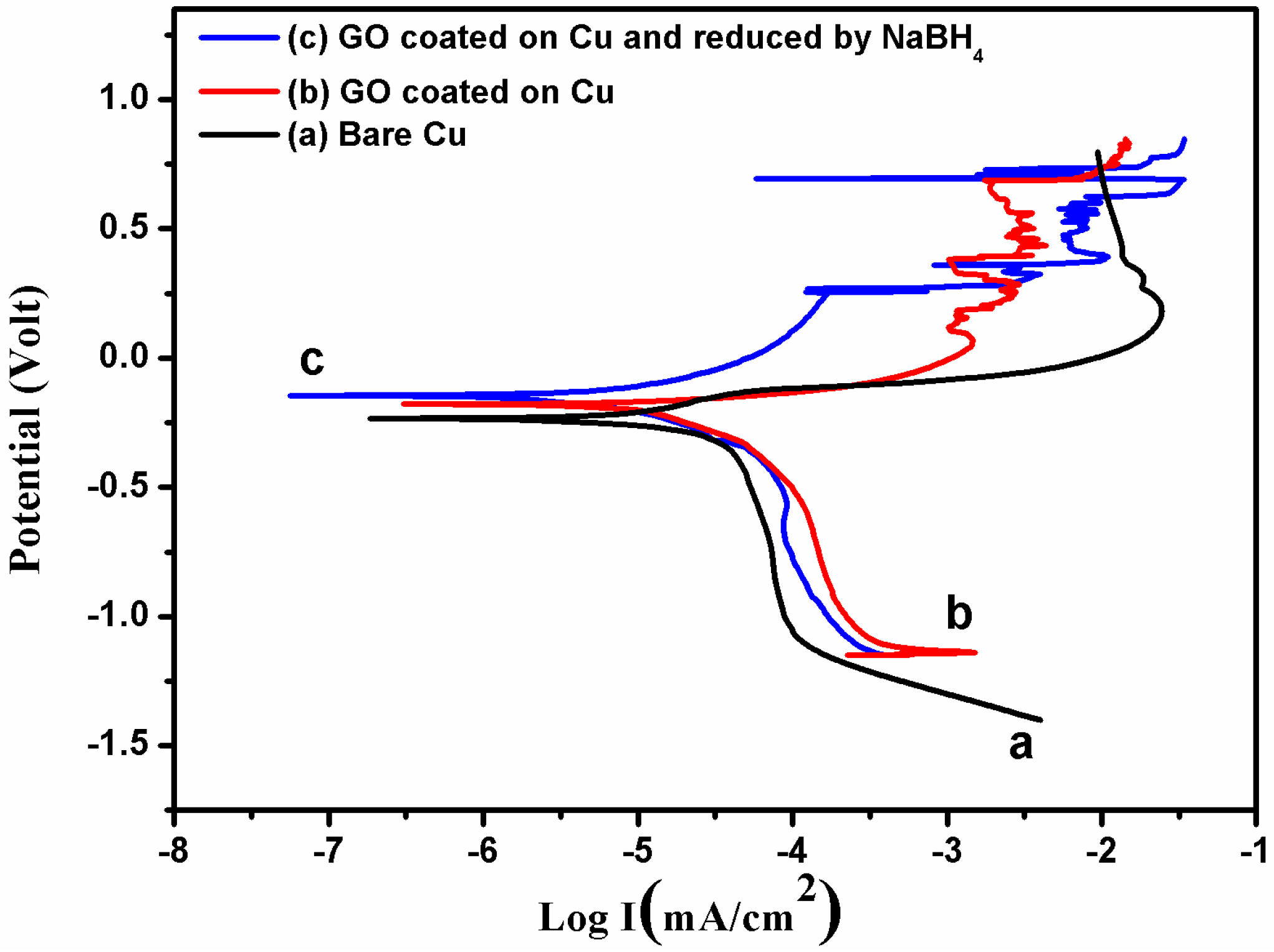

| Sample | Ecorr (mV) | Icorr (µA/cm2) | Corrosion Rate (mm/year) | Inhibition Efficiency (%) |
|---|---|---|---|---|
| Bare copper | −235.3 | 15.375 | 0.18 | – |
| GO Coated copper | −182.2 | 12.44 | 0.15 | 19.08 |
| GO Coated Cu after reduction | −145.4 | 7.01 | 0.08 | 54.40 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Usha Kiran, N.; Dey, S.; Singh, B.P.; Besra, L. Graphene Coating on Copper by Electrophoretic Deposition for Corrosion Prevention. Coatings 2017, 7, 214. https://doi.org/10.3390/coatings7120214
Usha Kiran N, Dey S, Singh BP, Besra L. Graphene Coating on Copper by Electrophoretic Deposition for Corrosion Prevention. Coatings. 2017; 7(12):214. https://doi.org/10.3390/coatings7120214
Chicago/Turabian StyleUsha Kiran, N., Sanjukta Dey, Bimal P. Singh, and Laxmidhar Besra. 2017. "Graphene Coating on Copper by Electrophoretic Deposition for Corrosion Prevention" Coatings 7, no. 12: 214. https://doi.org/10.3390/coatings7120214
APA StyleUsha Kiran, N., Dey, S., Singh, B. P., & Besra, L. (2017). Graphene Coating on Copper by Electrophoretic Deposition for Corrosion Prevention. Coatings, 7(12), 214. https://doi.org/10.3390/coatings7120214





