Inhibitory Properties of Neomycin Thin Film Formed on Carbon Steel in Sulfuric Acid Solution: Electrochemical and AFM Investigation
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Electrochemical Measurements
2.3. Atomic Force Microscopy (AFM) Measurements
3. Results and Discussion
3.1. Potentiodynamic Polarization
3.2. Electrochemical Impedance Spectroscopy (EIS)
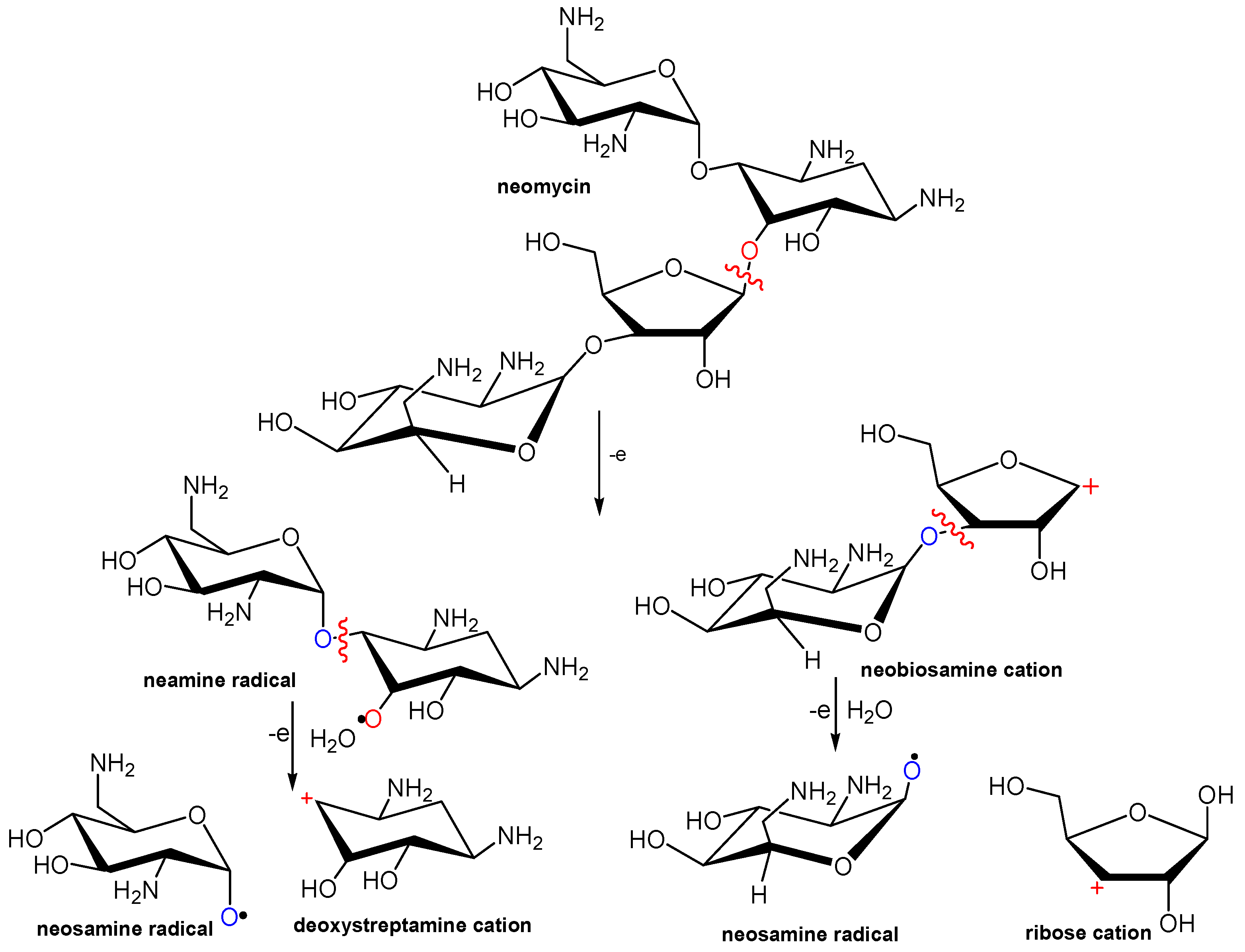
3.3. NMS Adsorption Mechanism
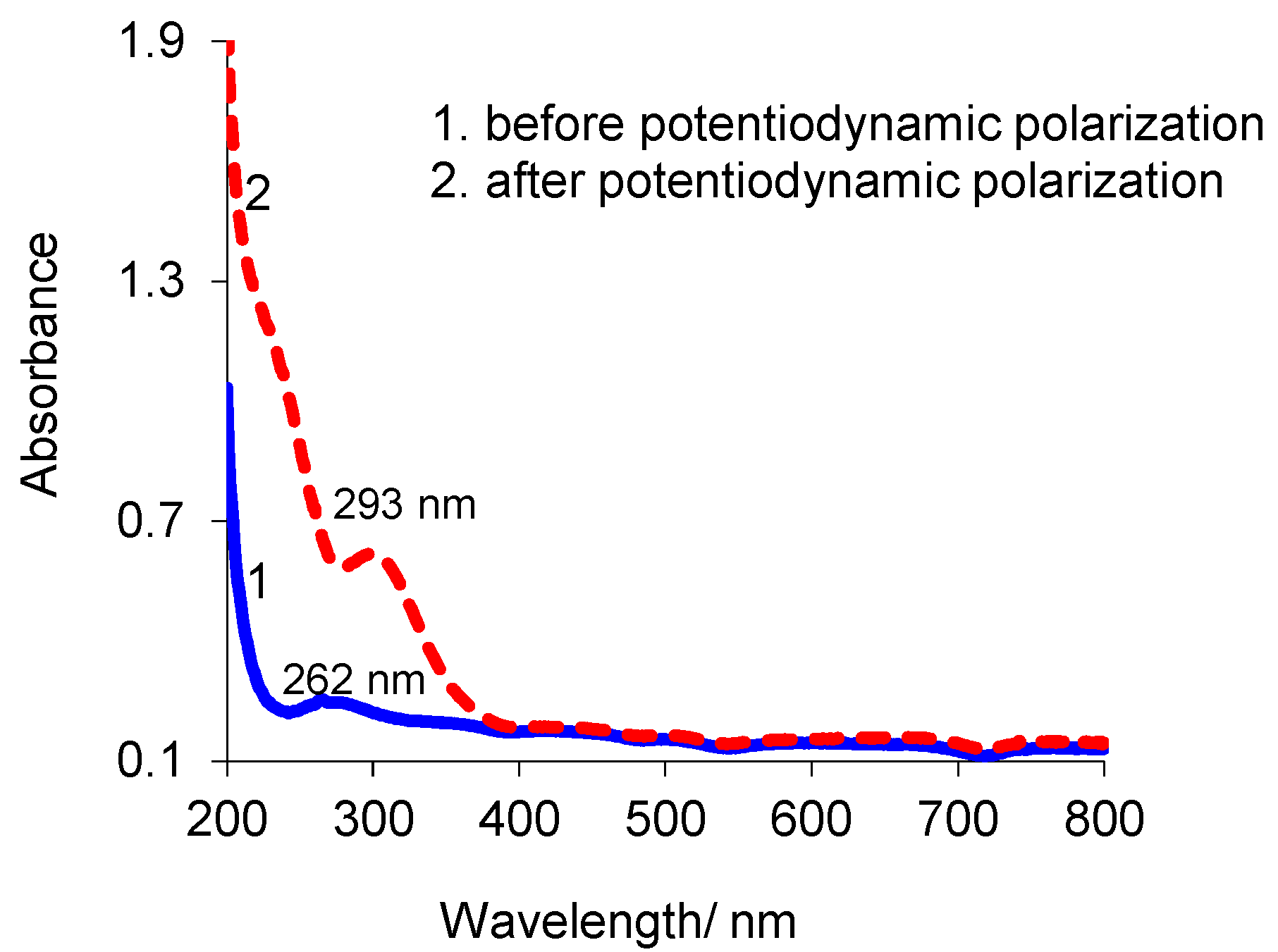
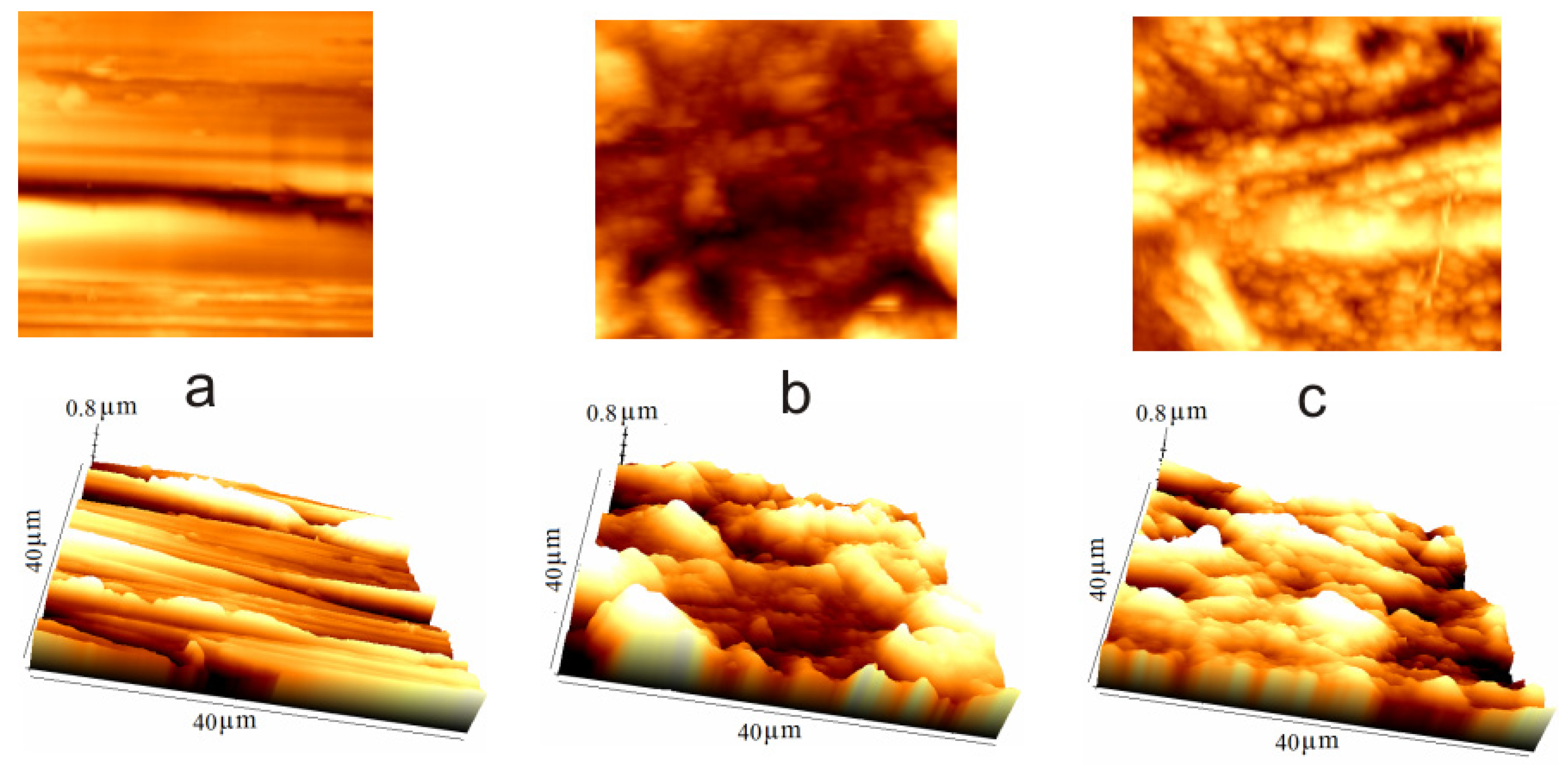
3.4. Atomic Force Microscopy (AFM) Technique
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Guo, L.; Obot, I.B.; Zheng, X.; Shen, X.; Qiang, Y.; Kaya, S.; Kaya, C. Theoretical insight into an empirical rule about organic corrosion inhibitors containing nitrogen, oxygen, and sulfur atoms. Appl. Surf. Sci. 2017, 406, 301–306. [Google Scholar] [CrossRef]
- Samide, A. A pharmaceutical product as corrosion inhibitor for carbon steel in acidic environments. J. Environ. Sci. Health 2013, 48, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Aziz, R.J. Study of some drugs as corrosion inhibitors for mild steel in 1 M H2SO4 solution. Int. J. Curr. Res. Chem. Pharm. Sci. 2016, 3, 1–7. [Google Scholar] [CrossRef]
- Samide, A.; Tutunaru, B. Quinine sulfate: A pharmaceutical product as effective corrosion inhibitor for carbon steel in hydrochloric acid solution. Cent. Eur. J. Chem. 2014, 12, 901–908. [Google Scholar] [CrossRef]
- Samide, A.; Tutunaru, B.; Ionescu, C.; Rotaru, P.; Simoiu, L. Aminophylline: Thermal characterization and its inhibitory properties for the carbon steel corrosion in acidic environment. J. Therm. Anal. Calorim. 2014, 118, 631–639. [Google Scholar] [CrossRef]
- Samide, A.; Tutunaru, B.; Negrilă, C.; Trandafir, I.; Maxut, A. Effect of sulfacetamide on the corrosion products formed onto carbon steel surface in hydrochloric acid. Dig. J. Nanomater. Biostruct. 2011, 6, 663–673. [Google Scholar]
- Samide, A.; Tutunaru, B.; Negrilă, C. Corrosion inhibition of carbon steel in hydrochloric acid solution using a sulfa drug. Chem. Biochem. Eng. Q. 2011, 25, 299–308. [Google Scholar]
- Samide, A.; Tutunaru, B.; Negrilă, C.; Prunaru, I. Surface analysis of inhibitor film formed by 4-amino-N-(1,3-thiazol-2-yl) benzene sulfonamide on carbon steel surface in acidic media. Spectrosc. Lett. 2012, 45, 55–64. [Google Scholar] [CrossRef]
- Gupta, N.K.; Gopal, C.S.A.; Srivastava, V.; Quraishi, M.A. Application of expired drugs in corrosion inhibition of mild steel. Int. J. Pharm. Chem. Anal. 2017, 4, 8–12. [Google Scholar]
- Gopiraman, M.; Sakunthala, P.; Kesavan, D.; Alexramani, V.; Kim, I.S.; Sulochana, N. An investigation of mild carbon steel corrosion inhibition in hydrochloric acid medium by environment friendly green inhibitors. J. Coat. Technol. Res. 2012, 9, 15–26. [Google Scholar] [CrossRef]
- Zhu, G.; Hou, J.; Zhu, H.; Qiu, R.; Xu, J. Electrochemical synthesis of poly(3,4-ethylenedioxythiophene) on stainless steel and its corrosion inhibition performance. J. Coat. Technol. Res. 2013, 10, 659–668. [Google Scholar] [CrossRef]
- Palimi, M.J.; Rostami, M.; Mahdavian, M.; Ramezanzadeh, B. A study on the corrosion inhibition properties of silane-modified Fe2O3 nanoparticle on mild steel and its effect on the anticorrosion properties of the polyurethane coating. J. Coat. Technol. Res. 2015, 12, 277–292. [Google Scholar] [CrossRef]
- Dolabella, L.M.P.; Oliveira, J.G.; Lins, V.; Matencio, T.; Vasconcelos, W.L. Ethanol extract of propolis as a protective coating for mild steel in chloride media. J. Coat. Technol. Res. 2016, 13, 543–555. [Google Scholar] [CrossRef]
- Samide, A.; Tutunaru, B.; Dobrițescu, A.; Ilea, P.; Vladu, A.C.; Tigae, C. Electrochemical and theoretical study of metronidazole drug as inhibitor for copper corrosion in hydrochloric acid solution. Int. J. Electrochem. Sci. 2016, 11, 5520–5534. [Google Scholar] [CrossRef]
- Karthikeyan, S. Drugs/Antibiotics as potential corrosion inhibitors for metals—A review. Int. J. ChemTech Res. 2016, 9, 251–259. [Google Scholar]
- Xhanari, K.; Finsgar, M.; Hrncic, M.K.; Maver, U.; Knez, Z.; Seiti, B. Green corrosion inhibitors for aluminium and its alloys: A review. RSC Adv. 2017, 7, 27299–27330. [Google Scholar] [CrossRef]
- Raja, K.; Jeeva, P.A.; Karthikeyan, S. Reduction of hydrogen embrittlement and green inhibition of stainless steel pipes in acid environment. Int. J. ChemTech. Res. 2015, 7, 2425–2431. [Google Scholar]
- Fouda, A.S.; Elmorsi, M.A.; Fayed, T.A.; Hassan, A.F.; Soltan, M. Corrosion inhibitors based on antibiotic derivatives for protection of carbon steel corrosion in hydrochloric acid solutions. Int. J. Adv. Res. 2014, 2, 788–807. [Google Scholar]
- Chitra, S.; Anand, B. Surface morphological and FTIR spectroscopic information on the corrosion inhibition of drugs on mild steel in chloride environment. J. Chem. Pharm. Sci. 2017, 10, 453–456. [Google Scholar]
- Zerga, B.; Attayibat, A.; Sfaira, M.; Taleb, M.; Hammouti, B.; Ebn Touhami, M.; Radi, S.; Rais, Z. Effect of some tripodal bipyrazolic compounds on C38 steel corrosion in hydrochloric acid solution. J. Appl. Electrochem. 2010, 40, 1575–1582. [Google Scholar] [CrossRef]
- Sobhi, M. Gatitloxacin as corrosion inhibitor for carbon steel in hydrochloric acid solutions. Prot. Met. Phys. Chem. Surf. 2014, 50, 825–883. [Google Scholar] [CrossRef]
- Abdallah, M.; Jahdaly, B.A.AL. Gentamicin, kanamycin and amikacin drugs as non-toxic inhibitors for corrosion of aluminium in l.0 M hydrochloric acid. Int. J. Electrochem. Sci. 2015, 10, 9808–9823. [Google Scholar]
- Samide, A.; Ilea, P.; Vladu, A.C. Metronidazole performance as corrosion inhibitor for carbon steel, 304L stainless steel and aluminium in hydrochloric acid solution. Int. J. Electrochem. Sci. 2017, 12, 5964–5983. [Google Scholar] [CrossRef]
- Bobina, M.; Kellenberger, A.; Millet, J.P.; Muntean, C.; Vaszilcsin, N. Corrosion resistance of carbon steel in weak acid solutions in the presence of l-histidine as corrosion inhibitor. Corros. Sci. 2013, 69, 389–395. [Google Scholar] [CrossRef]
- Nazeer, A.A.; El-Abbasy, H.M.; Fouda, A.S. Adsorption and corrosion inhibition behavior of carbon steel by cefoperazone as eco-friendly inhibitor in HCl. J. Mater. Eng. Perform. 2013, 22, 2314–2322. [Google Scholar]
- Fouda, A.S.; Mostafa, H.A.; El-Abbasy, H.M. Antibacterial drugs as inhibitors for the corrosion of stainless steel type 304 in HCl solution. J. Appl. Electrochem. 2010, 40, 163–173. [Google Scholar] [CrossRef]
- Samide, A.; Bibicu, I. Kinetics corrosion process of carbon steel in hydrochloric acid in absence and presence of 2-(cyclohexylaminomercapto) benzothiazole. Surf. Interface Anal. 2008, 40, 944–952. [Google Scholar] [CrossRef]
- Samide, A.; Rotaru, P.; Ionescu, C.; Tutunaru, B.; Moanţă, A.; Barragan-Montero, V. Thermal behaviour and adsorption properties of some benzothiazole derivatives. J. Therm. Anal. Calorim. 2014, 118, 651–659. [Google Scholar] [CrossRef]
- Clarot, I.; Regazzeti, A.; Auzeil, N.; Laadani, F.; Citton, M.; Netter, P.; Nicolas, A. Analysis of neomycin sulfate and framycetin sulfate by high-performance liquid chromatography using evaporative light scattering detection. J. Chromatogr. A 2005, 1087, 236–244. [Google Scholar] [CrossRef] [PubMed]
- Hanko, V.P.; Rohrer, J.S. Determination of neomycin sulfate and impurities using high-performance anion-exchange chromatography with integrated pulsed amperometric detection. J. Pharm. Biomed. 2007, 43, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Abraha, A.; Gholap, A.V.; Belay, A. Study self-association, optical transition properties and thermodynamic properties of neomycin sulfate using UV-Visible spectroscopy. Int. J. Biophys. 2016, 6, 16–20. [Google Scholar]
- Geetha, M.B.; Rajendran, S. Synergistic Inhibition of Corrosion of Mild Steel in Sulphuric acid by New Ternary System. Der. Pharm. Chem. 2016, 8, 194–201. [Google Scholar]
- Samide, A.; Iacobescu, G.E.; Tutunaru, B.; Tigae, C. Electrochemical and AFM study of inhibitory properties of thin film formed by tartrazine food additive on 304L stainless steel in saline solution. Int. J. Electrochem. Sci. 2017, 12, 2088–2101. [Google Scholar] [CrossRef]
- Samide, A.; Tutunaru, B. Eurovanillin thermal behaviour and its inhibitory properties on carbon steel corrosion in weakly acidic environments. J. Therm. Anal. Calorim. 2017, 127, 863–870. [Google Scholar] [CrossRef]
- Abdel Ghanyl, N.A.; El-Shenawy, A.E.; Hussien, W.A.M. The Inhibitive effect of some amino acids on the corrosion behaviour of 316L stainless steel in sulfuric acid solution. Mod. Appl. Sci. 2011, 5, 19–29. [Google Scholar]
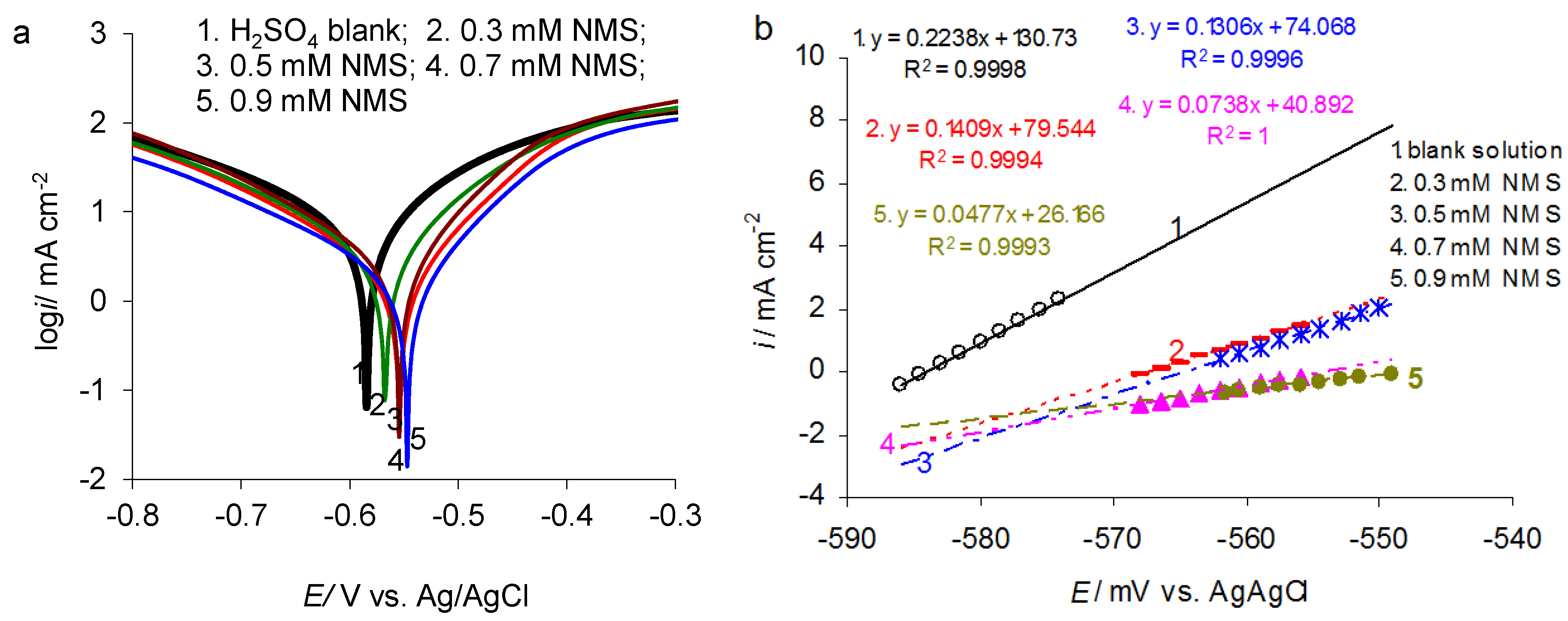
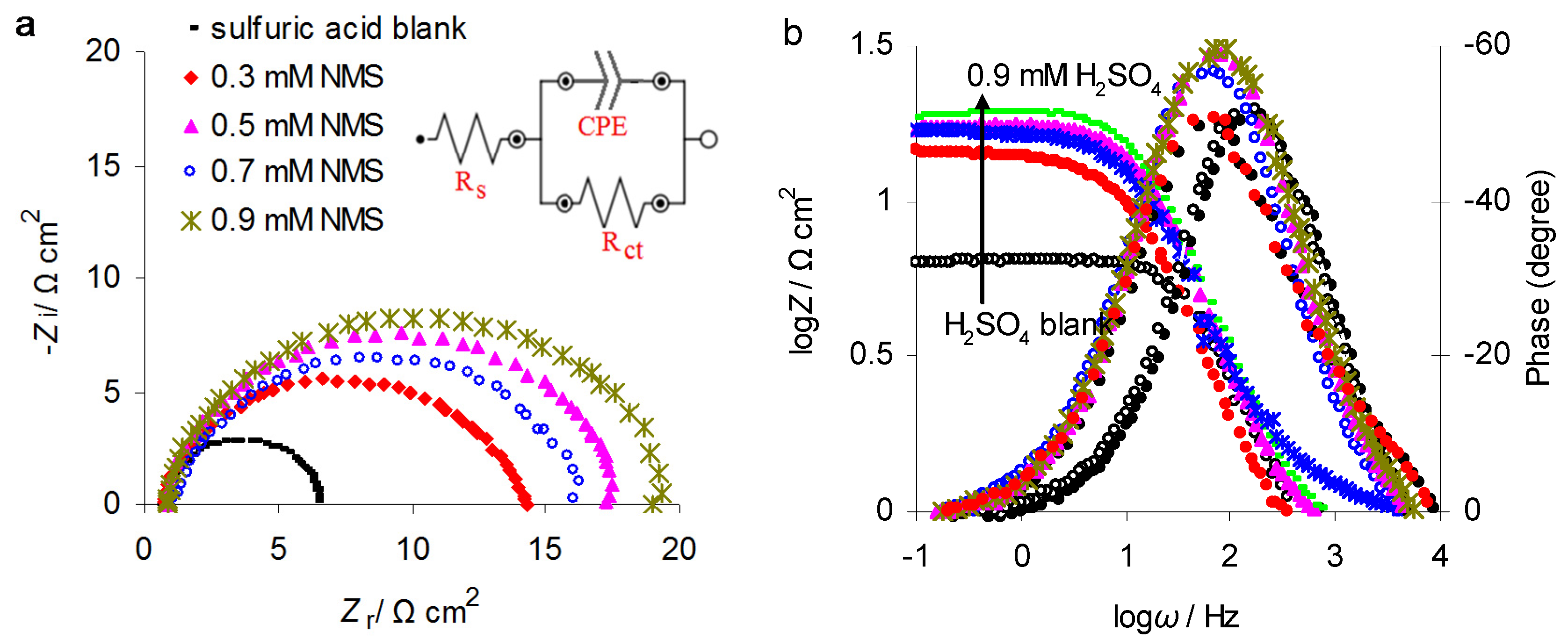
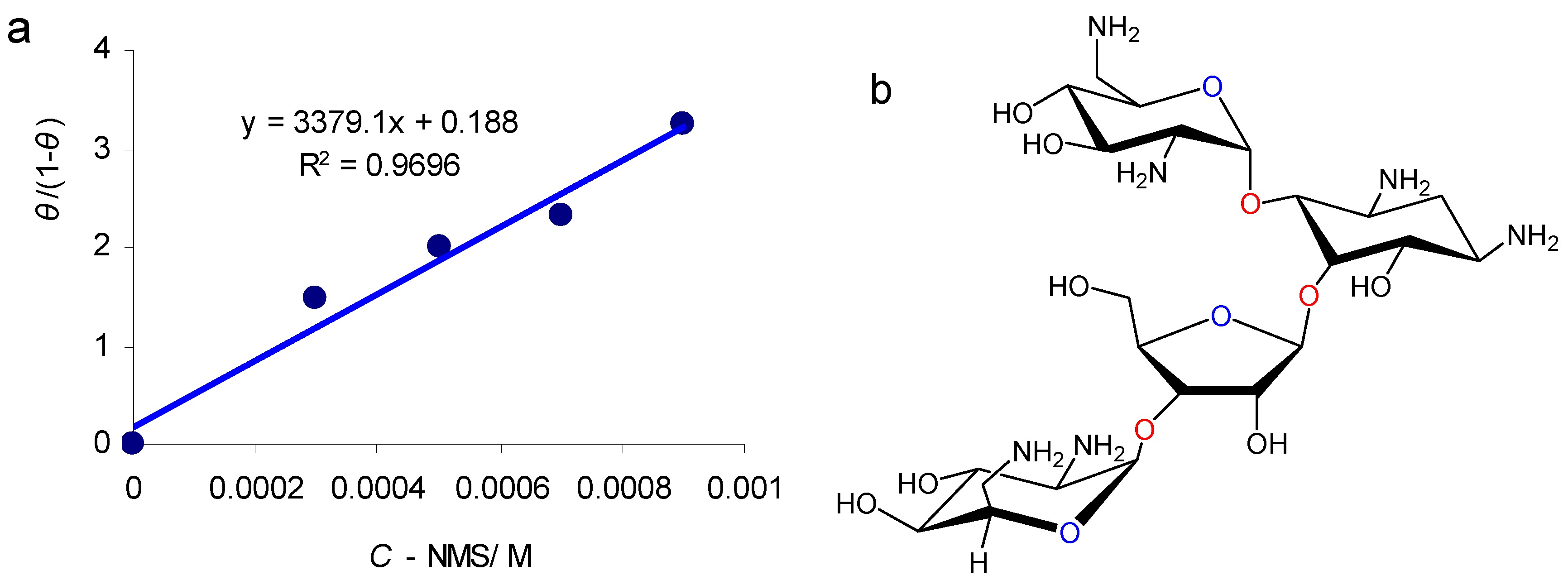
| C-NMS/mM | Ecorr/mV vs. Ag/AgCl | icorr/μA·cm−2 | Cdl/μF·cm−2 | n | Rct/Ω·cm2 | IE/% | ||
|---|---|---|---|---|---|---|---|---|
| From Tafel | From EIS | Average Values | ||||||
| 0 | −584.5 | 1560 | 520 | 0.963 | 5.3 | – | – | – |
| 0.3 | −568.0 | 690 | 290 | 0.981 | 14.4 | 55.8 | 63.2 | 59.5 ± 3.5 |
| 0.5 | −554.5 | 540 | 225 | 0.979 | 16.6 | 65.4 | 68.1 | 66.8 ± 1.3 |
| 0.7 | −554.0 | 460 | 197 | 0.975 | 17.2 | 70.5 | 69.2 | 69.9 ± 07 |
| 0.9 | −547.0 | 320 | 165 | 0.968 | 19.8 | 79.5 | 73.2 | 76.4 ± 3.2 |
| Sample | Rq/nm | Ra/nm | Rp−v/nm |
|---|---|---|---|
| Carbon steel, control sample | 125.1 | 95.6 | 647.4 |
| Carbon steel/1.0 M H2SO4 | 397.0 | 308.0 | 1634.0 |
| Carbon steel/1.0 M H2SO4/0.9 mM NMS | 170.1 | 127.1 | 896.6 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Samide, A.; Iacobescu, G.E.; Tutunaru, B.; Grecu, R.; Tigae, C.; Spînu, C. Inhibitory Properties of Neomycin Thin Film Formed on Carbon Steel in Sulfuric Acid Solution: Electrochemical and AFM Investigation. Coatings 2017, 7, 181. https://doi.org/10.3390/coatings7110181
Samide A, Iacobescu GE, Tutunaru B, Grecu R, Tigae C, Spînu C. Inhibitory Properties of Neomycin Thin Film Formed on Carbon Steel in Sulfuric Acid Solution: Electrochemical and AFM Investigation. Coatings. 2017; 7(11):181. https://doi.org/10.3390/coatings7110181
Chicago/Turabian StyleSamide, Adriana, Gabriela Eugenia Iacobescu, Bogdan Tutunaru, Roxana Grecu, Cristian Tigae, and Cezar Spînu. 2017. "Inhibitory Properties of Neomycin Thin Film Formed on Carbon Steel in Sulfuric Acid Solution: Electrochemical and AFM Investigation" Coatings 7, no. 11: 181. https://doi.org/10.3390/coatings7110181
APA StyleSamide, A., Iacobescu, G. E., Tutunaru, B., Grecu, R., Tigae, C., & Spînu, C. (2017). Inhibitory Properties of Neomycin Thin Film Formed on Carbon Steel in Sulfuric Acid Solution: Electrochemical and AFM Investigation. Coatings, 7(11), 181. https://doi.org/10.3390/coatings7110181






