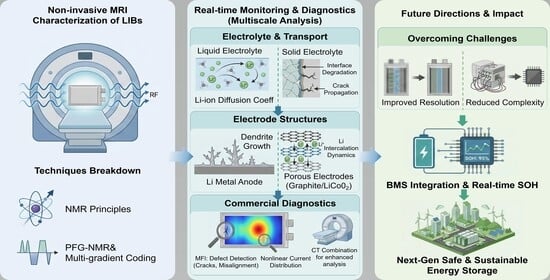
Research Progress of Non-Invasive Magnetic Resonance Imaging in Lithium-Ion Battery Detection
Abstract
1. Introduction
2. Principle of Magnetic Resonance Imaging Technology
2.1. The Basic Principles of Nuclear Magnetic Resonance
2.2. Spatial Resolution Generation of MRI
3. Application of MRI in Lithium-Ion Battery Detection
3.1. Micro-Scale Applications: Intrinsic Properties of Electrolytes and Electrode Materials and Interfacial Processes
3.1.1. Transport Properties and Ionic Distribution of Liquid Electrolytes
3.1.2. Interfacial Evolution and Volume Transport Properties of Solid-State Electrolytes
3.2. Mesoscale Applications: Visualization of Internal Processes in Laboratory Cells
3.3. Macro-Scale Applications: Indirect and Complementary Magnetic Techniques for Commercial-Format Cells
4. Recent Research Advances in MRI Techniques
4.1. Comparative Positioning of MRI Among Major Non-Destructive Diagnostic Techniques
4.2. Core Technical Bottlenecks and Quantitative Interpretation Challenges
4.3. Real-Time Monitoring of Current Distribution
4.4. Diagnosis of Battery Defects
5. Summary
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Britton, M.M. MRI of chemical reactions and processes. Prog. Nucl. Magn. Reson. Spectrosc. 2017, 101, 51–70. [Google Scholar] [CrossRef]
- Feindel, K.W. Spatially resolved chemical reaction monitoring using magnetic resonance imaging. Magn. Reson. Chem. 2016, 54, 429–436. [Google Scholar] [CrossRef]
- Mohammadi, M.; Jerschow, A. In situ and operando magnetic resonance imaging of electrochemical cells: A perspective. J. Magn. Reson. 2019, 308, 106600. [Google Scholar] [CrossRef]
- Schmuch, R.; Wagner, R.; Horpel, G.; Placke, T.; Winter, M. Performance and cost of materials for lithium-based rechargeable automotive batteries. Nat. Energy 2018, 3, 267–278. [Google Scholar] [CrossRef]
- Wu, F.X.; Maier, J.; Yu, Y. Guidelines and trends for next-generation rechargeable lithium and lithium-ion batteries. Chem. Soc. Rev. 2020, 49, 1569–1614. [Google Scholar] [CrossRef]
- Bhattacharyya, R.; Key, B.; Chen, H.; Best, A.S.; Hollenkamp, A.F.; Grey, C.P. In situ NMR observation of the formation of metallic lithium microstructures in lithium batteries. Nat. Mater. 2010, 9, 504–510. [Google Scholar] [CrossRef]
- Xiang, Y.; Tao, M.; Zhong, G.; Liang, Z.; Zheng, G.; Huang, X.; Liu, X.; Jin, Y.; Xu, N.; Armand, M.; et al. Quantitatively analyzing the failure processes of rechargeable Li metal batteries. Sci. Adv. 2021, 7, eabj3423. [Google Scholar] [CrossRef]
- Chen, R.; Jiao, J.; Chen, Z.; Wang, Y.; Deng, T.; Di, W.; Zhu, S.; Gong, M.; Lu, L.; Xie, X.; et al. Power Batteries Health Monitoring: A Magnetic Imaging Method Based on Magnetoelectric Sensors. Materials 2022, 15, 1980. [Google Scholar] [CrossRef]
- Ding, T.; Han, J.; Lv, Y.; Xiao, H.; Li, L. Development of battery test platform for long pulse high magnetic field. Chin. J. Power Sources 2016, 40, 1094–1097. [Google Scholar]
- Green, J.E.; Stone, D.A.; Foster, M.P.; Tennant, A. Spatially Resolved Measurements of Magnetic Fields Applied to Current Distribution Problems in Batteries. IEEE Trans. Instrum. Meas. 2015, 64, 951–958. [Google Scholar] [CrossRef]
- Khanna, R. Generation of magnetic fields by a gravitomagnetic plasma battery. Mon. Not. R. Astron. Soc. 1998, 295, L6–L10. [Google Scholar] [CrossRef][Green Version]
- Khare, N.; Singh, P.; Vassiliou, J.K. A novel magnetic field probing technique for determining state of health of sealed lead-acid batteries. J. Power Sources 2012, 218, 462–473. [Google Scholar] [CrossRef]
- Lee, M.; Shin, Y.; Chang, H.; Jin, D.; Lee, H.; Lim, M.; Seo, J.; Band, T.; Kaufmann, K.; Moon, J.; et al. Diagnosis of Current Flow Patterns Inside Fault-Simulated Li-Ion Batteries via Non-Invasive, In Operando Magnetic Field Imaging. Small Methods 2023, 7, 2300748. [Google Scholar] [CrossRef]
- Martinez, S. Investigating Lithium-Ion Batteries Using Low-Field Nuclear Magnetic Resonance and Relaxometry: A Short Story; University of California: Davis, CA, USA, 2022. [Google Scholar]
- Lin, H.; Jin, Y.; Tao, M.; Zhou, Y.; Shan, P.; Zhao, D.; Yang, Y. Magnetic resonance imaging techniques for lithium-ion batteries: Principles and applications. Magn. Reson. Lett. 2024, 4, 200113. [Google Scholar] [CrossRef]
- Li, Y.; Ye, M.; Wang, Q.; Lian, G.; Xia, B. An improved model combining machine learning and Kalman filtering architecture for state of charge estimation of lithium-ion batteries. Green Energy Intell. Transp. 2024, 3, 100163. [Google Scholar] [CrossRef]
- Yu, C.; Zhu, J.; Liu, W.; Dai, H.; Wei, X. Co-estimation of state-of-charge and state-of-temperature for large-format lithium-ion batteries based on a novel electrothermal model. Green Energy Intell. Transp. 2024, 3, 100152. [Google Scholar] [CrossRef]
- Zhu, J.; Weng, W.; You, H.; Zhang, J.; Wang, Y.; Jiang, B.; Ji, C.; Wei, X.; Dai, H. Lithium-ion battery end of life prediction based on the decelerating aging point. Appl. Energy 2025, 401, 126692. [Google Scholar] [CrossRef]
- Eto, A.; Akimoto, Y.; Okajima, K.; Okano, J.; Onoue, Y. Evaluation of lithium-ion batteries with different structures using magnetic field measurement for onboard battery identification. Green Energy Intell. Transp. 2025, 4, 100257. [Google Scholar] [CrossRef]
- Zhao, S.; Sun, X.; An, Y.; Guo, Z.; Li, C.; Xu, Y.; Li, Y.; Li, Z.; Zhang, X.; Wang, K.; et al. Lithium plating accurate detection of lithium-ion capacitors upon high-rate charging. Green Energy Intell. Transp. 2025, 4, 100268. [Google Scholar] [CrossRef]
- Liao, L.; Koettig, F. A hybrid framework combining data-driven and model-based methods for system remaining useful life prediction. Appl. Soft Comput. 2016, 44, 191–199. [Google Scholar] [CrossRef]
- Romanenko, K.; Kuchel, P.W.; Jerschow, A. Accurate Visualization of Operating Commercial Batteries Using Specialized Magnetic Resonance Imaging with Magnetic Field Sensing. Chem. Mater. 2020, 32, 2107–2113. [Google Scholar] [CrossRef]
- Haase, A.; Frahm, J.; Matthaei, D.; Hanicke, W.; Merboldt, K.D. FLASH imaging. Rapid NMR imaging using low flip-angle pulses. J. Magn. Reson. (1969) 1986, 67, 258–266. [Google Scholar] [CrossRef]
- Ruan, G.; Hua, J.; Hu, X.; Yu, C. Effect of magnetic field on the lithium-ion battery performance. Energy Storage Sci. Technol. 2022, 11, 265–274. [Google Scholar]
- Ruan, G.; Hua, J.; Hu, X.; Yu, C. Study on the influence of magnetic field on the performance of lithium-ion batteries. Energy Rep. 2022, 8, 1294–1304. [Google Scholar] [CrossRef]
- Grey, C.P.; Dupré, N. NMR studies of cathode materials for lithium-ion rechargeable batteries. Chem. Rev. 2004, 104, 4493–4512. [Google Scholar] [CrossRef]
- Gallagher, T.A.; Nemeth, A.J.; Hacein-Bey, L. An Introduction to the Fourier Transform: Relationship to MRI. Am. J. Roentgenol. 2008, 190, 1396–1405. [Google Scholar] [CrossRef]
- Bernstein, M.A.; Huston, J., III; Ward, H.A. Imaging artifacts at 3. Reson. Imaging 2006, 24, 735–746. [Google Scholar] [CrossRef]
- Brateman, L. Chemical shift imaging: A review. Am. J. Roentgenol. 1986, 146, 971–980. [Google Scholar] [CrossRef] [PubMed]
- Moser, E.; Laistler, E.; Schmitt, F.; Kontaxis, G. Ultra-High Field NMR and MRI-The Role of Magnet Technology to Increase Sensitivity and Specificity. Front. Phys. (In English) 2017, 5, 33. [Google Scholar]
- Singh, D.; Monga, A.; de Moura, H.L.; Zhang, X.; Zibetti, M.V.W.; Regatte, R.R. Emerging Trends in Fast MRI Using Deep-Learning Reconstruction on Undersampled k-Space Data: A Systematic Review. Bioengineering 2023, 10, 1012. [Google Scholar] [CrossRef]
- Winkler, S.A.; Schmitt, F.; Landes, H.; de Bever, J.; Wade, T.; Alejski, A.; Rutt, B.K. Gradient and shim technologies for ultra high field MRI. NeuroImage 2018, 168, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Gudino, N.; Littin, S. Advancements in Gradient System Performance for Clinical and Research MRI. J. Magn. Reson. Imaging 2023, 57, 57–70. [Google Scholar] [CrossRef]
- Hori, S.; Taminato, S.; Suzuki, K.; Hirayama, M.; Kato, Y.; Kanno, R. Structure-property relationships in lithium superionic conductors having a Li10GeP2S12-type structure. Acta Crystallogr. Sect. B-Struct. Sci. Cryst. Eng. Mater. 2015, 71, 727–736. [Google Scholar] [CrossRef]
- Liu, X.; Liang, Z.; Xiang, Y.; Lin, M.; Li, Q.; Liu, Z.; Zhong, G.; Fu, R.; Yang, Y. Solid-State NMR and MRI Spectroscopy for Li/Na Batteries: Materials, Interface, and In Situ Characterization. Adv. Mater. 2021, 33, 2005878. [Google Scholar] [CrossRef]
- McEvoy, T.M.; Stevenson, K.J. Spatially resolved imaging of inhomogeneous charge transfer behavior in polymorphous molybdenum oxide. II. Correlation of localized coloration/insertion properties using spectroelectrochemical microscopy. Langmuir 2005, 21, 3529–3538. [Google Scholar] [CrossRef]
- Mohammadi, M. Characterization of Li-Ion Batteries and Their Components Using Nuclear Magnetic Resonance Spectroscopy and Imaging; New York University: New York, NY, USA, 2020. [Google Scholar]
- Wang, G.; Jiang, B.; Liu, Y.; Wang, L.; Zhang, Y.; Yan, J.; Wang, K. Source-Load Coordinated Optimization Framework for Distributed Energy Systems Using Quasi-Potential Game Method. Prot. Control Mod. Power Syst. 2025, 10, 103–122. [Google Scholar] [CrossRef]
- Jiang, W.; Wang, J.; Guo, R.; Wang, J.; Song, J.; Wang, K. Electrode Materials and Prediction of Cycle Stability and Remaining Service Life of Supercapacitors. Coatings 2026, 16, 41. [Google Scholar] [CrossRef]
- Jiang, W.; Tan, C.; Su, E.; Lu, J.; Shi, H.; Wang, Y.; Song, J.; Wang, K. Advanced Electronic Materials for Liquid Thermal Management of Lithium-Ion Batteries: Mechanisms, Materials and Future Development Directions. Coatings 2026, 16, 59. [Google Scholar] [CrossRef]
- Lou, C.; Zhang, J.; Mu, X.; Zeng, F.; Wang, K. Innovative deep learning method for predicting the state of health of lithium-ion batteries based on electrochemical impedance spectroscopy and attention mechanisms. Front. Chem. Sci. Eng. 2025, 19, 52. [Google Scholar] [CrossRef]
- Li, J.; Li, Y.; Wang, Y.; Wang, X.; Wang, P.; Ci, L.; Liu, Z. Preparation, design and interfacial modification of sulfide solid electrolytes for all-solid-state lithium metal batteries. Energy Storage Mater. 2025, 74, 103962. [Google Scholar] [CrossRef]
- Thienenkamp, J.H.; Lennartz, P.; Winter, M.; Brunklaus, G. Experimental correlation of anionic mass transport and lithium dendrite growth in solid-state polymer-based lithium metal batteries. Cell Rep. Phys. Sci. 2024, 5, 102340. [Google Scholar] [CrossRef]
- Akchach, A.; Bayle, P.A.; Buzlukov, A.; Chandesris, M.; Genies, S.; Bardet, M. Interplay between Lithium Intercalation and Plating during Fast Charging of Lithium-Ion Batteries Investigated by Operando NMR Spectroscopy. Batter. Supercaps 2025, 8, e202500208. [Google Scholar] [CrossRef]
- Liu, M.; Song, A.; Zhang, X.; Wang, J.; Fan, Y.; Wang, G.; Tian, H.; Ma, Z.; Shao, G. Interfacial Lithium-Ion Transportation in Solid-State Batteries: Challenges and Prospects. Nano Energy 2025, 136, 110749. [Google Scholar] [CrossRef]
- Shan, P.; Chen, J.; Tao, M.; Zhao, D.; Lin, H.; Fu, R.; Yang, Y. The applications of solid-state NMR and MRI techniques in the study of rechargeable sodium-ion batteries. J. Magn. Reson. 2023, 353, 107516. [Google Scholar] [CrossRef]
- Chandrashekar, S.; Trease, N.M.; Chang, H.J.; Du, L.-S.; Grey, C.P.; Jerschow, A. 7Li MRI of Li batteries reveals location of microstructural lithium. Nat. Mater. 2012, 11, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Shan, W.; He, Q.; Cao, Z.; Zhang, Z.; Dai, Z.; Zheng, L.; Li, X. Numerical study of multi-nozzle inlet structure optimization for immersion cooling systems of large-scale lithium-ion battery pack. J. Energy Storage 2026, 153, 120872. [Google Scholar] [CrossRef]
- Markert, A.; Morales, M.; Guntlin, C.; Nirschl, H.; Guthausen, G. Simultaneous Electrochemical Impedance Spectroscopy and Magnetic Resonance Imaging analysis of lithium-ion batteries. Int. J. Electrochem. Sci. 2025, 20, 101129. [Google Scholar] [CrossRef]
- Leifer, N.; Aurbach, D.; Greenbaum, S.G. NMR studies of lithium and sodium battery electrolytes. Prog. Nucl. Magn. Reson. Spectrosc. 2024, 142–143, 1–54. [Google Scholar] [CrossRef]
- Mohammadi, M.; Silletta, E.V.; Ilott, A.J.; Jerschow, A. Diagnosing current distributions in batteries with magnetic resonance imaging. J. Magn. Reson. 2019, 309, 106601. [Google Scholar] [CrossRef]
- Zia, A.W.; Rasul, S.; Asim, M.; Samad, Y.A.; Shakoor, R.A.; Masood, T. The potential of plasma-derived hard carbon for sodium-ion batteries. J. Energy Storage 2024, 84, 110844. [Google Scholar] [CrossRef]
- Romanenko, K.; Jerschow, A. Numerical modeling of Surface-Scan MRI experiments for improved diagnostics of commercial battery cells. J. Magn. Reson. Open 2022, 10–11, 100061. [Google Scholar] [CrossRef]
- Griffith, K.J.; Griffin, J.M. Solid-state NMR of energy storage materials. In Comprehensive Inorganic Chemistry III, 3rd ed.; Reedijk, J., Poeppelmeier, K.R., Eds.; Elsevier: Oxford, UK, 2023; pp. 282–329. [Google Scholar]
- Aguilera, A.R.; Marica, F.; Sanders, K.J.; Al Raihan, M.; Dyker, C.A.; Goward, G.R.; Balcom, B.J. Multinuclear MR and MRI study of lithium-ion cells using a variable field magnet and a fixed frequency RF probe. Magn. Reson. Lett. 2024, 4, 100090. [Google Scholar] [CrossRef] [PubMed]
- Santos, É.A.; Amaral, M.M.; Damasceno, B.S.; Da Silva, L.M.; Zanin, H.G.; Weker, J.N.; Rodella, C.B. Advanced in situ/operando characterizations of lithium-sulfur batteries: A sine qua non. Nano Energy 2024, 130, 110098. [Google Scholar] [CrossRef]
- Bai, X.; Yu, T.; Ren, Z.; Gong, S.; Yang, R.; Zhao, C. Key issues and emerging trends in sulfide all solid state lithium battery. Energy Storage Mater. 2022, 51, 527–549. [Google Scholar] [CrossRef]
- Baek, J.; Kim, S.; Kim, H.-T.; Han, O.H. Postmortem 7Li NMR analysis for assessing the reversibility of lithium metal electrodes in lithium metal batteries. J. Energy Chem. 2024, 94, 430–440. [Google Scholar] [CrossRef]
- Peklar, R.; Mikac, U.; Serša, I. Observation of Electroplating in a Lithium-Metal Battery Model Using Magnetic Resonance Microscopy. Molecules 2025, 30, 2733. [Google Scholar] [CrossRef]
- Peklar, R.; Mikac, U.; Serša, I. The Effect of Battery Configuration on Dendritic Growth: A Magnetic Resonance Microscopy Study on Symmetric Lithium Cells. Batteries 2024, 10, 165. [Google Scholar] [CrossRef]
- Romanenko, K.; Avdievich, N. Unilateral RF sensors based on parallel-plate architecture for improved surface-scan MRI analysis of commercial pouch cells. J. Magn. Reson. Open 2024, 18, 100141. [Google Scholar] [CrossRef]
- Wang, Y.; Lai, X.; Chen, Q.; Han, X.; Lu, L.; Ouyang, M.; Zheng, Y. Progress and challenges in ultrasonic technology for state estimation and defect detection of lithium-ion batteries. Energy Storage Mater. 2024, 69, 103430. [Google Scholar] [CrossRef]
- Cleland, M.J. Rechargeable lithium batteries for medical applications. In Rechargeable Lithium Batteries; Franco, A.A., Ed.; Woodhead Publishing: Cambridge, UK, 2015; pp. 353–367. [Google Scholar]
- Brauchle, F.; Grimsmann, F.; von Kessel, O.; Birke, K.P. Defect detection in lithium ion cells by magnetic field imaging and current reconstruction. J. Power Sources 2023, 558, 232587. [Google Scholar] [CrossRef]
- Biller, J.R.; Delluva, A.; Finch, K. Magnetic Resonance and Magnetometry: Complimentary Tools for Probing Different Size Scales in Lithium-Ion Batteries. Appl. Magn. Reson. 2025, 56, 9–31. [Google Scholar] [CrossRef]
- Tao, M.; Chen, J.; Lin, H.; Zhou, Y.; Zhao, D.; Shan, P.; Jin, Y.; Yang, Y. Recent advances in quantifying the inactive lithium and failure mechanism of Li anodes in rechargeable lithium metal batteries. J. Energy Chem. 2024, 96, 226–248. [Google Scholar] [CrossRef]
- Wang, W.; Yu, T.; Cheng, Y.; Lei, X.; Wang, B.; Guo, R.; Liu, X.; You, J.; Wang, X.; Zhang, H. Field-assisted metal-air batteries: Recent progress, mechanisms, and challenges. Nano Energy 2024, 125, 109550. [Google Scholar] [CrossRef]
- Ogata, K.; Salager, E.; Kerr, C.; Fraser, A.; Ducati, C.; Morris, A.; Hofmann, S.; Grey, C. Revealing lithium–silicide phase transformations in nano-structured silicon-based lithium ion batteries via in situ NMR spectroscopy. Nat. Commun. 2014, 5, 3217. [Google Scholar] [CrossRef]
- Shi, H.; Chu, F.; Zhang, Y.; Liu, Y.; Hou, L.; Li, X.; Yuan, C. Anode-less Li-based metal batteries and beyond: Challenges, strategies, and prospects. Mater. Today 2025, 83, 446–483. [Google Scholar] [CrossRef]
- Zhao, H.; Zhan, Z.; Cui, B.; Wang, Y.; Yin, G.; Han, G.; Xiang, L.; Du, C. Non-destructive detection techniques for lithium-ion batteries based on magnetic field characteristics-A model-based study. J. Power Sources 2024, 604, 234511. [Google Scholar] [CrossRef]
- Gou, Y.; Yan, Y.; Lyu, Y.; Chen, S.; Li, J.; Liu, Y. Advances in acoustic techniques for evaluating defects and properties in lithium-ion batteries: A review. Ultrasonics 2024, 142, 107400. [Google Scholar] [CrossRef]
- Ma, S.; Sun, B.; Chen, X.; Zhang, X.; Zhang, X.; Zhang, W.; Ruan, H.; Zhao, X. Machine learning and feature engineering-based anode potential estimation method for lithium-ion batteries with application. J. Energy Storage 2024, 103, 114387. [Google Scholar] [CrossRef]
- Meddings, N.; Heinrich, M.; Overney, F.; Lee, J.-S.; Ruiz, V.; Napolitano, E.; Seitz, S.; Hinds, G.; Raccichini, R.; Gaberšček, M.; et al. Application of electrochemical impedance spectroscopy to commercial Li-ion cells: A review. J. Power Sources 2020, 480, 228742. [Google Scholar] [CrossRef]
- Gunnarsdóttir, A.B.; Amanchukwu, C.V.; Menkin, S.; Grey, C.P. Noninvasive in situ NMR study of “dead lithium” formation and lithium corrosion in full-cell lithium metal batteries. J. Am. Chem. Soc. 2020, 142, 20814–20827. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Yao, S.; Xu, K.; Wang, K. Blockchain and IoT-Driven Sustainable Battery Recycling: Integration and Challenges. CHAIN 2025, 2, 81–103. [Google Scholar] [CrossRef]
- Lei, M.; Zhang, M.; Wang, K. Research on bidding optimization strategy for virtual power plants with wind–solar–storage systems based on IGDT–DRO. Electr. Eng. 2026, 108, 208. [Google Scholar] [CrossRef]
- Xiang, Y.; Li, X.; Cheng, Y.; Sun, X.; Yang, Y. Advanced characterization techniques for solid state lithium battery research. Mater. Today 2020, 36, 139–157. [Google Scholar] [CrossRef]
- Zhang, G.; Wei, X.; Tang, X.; Zhu, J.; Chen, S.; Dai, H. Internal short circuit mechanisms, experimental approaches and detection methods of lithium-ion batteries for electric vehicles: A review. Renew. Sustain. Energy Rev. 2021, 141, 110790. [Google Scholar] [CrossRef]
- Li, J.; Wang, Q. Multi-modal bioelectrical signal fusion analysis based on different acquisition devices and scene settings: Overview, challenges, and novel orientation. Inf. Fusion 2022, 79, 229–247. [Google Scholar] [CrossRef]
- Durmaz, A.R.; Müller, M.; Lei, B.; Thomas, A.; Britz, D.; Holm, E.A.; Eberl, C.; Mücklich, F.; Gumbsch, P. A deep learning approach for complex microstructure inference. Nat. Commun. 2021, 12, 6272. [Google Scholar] [CrossRef]
- Fritz, F.J.; Sengupta, S.; Harms, R.L.; Tse, D.H.; Poser, B.A.; Roebroeck, A. Ultra-high resolution and multi-shell diffusion MRI of intact ex vivo human brains using kT-dSTEAM at 9.4T. NeuroImage 2019, 202, 116087. [Google Scholar] [CrossRef] [PubMed]
- Anoardo, E.; Rodriguez, G.G. New challenges and opportunities for low-field MRI. J. Magn. Reson. Open 2023, 14–15, 100086. [Google Scholar] [CrossRef]
- Chen-Wiegart, Y.-C.K. Multimodal and Operando Synchrotron X-ray Characterization for Advanced Energy Materials. Microsc. Microanal. 2024, 30, ozae044.226. [Google Scholar] [CrossRef]
- Ziesche, R.F.; Kardjilov, N.; Kockelmann, W.; Brett, D.J.L.; Shearing, P.R. Neutron imaging of lithium batteries. Joule 2022, 6, 35–52. [Google Scholar] [CrossRef]
- Yang, H.; Guo, C.; Naveed, A.; Lei, J.; Yang, J.; Nuli, Y.; Wang, J. Recent progress and perspective on lithium metal anode protection. Energy Storage Mater. 2018, 14, 199–221. [Google Scholar] [CrossRef]
- Britton, M.M.; Bayley, P.M.; Howlett, P.C.; Davenport, A.J.; Forsyth, M. In Situ, Real-Time Visualization of Electrochemistry Using Magnetic Resonance Imaging. J. Phys. Chem. Lett. 2013, 4, 3019–3023. [Google Scholar] [CrossRef][Green Version]
- Li, Y.; Yang, M.; Bian, T.; Wu, H. MRI Super-Resolution Analysis via MRISR: Deep Learning for Low-Field Imaging. Information 2024, 15, 655. [Google Scholar] [CrossRef]
- Lv, X.; Cui, S.; Wang, Y.; Lu, J.; Yu, P.; Wang, K. Patch Time Series Transformer−Based Short−Term Photovoltaic Power Prediction Enhanced by Artificial Fish. Energies 2026, 19, 284. [Google Scholar] [CrossRef]
- Höltschi, L.; Borca, C.N.; Huthwelker, T.; Marone, F.; Schlepütz, C.M.; Pelé, V.; Jordy, C.; Villevieille, C.; Kazzi, M.E.; Novák, P. Performance-limiting factors of graphite in sulfide-based all-solid-state lithium-ion batteries. Electrochim. Acta 2021, 389, 138735. [Google Scholar] [CrossRef]















| Technique | Primary Measurable Parameters | Typical Spatial Resolution | Typical Temporal Resolution | Applicable Scale | Key Advantages | Key Limitations |
|---|---|---|---|---|---|---|
| 7Li/1H MRI [22,23,24,25] | Li+/H+ concentration distribution, chemical environment mapping | 10–100 μm | Minutes–Hours | Micro-, Meso-scale | Chemical specificity, 3D spatial encoding, non-invasive. | Low intrinsic sensitivity for 7Li; relaxation-dependent quantification; severe artifacts near metal. |
| PFG-NMR [37] | Ionic self-diffusion coefficients, transport properties. | N/A (bulk measurement) | Seconds–Minutes | Micro-scale (bulk properties) | Highly accurate for diffusion coefficients; model-free for self-diffusion. | No inherent spatial resolution; requires correlation with electrochemical models for applied fields |
| CSI [1] | Spatially resolved chemical spectra, distribution of different nuclei/species. | 100 μm–1 mm | Hours | Meso-scale | Simultaneous spatial and spectroscopic information; identifies chemical states. | Very long acquisition times; low signal-to-noise ratio; limited spatial resolution. |
| MFI/Surface-Scan MRI [37] | External magnetic field perturbations, internal current density reconstruction, defect location | 1–10 mm; limited depth resolution | Seconds–Minutes | Macro-scale | Fast, truly non-invasive, compatible with commercial packaged cells; no strong magnetic field required. | Poor depth resolution; current reconstruction is an ill-posed inverse problem. |
| Operando MRI [64] | Dynamic processes: ion transport, plating/stripping, phase transformations. | 10–500 μm | Minutes–Hours | All scales | Provides direct in situ correlation between structure/chemistry and performance. | Complex cell design required; trade-off between temporal resolution and data quality. |
| Technique | Spatial Resolution | Temporal Resolution/Scan Time | Relative Cost | Key Advantages | Key Limitations |
|---|---|---|---|---|---|
| Direct MRI/operando NMR-MRI [6] | 10–100 μm | Minutes to hours | High | Non-invasive, operando, chemical specificity (e.g., 7Li speciation), quantitative ion transport | Low sensitivity to 7Li, long acquisition, artifacts in conductive cells |
| X-ray Computed Tomography [8] | <1–10 μm | Seconds to minutes | High | High resolution, 3D structural imaging, fast | Radiation damage risk, limited chemical contrast, ex situ preference |
| Neutron Imaging/Tomography [1] | 10–100 μm | Minutes to hours | Very High (facility-based) | Excellent lithium contrast, deep penetration | Limited access, low temporal resolution, radiation |
| Ultrasonic Imaging/Acoustics [71] | 50–500 μm | Seconds to minutes | Low–Medium | Fast, low-cost, sensitive to defects/gas evolution | Lower resolution, limited chemical info, interpretation challenges |
| Magnetic Field Imaging (MFI) [37] | Macroscopic (~mm) | Seconds | Medium | Fast, non-invasive defect/current mapping | Poor spatial resolution, surface-sensitive only |
| Electrochemical Impedance Spectroscopy (EIS) [73] | None (bulk) | Seconds to minutes | Low | Fast, sensitive to interfaces/kinetics | No spatial information, model-dependent |
| Reference | Technique | Key Finding | Research Scale |
|---|---|---|---|
| J. E. Green et al., 2015 [10] | 1D 7Li MRI + Modeling | First visualization of Li+ concentration gradient under polarization; enabled transference number determination. | Micro (Electrolyte) |
| Chandrashekar et al., 2012 [47] | 7Li MRI | Localized high-surface-area “mossy” lithium formation on anode due to skin effect. | Micro (Interface) |
| R. Khanna et al., 1998 [11] | 3D 7Li MRI (FLASH) | Revealed microscopic inhomogeneity and mossy morphology of Li metal deposits. | Micro/Meso (Electrode) |
| Tang et al., 2019 [78] | STRAFI NMR | Mapped Li+ intercalation kinetics and concentration gradients in thick electrodes. | Meso (Electrode) |
| Britton et al., 2013 [86] | MRI relaxation maps | In situ, real-time visualization of Zn(OH)xy−, OH−, and Zn distribution in an alkaline zinc electrochemical cell during discharge; demonstrated species-specific gradients and electrochemical processes | Micro (Electrolyte/Interface) |
| Romanenko et al., 2020 [22] | Surface-Scan MRI | Achieved non-destructive imaging and diagnosis of internal state in packaged commercial cells. | Macro (Full Cell) |
| Mohammadi et al., 2020 [37] | MFI (based on 1H MRI) | Mapped nonlinear, SOC-dependent internal current distribution in commercial pouch cells. | Macro (Full Cell) |
| Brauchle et al., 2023 [64] | MFI (based on AMR sensors) | Enabled non-invasive detection and localization of manufacturing defects. | Macro (Quality Control) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Jiang, W.; Deng, Y.; Li, W.; Song, J.; Che, S.; Wang, K. Research Progress of Non-Invasive Magnetic Resonance Imaging in Lithium-Ion Battery Detection. Coatings 2026, 16, 453. https://doi.org/10.3390/coatings16040453
Jiang W, Deng Y, Li W, Song J, Che S, Wang K. Research Progress of Non-Invasive Magnetic Resonance Imaging in Lithium-Ion Battery Detection. Coatings. 2026; 16(4):453. https://doi.org/10.3390/coatings16040453
Chicago/Turabian StyleJiang, Wen, Yunyi Deng, Wentao Li, Jilong Song, Songtao Che, and Kai Wang. 2026. "Research Progress of Non-Invasive Magnetic Resonance Imaging in Lithium-Ion Battery Detection" Coatings 16, no. 4: 453. https://doi.org/10.3390/coatings16040453
APA StyleJiang, W., Deng, Y., Li, W., Song, J., Che, S., & Wang, K. (2026). Research Progress of Non-Invasive Magnetic Resonance Imaging in Lithium-Ion Battery Detection. Coatings, 16(4), 453. https://doi.org/10.3390/coatings16040453