Experimental Study on Anti-Crystallization Performance of Tunnel Drainage Pipes Based on Magnetic Powder Effect
Abstract
1. Introduction
2. Experimental Design
2.1. Experimental Materials and Apparatus
2.1.1. Preparation of Magnetic Drainage Pipes
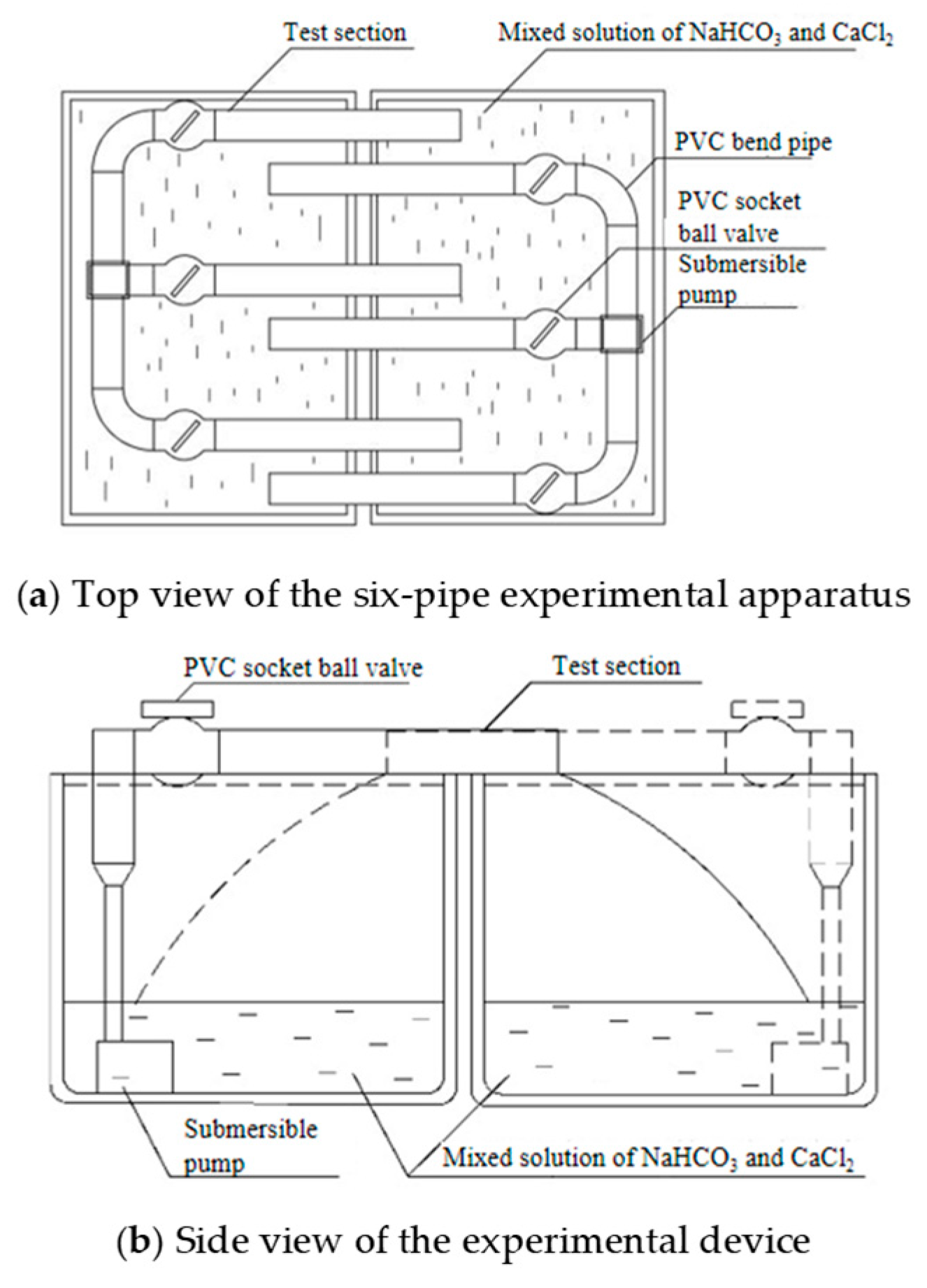
2.1.2. Test Solution and Apparatus Design
2.2. Experimental Plan
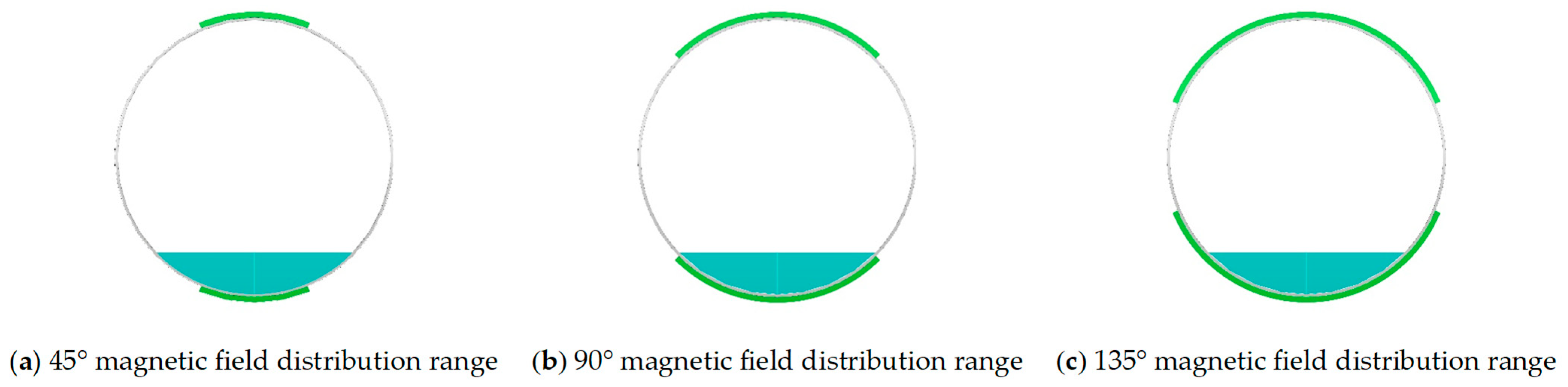
2.2.1. Crystallization Behavior Under Different Magnetic Field Coverage Angles
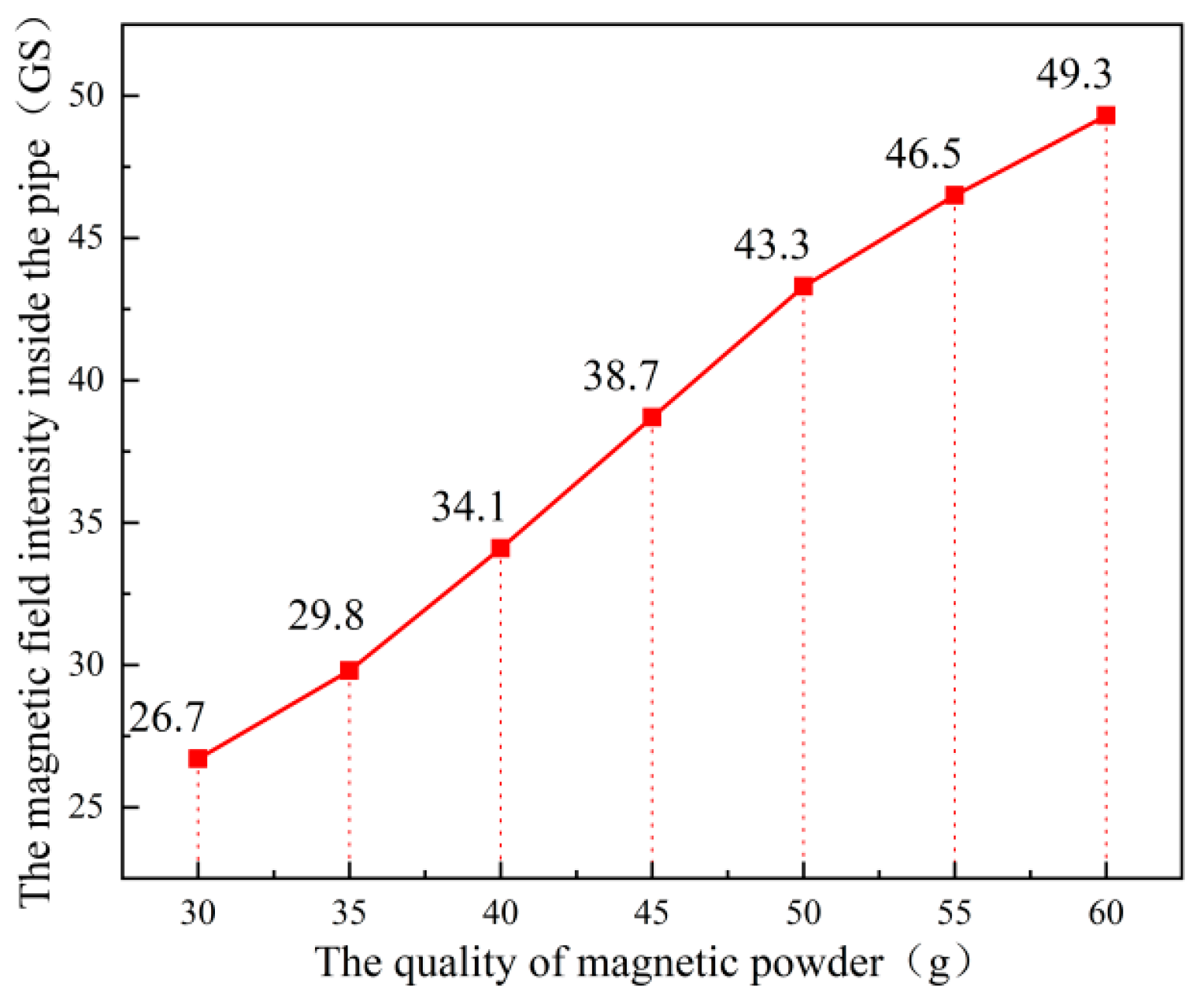
2.2.2. Crystallization Behavior Under Different Magnetic Field Intensities
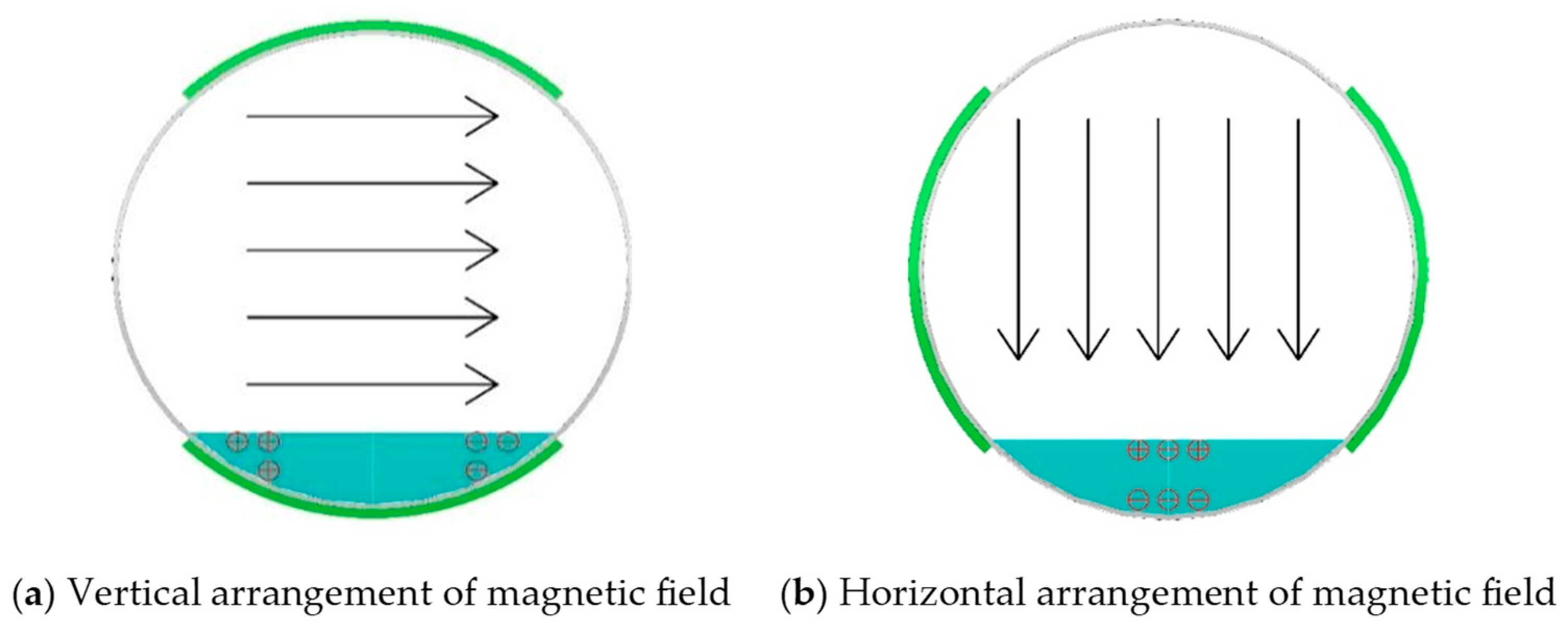
2.2.3. Crystallization Behavior Under Different Flow Rates and Magnetic Field Orientations
3. Experimental Results and Discussion
3.1. Effect of Magnetic Field Coverage Angle on Crystallization Behavior in Tunnel Drainage Pipes
3.1.1. Analysis of Laboratory Test Results
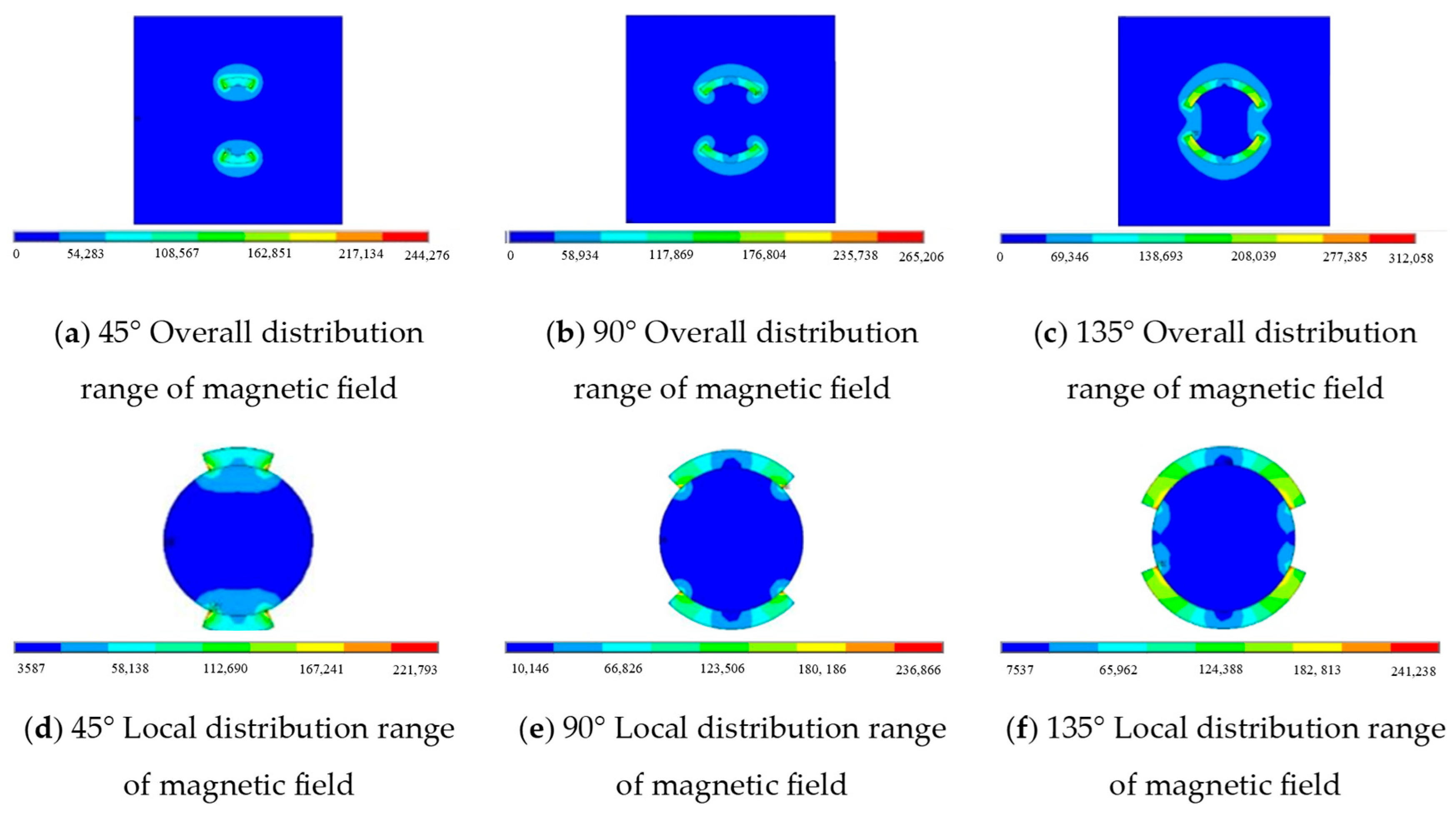
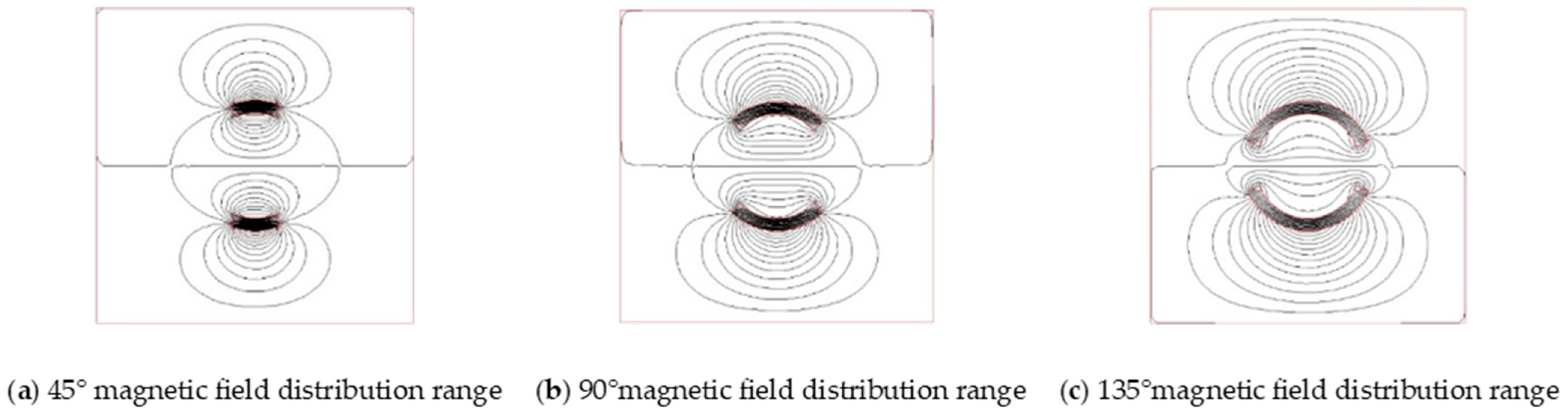
3.1.2. Numerical Simulation Validation
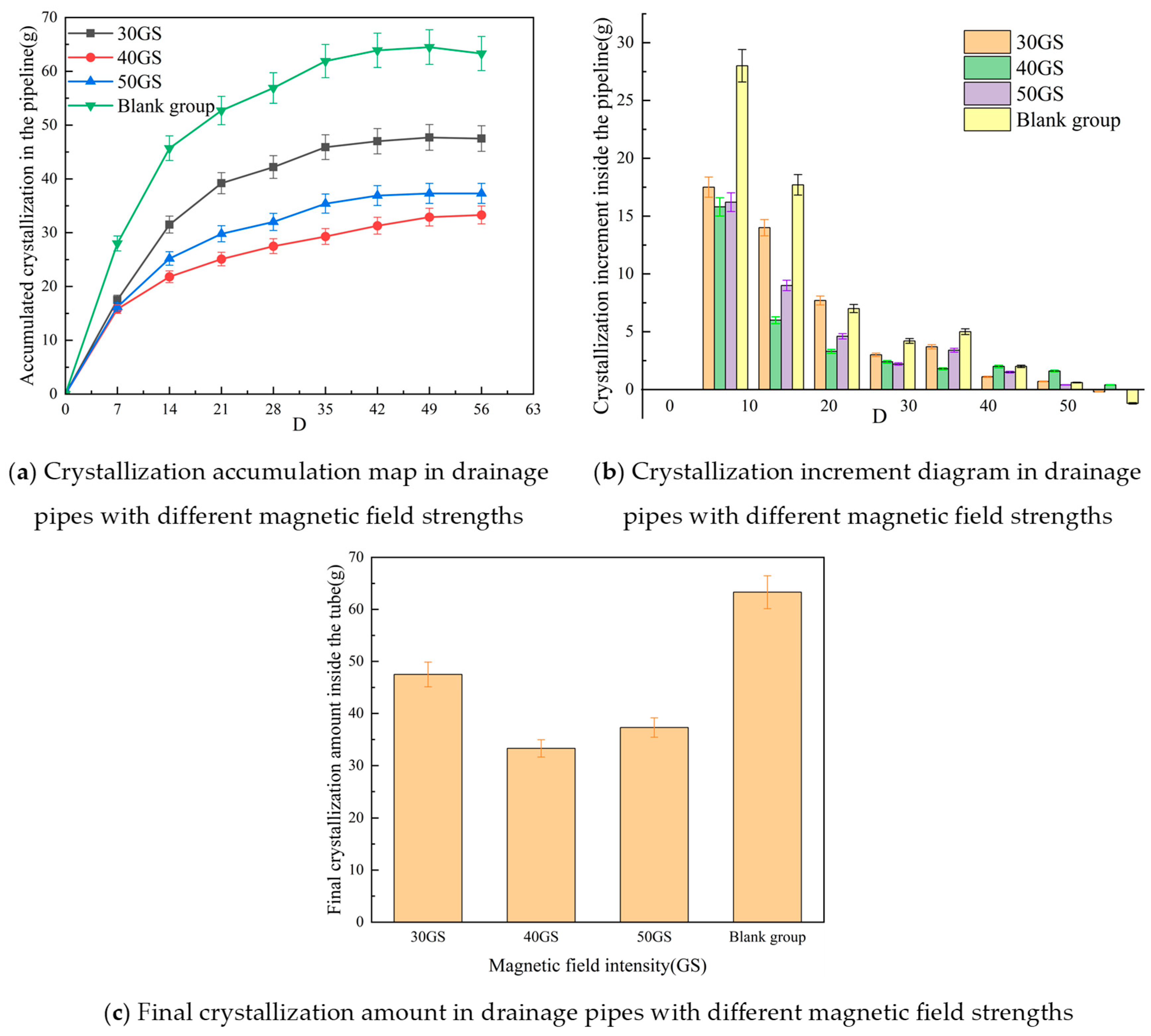
3.2. Influence of Magnetic Field Intensity on Crystallization Behavior in Tunnel Drainage Pipes
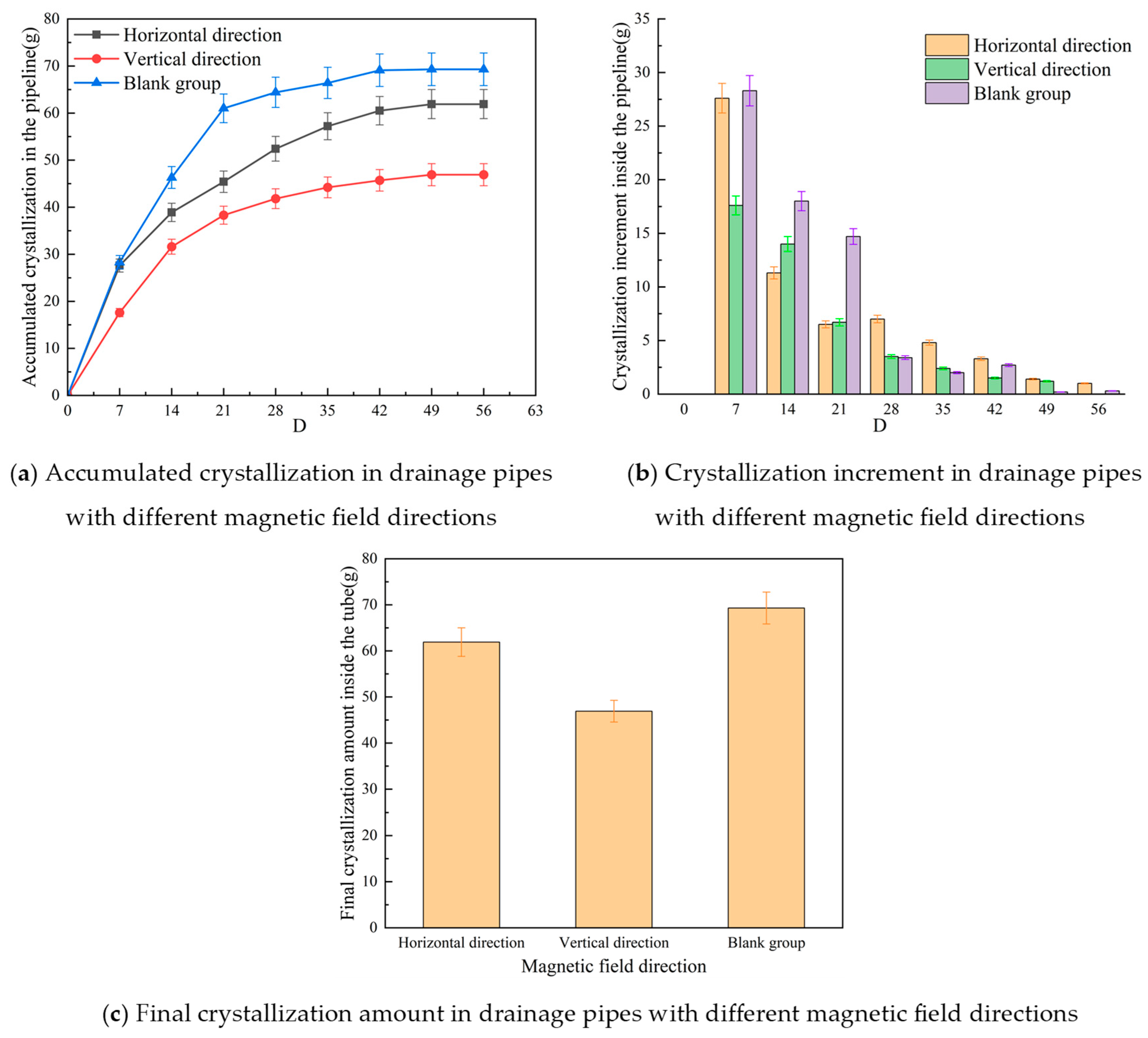
3.3. Influence of Magnetic Field Orientation on Crystallization Behavior in Tunnel Drainage Pipes
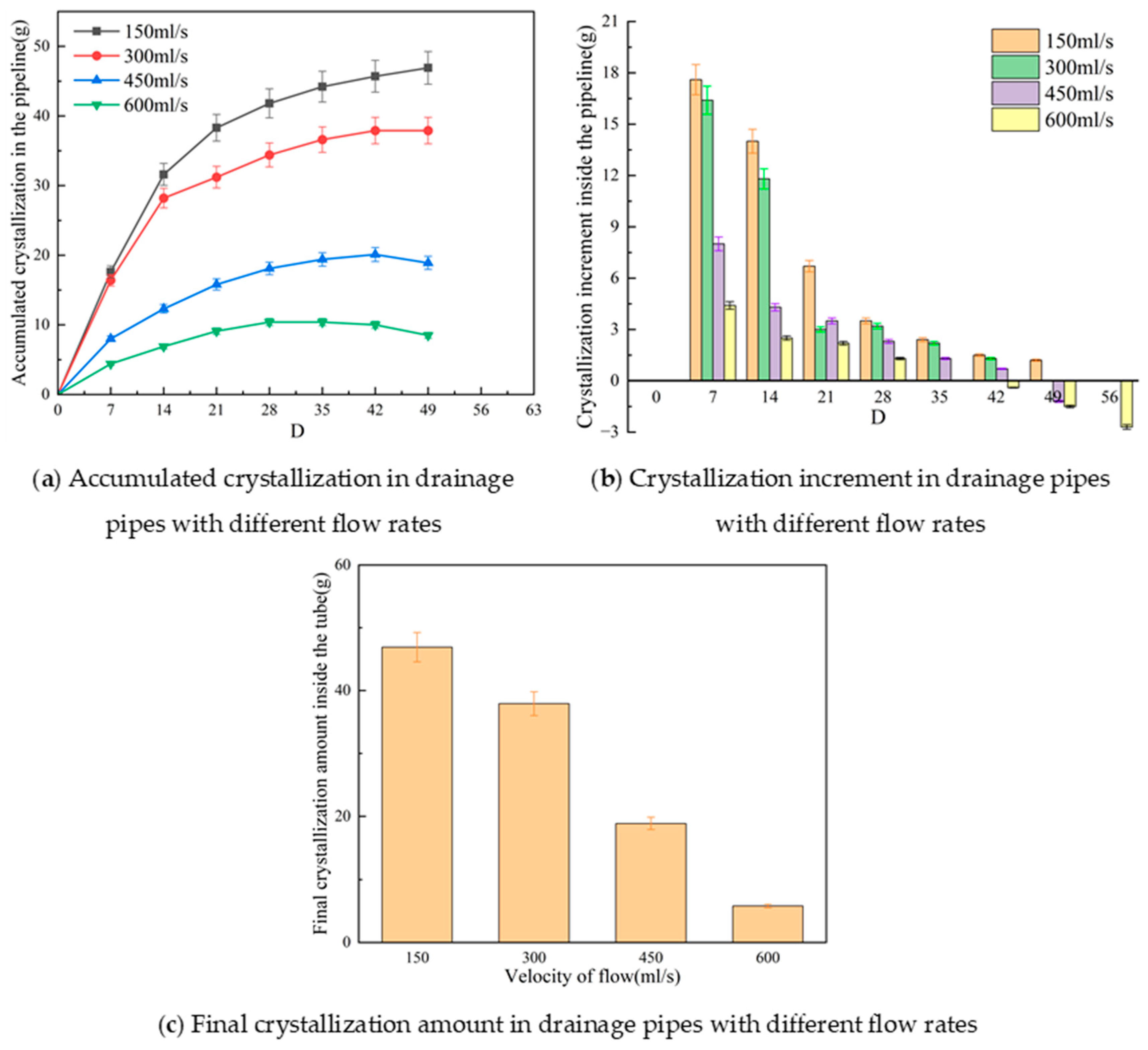
3.4. Influence of Internal Flow Rate on Crystallization Behavior in Tunnel Drainage Pipes
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Xu, S.; Ma, E.; Lai, J.; Yang, Y.; Liu, H.; Yang, C.; Hu, Q. Diseases failures characteristics and countermeasures of expressway tunnel of water-rich strata: A case study. Eng. Fail. Anal. 2022, 134, 106056. [Google Scholar] [CrossRef]
- Tan, Z.; Li, S.; Yang, Y.; Wang, J. Large deformation characteristics and controlling measures of steeply inclined and layered soft rock of tunnels in plate suture zones. Eng. Fail. Anal. 2022, 131, 105831. [Google Scholar] [CrossRef]
- Yoo, C. Hydraulic deterioration of geosynthetic filter drainage system in tunnels–its impact on structural performance of tunnel linings. Geosynth. Int. 2016, 23, 463–480. [Google Scholar] [CrossRef]
- Seki, S.; Kaise, S.; Morisaki, Y.; Azetaka, S.; Jiang, Y. Model experiments for examining heaving phenomenon in tunnels. Tunn. Undergr. Space Technol. 2008, 23, 128–138. [Google Scholar] [CrossRef]
- Zhang, S.; Xu, Q.; Yoo, C.; Min, B.; Liu, C.; Guan, X.; Li, P. Lining cracking mechanism of old highway tunnels caused by drainage system deterioration: A case study of Liwaiao Tunnel, Ningbo, China. Eng. Fail. Anal. 2022, 137, 106270. [Google Scholar] [CrossRef]
- Xu, Q.; Zhang, S.; Li, P.; Liu, C.; Bao, T. Lining failure performance of highway tunnels induced by the drainage system deterioration. Eng. Fail. Anal. 2023, 149, 107236. [Google Scholar] [CrossRef]
- Xu, C.; Chen, Y.; Yang, Y.; Li, P.; Wang, S.; Li, L. Experimental Investigation of Crystal Blocking in Drainage Pipes for Tunnels in the Karst Region. Appl. Sci. 2022, 12, 10928. [Google Scholar] [CrossRef]
- Jung, H.; Han, Y.; Chung, S.; Chun, B.; Lee, Y. Evaluation of advanced drainage treatment for old tunnel drainage system in Korea. Tunn. Undergr. Space Technol. 2013, 38, 476–486. [Google Scholar] [CrossRef]
- Jiang, Y.; Tong, Y.; Ye, F.; Zhang, J.; Tian, C.; Wu, B.; Cui, H. The failure mechanism of crystallization blockage in tunnel drainage system and preventive measure of scale inhibitor. Eng. Fail. Anal. 2024, 166, 108915. [Google Scholar] [CrossRef]
- Galan, I.; Baldermann, A.; Kusterle, W.; Dietzel, M.; Mittermayr, M. Durability of shotcrete for underground support–Review and update. Constr. Build. Mater. 2019, 202, 465–493. [Google Scholar] [CrossRef]
- Sun, M.; Cao, J.; Cao, J.; Zhang, S.; Chen, Y.; Bate, B. Discrete element modeling of shear wave propagation in carbonate precipitate–Cemented particles. Acta Geotech. 2022, 17, 2633–2649. [Google Scholar] [CrossRef]
- Eichinger, S.; Boch, R.; Leis, A.; Koraimann, G.; Grengg, C.; Domberger, G.; Nachtnebel, M.; Schwab, C.; Dietzel, M. Scale deposits in tunnel drainage systems–A study on fabrics and formation mechanisms. Sci. Total Environ. 2020, 718, 137140. [Google Scholar] [CrossRef]
- Wang, Y.D.; Liu, Y.; Qi, C.F.; Zhou, T.Y.; Ye, M.; Wang, T. Crystallization law of karst water in tunnel drainage system based on DBL theory. Open Phys. 2021, 19, 241–255. [Google Scholar] [CrossRef]
- Liu, D.G.; Yang, Y.; Mao, C.J.; Wu, J.F.; Wu, J.C. A comparative study on hydrodynamics and hydrochemistry coupled simulations of drainage pipe crystallization blockage in karst tunnels. J. Earth Sci. 2022, 33, 1179–1189. [Google Scholar] [CrossRef]
- Zhao, K.; Yu, B.; Yang, Y.; Li, S.; Li, D. Causes, influencing factors, and prediction modeling of crystallization blockage of tunnel drainage blind pipe in non-karst area. Case Stud. Constr. Mater. 2024, 21, e04041. [Google Scholar] [CrossRef]
- Dietzel, M.; Rinder, T.; Leis, A.; Reichl, P.; Sellner, P.; Draschitz, C.; Plank, G.; Klammer, D.; Schofer, H. Koralm tunnel as a case study for sinter formation in drainage systems–Precipitation mechanisms and retaliatory action. Geomech. Tunnelbau Geomech. Tunnelbau 2008, 1, 271–278. [Google Scholar] [CrossRef]
- Xu, Z.; Wang, X.; Zhang, C.; Zeng, D.; Liu, C. Scaling and clogging treatment of aging tunnel drainage pipes in karst areas using eco-friendly acid agent. J. Rock Mech. Geotech. Eng. 2023, 15, 896–910. [Google Scholar] [CrossRef]
- Tian, C.; Tong, Y.; Zhang, J.; Ye, F.; Song, G.; Jiang, Y.; Zhao, M. Experimental study on mix proportion optimization of anti-calcium dissolution shotcrete for tunnels based on response surface methodology. Undergr. Space 2024, 15, 203–220. [Google Scholar] [CrossRef]
- Lin, Z.; Liu, K.; Zhang, X.; Chen, X.; Xie, P.; Tan, Y. Optimizations for tunnel drainage water system based on locations of crystallizations. Desalination Water Treat. 2023, 299, 232–238. [Google Scholar] [CrossRef]
- Wei, L.; Guo, J.; Zhang, X.; Li, Y.; Huang, H. Study of electric field impact on crystallization in tunnel drainage pipes in hard water area. Adv. Civ. Eng. 2021, 2021, 5584229. [Google Scholar] [CrossRef]
- Hou, Z.; Liu, Q.; Sun, Y.; Han, F.; Zhang, J.; Liu, S.; Liu, Z. Influence of Electromagnetic Field on Crystallization of Tunnel Drainage Pipes. J. Appl. Sci. Eng. 2022, 26, 517–527. [Google Scholar]
- Leng, H.; Lv, H.; Han, F.; Liu, F.; Liu, T. Permanent magnet treatment technology for crystal blockage of tunnel drainage pipes. Desalination Water Treat. 2021, 243, 211–220. [Google Scholar] [CrossRef]
- Xin, Z.; Moon, J.H.; Kim, Y.U. Reduction of adherent forces of sedimentous contaminants in tunnel drainage using vibrations from flexible and transparent organic films. KSCE J. Civ. Eng. 2018, 22, 2619–2622. [Google Scholar] [CrossRef]
- Xin, Z.; Moon, J.H.; Kim, Y.U. Effect of vibration of flexible transducer on shear resistance of scaling in tunnel drainage. KSCE J. Civ. Eng. 2017, 21, 1929–1932. [Google Scholar] [CrossRef]
- Jiang, Y.; Zhang, J.; Ye, F.; Tong, Y.; Tian, C.; Li, Y. Anti-crystallisation coating for tunnel drainage system: Investigation, experiment, and mechanism. Eng. Fail. Anal. 2024, 157, 107903. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, X.; Wei, L.; Liu, S.; Zhang, B.; Zhou, C. Experimental study on prevention of calcium carbonate crystallizing in drainage pipe of tunnel engineering. Adv. Civ. Eng. 2018, 2018, 9430517. [Google Scholar] [CrossRef]
- Eichinger, S.; Boch, R.; Leis, A.; Baldermann, A.; Domberger, G.; Schwab, C.; Dietzel, M. Green inhibitors reduce unwanted calcium carbonate precipitation: Implications for technical settings. Water Res. 2022, 208, 117850. [Google Scholar] [CrossRef]
- Liu, H.; Zhao, Y.; Peng, C.; Song, S.; López-Valdivieso, A. Lime mortars–The role of carboxymethyl cellulose on the crystallization of calcium carbonate. Constr. Build. Mater. 2018, 168, 169–177. [Google Scholar] [CrossRef]
- Liu, Y.; Cui, Y.J.; Mao, H.Y.; Guo, R. Calcium carbonate crystallization in the presence of casein. Cryst. Growth Des. 2012, 12, 4720–4726. [Google Scholar] [CrossRef]
- Liu, S.; Zhang, X.; Chen, X.; Wang, C.; Chen, Y. Exploratory Research on Drainage Structure of Highway Tunnel Based on Reducing the Risk of Crystallization Blockage. Processes 2022, 10, 1319. [Google Scholar] [CrossRef]
- Liu, S.; Zhang, X. Fault Diagnosis and Maintenance Countermeasures of Transverse Drainage Pipe in Subway Tunnel Based on Fault Tree Analysis. Int. J. Environ. Res. Public Health 2022, 19, 15471. [Google Scholar] [CrossRef]
- Goncharuk, V.V.; Bagrii, V.A.; Bashtan, S.Y. Crystallization of calcium carbonate from aqueous solutions in superimposition of electric and magnetic fields. J. Water Chem. Technol. 2012, 34, 133–135. [Google Scholar] [CrossRef]
- Mahmoud, B.; Yosra, M.; Nadia, A. Effects of magnetic treatment on scaling power of hard waters. Sep. Purif. Technol. 2016, 171, 88–92. [Google Scholar] [CrossRef]
- Sohaili, J.; Shi, H.S.; Zardari, N.H.; Ahmad, N.; Muniyandi, S.K. Removal of scale deposition on pipe walls by using magnetic field treatment and the effects of magnetic strength. J. Clean. Prod. 2016, 139, 1393–1399. [Google Scholar] [CrossRef]
- Latifa, S.B.; Cheap-Charpentier, H.; Perrot, H.; Amor, Y.B. Effects of magnetic field on homogeneous and heterogeneous precipitation of calcium carbonate. ChemElectroChem 2023, 10, e202300105. [Google Scholar] [CrossRef]
- Turner, C.W.; Smith, D.W. Calcium carbonate scaling kinetics determined from radiotracer experiments with calcium-47. Ind. Eng. Chem. Res. 1998, 37, 439–448. [Google Scholar] [CrossRef]
- Xu, X.; Zhang, Y.; Li, L.; Xu, C.B. Model test on crystallization law of tunnel drainage pipe in karst area. Highw. Traffic Sci. Technol. 2022, 39, 133–139. [Google Scholar]
- Lv, J.; Wu, H.; Li, J.; Wan, X.; Fu, H. Analysis of Fouling Pattern of Tunnel Drainage Pipe in Karst Areas. Adv. Civ. Eng. 2023, 2023, 8881333. [Google Scholar] [CrossRef]






| Simulation Materials | Relative Permeability | Coercivity (A/m) |
|---|---|---|
| Air | 1 | - |
| Neodymium iron–boron magnetic powder | 1.05 | 220,000 (220 Gs) |
| Coercivity (A/m) | Magnetic Field Intensity (Gs) |
|---|---|
| 30,000 | 30 |
| 40,000 | 40 |
| 50,000 | 50 |
| Working | Test Section Number | Magnetic Field Intensity (Gs) | Distribution Range (°) | Magnetic Field Direction | Water Flow Rate (mL/s) | |
|---|---|---|---|---|---|---|
| Condition Water Tank | ||||||
| A | 1 | 30 | 90 | vertical direction | 600 | |
| 2 | 0 | 0 | - | 150 | ||
| 3 | 30 | 90 | Horizontal direction | 150 | ||
| B | 4 | 30 | 90 | vertical direction | 450 | |
| 5 | 30 | 90 | vertical direction | 300 | ||
| 6 | 30 | 90 | vertical direction | 150 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xiao, D.; Liu, B.; Liu, S.; Wang, C.; Huang, K.; Yu, X.; Wu, W. Experimental Study on Anti-Crystallization Performance of Tunnel Drainage Pipes Based on Magnetic Powder Effect. Coatings 2025, 15, 1005. https://doi.org/10.3390/coatings15091005
Xiao D, Liu B, Liu S, Wang C, Huang K, Yu X, Wu W. Experimental Study on Anti-Crystallization Performance of Tunnel Drainage Pipes Based on Magnetic Powder Effect. Coatings. 2025; 15(9):1005. https://doi.org/10.3390/coatings15091005
Chicago/Turabian StyleXiao, Donghui, Benhua Liu, Shiyang Liu, Cheng Wang, Kun Huang, Xingjie Yu, and Wenzhen Wu. 2025. "Experimental Study on Anti-Crystallization Performance of Tunnel Drainage Pipes Based on Magnetic Powder Effect" Coatings 15, no. 9: 1005. https://doi.org/10.3390/coatings15091005
APA StyleXiao, D., Liu, B., Liu, S., Wang, C., Huang, K., Yu, X., & Wu, W. (2025). Experimental Study on Anti-Crystallization Performance of Tunnel Drainage Pipes Based on Magnetic Powder Effect. Coatings, 15(9), 1005. https://doi.org/10.3390/coatings15091005






