Thermally Induced Polymorphic Changes in Poly(vinylidene Fluoride) Thin Layer Investigated Using Micro-Raman Spectroscopy
Abstract
1. Introduction
2. Materials and Methods
2.1. Material
2.2. Methodologies
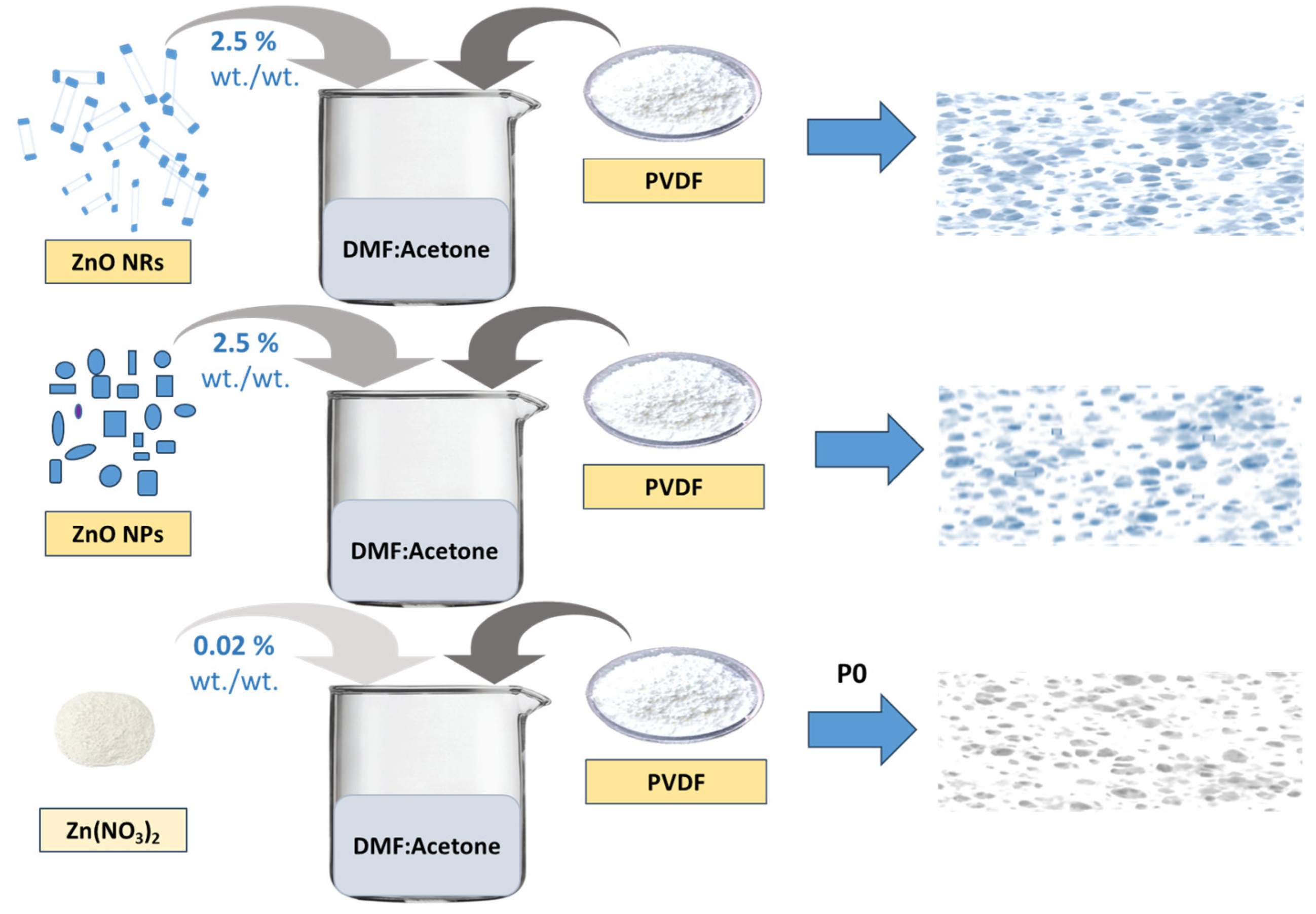
2.2.1. Synthesis of PVDF Thin Films
2.2.2. Experimental Setup
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dallaev, R.; Pisarenko, T.; Sobola, D.; Orudzhev, F.; Ramazanov, S.; Trčka, T. Brief review of PVDF properties and applications potential. Polymers 2022, 14, 4793. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Han, X.; Shen, Q.D. PVDF-based ferroelectric polymers in modern flexible electronics. Adv. Electron. Mater. 2017, 3, 1600460. [Google Scholar] [CrossRef]
- Wan, C.; Bowen, C.R. Multiscale-structuring of polyvinylidene fluoride for energy harvesting: The impact of molecular-, micro-and macro-structure. J. Mater. Chem. A 2017, 5, 3091–3128. [Google Scholar] [CrossRef]
- Veved, A.; Ejuh, G.W.; Djongyang, N. Review of emerging materials for PVDF-based energy harvesting. Energy Rep. 2022, 8, 12853–12870. [Google Scholar] [CrossRef]
- Ruan, L.; Yao, X.; Chang, Y.; Zhou, L.; Qin, G.; Zhang, X. Properties and applications of the β phase poly(vinylidene fluoride). Polymers 2018, 10, 228. [Google Scholar] [CrossRef]
- Concha, V.O.; Timóteo, L.; Duarte, L.A.; Bahú, J.O.; Munoz, F.L.; Silva, A.P.; Lodi, L.; Severino, P.; León-Pulido, J.; Souto, E.B. Properties, characterization and biomedical applications of polyvinylidene fluoride (PVDF): A review. J. Mater. Sci. 2024, 59, 14185–14204. [Google Scholar] [CrossRef]
- Mohammadpourfazeli, S.; Arash, S.; Ansari, A.; Yang, S.; Mallick, K.; Bagherzadeh, R. Future prospects and recent developments of polyvinylidene fluoride (PVDF) piezoelectric polymer; fabrication methods, structure, and electro-mechanical properties. RSC Adv. 2023, 13, 370–387. [Google Scholar] [CrossRef]
- Saxena, P.; Shukla, P. A comprehensive review on fundamental properties and applications of poly(vinylidene fluoride)(PVDF). Adv. Compos. Hybrid Mater. 2021, 4, 8–26. [Google Scholar] [CrossRef]
- Xu, F.; Zhang, K.; Zhou, Y.; Qu, Z.; Wang, H.; Zhang, Y.; Zhou, H.; Yan, C. Facile preparation of highly oriented poly(vinylidene fluoride) uniform films and their ferro-and piezoelectric properties. RSC Adv. 2017, 7, 17038–17043. [Google Scholar] [CrossRef]
- Jia, N.; Xing, Q.; Liu, X.; Sun, J.; Xia, G.; Huang, W.; Song, R. Enhanced electroactive and mechanical properties of poly(vinylidene fluoride) by controlling crystallization and interfacial interactions with low loading polydopamine coated BaTiO3. J. Colloid Interface Sci. 2015, 453, 169–176. [Google Scholar] [CrossRef]
- He, F.; Lau, S.; Chan, H.L.; Fan, J. High dielectric permittivity and low percolation threshold in nanocomposites based on poly(vinylidene fluoride) and exfoliated graphite nanoplates. Adv. Mater. 2009, 21, 710–715. [Google Scholar] [CrossRef]
- Kushwah, M.; Sagar, R.; Rogachev, A.; Gaur, M. Dielectric, pyroelectric and polarization behavior of polyvinylidene fluoride (PVDF)-gold nanoparticles (AuNPs) nanocomposites. Vacuum 2019, 166, 298–306. [Google Scholar] [CrossRef]
- Lin, B.; Chen, G.-D.; He, F.-A.; Li, Y.; Yang, Y.; Shi, B.; Feng, F.-R.; Chen, S.-Y.; Lam, K.-H. Preparation of MWCNTs/PVDF composites with high-content β form crystalline of PVDF and enhanced dielectric constant by electrospinning-hot pressing method. Diam. Relat. Mater. 2023, 131, 109556. [Google Scholar] [CrossRef]
- Chiu, F.-C. Comparisons of phase morphology and physical properties of PVDF nanocomposites filled with organoclay and/or multi-walled carbon nanotubes. Mater. Chem. Phys. 2014, 143, 681–692. [Google Scholar] [CrossRef]
- Kadir, E.; Gayen, R. Improved UV sensitivity in solution-processed PVDF/ZnO nanocomposites via piezo-phototronic effect. Mater. Today Commun. 2024, 39, 109174. [Google Scholar] [CrossRef]
- Sabry, R.S.; Hussein, A.D. Nanogenerator based on nanocomposites PVDF/ZnO with different concentrations. Mater. Res. Express 2019, 6, 105549. [Google Scholar] [CrossRef]
- Indolia, A.P.; Gaur, M. Optical properties of solution grown PVDF-ZnO nanocomposite thin films. J. Polym. Res. 2013, 20, 43. [Google Scholar] [CrossRef]
- Satapathy, S.; Pawar, S.; Gupta, P.; Varma, K. Effect of annealing on phase transition in poly(vinylidene fluoride) films prepared using polar solvent. Bull. Mater. Sci. 2011, 34, 727–733. [Google Scholar] [CrossRef]
- Chinaglia, D.L.; Gregorio Jr, R.; Stefanello, J.C.; Pisani Altafim, R.A.; Wirges, W.; Wang, F.; Gerhard, R. Influence of the solvent evaporation rate on the crystalline phases of solution-cast poly(vinylidene fluoride) films. J. Appl. Polym. Sci. 2010, 116, 785–791. [Google Scholar] [CrossRef]
- Ginghina, R.-E.; Toader, G.; Purica, M.; Bratu, A.-E.; Lazaroaie, C.; Tiganescu, T.-V.; Oncioiu, R.-E.; Iorga, G.-O.; Zorila, F.-L.; Constantin, M. Antimicrobial activity and degradation ability study on nanoparticle-enriched formulations specially designed for the neutralization of real and simulated biological and chemical warfare agents. Pharmaceuticals 2022, 15, 97. [Google Scholar] [CrossRef]
- Dediu, V.; Busila, M.; Tucureanu, V.; Bucur, F.I.; Iliescu, F.S.; Brincoveanu, O.; Iliescu, C. Synthesis of ZnO/Au nanocomposite for antibacterial applications. Nanomaterials 2022, 12, 3832. [Google Scholar] [CrossRef] [PubMed]
- Benz, M.; Euler, W.B. Determination of the crystalline phases of poly(vinylidene fluoride) under different preparation conditions using differential scanning calorimetry and infrared spectroscopy. J. Appl. Polym. Sci. 2003, 89, 1093–1100. [Google Scholar] [CrossRef]
- Hasegawa, R.; Kobayashi, M.; Tadokoro, H. Molecular conformation and packing of poly(vinylidene fluoride). Stability of three crystalline forms and the effect of high pressure. Polym. J. 1972, 3, 591–599. [Google Scholar] [CrossRef]
- Ievlev, A.V.; Susner, M.A.; McGuire, M.A.; Maksymovych, P.; Kalinin, S.V. Quantitative analysis of the local phase transitions induced by laser heating. ACS Nano 2015, 9, 12442–12450. [Google Scholar] [CrossRef]
- Zheng, J.; He, A.; Li, J.; Han, C.C. Polymorphism control of poly(vinylidene fluoride) through electrospinning. Macromol. Rapid Commun. 2007, 28, 2159–2162. [Google Scholar] [CrossRef]
- Constantino, C.; Job, A.E.; Simões, R.; Giacometti, J.A.; Zucolotto, V.; Oliveira Jr, O.; Gozzi, G.; Chinaglia, D. Phase transition in poly(vinylidene fluoride) investigated with micro-Raman spectroscopy. Appl. Spectrosc. 2005, 59, 275–279. [Google Scholar] [CrossRef]
- Silva, M.; Sencadas, V.; Botelho, G.; Machado, A.; Rolo, A.; Rocha, J.G.; Lanceros-Méndez, S. α-and γ-PVDF: Crystallization kinetics, microstructural variations and thermal behaviour. Mater. Chem. Phys. 2010, 122, 87–92. [Google Scholar] [CrossRef]
- Bormashenko, Y.; Pogreb, R.; Stanevsky, O.; Bormashenko, E. Vibrational spectrum of PVDF and its interpretation. Polym. Test. 2004, 23, 791–796. [Google Scholar] [CrossRef]
- Tan, K.; Gan, W.; Velayutham, T.; Abd Majid, W. Pyroelectricity enhancement of PVDF nanocomposite thin films doped with ZnO nanoparticles. Smart Mater. Struct. 2014, 23, 125006. [Google Scholar] [CrossRef]
- Neidhöfer, M.; Beaume, F.; Ibos, L.; Bernes, A.; Lacabanne, C. Structural evolution of PVDF during storage or annealing. Polymer 2004, 45, 1679–1688. [Google Scholar] [CrossRef]
- Purushothaman, S.M.; Tronco, M.F.; Ponçot, M.; CS, C.; Guigo, N.; Malfois, M.; Kalarikkal, N.; Thomas, S.; Royaud, I.; Rouxel, D. Quantifying the Crystalline Polymorphism in PVDF: Comparative Criteria Using DSC, WAXS, FT-IR, and Raman Spectroscopy. ACS Appl. Polym. Mater. 2024, 6, 8291–8305. [Google Scholar] [CrossRef]



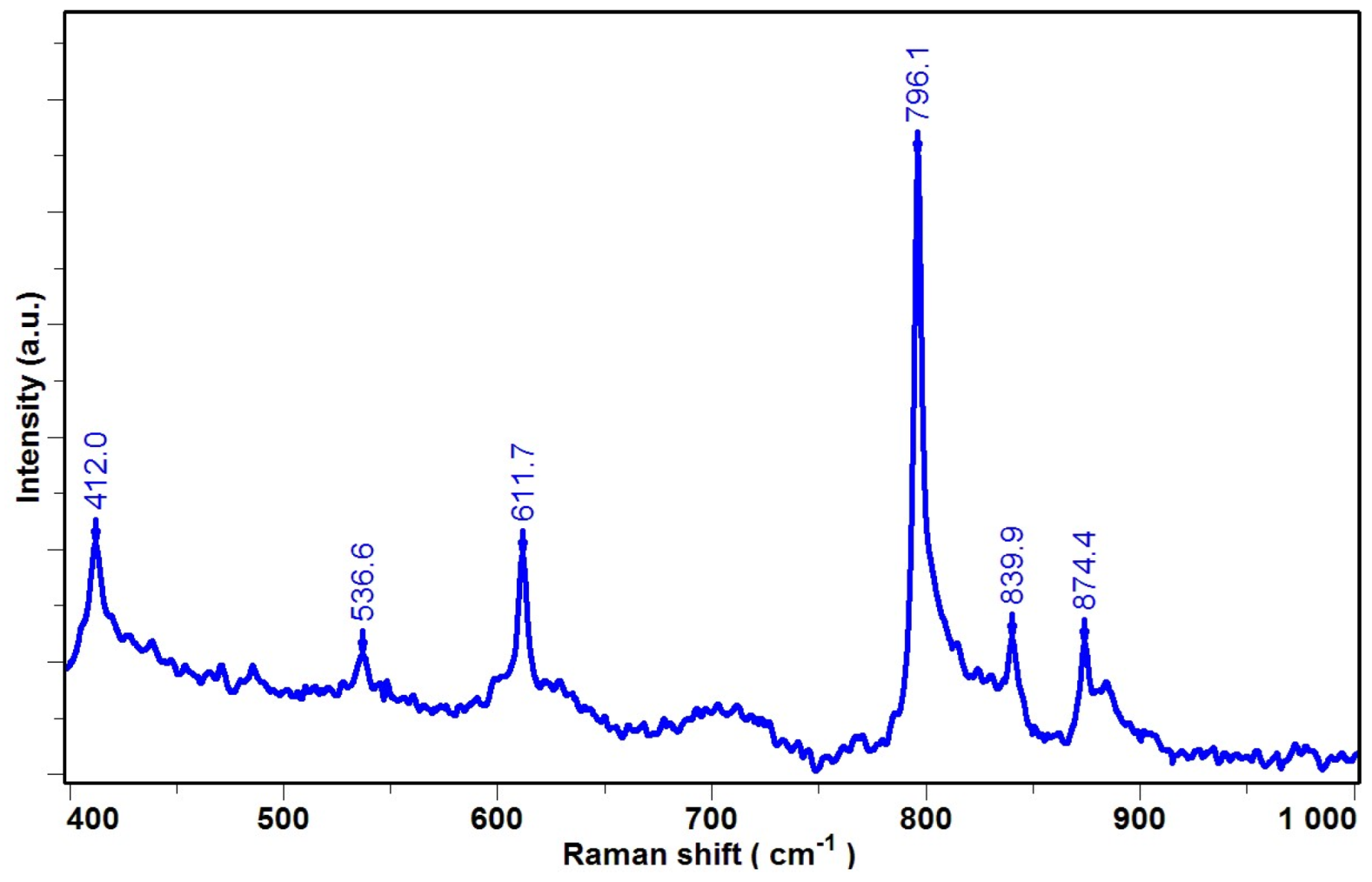
| Raman Peaks [cm−1] | References | Phase | Experimental Micro-Raman Peaks Position (λ = 632.8 nm) |
|---|---|---|---|
| 485 | [22,23] | γ | 484.5 |
| 513 | [22,23,24] | β | 513.7 |
| 610 | [22,25,26] | α | 606.2 |
| 795 | [22,23,27] | α | 798.5 |
| 812 | [22,23] | γ | 813.5 |
| 840 | [22,23,27] | β | 840.5 |
| 881 | [25,26] | α, β | 882.0 |
| 1076–3000 | [27,28] | CC, CH2 Vibrations, common to the phases | |
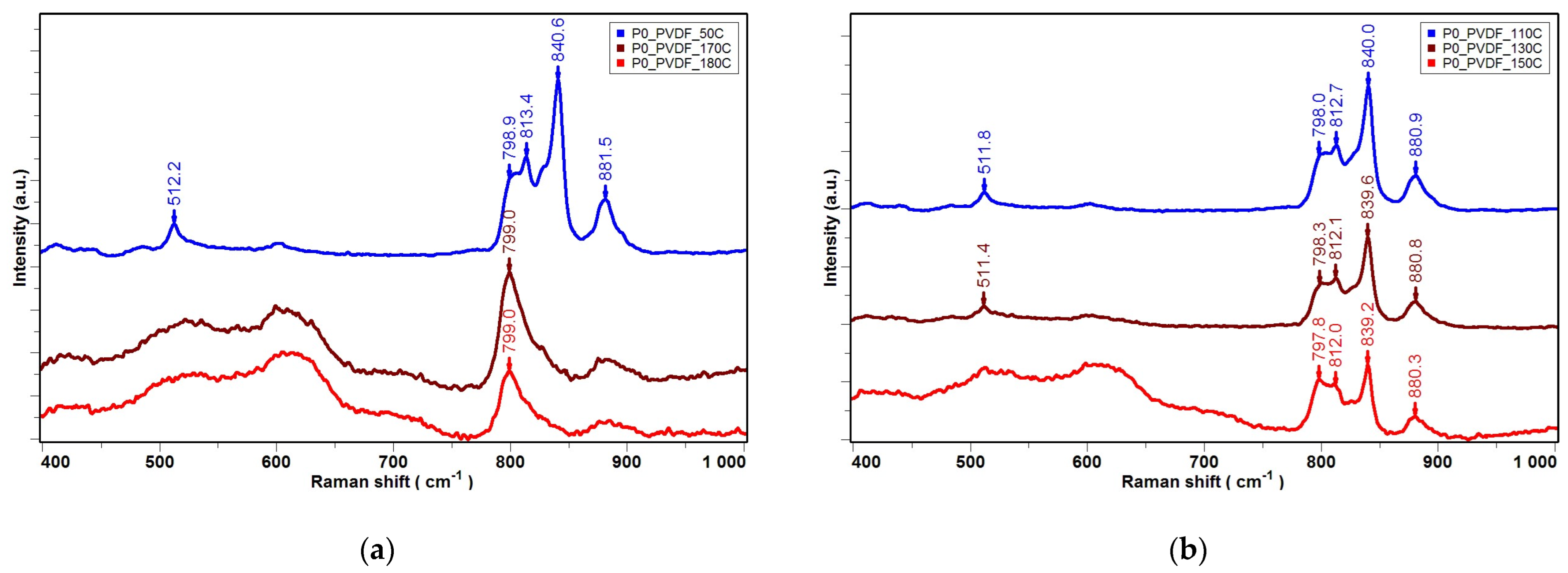
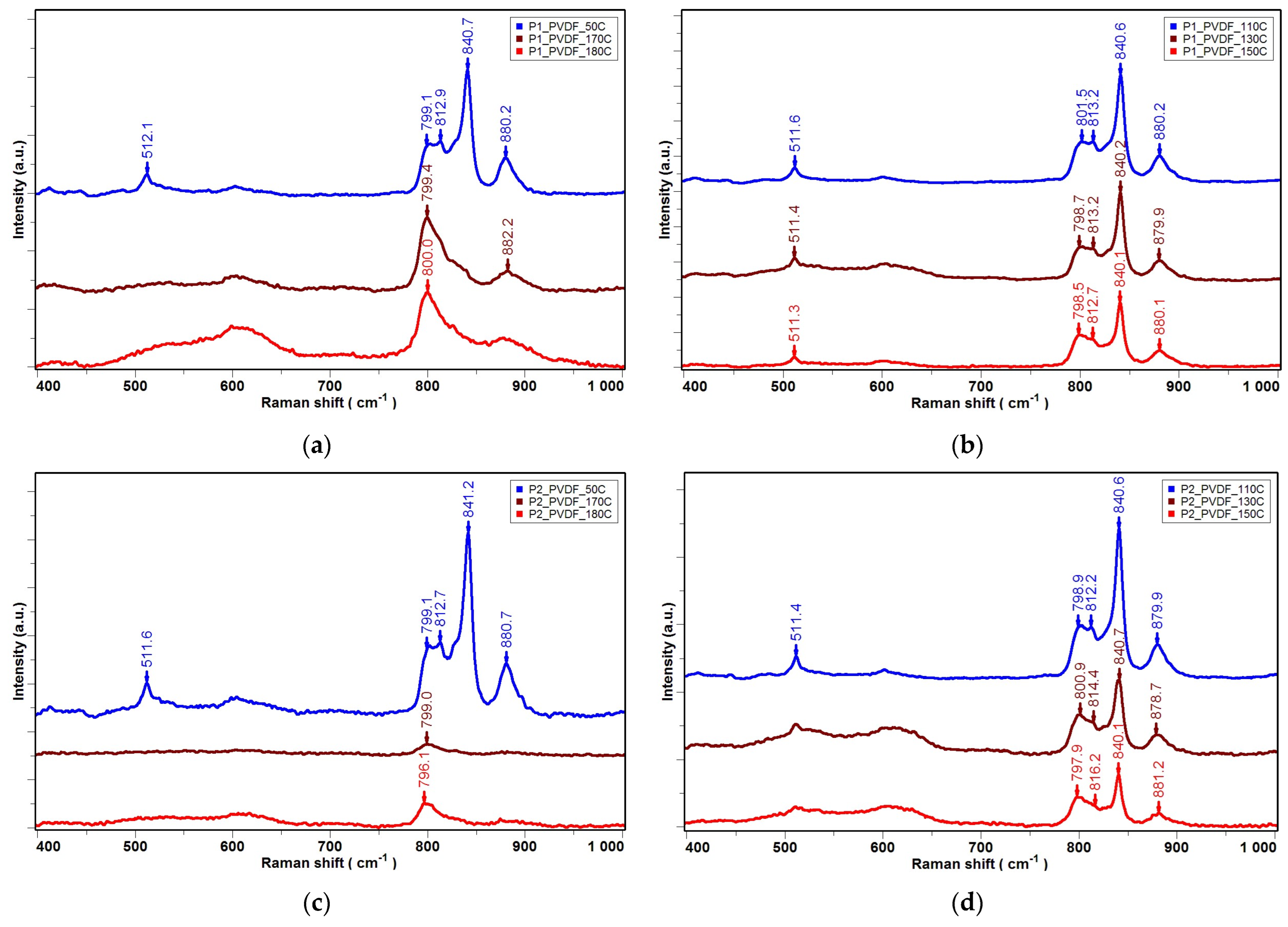
| Temperature T [°C] | 50 °C | 110 °C | 130 °C | 150 °C |
|---|---|---|---|---|
| Iβ/Iα- sample P0 | 1.42 | 2.38 | 2.38 | 2.17 |
| Iβ/Iα- sample P1 | 3.21 | 3.82 | 3.92 | 4.58 |
| Iβ/Iα- sample P2 | 3.62 | 3.86 | 4.16 | 5.56 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Purica, M.; Comanescu, F.C.; Dediu, V. Thermally Induced Polymorphic Changes in Poly(vinylidene Fluoride) Thin Layer Investigated Using Micro-Raman Spectroscopy. Coatings 2024, 14, 1520. https://doi.org/10.3390/coatings14121520
Purica M, Comanescu FC, Dediu V. Thermally Induced Polymorphic Changes in Poly(vinylidene Fluoride) Thin Layer Investigated Using Micro-Raman Spectroscopy. Coatings. 2024; 14(12):1520. https://doi.org/10.3390/coatings14121520
Chicago/Turabian StylePurica, Munizer, Florin Constantin Comanescu, and Violeta Dediu. 2024. "Thermally Induced Polymorphic Changes in Poly(vinylidene Fluoride) Thin Layer Investigated Using Micro-Raman Spectroscopy" Coatings 14, no. 12: 1520. https://doi.org/10.3390/coatings14121520
APA StylePurica, M., Comanescu, F. C., & Dediu, V. (2024). Thermally Induced Polymorphic Changes in Poly(vinylidene Fluoride) Thin Layer Investigated Using Micro-Raman Spectroscopy. Coatings, 14(12), 1520. https://doi.org/10.3390/coatings14121520







