3.1. Microstructural Analyses of As-Received and Coated Particles
As-received iron oxide particles were investigated via SEM to reveal the details of the microstructure. SEM micrographs, in addition to optical microscopy images of the as-received iron oxide particles, are represented in
Figure 2.
The optical microscopy image in
Figure 2a shows the appearance of as-received iron oxide particles highlighting their grey-brownish color and the pronounced surface roughness. Microstructures of the surface and cross sections in
Figure 2b,c exhibit typical granulation morphology, where primary sub-micron-sized grains agglomerate into the spherical-like particles with a certain amount of porosity.
In order to assess the potential of increased solar absorptance by additional surface modification, iron oxide-rich particles were coated with deep black pigment using the same coating and sintering parameters for bauxite proppants, as reported in our previous study [
14].
Figure 3 depicts the visual appearance and SEM/EDS mapping of iron oxide particle cross sections at the surface after deep black pigment coating.
Except for a shiny appearance, visual appearance and optical microscopy indicates very little change in surface color after deep black coating of gray-brownish iron oxide particles (
Figure 3a). SEM cross sections also do not show formation of a clearly distinguishable coating layer containing cations from the black pigment, in particular Cu. The EDS maps in
Figure 3d reveal Cu diffusion deep into the particle’s inner volume, where accumulation of Cu seems to occur preferably in Al-rich regions. This rapid diffusion of key black pigment components into the bulk material is evidently unfavorable in terms of improved optical properties. This behavior is in strong contrast to the coating of bauxite proppants, where the formation of a well-defined crystalline Fe-Al-Mn-Mg-Cu spinel layer was observed at the surface of the proppants.
In case of bauxite proppants, a high-temperature stable Fe-Al-Mn-Mg-Cu spinel formed through the reaction between black pigment (Fe-Mn-Cu) and Al and Mg components of bauxite. Considering the much lower initial Al content of new iron oxide particles, a new coating composition was developed in which external Al resource was introduced to the initial deep black powder. Through Al incorporation, mimicking of the high-temperature stable Fe-Cu-Mn-Al spinel was targeted. Metallic Al was preferred to alumina powders to utilize the melting of Al beyond 660 °C, which should lead to a more uniform and homogenous coating layer and may contribute to a better reaction and eventually incorporation of Al to the deep black pigment.
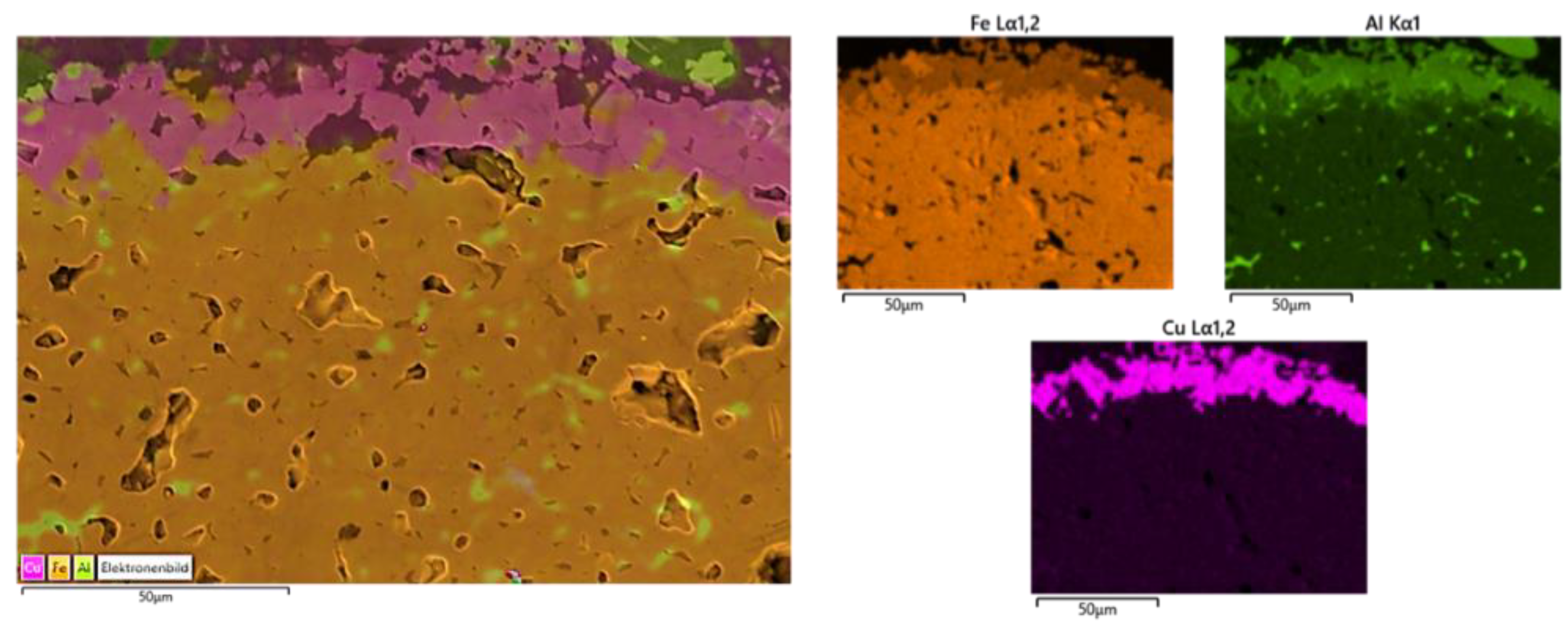
Figure 4 illustrates the appearance of iron oxide particles coated with Al-deep black mixture and the SEM/EDS mapping of cross sections.
The optical microscopy image reveals the successful coverage of the iron oxide particles with the coating mixture of (85 wt.%) deep black pigment and (15 wt.%) Al, where a darker color and smoother surface can be observed. Moreover, EDS mappings reveal a well-defined Cu-rich coating layer at the surface of particles, on the contrary to the particles coated with deep black pigment only (
Figure 3). These results indicate that it is possible to prevent the diffusion of pigment cations, in particular Cu, to the inner structures of the particle and promote the Al-rich spinel formation through the addition of metallic Al.
3.2. Solar Absorptance and Its Long-Term Evolution
Beyond qualitative analysis provided by SEM cross sections and macro examination, the solar absorptance was measured via spectrometry to quantify the optical properties.
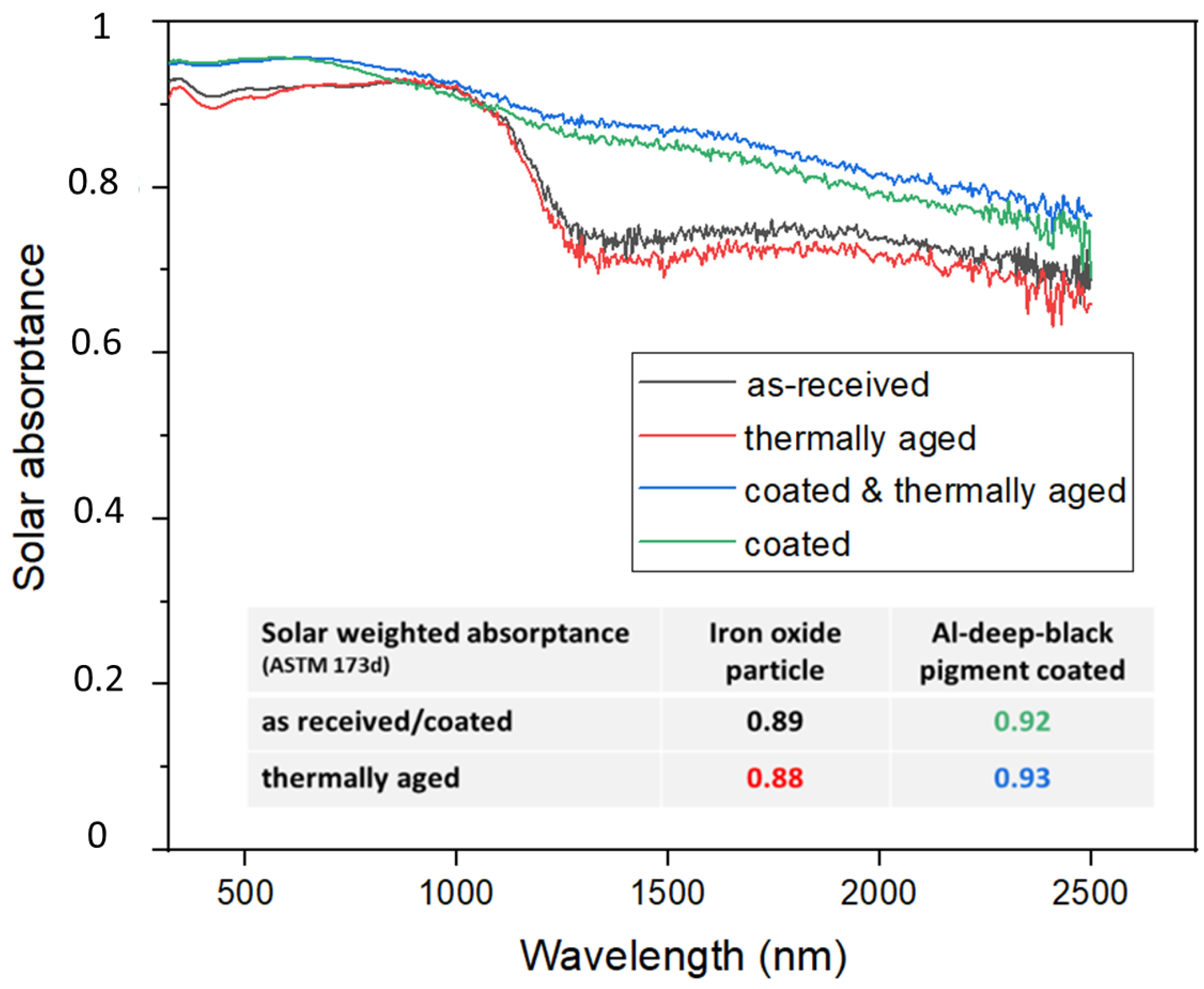
Figure 5 represents the solar absorptance in the 320–2500 nm range for as-received, ‘Al-deep black’ coated and thermally aged iron oxide particles, respectively. Moreover, solar weighted absorptance of the particles was calculated according to ASTM 173d standards and is listed in the inset of
Figure 5.
Iron oxide particles in as-received condition exhibit a solar weighted absorptance of 0.89, which decreases slightly after 1-week thermal aging at 1000 °C to 0.88. To begin with, these new particles exhibit negligible absorptance loss when compared with bauxite proppants, which had lost almost 15% of as-received solar absorptance as previously reported. [
13,
14]. Beyond highly promising optical performance of as-received iron oxide particles, spectrometric analysis given in
Figure 5 proved their further enhancement through the ’Al-deep black´ coating from 0.89 and 0.92. These spectrometric analysis results are in line with SEM/EDS mappings and optical microscopy findings (
Figure 4), in which a surface-localized coating layer had been observed. Coated iron oxide particles were also exposed to thermal aging in identical conditions with as-received particles.
Figure 5 revealed that such additional high-temperature annealing increased the solar absorptance of coated particles slightly from 0.92 to 0.93. This ‘in-operando’ blackening effect indicated that thermal storage provided slight improvement rather than thermal degradation, as observed for uncoated particles.
Low-vacuum SEM was used to provide the details of the surface structure of the as-received, coated and thermally aged samples to better understand the quantitative results obtained via spectrometry. The surface microstructures of as-received particles before and after thermal aging and EDS mapping of coated surfaces before and after thermal aging are given in
Figure 6. Well-defined surface crystals with relatively fine size distribution are observed in the as-received state, as given in
Figure 6a. After thermal aging at 1000 °C for one week, significant grain growth and slight pore coalescence can be observed (
Figure 6b), which may be responsible for the minor decrease in solar absorptance. Propitious long-term optical properties are owing to their more stable and well-crystallized surface features revealed via surface analysis.
Figure 6c provides further insight to the beneficial effect of ‘Al-deep black’ coating, in addition to the cross section mappings in
Figure 4. It is possible to observe a poorly packed coating layer with non-uniform porosity distribution (
Figure 6a). Higher porosities are observed around Al-rich spots, which are marked by the green color (
Figure 6c). Less porous regions exhibited a similar structure to that observed in our previous study [
10], where the reaction between deep black pigment and Al
2O
3 to stable spinel grains was completed. In the present case, several Al
2O
3-rich spots with different surrounding morphology at the surface point out an incomplete Al incorporation to spinel and, thus, a relatively slow formation of a stable, chemically homogeneous coating. After exposure to 1000 °C for one week, a more compact surface layer was achieved as represented in
Figure 5d. Moreover, it is also possible to observe a more chemically and structurally homogenous nature of the coating after thermal aging with respect to the as-received particles (
Figure 6d).
In the light of the spectroscopic analysis and SEM/EDS results, it can be assumed that the addition of Al powders to the black pigment changes the coating microstructural evolution and reaction path in such a way that significantly longer sintering times are required to reach the anticipated equilibrium state. This effect may also apply to the slightly improved optical properties after thermal aging (
Figure 5), which can be regarded as a further sintering step improving surface homogeneity and associated solar absorptance of coated particles rather than their thermal degradation. To verify this concept, a two-hour additional sintering at 1200 °C was applied to the samples in order to provide enough time for the final stabilization of the coating; the corresponding EDS mappings of the cross-sections are presented in
Figure 7.
Through the additional sintering step, a denser, well-consolidated and integrated coating layer is achieved when compared with one-step sintered particles (
Figure 4). It is also worth emphasizing that, despite elongated sintering time, a well-defined interface between the coating layer and particle in EDS mappings confirms that diffusion of pigment cations, in particular Cu, into the particle volume is effectively mitigated by added Al. Al-modified deep black coatings with elongated sintering time can thus be considered very promising in terms of long-term optical stability and process efficiency.
3.3. Elucidating the Role of Added Al in Reactions and Microstructural Evolution of Deep Black Pigment
Obviously, Al incorporation into the initial pigment mixture changed the coating mechanism, postponed the completion of equilibrium phase formation, and required a longer sintering time. Metallic aluminum in the coating mixture may be acting as a reducing agent for Fe, Mn and Cu components of the pigment due to its strong oxygen affinity [
17]. On the other hand, the formation of intermediate compounds may also prolong the formation of thermally stable spinel formation. In order to better understand the effect of the Al on the coating mechanism, additional analyses were performed.
In order to monitor the effect of metallic Al on the spinel formation during the sintering stage, the sinter process was mimicked and simultaneous phase changes were recorded in situ by HT-XRD. Iron oxide particles with a dry-coated Al-deep black pigment layer were heated up to 1200 °C and this temperature was maintained for 2 h. No evidence for the formation of α-Al
2O
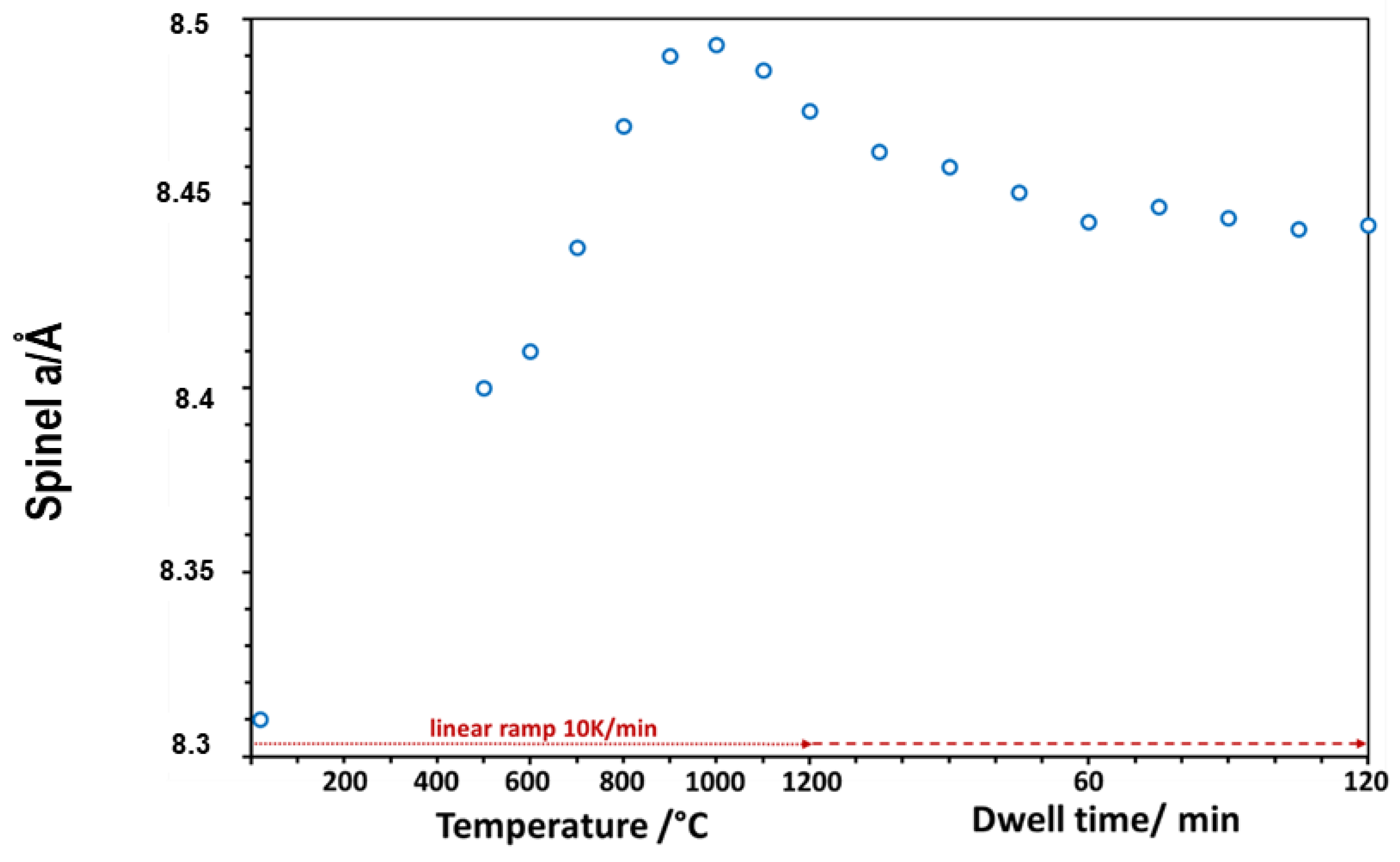
3 (corundum) was found in HT-XRD-profiles. The change in the cubic spinel lattice parameter was recorded as a function of increasing temperature by the cell tuning function of EVA software and is plotted in
Figure 8. Upon heating from room temperature, the increasing lattice parameter of the spinel occurred due to normal thermal expansion at elevated temperatures. However, this linear trend obviously faded out above 800 °C. A peak lattice parameter was reached at 1000 °C, which also marked the onset of a lattice parameter decrease in spinel up to the temperature maximum of 1200 °C. During the first 60 min of the dwell period at 1200 °C, a further significant decrease in the lattice parameter was observed. After 60 min, a steady state seemed to be evolving.
A plausible reason for the observed evolution of the lattice parameter is obviously the progressive incorporation of Al having a smaller atomic size to the Fe, Cu, Mn spinel structure. In order to detect if there is an intermediate compound formation and to better understand the Al-pigment-iron oxide interaction, a quenching experiment was performed at the onset temperature of lattice parameter decrease determined by HT-XRD analysis. Particles are taken out of the furnace at 1000 °C to allow fast cooling rates to keep metastable phases at RT. SEM/EDS mapping of the quenched particle’s cross section is given in
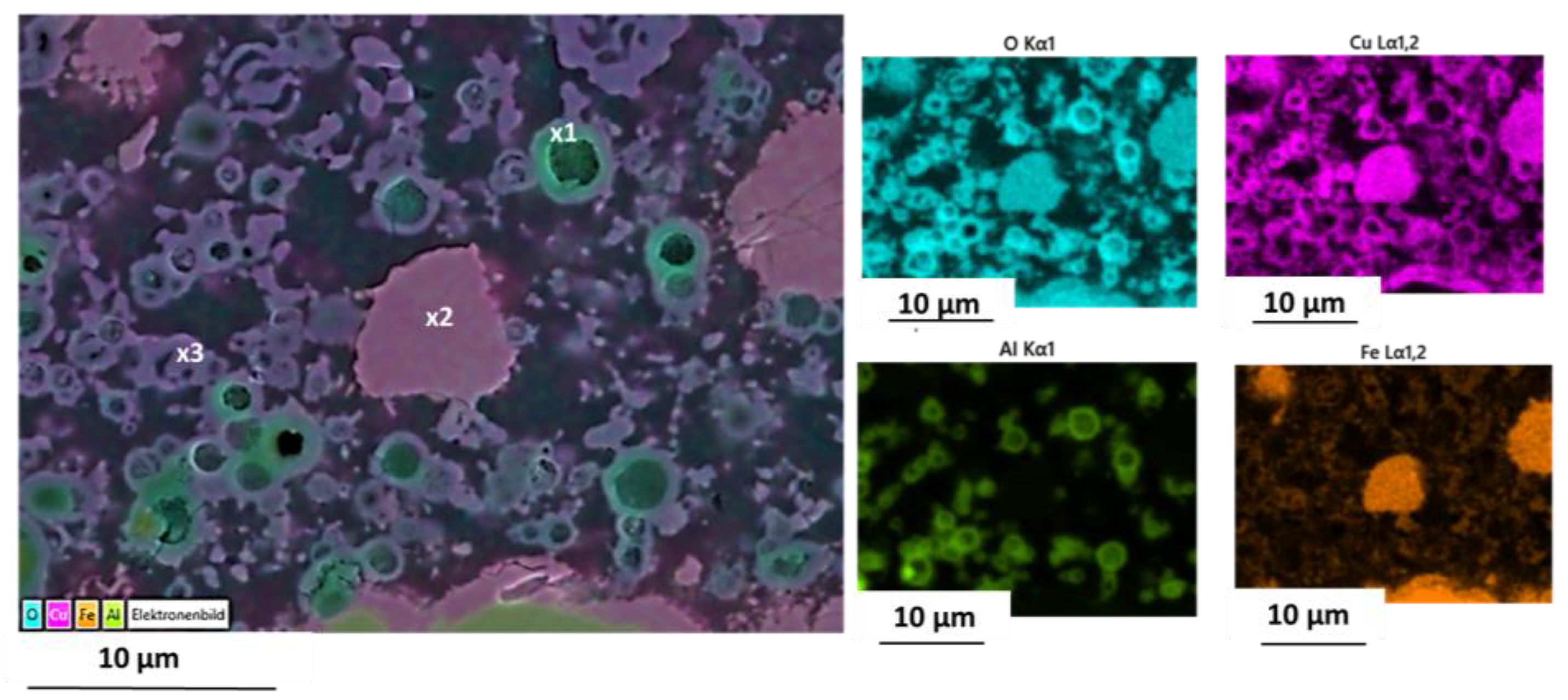
Figure 9.
Firstly, it is worth noting that there is no metallic Al or other metallic pigment components in the system. All components were oxidized and no partial reduction of Cu, Fe, Mn was detected despite the high oxygen affinity of Al.
EDS spot analyses of quenched particles marked in the EDS mapping (
Figure 9) are given together with the cation composition of initial coating powder mixture and as-coated particles (
Figure 4) in
Table 1 for direct comparison purposes.
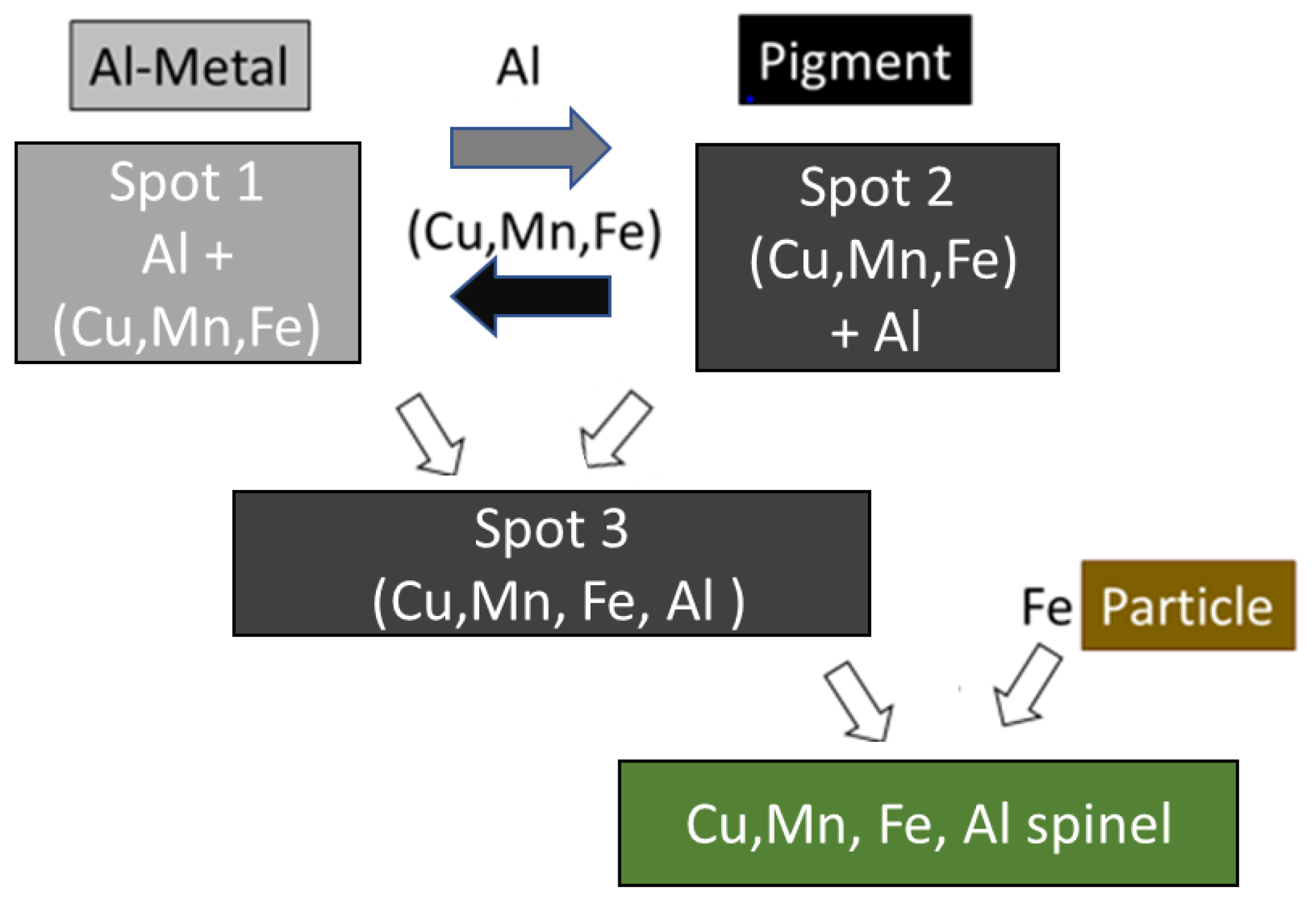
EDS mapping of quenched coated particles revealed an inhomogeneous chemical composition with three typical variants. High-Al areas are frequently found close to larger former Al particles and are enriched in Cu with respect to Mn and Fe (spot #1). Low-Al areas (spot #2) also show some relative depletion of Cu, which is seemingly replaced by Al. Intermediate-Al areas (spot #3) show a composition where all other cations are detected in similar amounts. It must be emphasized, however, that spot analyses from the 1000 °C quenched sample are only a snapshot of ongoing cation migration and should only be regarded as a general trend. A simplified schematic of a plausible cation migration sequence is presented in
Figure 10.
The relative enrichment of Cu at Al as well the enrichment of Al in (Cu,Mn,Fe)-rich grains, obviously former pigment, can be explained as interdiffusion caused by a relative high mutual affinity of Cu and Al. It is reported in previous studies that when the defect spinel γ-Al
2O
3 polymorph of alumina and CuO co-exist, the formation of a CuAl spinel is thermodynamically favorable [
18]. During sintering of the Al-deep black pigment powder mixture, the melting of Al initially takes place. Upon subsequent Al oxidation, the presence of Cu may stabilize an Al-rich, spinel-type polymorph (spot #1) similar to γ-Al
2O
3 rather than corundum [
19]. The other, low Al-spinel phase can be regarded as deep black pigment after out-diffusion of Cu and corresponding in-diffusion of Al (spot #2). In between both areas, the chemical composition may be explained by progressive interdiffusion of low-Al and high-Al spinels (spot #3). After processing at 1200 °C, a uniform Cu, Mn, Fe, Al-spinel coating can be achieved via mostly finished cation interdiffusion (
Table 1). Due to the affinity between spinel-type alumina and Cu oxide, Cu in particular can effectively be captured at the surface and high-temperature stable Fe-Mn-Cu-Al spinel formation occurs without the loss of pigment components by diffusion into the bulk iron oxide particle. On the other hand, chemical homogenization requires a significant amount of time; therefore, better results were obtained during a second sintering step at 1200 °C. As-processed coatings show additional incorporation of Fe rather than inward diffusion of other cations. This can be explained by the different crystal structures, which favor the stabilization of a multi-cation, spinel solid solution rather than a α-Fe
2O
3-type solid solution. A further analysis including a detailed discussion of cation oxidation states or site preference in spinel- or α-Fe
2O
3-type structures is evidently highly complex and would require much more experimental effort. This is beyond the scope of this work. Here, we can only focus on our general concept, which involves specific modification of Cu-rich black pigment with Al, an effective stabilization of a high-temperature stable spinel-type phase with favorable optical properties.